Introduction
Obesity remains a growing public health concern in the paediatric population. Between 2000 and 2015, obesity in the paediatric population increased from 13.9% to 18.5% nationwide. Reference Hales, Carroll and Fryar1,Reference Geserick, Vogel and Gausche2 Importantly, half of obese adolescents were first found to be overweight or obese at age 5, with the majority of young obese children continuing to be obese as adolescents. Reference Geserick, Vogel and Gausche2 Furthermore, these adolescents are likely to continue to be overweight or obese as adults. Obesity is a source of significant morbidity, among which is an increase in cardiovascular disease, including atherosclerosis, hypertension, and coronary vascular disease. Reference Allcock, Gardner and Sowers3,Reference Umer, Kelley, Cottrell and Giacobbi4
The CHD population is a unique cohort of the paediatric population, as CHD is independently associated with an underlying increased risk for cardiovascular complications, including arrhythmia, cardiac infection, myocardial infarction, and hypertension. Reference Verheugt, Uiterwaal and Velde5 Anatomic variation due to CHD may cause cardiovascular risks to be more pronounced. For example, early coronary artery atherosclerosis is seen in patients with obesity following arterial switch operation, Reference Pasquali, Marino and Powell6 and obesity in patients with surgically corrected tetralogy of Fallot increases risk of tachyarrhythmia. Reference Beurskens, Hagdorn and Gorter7 Several common causes of CHD, including ventricular septal defect, coarctation of the aorta, d-transposition of the great arteries, and tetralogy of Fallot, have varied findings regarding their individual risk for obesity following surgical correction of the defect, which may have implications for subsequent management of these populations. Reference Pasquali, Marino and Powell6,Reference O’Byrne, Mcbride and Paridon8,Reference Kim, Jae and Choo9
Several recent studies have demonstrated that the CHD population is similarly affected by the rise in obesity among paediatric and adult populations with CHD. Reference Deen, Krieger and Slee10–Reference Andonian, Langer and Beckmann13 In this population, however, they experience unique challenges, including both recommended and perceived limitations to competitive and low-intensity exercise, concerns about risks associated with exercise, and frequent scrutiny of weight and feeding patterns, including the possibility of multiple interventions such as enteric feeds focused on weight gain in early life. Reference Steele, Preminger and Erenberg14,Reference Willinger, Brudy and Meyer21,Reference Shustak and Mcguire22 Pre-operative nutritional status has been shown to be important for surgical outcomes, and optimising post-operative nutrition is associated with improved surgical outcomes and faster resolution of failure to thrive. Reference Toole, Toole and Kyle35,Reference Kogon, Ramaswamy and Todd37,42 However, many infants require long-term tube feeding support at discharge and report long-term oral aversion as well as behavioural aversion to mealtime. Reference Herridge, Tedesco-bruce and Gray43,Reference Norman, Zühlke and Murray44 Fewer studies describe the subsequent nutritional status of children with repaired CHD after normalisation of weight. As in the general population, a 2019 study shows that the risk of obesity in CHD has increased in recent years, both with higher prevalence and earlier age of onset. Reference Steele, Preminger and Erenberg14 The complex aetiology of obesity includes many intrinsic and extrinsic factors including race, socioeconomic status, genetic predisposition, food access, and confounding interactions. Reference Abdelaal, Roux and Docherty15–Reference Baleilevuka-hart, Khader and Gonzalez De Alba18 Exercise within recommended guidelines is safe and is associated with decreased Body Mass Index. Reference Baleilevuka-hart, Khader and Gonzalez De Alba18–Reference Willinger, Brudy and Meyer21 This paper aims to describe the incidence of obesity in a single-centre cohort of patients undergoing surgical correction of CHD.
Materials and methods
For this single-centre, longitudinal, retrospective cohort study, patient data were identified between 2004 and 2020 and approved by the Institutional Review Board at Oregon Health and Science University. Eligible patients were first identified by operative records, with inclusionary criteria of congenital heart surgery within the first 6 months of life and post-operative diagnosis of ventricular septal defect, d-transposition of the great arteries, coarctation of the aorta, or tetralogy of Fallot. Exclusion criteria included the following: additional structural heart lesions, trisomy 21 diagnosis, a diagnosis of other genetic syndromes known to affect growth, multiple corrective surgeries, or significant post-operative complications (including death).
Body Mass Index scores were identified for a minimum of 2 years up to the most recent recorded height and weight for each patient as of January 1, 2021. Additional data gathered includes sex at birth, gestational age, birth weight, ethnicity (Hispanic or non-Hispanic), and insurance payer (Medicaid or private insurance). Overweight was defined as a Body Mass Index between the 85th and 94th percentile, and obesity was defined as a Body Mass Index at or above the 95th for age and sex. Time-to-overweight and time-to-obesity were defined as the time lapsed from birth to the first identified overweight or obese Body Mass Index, respectively.
Data cohorts were evaluated based on birth year to analyse the risk of obesity by over time. Patients were split into three time cohorts based on birth year from 2004–2008, 2009–2013, and 2014–2017. A survival curve and hazard curve were constructed, and a hazard ratio was calculated to determine risk of obesity compared between birth cohorts, as well as to analyse time-to-obesity. Additionally, subjects were grouped postoperative diagnosis and analysed for time-to-obesity and rate of obesity by time cohort. Data was similarly analysed for outcomes of time-to-overweight. Descriptive analysis was performed on cohort demographics. Chi-squared analysis was performed to evaluate categorical data in relation to the development of overweight or obesity.
To evaluate the prevalence of obesity in this study compared to the general population, further chi-squared analysis was performed between an expected prevalence of obesity in the general paediatric population in the state of Oregon of 13.7%, as identified by the National State of Children“s Health data for the state of Oregon 2019–2020. Reference Hill, Silverman and Noel45 An additional chi-squared calculation was performed to evaluate the overall prevalence of obesity in the study and within each diagnosis. Analysis was performed using the SPSS Statistics model.
Results
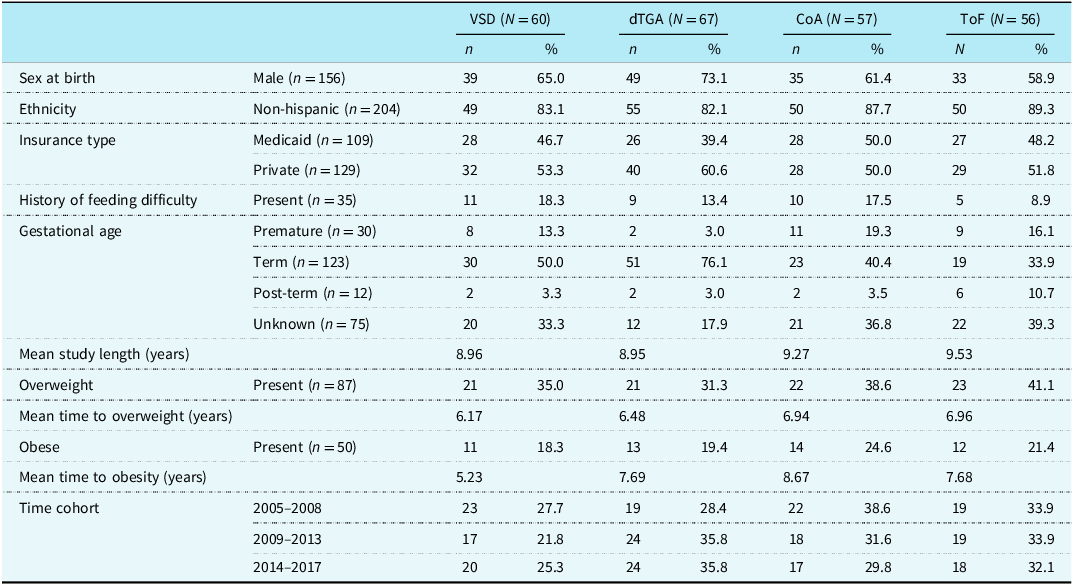
484 patients were screened, with 244 excluded due to not meeting the inclusion criteria or being lost to follow-up. A total of 240 patients were included in the cohort. Of those, 60 (25%) had a diagnosis of ventricular septal defect, 67 (25.4%) had d-transposition of the great arteries, 57 (26.2%) had coarctation of the aorta, and 56 (23.3%) had tetralogy of Fallot. A total of 83 patients were assigned to birth cohort 1 (2004–2008), 78 to cohort 2 (2009–2013), and 79 to cohort 3 (2014–2017). 156 subjects were male at birth (65%), 35% identified as Hispanic (14.6%), and 111 had Medicaid as their primary form of insurance (46.3%). Mean gestational age was 38 weeks and 2 days, with 30 patients (18%) born before 37 weeks (75 subjects did not have gestational age available). Mean birth weight was 3229g, with 26% of patients born at a weight below the 10th percentile for gestational age. Thirty-five subjects (14.5%) were found to have diagnoses of feeding difficulty (Table 1).
Table 1. Demographics of patient cohort by underlying congenital heart disease

VSD = ventricular septal defect; dTGA = d-transposition of the great arteries; CoA = coarctation of the aorta; ToF = tetralogy of Fallot.
A total of 87 (36.2%) subjects became overweight, with 50 (20.8%) of those subsequently developing obesity (Chart 1), which was significantly higher than the expected prevalence of 13.7% (p < 0.01). When separated by diagnosis, coarctation of the aorta had a prevalence of obesity (24.6%) significantly higher than expected (p < 0.01). Ventricular septal defect (18.3%), d-transposition of the great arteries (19.4%), and tetralogy of Fallot (21.4%) were not found to have significantly higher prevalences than expected (Table 2). Mean time-to-overweight was 6.65 years (interquartile range [IQR] 5.54 years), and mean time-to-obesity was 7.42 years of life (IQR 4.34 years).
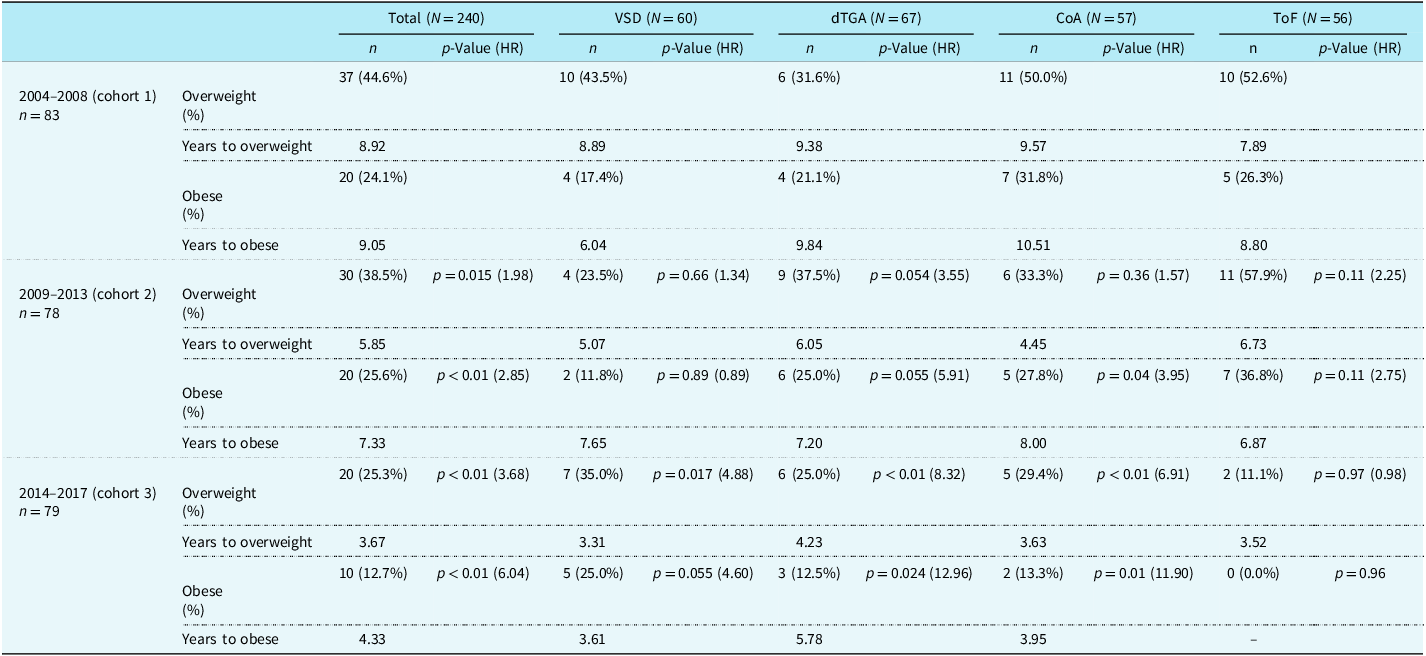
Table 2. Overweight and obesity by diagnosis and time cohort

VSD = ventricular septal defect; dTGA = d-transposition of the great arteries; CoA = coarctation of the aorta; ToF = tetralogy of Fallot.
Analysis of time-to-obesity was initially performed as an aggregated cohort, followed by analysis of individual diagnoses comparing obesity across birth cohorts: cohort 1 (2004–2008), cohort 2 (2009–2013), and cohort 3 (2014–2017). When aggregate CHD was analysed, there was an increased risk of developing obesity in cohort 2 compared to those born in cohort 1 (HR [CI]: 2.85 [1.37–5.93], p < 0.01), and when comparing cohort 3 to those born in cohort 1 (HR: 6.048 [2.25–16.26], p < 0.01) (Figure 1). However, there was no significantly increased risk of obesity with comparison of cohorts 2 and 3 (Figure 2).

Figure 1. Overweight and obese status of all diagnoses by time cohort.

Figure 2. Cumulative hazard of time to obesity among all diagnoses by time cohort.
Further analysis was performed to describe time-to-obesity in the era cohorts (as defined above) by CHD diagnosis. Regarding coarctation of the aorta, there was an increased risk of obesity when comparing era cohorts 1 and 2 (HR: 3.95 [1.03–15.17], p = 0.045) as well as era cohorts 1 and 3 (HR: 11.91 [1.81–77.98], p < 0.01). In d-transposition of the great arteries, there was an increase in risk of obesity comparing era cohort 1 and 3 (HR: 12.96 [1.40–119.93], p = 0.024). There were no significant differences in era cohorts when analysing ventricular septal defect or tetralogy of Fallot, though notably there were 0 subjects with tetralogy of Fallot who developed obesity in time cohort 1, thus comparisons with this group were not performed.
Time-to-overweight analysis was similarly performed on both the aggregated CHD cohort and separated by diagnosis. For all patients, there is an increased risk of being overweight when comparing time cohort 1 and 2 (HR: 1.98 [1.14–3.42], p = 0.015), as well as time cohort 1 and 3 (HR: 3.68 [1.85–7.31], p < 0.01). There was no significant relationship between time cohorts 2 and 3. Time-to-overweight was evaluated by subgroups of CHD diagnosis. Subjects diagnosed with coarctation of the aorta showed a significant increase in risk between time cohorts 1 and 3 (HR: 6.91 [2.01–23.72], p < 0.01), as well as time cohorts 2 and 3 (HR: 4.40 [1.37–14.11], p = 0.013). As with time-to-obese, subjects with d-transposition of the great arteries demonstrated an increased risk between time cohorts 1 and 3 (HR: 8.32 [1.87–37.11], p < 0.01). In subjects with ventricular septal defect diagnosis, there is a statistically increased risk of overweight between time cohort 1 and 3 (HR: 4.88 [1.33–17.92], p = 0.017). Subjects with tetralogy of Fallot did not demonstrate an increased risk of overweight at any time cohort.
Discussion
This single-centre, retrospective cohort study sought to describe the prevalence and evaluate the risk of obesity in a subset of surgically corrected CHD subjects between 2004 and 2017. Obesity and overweight have arisen as a significant public health concern over the past several decades, with a unique risk to children with CHD. Studies have demonstrated that paediatric patients with CHD generally have elevated prevalences of obesity compared to the general population. Reference Briston, Sabanayagam and Zaidi11–Reference Steele, Preminger and Erenberg14 This study re-demonstrated this significantly increased prevalence of both overweight (36.2%) and obesity (20.8%). Furthermore, this study showed that in comparing recent eras, the risk of development of both overweight and obesity for all CHD subjects increased with time. This is a similar trend to what is seen nationally, Reference Stierman, Afful and Carroll23 and emphasises the growing evidence that overweight and obesity are prominent sources of morbidity in CHD. Perhaps more concerning is the progressive trend of earlier onset of overweight and obesity as seen with this cohort.
Coarctation of the aorta was the only diagnosis of those included that was found to have a significantly higher prevalence of obesity compared to other forms of CHD. This subgroup is particularly vulnerable to the increasing risk of obesity in the more modern era cohorts. There is no evident physiologic reason for this difference, and study criteria excluded those with underlying genetic diagnoses predisposing to elevated Body Mass Index, as well as those with significant concomitant cardiac anomalies such as a bicuspid aortic valve. Individuals with coarctation of the aorta do remain at risk of hypertension at rest and with exercise, as well as more devastating complications such as aortic aneurysm, dissection, or rupture. Reference Van Hare, Ackerman and Evangelista24 The strong correlation between obesity and hypertension, left ventricular hypertrophy, and aortic dilation Reference Jackson, Ph and Fox26 may only further increase these risks. These patients may be at particular risk for poor outcomes from obesity-related morbidity.
The distribution of obesity across different eras was comparable to that of general population and prior studies. Our cohort found the overall prevalence of obesity was lower (12.6%) between 2014–2017 compared to the overall prevalence 20.8% (2004–2017), and is near identical to the prevalence of obesity found in the NHANE 2017–2020 survey in those ages 2 to 5 years old (12.7%). Reference Stierman, Afful and Carroll23 These subjects would be expected to have a lower prevalence of obesity as the steepest rise in Body Mass Index has previously been found to occur between ages 6 and 9, and further increase into adolescence. Reference Umer, Kelley, Cottrell and Giacobbi4,Reference Kim, Jae and Choo9,Reference Stierman, Afful and Carroll23,Reference Cunningham, Kramer and Narayan27 Older cohorts of 2004–2008 as well as 2009–2013 (24.1% and 25.3%, respectively) were found to have a prevalence of obesity similar to prior studies. Reference Kim, Jae and Choo9
Several plausible hypotheses regarding this increased risk of obesity have been posed previously. It is possible that this association is driven by environmental factors affecting exercise. Reference O’Byrne, Mcbride and Paridon8 The American Heart Association has published recommendations Reference Van Hare, Ackerman and Evangelista24 for sports participation in children with CHD. Subjects with repaired ventricular septal defect are recommended for full participation if there is little to no residual defect and absence of pulmonary hypertension. Subjects repaired tetralogy of Fallot and d-transposition of the great arteries are recommended to have evaluation with electrocardiography, ventricular function and exercise testing, with exercise restriction if evidence is found of exercise-induced arrhythmias, hypotension, ischaemia, or ventricular dysfunction. In addition, subjects with repaired coarctation of the aorta are recommended to undergo similar testing, including either magnetic resonance imaging or computed tomography angiography, as well as gradient systolic blood pressure measurements prior to participation. Reference Van Hare, Ackerman and Evangelista24,Reference Curran, Hansen and Lindsey25 For all repaired CHD, recommendations for thorough evaluation, especially those requiring specialised testing, may delay or inhibit clearance for sport participation. This barrier is particularly high for the coarctation of the aorta population, with restricted exercise limited to low-intensity activities only. Indeed, in the absence of specific exercise restrictions from a cardiac provider, some patients and families may elect to self-restrict due to ambiguity or concerns.
This rigour of testing may delay or fully deter some from engaging in appropriately intense exercise. A study of exercise tolerance and risk aversion in the Fontan population demonstrated that around half of parents are concerned about sports participation for their child, though there was notably no significant difference between athletes and non-athletes. Reference Baleilevuka-hart, Khader and Gonzalez De Alba18 This risk aversion may similarly hinder involvement by children with coarctation of the aorta, even when recommended for full participation. Exercise in childhood is an integral component to many proposed and studied weight interventions, Reference Wang, Cai and Wu28,Reference Motevalli, Drenowatz and Tanous29 leads to further involvement in exercise in adulthood, Reference Perkins, Jacobs and Barber30,Reference Jose, Blizzard and Dwyer31 and has been associated with decreased Body Mass Index, Reference Logan, Lloyd and Schafer-kalkho32 improved quality of life, Reference Vella, Cliff and Magee33 and decreased cardiovascular risk. Reference Hebert, Klakk and Møller34 While sport restriction may be warranted following specialist evaluation, it is similarly important to identify safe exercise and promote regular involvement in these activities. Reference Baleilevuka-hart, Khader and Gonzalez De Alba18,Reference Longmuir, Brothers and de Ferranti20,Reference Perkins, Jacobs and Barber30 Active practice of activity prescriptions by cardiac providers may further augment exercise participation.
Nutritional status is an area of close scrutiny as children with CHD grow. In both the pre-operative period and subsequent first years of life, there is an emphasis on promoting nutritional status as measured by weight gain to manage the perioperative state and optimise cognitive growth. Reference Toole, Toole and Kyle35–Reference Kogon, Ramaswamy and Todd37 This was seen in our cohort, though interestingly at lower prevalences than previously reported prevalences of 40–60%. Reference Toole, Toole and Kyle35,Reference Kogon, Ramaswamy and Todd37 These feeding difficulties are a source of family anxiety and patient morbidity, particularly in the post-operative period. Reference Wright, Phillips and Matthews38 As patients pass this critical period of nutritional intervention, focus within multidisciplinary CHD clinics may shift away from preemptive management of weight. Meanwhile, patient and family concerns regarding weight continue throughout early childhood and adolescence, including culturally driven views of higher weight indicating improved health. Reference Kaufman and Karpati39,Reference Baughcum, Burklow and Deeks40 Regular specialist clinic visits throughout childhood remain an opportunity for intervention by multidisciplinary teams, including education regarding the unique importance of normal weight in this population.
This study had several limitations. First, the data were sampled from a single tertiary centre, limiting the diversity of the patient population as well as the overall sample size. This is a limited robust analysis of subgroups of diagnosis and era. Most notably, this was seen in the analysis of tetralogy of Fallot, which had an incidence of 0% obesity in the time cohort 2014–2017. While this does limit the power of analysis, this centre serves a large number of post-surgical cardiac patients in the state of Oregon and Southwest Washington, potentially decreasing sampling bias. This study was retrospective in nature; thus, there were no specific time points of data to indicate the onset of overweight and obesity. It is possible that patients met criteria for these categories prior to their otherwise scheduled hospital encounters. This is alleviated to some extent by the need for frequent follow-up for corrected CHD patients, particularly in the first several years of life. Third, Body Mass Index was compared to a fixed prevalence of 13.7% as published for the State of Oregon in a 2019–2020 survey, as opposed to matched healthy controls. 42 This may have allowed for more descriptive analysis of the change in obesity, both for the general population as well as those with CHD. While these controls could have been identified, they lacked adequate follow-up to identify the onset of overweight or obesity with accuracy. As this population largely contains individuals living in Oregon, this fixed prevalence is believed to closely represent the expected prevalence of obesity for this cohort. Finally, Body Mass Index is known to be an imperfect surrogate for overall health and cardiovascular comorbidities. Prior studies, however, have demonstrated a relationship between paediatric Body Mass Index and the development of early atherosclerosis on autopsy evaluation, Reference Herridge, Tedesco-bruce and Gray43 cardiovascular morbidity in childhood, and subsequent cardiovascular disease in adulthood. Reference Umer, Kelley, Cottrell and Giacobbi4 While other methods may more directly measure the progression of cardiovascular risk in this at-risk population, this surrogate demonstrates the ongoing disparity between those with CHD and the general population.
Conclusions
Our findings identify that over a 14-year cohort of children with surgically repaired CHD, the prevalence of obesity was higher than expected in our study population as compared to the general population overall, as well as having an increasing risk in more modern cohorts. This effect was most prominent for subjects with coarctation of the aorta, both in terms of overall obesity prevalence and increased risk of developing obesity with more modern time cohorts. Further research is warranted to elucidate the unique risk seemingly present in patients with coarctation of the aorta. Early subspecialist and primary care provider intervention in exercise counselling and sports engagement may bring a substantial benefit to both childhood and eventual adult cardiovascular outcomes. Future prospective research is needed to identify other categories of CHD at particular risk for the development of obesity.
Financial support
None.
Competing interests
None.