Introduction
An estimated 1.3 million persons receive care from nursing homes in the United States each year. 1,Reference Slayton, Toth and Lee2 Nursing home residents are have a high colonization prevalence and risk of infection with multidrug-resistant organisms (MDROs) such as methicillin-resistant S. aureus (MRSA), Vancomycin-resistant Enterococcus (VRE), extended-spectrum beta-lactamase producers (ESBLs), and Carbapenem-resistant Enterobacterales (CRE). Reference Bradley, Terpenning and Ramsey3–Reference Muder, Brennen and Wagener5,Reference Gussin, McKinnell and Singh11 With such extensive MDRO prevalence, it is not surprising that MDRO environmental contamination has been reported to be as high as 50% on some surfaces. Reference McKinnell, Miller and Singh12,Reference Kanamori, Rutala, Sickbert-Bennett and Weber13
Trials of decolonization of patients in intensive care units, patients with medical devices, MRSA-colonized patients following hospital discharge, and, most recently, nursing homes residents using routine chlorhexidine gluconate (CHG) bathing and topical nasal products have reduced MDRO carriage and rates of serious infections. Reference Huang, Septimus and Kleinman6–Reference Gussin, McKinnell and Singh11,Reference Miller, McKinnell and Singh14 However, the effect of body decolonization in nursing home residents on environmental MDRO contamination in these facilities is less well understood, and previously studied in hospital settings. Reference Vernon, Hayden, Trick, Hayes, Blom and Weinstein15 We explore the ability of routine decolonization to affect MDRO environmental contamination and body colonization in nursing homes.
Methods
We evaluated retrospective data collected from June 2015-March 2016 in 3 nursing homes in Southern California to study the impact of CHG and nasal iodophor. This study involved: (1) a 3-month baseline data collection period, (2) a 3-month phase-in period when CHG antiseptic bathing/shower soap and nasal iodophor were introduced, and staff were trained on proper application, and (3) a 3-month period of continued universal decolonization. The universal decolonization intervention involved administration of (1) nasal iodophor to all residents twice daily Monday through Friday every other week plus, (2) routine use of 4% rinse-off CHG in the shower and 2% leave-on CHG for bed baths. Reference Huang, Septimus and Kleinman10 Most residents were showered 2 or 3 times a week, as per institutional standard, and received periodic bed baths or partial wipe downs, as needed. The usual frequency of resident bathing in participating nursing homes was not changed during the intervention. Additionally, study staff trained facility nurses prior to swabbing and accompanied facility nurses during the swabbing to provide logistical support (ie, bagging and transporting specimens). This study was reviewed and approved by the institutional review boards at the University of California, Irvine, Providence Little Company of Mary Medical Center, and The Lundquist Institute at Harbor-UCLA Medical Center.
Each nursing home performed 6 point prevalence samplings of residents and the environment in the baseline period, and another 6 point prevalence samplings in the intervention period. For each resident sampling, 50 occupied beds were randomly selected and facility nurses were trained to perform body site collection. A bilateral nares swab was assessed for MRSA, and a combined bilateral axilla/groin swab was assessed for MRSA, VRE, ESBL, and CRE, as described previously. Reference McKinnell, Miller and Singh12
For assessing environmental contamination, we swabbed a standardized set of 5 high-touch fomites in rooms of residents who tested positive for any MDRO on the most recent point-prevalence screening. We did not sample any rooms of residents who tested negative for MDROs. Surfaces tested were: (1) bedside table/bedrail; (2) call button/television remote control/phone; (3) doorknobs; (4) light switch; and (5) bathroom rail/sink/toilet flush handle). Within six hours of collection, swabs were enriched in liquid Tryptic Soy Broth for 10–12 hours. Screening for MDROs utilized: Spectra MRSA (Thermo Scientific) for MRSA; Campylobacter agar (10% Sheep blood with 5 antibiotics, vancomycin 10 ug, cephalothin 15 ug, trimethoprim 5 ug, Polymyxin B 2.5 units, Amphotericin B 2 ug) for VRE; MacConkey with cefpodoxime disk (2 ug) for ESBL organisms; and MacConkey Agar with meropenem disk (2ug) for CRE. We defined environmental contamination of a room as isolation of 1 or more MDROs among the five high-touch surfaces sampled within a given room.
We collected limited clinical data on each resident during the body swabbing process including bed bound status, incontinence, lucidity, diabetes, hemodialysis, presence of wounds, central venous catheters, urinary catheters, and other medical devices (eg, feeding devices). Group comparisons between baseline and intervention period were performed using Chi-squared test or Fisher’s exact test, as appropriate (we did not analyze the phase in period). We then created multivariable logistic regression models for the outcomes of MDRO carriage and MDRO object contamination, adjusting for period (baseline and intervention) and collected variables. MDRO carriage models were clustered by resident and nursing home. MDRO contamination models controlled for clustering by room and nursing home. Statistical analyses were conducted using SAS version 9.4.
Results
Resident MDRO colonization
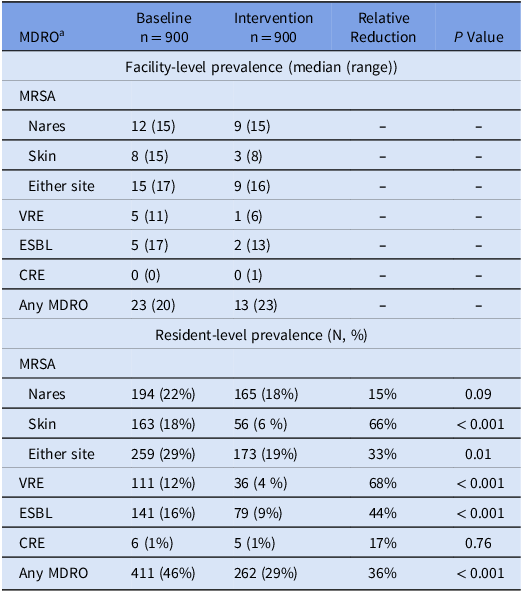
During the baseline period, overall resident MDRO prevalence was 46% (411/900). During the intervention period, MDRO prevalence decreased to 29% (262/900, P < 0.001). Compared to the baseline period, during the intervention period, MRSA decreased from 29% (259/900) to 19% (173/900) (P = 0.01), VRE 12% (111/900) to 4% (36/900) (P < 0.001), and ESBL 16% (141/900) to 9% (79/900) (P < 0.001). CRE prevalence did not significantly change (1% (6/900) to 1% (5/900, P = 0.76). Further details on colonization are summarized in Table 1.
Table 1. Prevalence of MDRO colonization before and after decolonization intervention

a Abbreviations: MDRO, multidrug-resistant organisms; MRSA, Methicillin-resistant Staphylococcus aureus; VRE, Vancomycin-resistant Enterococcus; ESBL, extended-spectrum beta-lactamase producers; CRE, carbapenem-resistant Enterobacteriaceae.
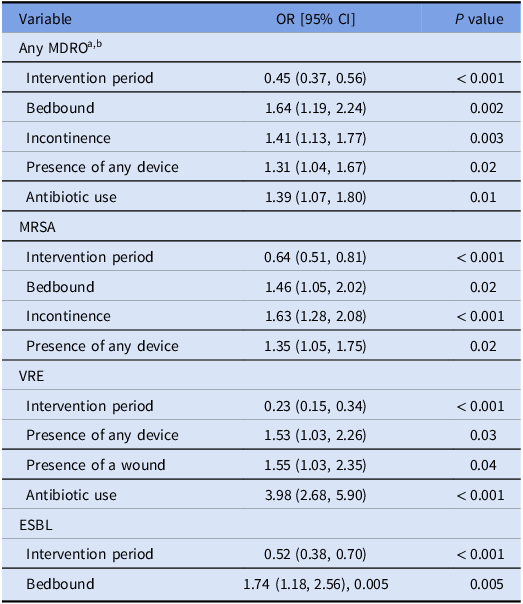
In a multivariable model, MDROs prevalence was positively associated with bed bound status (odds ratio (OR) 1.64 [95% confidence interval (CI): 1.19–2.24]), incontinence (OR 1.41 [95% CI: 1.13–1.77]), and the presence of any device (OR 1.31 [95% CI 1.04–1.67]), and inversely associated with the intervention period (OR 0.45 [95% CI 0.37–0.56]) (Table 2). In MRSA, VRE, and ESBL-specific models, body colonization was inversely associated with the intervention period in the MRSA model (OR 0.64 [95% CI 0.51–0.81]), in the VRE model (OR 0.23 [95% CI 0.15–0.34]), and the ESBL model (OR 0.52 [95% CI 0.38–0.70]). Pathogen-specific models are outlined in Table 2. We did not perform a CRE model given the low number of cases (n = 15).
Table 2. Multivariable model of factors associated with body site colonization in 3 nursing homes

a Abbreviations: MDRO, multidrug-resistant organisms; MRSA, Methicillin-resistant Staphylococcus aureus; VRE, Vancomycin-resistant Enterococcus; ESBL, extended-spectrum beta-lactamase producers; CRE, carbapenem-resistant Enterobacteriaceae.
b MDRO models included diabetes, lucidity, and presence of wounds.
Environmental contamination
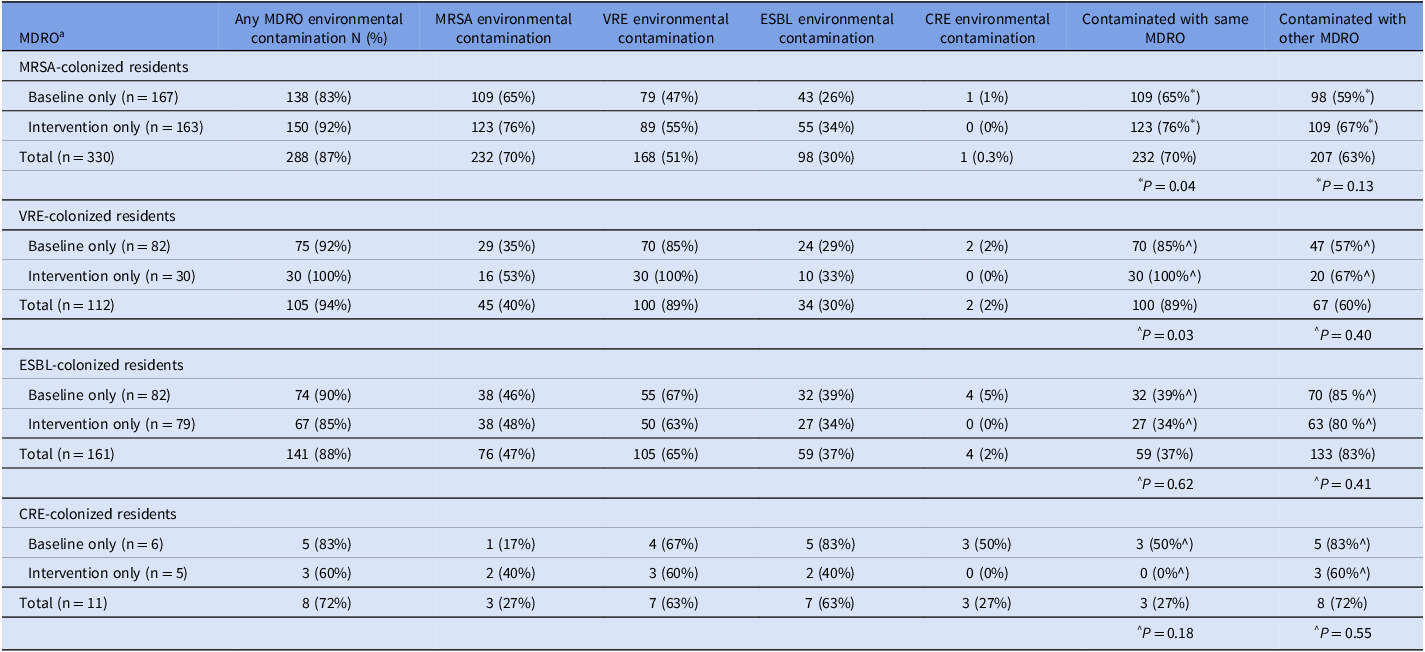
Among residents who were colonized with any MDRO, 288/330 (87%) of their room environments were contaminated with ≥1 MDRO (Table 3).
Among rooms of MRSA-colonized residents, 232/330 (70%) were contaminated with MRSA, 168/330 (51%) were contaminated with VRE, 98/330 (30%) with ESBL, and 1/330 (0.3%) with CRE. Thus, for MRSA-colonized residents’ rooms, 70% (232/330) of their room environments were contaminated with the same MDRO isolated from the resident and 63% (207/330) were contaminated with a non-MRSA MDRO. Overall, 288/330 (87%) of the rooms of MRSA carriers were contaminated with ≥1 MDRO. Similar results for other MDRO carriers are found in Table 3.
Table 3. Fomite contamination of residents’ room among residents who were colonized with one or more multidrug-resistant organisms

$ Chi-square test comparing the proportion of rooms with MDRO-specific contamination in the baseline versus intervention periods.
^ Fisher exact test comparing the proportion of rooms with MDRO-specific contamination in the baseline versus intervention periods.
a Abbreviations: MDRO: multidrug-resistant organisms, MRSA: Methicillin-resistant Staphylococcus aureus, VRE: Vancomycin-resistant Enterococcus, ESBL: extended-spectrum beta-lactamase producers, CRE: carbapenem-resistant Enterobacteriaceae.
Looking at each study period separately, MDRO contamination of high-touch environmental surfaces in the rooms of MDRO carriers did not decrease between the baseline and intervention periods (Table 3). In fact, for rooms of residents that remained MRSA and VRE carriers despite decolonization, MRSA and VRE room contamination, respectively, was higher during the intervention versus baseline period (Table 3), while it was unchanged for other MDROs.
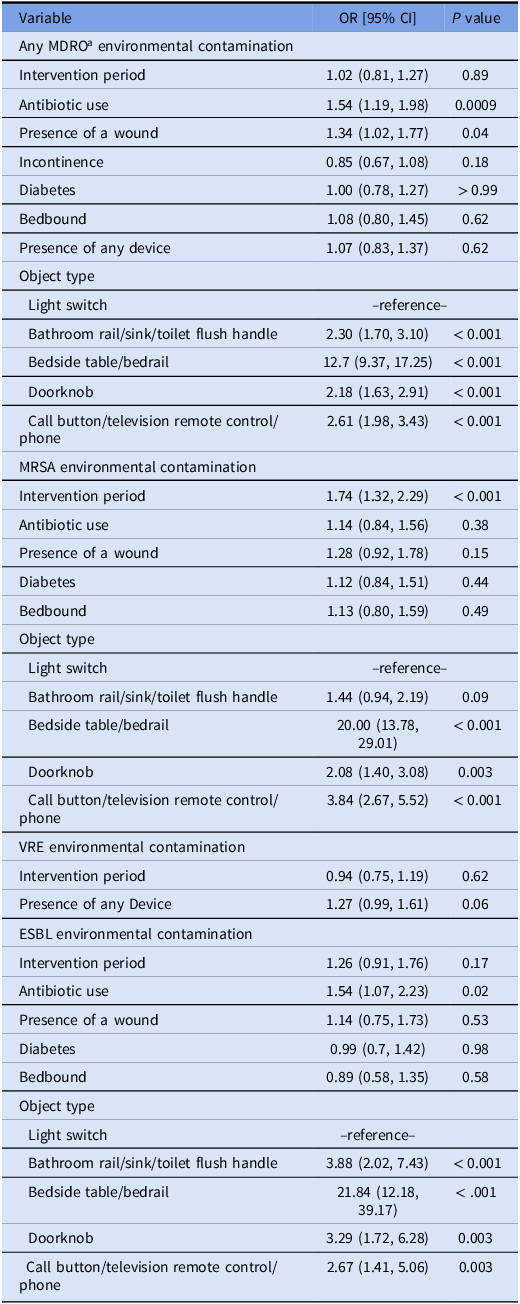
In multivariable models, MDRO room contamination was associated with antibiotic use (OR 1.54 [95% CI 1.19–1.98]), presence of a wound (OR 1.34 [95% CI 1.02–1.77]), and specific objects (bathroom rail/sink/toilet flush handle (OR 2.30 [95% CI 1.70– 3.10]), bedside table/bedrail (OR 12.7 [95% CI 9.37–17.25]), doorknob (OR 2.18 [95% CI 1.63, 2.91]), and call button/television remote control/phone (OR 2.61 [95% CI 1.98– 3.43], using the light switch as the referent). There was no relationship between fomite contamination and the intervention period (OR 1.02 [95% CI 0.81, 1.27]) (Table 4).
Table 4. Multivariable model of factors associated with fomite contamination among residents with MDRO colonization

a Abbreviations: MDRO, multidrug-resistant organisms; MRSA, Methicillin-resistant Staphylococcus aureus; VRE, Vancomycin-resistant Enterococcus; ESBL, extended-spectrum beta-lactamase producers.
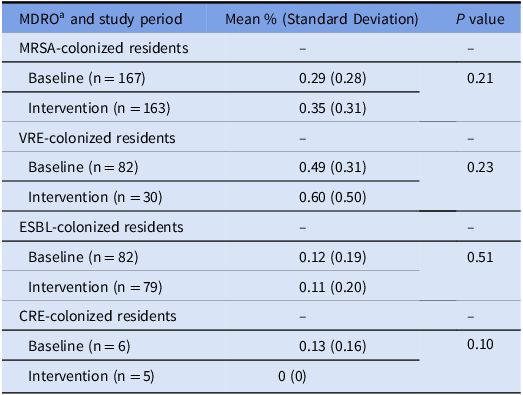
Results of MRSA, VRE, and ESBL-specific models of environmental contamination are summarized in Table 4. Of note, we found fomite contamination was significantly associated with the intervention period in the MRSA specific model (OR 1.74 [95% CI [1.32–2.29]), but not in the VRE and ESBL specific models (OR 0.94 [95% [0.75–1.19], and OR 1.26 [0.91–1.76], respectively) (Table 4). Additionally, we did not find any significant differences between baseline and intervention periods in the mean number of room objects that tested positive for ≥1 MDRO in rooms of residents who were MDRO colonized in for each MDRO tested (MRSA, VRE, ESBL, and CRE; P > 0.05 for all comparisons, Table 5).
Table 5. Mean percent of room objects that test positive for 1 or more MDROs in rooms of residents who are MDRO colonized

a Abbreviations: MDRO, multidrug-resistant organisms; MRSA, Methicillin-resistant Staphylococcus aureus; VRE, Vancomycin-resistant Enterococcus; ESBL, extended-spectrum beta-lactamase producers; CRE, carbapenem-resistant Enterobacteriaceae.
Discussion
We found that when use of CHG and nasal iodophor was pragmatically adopted into routine bathing and showering care in nursing homes, MDRO colonization with MRSA, VRE, and ESBL Gram-negative bacilli significantly decreased among residents. However, for residents that remained carriers despite decolonization efforts, we found no reduction of MDRO contamination of high-touch environmental fomites in their rooms. In fact, MRSA room contamination was significantly higher when MRSA carriers failed to clear colonization with routine decolonization. Although the exact reason for lack of MDRO environmental contamination reduction in rooms of residents with MDRO colonization during the decolonization period is difficult to determine, our result may suggest that those residents who failed MDRO decolonization may be more likely to shed their MDRO in the environment.
It is important to highlight that we only swabbed environmental surfaces in rooms of MDRO-colonized patients. Thus, we cannot conclude that de-colonization did not decrease overall MDRO contamination of environmental surfaces throughout the nursing homes since we did not survey a random sample of these rooms. Since fewer residents remained colonized with MDROs in the decolonization period, fewer rooms were available to be sampled under our design. We cannot know if rooms of residents who had been previously MDRO-positive but turned MDRO-negative on our culture results were less contaminated.
What we do know is that persistence of carriage is associated with persistent environmental contamination. These findings suggest that environmental surfaces in rooms of MDRO carriers may be important reservoirs of MDROs and require special attention for environmental decontamination despite decolonization strategies. Given that hand hygiene compliance in nursing homes has found to be generally suboptimal and as low as 5%, Reference Haenen, de Greeff, Voss, Liefers, Hulscher and Huis16 efforts to improve hand hygiene in nursing homes may also help minimize spread of MDROs from MDRO-colonized residents’ rooms to other environments and residents.
Interestingly, our findings differ from that of Mody et al, who performed a multi-component intervention to prevent infections in nursing homes. In their cluster randomized trial, the use of enhanced barrier precautions, CHG bathing, MDRO surveillance, environmental cleaning education and feedback, hand hygiene promotion, and healthcare worker education and feedback reduced the odds of MDROs in patients’ environment by 43% but had no significant effect on residents’ body colonization. Reference Mayer, Stone and Leecaster17 However, we found that introducing body decolonization without other interventions reduced MDRO body colonization.
The reasons behind the discrepancies in the two studies are not clear, but may relate to adherence and the quality of specific interventions. Perhaps our use of nasal iodophor helped drive the decrease in MRSA carriage, but we also found significant decreases in VRE and ESBL carriage, which would not be affected by nasal iodophor. Perhaps Mody et al’s emphasis of daily CHG bathing for high infection risk residents but only 2 days per week for low-risk residents created a complex system that was harder to implement and achieve adherence compared to the system used by our nursing homes in which bathing with soap and water was replaced by CHG for the typical frequency of bathing at 3 times per week for all residents. In support of this, Mayer et al Reference Mayer, Stone and Leecaster17 found that implementation of less complex bundles, eg, ours compared to Mody et al, Reference Mody, Gontjes and Cassone18 may be easier to implement successfully and may explain why in our study, body colonization was significantly reduced. The positive effect of the multifaceted bundle by Mody et al on environmental contamination may be related to the fact that their intervention included directed campaigns to increase cleaning; however, since we did not sample rooms of non-MDRO-colonized residents, we cannot directly compare our results to theirs.
Our analysis had several insights into variables associated with MDRO carriage in nursing home residents. First, the prevalence of MDRO-colonized residents was very high (42%), and prior reports have noted that the vast majority of MDRO-colonized residents are not known by staff to be MDRO colonized. Reference McKinnell, Miller and Singh19 In addition, we found MDRO carriage was higher among those with bed bound status, incontinence, and the presence of devices, even during decolonization. These findings are consistent with other studies. Reference Rodríguez-Villodres, Martín-Gandul and Peñalva21,Reference Gontjes, Gibson and Lansing22 We hypothesize that bedbound status makes bathing more challenging and thus these residents have a higher chance for MDRO carriage to persist. Given CHG and nasal iodophor do not decolonize MDRO from gut reservoirs Reference McKinnell, Miller and Singh12 we also hypothesize that incontinence allowed intestinal MDROs to spread from the gastrointestinal tract to the skin, resulting in higher MDRO prevalence in bedbound residents.
Our analysis also found that rooms of MRSA carriers were not only commonly contaminated with MRSA but were also commonly contaminated with VRE and ESBLs. Likewise, rooms of VRE carriers’ rooms were commonly contaminated with MRSA and ESBL. This cross-contamination may be explained by insensitivity of sampling only a few body sites (eg, residents may, in fact, be carriers of the other MDROs since MDROs are correlated and share common associated variables such as antibiotic use), residual contamination from prior occupants, or transmission from roommates or contaminated healthcare workers hands or shared objects. Additional efforts to ensure effective environmental decontamination is warranted to reduce overall MDRO burden in nursing homes.
We also note that Rutala et al found that rooms of MDRO carriers were contaminated with the same MDRO 30% of the time, Reference Rutala, DiBiase and Powell20 while we found that the frequency of contamination was much greater in this study. This may be due to differences in the quality of cleaning in nursing homes. It may also reflect differences in identifying carriers as they sampled residents who were known carriers, and knowledge may have led to cleaning or other interventions. In contrast, we performed active surveillance to identify otherwise unidentified MDRO carriers. Thus, our survey may more accurately reflect environmental contamination of MDRO-colonized nursing home residents.
Similar to Gontjes et al, Reference Gontjes, Gibson and Lansing22 we found that antibiotic use was associated with increased environmental contamination, possibly by maintaining selective pressure for MDRO colonization. These findings suggest that antibiotic stewardship in nursing homes may have further benefits, although such a relationship would need dedicated studies to prove. Additionally, similar to Mody et al, we found that wounds were associated with increased environmental contamination, likely due to colonization of non-intact skin, drainage, and increased handling by staff for wound care.
Similar to our prior work, Reference McKinnell, Miller and Singh19 we found that MDRO contamination was more commonly found on doorknobs, call buttons, remote controls, phones, toilet flush handles, and especially bedside tables and bedrails, compared to light switches. Regardless of the mechanism, the high frequency of MDRO environmental contamination suggests that those fomites are challenging to clean in the constant presence of a shedding MDRO carrier with typical cleaning protocols in nursing homes. We have recently shown that contamination by carriers after cleaning is swift and extensive. Reference Sansom, Gussin and Schoeny23 Other efforts to enhance environmental cleaning are needed, perhaps via the further development of agents that allow residual disinfection after application given that the first to market has been withdrawn. Reference Redmond, Cadnum and Silva24
Our study has limitations. First, as noted above, we only performed environmental MDRO surveys in rooms of MDRO-colonized residents. We cannot conclude what happened in terms of environmental contamination in rooms of residents who were not colonized either at baseline or during the decolonization intervention. Second, we only surveyed three body sites (axilla/groin and nares) and surveying more body sites may have identified more carriers and possibly explained more of the discordant MDRO environmental contamination that was found. Third, we did not quantify carriage or assess the quality of decolonization for residents in the rooms sampled. Thus, we are unable to know whether persistent carriage was due to decolonization failure or poor quality/lack of decolonization. We are also unable to determine if bioburden reduction without clearance has an impact on the degree of environmental contamination. Finally, we did not quantify adverse events to chlorhexidine, although much larger studies showed that adverse effects of chlorhexidine were rare and mild. Reference Miller, McKinnell and Singh25
In summary, we found that routine decolonization with chlorhexidine and nasal iodophor in nursing home residents reduced body colonization. However, among persistent MDRO carriers, environmental contamination of high-touch surfaces in resident rooms remained extensive and included the carried MDRO and other MDROs. Efforts to ensure high-quality cleaning is needed when MDRO carriage exists.
Financial support
The project was funded by the CDC Epicenters program (U54 CK000172 Supplement to MPI: Huang/Platt) for conduct and NIH (U19 AI172725, PI: Huang) for analysis.
Competing interests
S.H., R.S., and G.M.G., conduct clinical studies and trials in which participating hospitals and nursing homes receive contributed products from Xttrium Laboratories.