Introduction
Water occurs as trace hydrogen (H) and much rarer deuterium (D, at 0.0156%) in nominally anhydrous minerals throughout the Earth’s mantle. Hydrogen occupies structural point defects in these nominally anhydrous minerals at concentrations that commonly range from zero to a few hundred ppm (Mosenfelder et al., Reference Mosenfelder, Bureau and Withers2024). Regardless of such low H concentrations at the crystal-scale, the mantle represents an immense water reservoir because of its vast volume (Bell and Rossman, Reference Bell and Rossman1992). The mantle reservoir is a major sink and source for Earth’s petrological, hydrological and atmospheric water cycles through time (Hirschmann, Reference Hirschmann2006). The presence of water as trace H in nominally anhydrous minerals greatly influences the geodynamic and geochemical evolution of the mantle. Increasing H contents reduces mineral strength and viscosity, and consequently influences mantle deformation and plate tectonics (e.g. Hirth and Kohlstedt, Reference Hirth and Kohlstedt1996; Warren and Hansen, Reference Warren and Hansen2023). Increasing H modifies the seismic and electrical properties of the mantle and as a result influences how geophysicists study mantle processes (Karato, Reference Karato2011; Jones et al., Reference Jones, Fullea, Evans and Muller2012). Increasing H contents also lowers the solidus temperature of the mantle, promoting melt formation and magmatism (Hirschmann, Reference Hirschmann2006). Decades of research on the water content of the mantle revealed that water is distributed heterogeneously throughout the mantle, and its concentration in any single position might also vary over geological timescales (e.g. Peslier et al., Reference Peslier, Schönbächler, Busemann and Karato2017). Water is added to the mantle by subduction-related fluids. Its concentration can also be modified by contributions delivered from deeper in the mantle carried upwards by metasomatic fluids, silicate melts or carbonatitic melts (Williams and Hemley, Reference Williams and Hemley2001). Water loss occurs during melting because trace H behaves as an incompatible element (Aubaud et al., Reference Aubaud, Hauri and Hirschmann2004). Diffusion attempts to homogenise water concentrations locally, but diffusivities might be outpaced by these mantle mechanisms (Shaw et al., Reference Shaw, Hauri, Behn, Hilton, Macpherson and Sinton2012; Marshall et al., Reference Marshall, Lassiter and Barnes2018). Together those processes create and maintain H and D heterogeneities in the mantle, which in turn, regulate the mantle’s geochemical properties, geodynamic behaviour and water-storing capacity.
The ratio of the isotopes of D and H is the hydrogen isotope composition of a specimen, commonly reported as δD (in per mille or ‰), where δD is reported with respect to a reference standard. Hydrogen isotopes are used to understand the behaviour of water as a fluid, gas or dissolved in minerals. Hydrogen isotope ratios can be used to better understand geological processes involved in melting, degassing, dehydration and metasomatic hydration. Because of their large mass difference, H isotopes can fractionate to a considerable extent. In one approach, the δD of mantle minerals has been measured to place constraints on the origin of Earth’s water reservoirs and learn about active processes in Earth’s primeval history (e.g. Hallis et al., Reference Hallis, Huss, Nagashima, Taylor, Halldórsson and Hilton2015). Other studies investigated mantle domains in which the δD values in mantle minerals could have been modified by episodes of tectonism or metasomatism (Shaw et al., Reference Shaw, Hauri, Behn, Hilton, Macpherson and Sinton2012; Walowski et al., Reference Walowski, Wallace, Hauri, Wada and Clynne2015; Banerjee et al., Reference Banerjee, Kyser and Mitchell2018; Moine et al., Reference Moine, Bolfan-Casanova, Radu, Ionov, Costin and Korsakov2020). Data resulting from such samples must be interpreted in the context of potential isotopic modifications introduced by melting, volcanism, diffusion or alteration.
In the present study, the authors combined NanoSIMS with traditional Fourier-Transform Infrared Spectroscopy (FTIR) to analyse D and H in domains of single, nominally anhydrous olivine and orthopyroxene crystals. Importantly, the olivine and orthopyroxene are protogenetic inclusions hosted within diamonds derived from the cratonic lithospheric mantle of northern South America. Diamond-hosted mineral inclusions are important to study because they provide exclusive evidence to understand the asthenosphere and lithospheric mantle (Stachel et al., Reference Stachel, Aulbach and Harris2022). Inclusions in diamond were first studied by prospecting geologists seeking to learn where diamonds form so they could better discover economic deposits. Because diamond acts as a protective containment vessel that isolates the inclusions from alteration they are now used as a primary constraint on the composition, age, and behaviour of the mantle (e.g. Shirey et al., Reference Shirey, Cartigny, Frost, Kshav, Nestola and Pearson2013). Inclusion mineralogy reveals a paragenesis, which encompasses a diamond’s petrological and tectonic history (Stachel and Harris, Reference Stachel and Harris2009). Major element, trace element and stable isotope compositions of inclusions can be used for geothermobarometric assessments of diamond crystallisation, phase stability, to establish the geothermal gradient, and to infer the nature of metasomatic fluids that migrate through the mantle (Walter et al., Reference Walter, Thomson and Smith2022). Radiogenic isotopes in diamond-hosted inclusions are used to determine the age of diamond crystallisation and/or eruptive emplacement (Smit et al., Reference Smit, Timmerman, Aulbach, Shirey, Richardson, Phillips and Pearson2022). Our NanoSIMS approach allowed us to measure the stable isotope compositions of hydrogen in ∼500 µm3 domains of single crystals that were probably shielded from post-entrapment modification by their protective host diamond jackets. We studied these diamond-hosted silicate inclusions to understand better the origin of the Earth’s water and metasomatic processes that occur in the mantle. We found that the diamond-hosted olivine and orthopyroxene from the lithospheric mantle of northern South America preserve a population average δD of –31 ± 56‰ (VSMOW). This value is D-enriched relative to bulk mantle (Kyser and O’Neil, Reference Kyser and O’Neil1984; Dixon et al., Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017). Our results represent a diamond-protected estimate of the composition of the regional mantle >2 billion years ago, which differs from present day expectations. Alternatively, the δD of the olivine and orthopyroxene inclusions might reflect the isotopic composition of fluids that preceded and/or precipitated the host diamonds. In this scenario the nominally anhydrous inclusions sample spatially restricted domains overprinted by metasomatic, carbonatitic or silicate fluids.
Geological setting
The authors collected 665 diamonds from Guyana during field work and with the assistance of artisanal miners and a local diamond merchant (Fig. 1). All of the diamonds were recovered from placer deposits along alluvial drainages and Pleistocene terraces within the western Guyana Amazon rainforest (Bassoo et al., Reference Bassoo, Befus, Liang, Forman and Sharman2021) (Fig. 1b). These predominantly colourlesss diamonds range from 0.3–2.7 mm in diameter, and mainly occur as octahedra and dodecahedra (Fig. 1c). Most stones present evidence for mild abrasion during alluvial transport. Detrital zircon geochronology of the source placer deposits is dominated by ages between 2.0 and 2.2 Ga (Bassoo et al., Reference Bassoo, Befus, Liang, Forman and Sharman2021). This suggests that the diamonds were eroded from the sedimentary Paleoproterozoic Roraima Supergroup. The diamonds were probably emplaced during an unidentified episode of kimberlite-related volcanism that erupted through the Guiana Shield more than 2 billion years ago. That such provenance removes some geological context is acknowledged, but these are the samples that were available for this pilot study.
(a) Topographic map of Guyana in northern South America using Shuttle Radar Topography Mission. (NASA/JPL/NIMA). Diamonds in this study were collected in the northwestern third of the country. (b) Author R. Bassoo collecting mineral samples from Pleistocene terrace gravels. The peak in the background is a tepui produced by the cliff-forming Roraima Formation. (c) Examples of the form, size and optics of our collection of rough Guyanese diamonds.

Approximately 10% of the Guyanese placer diamonds contained silicate inclusions (Fig. 2). Olivine occurs most commonly, followed by orthopyroxene. Both minerals range in size from 5–500 µm. We do not know the crystallisation age of the diamonds in our collection, but we consider 2 Ga to be a minimum age for inclusion entrapment because of the co-occurrence with 2.0–2.2 Ga detrital zircons. The most common inclusions reflect a harzburgitic paragenesis from the cratonic lithospheric mantle (Bassoo and Befus, Reference Bassoo and Befus2021). The diamond-hosted inclusions may be environmental recorders of the early subcontinental lithospheric mantle of the Guiana Shield.
Photomicrographs of Guyanese diamond-hosted inclusions. (a) Olivine inclusion Guy003 in the octahedral diamond host, prior to grinding and polishing. Panels (b) and (c) show other mineral inclusions in the polished wafer of a different diamond. The olivine in (b) is exposed at the surface.

Materials and methods
The authors selected ∼35 inclusion-bearing diamonds for analysis. These were chosen because they hosted colourless silicate inclusions that were >50 µm in diameter, and the inclusions were well positioned within the diamond’s interior, i.e. unaffected by alteration and terrestrial contamination. The mineralogy of each inclusion was confirmed by matching their Raman spectra with those in the RRUFF spectral database (Lafuente et al., Reference Lafuente, Downs, Yang, Stone, Armbruster and Danisi2015). Using a high rpm diamond-cutting scaife, we fashioned each diamond into a doubly-polished plate that fully encased the target inclusion. These plates were 450–1300 µm in thickness. We then analysed the fully encased inclusions using FTIR. Following FTIR analysis, we returned the diamonds to the scaife and carefully polished one side of the diamond wafer until a silicate inclusion was exposed. The sectioned, singly-exposed inclusion was then ready for additional analyses. Only 11 of the initial ∼35 inclusion-bearing diamonds survived this demanding sample-preparation process. Cathodoluminescence and electron microprobe analyses for these specimens were described previously by Bassoo and Befus (Reference Bassoo and Befus2021), who established the diamonds’ interior structure and major element composition of the inclusions. These techniques do not play a prominent role in this study and so their description is not repeated here. FTIR analyses were also described by Bassoo and Befus (Reference Bassoo and Befus2021), and these measurements are central to this narrative. The lattice-bound hydrogen contents (H2O ppm) of silicate inclusions were estimated using unpolarised FTIR spectra. Orientated, polarised measurements of the three crystallographic directions of the inclusions were not possible (e.g. Bell et al., Reference Bell, Ihinger and Rossman1995). Absorbance was measured as total integrated area in the OH bond active range between 3100 and 3800 cm–1 using a linear baseline. We then used the Beer-Lambert equation to calculate H2O ppm (using absorption coefficients from Bell et al., Reference Bell, Ihinger and Rossman1995, Reference Bell, Rossman, Maldener, Endisch and Rauch2003). We applied a 3× absorption coefficient multiplier calibration for unpolarised IR spectra (e.g. Libowitzky and Rossman, Reference Libowitzky and Rossman1997). This approach generates concentrations that are ∼2/3 the value calculated from the calibration by Withers et al. (Reference Withers, Bureau, Raepsaet and Hirschmann2012).
The polished diamond wafers were next mounted for nano secondary ion mass spectrometry analyses (NanoSIMS). The diamond wafers were arranged loosely on indium in a 1-inch aluminium holder. The samples and indium were then firmly, yet carefully, pressed using a vice to produce a flat surface. The mount was then sputtered with a gold coat. The hydrogen isotope compositions of the diamond-hosted olivine and orthopyroxene inclusions were measured by analysing D and H using the Cameca Ametek NanoSIMS 50L instrument at Arizona State University. All D/H ratios are reported in standard δD (‰) notation relative to Vienna Standard Mean Ocean Water. Operating conditions included a focused, ∼250 pA Cs+ primary beam accelerated at 16 keV. Before taking measurements on the samples, we first assessed the beam size at 2 nA, 500 pA and 250 pA, confirming that the 250 pA setting generated the desired small probe suitable for the analysis of the silicate phases. The target minerals were pre-sputtered for 5–6 min to remove surface contamination and implant Cs to enhance emission of secondary ions. Secondary ions of 1H–, D–, and 12C– were collected in the multicollection mode with ∼6% blanking using electronic gating with entrance slit 1 or no aperture slit. The 12C– signal was monitored to ensure that the analysed areas were free of cracks or pits. A typical analysis took ∼2 h to complete with 225 cycles (45 blocks of five measurements per block) (Fig. 3). We measured two secondary standards: pyroxene PMR53 and basaltic glass ALV519 (Bell et al., Reference Bell, Ihinger and Rossman1995; Bell and Ihinger, Reference Bell and Ihinger2000; Le Voyer et al., Reference Le Voyer, Cottrell, Kelley, Brounce and Hauri2015). Pyroxene PMR53 is the ‘matrix equivalent’ isotopic reference standard to our silicates because of similar crystal structure, composition and water content. Using PMR53 the isotopic instrumental mass fractionation was 0.64 for the entire analytical session of 7 days. Standards were analysed at the start, end and throughout the analytical session. The 1σ uncertainty of our δD measurements ranged from 71 to 74‰.
Example NanoSIMS analysis. (a) NanoSIMS ablation pit in polished olivine inclusion. (b–d) Counts of H, D and ratio of D/H in panels.

Results
The diamond-hosted olivine analysed ranged in size from 50 µm × 55 µm to 100 µm × 210 µm, and the orthopyroxene inclusions ranged in size from 30 µm × 40 µm to 50 µm × 80 µm (Table 1). All inclusions are colourless, prismatic cubo-octohedrons, which might be elongated along one direction (Fig. 2). They occur in isolation or grouped in clusters. The host diamonds preserve green, blue, turquoise, yellow and inert cathodoluminescent growth bands (Fig. 4). These bands wrap the inclusions. We see no evidence for healed cracks near the inclusions in plane light, reflected light or cathodoluminescence.
Cathodoluminescence photomicrographs reveal the internal structure of the diamond hosts near olivine and orthopyroxene inclusions.

Dimensions, composition, FTIR parameters, and the hydrogen isotopic composition of standards and diamond-hosted silicate inclusions

Notes: Values in parentheses represent 1σ standard deviations. – indicates not detected; H2O (ppm) and Mg# are from Bassoo and Befus (Reference Bassoo and Befus2021); the fractionation factor for PMR53 for this analytical session was 0.64.
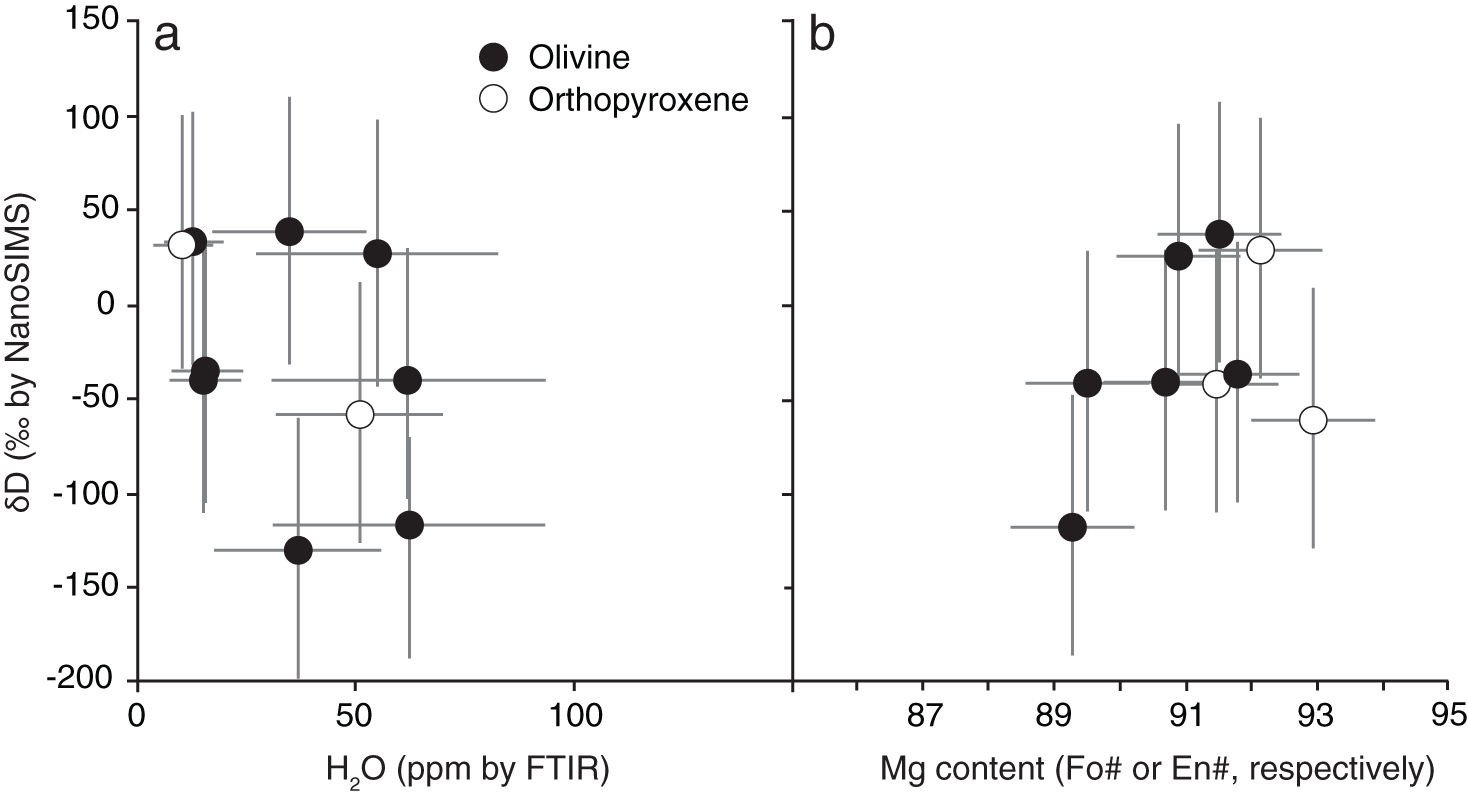
The olivine inclusions are forsterite that range from Fo89 to Fo92. The three orthopyroxene inclusions are enstatite and yield a homogeneous composition of En92Fs7Wo1. FTIR spectra present small absorbances for both olivine and orthopyroxene (Table 1). Olivine inclusions contain 13 ± 3 to 62 ± 12 ppm H2O whereas the two orthopyroxene we analysed contain 10 ± 2 to 51 ± 10 ppm H2O (Table 1). Water contents in the inclusions do not correlate with their size, or their position, in the host diamond. D/H ratios were very low and ranged from 9×10–5 to 1×10–4. The hydrogen isotopic composition of the olivine and orthopyroxene together range from –131 ± 71‰ to 26 ± 73‰ (Fig. 5, Table 1). The average of the 11 inclusions is –31 ± 59‰ (1σ of population). Separated by phase, the eight olivine inclusions encompass the full spread from –131±71‰ to 38 ± 73‰, and average –34 ± 65‰. The three orthopyroxenes range from –60±72‰ to 29 ± 73‰, and average –24 ± 47‰. We do not distinguish D/H fractionation between olivine and orthopyroxene (e.g. Δol-opx, Bell and Ihinger, Reference Bell and Ihinger2000), although we acknowledge the small number of samples here in our pilot study. δD does not correlate with inclusion size, position in the host diamond, inclusion composition, or FTIR-derived H2O content (Fig. 4).
Hydrogen isotope compositions (δD) of diamond-hosted olivine (black circles) and orthopyroxene (white circles) plotted against (a) H2O measured by FTIR and (b) mineral composition (Mg#). Not all δD have corresponding H2O or Mg#. 1σ error bars are shown in grey.

Discussion
Diamond-hosted olivine and orthopyroxene inclusions from the harzburgitic subcontinental lithospheric mantle of the Guiana Shield are relatively dry, containing only a few ppm to <165 ppm lattice-bound H2O (Bassoo and Befus, Reference Bassoo and Befus2021). The water content of the inclusions is similar to that measured in lithospheric xenoliths and diamond-hosted silicate inclusions from other cratons (Demouchy and Bolfan Casanova, Reference Demouchy and Bolfan-Casanova2016; Peslier, Reference Peslier, Schönbächler, Busemann and Karato2017; Chin and Palin, Reference Chin and Palin2022; Curtolo et al., Reference Curtolo, Day, Innocenzi, Bolfan-Casanova, Pamato and Falvard2025).
We analysed the D/H ratios of olivine and orthopyroxene inclusions to learn more about the lithospheric mantle in the cratonic root of the Guiana Shield. δD is commonly used to infer geological processes because the large mass difference between D and H produces significant fractionation. δD may also vary significantly between reservoirs exposed to disparate histories. Our overarching hypothesis was that diamond-encapsulated olivine and orthopyroxene would have a δD indistinguishable from previous estimates of the bulk upper mantle (Kyser and O’Neil, Reference Kyser and O’Neil1984; Poreda et al., Reference Poreda, Schilling and Craig1986; Dixon et al., Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017). The olivine and orthopyroxene inclusions from the harzburgitic subcontinental lithospheric mantle of the Guiana Shield contest that hypothesis. The inclusions preserve a δD of –31 ± 59‰ (1σ of population average) although we acknowledge that individual inclusions span a wide range from –131±71‰ to 26 ± 73‰ (1σ analytical errors) (Fig. 4). Most of the inclusions fall between –60 and 40‰, which is more D-enriched than hotspot basalts, mid-ocean ridge basalts, and nominally anhydrous minerals. Olivine inclusions Guy003 and Guy580 show a δD of –118‰ and –131‰, respectively, and are the most D-depleted outliers in the dataset, overlapping with typical nominally anhydrous minerals. We present our discussion based on the population average rather than a point-by-point basis. We interpret individual variability between samples, especially the more negative endmembers, to reflect natural variability mixed by kimberlitic melts sampling along the full length of their eruptive transect. Our analytical uncertainties are large, owing largely to trace amounts of water in the minerals and the small areas measured. We emphasise that our uncertainties are typical of D/H measurements of nominally anhydrous materials using NanoSIMS.
As δD does not correlate with H2O content (Fig. 5). The lack of trends between δD and H2O suggests that isotopic fractionation of D and H by geological processes are not apparent in the sample suite (e.g. Kyser and O’Neil, Reference Kyser and O’Neil1984). We find no evidence for diffusive loss of H from the inclusions, which would produce a trend to higher δD and lower H2O. The diamond hosts might have shielded the inclusions from diffusive H loss, and/or the inclusions could not have experienced a concentration gradient required for diffusive re-equilibration (Demouchy and Bolfan Casanova, Reference Demouchy and Bolfan-Casanova2016; Taylor et al., Reference Taylor, Logvinova, Howarth, Liu, Peslier and Guan2016; Vangu et al., Reference Vangu, Bureau, Khodja, Charrondiere, Esteve and Béneut2023; Curtolo et al., Reference Curtolo, Day, Innocenzi, Bolfan-Casanova, Pamato and Falvard2025). We attribute the compositional spread to heterogeneous eruptive sampling. Kimberlites are known to deliver xenoliths/xenocrysts to the Earth’s surface from the full length of their eruptive transect. We should thus expect the Guyanese diamonds to represent a composite sample from the subcratonic lithospheric mantle.
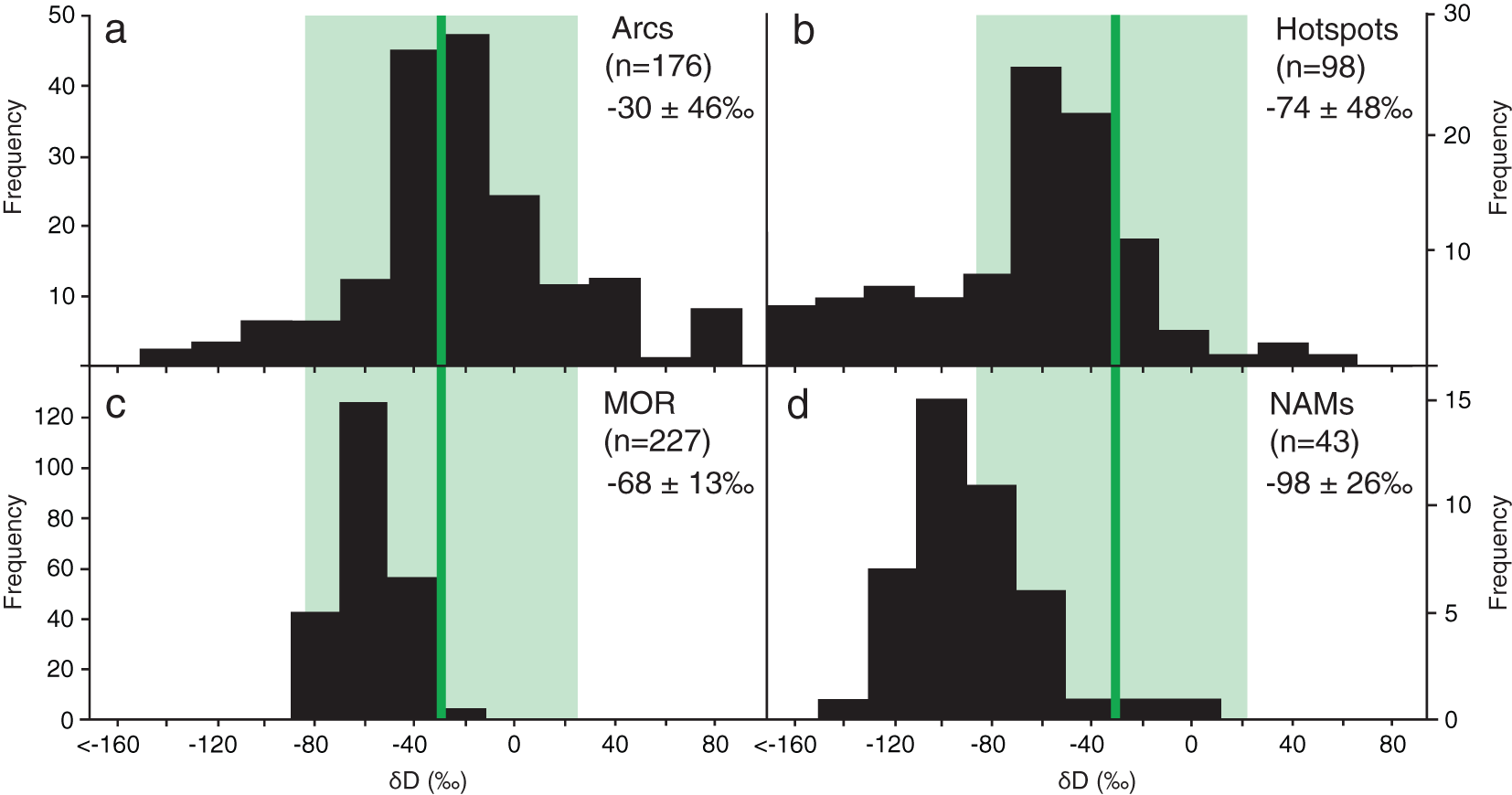
The population of Guyanese diamond-hosted inclusions preserve a hydrogen isotopic composition that is, on average, tens of per mille more positively D-enriched, than bulk upper mantle. They are also, on average, more positive, D-enriched than hotspot basalts, mid-ocean ridge basalts, or other mantle nominally anhydrous minerals (NAMs, Fig. 6). The δD of the diamond-hosted inclusions is most similar to the hydrogen isotopic composition of arc basalts (Fig. 6a). To offer context to our first-of-its-kind data set, we offer three separate explanations for elevated D/H in the diamond-hosted inclusions from the lithospheric mantle of the Guiana Shield. Because of the similarity with arc basalts, all three explanations rely upon the significant isotopic fractionation induced by hydrous mineral breakdown. When hydrous minerals break down they preferentially liberate D, producing a D-enriched fluid phase, which we contend is important here (Suzuoki and Epstein, Reference Suzuoki and Epstein1976; Wallace, Reference Wallace2005). The residual mineral(s) become enriched in H, and these minerals are not part of this study. Almost all water is removed by prograde dehydration at subduction zones. Some trace amounts of water could remain within the subducting nominally anhydrous minerals, and be a source for D-enriched fluids during later mantle dynamics.
Literature compilation of previously published hydrogen isotope compositions of mafic glasses from arcs (a), mantle plume hotspots (b), and mid-ocean ridge systems (c), as well as nominally anhydrous mantle minerals (d). The range of δD (with an average of –31± 59‰) for Guyanese diamond-hosted inclusions are superimposed in green. Arc data compiled from Shaw et al. (Reference Shaw, Hauri, Fischer, Hilton and Kelley2008), Bouvier et al. (Reference Bouvier, Métrich and Deloule2010), Gaetani et al. (Reference Gaetani, O’Leary, Shimizu, Bucholz and Newville2012), Bucholz et al. (Reference Bucholz, Gaetani, Behn and Shimizu2013), Métrich and Deloule (Reference Métrich and Deloule2014), Walowski et al. (Reference Walowski, Wallace, Hauri, Wada and Clynne2015), Loewen et al. (Reference Loewen, Graham, Bindeman, Lupton and Garcia2019), Venugopal et al. (Reference Venugopal, Moune, Williams-Jones, Druitt, Vigouroux, Wilson and Russell2020). Hotspots data from Hauri (Reference Hauri2002) Kingsley et al. (Reference Kingsley, Schilling, Dixon, Swart, Poreda and Simons2002), Aubaud et al. (Reference Aubaud, Pineau, Hékinian and Javoy2005), Hallis et al. (Reference Hallis, Huss, Nagashima, Taylor, Halldórsson and Hilton2015), Wu et al. (Reference Wu, Guo, Wang, Wilde and Fan2020). Midocean ridge data from Kyser and O’Neil (Reference Kyser and O’Neil1984), Poreda et al. (Reference Poreda, Schilling and Craig1986), Kingsley et al. (Reference Kingsley, Schilling, Dixon, Swart, Poreda and Simons2002), Cartigny et al. (Reference Cartigny, Pineau, Aubaud and Javoy2008), Bindeman et al. (Reference Bindeman, Kamenetsky, Palandri and Vennemann2012), Clog et al. (Reference Clog, Aubaud, Cartigny and Dosso2013), Dixon et al. (Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017), Loewen et al. (Reference Loewen, Graham, Bindeman, Lupton and Garcia2019). Nominally anhydrous mantle mineral data from Bell and Ihinger (Reference Bell and Ihinger2000), Huimin et al. (Reference Huimin, Qunke, Deloule and Xiaozhi2005), O’Leary (Reference O’Leary2007), Fourel et al. (Reference Fourel, Lécuyer, Demeny, Boulvais, Lange, Jacob and Kovacs2017), Moine et al. (Reference Moine, Bolfan-Casanova, Radu, Ionov, Costin and Korsakov2020).

Our first explanation is that the upper mantle could simply be more heterogeneous with respect to δD than commonly thought. Many studies have proposed a δD of –80 ± 10‰ for the upper Earth’s mantle (e.g. Kyser and O’Neil, Reference Kyser and O’Neil1984; Poreda et al., Reference Poreda, Schilling and Craig1986). However, Dixon et al. (Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017) argued the upper mantle is heterogeneous on the basis of the δD of mafic glasses from midocean ridge systems. Individual mid-ocean ridge segments preserve different δD values (Dixon et al. Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017). Subducting slabs might sometimes carry H2O to great depths in the mantle. If that H2O is liberated at depths ranging from 200 to >400 km, then it would be available to enrich the mantle in D far from magmatic arcs. This process would produce a more heterogeneous mantle in δD, and the resulting melts from those hydrated mantle domains would be D-enriched. The δD in Guyanese inclusions could be a vestige of such deeply subducted H2O, that either modified the ancient cratonic mantle beneath northern South America or was a product of initial cratonisation by slab imbrication (e.g. Lee et al., Reference Lee, Luffi and Chin2011). This subduction hypothesis implies that the slabs ‘parked’ beneath Guyana were cold because they allowed serpentine and other hydrous minerals to remain stable to great depths. Eclogitic parageneses would be expected to be more associated with slab imbrication, but most of the Guyanese silicate inclusions reflect a relatively shallow peridotite paragenesis instead (e.g. Dixon et al., Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017; Bassoo and Befus, Reference Bassoo and Befus2021).
Our second explanation is the temporal evolution of D/H ratios in Earth’s mantle. Shaw et al. (Reference Shaw, Hauri, Fischer, Hilton and Kelley2008) proposed that the δD of the mantle has decreased through time because of convective assimilation of subduction inputs. In their D/H exchange model they show mantle δD evolved gradually to more negative, lighter values, over Earth’s history. The Guyanese inclusions may provide an opportunity to evaluate this idea: as we know that entrapment was prior to 2 Ga, and the inclusions preserve a δD of –31 ± 59‰. The population average of the diamond-hosted inclusions thus supports an evolution of H-enrichment and D-depletion in the mantle. Shaw et al. (Reference Shaw, Hauri, Fischer, Hilton and Kelley2008) calculated D/H evolution rates that range from –10‰ to –40‰ per billion years. The Guyanese inclusions yield a D/H evolution rate of –15‰ to –25‰ per billion years, in agreement with the range predicted by Shaw et al. (Reference Shaw, Hauri, Fischer, Hilton and Kelley2008). Such D/H exchange calculations contextualise these NanoSIMS measurements in the framework of an existing global model. If applicable, then the Palaeoproterozoic, >2Ga old diamond-hosted inclusions can represent the oldest direct measurement of the mantle’s evolving D/H ratio. When individual samples are considered, this explanation is valid for those near the population average. A different model is required to explain samples with the most negative and most positive δD values.
The third explanation is that the elevated D/H in the diamond-hosted inclusions might reveal localised metasomatic D-enrichment rather than steady, mantle-wide evolution towards H-enrichment (Thomassot et al., Reference Thomassot, Cartigny, Harris and Viljoen2007). Guyanese olivine inclusions are Mg-depleted (∼Fo90). Both olivine and orthopyroxene are enriched in Mn (Bassoo and Befus, Reference Bassoo and Befus2021). The major and trace element compositions of the inclusions provide geochemical evidence for metasomatic alteration. The metasomatising fluid altered the protogenetic inclusions prior to, and during, diamond growth (e.g. Stachel et al., Reference Stachel, Chacko and Luth2017; Bruno et al., Reference Bruno, Ghignone, Aquilano and Nestola2024). Because the olivine and orthopyroxene inclusions contain only a few 10s of ppm of lattice-bound H2O, the evidence suggests that the metasomatism was produced by a water-poor fluid. We speculate here that the hydrogen isotopic composition observed in diamond-hosted inclusions could be produced during hydration by metasomatic fluids produced by mineral dehydration from deeper phases in the mantle-transition zone, which is known to be a large reservoir of water. High-pressure phases like ringwoodite and wadsleyite can hold 1–3 wt.% H2O (Kohlstedt et al., Reference Kohlstedt, Keppler and Rubie1996; Williams and Hemley, Reference Williams and Hemley2001; Mosenfelder et al., Reference Mosenfelder, Bureau and Withers2024). Those minerals dehydrate if they become unstable in response to mantle advection (e.g. Bercovici and Karato, Reference Bercovici and Karato2003). Dehydration produces isotopic fractionation, and the resulting fluid from the mantle transition zone should be D-enriched, in common with fluids released from the slab at subduction zones. Kinetic and equilibrium isotopic fractionation between water and nominally anhydrous minerals at mantle conditions remain unknown, but are an active area of investigation. Equilibrium modelling suggests that the fractionation should be ∼±5‰ at mantle temperatures (Suzuoki and Epstein, Reference Suzuoki and Epstein1976; Bell and Ihinger, Reference Bell and Ihinger2000). If correct, then the Guyana D-enriched diamond-hosted inclusions might be a tracer of the hydrogen isotopic composition of fluids derived from the transition zone, and subsequently provide a pioneering constraint on water and metasomatic fluids from a relatively unknown portion of the mantle. In summary, for this third idea, we hypothesise that the scatter observed in δD and H2O within Guyanese diamond-hosted inclusions reflect distinct metasomatic fluids. The fluid overprint varies as the fluids vary in space, time and composition (e.g. Thomassot et al., Reference Thomassot, Cartigny, Harris and Viljoen2007; Jaques et al., Reference Jaques, Luguet, Smith, Pearson, Yaxley and Kobussen2018). Separate rising kimberlitic melts then sampled diamonds from the various depths and mixed them thoroughly as they were eruptively transported to the Earth’s surface.
Conclusions
The D/H measurements presented here are the first of their kind. We offer no single, conclusive interpretation for why the diamond-hosted inclusions are D-enriched relative to previous estimates of the bulk upper mantle. A heterogeneous upper mantle with respect to D/H in both time and space might be the simplest explanation (e.g. Dixon et al., Reference Dixon, Bindeman, Kingsley, Simons, Le Roux and Hajewski2017). Subduction, partial melting and mantle advection at hotspots and tectonic boundaries can change the abundance and isotopic composition of water in the mantle. Such a mantle-scale explanation may, or may not, be expanded by including effects introduced by localised fluids. Diamonds crystallise from metasomatic silicate, carbonatitic, or C-O-H-N-S fluids. The D-enrichment observed in the diamond-hosted orthopyroxene and olivine might have been produced by metasomatic event(s) in the mantle beneath northern South America. We acknowledge that our diamonds are from placers with a modest petrological context compared to other diamond sources. Future studies could test our research using diamonds and suites of xenoliths from well-characterised primary kimberlite/lamproite source(s).
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2026.10191.
Acknowledgements
We thank the Editors Roger Mitchell and Caterina Melai, along with Tony Withers and an anonymous reviewer for guidance and edits.
Financial statement
We acknowledge the use of the Arizona State University SIMS Facility supported by a grant from the US National Science Foundation (EAR-1819550).
Competing interests
The authors declare none.