Introduction
Some complex CHDs are associated with a smaller brain size at birth and neurodevelopmental impairment later in life. Reference Clouchoux, du Plessis and Bouyssi-Kobar1–Reference Omann, Kristensen, Tabor, Gaynor, Hjortdal and Nyboe3 Although incompletely understood, both genetic and circulatory factors are likely involved. Reference Hoang, Goldmuntz and Roberts4–Reference Vena, Manganaro and D'Ambrosis6 Apart from affecting brain development, fetal circulatory alterations may also affect the development and size of other organs. Both fetuses with Tetralogy of Fallot and/or pulmonary stenosis have smaller lung volumes compared to fetuses without CHD. Reference Mlczoch, Schmidt and Schmid7–Reference Guo, Liu, Gu, Zhang, Sun and He9 In both, reduced pulmonary blood flow during fetal development may explain impaired lung growth. Reference Sun, van Amerom and Marini10 In contrast, the fetal circulation in transposition of the great arteries is characterised by a relative abundance of well-oxygenated blood being directed towards the lungs. In a small study, unable to differentiate between those with or without a ventricular septal defect, we recently described larger lung volumes in fetuses with transposition of the great arteries compared to healthy controls. Reference Krogh, Ringgaard and Kelly11
In this early report, we hypothesised that the existence of a ventricular septal defect might alter fetal blood flow, and subsequently organ development. The aim of the current study was to compare the lung, liver, kidney and intracranial volume in fetuses with transposition of the great arteries with and without a ventricular septal defect with healthy controls.
Materials and methods
This was a retrospective, single-centre, cohort study utilising fetal MRI scans from two prior longitudinal MRI studies, originally looking at fetal haemodynamics and brain and body growth. These studies were conducted from 2016 to 2017 Reference Jørgensen, Tabor and Rode12 and 2021 to 2023, Reference Hellmuth, Jørgensen and Wright13 at the Center of Pregnancy and Fetal Medicine, Copenhagen University Hospital—Rigshospitalet, Denmark.
Population
Inclusion criteria for both studies were as follows: no known maternal health constraints, age between 18 and 40 years, body mass index between 18 and 30, singleton pregnancy, no smoking in the previous 6 months, no previous preterm delivery (<37 weeks) or a birth weight <2500 g, and normal first-trimester screening. Exclusion criteria for both studies were any malformation other than CHD detected at the 18–21-week malformation scan, any abnormal genetic findings on a chromosomal microarray, first-trimester risk assessment for trisomy 21 greater than 1:300, implanted magnetic material (cerebral ferromagnetic clips, pacemaker/ICD-unit) or severe claustrophobia.
All fetuses from the early study conducted between 2016 and 2017 were included, comprising five fetuses diagnosed with transposition of the great arteries and 10 healthy ones. Reference Jørgensen, Tabor and Rode12 From the later study conducted during the period 2021–2023, all fetuses who had been enrolled at the time of this study’s execution were included, comprising six fetuses with transposition of the great arteries and twelve healthy controls. Reference Hellmuth, Jørgensen and Wright13 This resulted in a cohort of eleven fetuses with transposition of the great arteries, without chromosomal abnormalities or concomitant extracardiac disease, and 22 healthy controls, which were scanned between 1 and 3 times at gestational age 27 through 38 weeks. Of the 11 fetuses with transposition of the great arteries, six had a ventricular septal defect (transposition of the great arteries +ventricular septal defect), and five had an intact ventricular septum (transposition of the great arteries +intact ventricular septum).
The studies were approved by the Danish Data Protection Agency (30-1404 and P-2020-711) and by the Regional Research Ethics Committees (H-4-2014-078 and H-20015468). All participating mothers gave written informed consent.
MRI acquisition
A 1.5 tesla MR scanner (Siemens Avanto, Siemens, Erlangen, Germany) was used for all scans. The MR sequence was a 3D steady-state-free-precession sequence with the following sequence parameters: a scan duration (TA) of 14–17s, slice thickness of 2 mm, voxel size of 0.8 × 0.8 × 2.0 mm3, echo time (TE) of 1.74 ms, repetition time (TR) of 4.00 ms, flip angle of 54°, and field-of-view of 400 × 325 mm.
Estimated fetal weight
Estimated fetal weight was calculated by MRI measurements of total fetal volume, using the Baker formula. Reference Baker, Johnson and Gowland14
Organ volumetry
All organ volume measurements were performed in the open-source software platform, 3D Slicer (https://www.slicer.org/). A blinded observer (EK) measured total intracranial, lung, liver, and kidney volumes. Total intracranial volume was measured by tracing regions of interest and computing the area around the inside of the cranium, covering the brain, cerebellum, meninges, and cerebrospinal fluid in every slice of the fetal MRI scan. Lung volumes were measured by tracing the lung border against the thorax wall, the diaphragm, and the mediastinum. Liver volumes were traced based on the border to the diaphragm and the other abdominal organs. Kidney volumes were obtained the same way, based on the border between the kidney and the other abdominal organs. Both kidneys were measured in all fetuses, and the sum of the two was used in the analysis. The fetal organ volumes were then calculated by multiplying the total regions of interest areas by the slice thickness of the fetal MRI. Below is an example of defining total intracranial volume, lung, liver, and kidney regions of interest and computing the volume in 3D Slicer (Figure 1).
(a) Fetal MR image, (b) organ volumetry measurements of total intracranial volume (green), lungs (yellow), liver (red), and kidney (blue and grey), and (c) a 3D model of all organ volumes.

Statistics
For maternal and fetal characteristics, all continuous variables are shown as median (interquartile range) and number (%) for all categorical variables (Table 1). A statistical comparison between the three groups was performed, using either a t-test or a Wilcoxon rank-sum test depending on whether data followed a normal distribution.
Maternal and fetal characteristics of the groups with and without transposition of the great arteries. Age, BMI, EFW, and GA are shown as medians (interquartile range); the remaining are absolute numbers (%). CHD, congenital heart defect, n, number of fetuses, TGA, transposition of the great arteries, VSD, ventricular septal defect, IVS, intact ventricular septum, BMI, Body Mass Index, smoking, smoking during pregnancy, alcohol, alcohol during pregnancy, EFW, estimated fetal weight at the time of MRI, GA, gestational age at the time of MRI. *(n = 4). No statistically significant differences in maternal or fetal characteristics were found between the control group, TGA+VSD and TGA+IVS

To assess organ volume, mixed effects regression analysis was used with organ volume as the outcome. A random intercept was fitted for the individual fetus ID to account for repeated measurements of fetuses. Fixed effects for all initial fetal and maternal characteristics were included: fetal characteristics (estimated fetal weight, gestational age, ventricular septal defect/intact ventricular septum status, and fetal sex) and the maternal characteristics (maternal age, BMI, ethnicity, smoking during pregnancy, drinking alcohol during pregnancy, and parity). Backwards elimination was used for model selection. The included variables in the final regressions, following backwards elimination, were ventricular septal defect/intact ventricular septum status, estimated fetal weight, and gestational age. All analyses were performed using R Statistical Software (v. 4.1.3; R Core Team, 2021) and the package lme4. Reference Bates, Mächler, Bolker and Walker15
Results
A total of 56 fetal MR scans were included in this study: 36 scans from 22 healthy fetuses and 20 scans from 11 fetuses diagnosed with transposition of the great arteries. Among the transposition of the great arteries group, eleven scans were from six fetuses with transposition of the great arteries+ventricular septal defect, and nine scans were from five fetuses with transposition of the great arteries+intact ventricular septum. The six transposition of the great arteries+ventricular septal defect fetuses underwent a total of eleven scans, while the five transposition of the great arteries+intact ventricular septum fetuses underwent a total of nine scans. Specifically, one fetus in each group (transposition of the great arteries+ventricular septal defect and transposition of the great arteries+intact ventricular septum) was scanned three times; three transposition of the great arteries+ventricular septal defect and two transposition of the great arteries+intact ventricular septum fetuses were scanned twice; and two fetuses in each group were scanned once. Maternal and fetal characteristics are shown in Table 1.
Fetal organ development
To depict the fetal organ volume development, organ volumes were plotted as a function of gestational age. This was done separately for lungs (Figure 2), total intracranial volume (Supplementary figure S1), liver (Supplementary figure S2), and combined kidney volume (Supplementary figure S3) in fetuses with transposition of the great arteries+ventricular septal defect, transposition of the great arteries+intact ventricular septum, and the healthy controls. Fetuses with transposition of the great arteries+intact ventricular septum had significantly larger lungs compared to healthy controls and fetuses with transposition of the great arteries+ventricular septal defect.
Lung volume as a function of gestational age. Blue dots represent fetuses with TGA and an intact ventricular septum (TGA+IVS), red dots represent fetuses with transposition of the great arteries and a ventricular septal defect (TGA+VSD), and grey dots represent healthy control fetuses. Solid lines represent linear mixed model regression analysis. TGA+IVS had significantly larger lungs throughout the studied gestational ages than the two other groups. No other known variables apart from gestational age and estimated fetal weight impacted the organ volumes.

No difference in total intracranial volume, liver, or combined kidney volume was found between the groups. Postnatally, one transposition of the great arteries+intact ventricular septum presented with a value of 7 on APGAR at 1 minute, and all other APGAR scores, including APGAR at 5 minutes, were above 7. None of the children in the cohort presented with evidence of pulmonary hypertension.
None of the maternal and fetal characteristics, apart from gestational age, estimated fetal weight, and the absence of a ventricular septal defect in a fetus with transposition of the great arteries, significantly affected lung volume, and only the presence or absence of a ventricular septal defect impacted organ size differently between fetuses with transposition of the great arteries. Additionally, none of the above-mentioned maternal and fetal characteristics, apart from gestational age and estimated fetal weight, significantly impacted total intracranial volume, liver volume, or kidney volume.
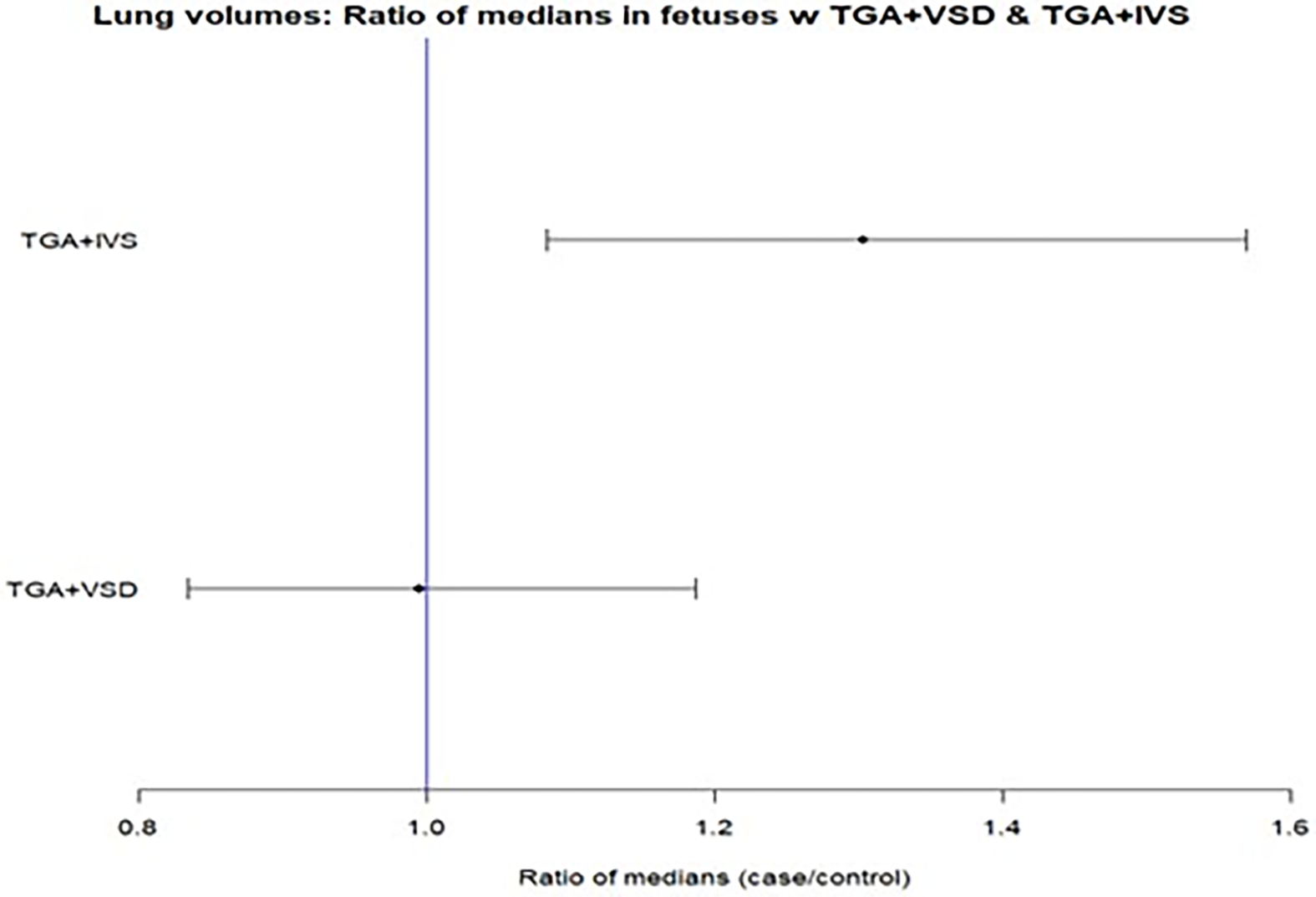
A forest plot was created to illustrate the difference in lung volume between fetuses with transposition of the great arteries+ventricular septal defect, transposition of the great arteries+intact ventricular septum, and healthy controls (Figure 3).
Forest plot showing the median lung size with 95% confidence intervals for the estimated differences in fetuses with transposition of the great arteries and an intact ventricular septum (TGA+IVS) and those with a ventricular septal defect (TGA+ VSD), compared to healthy controls. Values are adjusted for estimated fetal weight and gestational age.

When corrected for estimated fetal weight and gestational age, the ratio of median lung size in fetuses with transposition of the great arteries+intact ventricular septum compared to controls is 1.30 (95% CI: 1.08–1.57, p = 0.005), suggesting that median lung volume is on average 30% higher in fetuses with transposition of the great arteries+intact ventricular septum compared to controls. The lung volume of fetuses with transposition of the great arteries+ventricular septal defect was not different from that of the controls (median lung volume ratio 0.99 (95% CI: 0.83–1.19, p = 0.95)). There was no difference in total intracranial volume, liver volume, or combined kidney volume between transposition of the great arteries+ventricular septal defect, transposition of the great arteries+ intact ventricular septum, and healthy controls.
Discussion
In this preliminary study, we found significantly larger lung volumes in late-gestation fetuses with transposition of the great arteries+intact ventricular septum compared to healthy controls. When comparing the lung volume of those with transposition of the great arteries+ventricular septal defect with controls, no differences were found. The results indicate altered haemodynamics and circulatory factors play a role in the increased lung volume, since the changes were mitigated by the presence of a ventricular septal defect.
We observed a 30% increase in median lung volume in fetuses with transposition of the great arteries+intact ventricular septum compared to controls. However, the 95% CI for this estimate was wide, ranging from an 8% to 57% difference. We have previously found an increase in lung volume of 8.1 cm3/kg estimated fetal weight in eight fetuses with transposition of the great arteries compared to healthy controls, and the present study confirms and elaborates on our preliminary findings. Reference Krogh, Ringgaard and Kelly11 In contrast to our preliminary data, the number of fetuses included in this current study enables us to demonstrate that fetuses with transposition of the great arteries+ventricular septal defect have a lung size closer to normal than fetuses with transposition of the great arteries+intact ventricular septum, indicating that the perfusion pattern of the lungs in fetal life is important for normal organ size.
In fetuses with transposition of the great arteries+intact ventricular septum, the flow of well-oxygenated blood from the placenta runs, due to preferential streaming, through the oval foramen into the left side of the heart and the transposed pulmonary artery, while the more desaturated blood from the caval veins is directed to the right ventricle and through the transposed aorta to the brain. Reference Sun, van Amerom and Marini10,Reference Lachaud, Dionne and Brassard16 Increased blood oxygen saturation in the pulmonary circulation reduces pulmonary vascular resistance, Reference Accurso, Alpert, Wilkening, Petersen and Meschia17 and the consequent increase in pulmonary blood flow may explain the observed larger lung size. Using ultrasound and modelled blood flow based on MRI, higher blood flow to the lungs has been demonstrated in fetuses with transposition of the great arteries+ intact ventricular septum compared to both healthy controls and fetuses with transposition of the great arteries+ventricular septal defect. Reference Sun, van Amerom and Marini10,Reference Lachaud, Dionne and Brassard16 Normal transpulmonary flow is noticed in transposition of the great arteries with a ventricular septal defect. Reference Sun, van Amerom and Marini10,Reference Lachaud, Dionne and Brassard16 In other words, a ventricular septal defect may normalise fetal pulmonary blood flow in fetuses with transposition of the great arteries. Knowledge from CHD with reduced pulmonary flow supports the hypothesis that fetal blood flow and organ size are associated. In support of the hypothesis, fetuses with Tetralogy of Fallot or pulmonary stenosis have smaller lung volumes when compared to healthy controls. Reference Mlczoch, Schmidt and Schmid7–Reference Guo, Liu, Gu, Zhang, Sun and He9
It is well known that children born with a major CHD suffer from organ complications other than the heart and the brain, e.g., kidney insufficiency and pulmonary hypertension. Reference Hoskote and Burch18–Reference Miranda, Jain and Conolly20 With the findings of this study, it is interesting to speculate whether the increased fetal lung volume could be a part of pulmonary complications later in life. For instance, infants born with transposition of the great arteries are at risk of developing pulmonary hypertension. Reference Goissen, Ghyselen and Tourneux21,Reference Zijlstra, Elmasry and Pepplinkhuizen22 Furthermore, patients with transposition of the great arteries corrected by an arterial switch operation have reduced exercise capacity, which is mostly believed to be due to chronotropic incompetence, residual right ventricular outflow tract obstruction after surgery, narrowing of the main pulmonary arteries, and coronary abnormalities. Reference Giardini, Khambadkone and Rizzo23,Reference Khairy, Clair and Fernandes24 Could an increased lung volume play a role in the reduced exercise capacity? To answer that, more knowledge is needed about lung growth both pre- and postnatally, including other CHDs such as hypoplastic left heart syndrome, where the dominant right side pumps considerably more blood into the pulmonary circuit than in healthy controls. Reference Sun, van Amerom and Marini10
We found no difference in total intracranial volume in fetuses with transposition of the great arteries compared to healthy controls, also when correcting for ventricular septal defect/intact ventricular septum and other maternal and fetal parameters. Decreased intracranial volume in fetuses with transposition of the great arteries has been described in a part of the current data set, although it is only significant through gestational age 26–33. Reference Jørgensen, Tabor and Rode12 Some ultrasound and MRI studies report smaller brain volumes in late gestation for fetuses with transposition of the great arteries, Reference Jørgensen, Tabor and Rode12,Reference Zeng, Zhou, Zhou, Li, Long and Peng25,Reference Limperopoulos, Tworetzky and McElhinney26 while other studies report no difference in head circumference during gestation in transposition of the great arteries fetuses when compared to healthy controls. Reference Lauridsen, Uldbjerg and Henriksen27,Reference Mebius, Clur and Vink28 In these papers, it is not mentioned how many of the transposition of the great arteries fetuses had an additional ventricular septal defect.
In line with existing research, Reference Schiessl, Fakler, Vogt, Friese, Hess and Oberhoffer29 we found no difference in fetal liver volume between transposition of the great arteries and healthy controls, and the same holds true for kidney volumes, although no studies are available for comparison. In our study, the presence of a ventricular septal defect/intact ventricular septum made no difference in the size of kidneys and liver volumes in the fetuses with transposition of the great arteries. We found no studies investigating fetal kidney and liver volume in fetuses with transposition of the great arteries+ventricular septal defect and transposition of the great arteries+intact ventricular septum.
Combining the tendencies displayed in Figure 2, Figure 3, and supplementary S1–S3, and acknowledging that a statistically significant difference was only found in the lungs, one might get the impression that reduction in total intracranial volume is most likely in transposition of the great arteries-intact ventricular septum, where brain oxygen saturation is lowest, and that organs in the abdomen (liver and kidneys) tended to be smaller in transposition of the great arteries-ventricular septal defect. But this is all speculative.
Fetal sex may affect organ volumes. Total intracranial volume in late gestation has been found to be larger in male fetuses, while no sex differences have been found in the lungs and liver volumes. Reference Griffiths, Jarvis, Mooney and Campbell30–Reference Szpinda, Siedlaczek, Szpinda, Woźniak, Mila-Kierzenkowska and Wiśniewski32 Parity impacts fetal growth, with the birth weight of infants of nulliparous pregnancies being smaller. Reference Melamed, Aviram, Barg and Mei-Dan33,Reference Hinkle, Albert and Mendola34 Neither sex nor parity had an impact in the current study.
Strengths and limitations
Using a healthy control group scanned with the same equipment and personnel is preferable to published reference values, which are affected by inter-observer variability. Reference Di Mascio, Khalil and Rizzo35,Reference Deshmukh, Rubesova and Barth36 The sequence was optimised for volumetry with thin slices and short scan duration, and all measurements were performed by a blinded operator to minimise bias. While the use of anisotropic voxels is a limitation, the consistent use of 3D SSFP across the study population ensures comparability. However, 3D reconstructed single-shot T2 sequences are now often applied to achieve isotropic volumes. Reference Di Mascio, Khalil and Rizzo35,Reference Deshmukh, Rubesova and Barth36
The conclusions are limited by the small sample size, diminishing the statistical power of our findings. However, we found a significant difference, indicating that the difference in lung volumes is substantial, also when corrected for estimated fetal weight and gestational age. It should be noted, however, due to the absence of transposition of the great arteries-intact ventricular septum scans <30 weeks; that our findings reflect organ size at ∼30–38 weeks’ gestation. Another limitation of the current study was the decision to only analyse and report total intracranial volume instead of brain volume. This was done to ease the burden of analysis and due to prioritisation of the extra-cerebral organs. The choice may mask a possible association between transposition of the great arteries and smaller brain volumes. Jørgensen et al. suggested that the brain/intracranial cavity ratio in late-gestation fetuses with transposition of the great arteries is smaller than the healthy controls. Reference Jørgensen, Tabor and Rode12 Future research should seek to include both volume measurements.
Conclusion
In this preliminary study, we report a 30% increased lung volume in late-gestation fetuses with transposition of the great arteries+intact ventricular septum compared to fetuses with transposition of the great arteries+ventricular septal defect and healthy controls. Combined with the current knowledge about higher fetal pulmonary blood flow and increased saturation of the blood reaching the lungs in transposition of the great arteries+intact ventricular septum, a relationship between blood flow, oxygen delivery and organ volume is suggested.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S1047951126112281.
Acknowledgements
None.
Financial support
EK, OBP, and VH were employed on a grant from NNF17SA0030576.
Competing interests
None.