1. Introduction
As plants are unable to move to relocate themselves, they need to be able to adapt and adjust to their constantly changing environment. The plasticity of plants is the key to their success, and this requires an ability for plant cells to maintain an internal stability or ‘homeostasis’ even when the external conditions vary widely. Cellular homeostasis requires the preservation of a physiological variable at a set point that is an optimal value for physiology, despite external and internal disturbances. The set point concept comes from control theory, requiring a sensing system that detects deviations from the set point. A regulatory network usually acts via negative feedback to restore the variable towards the set point. In this review, we explore the core idea of homeostasis of plant nitrogen (N), more specifically the inorganic N ions ammonium (NH4 +) and nitrate (NO3 −) within cells. We identify some of the control theory components in plants for NO3 − and NH4 + that might define N homeostasis. Transport and assimilation are the main drivers for plant N homeostasis, and the balance between these two processes drives growth and yield. These topics have been extensively reviewed previously, and for brevity, recent overviews are cited.
2. Whole plant N homeostasis
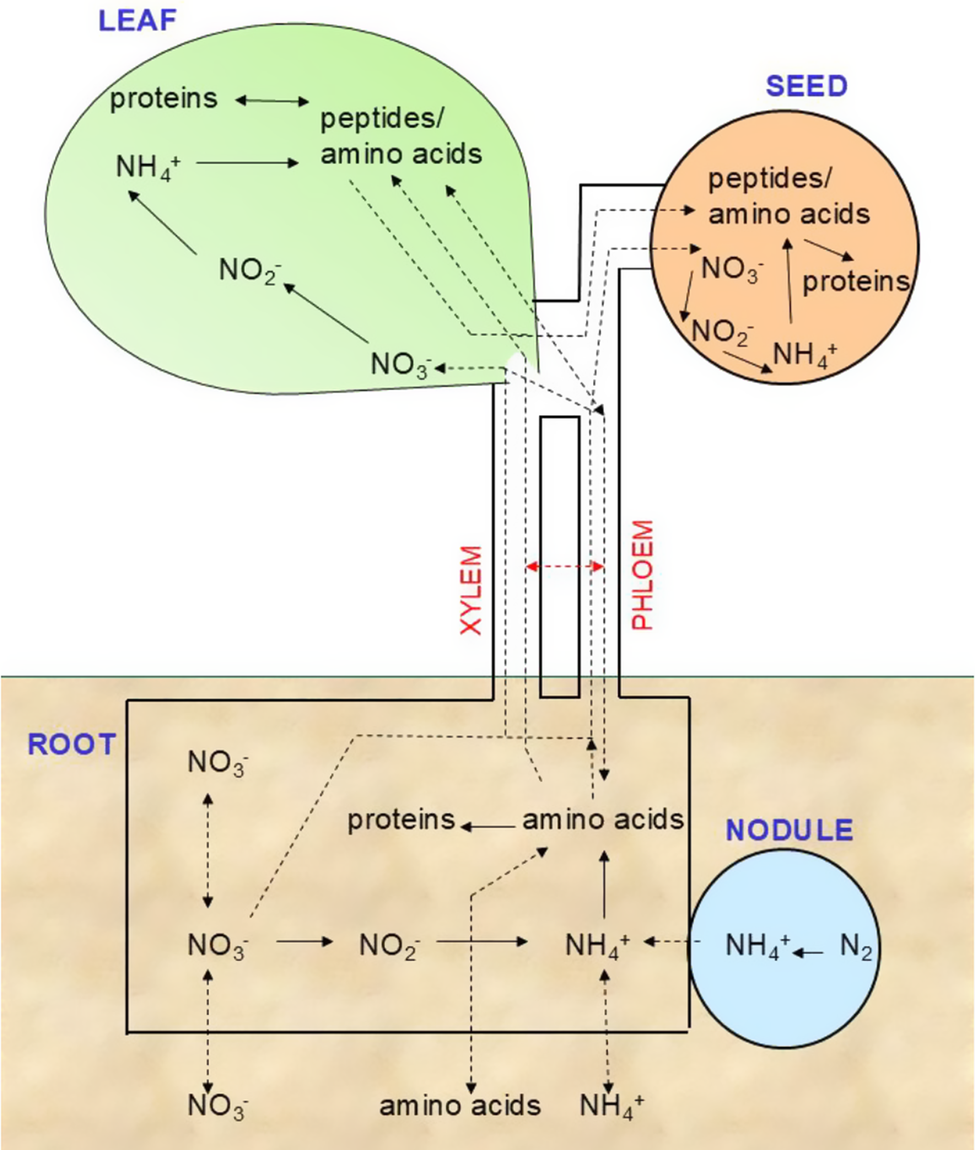
N is an essential macronutrient for plant growth, and the optimisation of agricultural production depends on the addition of N fertiliser. Chemical fertilisers are often applied with N available as NH4 + or NO3 −, and other forms of organic N, such as urea, are rapidly converted by soil microbes into these inorganic forms. Plants can take up both inorganic N forms: NO3 − and NH4 +. Legume plants can capture gaseous N2 through symbioses with N-fixing microorganisms, forming the neutral molecule ammonia (NH3), which can be transferred to the host. Gaseous NH3 can be captured by plants and can diffuse through cell membranes, but this environmental source of N is not usually important for growth (Xu et al., Reference Xu, Fan and Miller2012). Organic sources of N in the soil, including amino acids, urea and ureides, can be taken up and used by plants. These forms of N are particularly important for plants growing in impoverished soils. N-starved plants have systemic long-distance signalling mechanisms mediated by phloem-mobile microRNAs (miRNAs) (Liang et al., Reference Liang, He and Yu2012) and peptides (C-terminally encoded peptide or CEP) in the phloem (Ohkubo et al., Reference Ohkubo, Tanaka, Tabata, Ogawa-Ohnishi and Matsubayashi2017). Circulating peptide signals are perceived by specific receptors and can modify strigolactone hormone levels to alter root (branching) and shoot (tillering) architecture. In contrast, N-replete status is mediated by circulating cytokinin hormone levels (Poitout et al., Reference Poitout, Crabos, Petřík, Novák, Krouk, Lacombe and Ruffel2018). Insufficient N levels can lead to restricted plant growth and a shift from the vegetative to the reproductive developmental phase. Interestingly, N deficiency encourages extensive root development, while sufficiency promotes resource allocation for enhanced vegetative growth (Xu et al., Reference Xu, Fan and Miller2012). The process of N allocation is heavily influenced by transporter activity, not only through uptake from the soil but also for the movement of N around the plant. During plant development the distribution of N changes as vegetative growth builds the canopy that later switches to florescence and seed filling (see Figure 1). Throughout these developmental changes the tissue pools of inorganic N ions could alter but only NO3 − changes directly indicate the overall N status of the plant. For this reason, the crop N status testing applied by farmers uses tissue sap NO3 −, not NH4 + as an indicator ion species when deciding on fertiliser application rates. This highlights the crucial role of NO3 − as an indicator ion species and therefore identifies it as a signal in coordinating N utilisation and plant growth. Undeniably, NO3 − signalling triggers the activation of genes associated with various metabolic pathways and developmental processes (reviewed in Zhao et al., Reference Zhao, Liu, Crawford and Wang2018).
Whole plant N homeostasis pools.

Whole plant N homeostasis set points are controlled by variables such as metabolism, growth and signalling to stay balanced. Assimilation, uptake from the soil and long-distance transport within the plant in the phloem and xylem are key processes. The pathways and key enzymes for the assimilation of NO3 − and NH4 + are well characterised (Xu et al., Reference Xu, Fan and Miller2012). Growth and yield are optimised when N assimilation into amino acids and proteins is most active, and this occurs during periods of maximised photosynthetic carbon assimilation. Nitrate uptake by roots and movement of nitrate to the shoot remains high during the light period and decreases slightly at night. The NO3 − that is taken up during the nightis chiefly used to replenish the leaf NO3 − pool (Matt et al., Reference Matt, Geiger, Walch-Liu, Engels, Krapp and Stitt2001). Once transported to the leaf, NO3 − is primarily stored within the vacuole, accounting for 58–99% of the total NO3 − pool (Granstedt & Huffaker, Reference Granstedt and Huffaker1982). Therefore, throughout the day and night, the pool of NO3 − primarily localised in leaf vacuoles, is continually changing and responding to both the soil supply and assimilation activity. Throughout these fluctuations in NO3 −, tissue NH4 + concentrations usually remain low, indicating a tight coupling between NO3 − and NH4 + assimilation, primarily GS activity. The set points for N homeostasis are fundamentally linked to those of other elements; these are often expressed as ratios, such as C:N (reviewed by Fañanás-Pueyo et al., Reference Fañanás-Pueyo, Carrera-Castaño, Pernas and Oñate-Sánchez2025). The C:N ratio changes in tissues during the switch from vegetative to floral ontogeny, from 15-20:1 to 80-100:1, and two conserved kinases (TOR and SnRK1) play key parts in signalling in all eukaryotes. Other elemental ratios have been studied in much less detail, for example, N:P, although these are linked to the type of plant, higher in graminoids than forbs, and linked to stress tolerance (Güsewell, Reference Güsewell2004). Long-distance transport is also closely coupled with the need for electrical charge balance and for example, potassium is well known to counter NO3 − movement in the xylem and phloem (Drechsler et al., Reference Drechsler, Zheng, Bohner, Nobmann, von Wirén, Kunze and Rausch2015).
3. Cellular homeostasis of N
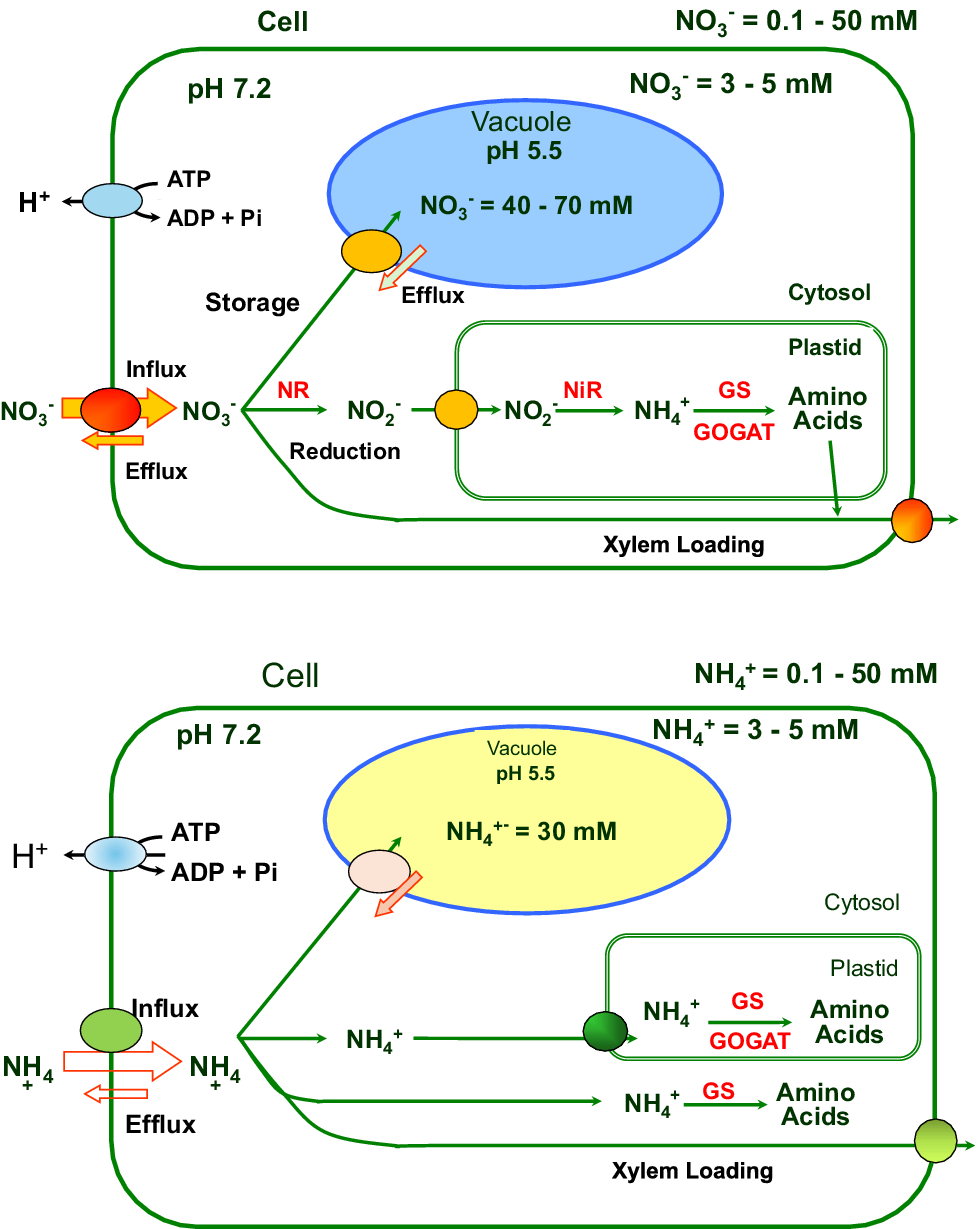
The set points for cytosolic homeostasis of NO3 − and NH4 + seem to be similar, with both in the low mM range for the cytosol and an order of magnitude greater for the vacuole (see Figure 2). Although the published values can vary hugely over several orders of magnitude for NH4 + from 0.02 to 0.2 M, depending on the supply and method used (Miller et al., Reference Miller, Cookson, Smith and Wells2001). There can be fundamental metabolic consequences of NO3 − and NH4 + use by plant cells that could directly act on intracellular homeostasis through regulatory mechanisms for pH and redox regulation. For example, excessive NO3 − transport can acidify the cytosol and activate proton pumps to restore pH homeostasis (Feng et al., Reference Feng, Fan, Miller and Xu2020). Nitrate assimilation consumes reducing power in the cell through NADH- or NADPH-mediated transfer of reductant. In Arabidopsis leaf cells, both photosynthesis and nitrate reductase activity were shown to alter the cytosolic NO3 − set point (Cookson et al., Reference Cookson, Williams and Miller2005). These primary assimilatory processes may provide another level of control that is important when there is an excessive supply of either of these forms of N supply.
Diagrammatic overview of plant cell ammonium and nitrate homeostasis showing metabolism and transport. Upper, nitrate; Lower, ammonium.

When plants are N replete, the few measurements which have been made suggest that cytosolic NH4 + and NO3 − activities are maintained at mM activities that are independent of the external supply (e.g. NO3 −: Miller & Smith, Reference Miller and Smith2008; NH4 +: Wells & Miller, Reference Wells and Miller2000; Zhou et al., Reference Zhou, Bai and Song2015). These homeostatic set point values are higher than the affinities of the enzymes that assimilate the ions; GS/GOGAT for NH4 + and NO3 − reductase for NO3 − (see Figure 2). Suggesting that assimilatory enzyme activities are not directly determining the cytosolic set points. A range of set point values has been reported, depending largely on the type of method used, and as discussed above, more measurements and new techniques are needed.
3.1. The evidence for set points of cellular NH4 + and NO3 −
A previous review (Miller & Smith, Reference Miller and Smith2008) described the evidence that cytosolic NO3 − homeostasis is regulated and has a potential role in nutrient sensing and signalling. It was suggested that root tips may be particularly important in this sensing role. Nitrate is essential for plant growth and development, even in organs like seeds where its nutritional role is unclear (Chopin et al., Reference Chopin, Orsel, Dorbe, Chardon, Truong, Miller, Krapp and Daniel-Vedele2007). It can act as a signal for germination and shoot–root growth balance. Different cell types and tissues can show varying NO3 − concentrations, with vacuolar NO3 − pools acting as reservoirs to maintain cytosolic NO3 − levels. Twenty years ago, the evidence for these ideas was largely based on measurements using intracellular NO3 −-selective electrodes. Nitrate-selective microelectrodes measurements have shown environmental conditions when cytosolic nitrate changed, for example, during light/dark changes in leaf cells, NO3 −-starvation and when NO3 − and ammonium or glutamine were supplied together as a mixed supply of N (Miller & Smith, Reference Miller and Smith2008).
The stability of cytosolic NO3 − is controversial, and even now, the methods to measure it are limited – new techniques are needed, and despite progress in using genetically encoded sensors for other ions (Sadoine et al., Reference Sadoine, De Michele, Župunski, Grossmann and Castro-Rodríguez2023), obtaining quantitative NO3 − measurements remains a tricky nut to crack (Clophensor Demes et al., Reference Demes, Besse, Cubero-Font, Satiat-Jeunemaitre, Thomine and De Angeli2020; NitraMeter3.0 Chen et al., Reference Chen, Cartwright and C-H2022; NitrOFF Cook et al., Reference Cook, Smailys, Ji, Phelps, Tutol, Kim, Ong, Peng, Maydew, Zhang and Dodani2025). These genetically encoded sensors are often pH sensitive, but in the cytoplasm the tight regulation of pH should minimise this problem. The high affinity of the bacterial-derived sensors, for example, the nitrate recognition domain NreA from Staphylococcus carnosus (Kd=9 μM) of NitrOFF, should be saturated at the mM concentrations suggested from microelectrode measurements. For a comparison of the properties of genetically encoded sensors for NO3 −, see Table S1 in Cook et al. (Reference Cook, Smailys, Ji, Phelps, Tutol, Kim, Ong, Peng, Maydew, Zhang and Dodani2025).
The toxicity of NH4 + accumulation in biology is widely accepted and therefore the need for plants to regulate their cellular concentration is less controversial (Bittsánszky et al., Reference Bittsánszky, Pilinszky, Gyulai and Komives2015).
3.2. Nitrate and ammonium transporters
Four gene families have been identified as NO3 − transporters; these are proton-coupled symporters NRT1s (NPFs) and NRT2s (Wang et al., Reference Wang, Hsu and Tsay2012), proton/anion antiporters CLCs and the NAXT/SLAC/SLAH anion channels (Kollist et al., Reference Kollist, Jossier, Laanemets and Thomine2011). Many NRT1 (NPF) and NRT2 NO3 − transporters have been characterised in detail, they are proton-coupled cotransporters that can operate efficiently over a large range of NO3 − concentrations (reviewed in Wang et al., Reference Wang, Hsu and Tsay2012; Xu et al., Reference Xu, Fan and Miller2012). Two families of NH4 + transporters (AMT1 and 2) are well described and, like the NRTs, also have wide-ranging affinities for NH4 + (Xu et al., Reference Xu, Fan and Miller2012). The entry of NH4 + into cells can occur via non-selective cation channels, driven by the large negative membrane potential across the plasma membrane (Zhou et al., Reference Zhou, Bai and Song2015). Both types of transporters, NO3 − and NH4 +, have proton-coupled mechanisms to drive uptake from low external concentrations of the ions. In Figure 2, we have summarised the main cellular pathways for both ions, NO3 − and NH4 +. At the cell plasma membrane, we show efflux mechanisms for both ions, but the precise genetic identity is not clear. Stretch-activated anion channels for NO3 − and outwardly directed cation channels for NH4 + have been functionally characterised (Hedrich, Reference Hedrich2012). At the plasma membrane, nitrate efflux is mediated by AtNRT1.5,1.8,1.9 (Wang et al., Reference Wang, Hsu and Tsay2012), SLAC/SLAH anion channels, and these are important for xylem loading and stomata guard cell function (Hedrich & Geiger, Reference Hedrich and Geiger2017). The aluminium-activated malate transporters are generally considered organic anion channels, but some family members are permeable to NO3 − (e.g. rice OsALMT7, Heng et al., Reference Heng, Wu, Long, Luo, Ma, Chen, Liu, Zhang, Ren, Wang, Tan, Zhu, Wang, Lei, Zhang, Guo, Wang, Cheng and Wan2018).
In addition to regulation of transcript levels, both types of transporters, NO3 − and NH4 +, have post-translational mechanisms (e.g. phosphorylation) to regulate their activity (see Section 4) and this fact supports the view that transport is a key factor in plant N homeostasis (e.g. NO3 −: Yue et al., Reference Yue, Liu, Liao, Zhang, Wu and Wang2025; NH4 +: Wang et al., Reference Wang, Li, Zhang and Chu2021).
3.3. Vacuolar transporters
There are three protein families involved in NO3 − transport through the vacuolar membrane, CLCs (De Angeli et al., Reference De Angeli, Monachello, Ephritikhine, Frachisse, Thomine, Gambale and Barbier-Brygoo2006; Yang et al., Reference Yang, Zhang, Ye, Zheng, Huang, Yu, Chen, Cai and Zhang2023), NRT2s (e.g. Chopin et al., Reference Chopin, Orsel, Dorbe, Chardon, Truong, Miller, Krapp and Daniel-Vedele2007) and NPFs (e.g. He et al., Reference He, Peng, Cai, Liu, Guan, Yi and Gong2017). Plant tissue NO3 − concentrations directly follow the storage concentrations in the vacuole, which in turn generally reflect the changing levels in the soil. Remobilisation of vacuolar-stored NO3 − is important for NUE (Chen et al., Reference Chen, Chen, Tseng and Tsay2020) and this is an important target for improving NUE that may also have consequences for water use efficiency as vacuolar NO3 − is an important osmoticum (Hodin et al., Reference Hodin, Lind, Marmagne, Espagne, Bianchi, De Angeli, Abou-Choucha, Bourge, Chardon, Thomine and Filleur2023). In Arabidopsis, vacuolar AtCLCa activity is regulated by phospholipids and nucleotides like ATP (Yang et al., Reference Yang, Zhang, Ye, Zheng, Huang, Yu, Chen, Cai and Zhang2023).
Information for NH4 + transport at the vacuole is complicated by the fact that NH3 is trapped in this acidic compartment. Ammonia, due to the pH gradient between the cytoplasm and vacuole, diffuses across the tonoplast into the acidic vacuole, where it is trapped as the charged NH+ 4 ion in the acid compartment. In the vacuolar membrane, the tonoplast intrinsic proteins (TIPs) that can mediate water movement have also been shown to facilitate NH3 transport into the vacuole (Loqué et al., Reference Loqué, Ludewig, Yuan and von Wirén2005). Both NH4 + and NH3 may be exported from the vacuole with the possibility for post-translational regulation of the direction of transport. This topic is important and needs more research effort.
4. Regulatory network: restoration of the set point
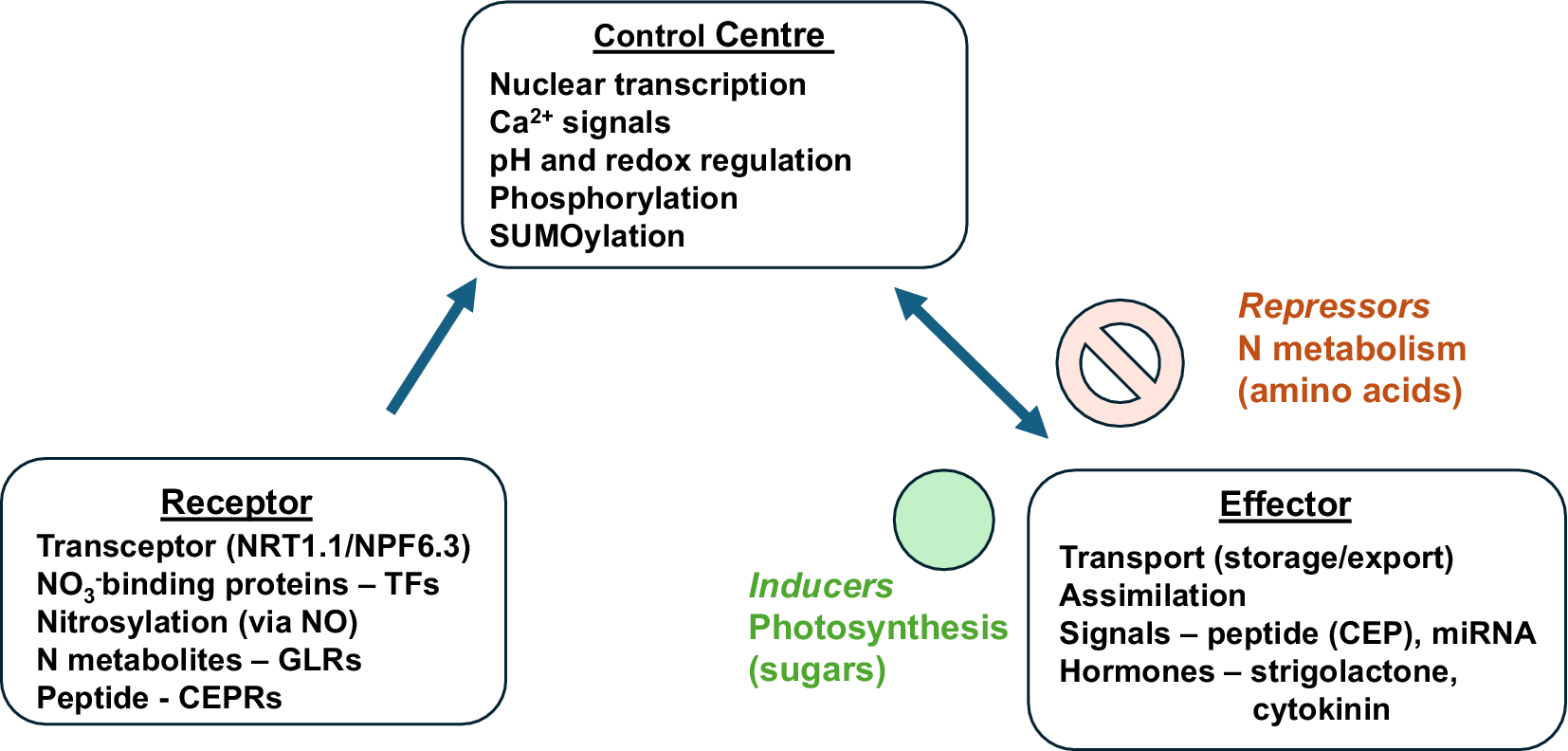
We can accept and build on the tenet that all homeostatic control mechanisms have a minimum of three interdependent components for the variable being regulated: a receptor, a control centre and an effector. This textbook homeostatic model has been formulated for animal cells (Billman, Reference Billman2020). Applying these fundamental questions to the plant cell model, we can look for the three components.
4.1. Cell N sensing
Are there receptors for NO3 − or NH4 + in plant cells? The transporter NRT1.1 has been described as a NO3 − transceptor linked to calcium signals via kinase activity (Wang et al., Reference Wang, Hsu and Tsay2012). A receptor should have some capacity to specifically bind either ion with an affinity that perhaps matches the range of possible homeostatic values of the cytosolic activities of these ions. Nitrate-binding proteins that are regulators of transcription have been identified in the nucleus, for example, NLP7 (Cheng et al., Reference Cheng, Durand, Brehaut, Hsu, Kelemen, Texier, Krapp and Tsay2023; Liu et al., Reference Liu, Liu, Lin, Wang, Chen, Liu, Guo, Konishi, Yanagisawa, Wagner and Sheen2022). The nucleus may seem an odd place to perceive external environmental changes in N supply, but if there is homeostasis in the cytosol and this depends on the plant’s N status, then this makes sense. The nitrate-binding domain of NLP7 has been identified and a genetically encoded fluorescent split biosensor, mCitrine-NLP7, enabled visualisation of single-cell nitrate dynamics in planta (Liu et al., Reference Liu, Liu, Lin, Wang, Chen, Liu, Guo, Konishi, Yanagisawa, Wagner and Sheen2022). As amino acids are the primary product of N assimilation, they have been proposed as potential negative feedback signals for N status. To this end, plant glutamate receptor-like genes have been identified as Ca2+ channels that bind amino acids, but their functions appear more widespread, including long-distance electrical signalling and redox sensing (Simon et al., Reference Simon, Navarro-Retamal and Feijó2023).
There are many examples of post-translational modifications of proteins involved in the transport and assimilation of NH4 + and NO3 −, this tight regulation provides indirect evidence for homeostasis and set points in the N physiology.
4.2. Regulation of assimilation enzymes
The activity of the primary N assimilatory enzymes is carefully regulated by phosphorylation; for example, NR is regulated by phosphorylation and subsequent 14-3-3 protein binding (Lambeck et al., Reference Lambeck, Fischer-Schrader, Niks, Roeper, Chi, Hille and Schwarz2012) and GS (Finnemann & Schjoerring, Reference Finnemann and Schjoerring2000). These are enzymes that catalyse key steps in N assimilation and homeostasis, as the former generates toxic nitrite (NO2 -) and the latter is the confluence of C and N assimilation. NR activity can generate nitric oxide (NO) when NO2 - concentrations increase, and this reactive oxygen species can result in S-nitrosylation, reacting with cysteine thiols in proteins, an indicator of stress, redox status and an imbalance in N homeostasis. This reaction can also regulate protein SUMOylation linking N status to plant immunity (Borrowman et al., Reference Borrowman, Kapuganti and Loake2023). This mechanism is a reversible post-translational modification where SUMO (small ubiquitin-like modifier) proteins are covalently attached to lysine residues on target proteins.
4.3. Regulation of NH4 + and NO3 − transporters
Phosphorylation of transporters is a common regulatory mechanism and there are many examples (Hao et al., Reference Hao, Li, Kong, Chen, Liu, Guo and Zhou2023). The calcium-dependent protein kinase and CIPK families of plant protein kinases function in calcium signalling pathways and are important in the regulation of N cellular homeostasis. Both NH4 + (AMT1.1/2) and NO3 − (NRT1.1) transporter activity in the model plant Arabidopsis are regulated by the activity of CIPK23 (reviewed by Ródenas & Vert, Reference Ródenas and Vert2020). At high NH4 + concentrations, the AMT transport activity is inactivated by phosphorylation. In contrast, at low NO3 − concentrations, AtNRT1.1 transport activity is activated by CIPK23 phosphorylation. There is allosteric regulation of plant AMTs, involving a phosphorylation switch that functions in a feedback loop to restrict NH4 + uptake (Lanquar et al., Reference Lanquar, Loqué, Hörmann, Yuan, Bohner, Engelsberger, Lalonde, Schulze, von Wirén and Frommer2009).
The activity of NRT2 transporters is also regulated by phosphorylation, including their interaction with the Nar2 partner proteins that are required for function (Jacquot et al., Reference Jacquot, Chaput, Mauries, Li, Tillard, Fizames, Bonillo, Bellegarde, Laugier, Santoni, Hem, Martin, Gojon, Schulze and Lejay2020; Li et al., Reference Li, Wu, Jaquot, Chaput, Adamo, Neuhäuser, Straub, Lejay and Schulze2024). Taken together there are multiple levels of control for the entry of inorganic N into non-leguminous plants. In legumes, the situation is further complicated by the activity of the symbiotic bacteria living in nodules and generating NH3 from gaseous atmospheric N2.
4.4. Regulation of vacuolar transporters
All three protein families of vacuolar NO3 − transporters can be post-translationally regulated. In Arabidopsis, vacuolar AtCLCa NO3 − selectivity is determined by a proline residue (Wege et al., 2010) and activity is regulated by phospholipids and nucleotides like ATP (De Angeli et al., Reference De Angeli, Moran, Wege, Filleur, Ephritikhine, Thomine, Barbier-Brygoo and Gambale2009; Yang et al., Reference Yang, Zhang, Ye, Zheng, Huang, Yu, Chen, Cai and Zhang2023). The vacuolar CLCs also have an important role in cytosolic pH regulation (Demes et al., Reference Demes, Besse, Cubero-Font, Satiat-Jeunemaitre, Thomine and De Angeli2020), which is key for pH balance during the switch between NH+ 4 and NO3 − external supply. This provides an interesting direct link between N supply, homeostasis and pH regulation that is worthy of future investigation. The activity of the aquaporin TIPs that facilitate NH3 transport (Loqué et al., Reference Loqué, Ludewig, Yuan and von Wirén2005) has also been shown to be regulated by phosphorylation of the protein (Maurel et al., Reference Maurel, Kado, Guern and Chrispeels1995). The direction of tonoplast transport of NH4 + and NH3 may be post-translationally regulated and vacuolar accumulation of NH4 + is regulated by CAP1 (Bai et al., Reference Bai, Ma, Zhang, Song, Zhou, Gao, Miao and Song2014).
4.5. Transcription factors
Plant transcription factors (TFs) are important molecular regulatory systems for N homeostasis, typically comprising 5% of the genome (Blanc-Mathieu et al., Reference Blanc-Mathieu, Dumas, Turchi, Lucas and Parcy2024), they coordinate N transport and assimilation (e.g. NO3 −, Sámano et al., Reference Sámano, Nanjareddy and Arthikala2024). There are several well-known examples, for example, HY5 is important for C:N partitioning (Chen et al., Reference Chen, Yao, Gao, Jiang, Harberd and Fu2016). The NIN-like proteins (NLPs) are a group of TFs belonging to the RWP-RK gene family, they act as major nitrate sensors and are implicated in the primary NO3 − response within the nucleus of both non-leguminous and leguminous plants through their RWP-RK domains. The NLPs can act as intracellular nitrate sensors, as they can bind NO3 − and thereby alter the expression of other transcripts linked to the N status of the cell. In the model plant, Arabidopsis, there are nine NLP genes expressed in Arabidopsis shoots, and the role of some family members in binding NO3 − to alter transcription is well established (Sámano et al., Reference Sámano, Nanjareddy and Arthikala2024). In a legume, NO3 − induces SUMOylation of the NLP1 TF (Liu et al., Reference Liu, Luo, Wang, Lin and Xie2026). This mechanism may yet prove to be more widely found among plants, providing a link between N homeostasis and TFs.
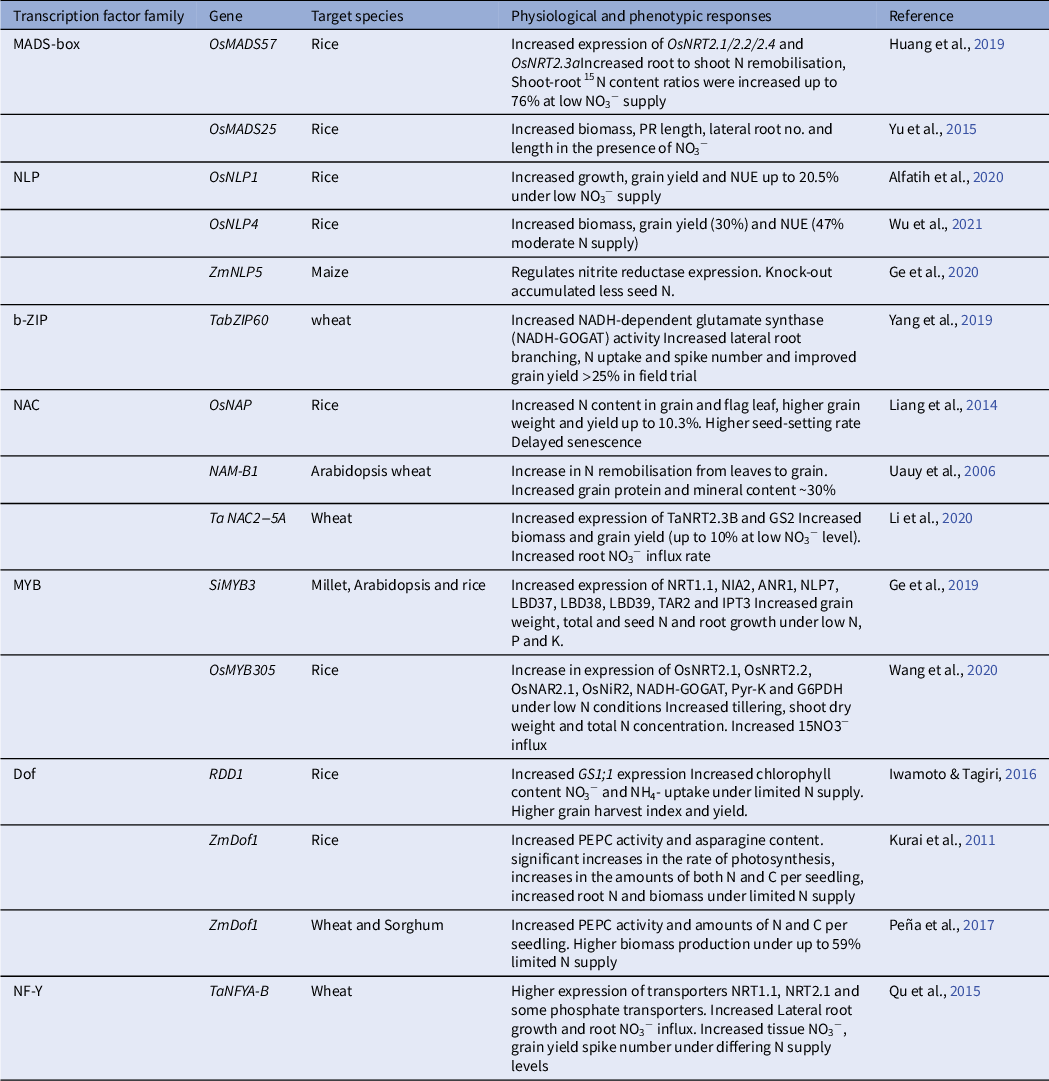
As TFs are more general regulators of N acquisition and homeostasis, they have been targets for efforts to improve crop fertiliser use (Maurya et al., Reference Maurya, Bandyopadhyay and Prasad2020). There are now many published examples of altered expression of N-responsive TFs that have been used in crops to have significant effects on NUE (see Table 1).
Some N-related TFs used in crop plants to improve NUE (redrawn from Maurya et al., Reference Maurya, Bandyopadhyay and Prasad2020)

5. Future perspectives and N use efficiency
Improving N use efficiency (NUE) is the target of major research efforts. Plant N homeostasis and NUE are interdependent and closely linked. Altering N set points is a key component of attempts to improve NUE and here an opportunity can be provided by natural ecosystems as research using rapidly growing species adapted to nutrient-poor environments signpost the route to better crops. As described above, TFs are targets for altering C:N ratios and NUE as they offer a high tier of control (Figure 3) and there are many candidates (Table 1). It remains to be tested to what extent the effects of each TF may be additive, and this may provide a breeding opportunity. Both N assimilation and uptake are important components of NUE. The assimilation of N is limited by the photosynthetic production of sugars for amino acid synthesis by GS/GOGAT activity. The energy from carbon metabolism and supply of reductant from photosynthesis power the uptake and assimilation of N. Therefore, plant N homeostasis and NUE are directly linked to carbon assimilation and future efforts to improve crops must recognise this fact and integrate both traits into breeding programmes. The supply of N to cells, maintaining an optimal concentration for assimilation, is a fundamental driver for homeostasis both at the cellular and whole plant levels. If N homeostasis breaks down this is likely to be very detrimental for growth, but when it is sub-optimal then crop productivity is below ideal. Breeding future crops to improve the plasticity of N homeostasis is likely to be a promising strategy, particularly with the threat of climate change extreme weather events. Therefore, a better understanding of plant N homeostasis and what can limit it in cells and the whole plant is fundamentally important for improving NUE. A holistic view that includes N and C cycling is important for this progress.
Identifying the model components of plant N homeostasis.

In the future, a better understanding of factors extending beyond the plant, into the rhizosphere and the soil, that influence N homeostasis is important. For example, soil C supply is important for nutrient cycling and the slow release of N for crops. Maintaining a slow, steady N supply that matches the changing needs of the crops and balancing homeostasis can directly depend on root activity. Root exudates can modify soil microbial activities to influence the form of N supply and optimise growth. Optimising the balance in the N supply, NO3 − and NH4 + (Feng et al., Reference Feng, Fan, Miller and Xu2020) for the crop during plant development is likely to be an important aspect of improving crop NUE. To achieve this soil delivery target, better methods for monitoring soil nutrient delivery are needed. There are also exciting opportunities for phage engineering of the rhizosphere microbiome (Quirós et al., Reference Quirós, Sala-Comorera, Gómez-Gómez, Ramos-Barbero, Rodríguez-Rubio, Vique, Yance-Chávez, Atarés, García-Gutierrez, García-Marco, Vallejo, Salaet and Muniesa2023) to better optimise microbially mediated N cycling to balance the soil supply of NO3 − and NH4 + to crops. A holistic view of N homeostasis should extend beyond the whole plant and cells into the soil for improving NUE.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/qpb.2026.10042.
Abbreviations and Glossary
-
ALMT Aluminium-activated Malate Transporter
-
CAP1 Adenylate Cyclase-Associated Protein 1 (actin-binding protein)
-
CBL Calcineurin B-like protein
-
CEP C-terminally encoded peptide
-
CEPR CEP Receptor
-
CIPK CBL-interacting protein kinase
-
CPK Calcium-dependent protein kinase
-
GOGAT glutamate synthase
-
GS glutamine synthetase
-
NAD Nicotinamide Adenine Dinucleotide
-
NADPH nicotinamide adenine dinucleotide phosphate
-
NIN Nodule inception TF
-
NiR nitrite reductase.
-
NLP NIN-like proteins
-
NR nitrate reductase
-
RWP-RK a family of TFs
-
SLAC Slow-type anion channel
-
SLAH SLAC-1 homolog anion channel
-
SnRK1 SNF1-related protein kinase 1, a key cellular energy-sensing protein kinase.
-
SUMO Small Ubiquitin-like Modifier
-
TF transcription factor – a highly conserved serine/threonine protein kinase
-
TOR Target of rapamycin
Data availability statement
No new data or coding was used in preparing this manuscript.
Acknowledgements
Y.C. and T.M. are funded by the BBSRC Institute Strategic Programmes: Plant Health (grant no. BB/P012574/1) and Advancing Plant Health (grant no. BB/X010996/1); Harnessing Biosynthesis for Sustainable Food and Health (grant no. BB/X01097X/1).
Author contributions
Y.C. and T.M. planned and wrote the review.
Funding statement
WishRoots: The BBSRC Wish-Roots 21EJP Soil: Tuning the wheat root microbiome to improve soil health and optimise rhizosphere nitrogen cycling and availability (grant no. BB/X003000/1). APH: The BBSRC Institute Strategic Programmes: Plant Health (grant no. BB/P012574/1) and Advancing Plant Health (grant no. BB/X010996/1). HBio: The BBSRC Institute Strategic Programme: Harnessing Biosynthesis For Sustainable Food and Health (grant no. BB/X01097X/1).
Competing interest
The authors declare no conflict of interest.



Comments
Quantitative Plant Biology,
Cambridge University Press.
Dear Editor,
I enclose a review manuscript titled “Homeostasis of ammonium and nitrate in plants” to be considered for publication.
The manuscript includes three figures and one table. As the table cites 15 more references, these are included below the table and submitted as Supplemental material. We welcome your and the Reviewers’ opinions on how best to include this information.
Nitrogen is a frequently reviewed topic; therefore, we have heavily drawn on other reviews, referring the reader to these for much of the background information on the subject.
We hope the manuscript will be of interest to the readership of Quantitative Plant Biology, and as pointed out in the text, it is difficult to be precisely quantitative about the set points for cellular concentrations within plant cells.
Kind regards,
Drs Yi Chen and Tony Miller