Introduction
Cognitive symptoms, such as forgetfulness, distractibility, and disrupted goal-driven behaviours, are commonly reported by patients with functional neurological disorder (FND) (APA, 2013; Butler et al., Reference Butler, Shipston-Sharman, Seynaeve, Bao, Pick, Bradley-Westguard and Nicholson2021; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Teodoro, Edwards, & Isaacs, Reference Teodoro, Edwards and Isaacs2018). Occurring as the primary concern in functional cognitive disorder (FCD) (Teodoro et al., Reference Teodoro, Edwards and Isaacs2018), or alongside other FND presentations such as functional seizures (FS) and functional motor symptoms (FMS) (Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018), self-reported cognitive symptoms have been associated with greater psychological symptom load and compromised quality of life (Forejtova et al., Reference Forejtova, Serranova, Sieger, Slovak, Novakova, Vechetova and Edwards2023; Millman, Williams, Jungilligens, & Pick, Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Vechetova et al., Reference Vechetova, Slovak, Kemlink, Hanzlikova, Dusek, Nikolai and Serranova2018).
Despite the prevalence and apparent impact of self-reported cognitive difficulties in FND, neuropsychological testing studies have provided inconsistent evidence for cognitive performance deficits across FND phenotypes. One systematic review and meta-analysis reported significant performance impairments across cognitive domains in patients with FS, with moderate-large effect sizes compared to healthy controls (Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a). Compromised cognitive performance may also be related to poorer mental health and quality-of-life in FS (Van Patten et al., Reference Van Patten, Lu, Austin, Cotton, Chan, Bellone and LaFrance2024b). However, patients with FS displayed superior performance to epilepsy controls on tests of general cognitive functioning, language, and long-term memory (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a). The broader neuropsychological literature across FND phenotypes has also provided mixed evidence for intact, impaired, or superior cognitive test performance compared to neurological, psychiatric, and/or healthy controls (Alluri et al., Reference Alluri, Solit, Leveroni, Goldberg, Vehar, Pollak and Perez2020; Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a). While a considerable number of investigations have examined cognitive performance in FS samples, fewer have focused on FMS, FCD, or mixed FND presentations (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a). It therefore remains unclear whether cognitive performance impairments are present to a similar extent across FND phenotypes, and which cognitive domains are most affected.
Beyond core cognitive functions, emerging findings indicate potential differences in metacognition in FND (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018). Metacognition refers to awareness and control of one’s own cognitive processes, and adaptive use of this knowledge in goal-driven behaviours (Fleming, Reference Fleming2024). Metacognitive deficits are associated transdiagnostically with mental health symptoms and maladaptive behaviour, and may be important treatment targets in neuropsychiatric disorders (Seow, Rouault, Gillan, & Fleming, Reference Seow, Rouault, Gillan and Fleming2021). ‘Global metacognition’ involves broad self-evaluations of cognitive performance, often assessed with self-report measures of cognitive symptoms or aspects of cognitive functioning in everyday life. ‘Local metacognition’, on the other hand, relates to shorter-term assessments of performance or specific decisions/actions within a particular cognitive task/activity (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025; Seow et al., Reference Seow, Rouault, Gillan and Fleming2021). In FND, altered global metacognition has been indicated by the presence of elevated self-reported cognitive symptoms and/or poorer self-evaluations of everyday cognitive functioning, in the absence of observable performance deficits (Alluri et al., Reference Alluri, Solit, Leveroni, Goldberg, Vehar, Pollak and Perez2020; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018). Reduced concordance between neurocognitive test performance and post-diction self-evaluations in some FND samples has also indicated potentially diminished local metacognition (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018), although a limited number of performance-controlled experimental studies have yielded mixed findings on local metacognition in FND samples to date (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025).
The disparate findings relating to cognitive functioning in FND may be due in part to methodological issues, such as suboptimal consideration of confounding variables (e.g., age, education, medication, and performance validity), the wide range and variable quality of measures employed, and differing comparison groups (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a,Reference Van Patten, Lu, Austin, Cotton, Chan, Bellone and LaFrance2024b). As such, studies with improved methodological rigor are needed to establish with greater certainty whether cognitive and metacognitive differences are related directly to the presence of FND, rather than to non-specific factors that are merely associated with the disorder. Additionally, few studies have directly compared cognitive and metacognitive profiles in specific FND subgroups using the same measures, which could highlight shared and distinct cognitive/metacognitive features, with clinical and mechanistic relevance.
Aims and hypotheses
Within a larger research program examining aetiological factors and mechanisms specifically in FS and FMS (Pick et al., Reference Pick, David, Edwards, Goldstein, Hodsoll, Millman and Hotopf2024), this study aimed to assess self-reported cognitive symptoms, neuropsychological test performance profiles, and post-diction metacognitive confidence in patients with a primary diagnosis of FS or FMS. We sought to compare the FS and FMS groups to healthy controls without physical or mental health disorders (HC), and clinical controls (CC) with major depression and/or anxiety disorders, to better understand the neurocognitive features that distinguish those with FS and FMS from the general population, and from those with common mental health symptoms. This project was not designed to investigate mechanisms in FCD, but rather to improve the understanding of cognitive and metacognitive functioning in FS and FMS – two of the most common FND presentations.
The objectives were to examine overall between-group differences (FS/FMS/CC/HC) and FND subgroup-specific (FS/FMS) outcome profiles in cognitive domains with clinical and mechanistic relevance in FND (Brown & Reuber, Reference Brown and Reuber2016; Drane et al., Reference Drane, Fani, Hallett, Khalsa, Perez and Roberts2020; Edwards et al., Reference Edwards, Adams, Brown, Parees and Friston2012; Pick, Goldstein, Perez, & Nicholson, Reference Pick, Goldstein, Perez and Nicholson2019), including (1) attention, executive functioning, working memory, and social–emotional processing test performance, (2) test-specific post-diction metacognitive confidence, and (3) self-reported cognitive symptoms. We also sought to explore within-group relationships between cognitive/metacognitive outcomes and clinical characteristics (FS/FMS/CC).
Compared to HC and CC, we predicted that the FS and/or FMS groups would display:
-
• Elevated self-reported cognitive symptoms (Butler et al., Reference Butler, Shipston-Sharman, Seynaeve, Bao, Pick, Bradley-Westguard and Nicholson2021; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023).
-
• Altered performance on tests of:
-
a. executive functioning (i.e., diminished sustained attention, set-shifting, response inhibition) (Alluri et al., Reference Alluri, Solit, Leveroni, Goldberg, Vehar, Pollak and Perez2020; Brown & Reuber, Reference Brown and Reuber2016; Edwards et al., Reference Edwards, Adams, Brown, Parees and Friston2012; Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Goldstein, Perez and Nicholson2019; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a);
-
b. working memory (poorer performance) (Alluri et al., Reference Alluri, Solit, Leveroni, Goldberg, Vehar, Pollak and Perez2020; Jungilligens et al., Reference Jungilligens, Paredes-Echeverri, Popkirov, Barrett and Perez2022; Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a);
-
c. social–emotional processing (enhanced emotional bias/reduced emotion recognition) (Brown & Reuber, Reference Brown and Reuber2016; Pick et al., Reference Pick, Goldstein, Perez and Nicholson2019);
-
-
• Reduced post-diction metacognitive confidence (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025).
Methods
Study design and participants
This single-centre, case–control study was based at King’s College London (KCL), conducted within a larger research program (Pick et al., Reference Pick, David, Edwards, Goldstein, Hodsoll, Millman and Hotopf2024). Ethical approval was granted by the North-West Greater Manchester South National Health Service (NHS) Research Ethics Committee, United Kingdom (UK, Reference: IRAS 322652). Recruitment and data collection proceeded from 11/2023 to 04/2025.
Participants with FMS (n = 50) or FS (n = 50) were recruited from neuropsychiatry/neurology services at King’s College Hospital, South London, and Maudsley, and St George’s University Hospital NHS Foundation Trusts (London, UK). Most of the FS (82%) and FMS (96%) groups self-referred to the study following adverts shared by clinicians and/or through FND Hope UK. When self-referring to the study, participants in the FND groups were asked to provide documentary evidence of an FND diagnosis from a specialist clinician. Healthy (n = 50) and clinical (n = 50) controls were recruited using advertisements on community webpages and participant registers. Control participants were selected to frequency-match groups for age and gender (1:1 cases/controls). An a-priori power calculation indicated that a between-group (FS/FMS/CC/HC) ANOVA/ANCOVA, used to test the primary hypotheses, would require a sample of 180 to achieve 80% power for the detection of a small effect (f = 0.1, p < 0.05).
Inclusion criteria required that participants were aged 18–65 years, fluent in English, and had normal/corrected eyesight. FND samples were required to have a confirmed DSM-5 (Association, 2013) diagnosis with FS or FMS as their most prominent symptom. Clinical controls met criteria for a diagnosis of major depression and/or an anxiety disorder, confirmed with a structured clinical interview (‘Materials and Measures’). We excluded candidates with active severe psychiatric disorder (i.e., severe alcohol/substance use disorder, psychosis) and current major neurological diseases and/or lesions (e.g., neurodegenerative disorder, active epilepsy). Other exclusion criteria were being unable to perform tasks due to significant physical/cognitive impairment, current participation in an interventional trial, lifetime diagnosis of FND (HC/CC), or the presence of concurrent FS and FMS within the previous 12 months (FND). Individuals with a primary diagnosis of FCD were ineligible.
Materials and measures
Clinical measures (interview, self-report questionnaires)
A medical history interview was conducted at baseline (SP/LSMM/SB), including modules from the Quick Structured Clinical Interview for DSM-5 Disorders (First & First, Reference First and First2021) (Psychosis, Alcohol/Substance Use Disorder, Affective Disorders, Anxiety Disorders). Clinical self-report questionnaires (Supplementary Table 1) assessed depression, generalised anxiety, psychological/somatoform dissociation, physical symptoms, health-related quality-of-life, and general functioning (Pick et al., Reference Pick, David, Edwards, Goldstein, Hodsoll, Millman and Hotopf2024). A study-specific scale was developed to quantify FND symptom severity/impact (Supplementary Table 2).
The Cognitive Failures Questionnaire (CFQ) (Broadbent, Cooper, FitzGerald, & Parkes, Reference Broadbent, Cooper, FitzGerald and Parkes1982) assessed self-reported cognitive symptoms over the preceding 6 months, such as everyday memory and attention lapses, and disrupted action sequences. Higher scores signify poorer subjective cognitive functioning. Satisfactory psychometric properties have been demonstrated for this measure (Bridger, Johnsen, & Brasher, Reference Bridger, Johnsen and Brasher2013; Broadbent et al., Reference Broadbent, Cooper, FitzGerald and Parkes1982; Wallace, Kass, & Stanny, Reference Wallace, Kass and Stanny2002).
Medical Symptom Validity Test (MSVT)
The MSVT is a computerised test designed to examine neuropsychological test performance validity, with sound psychometric properties (Green, Reference Green2005; Green & Flaro, Reference Green and Flaro2016; Green, Montijo, & Brockhaus, Reference Green, Montijo and Brockhaus2011). Performance validity indicators are Immediate and Delayed Recall scores, and immediate-delayed Consistency scores (test failure indicated by scores ≤85%). This test was included to assess task engagement/negative response biases across groups (McWhirter, Ritchie, Stone, & Carson, Reference McWhirter, Ritchie, Stone and Carson2020). A pooled failure base-rate of 33% has been reported in previous FS samples (Roor, Peters, Dandachi-FitzGerald, & Ponds, Reference Roor, Peters, Dandachi-FitzGerald and Ponds2024).
Wechsler Abbreviated Scale of Intelligence – 2nd edition (WASI-II)
The WASI-II two-subtest version was administered to test non-verbal and verbal intellectual abilities (Matrix Reasoning/Vocabulary) and to provide an estimated Full-Scale Intellectual Quotient Score (WASI-II FSIQ-2). This version has strong inter-rater reliability (.95–.99), internal consistency (0.94), and test–retest stability (.94) (Wechsler, Reference Wechsler2011).
Cambridge Neuropsychological Test Automated Battery (CANTAB) Connect
The CANTAB Connect platform is a modified version of the original battery, administered using a touchscreen device. Selected subtests probed motor/cognitive response times, attention, executive functioning, social–emotional cognition, and memory (Table 1, Supplementary Table 3). The original battery has strong psychometric properties in clinical and non-clinical samples (Robbins et al., Reference Robbins, James, Owen, Sahakian, McInnes and Rabbitt1994, Reference Robbins, James, Owen, Sahakian, Lawrence, McInnes and Rabbitt1998), although psychometric data for the Connect version are not currently available.
CANTAB Connect subtests

Adapted from Pick et al. (Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023).
Metacognitive confidence ratings
Post-diction metacognitive confidence ratings were obtained immediately after every test (1 = Very poor performance; 2 = Poor performance; 3 = Below average; 4 = Average; 5 = Above average; 6 = Superior; 7 = Very superior). Such metacognitive assessments have been used widely in other clinical disorders (Seow et al., Reference Seow, Rouault, Gillan and Fleming2021), capturing short-term, test-level metacognitive judgements.
Procedure
The recruitment and screening process is detailed in the study protocol (Pick et al., Reference Pick, David, Edwards, Goldstein, Hodsoll, Millman and Hotopf2024). After obtaining written consent, the medical history interview was conducted remotely. Eligible participants were sent a link to the online questionnaires to be completed within 48 hours of the research visit. The cognitive battery was administered in a quiet testing room, between 10 am and 12 pm for most participants. The session commenced with the WASI-II, followed by the CANTAB battery (fixed order, Table 1), then the MSVT. Participants were provided with standardised instructions for each test. Abstinence from caffeine/nicotine for >2 hours before the visit was requested of all participants. Those on psychotropic medications that might alter cognition were asked to abstain for 12–24 hours, provided abstinence was unlikely to cause adverse consequences (self-report/judgement of principal investigator/research group). Participants received a £50 shopping voucher on completion of the session.
Data processing and statistical analyses
Data analyses were conducted in SPSS (v29, IBM, 2022) by SP and EGG independently, following a pre-registered analysis plan (https://osf.io/grh89/). Assumptions were checked for each test, and if violated, suitable corrections were implemented (‘Results’). We defined outliers as scores of 2.5 standard deviations (+/−) from the group mean for each variable. Analyses were rerun with outliers winsorised in sensitivity analyses. Some participants failed to complete tests due to technicalities, symptoms, or declining (‘Results’). There were no within-test missing data points because each test required all trials/elements to be completed to yield a valid score.
Categorical variables were examined with Fisher’s exact/chi-squared tests. One-way Analysis of Variance (ANOVA) was used for between-group comparisons with most continuous variables. Mixed-model ANOVA was used for the CANTAB Emotion Recognition Test, with group as the between-groups factor (FMS/FS/CC/HC) and emotion (happiness, disgust, anger, fear, sadness, surprise) as the within-subjects factor. Significant main effects/interactions were examined with Bonferroni or Games–Howell post hoc tests for ANOVA/Welch’s ANOVA. Cramer’s V, eta-squared, partial eta-squared, and omega-squared were calculated as effect sizes, as appropriate. Analysis of Covariance (ANCOVA) was applied in sensitivity analyses to examine the influence of potentially relevant confounding variables, such as performance validity, education, intellectual functioning, age, gender, and psychotropic medication. Results that did not withstand sensitivity analyses were considered uninterpretable and not considered further.
Exploratory Spearman’s correlations were run between variables that differed significantly between groups. Benjamini–Hochberg corrections were applied within each set of tests to control the false discovery rate (5%). Results remaining significant after correction are reported.
Results
Sample characteristics
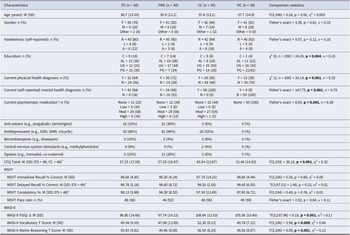
Sociodemographic and clinical characteristics of the 200 participants are presented in Table 2, including common psychotropic medications and comorbid diagnoses (additional details provided in Supplementary Tables 4–5). The groups were comparable in age, gender, and handedness. Significant overall group differences were found for self-reported education, psychotropic medication, comorbid physical/mental health diagnoses, anxiety, depression, somatoform and psychological dissociation, physical symptoms, health-related quality-of-life, and work/social functioning. These results largely reflected expected differences between HC and FS/FMS/CC groups (https://osf.io/preprints/psyarxiv/8vtpe_v1). The CC and FND groups were similar in rates of physical health comorbidities (p = 0.25), anxiety (p = 0.55), and depression severity (p = 0.50), but they differed significantly in education (p = 0.01) and psychotropic medication use (p < 0.001).
Sample characteristics, cognitive symptoms, performance validity, and intellectual functioning

Note: A, ambidextrous; AL, A levels or equivalent; C, compulsory; CC, clinical control; CFQ, Cognitive Failures Questionnaire; CGI, Clinical Global Impression; FMS, functional motor symptoms; FS, functional seizures; HC, healthy control; L, left; M, mean; Mod, Moderate; MSVT, Medical Symptom Validity Test; N, no; NA, not applicable; O, other; PG, postgraduate degree; R, right; SD, standard deviation; SNRI, selective serotonin and noradrenalin reuptake inhibitors; SSRI, selective serotonin reuptake inhibitors; UG, undergraduate degree; WASI-II FSIQ-2, Wechsler Abbreviated Scale of Intelligence–Full-Scale Intelligence Quotient 2 sub-test; Y, yes. Bold values indicate statistically significant (p < 0.05).
a Low, ‘Occasional use/very low daily dose 1–2 psychotropics’; Moderate, ‘medium-high (therapeutic) doses of 1–3 psychotropics (daily/multiple daily)’; High, ‘high-very high doses of 3 or more psychotropics (daily/multiple daily/continuous)’.
b Sample size deviated due to missing data.
c Welch’s ANOVA.
Cognitive symptoms
Self-reported cognitive symptoms (CFQ-Total) differed significantly between groups (Table 2, Figure 1), withstanding sensitivity analyses (Supplementary Table 6). Bonferroni post hoc tests (Supplementary Table 7) showed that CFQ-Total scores were elevated in the FND groups compared to HC and CC, and they were also higher in the CC group relative to HC (p-values = 0.002- < 0.001).
Cohen’s d (effect size) for cognitive symptoms (CFQ), Motor/Reaction Time (MST/RTT), Sustained Attention (RVIP), and Set-Shifting (IEDSS) tasks.

Performance validity
The MSVT performance validity test Pass rate was comparable between groups, and there were no significant differences between groups in Immediate Recall, Delayed Recall, and Consistency scores (Table 2).
General intellectual functioning
On the WASI-II test (Table 2), overall intellectual functioning scores (FSIQ-2) were in the average range in all groups. There were significant differences between groups for FSIQ-2, Vocabulary, and Matrix Reasoning scores. Pairwise comparisons (Supplementary Table 8) indicated that performance was poorer in the FS group than in CC for FSIQ-2 (p = 0.005) and Vocabulary (p = 0.006). Both FS and FMS groups showed reduced performance on Matrix Reasoning compared to HC and CC (p-values = 0.005- < 0.01). The group differences in WASI-II performance survived all sensitivity analyses (Supplementary Table 9).
CANTAB test performance
Statistical values for the CANTAB performance outcomes that varied between groups are shown in Table 3 and Figure 1.
CANTAB results – Motor/reaction time, sustained attention, and set-shifting

Note: CC, ‘clinical controls’; FS, ‘functional seizures’; FMS, ‘functional motor symptoms’; HC, ‘healthy controls’; IEDSS = Intra-Extra Dimensional Set-Shift’; M, ‘mean’; ms, ‘milliseconds’; MST, ‘Motor Screening Test’; RTT, ‘Reaction Time Test’; RVIP, ‘Rapid Visual Information Processing’; SD, ‘standard deviation’. Bold values indicate statistically significant (p < 0.05).
a Welch’s ANOVA.
CANTAB Motor Screening Test (MST) – Mean Motor Latency diverged significantly between groups (Table 3), remaining so in all sensitivity analyses (Supplementary Table 10). Games–Howell post hoc tests (Supplementary Table 11) showed extended latency in the FMS group compared to both HC (p < 0.001) and CC groups (p < 0.001), and in FS relative to CCs (p = 0.02).
CANTAB Reaction Time Test (RTT) – Mean Reaction Time and Mean Movement Time scores were significantly different across groups (Table 3). Games–Howell pairwise tests (Supplementary Table 11) demonstrated significantly elevated scores (slower responses) in both FND groups compared to HC/CC (p-values = 0.01− < 0.001). As delayed response speed in the FND groups could unduly impair performance on several other CANTAB tests, MST and RTT variables were entered as covariates in sensitivity analyses for all other CANTAB outcomes.
Sustained attention test performance (CANTAB RVIP) differed significantly between groups on several indices – Ability, Total Misses, Probability of Hit, and Median Response Latency (Table 3). These differences endured all sensitivity analyses (Supplementary Table 12), aside from the group effect on RVIP Median Response Latency, which became non-significant after excluding MSVT fails and/or including RTT Mean Reaction Time as a covariate. Both FND groups displayed significantly inferior performance to HC and CCs on all other RVIP outcomes, assessed with Bonferroni post hoc tests (Supplementary Table 13, p-values = 0.03- < 0.001).
Set-shifting test performance (CANTAB IEDSS) deviated significantly between-groups on Total Errors, Adjusted Errors, Pre-Extra-Dimensional Shift Errors, Total Trials Completed, and Response Latency (Table 3). These results remained stable in sensitivity analyses (Supplementary Table 14), except Adjusted Error and Pre-Extra-Dimensional Shift Errors, which dissipated after controlling for MSVT fails, education, gender, and/or response latency (CANTAB MST/RTT variables). Post hoc analyses confirmed that the FS and FMS groups had significantly more IEDSS Total Errors and Total Trials Completed, and showed extended Response Latency, compared to CCs (Supplementary Table 15, p-values = 0.01−0.001).
None of the significant group main effects and/or interactions on CANTAB Spatial Span, Stop Signal, Emotion Bias, or Emotion Recognition test performance remained stable in sensitivity analyses accounting for MSVT fails, education, FSIQ-2 performance, psychotropic medication, and/or CANTAB MST/RTT scores (Supplementary Tables 16–22).
Metacognitive confidence ratings
Both FND groups expressed significantly lower post-diction confidence for sustained attention test (CANTAB RVIP) performance than HCs, and the FMS group also reported reduced confidence on this task compared to CCs (Supplementary Tables 23–25, p-values<0.001). The groups did not differ in post-diction confidence ratings for any other test.
Exploratory correlational analyses
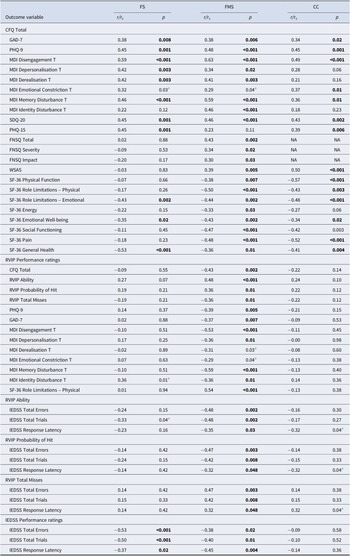
Within-groups (FS/FMS/CC) correlations examined potential associations between sustained attention and set-shifting test outcomes (CANTAB RVIP/IEDSS), post-diction metacognitive confidence ratings, self-reported cognitive symptoms, and other clinical variables (Table 4). Only correlations that remained significant after Benjamini–Hochberg correction are reported.
Significant within-group correlations

Note: CC, ‘clinical controls’; CFQ, ‘Cognitive Failures Questionnaire’; FMS, ‘functional motor symptoms’; FNSQ, ‘Functional Neurological Symptoms Questionnaire’; FS, ‘functional seizures’; GAD-7, ‘Generalised Anxiety Disorder-7’; IEDSS=Intra-Extra Dimensional Set Shift’; MDI, ‘Multiscale Dissociation Inventory’; PHQ, ‘Patient Health Questionnaire’; RVIP, ‘Rapid Visual Information Processing’; SDQ-20, ‘Somatoform Dissociation Questionnaire-20’; SF-36-Short-Form Health Survey-36 item’; WSAS, ‘Work and Social Adjustment Scale’. Bold values indicate statistically significant (p < 0.05).
a Did not survive Benjamini–Hochberg correction (5%).
Sustained attention and set-shifting performance outcomes
In the FMS group, significant associations were found between performance outcomes on both the sustained attention (CANTAB RVIP) and set-shifting tasks (CANTAB IEDSS).
Performance outcomes on the sustained attention (CANTAB RVIP) and set-shifting (CANTAB IEDSS) tests did not correlate significantly with self-reported cognitive symptoms (CFQ Total) in any group. However, in both FMS and FS groups, poorer set-shifting task outcomes (CANTAB IEDSS) were significantly associated with reduced post-diction confidence on that test. Additionally, in the FMS group, worse performance on the sustained attention test outcomes (CANTAB RVIP) correlated with lower post-diction confidence ratings for the task.
Clinical variables
Self-reported cognitive symptoms (CFQ-Total) correlated positively with several clinical features in the FND/CC samples, including anxiety, depression, and psychological/somatoform dissociation. CFQ-Total scores were also related to physical symptoms in the FS/CC groups, and to greater FNS severity and impact in the FMS group. Furthermore, CFQ-Total scores were associated with work/social functioning in the FMS/CC groups, and quality-of-life in FMS/FS/CC groups.
In the FMS sample only, post-diction metacognitive confidence for sustained attention performance (RVIP) correlated with self-reported cognitive symptoms, psychological dissociation, depression, anxiety, and role limitations due to physical factors.
Discussion
We examined self-reported cognitive difficulties, neurocognitive test performance, and metacognitive confidence in patients with FMS or FS, compared to healthy and clinical controls (HC/CC). As predicted, the FND groups reported greater self-reported cognitive symptom burden, combined with localised performance impairments on tests of sustained attention and set-shifting. In contrast to our hypotheses, there were no consistent group differences in performance on tests of response inhibition, working memory, or social–emotional processing. In the FMS and/or FS groups, impaired test performance in sustained attention and/or set-shifting was associated with reduced metacognitive confidence in those domains, suggesting broadly accurate self-appraisals in those domains.
Self-reported cognitive symptoms
Elevated self-reported cognitive symptoms in our FS and FMS samples replicated several studies (Butler et al., Reference Butler, Shipston-Sharman, Seynaeve, Bao, Pick, Bradley-Westguard and Nicholson2021; Heintz et al., Reference Heintz, van Tricht, van der Salm, van Rootselaar, Cath, Schmand and Tijssen2013; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Teodoro et al., Reference Teodoro, Edwards and Isaacs2018; Vechetova et al., Reference Vechetova, Nikolai, Slovak, Forejtova, Vranka, Strakova and Serranova2022), showing that subjective cognitive symptoms are a significant challenge for individuals with FND, beyond those with a primary diagnosis of FCD. As found in other populations, cognitive difficulties were significantly related to a range of clinical features in our FS/FMS samples, but they did not correlate meaningfully with test performance deficits (Forejtova et al., Reference Forejtova, Serranova, Sieger, Slovak, Novakova, Vechetova and Edwards2023; Goodman, Timpano, Llabre, & Bainter, Reference Goodman, Timpano, Llabre and Bainter2022; Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023; Van Patten et al., Reference Van Patten, Mulhauser, Austin, Bellone, Cotton, Chan and LaFrance2025). Clinical correlates of self-reported cognitive symptoms included depression, anxiety, dissociation, physical symptoms, quality-of-life, and work/social functioning, with some of these associations more prominent in our FMS sample. These self-reported cognitive concerns were heightened in our FS/FMS samples compared to CCs, who were comparable in anxiety and depression severity.
Together, these findings underscore the prominence and potential impact of cognitive concerns in FND and provide preliminary evidence that these difficulties may be particularly relevant in patients with FMS. Our results also indicate that self-reported cognitive symptoms in FND may not be due solely to the presence of elevated psychological distress (i.e., anxiety/depression). Future research should evaluate the extent to which self-reported cognitive symptoms in FND are related to observable cognitive deficits in real-world settings, in addition to ascertaining the direction of influence between self-reported cognitive symptoms and other clinical outcomes. Importantly, patients with FMS and FS may require targeted interventions to facilitate management of cognitive concerns and their impact in everyday life (Poole et al., Reference Poole, Cope, Vanzan, Duffus, Williams, Mantovani and Howard2025).
Neurocognitive test performance
Test performance indicated reduced intellectual functioning and extended motor/cognitive response speed in one or both FND groups in this study, corresponding with some previous reports (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a), although intellectual functioning was in the normal range in all groups in this study. Nonetheless, some of our primary results failed to survive sensitivity analyses, including one or more of these variables, reinforcing the importance of assessing their influence in future neuropsychological studies in FND.
The lack of robust group differences in performance on tests of response inhibition, working memory, and social–emotional processing tests countered our predictions, adding to the inconsistent findings in these domains (Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Pick et al., Reference Pick, Goldstein, Perez and Nicholson2019; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a). More thorough assessment of constituent processes with multiple tests may be necessary to uncover disorder-specific deficits in these cognitive functions.
Performance deficits on sustained attention (CANTAB RVIP) and set-shifting (CANTAB IEDSS) tasks exhibited by our FS and FMS samples withstood sensitivity analyses, therefore representing compelling evidence for demonstrable, specific executive function difficulties in these groups. The findings relating to sustained attention performance align with prior accounts of attentional control impairments in FND samples (Alluri et al., Reference Alluri, Solit, Leveroni, Goldberg, Vehar, Pollak and Perez2020; Millman et al., Reference Millman, Williams, Jungilligens and Pick2025; Van Patten et al., Reference Van Patten, Austin, Cotton, Chan, Bellone, Mordecai and LaFrance2024a; Vechetova et al., Reference Vechetova, Nikolai, Slovak, Forejtova, Vranka, Strakova and Serranova2022). Our observation of weaker performance on the set-shifting task (CANTAB IEDSS) in the FND groups differed from two previous studies that used the same test (O’Brien et al., Reference O’Brien, Fortune, Dicker, O’Hanlon, Cassidy, Delanty and Murphy2015; Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023), although the pattern of deficits seen here suggested overall rule-learning difficulties during the task, rather than set-shifting performance specifically. Effect sizes were large for sustained attention test performance in this sample, whereas they were small to medium for set-shifting performance, demonstrating that differences in the former domain might be more clinically meaningful.
Impaired attentional control might represent a valuable neurocognitive marker in FS and FMS, characterised by diminished maintenance of attentional focus, together with difficulties in learning implicit rules and/or voluntarily switching from a particular cognitive set. However, given that test performance in these domains did not correlate meaningfully with clinical outcomes (e.g., FND, cognitive, psychological, and physical symptoms, quality-of-life, work/social functioning), the relevance of these differences to FND should be scrutinised in greater detail. Investigators may also examine executive function performance in FS/FMS relative to patients with FCD, to identify possible shared and distinctive profiles and mechanisms in these subgroups.
Performance validity
A minority of participants in this study failed the performance validity test (MSVT), although the proportion was not significantly different between the FS/FMS/CC/HC groups (mean failure rates = 2–8%), consistent with a number of other studies providing no clear evidence of weaker performance validity in FND than in other clinical groups (McWhirter et al., Reference McWhirter, Ritchie, Stone and Carson2020). The rates of MSVT failure in the present study were considerably lower than pooled performance validity test failure base-rates observed across clinical populations (16%) and in FS specifically (33%), although these values were based on patients being evaluated in clinical settings (Roor et al., Reference Roor, Peters, Dandachi-FitzGerald and Ponds2024). As previously noted, various methodological issues may introduce bias in performance validity testing studies, such as inclusion of individuals with external motivation (e.g., ongoing litigation), medication-related impairments, and/or with psychiatric, cognitive and/or physical symptoms that might alter motivation or attentional focus on the tasks (McWhirter et al., Reference McWhirter, Ritchie, Stone and Carson2020; Roor et al., Reference Roor, Peters, Dandachi-FitzGerald and Ponds2024).
With the inclusion of both HC and CC groups in this statistically powered study, we were able to demonstrate that performance validity was not unduly disrupted in our FS and FMS samples, while accounting for the presence of psychotropic medications and general psychiatric symptoms (i.e., depression, anxiety). Our findings strengthen the literature showing intact effort and engagement in neuropsychological testing in FND (McWhirter et al., Reference McWhirter, Ritchie, Stone and Carson2020). Nonetheless, it is notable that most previous studies focused on performance validity in FS samples only, with less data available for FMS or other FND subgroups. Inclusion of performance validity measures in future studies in all FND phenotypes would be valuable to ensure that this potential confound is consistently accounted for.
Metacognitive confidence
Participants with FS and FMS expressed markedly reduced post-diction metacognitive confidence on the sustained attention task, mirroring the performance deficit displayed on this measure. Actual performance and post-diction confidence ratings on the set-shifting task (CANTAB IEDSS) were correlated within both FND groups, but not in HC/CC. Furthermore, sustained attention performance (CANTAB RVIP) correlated with post-diction confidence in the FMS group only, not in FS/CC/HC groups. These results indicate that the FND groups were generally accurate in appraising their performance on these measures, and possibly more so than the CC/HC comparison groups, suggesting intact metacognitive sensitivity in the domain of cognitive control (i.e., the statistical relationship between test performance and confidence ratings). This contrasts with our hypothesis and some theoretical perspectives, although it is consistent with several previous studies that have not supported the presence of local metacognitive deficits in FND samples (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025). As such, the emerging picture is that second-order metacognition may be unimpaired in FND, countering the proposition that altered self-evaluation of cognitive processing is an underlying mechanism in the disorder (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025).
In this study, post-diction metacognitive confidence ratings for sustained attention test performance, but not actual performance on that test, correlated significantly with self-reported cognitive symptoms in the FMS group, replicating our pilot findings (Pick et al., Reference Pick, Millman, Sun, Short, Stanton, Winston and Chalder2023). Lower post-diction confidence for sustained attention performance was also associated with other adverse clinical characteristics in the FMS group. Therefore, reduced local metacognitive confidence in attentional control might contribute to generally increased subjective cognitive concerns and/or other clinical outcomes in FMS, beyond the degree of actual performance deficits displayed. If replicated, these results imply that short-term (local) attentional performance self-appraisals may be important treatment targets in cognitive rehabilitation programs for patients with FND experiencing disruptive cognitive symptoms.
Our findings highlight some clear directions for future research on metacognition in FND. More rigorous experimental studies are needed to examine local metacognition on a trial-by-trial basis, to examine metacognitive efficiency (i.e., performance-controlled assessment of metacognitive sensitivity), and to utilise a comprehensive range of established local metacognitive metrics (Fleming & Lau, Reference Fleming and Lau2014), with careful consideration of potential confounds (Sadnicka et al., Reference Sadnicka, Strudwick, Grogan, Manohar and Nielsen2025). These experimental measures could be combined with post-diction test-level confidence ratings and measures of global metacognition, as used in the present study, to provide multidimensional assessments in specific FND subgroups (e.g., FS, FMS, FCD, functional sensory symptoms). Further investigations should also clarify the direction of effects between local metacognition for attentional control and relevant clinical outcomes (e.g., self-reported cognitive concerns, depression, anxiety). Finally, an examination of the potential benefits of targeted metacognitive training on cognitive symptoms and other clinical outcomes seems warranted, with a particular focus on improving local metacognitive confidence in cognitive control.
Strengths and limitations
The major strength of this study was the direct comparison of two common FND subgroups using the same test battery, with a sample size providing adequate statistical power to detect small effects. Inclusion of HC and CC groups provided insights into cognitive features in FND that were distinct from the general population, while controlling for elevated psychological distress. We employed a range of measures to comprehensively assess cognitive performance, along with two measures of metacognitive functioning (self-reported cognitive symptoms, test-specific post-diction confidence), in addition to a test of performance validity. Our groups were comparable on most relevant background characteristics (e.g., age, gender, handedness), and we accounted for any pertinent group differences within our analyses (e.g., education, response speed, psychotropic medications). Examining potential relationships between cognitive variables and other clinical features allowed us to explore the clinical significance of neurocognitive differences in our FS/FMS/CC samples.
Some limitations of this study should be considered. We did not include a group with FCD because this was beyond the remit of the broader research program within which this study took place. While we controlled statistically for the influence of general intellectual functioning, education, psychotropic medications, and other confounds, there may have been residual effects on our results. A minority of our FND/CC samples reported diagnoses of attention-deficit hyperactivity disorder, which may have affected attentional performance. The use of a retrospective questionnaire to assess self-reported cognitive symptoms may have resulted in recall biases. Finally, the CANTAB Connect test battery currently lacks robust psychometric evaluation and has limited ecological validity.
Conclusions
Our findings provide robust evidence of performance deficits on tests of sustained attention and set-shifting in FS and FMS, which were associated with reduced post-diction metacognitive confidence in these domains, and which were accompanied by elevated self-reported cognitive symptoms in daily life. Poorer subjective cognitive functioning was associated with greater physical and psychological symptom burden, decreased work and social functioning, and worse quality-of-life, in patients with FND and/or common psychological symptoms (anxiety/depression). Neuropsychological rehabilitation approaches might hold promise for some patients with FMS and FS, specifically targeting cognitive control and associated self-appraisals in everyday activities, and facilitating self-management of subjective cognitive symptoms and their impact.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103420.
Data availability statement
The anonymised dataset will be made available in response to reasonable requests, on a case-by-case basis. Completion of a Data Sharing Agreement will be required, facilitated by the King’s College London Data Management team.
Acknowledgements
We are grateful to all participants and the FND Patient and Carer Advisory Panel who oversaw the study. With thanks to FND Hope UK, clinicians and administrative staff at King’s College Hospital, South London and Maudsley, and St George’s University Hospital NHS Foundation Trusts (including Dr Simon Harrison, Dr Albert Joseph, Dr Sotiris Posporelis, Dr Norman Poole, Dr Tim Segal), and the RADAR-MDD team, for contributions to recruitment and screening. We thank NIHR BioResource volunteers for their participation, and gratefully acknowledge NIHR BioResource centres, NHS Trusts, and staff for their contribution. Thanks also to the research assistants who assisted in some testing sessions (Chrystel-Jazz Campbell, Akshaana Manoharan).
Author contribution
Conceptualisation: S.P.; Data Curation (Data Processing/Preparation): L.S.M.M., E.G.G., S.P.; Formal Analysis: S.P., E.G.G.; Funding Acquisition: S.P., M.H., T.C.; Investigation (Data Collection/Recruitment): L.S.M.M., S.P., Y.B., J.U.J., J.D., L.B., S.B., J.C., B.S.; Methodology: S.P., M.H., T.C., A.S.D., M.J.E., L.H.G., M.A.M., T.R.N., B.S., J.S.W.; Project Administration: S.P., L.S.M.M.; Resources: S.P., T.C., M.H.; Supervision: S.P., T.C., M.H., A.S.D., M.J.E., L.H.G., M.A.M., T.R.N., B.S., J.S.W.; Validation: S.P., E.G.G.; Visualisation: S.P.; Writing (Original Draft): S.P. (Introduction, Methods, Results, Discussion, Supplementary Materials), E.G.G. (Results/Supplementary Materials); Writing (Review/Editing): S.P., T.C., J.C., A.S.D., M.J.E., L.H.G., J.H., M.H., M.A.M., L.S.M.M., B.S., J.S.W.
Funding statement
The study was funded by a Medical Research Council Career Development Award to SP [MR/V032771/1]. This paper represents independent research part-funded by the National Institute for Health and Care Research (NIHR) Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care. The funders of this study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Competing interests
The authors declare none.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.