Introduction
The traditional radiation oncology workflow requires a computed tomography simulation (CT-sim) appointment. This is the foundation of the treatment planning paradigm, as the CT-sim provides accurate electron density information for dose calculation and high anatomical visualisation for delineation. Scanning the patient in the treatment position also allows for reproducible treatment delivery through the entirety of the treatment course to reduce margins. In recent years, direct-to-unit (DTU) workflows have increased in popularity. DTU, also referred to as simulation-free or diagnostic scan-based planning, involves forgoing CT-sim and using a patient’s diagnostic imaging (DI) to create a treatment plan. Reference Schiff, Zhao and Huang1–Reference Roderick, Wong and Kejda3 Several experiences have been published, ranging from in silico planning studies to the clinical use of DTU for online adaptive intensity modulated radiation therapy (IMRT) treatments (ART DTU) (Table 1). Reference Schiff, Zhao and Huang1,Reference O’Neil, Laba and Nguyen2,Reference Glober, Kubli and Kielbasa4–Reference Nelissen, Verbakel, Slotman, Visser and Versteijne7 These studies demonstrate that DTU is feasible and decreases the time burden of radiation as well as improving patient satisfaction. Given the increased interest in implementing DTU clinically, and specifically ART DTU, we herein aim to delineate practical considerations for implementing DTU workflows in the clinic.
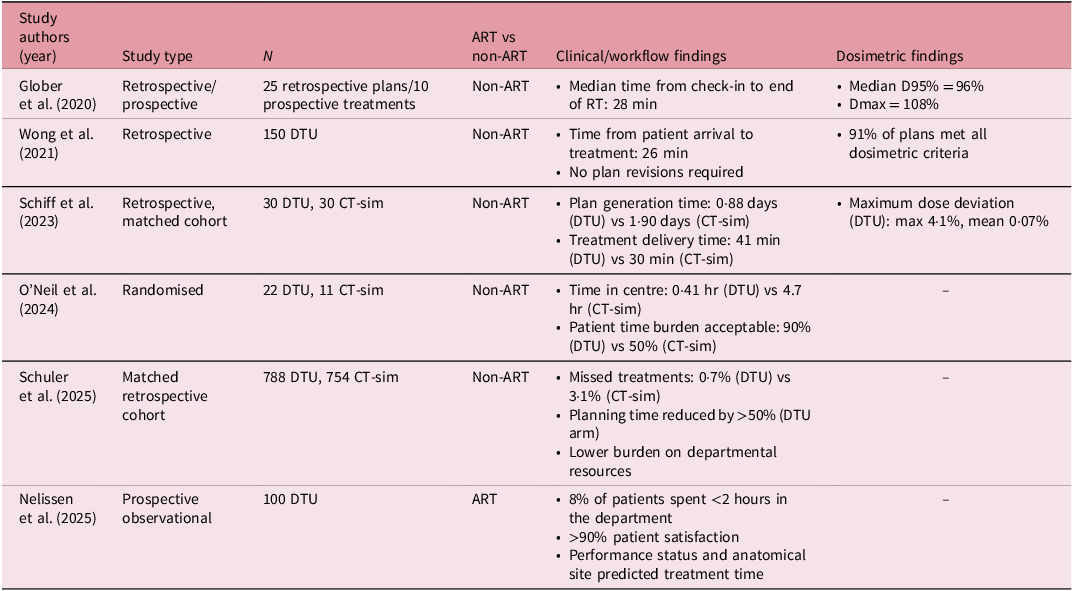
Direct-to-unit studies. A non-exhaustive list of both adaptive (ART) and non-adaptive (non-ART) DTU studies sampled from PubMed. These studies were identified through a targeted, non-exhaustive search of the PubMed database. Search terms included combinations of ‘direct-to-unit’, ‘simulation-free radiotherapy’ and ‘adaptive radiotherapy’, with a focus on clinical feasibility, workflow efficiency and dosimetric outcomes. The intent of this table is to provide a representative overview of the evolving DTU literature rather than a comprehensive or systematic review

Innovation
Basic workflow
Both ART and non-ART DTU workflows begin with the patient consultation. An overview of the workflow is demonstrated in Figure 1. From there, both physician and physicist typically work to confirm an appropriate DI to use for treatment planning. The DI image is then imported into the treatment planning system, and contours are drawn. From here, the workflows for non-ART and ART DTU diverge somewhat, and therefore, it is essential to understand the conceptual and operational differences between the two.
Direct-to-unit workflow. An overview of the DTU workflow. Note that the workflows for both ART and non-ART DTU are fairly similar until the day of treatment.

Non-adaptive DTU workflow
In a non-ART DTU workflow, the DI is used to create a single treatment plan intended for delivery without modifications. This approach relies on appropriate contouring and larger margin selection that must fully account for anatomical variability between the DI and treatment day as well as throughout the treatment course. Reference Schiff, Zhao and Huang1 Patients chosen for non-ART DTU are usually treated with conventional palliative radiotherapy, often for metastatic disease, where anatomy is expected to remain relatively stable. In this context, both the physician and physicist verify that the DI is of adequate quality, recency and field-of-view to support contouring, beam arrangement and dose calculation, as no plan adjustments will be made on the table. On the day of treatment, advanced on-board imaging is acquired for alignment. At this time, the therapist may mark the patient if a multi-fraction treatment course is planned, so less advanced on-board imaging can be used for future fractions.
Adaptive DTU workflow
In contrast, ART DTU accounts for day-to-day anatomical variability. For ART DTU, requirements necessary during non-ART DTU have the potential to be relaxed during the ART workflow due to the ability to account for discrepancies with ART planning. Typically, ART DTU can reduce planning target volume (PTV) margins used to account for uncertainty in positional and anatomical differences between the DI and patient positioning, which would have been addressed with simulation immobilisation in the traditional CT-sim workflow. Reference Fuller, Osbourne and Beldham-Collins8–Reference Hayworth, Hotsinpiller and Pogue10 However, it is important to ensure that both the DI image and treatment image are of sufficient quality to clearly delineate both targets and organs at risk (OARs), as contouring errors are a concern in ART. Reference Noel, Santanam, Parikh and Mutic11–Reference Noel, Parikh and Spencer15 With ART DTU, daily position or differences from DI can be accounted for, reducing the need for extensive beam placement adjustments to minimise these uncertainties, which contrasts with non-ART DTU recommendations. Reference Roderick, Wong and Kejda3 Instead, considerations should be based on delivery time versus calculation time for the ART plan.
On the day of treatment, ART DTU requires a structured, time-sensitive workflow that begins with the acquisition of high-quality on-board imaging to confirm setup. The physicist performs a rapid assessment of image quality, registration fidelity and anatomical suitability for adaptation. The physician then performs on-table contour review and edits, and the physicist subsequently initiates real-time plan re-optimisation using the adaptive planning system. In non-DTU workflows, the ART plan is not always used if the initial plan meets OAR constraints and/or target metrics. However, in ART DTU, the adaptive plan is almost always used, as the adaptive nature of the treatment accounts for differences in the treatment plan resulting from changes from the DI to the day of treatment imaging. Lastly, while marking the patient may facilitate more rapid setup for future appointments in multi-fraction treatment courses, advanced on-board imaging will always be acquired to allow for the ART process.
Clinical considerations
When identifying appropriate candidates for DTU, one can separate the clinical criteria into patient and image factors. Ideal patients for DTU have medial target locations, as more lateralised targets, particularly of the extremities, tend to be harder to align to on the day of treatment. Patients who have undergone major anatomic interventions in the area adjacent to the target from the day of the DI to the day of treatment (e.g., thoracentesis for pleural effusion adjacent to a thoracic target) may be less suitable for DTU. This is less of a concern with ART, as these changes can be accounted for in adaptive planning. Patient comfort is a major factor with DTU treatments. DTU time-on-table is typically longer than for conventional palliative radiation. Reference Nelissen, Verbakel, Slotman, Visser and Versteijne7 For patients who struggle with pain, DTU may not be the best option for them, and using padding on the day of DTU may be helpful for all comers. This is even more of a concern for ART DTU, as ART is generally a relatively lengthy process and must be weighed against the benefit of ART.
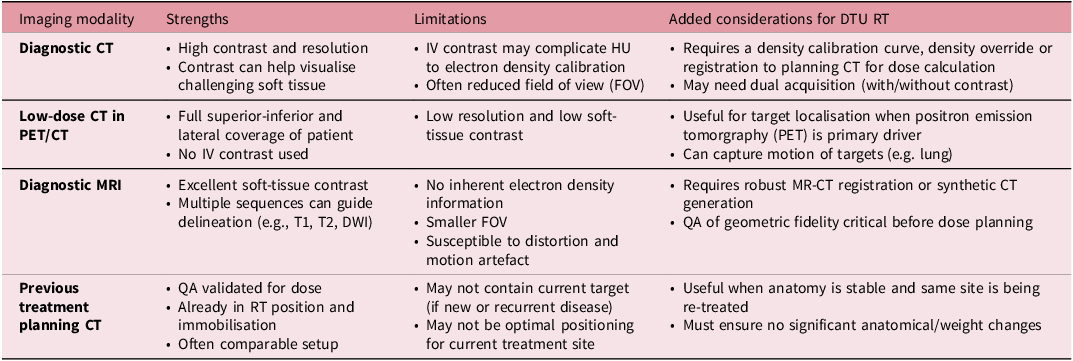
Selecting an appropriate DI is critical for DTU. It is recommended that this be done by both physician and physicist in the upfront setting to confirm that the DI is adequate for DTU prior to treatment planning. In general, DI for DTU should fully capture the body contour at the planes of the target to allow for maximal flexibility in beam arrangements and appropriate assessment of dose deposition to surrounding tissue. The recency of the DI is also an important consideration. It is generally recommended that the DI be from no greater than one to two months prior to the time of DTU. Reference Roderick, Wong and Kejda3 Further considerations are presented in Table 2.
Pre-plan image selection. A list of strengths, limitations and considerations for DTU pre-plan image selection

Taken together, these clinical factors highlight the different demands of non-ART and ART DTU workflows. Non-ART DTU depends on high-quality, representative DI and sufficient anatomical stability to support a fixed plan used across all fractions. ART-DTU, on the other hand, uses daily imaging and on-table assessment to adjust to shifts or changes in target and OAR anatomy, offering greater flexibility but requiring more staff, time and coordination.
The need for a DTU workflow in the clinic should be considered prior to implementation. Certain clinics have busy CT-sim schedules, and getting emergent patients onto treatment can be hastened in the absence of CT-sim appointments. Clinics with wide catchment areas or that provide for patients with socioeconomic challenges might find that eliminating a CT-sim appointment enables easier treatment for patients. Lastly, in resource-limited environments in which CT-sim downtime can lead to delays for patients with no other treatment options, DTU workflows can keep patients on-beam. Reference Dykstra, Netherton and Lasebikan16 However, clinics may not suffer from any of these issues, and DTU workflows might be of less interest to them.
In the United States in particular, billing is often an issue with regard to DTU treatments. CT-sim charges generally are not a part of inpatient billing, and so inpatient DTU is often not a concern for billing departments. In the outpatient setting, DTU does cause departments to lose out on the CT-sim billing charge, and this should be accounted for when creating departmental guidelines on optimal patients for DTU. However, despite the lack of CT-sim, DTU is a complex treatment planning process, and it would be of long-term interest to the field to advocate for billing codes that capture that complexity.
DTU implementation framework
Establishing a DTU workflow requires coordinated clinical, operational and technical preparation. To assist centres aiming to develop DTU capabilities, we outline a set of practical steps below. First, the clinic must assess whether there is a need for a DTU workflow. DTU can provide significant value in areas with high urgent-care demand, long CT-simulation wait times or geographically diverse patient populations in which limiting CT-simulation appointments may be of value to the patient and/or clinic flow. Next, imaging and planning infrastructure must be assessed. DTU implementation requires reliable DI, a planning system capable of accepting and processing diverse DI sources and onboard imaging of sufficient quality for registration and/or contouring. Then, physicians and physicists should develop a patient-selection protocol. Patient appropriateness should be determined based on anatomical, clinical and workflow factors, including expected anatomical stability, time-on-table tolerance, pain control needs and target laterality. From there, operational workflows can be built. Both ART and non-ART DTU require efficient coordination. Centres should create process maps detailing the necessary steps from patient arrival to treatment delivery. Physicists must also implement appropriate QA procedures. ART DTU workflows need quick QA steps designed for on-table execution without compromising safety. Next, staff training can commence. Therapists, physicists and physicians should participate in training sessions and mock DTU cases to ensure familiarity with the workflow. All centres have access to DI and cone beam CT (CBCTs) to mock these workflows and cultivate a true familiarity with anticipated workflows. Finally, the workflow may be piloted in the clinic. Using a small, well-defined patient cohort for the initial rollout with additional allocated time for potential longer treatment times during the initial few cases, then evaluate workflow efficiency and adjust protocols before expanding to broader implementation.
Physics considerations
Dose calculation, patient setup, structure delineation and quality assurance are the predominant process steps that have additional considerations or alterations when transitioning to DTU workflows. Dose calculation accuracy should be carefully considered when implementing ART DTU treatments, as it depends on the patient’s clinical situation. In the setting of CBCT-guided ART DTU, multiple methods are available to obtain electron density information, including bulk density override, deformed reference images on CBCT, electron density-accurate CBCTs and algorithmically driven approaches. All of these approaches have demonstrated remarkably similar HU values between image data sets or dose distributions and target coverage metrics, with minimal impact from the dose calculation approach. The use of an increased number of beam angles enhances the robustness of plan calculations compared to simple field arrangements, as it minimises the local impact due to tissue surface heterogeneity through the increased number of beam entry points.
As for dose calculation commissioning, we suggest creating electron density to Hounsfield Unit (ED-HU) tables specific to the scanners (both DI and CBCT) used within your hospital network and uploading them into your treatment planning system. This may require collaboration with the diagnostic imaging team to coalesce that information. Based on the size of the hospital network, it may be impractical to have specific ED-HU tables for each scanner, and thus a generic ED-HU table can be generated for DI. For CBCT within your clinic, it would be appropriate and suggested to create an ED-HU table for each CBCT used in an ART DTU workflow. However, the magnitude of uncertainty must be characterised, and the dose calculation accuracy should be within 5% for common tissues encountered in patients, such as lung, muscle, fat and bone, when comparing to dose calculation on a DI, CBCT and CT-sim. Reference Zhu, Stathakis and Clark17,Reference Geurts, Jacqmin and Jones18 Understanding the magnitude of uncertainty in varying tissue densities may dictate beam selection or treatment selection depending on plan complexity or prescribed dose.
Traditional radiation therapy workflows rely on CT-sim to create immobilisation. In the setting of DTU, increasing treatment time slots can be done for immobilisation creation or when using ART. Studies have reported increased on-table time in non-ART DTU compared to conventional workflows, so comfort in combination with immobilisation should be considered. Reference Schiff, Zhao and Huang1 With ART-DTU, lack of inter-fractional immobilisation can be accounted for with daily structure delineation. In addition, at the time of the DI review, immobilisation can also be suggested for the treatment time. For example, headrests could be identified based on the curvature of the patient’s neck on DI. Finally, based on disease site, a standard immobilisation approach should be used for all patients to limit resource allocation in treatment time slots for both the patient’s experience and for operation efficiency.
For DTU localisation and delineation, various studies have demonstrated that both visibility and delineation are non-inferior to CT-sim. Reference Price, Kang and Reynoso14,Reference Noel, Parikh and Spencer15,Reference Henke, Fischer-Valuck and Rudra19 This is especially true with next-generation CBCT systems, which are approaching equivalence to that of CT-sim. Reference Kunnen, J A J van de Schoot and Fremeijer12 We suggest delineating targets and OARs on both the DI and CBCT images of patients previously treated on a system identified for DTU treatment for commissioning. This should be done to understand differences in contouring between the two imaging systems. Assessment of how patient positioning, image quality and patient motion impact DTU alignment and/or structure delineation can be performed using quantitative methods, such as Dice similarity coefficients or asymmetric surface distances, or qualitative methods, such as physician or therapist review. If there are limited field-of-view images or poor image quality that omit important structure information, such as the target, DTU would be inappropriate, especially if not using ART DTU. Additionally, avoid beam selection in those omitted areas, again if not using ART DTU, as you will underestimate the dose to the target. However, even in the setting of ART DTU, one should expect the significant MU changes in the plan as the beam path length changes within the field-of-view, so it would be appropriate to have a range of relevant MUs based on the treatment site and prescribed dose.
Finally, your DTU workflow should be exhaustively commissioned and have relevant QA procedures in place. Reference Roderick, Wong and Kejda3,Reference O’Neil, Tan and Schuler20 Multiple end-to-end tests from DI to treatment in the relevant diseases and planning approaches are encouraged with both point- and volumetric-dose comparisons that follow consensus guidelines for treatment planning commissioning. Reference Mutic, Palta and Butker21,Reference Fraass, Doppke and Hunt22 In the early phases of DTU deployment, if feasible, pre-treatment IMRT QA should be done for scheduled non-urgent intensity-modulated treatment plans. If performing ART DTU, pre-treatment volumetric secondary dose calculation comparisons and post-treatment IMRT QA should be performed until confidence (roughly 10–20 plans) is built that the ART plans developed are technically deliverable. There are publications noting this approach. Reference Zhao, Stanley, Cardenas, Harms and Popple23–Reference Wang, Mazur, Park, Yang, Mutic and Li25 Finally, institute approaches to identify potential failure modes during the commissioning phase. This can include HU verification in varying OARs, MU ranges per intent, secondary target review or plan review like brachytherapy, beams-eye-view to target projection verification and well-structured time-out procedures.
Discussion
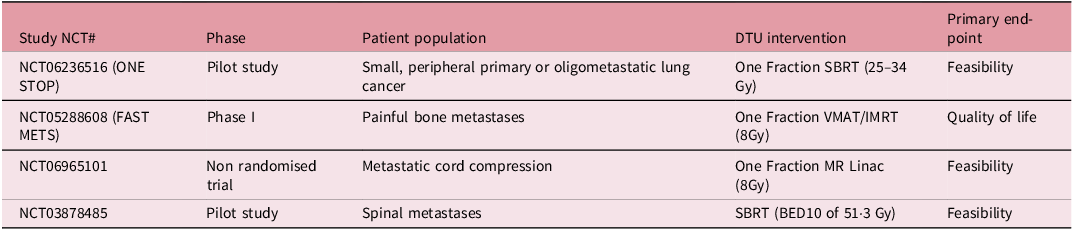
As experience continues to expand with DTU, so too will the complexity of how it is approached, especially with ART DTU. For example, setting the magnetic resonance imaging (MRI) image as the reference image or even removing the need for a DI altogether are possible with current technology. Reference Price, Kang and Reynoso14,Reference Kang, Price and Reynoso26 Additionally, as with increasing stereotactic body radiotherapy (SBRT) volume in radiation oncology, ART DTU should expect to see usage of SBRT as well, especially in the setting of single fraction SBRT for oligometastases. Reference Guevara, Rezaei and Baydoun27,Reference Price, Schiff and Silberstein28 We anticipate more prospective evidence to come on DTU in the coming years, and Table 3 highlights some ongoing prospective clinical trials in this space.
Direct-to-unit trials. A non-exhaustive list of ongoing prospective DTU clinical trials

Recommendations
Herein is a high-level overview of the DTU workflow to aid clinics in implementing this technique. DTU workflows can be very beneficial for patients, particularly when getting patients onbeam quickly is a primary concern. Clinical and physics considerations should be evaluated on a clinic-by-clinic basis to identify the optimal specific DTU workflow for your specific clinic.
Acknowledgements
Not applicable.
Financial support
Not applicable.
Competing interests
Joshua P Schiff has no competing interests. Amir Ahari has no competing interests. Lauren Henke receives grants from Varian Medical Systems, consulting fees from Varian Medical Systems and Radialogica and honoraria from ViewRay, Varian Medical Systems and LusoPalex and participates on a safety board for ViewRay. Alex Price receives grants from Varian Medical Systems and consults for Atrium Health.