Impact Statement
Tuvalu is one of the world’s smallest and most isolated nations. Despite this, our study shows microplastics are present in critical food fishes from the main atoll, Funafuti’s lagoon. We sampled 201 fish and found 37% of the individuals contained microplastic particles. By establishing the first baseline for Tuvalu, analyzing locally caught fish across 44 species, we provide clear evidence that microplastics have infiltrated subsistence diets in a setting with minimal industry, limited land and vast distance from major population centers. Given the reliance of people in Tuvalu on fish for daily nutrition, livelihoods and culture, these results are particularly concerning. The findings here help to correct geographic gaps in global microplastics research and bring the lived realities of remote atoll communities into regional and international policy conversations. Action to reduce this problem will require upstream solutions to reduce plastic at its source, not just downstream clean-ups, making these findings relevant for discussions in the Global Plastics Treaty negotiations.
Introduction
Microplastics, defined as plastic particles smaller than 5 mm, are ubiquitous in the environment (Andrady, Reference Andrady2011; Ivar do Sul and Costa, Reference Ivar do Sul and Costa2014). Numerous studies have quantified their presence and highlighted the potential risks they pose to ecosystems and human health (Wright et al., Reference Wright, Thompson and Galloway2013; Sharma and Chatterjee, Reference Sharma and Chatterjee2017; Moyo, Reference Moyo2022), yet only 44 (22.9%) of countries are represented in microplastic research (Ajith et al., Reference Ajith, Arumugam, Parthasarathy, Manupoori and Janakiraman2020), highlighting broad geographic gaps. These particles can originate from the breakdown of larger plastic debris (secondary microplastics) or be produced directly from sources such as industrial abrasives and personal care products (primary microplastics). Once released into the environment, microplastics are dispersed by waterways, currents, wind and runoff; pathways strongly influenced by particle shape, size and density (Shamskhany et al., Reference Shamskhany, Li, Patel and Karimpour2021). These dynamics ultimately lead to their accumulation in marine ecosystems and entry into food webs. In fish, ingested microplastics have been associated with structural damage to organs such as the intestines, as well as changes in metabolism, behavior and fertility (Zolotova et al., Reference Zolotova, Kosyreva, Dzhalilova, Fokichev and Makarova2022). These particles can bioaccumulate within organisms (Miller et al., Reference Miller, Hamann and Kroon2020), amplifying their ecological and health impacts, with the potential to transfer through trophic levels.
Understanding how microplastics are distributed within marine ecosystems, particularly in fish, is vital as these species may play a crucial role in transferring microplastics and related contaminants back into the human food chain (Amelia et al., Reference Amelia, Khalik, Ong, Shao, H-J and Bhubalan2021; Makhdoumi et al., Reference Makhdoumi, Hossini and Pirsaheb2023). Fish are important indicators of microplastic pollution due to their central role in food webs and their significance in human diets (de Souza et al., Reference de Souza, de Pinho, de Almeida Rodrigues, de Melo, Bergsten-Torralba and Conte-Junior2024). Quantifying microplastics in fish provides valuable insights into pollution sources, pathways and the potential threats to food security and ecosystem services (Wang et al., Reference Wang, Chen, He, Liang and Jiang2024). The impact of microplastics is of particular concern for regions such as the Pacific Islands, where marine resources are essential for food security and livelihoods (Gillett, Reference Gillett2009).
While research on microplastic pollution has been conducted in countries such as Fiji (Dehm et al., Reference Dehm, Singh, Ferreira and Piovano2020; Ferreira et al., Reference Ferreira, Thompson, Paris, Rohindra and Rico2020; Wootton et al., Reference Wootton, Ferreira, Reis-Santos and Gillanders2021a), many of the smaller nations such as Tuvalu, remain largely underexplored. As a low-lying island nation, Tuvalu is highly vulnerable to environmental pollution (Connell, Reference Connell1999), relying heavily on its marine ecosystems for sustenance and economic stability (Sauni and Sauni, Reference Sauni and Sauni2005). In Funafuti, the most populous atoll where 62% of the population lives (SPC and CSD, 2025), there is one centralized landfill, with 6–10 and 750–1,000 m3 of household and non-household waste entering the landfill daily (with an estimated 3,000 kg of waste generated daily) (Sagapolutele et al., Reference Sagapolutele, Wander and Prince2019). Household waste generation falls below global averages (Hoornweg and Bhada-Tata, Reference Hoornweg and Bhada-Tata2012) but is caveated by the lack of recycling capacities and very limited space for waste management, with illegal waste dumps and burning still prevalent (SPREP, 2016). Furthermore, landfills are not lined, and specific coastal conditions, such as lagoon hydrodynamics, may exacerbate the accumulation of degrading plastic products (Garcés-Ordóñez et al., Reference Garcés-Ordóñez, Saldarriaga-Vélez, Espinosa-Díaz, Canals, Sánchez-Vidal and Thiel2022). Despite its vulnerability to plastic pollution, no studies to date have assessed the level of microplastics in the environment in Tuvalu. Establishing baseline data on microplastic contamination is critical to assess the extent of the problem, evaluate risks to marine life and develop targeted mitigation strategies for island communities. In Tuvalu, where subsistence fishing is vital for both nutrition and cultural heritage, establishing baseline data on microplastic ingestion in fish is particularly urgent. Without such information, assessing the full scope of microplastic pollution and its potential impacts on marine ecosystems and communities becomes challenging.
This study provides foundational data on microplastic contamination in fish caught from the coastal waters of Funafuti atoll; Tuvalu’s capital and most populous atoll. It represents the first published assessment of microplastics in fish from Tuvalu, filling a critical gap in data for one of the world’s most remote island nations. The study’s objective was to quantify the frequency of occurrence and microplastic load in fish species commonly targeted by subsistence fishers. By directly sampling fish landed through subsistence fisheries, our approach ensures cultural and dietary relevance, and provides key baseline information to inform both scientific and policy dialogs.
Methodology
Sample collection
Fish samples were collected from three islets around Funafuti Atoll – Fongafale (the primary settlement where the majority of the population lives), and the smaller islets Papaelise and Funafala (Figure 1) – between July and October 2024. All fish were caught from within the Funafuti Lagoon. A total of 201 fish (133 from Fongafale, 68 from Papaelise/Funafala) from 44 species were caught by local fishers within the lagoon adjacent to these communities. Upon landing, the gastrointestinal tracts (GITs) of the fish were extracted, labeled, isolated and preserved at −20 °C at the Tuvalu Fisheries Authority laboratory. All dissections took place within a confined air-conditioned lab with stainless steel scalpels and scissors, which were thoroughly rinsed after each dissection. No plastic-based materials were used in the process. Data on fish species, local names and morphometric measurements (total length, standard length and weight) are summarized in Table 1. The complete collection record dataset is available through the Global Biodiversity Information Facility (GBIF) (Nivaga et al., Reference Nivaga, Kaitu, Lopati, Makolo and Alefaio2024). The fish GITs were then frozen and air-freighted to the University of the South Pacific in Suva (Fiji) for subsequent analysis.
Map of Funafuti Lagoon.

Summary of fish species sampled from Funafuti atoll, Tuvalu, including morphometric measurements (mean ± SD) for total length, fork length and total weight

Note: Note that fish from Papaelise and Funafala were collected from a collective area towards the southern end of the atoll, thus ‘Papaelise/Funafala’ in the table. NA indicates data are not available for certain measurements.
Microplastic analysis
Microplastic extraction and analysis methods were adapted from previous studies (Foekema et al., Reference Foekema, De Gruijter, Mergia, van Franeker, Murk and Koelmans2013; Rochman et al., Reference Rochman, Tahir, Williams, Baxa, Lam, Miller, Teh, Werorilangi and Teh2015; Wootton et al., Reference Wootton, Ferreira, Reis-Santos and Gillanders2021a) and were consistent with other recent studies on microplastics in marine biota from the region (Botleng et al., Reference Botleng, Drova, Kotra, Sokach, Feremaito, Ham, Kalmer, Kaku, Molitaviti, Varea, Brown, Ford and Dehm2025; Drova et al., Reference Drova, Tuqiri, Prasad, Rosi, Gadekilakeba, Bai, Tunaka, Williams, Varea, Brown and Dehm2025; Fe’ao et al., Reference Fe’ao, Drova, Matoto, Afu, Kautoke, Fatongiatau, Botleng, Varea, Dehm, Morris, Brown and Ford2025; Powell et al., Reference Powell, Varea, Williams, Naivalurua, Brown and Dehm2025) to allow for comparability. Each GIT was placed in a glass beaker and digested with 30% hydrogen peroxide (H₂O₂), using a volume three times that of the GIT. The samples were incubated at 60 °C for approximately 12 h until all soft tissues were fully digested. The resulting liquid was filtered through a series of stainless-steel sieves with mesh sizes of 1 mm, 250 μm and 125 μm. The sieved residues were examined under a stereo microscope (Prism Optical, Banksia Scientific; up to 100x magnification) using a systematic transect approach to identify and categorize potential microplastics by type, form and size. Microplastics were classified according to size ranges based on the sieve mesh sizes (125–250 μm, 250 μm–1 mm and > 1 mm). Microplastic loads are presented as frequency of occurrence (%; per species, location and overall) and concentration (mean ± SD; per species, location and overall). A subset of identified microplastics was analyzed for polymer composition using Fourier Transform Infrared Spectroscopy (FTIR) (Spectrum Two, Perkin Elmer), following the methods outlined in previous studies (Jung et al., Reference Jung, Horgen, Orski, Rodriguez, Beers, Balazs, Jones, Work, Brignac, Royer, Hyrenbach, Jensen and Lynch2018; Dehm et al., Reference Dehm, Singh, Ferreira and Piovano2020; Ferreira et al., Reference Ferreira, Thompson, Paris, Rohindra and Rico2020), with spectra compared to reference data for microplastics as defined by Jung et al. (Reference Jung, Horgen, Orski, Rodriguez, Beers, Balazs, Jones, Work, Brignac, Royer, Hyrenbach, Jensen and Lynch2018). Spectra were collected over a range of 4,000–450 cm⁻1 at a resolution of 4 cm⁻1, following Jung et al. (Reference Jung, Horgen, Orski, Rodriguez, Beers, Balazs, Jones, Work, Brignac, Royer, Hyrenbach, Jensen and Lynch2018). All visually identified microplastics larger than 250 μm were targeted for FTIR analysis; however, only particles that were sufficiently large to be recovered and successfully transferred from the sieves to the ATR crystal were ultimately analyzed (n = 30).
Quality control
To minimize contamination, all digestion, extraction and analysis procedures were conducted in controlled laboratory conditions in batches of 30–35 samples. Cotton lab coats were worn and equipment used during the extraction was thoroughly rinsed with filtered distilled water. GIT samples, beakers and solutions were covered with aluminum foil throughout the digestion and extraction processes to reduce exposure to open air, and all solution transfers were conducted within laminar flow hoods. Additionally, at least two process control samples (exposed and handled in the same manner and for comparable durations as samples) were included in each batch, totaling 14 process control samples. No plastic-like particles were observed in the process control samples, indicating no detectable contamination throughout the study.
Statistical analyses
To compare differences among taxa and location, the data were filtered to species with at least ten individuals, resulting in seven species: Acanthurus triostegus, Epinephelus maculatus, Epinephelus polyphekadion, Gnathodentex aureolineatus, Lethrinus harak, Monotaxis grandoculis and Parupeneus barberinus. Shapiro-Wilk tests indicated that total microplastic counts were non-normally distributed (p < 0.0001). The effect of species and location differences were thus analyzed using a negative binomial generalized linear model (GLM) with species and locality (Fongafale vs. Papaelise/Funafala, with the latter two combined due to fish being collected from a collective area towards the southern end of the atoll) included as fixed effects, implemented using the ‘glmmTMB’ package (Brooks et al., Reference Brooks, Kristensen, van Benthem, Magnusson, Berg, Nielsen, Skaug, Mächler and Bolker2017). Model fit was evaluated with simulation-based residual diagnostics in the ‘DHARMa’ package (Hartig, Reference Hartig2020), confirming that model assumptions were not violated (Supplementary Figure S1). Because the seven species differed in body size, total length and total weight were compared among species using Kruskal–Wallis tests (Supplementary Table S1) followed by post-hoc pairwise comparisons using a Dunn’s test with Benjamini–Hochberg correction in the ‘FSA’ package (Ogle et al., Reference Ogle, Doll, Wheeler and Dinno2025). Correlations between microplastic load and fish body size (total weight and total length) were evaluated using Spearman’s rank correlation separately within the seven species. All data analyses were conducted in R-4.5.0 (R Core Team, 2025), and plots were drawn using the ‘ggplot2’ package (Wickham, Reference Wickham2016).
Results and discussion
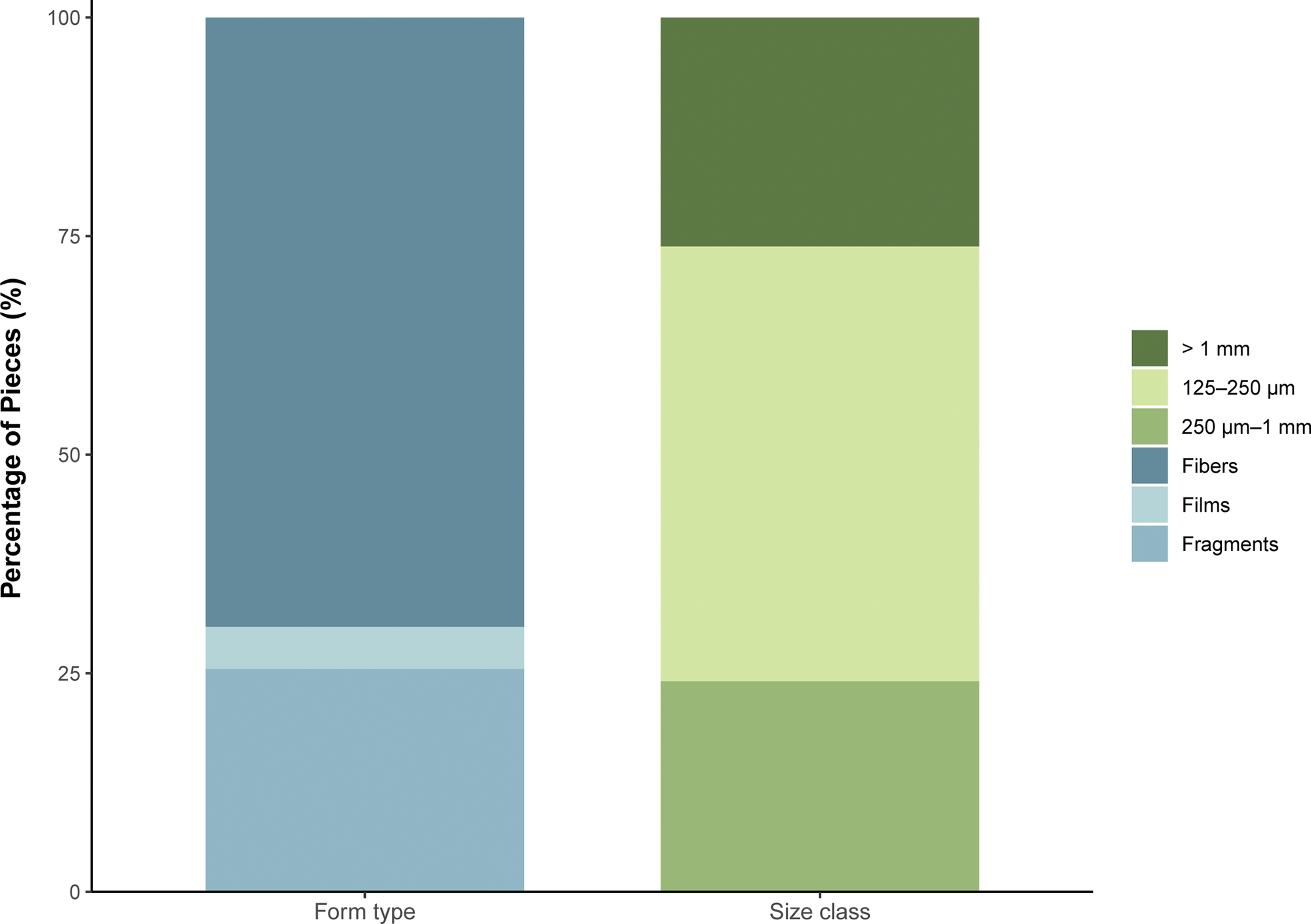
Of the 201 fish sampled, microplastics were found in 75 individuals, resulting in an overall frequency of occurrence of 37%, with microplastics occurring in 47% (n = 63) of Fongafale samples and 18% (n = 12) of Papaelise/Funafala samples. Overall in Funafuti Lagoon, a total of 145 microplastic particles were identified from the samples and comprised three form types, of which fibers were most frequent, comprising 70% (101 particles) of the samples, followed by fragments at 26% (37 particles), and films as the least frequent (only seven particles). Furthermore, particles in the 125–250 μm range were the most abundant, representing 48% (70 particles) of the total, followed by particles in the 250 μm–1 mm range, which accounted for 28% (41 particles). Microplastics larger than 1 mm constituted 24% (34 particles) of the total.
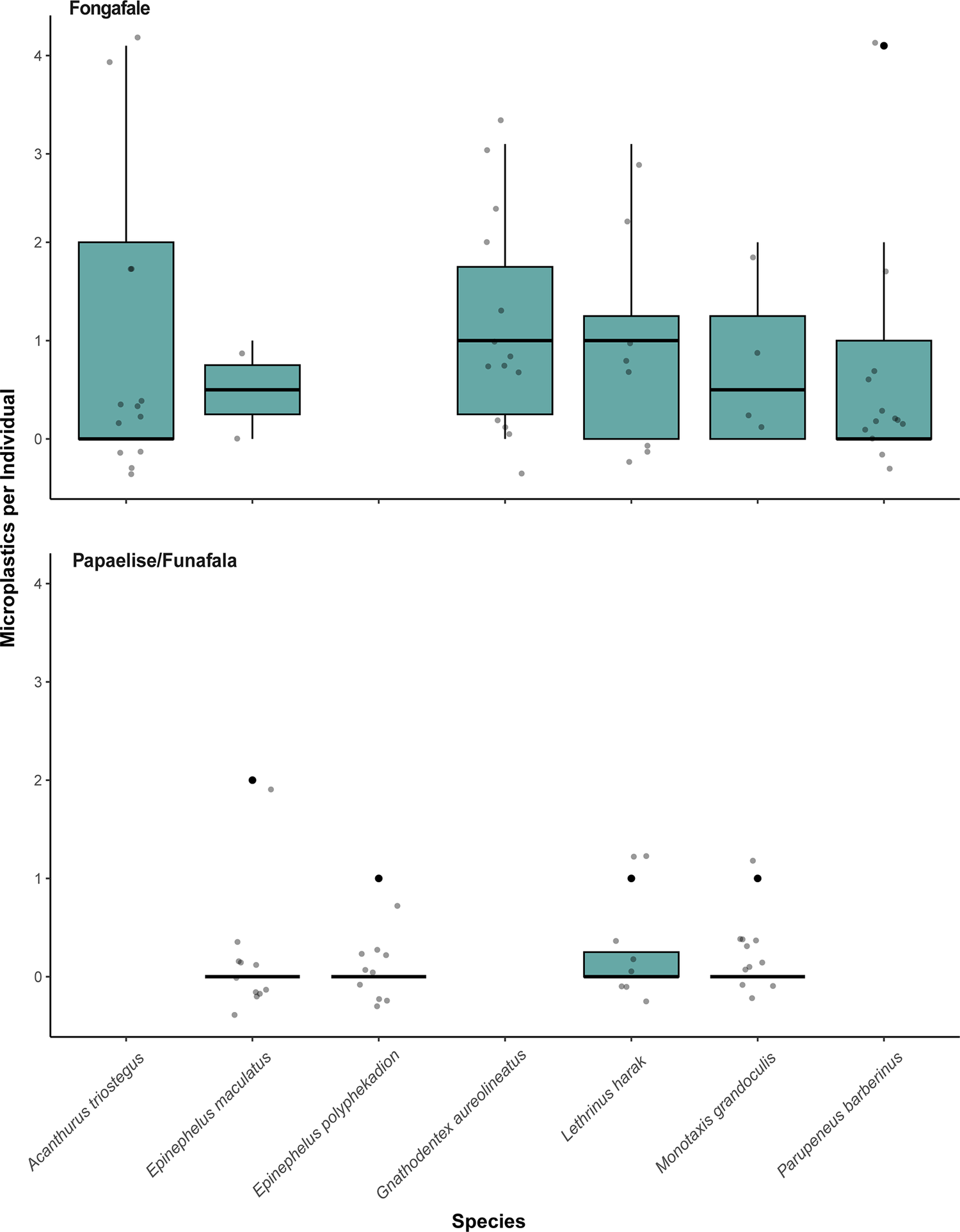
Within species, frequencies of occurrence ranged from 0% (e.g., Acanthurus xanthopterus, Caesio teres and Siganus punctatus) to 100% in others, such as Acanthurus auranticavus, Epinephelus spilotoceps, and Mulloidichthys flavolineatus (Table 2). The highest count of microplastics was five particles found in individuals of Aphareus rutilans (from Papaelise/Funafala), M. flavolineatus, Mulloidichthys vanicolensis and Sargocentron spiniferum (all from Fongafale). Microplastic load across all samples was on average 0.72 ± 1.16 particles per individual (0.95 ± 1.26 in Fongafale; 0.28 ± 0.77 in Papaelise/Funafala) and across species ranged from 0 to a maximum of 2.66 ± 2.08 particles per individual in M. flavolineatus. For the seven species with at least ten replicates, the species with the highest mean microplastic load was G. aureolineatus (1.14 ± 1.03 particles per individual), with all individuals of this species collected around Fongafale (Figure 2). Interestingly, pairwise comparisons showed that G. aureolineatus was significantly lighter than three of the other species (E. maculatus, E. polyphekadion, M. grandoculis; all p < 0.001; Supplementary Table S1) suggesting that elevated ingestion in G. aureolineatus is not simply a function of body size. Both E. maculatus and E. polyphekadion feed primarily on fish (Froese and Pauly, Reference Froese and Pauly2025) by engulfing their prey whole, while G. aureolineatus and M. grandoculis primarily feed on invertebrates in the substratum (e.g., zoobenthos) (Froese and Pauly, Reference Froese and Pauly2025). However, despite these species covering a variety of diets and feeding strategies, negative binomial GLM analyses indicated that microplastic load did not differ significantly among species (all p > 0.46). In contrast, locality had a significant effect on microplastic abundance, with fish collected from Papaelise/Funafala exhibiting markedly lower microplastic loads than those from Fongafale (Est. = −1.51 ± 0.63 SE, z = −2.39, p = 0.017); a pattern consistent with Fongafale being the main urban area and where the formal landfill is located. No significant correlations emerged between total weight or total length with microplastics load (all p > 0.05; Figure S2).
Frequency of microplastic (MP) occurrence, average MP counts and MP characteristics (type and size) across fish species

Note: Note that fish from Papaelise and Funafala were collected from a collective area towards the southern end of the atoll, thus ‘Papaelise/Funafala’ in the table. Cells are left blank for zeroes.
Microplastic concentrations for species with more than 10 replicates. Boxes represent the interquartile range (IQR), horizontal lines indicate medians and whiskers extend to 1.5× the IQR.

Fibers were the predominant form type in most species (Figure 3). Notably, they accounted for 100% of the microplastics in several species including Epinephelus merra, Myripristis berndti and Siganus argenteus (Table 2). Fragments were abundant in species such as Hipposcarus longiceps (100%) and S. spiniferum (56%). Films, while less common overall, were present in species such as A. auranticavus (50%), M. vanicolensis (33%) and Lethrinus olivaceus (33%). Microplastic size distribution also showed notable variation among species (Figure 3). The smallest size class (125–250 μm) was dominant in species such as M. grandoculis (75%) and A. rutilans (67%) (Table 2). Microplastics in the intermediate range (250 μm–1 mm) were most prevalent in G. aureolineatus (56%) and E. polyphekadion (100%). Larger particles (>1 mm) were relatively less common but featured prominently in species like P. barberinus (63%) and M. flavolineatus (50%).
Distribution of microplastic types and size classes from across the 145 microplastics identified.

A subset of 30 microplastics, from the 145 particles visually identified in the fish gut samples from Tuvalu, were analyzed for polymer composition. Five distinct polymer types were identified, with polypropylene (PP) being the most prevalent (56% of fibers, 67% of films), accounting for ~43% of the particles (Figure 4); a polymer shown to induce oxidative stress in fish (Yedier et al., Reference Yedier, Yalçınkaya and Bostancı2023). Nylon and polyethylene terephthalate (PET; 56% of fragments) each represented a total of 20% of the subset, while polyethylene (PE) constituted ~13%. Polystyrene (PS) was the least common polymer, making up only 3% of the analyzed microplastics. Polymer identification was limited to the subset of particles that were retrievable and successfully transferred for FTIR analysis, and therefore may not fully represent the complete suite of visually identified microplastics. In addition, particles smaller than 250 μm could not be analyzed, which may have biased polymer representation and highlights the need for future studies to incorporate improved recovery and analytical methods.
Percentage composition of polymers identified from a subset of 30 microplastic particles.

The dominance of fibers and the identification of polypropylene (PP) as the primary polymer type (most of the fiber and film pieces analyzed) underscores the likely contribution of fishing gear, packaging materials and synthetic textiles (Mishra et al., Reference Mishra, Rath and Das2019; Sharma et al., Reference Sharma, Dhanker, Bhawna, Raza and Sharma2024). One challenge highlighted is the lack of a code of conduct for fishers inside the lagoon who commonly directly dispose of waste lines and nets at sea (S Alefaio pers. obs.). While similar observations have been made in the Pacific Islands (e.g. Drova et al., Reference Drova, Tuqiri, Prasad, Rosi, Gadekilakeba, Bai, Tunaka, Williams, Varea, Brown and Dehm2025) and across other regions (Jonathan et al. Reference Jonathan, Sujitha, Rodriguez-Gonzalez, Villegas, Hernández-Camacho and Sarkar2021), the authors would like to point out another potential key source: white woven plastic bulk bags used as sand bags to act as retaining walls and breakwater along many inundated strips of coastlines, as well as the potential use of geofabric along key reclamation areas as part of the Tuvalu Coastal Adaptation Project.
Importantly, while the frequency occurrence (37%) fell below the global average of 49% identified by (Wootton et al., Reference Wootton, Reis-Santos and Gillanders2021b), this value is surprisingly higher than the median value that the same review identified for Oceania. Tuvalu is one of the smallest countries in the world by population and land area, meaning one would reasonably expect lower levels of microplastics in the marine environment on a global scale. The frequency also exceeds that found in fish sampled previously from markets in Fiji (Wootton et al., Reference Wootton, Ferreira, Reis-Santos and Gillanders2021a); a significantly larger Pacific Island country. However, in terms of plastic load (on average < 1 particle per fish), the values are low when compared to other regions and studies (Wootton et al., Reference Wootton, Reis-Santos and Gillanders2021b; Zazouli et al., Reference Zazouli, Nejati, Hashempour, Dehbandi, Nam and Fakhri2022). While Tuvalu does have challenges with solid waste management (Filho et al., Reference Filho, Havea, Balogun, Boenecke, Maharaj, Ha’apio and Hemstock2019), there have been several externally supported initiatives that generate assistance to improve the whole management of solid waste on Funafuti through the Government, Kaupule (town council) and local communities, which may partially explain the low contamination loads (SPREP, 2016; Sagapolutele et al., Reference Sagapolutele, Wander and Prince2019). Other reasons for the relatively low concentrations may be explained by the very small amount of land, the low population size, the 2020 ban on single-use plastic, or the small size of many of the fish that were sampled.
This initial broad-scale screening study confirms for the first time the presence of microplastics in the gut contents of culturally and nutritionally important coastal food fish from the Funafuti lagoon in Tuvalu. While preliminary, the dataset is crucial for Tuvalu and the wider Pacific Islands, providing a foundation for further research in Tuvalu and enabling broader comparative analyses across the region. Our findings contribute to growing evidence of the omnipresence of microplastics across Pacific Island countries, despite their geographic isolation and relatively small populations. Some plastic debris may have originated from commercial fishing vessels operating in Tuvalu’s waters (Richardson et al., Reference Richardson, Haynes, Talouli and Donoghue2017) which anchor within the lagoon for transshipping. Additional inputs could arrive via ocean currents. During recent beach clean-up campaigns, large volumes of plastic bottles were collected from the ocean-side of Fongafale, but not the lagoon side, suggesting many plastics may be arriving from external sources (S Alefaio pers. obs. 2025). However, the samples presented in this study were collected within the lagoon with limited flushing and exposure to broader oceanic processes, and given the vast distances to neighboring islands (e.g. >800 km to Wallis and Futuna; >1,000 km to Fiji) and the significantly higher levels of microplastics close to the most populous islet Fongafale, it is likely that much of the plastic found within the lagoon originates locally.
We acknowledge that the dataset presented, while valuable in providing the first documentation of microplastics in culturally relevant fishes, is limited in its capacity for statistical inference or source attribution. Future research should prioritize beach and port litter audits to identify major contributors of plastic waste and to trace potential sources. We also recommend expanding this baseline to include a broader range of species (recognizing that nearshore fish are not the sole dietary contributors), seasonal variations and additional atolls and islets across Tuvalu. Standardized sampling with higher replication in key species (e.g. emperors, groupers, surgeonfishes) would enable more robust comparisons across species, functional groups, fish sizes and locations. We would also like to see similar studies expand to the outer islands of Tuvalu to verify the distribution of these waste impacts on key target food fishes in other atolls. Furthermore, whilst process control samples showed no plastic-like particles, indicating no detectable laboratory contamination, the potential influence of background contamination is proportionately more important when sample and particle counts are low, highlighting the need for cautious interpretation of single samples with low microplastic counts. Finally, future work should be designed to understand seasonal variability, and could expand to assessing microplastics <125 μm, allowing smaller microplastics and nanoplastics to be evaluated.
Conclusion
This study serves as an essential foundation for microplastics research in Tuvalu; a region where such data have been sparse. While the study is limited to the most populous atoll in Tuvalu, the findings highlight the pervasive nature of plastic pollution, even in some of the most isolated marine environments, and have global relevance as they provide critical evidence that microplastics are infiltrating food webs far from major industrial sources. Furthermore, as small island nations like Tuvalu rely heavily on reef fish for subsistence, economic livelihoods and cultural practices, these results underscore the urgent need for international collaboration on plastic waste management and ocean conservation. By quantifying the occurrence, form types and polymer composition of microplastics in fish species, this research contributes valuable baseline information for future investigations. By making this dataset available, these findings not only fill a critical knowledge gap but also provide a framework for expanding microplastic studies in Tuvalu as well as in the Pacific Island region. Establishing monitoring programs and adopting standardized methodologies, such as FTIR analysis, could enable a more detailed understanding of pollution trends and inform local and regional policies. This dataset also lays the groundwork for cross-disciplinary collaborations to assess the broader implications of microplastic pollution, including its socio-economic and ecological dimensions.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/plc.2026.10043.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/plc.2026.10043.
Data availability statement
All data is provided within this manuscript, and the fish data is published as an occurrence dataset on GBIF: https://doi.org/10.15468/4jedd8
Acknowledgements
The authors would like to express their gratitude to the Funafuti Kaupule for permitting us to conduct this research. We would also like to thank staff from the Department of Waste, Department of Environment and the Tuvalu Fisheries Authority, as well as community leaders who attended our initial workshop and shared their insights prior to us conducting our research. We would also like to acknowledge and thank the communities on Fongafale, Papaelise and Funafala for their cooperation and permitting us to collect samples from fish.
Author contribution
Semese Alefaio: Investigation, Methodology, Resources, Writing – review and editing; Lotokufaki Paka Kaitu: Investigation, Resources, Writing – review and editing; Lavata Nivaga: Investigation, Writing – review and editing; Paeniu Lopati: Investigation, Writing – review and editing; Filipo Makolo: Investigation, Writing – review and editing; Brian Stockwell: Investigation, Writing – review and editing; Daanish Ali: Investigation; Writing – review and editing; Eseta Drova: Investigation, Writing – review and editing; Jasha Dehm: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Visualization, Writing – original draft, Writing – review and editing; Amanda Ford: Conceptualization, Funding acquisition, Methodology, Project administration, Writing – original draft, Writing – review and editing.
Financial support
This study was funded by the Asia Pacific Network for Global Change Research (grant number CRRP2022-05MY-Ford) under the project “Establishing Baselines for Marine Plastics and Bridging Indigenous Knowledge with Ocean Policy to Improve Livelihood Security in the Pacific.”
Competing interests
The authors declare none.






Comments
Dear Editor-in-Chief,
I am pleased to submit our manuscript entitled “<b>First Evidence of Microplastic Contamination in Fish from the Remote Atoll Nation of Tuvalu</b>” for consideration as a Case Study article in Cambridge Prisms: Plastics. This study presents the first baseline assessment of microplastic contamination in reef fish from Tuvalu; a Pacific Island nation where data on plastic pollution and its ecological impacts are currently limited.
Given the ecological and socio-economic importance of coastal fisheries to small island states like Tuvalu, understanding the extent of microplastic contamination in reef-associated fish is vital. Our study examined the gastrointestinal tracts of 201 reef fish spanning 44 species and detected microplastics in 37% of individuals, with a mean abundance of 0.72 ± 1.16 particles per fish. The majority of microplastics were fibres, with polypropylene being the dominant polymer identified. These results provide critical baseline information on microplastic occurrence within Tuvalu’s lagoon ecosystem and highlight the potential exposure risks for reef fish populations and the communities dependent on them.
We believe this manuscript will be of interest to readers of this journal given that it fills a geographical gap in global microplastics research, providing the first evidence from this remote Pacific atoll nation and highlighting the ubiquitous nature of plastic pollution. The findings contribute to the broader understanding of microplastic dynamics in tropical coastal systems and provide a foundation for future research and management actions.
This manuscript has not been published nor is it under consideration elsewhere. All authors have approved the submission and have no conflicts of interest to declare. We thank you for considering our work and look forward to your review.
Sincerely,
Amanda Ford, on behalf of all authors