Introduction
Clinical trials are vital to advancements in the management of cancer and improved patient outcomes. Successful completion of clinical trials leads to faster development of new treatments, and ultimately improved survival rates with overall decreases in mortality; however, this has been hampered by low accrual rates [Reference Murthy, Krumholz and Gross1,Reference Friedman and Cain2]. A thorough understanding of the complexities associated with patient willingness and hesitation to participation in clinical trials is essential for developing robust strategies to improve engagement and participation of patients in clinical trials [Reference Hamm, Cavallo-Medved and Moudgil3–Reference Unger, Cook, Tai and Bleyer6].
Despite studies showing that half of patients would be willing to participate in clinical trials, this does not directly translate to accrual rates [Reference Unger, Hershman and Till7]. Accrual rates are far lower than reported willingness to participate ranging from 2–8% with higher participation at academic centers over community centers [Reference Unger, Hershman and Till7,Reference DasMahapatra, Raja, Gilbert and Wicks8]. Nearly half of patients that may have indicated willingness to participate are lost to follow up and do not participate highlighting additional systemic barriers that need to be addressed [Reference Unger, Hershman and Till7]. Participation is also intrinsically linked to perceived risk and potential benefits, with higher perceived benefits increasing trial participation [Reference King, Tan, Tan, Lai, Bujang and Voon9]. This points to the importance of awareness of the scientific research process and better trust and communication during the accrual process [Reference Brody, Dalen, Annett, Scherer and Turner10–Reference Yadav, Todd and Patel12].
Despite low accrual rates, patients report positive attitudes toward being approached about participating in clinical trials [Reference DasMahapatra, Raja, Gilbert and Wicks8,Reference Moorcraft, Marriott and Peckitt13]. However, factors such as low socioeconomic status, and the historical experiences of different ethnic backgrounds remain key barriers to patient accrual, with lower socioeconomic status associated with decreased willingness to participate in clinical trials [Reference Meng, McLaughlin, Pariera and Murphy14–Reference Sharrocks, Spicer, Camidge and Papa19]. Patients also cite lack of knowledge or general discomfort with the research and clinical process, fear of randomization as well as anxiety about the clinical trial process as impeding participation [Reference Unger, Vaidya, Hershman, Minasian and Fleury5,Reference Unger, Cook, Tai and Bleyer6,Reference Mills, Seely and Rachlis17,Reference Ginossar, Diaz Fuentes and Oetzel20]. Strategies to improve research literacy would aid in increasing interest and enrollment in clinical trials and alleviate fears and anxiety associated with the process [Reference Brody, Dalen, Annett, Scherer and Turner10–Reference Yadav, Todd and Patel12]. Among these efforts, few studies have assessed the impact of an interactive educational approach.
The purpose of this study was to gain a more thorough understanding of the factors influencing patient willingness and hesitation to participate in clinical trials and identify mechanisms to address these concerns and improve accrual rates. We provided patients with both written resources as well as hands-on engagement in research followed by a group discussion on the motivations and barriers to participating in clinical trials and identified key thematic areas to improve clinical trials accrual. This work adds to the expanding body of knowledge enforcing the need for a patient-centered approach to improve the overall quality and rigor of clinical research while elevating patient experience.
Methods
Participants
Ethics approval from the University of Windsor Research Ethics Board (REB# 23-024) was obtained. Interest in participating in a half-day, in person “behind the scenes” research experience on clinical trials in cancer research was added to a survey circulated for a separate study focused on attitudes toward science and clinical trials. The survey was widely distributed through social media accounts that targeted the local community as well as cancer patient support groups. Of the 81 participants that completed the initial survey, 9 consented to this part of the study, all of whom had lived experience with a cancer diagnosis. Of the 9 individuals; 8 identified as women and 1 identified as a man. All 9 participants also consented to participate in a 1-hour focus group following the research experience and lab tours.
Procedure
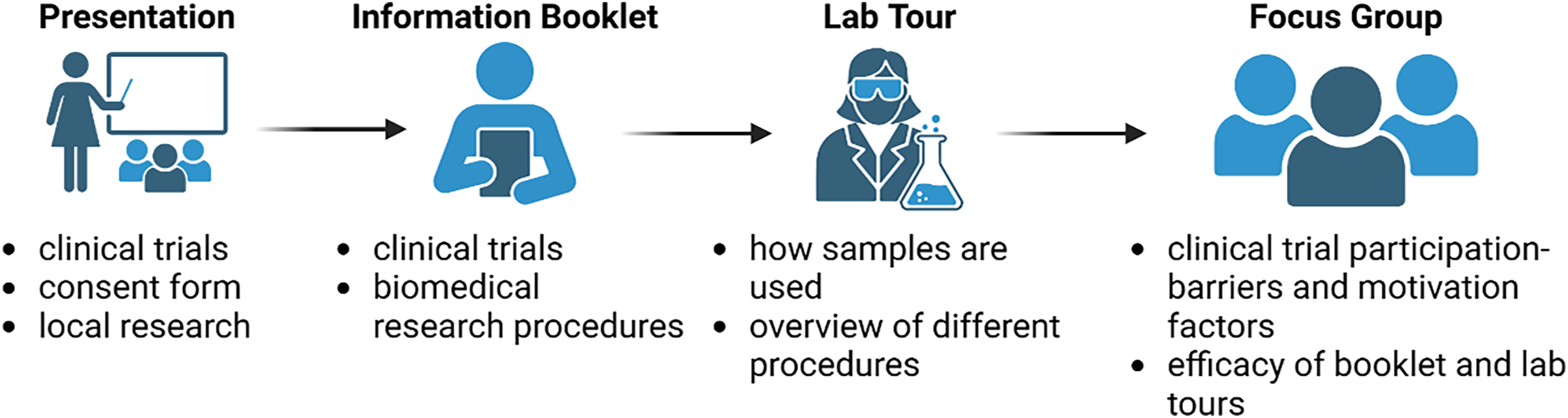
This study involved four components that took place during a one-day research experience and lab tour event in March 2023: (a) a presentation by local cancer researchers on clinical trials and research with a focus on local procedures in Windsor-Essex County, (b) distribution of a research information booklet regarding clinical trials and related research lab activities and procedures, (c) rotating research lab tour/demonstrations and (d) an hour-long focus group (Figure 1; Supplemental File 1). Participants were first provided with an overview of clinical trials and local biomedical research, followed by a tour of research labs highlighting techniques and tools used in cancer clinical trials by biomedical researchers (Figure 1). Following this, participants completed a focus group where they reflected on their experience of the day and group discussions surrounding clinical trials. The presentations featured an introduction to clinical trials and cancer research, including materials that cancer patients are given in recruitment efforts for clinical trials at Windsor Regional Hospital. An informational research handbook, “Bridging the Gap,” was created by the research team to highlight research concepts covered in the event. The research lab tour involved discussion of local research projects ongoing at the University of Windsor in the Department of Biomedical Science.
Overview of study design and components of research experience and focus group.

Those who chose to participate in the hour-long focus group were asked to discuss and provide feedback on the materials presented, the content and appropriateness for different learners of the research lab event and on clinical trials and share their perspectives on improving information for potential participants and increasing local interest (Supplemental File 1). Focus groups were moderated by two social science researchers with expertise in focus group delivery, and in the analysis of conversational data within health care. The focus group was recorded by Microsoft Teams. Transcripts were autogenerated, reviewed, corrected and de-identified by research assistants. Transcripts were reviewed by a second member of the research team, a research associate, for accuracy and used for documentation allowing for ease of cross-checking and referencing of the data.
Data analysis approach
Braun and Clarke’s 6-phased reflexive thematic analysis framework was the main strategy used to analyze the focus group data [Reference Braun and Clarke21–Reference Braun and Clarke23]. This involves familiarization with the data, generating initial codes, collating the codes into formed themes, reviewing and revising themes and defining and naming themes. Analysis was conducted in an inductive (bottom-up) manner where themes were driven by what was found in the data rather than by previous ideas or theoretical concepts.
The focus group was first transcribed into a Word document and the entire transcript was read and re-read by a research team member and a secondary coder who conducted the analysis. In phase 1, general ideas and observations were flagged and discussed, and team members listened to the recording for additional context. Initial reactions were discussed between the two coders and general ideas were brought back to the entire team for reflection. In phase 2, transcripts were coded line-by-line with information relevant to the research question coded. Needs, barriers, and hesitancies, as well as motivators and facilitators of clinical trial participation were recorded on the left side of the transcript by each coder independently; however both coders met weekly to discuss their developing codes.
In phases 3 and 4, recurring codes that reflected the same concept were grouped together to determine larger organizing themes. These themes were defined and labeled by each team member separately. Similarities and differences between the two team members were discussed and inconsistencies were resolved through discussion. Phases 5 and 6 included selecting appropriate quotes to contextualize the main findings and link them back to the literature. As two researchers coded the transcripts independently, they meticulously documented their own analytical decisions, interpretations, and reactions, which were discussed during meetings and represented an audit trail. As the coding team was comprised of researchers heavily involved in cancer research, these steps ensured the rigor of the analysis and that it remained grounded in the data and not extrapolated beyond participants’ experiences.
Results
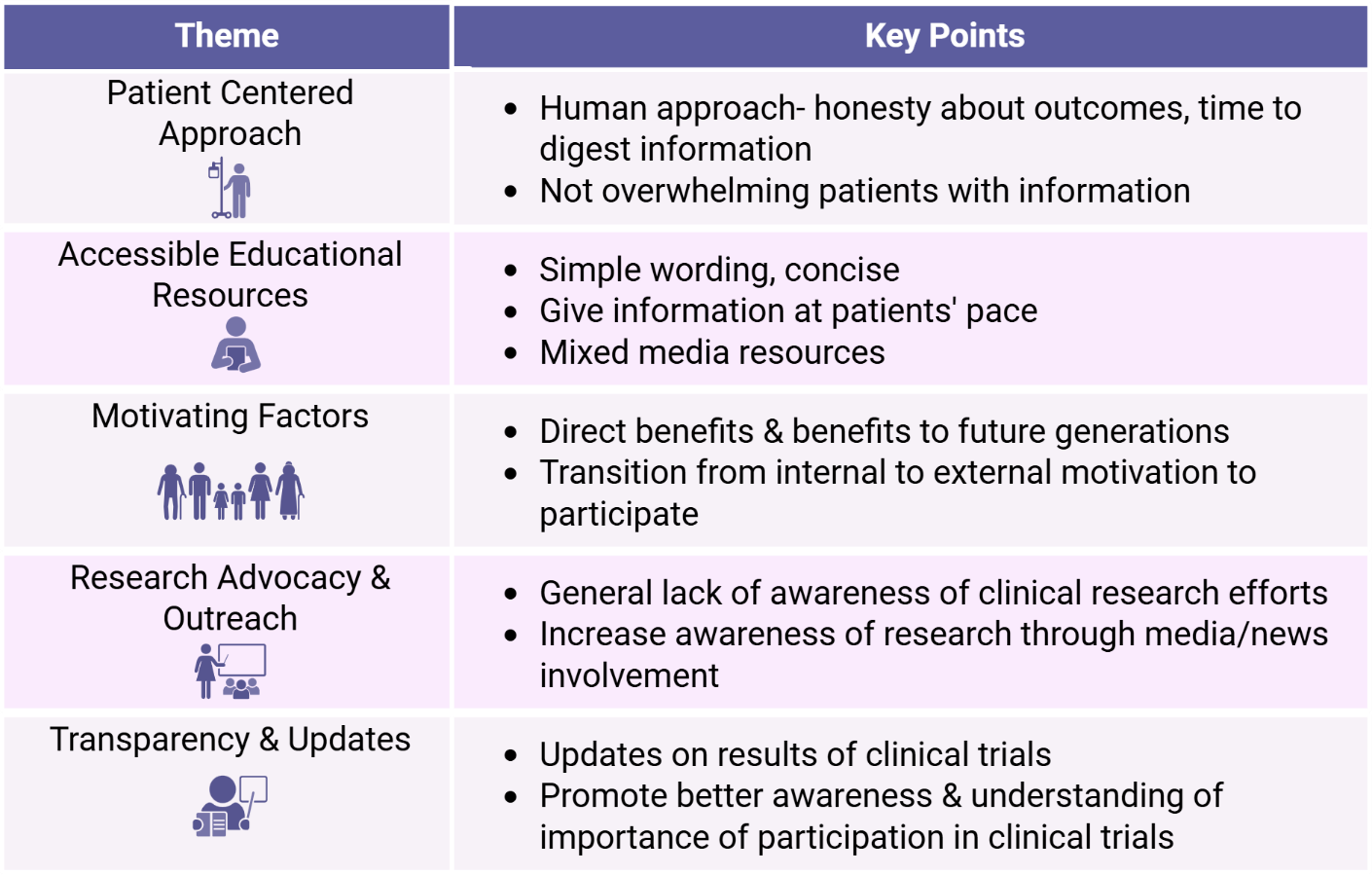
Five major areas of importance or need were identified as a theme from the focus group participants’ discussions surrounding clinical trials: (a) having a clear patient-centered approach, (b) the availability of digestible educational resources, (c) making a continual effort to understand patient’s motivation or hesitations, (d) local research advocacy and outreach, and (e) transparency and regular, ongoing updates for patients (Figure 2).
Overview of recurring themes and key points within each theme.
Theme 1: Importance of a patient centered approach
(“If you told me all of this at the beginning, I mean, I wouldn’t be there”)
Participants clearly stated that “it depends on who’s making the approach,” concerning the initial conversation when healthcare providers ask a patient if they would be interested in participating in a clinical trial. Participants stated they preferred the idea of being approached by someone that knew them, stating “if it’s someone internal [on the patient’s healthcare team]… you already have the relationship […] they’re already good at that,” in reference to understanding the patient’s needs. While flexibility in the approach is required for different patients as “people respond slightly differently,” it was mentioned that healthcare providers are “not without challenge,” as the same patient could react unpredictably and “the person that’s approaching would need to be able to have some insight into how that person is reacting and change their approach and yet still cover all the details.”
The approach is an initial event that can create lasting impressions for the patients [Reference A.Gregersen, Birkelund, Wolderslund, Dahl Steffensen and Ammentorp24]. Not only was the identity of the person who approached important, but their affect also, as “if they’re not exactly warm friendly people,” a participant said that ‘has a bit of an impact as well.’ They appreciated having “emotional support and that kind of person to guide you through.”
Participants also mentioned that “you have to have a different message,” when “you’re talking to somebody that’s 70 compared to somebody that’s 25.” They stated that “there [couldn’t] be a standard sort of approach,” in developing a way to ask patients to join a trial. When patients are broken down into age groups or by life priorities, it means “[healthcare workers have] focused on that particular group and where they’re coming from and what their expectations would be.” Data gathered showed a more personalized approach was favored over a generalized approach that didn’t consider their concerns and hesitations.
It was stated among multiple participants that healthcare professionals should be able to understand their patients and how much information they would be able to handle at any stage of their diagnosis and treatment. One participant discussed their outlook on receiving large amounts of information about their diagnosis and feeling overwhelmed:
Initially, it’s such a shock that I think coming at you with a clinical trial would be… you’re so overwhelmed in the beginning that you just need to settle. You find out you have so much to learn just at diagnosis… so that chat would be overwhelming for sure.
Participants collectively agreed that although patients want as much information about the condition as they can after diagnosis, everyone has an individual limit as to how much information they can handle before becoming overwhelmed. Participants stated they would appreciate “just being able to take the information with [them], home with [them], before [they] make a decision, cause then [they] have the time and being to not disagree with [joining a clinical trial].” It was agreed across all participants that a patient overwhelmed with too much information during their diagnosis and subsequent approach about a clinical trial would be less likely to process the information and less likely to participate.
Participants stated that healthcare professionals should also understand their patients’ present situation as they are “already in an overwhelming position.” Participants spoke about having “so much going on, and with trials and everything like that, so much on your mind and brain.” Specifically, someone brought up that they thought “wait times are a hard time,” as patients are waiting on chemotherapy, results or surgery and are in a highly stressed situation. As another participant said, healthcare professionals “have to be very compassionate, have a lot of empathy [and] know how to talk to you about what you’re going through.” It was described that “when you’re first diagnosed, you’re very self-focused;” the first part of receiving a diagnosis is “all about me,” and the “next piece is that it’d be wonderful to help the future.”
Participants also wanted to know that “the person that’s engaging [them] with that consent, they can answer all those questions right there.” As one participant described:
I found that several people that I talked to at that time were like really, I learned so much from them… and that’s what I would want, someone that really is professional, knows what they’re talking about, and is involved somehow in that research […] I want someone that really understands what it’s about to be talking, so no matter what question I asked them, they can answer more.
Theme 2: Need for digestible and accessible educational resources
(“Who do you talk to? The person that you’re dealing with in front of you? No, probably not”)
When discussing the consent forms and informational resources, participants were adamant about the need for clear vocabulary and explanations of unfamiliar terms. As one participant recalls: “I was reading through, you know, what they tell patients… that was excellent.” They mentioned that “whatever the words… they were simple and easy to understand and you can see the concept of how this is going to help me today and that is also going to be something for the future.” Others stated they did have some problems with the vocabulary and suggested the idea of “almost like a glossary,” or “a definition of terms,” to catch those who might have trouble comprehending.
Summarizing and sticking to main concepts were stated to be important when referencing the example clinical trial consent form that was read prior to the lab tour. A participant requested a summary of the main topics, focusing on “What? Why? When? Not now?” stating that including this would have helped with their understanding. There was an expectation that other healthcare professionals at the cancer center would have some knowledge of the ongoing research and trials. Participants thought “maybe the nurses have to be aware and up to date with what’s going on as well.” Participants expressed that they would appreciate being able to have more than just their doctor to talk to or ask questions.
A clear explanation of the potential risks and additional time required for participation was discussed as an important component of informed decision making and helping to alleviate any concerns and should be explained clearly. Participants stated that they “would want to know […] what [their ‘toxicity’] could be,” discussing the idea of “going to chemotherapy,” and how “they would tell you want your, what could happen to you years down the road because you took this chemo.” As one participant described, context of the clinical trial mattered:
I think sometimes people worry about the risk involved because you’re just giving your tissue… here it’s a donation and its not a risk to you or something […] if your clinical trial is for drugs or something then I think there’s more risk and that would be explained.
“How much time might be involved,” and the burden that they could place on caregivers was another concern. Participants worried about whether joining a trial would “involve more events or prodding and poking,” if it meant a sacrifice of “more of your own personal time,” in exchange for “more interaction with doctors and stuff like that,” and “extra meetings or time commitments.” As they discussed, “wait time are a hard time,” and most patients said they were in the mindset of “get this over with.” One participant described their experience with caregiver burden that resonated with the rest of the group:
So I found that with my grandma, she didn’t want to do like anything… she didn’t want to feel like a burden… like for the rest of my family […] you feel like taking that time from other people.
A large focus of discussion was on the difference between needing informational and emotional support to decide if participating in clinical trials was appropriate for them. While being able to develop proper informational resources for patients was stated to be helpful for knowledge gain, participants stated they would also want to know there was emotional support offered throughout the process as well. From the first approach, “it would be more positive,” if a healthcare professional were to approach someone and “they had their support person with them… like as opposed to being individual.” It then moved to speaking about the process, and how helpful it would be to have “somebody that you could talk to in that emotional way, an empathetic sort of situation… which is very important,” and someone to help “get away from the clinical dah-dah-dah.”
The idea of having a contact person for emotional support that had previously participated in a clinical trial was a recurring theme. Another participant acknowledged that this program would mean that patients “would have people essentially who have experience that they could talk to on an individual basis, confidentially,” and that though the “people would be chosen very carefully,” the concept had “seemed to be a successful approach,” for other groups in the past. Participants stated they would appreciate the support of being able to speak to someone who had previously had a good experience and could provide insight and some emotional guidance on the topic of participating in a clinical trial. A participant mentioned wanting “someone who can help navigate you know, like, the process, and they can connect you to like other services and resources,” when it came to understanding clinical trials and the options available.
Theme 3: Understanding patient’s motivating factors
(“I understand the power of research. Why should we not want to [participate]”)
The biggest factor discussed regarding motivation for participating in a clinical trial was the possible benefits to future patients with similar conditions. Participants stated that “its good and you will benefit someone,” and that when thinking “this [could] help someone down the road,” they found themselves “[thinking] of future generations.” It gave them hope that there could be “better outcomes for [the future patients].” One participant told their story of hearing about their participation in a clinical trial being used in future studies:
I had genetic testing and I didn’t have the gene… and then the London Science Center called me and said we’ve got more research – this is like 7 years later […] [they said] ‘we still have your tissue…’ they said “we”ve got the gold standard now’ or something they said “and do you mind, can we [use the patients stored tissue]? And I said absolutely […] absolutely it’s a go for it.
Another factor that was identified was that the results of the trial could help “advance research and benefit future generations through [patient participation].” Participants discussed the importance of understanding past research and how a patient could contribute to this process. One described it as “[being] thankful for the progress that has been made, and that [they’ve] benefitted from that.” Another participant brought up understanding “that there has been a lot of progress made [and that] even the state represented that […] we need to do more and contribute to the progress.” The idea of being a piece in the puzzle of a powerful research project had participants realizing that they “guess [they] are important.”
Framing the situation as potentially benefitting one’s own community was also discussed. As one participant described it, being a “part of a community research project that you yourself are important as a piece in this community effort… which then shows the macro as opposed to the micro.” The concept of helping a community that a patient identified with provided “a different perspective that people might like… ‘Oh okay, I’m doing something for other people too.” Participants thought that although it’s a difficult time, that “it’s exciting, to participate in a community effort,” and know that what they were contributing could possibly directly benefit those around them.
It was mentioned that throughout a patient’s diagnosis and treatment, participants felt that their motivation would change from internal (worry if trial can help them personally) to external (wonder if trial will help others). They acknowledged that for most patients it is likely that the motivation to help others “will come very quicky, but it’s not there,” right off the bat. As one participant described:
As you progress with it or you come through [the cancer diagnosis], then you [start] thinking about how [you] wanna make things better for future generations… [you] want to make things better for further research. Your motivation changes a bit… a lot actually.
There is also the idea that an individual’s life experiences or particular diagnosis could influence their motivation. As one participant described age-related differences:
I think when you’re 25, you’re like I want to live the rest of my life, so what can I do? And I think the older you get, the more you’re kind of like, you think for the future generations… you think more of that right, like your kids.
While another participant described the impact of their prognosis:
I think that, when I think of people that were faced with [a diagnosis] that maybe wasn’t ordinary, like a very challenging type of cancer, they would want to be in a clinical trial because it might give them that extra hope.
Theme 4: Spotlighting local research advocacy or outreach
(“Sometimes you just don’t think that’s going on in your own community here”)
Participants reflected on the experience they shared of getting to tour a biomedical sciences research lab. They mentioned getting “the sense that there’s so much going on,” and “the sense of hope.” Seeing students conducting research, they “thought ‘wow somethings got to come of this…’ this is how [they] make things happen.” Seeing the research happen made them realize that they “[couldn’t] really know the all of it, and [that they] think progress is going to come and it may come fast too, because there’s just so much going on.” Another participant agreed that “a lot of these processes, a lot of what’s going on…it makes progress.” There was overwhelming acknowledgement of previously being unaware of this research and how seeing it was “this really big thing to consider about your university and what’s building.”
In reflection of their exposure to local research, participants discussed the lack of awareness about local research. As they described, “a lot of people don’t know the kind of research that is going on in this university.” Another participant mentioned that “there hasn’t always been much relationship between results and advocacy,” in Windsor specifically. They stated that the University should focus on “the outreach part […] [including having] plans to have somebody to present to,” and that “it would be extremely good on the basis that there is something there, like you said before, there’s hope, something going on… this is who we are, this is what we’re doing.”
The participants brought up a need for more outreach to media and local news, stating that they should get “more media exposure,” as well as “every aspect involved – visuals, media, whatever.” As one participant discussed,
I think if you somehow just highlight it more, wherever you can, and advocate for it so the people know how important, and like you say, like the progress you’ve made is just wow
Another compared it to “seeing things on the news,” about another department at the university where “they go in there and they do a little thing… ‘oh what’s going on…’ “we”re developing this’.” The general consensus was that “some of [the clinical trial-based research] gets translated in the media, but not very often.” One participant brought up the idea of promotional material from other foundations where “they’ll say have a picture of someone and it says this is what this research helped me with and I’m alive today because of all this research that’s being done.”
Theme 5: Transparency and updates for patients
(“You are keeping up with the research… you’re a part of it… you are a part of that journey”)
One of the most frequently recurring themes throughout discussion was that participants wanted transparency about the study and continuity created through updates. A participant described that they “would want to be informed if [they’re] going to do that, like let me know where [they’re] at in your research.” They thought that “just a little blurb or something […] would reinforce [their] commitment,” talking about “updates, maybe […] what’s going on, a little update.” The idea of “having this ongoing conversation [where participants were] being informed […], having access to information and keeping the conversation with the researchers going and you know, being a part of that,” was strongly admired. Participants strongly believed that patients would want to be kept up-to-date about the research they contributed to: “the next step [the researchers would take when discovering new information in a research project] should be to inform [the patients participating].”
Participants discussed hesitancy surrounding how data and results were used, reporting “wanting to be sure where the data is going and how personal information is being used,” further highlighting the importance of transparency. Others were less concerned, citing that “you’ve given the permission,” and “you know they have access to your whole records.” There was consensus though that “the only issue there would be reassurance that the additional tissue [in reference to additional biopsies being performed to extract tissue for research] didn’t reflect that we didn’t get the right care in the first place.” Participants also believed there was uncertainty in understanding the possible outcomes for a specific trial, stating that its “the fact that it may or may not benefit them, that’s what its all about.” There was also “fear when you hear clinical trials,” as “why would I get in this group that’s not getting it,” in reference to a clinical trial that would trial a new drug or treatment with a placebo group. Regardless of these uncertainties, participants understood that no matter the form of participation, “its good and you will benefit someone…. I guess you’re important.”
The group also generally believed that patients want a breakdown of the clinical research barrier and a better understanding of what they’re participating in. As one participant said: “I think that would also help with participation in the future with other cancer areas.” Participants also supported patients getting to witness and feel better connected to the research they were taking part in. They suggested research labs taking the initiative to say “if [patients] are participating in a research project, come on in, we’re going to show you what we’re doing.” One participant phrased it as, “if you were participating, wouldn’t you come?.” They believed that seeing the research itself “might be more effective,” in gaining patient’s understanding of their contributions, motivation, and how they would speak to others about the experience, as “then it’s not just one of the things [patients] are doing.” Another participant suggested the implementation of “something like exactly what you did today,” in referring to allowing patients to take a tour of the research lab using patient-donated samples and that it could lead to a cascade of information as “people tell other people and it [could] become bring somebody with you… your family or anyone else that’s interested.”
Discussion
The decision to participate in a cancer clinical trial is complex. Participants in this study indicated that increased awareness, appropriate educational resources, transparency, and a patient centered approach that includes a personalized understanding of needs, hesitancies and fears are critical in decreasing barriers and increasing willingness to participate in clinical trials. This work supports that taking these key themes into consideration would lead to increased accrual rates. One specific outcome of these findings is the strong positive reinforcement that developing a framework for knowledge translation, that includes a range of easily digestible information and direct exposure to research has the potential to increase patient accrual rates for cancer clinical trials.
A common theme that was identified in this study and previous work, is that patients often decide to participate in clinical trials not only to benefit themselves, but for altruistic reasons with a desire to make a difference for future generations that may be impacted by their disease [Reference Moorcraft, Marriott and Peckitt13,Reference A.Gregersen, Birkelund, Wolderslund, Dahl Steffensen and Ammentorp24–Reference McAdams and Guo26]. Participants stated that motivations for participating in a clinical trial may change with age or circumstance. It was stated that older age groups may be more likely to participate for altruistic reasons whereas younger age groups may be more likely to consider personal benefits. This discussion is in line with previous studies [Reference Moorcraft, Marriott and Peckitt13,Reference A.Gregersen, Birkelund, Wolderslund, Dahl Steffensen and Ammentorp24].
While potential risks and benefits of clinical trial participation play a significant role in the decision-making process, how the patient is approached and asked about participation is equally important. Previous work has found that patients are guided by their emotions and relationships with their physician when making decisions regarding enrollment in clinical trials [Reference Mills, Seely and Rachlis17,Reference A.Gregersen, Birkelund, Wolderslund, Dahl Steffensen and Ammentorp24]. This work further supports these findings with participants stating that the approach is key and can leave a lasting impression. Participants stated a preference for the ask being made by someone they already had a relationship with who would be able to make the approach in a sensitive manner with an understanding of where they are at in their cancer journey. They felt this would help ensure information was delivered appropriately, taking into consideration the needs of the patient. Receiving a cancer diagnosis is a challenging and stressful time and having conversations surrounding more complex decisions regarding potential treatment or procedures with a trusted individual would be valuable in creating more trust in the process.
Participants also highlighted the need for more support and resources while considering participating. Having a trusted support person there when the ask was made was thought to be helpful, but beyond this, providing patients with information on support services during their journey was appreciated. Several participants shared their experiences with having a contact person for emotional support who had been through similar processes and could provide guidance and insight. While this was generally thought to be a positive experience, the need to carefully choose individuals to provide this level of support was highlighted to ensure it would be a positive experience. Beyond emotional support, the need for increased resources to provide information was stated. Information provided can be overwhelming. Participants stated having resources they could bring home to digest at their own pace would be welcomed. This goes hand in hand with hesitancies that were raised regarding how their data would be used and any potential risks associated with participating. Clear, concise informational resources would greatly benefit patients and lead to increased accrual and fully informed consent when choosing to participate in a clinical trial.
A lack of knowledge of the research process, how research decisions are made and how data and results are used are all contributors to reduced participation in clinical trials [Reference Unger, Cook, Tai and Bleyer6,Reference Mills, Seely and Rachlis17]. This study sought to address strategies to improve research knowledge by piloting an informational handbook regarding research processes used in a cancer clinical trial and providing participants with a lab tour of a biomedical research lab. Participants felt the handbook was a good resource to help demystify the research process and the lab tour was an overwhelmingly positive experience for all participants. Many stated they felt more work should be done to increase this type of awareness in the community. Seeing the work being done, and being educated on the progress made to date, clarified the benefits of clinical trials for participants and made them feel more invested and engaged in the process. The focus group agreed that clinical trial participants would gain a sense of purpose from having direct insight into the research that they have contributed to. This is in alignment with previous data that found that patients provided with an opportunity to interact with and see the research process firsthand stated this was a positive experience and they were empowered by the information received [Reference Connor, Hughes and Schäfer27]. Further insight into the types of initiatives and strategies that can be delivered to address this are needed to ensure the needs across all demographics are met without creating any additional barriers to participation.
There was also a desire for better transparency and updates with respect to communication of results of clinical trials, this sentiment has been noted in other studies [Reference Brody, Dalen, Annett, Scherer and Turner10–Reference Yadav, Todd and Patel12]. Participants expressed strong opinions on the importance of communicating results of the study with participants. Previous work has assessed how patients would prefer results to be delivered dependent on whether the results of the study were positive or negative [Reference Moorcraft, Marriott and Peckitt13,Reference Elzinga, Khan and Tang28–Reference Cox, Moghaddam, Bird and Elkan32]. Further work should be done in this area to develop standardized guidelines of addressing this unmet need. Currently, there are inconsistencies in ethical guidelines on how results should be delivered to participants with no specifications on exactly how, when and which projects this may apply to [Reference Darbyshire and Price29,Reference Fernandez, Kodish and Weijer31,Reference MacNeil and Fernandez33]. Further work in this area is critical to ensure results are disseminated according to standardized ethical guidelines that meet the needs of participants in cancer clinical trials.
While several studies have addressed patient identified barriers and hesitancies to cancer clinical trial participation, this study provides a new perspective by also implementing and evaluating the perceived benefits of developing programs aimed at increasing awareness and knowledge of the research process. Despite the small sample size, previously identified barriers and hesitancies towards participating in cancer clinical trials were found. An overarching theme uncovered in this study is the need for increasing awareness and knowledge of the research process with better communication of not only how clinical trials function and use the data but also increased transparency and communication of the results of the trial to participants. Developing new resources and programing that engage, educate and support patients before, during, and after the clinical trial will be key to increasing clinical trial accrual and make the experience positive. This work is also an example of how learning directly from patients can accelerate the development of these needed resources to ensure that accrual rates are increased more effectively and rapidly. Collectively, implementation of the findings from this focus group has the potential to ultimately lead to improved health outcomes for patients diagnosed with cancer for generations to come.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cts.2026.10695.
Acknowledgements
We thank Mr. Alan Cieslukowski, Mr. Philip Habashy, Ms. Emily Mailloux, Mr. Ali Nadi, Mr. Alireza Salimi Chirani, Ms. Jalyn Laliberte, Mr. Milan Lobana and Mr. Christopher Jaworski for their valuable role assisting with the research lab tours, as well as all participants in this study. We thank the WonderBroads for their continued support of local cancer research. We thank WE-SPARK Health Institute for REDCap support. Figures 1 and 2 were created with BioRender.com.
Author contributions
Bre-Anne Fifield: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review & editing; Allison Baker: Data curation, Formal analysis, Investigation, Validation, Visualization, Writing-original draft, Writing-review & editing; Omer Elkhidir: Conceptualization, Data curation, Investigation, Methodology, Writing-review & editing; Nick Philbin: Conceptualization, Data curation, Investigation, Methodology, Writing-review & editing; Isabelle Hinch: Data curation, Investigation, Methodology, Writing-review & editing; Nora McVinnie: Conceptualization, Investigation, Writing-review & editing; Amy Llancari: Investigation, Methodology, Writing-review & editing; Claudia Pecoraro: Investigation, Methodology, Writing-review & editing; Mahsa Rahimi: Investigation, Methodology, Writing-review & editing; Tiana Visconti: Investigation, Methodology, Writing-review & editing; Alexandra Shoust: Conceptualization, Writing-review & editing; Suzanne McMurphy: Conceptualization, Data curation, Investigation, Methodology, Writing-review & editing; Kendall Soucie: Conceptualization, Data curation, Investigation, Methodology, Writing-review & editing; Caroline Hamm: Conceptualization, Methodology, Writing-review & editing; Lisa A. Porter: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Visualization, Writing-original draft, Writing-review & editing.
Funding statement
This research is funded by the Breast Cancer Canada (grant 2022-007).
Competing interests
The authors declare none.