Introduction
For more than 10,000 years, wheat has been essential for food and nutritional security, making it the most widely grown crop worldwide. Globally, this crop accounts for approximately 20% of all protein and calories in the human diet but also provides a significant amount of fibre, minerals, vitamin B, and other micronutrients in the diet (Reynolds and Braun, Reference Reynolds and Braun2022). A set of properties, including healthiness, nutritional value, shelf life, organoleptic qualities, micronutrient concentration, and absence of contaminants, defines the quality and safety of wheat. Many of these properties depend on the production environment, particularly the soil from which plants derive nutrients for their growth. Consequently, elements of the soil that are bioavailable to wheat plants can potentially enter the animal and human food chain (Angon et al., Reference Angon, Islam, Das, Anjum, Poudel and Suchi2024; Benedetti and Mocali, Reference Benedetti and Mocali2009). Some elements, such as Co, Cu, Ni, and Zn, are essential for plant growth but can become phytotoxic at high concentrations. These potentially toxic elements (PTEs) can inhibit growth and affect photosynthesis, respiration, enzymatic activity, cell membranes, and DNA structure when their levels exceed specific thresholds, which vary by element, plant species, and soil characteristics (Ali et al., Reference Ali, Ahmed, Emara, Janjua and Alhafez2019; Shtangeeva, Reference Shtangeeva2020; Thalassinos et al., Reference Thalassinos, Petropoulos, Grammenou and Antoniadis2023). Other elements, such as As, Cd, Pb, V, and methylated forms of Hg, are not essential for plant growth, and their presence, even at very low concentrations, can be toxic to plants and humans. For this reason, they are classified as toxic elements (TEs) (Ali et al., Reference Ali, Ahmed, Emara, Janjua and Alhafez2019). High levels of PTEs and TEs in soil can compromise food safety due to the risk of translocation to edible parts and/or potential bioaccumulation along the food chain. In addition, they can negatively impact food quality and security, leading to phytotoxic effects that reduce crop yields (Legrand et al., Reference Legrand, Turmel, Sauvé and Courchesne2005; Seregin et al., Reference Seregin and Kozhevnikova2025; Takáč et al., Reference Takáč, Szabová, Kozáková, Benková and Takáč2009; Wuana and Okieimen, Reference Wuana and Okieimen2011). Plants have developed defence mechanisms that can limit the uptake and translocation of TEs and PTEs, thus reducing the effects of phytotoxicity. Roots can act as a barrier in the absorption or transport of TEs (Alkorta et al., Reference Alkorta, Hernandez-Allica, Becerril, Amezaga, Albizu and Garbisu2004; Ghuge et al., Reference Ghuge, Nikalje, Kadam, Suprasanna and Hong2023; Kabata-Pendias, Reference Kabata-Pendias2010; Laţo et al., Reference Laţo, Radulov, Berbecea, Lato and Crista2012). Furthermore, plant genotypes influence the absorption, translocation, accumulation, and use of elements in the plant, and this has been demonstrated for both essential and non-essential elements (Carcea et al., Reference Carcea, Aureli and Cubadda2007; Grant et al., Reference Grant, Clarke, Duguid and Chaney2008; Liu et al., 2009, Reference Liu, Liang, Zhang and Zhou2015; Singh et al., Reference Singh, Pratap, Tandon, Mishra, Tandon and Srivastava2020).
Durum wheat (Triticum turgidum L. subsp. durum) can accumulate more Cd than other cereals. Since the accumulation of Cd and Pb in durum wheat constitutes a risk to human health, according to the European Commission, by Regulation N. 2023/915 – repealing Regulation (EC) N. 1881/2006 (2023) established maximum levels of Cd and Pb in cereals, which must not exceed 0.1 and 0.2 mg kg−1, respectively. The presence of sources of pollution of TEs or PTEs and the use of phosphate fertilisers and amendment with Ca, K, and Mg fertilisers can affect the presence and mobility of elements in the soil. For this reason, the reaction of plants to chemical stresses that are caused by deficiencies and excesses of elements must always be investigated by studying the soil-plant system (Kabata-Pendias, Reference Kabata-Pendias2010; Seregin et al., Reference Seregin and Kozhevnikova2025; Thalassinos et al., Reference Thalassinos, Petropoulos, Grammenou and Antoniadis2023). Organic farming involves growing crops without chemical pesticides, herbicides, or fertilisers, primarily using crop rotation, organic fertilisers, and plant-based pesticides to improve soil productivity. The nutritional quality and safety of organically produced foods have been a topic of interest and debate for a long time. There are still few comparative studies on organic versus conventionally produced cereals, and the conclusions of these studies are often inconsistent (Alvarez, Reference Alvarez2021; Vrček et al., Reference Vrček, Čepo, Rašić, Peraica, Žuntar, Bojić, Mendaš and Medić-Šarić2014). In addition, the major and trace element profiles have been used in the literature to differentiate between organically and conventionally produced foods. The findings indicate that organic products, including pears, potatoes, wheat, and sweet corn, contain higher levels of essential mineral nutrients such as Ca, Fe, Mg, Mn, Na, and P compared to conventionally grown products (Magdas et al., Reference Magdas, Dehelean, Feher and Radu2017; Munné-Bosch and Bermejo, Reference Munné-Bosch and Bermejo2024; Popović-Djordjević et al., Reference Popović-Djordjević, Kostić, Rajković, Miljković, Krstić, Caruso, Moghaddam and Brčeski2021).
This research aimed to compare the multielemental profiles of soil (Al, As, Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, Ti, V, Zn) and the uptake and translocation of TEs (As, Cd, Cr, Ni, Pb, Ti) and PTEs (Co, Cu, V, Zn) in three durum wheat varieties grown in organic (OFS) and conventional (CFS) farming systems in field conditions to evaluate whether TEs and PTEs behave differently in the soil-plant system based on the cultivation system. To evaluate the adsorption and translocation of elements from the soil to the roots and from the roots to the aerial part (stems) of the plants, the biological absorption coefficient (BAC) and the translocation factor (TF) were calculated and interpreted.
Materials and methods
Soil and crop sample collection
Soil and crop samples were collected from two different experimental fields of CREA-IT – Research Centre for Engineering and Agro-Food Processing (Rome, Italy) in 2018. The following experimental fields of CREA-IT were considered for sampling: the ‘Inviolatella’ experimental field (41°57′23″N, 12°28′50″E) – organically managed soil (organic farming system – OFS) (Supplementary Material Figure S1a) and the ‘La Castellaccia’ experimental field (42°7′47″N, 12°38′23″E), conventionally managed soil (CFS) (Figure S1b). The straight-line distance between sampling points, calculated using geographic coordinates, was approximately 30 km. During the vegetative cycle of the plants, meteorological conditions, including temperature and precipitation, were monitored, and no significant differences were observed in either field (data not shown). The sampling period, in July, was selected considering its correspondence with the time when durum wheat is usually harvested in Italy. Two different soil preparation methods were applied before sowing. The CFS field was prepared using conventional tillage, which included deep plowing (approximately 30 cm) followed by harrowing to create a fine seedbed. The OFS field was managed under a minimum tillage system, consisting of shallow soil disturbance with a rotary harrow, without prior plowing. At sowing in the CFS field, 31 kg ha−1 of nitrogen and 20 kg ha−1 of phosphorus in the form of urea and diammonium phosphate were applied. Field beans were used in annual crop rotations in both conventional and organic farming systems. No irrigation, at any time of cultivation, was applied in either of the fields. The study focused on three durum wheat varieties: Antalis, Iride, and Simeto (Figure S1c). The collection of entire durum wheat plants and their rhizosphere soil resulted in the creation of individual soil-plant samples. For each variety, samples were collected in triplicate (3 plants with the rhizosphere soil per variety) at different points in the field. Furthermore, for each field, 5 soil samples named ‘bulk soil’ (totalling 10 samples) were collected from the central part of the field, spaced equidistantly from each other, to assess the homogeneity of the field and to carry out the physicochemical characterisation of the soil. A hand drill was used for the sampling, with careful attention paid to removing any soil portions that were in contact with the metal part of the drill. The samples were taken at a depth of 20 cm, and each soil sample weighed approximately 1 kg. Individual soil-plant samples were later separated into various components: rhizosphere soil, roots, stems (culm and leaves), and ears (flowers, spikelets, and grains). The grains were separated from the flowers and spikelets, which, together with the stem, constituted the stem samples (Figure S1d). In total, 12 samples of grains, stems, and roots were collected from various experimental fields, resulting in 36 samples. The collected samples were placed in paper bags and fully air-dried and then dried at room temperature.
Soil and plant sample pretreatment
Laboratory samples were subjected to a pre-treatment procedure to obtain representative portions to be used for the different analyses. Specifically, the pre-treatment procedure aimed to produce a homogeneous and stable fine powder that could then be analysed by Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES) and Inductively Coupled Plasma Mass Spectrometry (ICP-MS) after digestion in a high-pressure microwave system. Care was taken to avoid any contamination from the contact materials used during the pretreatment procedures. For this purpose, all equipment was thoroughly washed with ultrapure H2O (specific resistivity 18 mΩ cm), and anti-contamination contact materials (e.g., ceramic scissors) were used.
Each part was thoroughly rinsed with ultrapure H2O (specific resistivity 18 mΩ cm) and dried in a forced ventilation oven at 40°C for 48 hours. Each grain sample was weighed and subsequently ground using an Analysette 3 PRO_Fritsch mill (equipped with a grinding head for conversion to Vibratory Micro MIII PULVERISETTE). Each stem sample (stems + leaves + flower + spikelet) was weighed and then cut into small pieces with ceramic scissors before homogenisation. Root samples were separated from the plant along with the soil of the rhizosphere, then carefully washed with ultrapure water to remove any remaining soil residues and dried in a forced ventilation oven at 40°C. Each rhizosphere soil sample was meticulously separated from the roots and then crushed in a ceramic mortar to break the aggregates. Each ‘bulk soil’ sample was dried in a forced ventilation oven at 40°C, weighed, and homogenised. Specifically, each sample was passed through a 2 mm mesh sieve. The resulting sample (diameter < 2 mm) was ground in an agate ball mill. The physicochemical characteristics, including pH, soil texture, percentage of skeleton, humidity, pH in H2O and CaCl2, and electrical conductivity, were determined.
Determination of toxic and potentially toxic elements in soils and plants
Sample treatment
For each analysed matrix (grains, stems, roots, and soils), a sample dissolution procedure was developed using a high-pressure microwave system. Details about the mineralisation conditions of the samples, such as reagents, operating conditions, and the type of microwave used to dissolve the samples of the various matrices considered, are provided in Supplementary Material Table S1. The total duration of the digestion cycles was 25 min for the grains and 35 min for stems, roots, and soils. After digestion, the acid mixture was left to cool to room temperature and transferred to reach a final volume of 10 mL with ultrapure H2O. Reagent blanks were also prepared using the same reagents, and process blanks were prepared by applying the same dissolution procedure. The blanks were then subjected to elemental analysis under the same conditions as for the samples.
ICP-MS analysis of grains
TEs (As, Cd, Cr, Ni, Pb, Ti) and PTEs (Co, Cu, V, Zn) in grains were determined by ICP-MS using a Bruker Aurora M90 (90-degree ion mirror-ion optics, Collision Reaction Interface). The method was optimised based on AOAC Official Method 2015.01 (Briscoe, Reference Briscoe2015). The ICP-MS system adjustment was performed preliminarily using a tune solution composed of Ba, Be, Ce, Co, Mg, In, Pb, Th, and Ti at a concentration of 5 μg L−1. To evaluate and correct non-spectroscopic interferences during the optimisation phase, appropriate sample dilution was assessed to ensure that the total dissolved solids (TDS) was <0.2%.
The ICP-MS operating conditions were as follows: RF power 1.40 kW; plasma flow rate 18 L min−1; auxiliary gas flow rate 1.80 L min−1; nebuliser gas flow rate 1.00 L min−1; peak hopping scanning mode; integration time 240 s/sample; and dwell time 10,000–60,000 µs. The measured isotopes included As75, Cd111, Cd114, Co59, Cr52, Cu65, Ni60, Ni58, Ni64, Pb206, Pb207, Pb208, Sb121, Se76, Se78, Sn118, Ti49, V51, Zn64, Zn66 and Zn68.
An external calibration was performed for all elements using 5 solutions of the elements considered at known concentrations, along with the blank solution (ultrapure H2O acidulated with HNO3 1%). Calibration standards were prepared by diluting certified solutions of 1 g L−1 and brought to volume in class A graduated flasks with ultrapure acidified water with 1% HNO3. Each reading consisted of seven repetitions. The stability of the instruments was verified through a system that checks the validity of the calibration line during analysis sequences. To assess background interferences and study the effects of reagents and pretreatment, ultrapure H2O, reactive white, and process white were also examined.
ICP-AES analysis of stems, roots, and soils
The TEs (As, Cd, Cr, Ni, Pb, and Ti) and PTEs (Co, Cu, V, and Zn) were analysed in stems and roots. In soils, the multi-elemental profile (Al, As, Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, Sb, Sn, Ti, V, Zn) was evaluated according to Relić et al. (Reference Relić, Sakan, Anđelković, Popović and Đorđević2019). Analysis was performed with an ICP-AES spectrometer, model VISTA MPX, Varian (Meinhardt type, CCD detector). First, torch alignment and wavelength calibration were performed, reading two wavelength calibration solutions: an Mn standard solution (5 mg/L) and a multielement standard solution (Al, As, Ba, Cd, Co, Cr, Cu, Mn, Mo, Ni, Pb, Se, Sr, and Zn at 5 mg L−1; K at 50 mg L−1), respectively. To establish the method, preliminary tests were conducted that aimed to optimise the analytical conditions based on the signal-to-background ratio (S/B). A study of spectral interference was then carried out to select analytical wavelengths; where spectral interferences were absent, the most intense wavelength was chosen as the analytical one. The ICP-AES operating conditions and parameters for plant parts and soils were as follows: RF power 1.20–1.25 kW; plasma flow rate 15 L min−1; auxiliary gas flow rate 1.50 L min-1; nebuliser gas flow rate 1.10–0.8 L min−1; pump rate 30 rpm; internal stabilisation delay 10 s; and sample uptake delay 35 s. Each reading consisted of five repetitions. Signal stability tests were also performed using replicated measurements of the standard solutions.
Method validation
To evaluate the performance of each method, the following parameters were evaluated for each element: instrumental detection limit, method detection limit, linear dynamic range, and repeatability. Accuracy was determined based on the results obtained from the analysis of reference materials (RMs) analysed alongside the samples, following the same procedure. Specifically, the following RMs were used: NIST SRM 1570a ‘Road elements in Spinach Leaves’ and NIST SRM 1573a ‘Tomato leaves’, which are certified for the content of elements (macroelements, microelements, and trace elements), as well as NIST SRM 1567a ‘Wheat flour’, certified for the content of elements (minor constituents and trace elements). The recovery obtained was around 95% for all elements. The results were expressed in mg kg−1 as the mean of 3 replicate measurements. For all quantified elements, the RSD% ranged from 3% to 5%.
Translocation and biological absorption coefficient
The BAC was the index used to assess the absorption of elements from the soil. It was calculated as the ratio of the element content in the plant (aerial parts or roots) and in the soil. In this study, we calculated the BAC for soil/root transfer, which represents the initial phase of macronutrient and trace element absorption. BAC values are expressed as the mean of the three biological replicates of each variety (Ene et al., Reference Ene, Moraru, Moraru, Pantelica, Gosav and Ceoromila2024). The translocation factor (TF) was the index used to evaluate the transfer of elements from roots to aerial parts. This factor was determined by calculating the ratio of the content of each element in the roots and stems and in the roots and grains (Elbagory et al., Reference Elbagory, Zayed, El-Khateeb, El-Nahrawy, Omara, Mohamed, Elbyaly, El-Sharkawy, Singh, Dzaja, Mioč and Širić2025).
Statistical analysis
Statistical analysis was performed using the MATLAB R2015b software. One-way ANOVA and statistical significance, verified with the post hoc Newman-Keuls test (p < 0.05), were applied to compare the chemical-physical characteristics of the two soils and the multielemental content of the two soil types (conventional and organic). To highlight significant differences in the elemental profiles of roots, stems, and grains among varieties within the same field, a one-way ANOVA test was performed, and when differences between mean values were found, Tukey’s test (p < 0.05) was applied. The Pearson correlation matrix (presented as a colour map) was used to identify possible interactions between these parameters. In addition, a two-way ANOVA was computed to compare the varieties with each other, in addition to the type of soil. Subsequently, the analysis of the principal components (PCA) was carried out.
Results and discussion
Soil chemical-physical characterisation
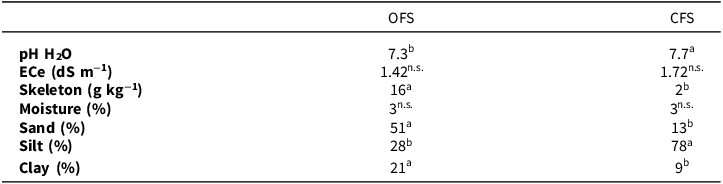
The results of the chemical-physical characterisation of soils show that the soils used for the cultivation of durum wheat in the two experimental fields, OFS and CFS, belong to two different textural classes, loam and silt-loam, respectively, with a significant difference in the fraction of skeleton, probably due to different soil management practices. Soil from OFS was neutral (pH 7.3), while soil from CFS was weakly alkaline (pH 7.7). Electrical conductivity at 25°C was for OFS 1.4 dS m−1 and for CFS 1.7 dS m−1; therefore, both soils were classified as non-saline (Table 1). The USDA soil texture triangle for both soils is shown in Supplementary Material Figure S2.
Main chemical-physical characteristics and texture of soils cultivated with durum wheat under organic (OFS) and conventional (CFS) farming systems

Different letters indicate statistical significance between OFS and CFS (p ≤ 0.05); ns = not significant.
Total contents of toxic and potentially toxic elements in bulk soil and rhizosphere soil
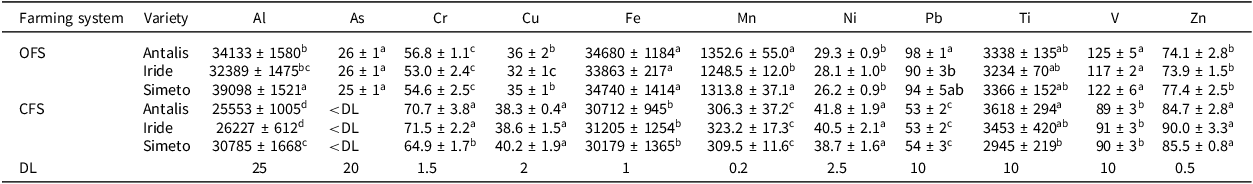
The results of the analysis of the total element content of the soils are reported in Table 2 for rhizosphere soil and in Supplementary Material Table S2 for bulk soils.
Total concentration of elements in rhizosphere soil (mg kg−1 dry matter) in organic (OFS) and conventional (CFS) farming systems

Values represent the average value concentration ± standard deviation of three replicates. Two-way ANOVA and statistical significance (post hoc Newman-Keuls test) were applied to compare the multielemental content of the three varieties in the experimental fields of OFS and CFS. For each element, different letters indicate significant differences at p ≤ 0.05. DL = Detection Limit.
Statistical analysis revealed that the concentrations of elements such as Al, Cr, Fe, Mn, Ni, Pb, V, and Zn were significantly different (p ≤ 0.05) in organic and conventionally managed soils. Among these, a higher content of Fe, Mn, Pb, V, and Zn was observed in organically managed soils, while the content of Cr and Ni was higher in conventionally managed soil. On the contrary, the content of Cu and Ti was negligible in both areas with substantially similar values. In both soils, the average contents of Al and Fe were consistent with those reported in the literature (Angelone and Bini, Reference Angelone, Bini and Adrian1992; Kabata-Pendias, Reference Kabata-Pendias2010) and below the limit values established by current Italian legislation (D.L. 152/2006 and D.M. 46/2019). Furthermore, the Mn content for OFS soil was slightly higher than the values reported in the literature for the Italian average (Angelone and Bini, Reference Angelone, Bini and Adrian1992), while the total content in both soils was lower than the world average and the European average, likely due to the characteristics of each substrate (Kabata-Pendias, Reference Kabata-Pendias2010). The levels of Pb and V in OFS bulk soil exceeded the limits of 100 mg kg−1 and 90 mg kg−1, respectively, imposed by D.M. 46/2019 but are consistent with the world average content (Kabata-Pendias, Reference Kabata-Pendias2010). The occurrence of V is strongly influenced by the type of parent rock, and its range of variation is extensive, particularly in magmatic rocks. The lack of baseline values for the experimental fields made it impossible to determine whether the presence of other elements was due to contamination from human activities.
Levels of Zn were below the limit values imposed by M. 46/2019 (300 mg kg−1) but aligned with the mean values of the Italian background (21 and 89 mg kg−1, respectively) (Angelone and Bini, Reference Angelone, Bini and Adrian1992), while Pb levels were higher than those reported in the literature (Angelone and Bini, Reference Angelone, Bini and Adrian1992; Kabata-Pendias, Reference Kabata-Pendias2010) (Table S2). Cr, Cu, and Ni were consistent with world average levels (Kabata-Pendias, Reference Kabata-Pendias2010) and, in any case, lower than the limits set by current legislation for agricultural area soils – D.M. 46/2019 (150, 200, and 120 mg kg−1, respectively). The total content of As (Table S2) for the soil of OFS is slightly below the limit of 30 mg kg−1 established by D.M. 46/2019 for agricultural area soils but aligned with data reported in the literature for Italian soils and specifically for the soils of the province of Rome, where the presence and concentration of this element are closely related to the volcanic origin of the soils (Quaternary pyroclastic deposits from Plinian-type eruptions) and to hydrothermalism (Chandramohan et al., Reference Chandramohan, da Silva and da Silva2024; Zuzolo et al., Reference Zuzolo, Cicchella, Demetriades, Birke, Albanese, Dinelli, Lima, Valera and De Vivo2020). However, no specific assessment has been made regarding potential sources of contamination of TEs and PTEs near the study areas. The total content of As was lower than the detection limit of the analytical technique (20 mg kg−1) for the soil of CFS (Table S2). Average data reported in the literature for agricultural soils in Italy show a variability in arsenic content ranging from 0.79 to 62.20 mg kg−1, with a median of 7.56 mg kg−1 (Zuzolo et al., Reference Zuzolo, Cicchella, Demetriades, Birke, Albanese, Dinelli, Lima, Valera and De Vivo2020). Cd, Co, Sb, and Sn were consistently below the detection limit of the analytical technique in both soils. Moreover, the elemental content also varied depending on the wheat variety (Table 2). In the OFS field, significant differences were observed for Al and Pb among all rhizosphere soils (p ≤ 0.05). The rhizosphere soil of the Iride variety exhibited significantly lower Cu and Mn contents compared to the rhizosphere soils of the Antalis and Simeto varieties. In the CFS field, the rhizosphere soil of the Simeto variety showed significant differences from the others for Cr and Al, while Ti levels differed among the three rhizosphere soils (Table 2).
Correlation between soil properties and element behaviour in bulk soil
Pearson’s correlation coefficients between total element concentrations and bulk soil properties in the OFS and CFS experimental fields are shown in Figure 1. The correlation matrices obtained from the two experimental fields reveal complex geochemical patterns that are consistent with the processes controlling the retention of trace elements in soils. In both sites (OFS and CFS), As, Cr, Cu, Pb, Ti, V, and Zn form a strongly positive cluster (0.8 ≤ r ≤ 0.9), indicative of a shared control dominated by the reactive surfaces of fine fractions and Fe/Mn oxides. This behaviour is well-documented in pedogeochemical literature, where ferric oxides represent a major sink for metals and metalloids through mechanisms of specific adsorption and co-precipitation (Fendorf et al., Reference Fendorf, Li and Sparks2017; Sparks, Reference Sparks2003; Sposito, Reference Sposito2008; Tipping et al., Reference Tipping, Lofts and Sonke2020; Violante et al., Reference Violante, Cozzolino, Perelomov, Caporale and Pigna2010). The strong correlation between Cr and V (r > 0.9) is particularly relevant, reflecting their common lithogenic origin linked to the weathering of ferromagnesian minerals and their low pedogenic mobility (Alloway, Reference Alloway2012; Kabata-Pendias, Reference Kabata-Pendias2010).
Pearson’s correlation matrix between chemical and physical soil properties and elemental concentrations in the OFS (organic farming system) (a) and CFS (conventional farming system) (b) experimental fields. The colour gradient represents the correlation coefficient, ranging from −1 (strong negative correlation, blue) to +1 (strong positive correlation, red). White cells indicate a weak or no correlation.

The general structure of the cluster is present in both fields, but the correlation intensity is significantly higher in the OFS (Figure 1a). In this site, element correlations are systematically stronger, indicating greater mineralogical homogeneity and a more uniform distribution of adsorptive phases within the profile. Such behaviours are typical of soils characterised by abundant Fe/Mn oxides and active clay (Cornell and Schwertmann, Reference Cornell and Schwertmann2003). In contrast, the CFS field (Figure 1b) exhibits greater heterogeneity in correlations, suggesting a more complex granulometric control in which silt and clay jointly contribute to elemental retention.
Soil texture emerges as one of the most influential factors in shaping element distribution. Negative correlations between sand and most elements in both sites are consistent with the low adsorptive capacity of sand and the dilutive effect of coarse fractions (Brady and Weil, Reference Brady, Weil and Fox2016). Conversely, clay and silt show pronounced positive correlations with metals and metalloids, confirming that the distribution of reactive surfaces and mineral colloids is the primary driver of elemental retention in soils (Sposito, Reference Sposito2008; Violante et al., Reference Violante, Pigna and Cozzolino2018). However, the relative contribution of fine fractions varies between the two sites: in OFS (Figure 1a), clay is clearly the main granulometric controller (with very high correlations with Cr, V, Pb, and Zn), whereas in CFS (Figure 1b), silt also plays a significant role (with high correlation with As, Cu, and Ti), suggesting a more mixed texture and higher microenvironmental variability.
Soil chemical properties show differential behaviour between the two fields. In CFS (Figure 1b), pH exhibits strong negative correlations with several elements (Cr, Pb, V, and Zn), indicating that acid–base conditions significantly influence adsorption mechanisms, surface protonation, and metal speciation (Fendorf et al., Reference Fendorf, Li and Sparks2017; McBride, Reference McBride1994; Sparks, Reference Sparks2003). In OFS (Figure 1a), pH exerts a lesser influence, while physical variables such as skeleton content and moisture are more relevant in modulating element distribution. ECe is more closely correlated with metals in OFS, suggesting a possible indirect influence of ionic strength and competition for adsorptive sites (Alloway, Reference Alloway2012; Tipping et al., Reference Tipping, Lofts and Sonke2020). Overall, the correlation matrices indicate that both soils share a strong common geochemical base dominated by fine fraction and Fe/Mn oxide control but differ in the relative weight of individual pedogenic processes. The OFS appears to be a more homogeneous system strongly structured around the role of clay and pH, whereas the CFS is characterised by greater granulometric heterogeneity and joint control of silt, clay, and physical variables. These differences in pedogeochemical structure suggest different responses to mobility, transfer, and stability processes of elements within the profile, with potential implications in the bioavailability of elements in the soil-plant system.
Principal component analysis to assess the relationship between TEs and PTEs concentrations and experimental fields
Principal component analysis (PCA) was employed to explore correlations and differences in the total elemental content of rhizosphere soils. The biplot (Figure 2) shows the relationships among the analysed elements (As, Cr, Cu, Ni, Pb, Ti, V, and Zn) and the distribution of soils from three durum wheat varieties across two experimental fields. The first two principal components account for 93% of the total variance, effectively summarising the main patterns in the dataset. PC1 distinctly separates the soils from the two fields, reflecting substantial differences in their elemental composition. Two clear groups of elements are observed: Cr, Cu, Ni, and Zn are positively correlated and more abundant in the CFS field, while As, Pb, Ti, and V are positively correlated and dominate in the OFS field. The distinct clustering of elements observed in the PCA biplot aligns with the patterns revealed by the correlation matrices. In the OFS soil, the elements As, Pb, Ti, and V are associated with a more homogeneous fine fraction, where clay and Fe/Mn oxides exert strong and coherent control on metal retention. In contrast, the grouping of Cr, Cu, Ni, and Zn in the CFS reflects the more heterogeneous granulometric structure of this field, where both silt and clay contribute to elemental retention, and physical variables such as moisture and skeleton content play an additional role. These field-specific elemental patterns may lead to differences in the mobility and bioavailability of trace elements, resulting in distinct behaviours of these elements within the soil/plant system.
Correlations of variables (levels of various elements in rhizosphere soil relative to the two test fields (OFS, organic farming system and CFS, conventional farming system) for each statistical unit (variety) to the main components PC1 and PC2. suo = soil.

Total content of TEs and PTEs in plants
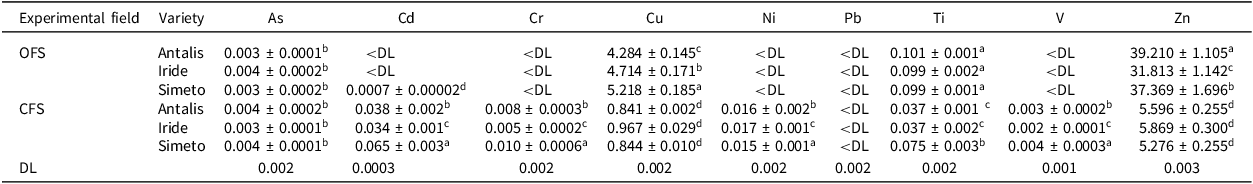
Each variety was studied in triplicate per experimental field to determine the total content and distribution of elements in different plant parts (roots, stems, and grains). The results obtained for stems, leaves, flowers, and spikelets are provided in Supplementary Material Tables S3 and S4. The grains of the OFS field were mainly characterised by the elements Cu, Ti, and Zn (Table 3), which are consistent with the average values reported in the literature for similar plant matrices (Kabata-Pendias, Reference Kabata-Pendias2010). However, Cd was above the detection limit of the analytical technique only in the Simeto variety, in any case, below the limit of 0.2 mg kg−1 imposed by EC Regulation 2023/915. As, Co, Cr, Ni, Pb, and V were below the detection limit of the analytical technique. In the case of the CFS field, the elements As, Cd, Cr, Cu, Ni, Ti, and Zn were consistent with the average values reported in the literature (Kabata-Pendias, Reference Kabata-Pendias2010). The V concentration in the grains from the CFS field was consistent with the values reported by Ertl et al. (Reference Ertl and Goessler2018) for European wheat grains and flours; instead, Pb was below the detection limit of the analytical technique.
Total element content in durum wheat grains (mg kg−1 dry matter) in organic (OFS) and conventional (CFS) farming systems

Values represent the average value concentration ± standard deviation from three replicates. Two-way ANOVA and statistical significance were applied to compare the multielemental content of the three varieties in the experimental fields of OFS and CFS. For each element, different letters indicate significant differences at p < 0.05. DL = Detection Limit.
According to the literature, organically grown wheat grains contain higher levels of essential mineral nutrients such as Cu and Zn than conventionally grown products (Magdas et al., Reference Magdas, Dehelean, Feher and Radu2017; Vrček et al., Reference Vrček, Čepo, Rašić, Peraica, Žuntar, Bojić, Mendaš and Medić-Šarić2014). On the other hand, higher levels of Cd, Cr, Ni, and V were observed in conventionally grown wheat grain. For Cd, this may be due to the use of mineral P fertilisers in conventionally cultivated soil (CFS), which has been linked to increased concentrations of Cd in soils and cereal grains (Park et al., Reference Park, Kim, Jung, Lee, Choi, Owens, Kumar, Yun and Hong2021; Suciu et al., Reference Suciu, De Vivo, Rizzati and Capri2022). Furthermore, mineral P fertilisers account for 45% of the total Cd contamination of croplands in Europe. Similarly, 55% of the average European consumer’s Cd intake is associated with Cd accumulation in soil (Suciu et al., Reference Suciu, De Vivo, Rizzati and Capri2022). These results are extremely important, especially from a food safety perspective. However, the durum wheat samples studied, from both organic and conventional farming systems, have never exceeded the threshold limits for Pb and Cd imposed by the European Union (EU 2023/915), which are 0.20 and 0.18 mg kg−1, respectively.
The elements in the stems from the fields OFS and CFS were Cu, Ti, and Zn, while As, Cd, Cr, Ni, Pb, and V were below the detection limit of the analytical technique (Supplementary Material Table S3), probably because the stem for most elements serves only as a transport channel from roots to grains in durum wheat plants. The roots of the OFS field were mainly characterised by the elements Cd, Cr, Cu, Ni, Ti, V, and Zn (Supplementary Material Table S4), while As and Pb were below the detection limit of the analytical technique. Otherwise, the elements in the roots included Cr, Cu, Ni, Pb, Ti, V, and Zn; As and Cd were below the detection limit of the analytical technique.
Evaluation of the uptake and translocation of TEs and PTEs in the soil-plant system
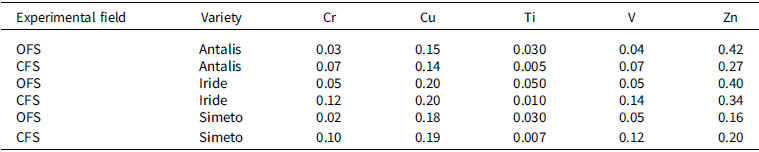
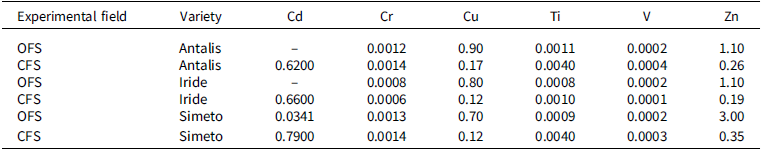
The BAC values were calculated for each variety to show the soil/root transfer. The mean values of BAC obtained from the three biological replicates of each variety are reported in Table 4 for OFS and CFS fields. The translocation factor (TF) value was calculated for each variety to show the translocation of elements from roots to stems and grains, respectively. The mean values of the TFs obtained from the three biological replicates of each variety are reported in Tables 5 and 6 for the experimental fields of OFS and CFS. BAC and TF indicated that Cu and Zn are easily absorbed and translocated to the stems and grains; for other non-essential elements such as Cr, Pb, Ti, and V, a barrier effect is consistently observed at the root level. In any case, the TF values indicate that Cr, Ti, and V are preferentially translocated to the stems, with limited or no translocation to the seeds. For these elements, the same behaviour was observed in all varieties across both experimental fields. On the other hand, Cr, Pb, Ti, and V, which are not nutrients, and Ni, which is not required in large quantities by plant organisms, are poorly absorbed by plant roots and are moved in very small quantities to other parts of the plant.
BAC (biological absorption coefficient) values for Cr, Cu, Ti, V, and Zn for each variety in organic (OFS) and conventional (CFS) farming systems

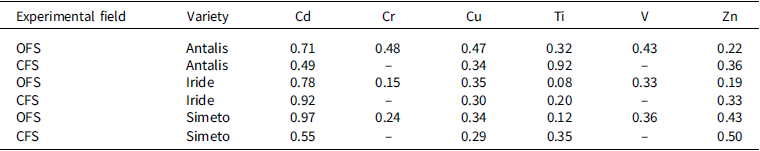
TF (translocation factor) values from roots to grains for Cd, Cr, Cu, Ti, V, and Zn for each variety in organic (OFS) and conventional (CFS) farming systems

‘–’ indicates that the TF could not be calculated because the concentration of the element was below the detection limit of the analytical technique.
TF (translocation factor) values from roots to stems for Cd, Cr, Cu, Ti, V, and Zn for each variety in organic (OFS) and conventional (CFS) farming systems

‘–’ indicates that the TF could not be calculated because the concentration of the element was below the detection limit of the analytical technique.
In both experimental fields, the Simeto variety exhibited a higher capacity to transport cadmium to the grains, the edible part. Numerous studies support this result, attributing it to genotypic differences among varieties. The genotypic variation that determines the accumulation of Cd in durum wheat grains is associated with the Cdu1 gene (Cadmium uptake 1), an important QTL that regulates Cd accumulation in wheat. The presence of this gene yields the phenotype ‘low grain Cd variety’, while its absence results in the phenotype ‘high grain Cd variety’. This gene controls the translocation of Cd from the roots to the grains through the xylem and phloem in the stem (Harris and Taylor, Reference Harris and Taylor2001, Reference Harris and Taylor2013; Zhang et al., Reference Zhang, Zhou, Teng, Chen and Li2024). It has been observed that the Iride and Antalis varieties are effective in blocking cadmium at the root level, as is confirmed by the results we have obtained. Previous studies have shown that the Iride variety can store significant amounts of Cd in the roots, preventing its movement to the grains (Mendez and Maier, Reference Mendez and Maier2008).
PCA was utilised as an exploratory technique to uncover correlations and differences in the total content of elements in the various parts of the plant (roots, stems, and grains) in each experimental field and variety. Although the first two principal components account for 72% of the data’s variance, the biplot reveals a clear distinction between the roots of durum wheat from the two experimental fields in PC1 (Figure 3a). Mn, Cd, Ti, and Zn are positively correlated and more prevalent in the roots from the organic experimental field. Conversely, Cu, Ni, V, and Cr are positively correlated and more present in the roots of the CFS experimental field (particularly in the roots of Iride and Simeto). Additionally, Fe is found to be more abundant in the roots of CFS (specifically those of the Antalis variety) and exhibits a slight negative correlation with Mn, Cd, Ti, and Zn. This pattern mirrors the geochemical differences previously observed in the rhizosphere soils, indicating that soil composition modulates the dynamics of elements within the root system.
Correlations of variables (levels of various elements in the roots (a), stems (b), and grains (c) of durum wheat relative to the two experimental fields, OFS, organic farming system, and CFS, conventional farming system) for each statistical unit (variety) regarding the main components PC1 and PC2. Rad = root; aer = stem; car = grain.

Figure 3b shows the correlations between the levels of various elements in the stems of different varieties in two experimental fields. The figure shows the vectors related to the elements that are under examination, such as Cu, Fe, Mn, Ti, and Zn, and the points relating to the stems of three durum wheat varieties, Antalis, Iride, and Simeto, in the two experimental fields. The first two principal components account for 69% of the total variance in the data. The biplot reveals that there is a separation of fields in the second principal component (PC2). However, there is no clear difference between the stems of the two experimental fields considered, probably because the stem for most elements serves only as a transport channel from roots to grains in durum wheat plants.
Figure 3c shows the correlations between the levels of various elements in the grains of different varieties in two experimental fields. The figure shows the vectors related to the elements that are under examination, such as Cd, Cu, Ti, and Zn, and the points relating to the grains of three durum wheat varieties, Antalis, Iride, and Simeto, in the two experimental fields. The biplot clearly shows a distinction between the two experimental fields on PC1, and the first two principal components explain 89% of the data variance. In particular, the samples of the experimental field CFS were grouped in the left square due to a higher concentration of Cd, while the elements that determined the cluster of the OFS experimental field were Cu, Ti, and Zn.
Interestingly, the spread of samples is narrower in the PCA of grains compared with soils and roots, suggesting a progressive physiological filtering effect occurring during translocation from roots to reproductive tissues. While soil geochemical differences strongly determine the element pools available to the plant, grain composition reflects a combination of environmental supply and plant-level regulation, resulting in a more constrained multielement pattern. This suggests that the varying mobility of elements in the soil and the type of cultivation (organic or conventional) may influence the total content of these elements in the soil-plant system and the plant parts.
To further contextualise these findings, recent literature on soil-to-plant transfer of potentially toxic elements (PTEs) has been considered, highlighting the roles of bioavailability, soil properties, agronomic practices and genotype.
Recent evidence indicates that the transfer of PTEs from soil to wheat grains is not solely determined by total soil concentrations but is strongly influenced by soil physicochemical properties, agronomic management, and soil amendments, which modulate metal bioavailability and plant uptake (Abd ElAzeim et al., Reference Abd ElAzeim, Menesi, Abd ElMageed, Lemanowicz and Haddad2022; Antonangelo et al., Reference Antonangelo, Zhang and Sitienei2023). For instance, a recent study on durum wheat reported considerable variability of metal accumulation among plant organs, with roots retaining most metals and limited translocation to grains under standard agricultural conditions (Berrettoni et al., Reference Berrettoni, Zamponi, Conti, Fattobene and Liu2023). This suggests that total soil metal content may overestimate the risks for edible parts if not coupled with bioavailability assessments.
Soil amendments, such as biochar and bentonite, have been shown to effectively reduce metal mobility and bioavailability. Application of 3–6% bentonite significantly decreased cadmium (Cd) uptake in both bread and durum wheat, while increasing biomass production (Özkutlu et al., Reference Özkutlu, Aydemir, Kocaman, Ece and Akgün2025). Similarly, biochar amendments reduced extractable fractions of Cd, Pb, and other metals, lowering their uptake by plants and improving plant physiological status (Antonangelo et al., Reference Antonangelo, Zhang and Sitienei2023; Algethami et al., Reference Algethami, Irshad, Javed, Alhamami and Ibrahim2023). These observations highlight that agronomic interventions can modulate metal uptake independently of total soil load, offering potential mitigation strategies for safer grain production.
Furthermore, soil properties, including cation exchange capacity (CEC), texture, organic matter content, and pH, play a central role in heavy-metal dynamics and accumulation in wheat grains. Field-scale studies have demonstrated strong correlations between CEC and Cd accumulation, whereas As accumulation was more associated with organic matter content (Bi et al., Reference Bi, Chu, Fu, Xu, Zhao, Zhong, Wang, Li and Zhang2023). Consequently, soils with similar total metal concentrations may differ significantly in bioavailable fractions and actual uptake by plants. In the present study, as only total soil concentrations were assessed, these factors must be considered when interpreting potential food-safety implications.
From an agronomic and food-safety perspective, these findings emphasise the need for integrated strategies. Ongoing analyses aim to quantify the bioavailable fraction of metals (e.g., via chemical extractants), which will allow for a more accurate assessment of uptake risk and clarify the combined effects of soil chemistry, texture, microbial communities, and plant genotype. Until these data are available, recommendations regarding specific wheat varieties or cultivation practices remain provisional. Ultimately, combining appropriate variety selection, soil amendments (e.g., biochar, bentonite), and sustainable soil management may mitigate the risk of toxic element accumulation while maintaining yield and grain quality.
Conclusions
This study provides a comprehensive assessment of the behaviour of toxic and potentially toxic elements in the soil–plant system of durum wheat cultivated under organic or conventional farming systems. Sampling individual durum wheat plants and their soil in the rhizosphere allowed a detailed assessment of the transfer capacity of elements within the soil-plant system. Having different varieties available in the same field minimised the variability due to pedoclimatic and agronomic conditions, allowing us to focus specifically on the effects related to the genotype itself. Furthermore, having the same varieties in two different experimental fields facilitated the evaluation of the impacts associated with organic versus conventional farming systems and soil characteristics. The results demonstrate that soil properties, cultivation practices, and varietal differences significantly influence the uptake and translocation of elements within plant tissues. Organic farming systems were associated with higher concentrations of essential micronutrients such as Cu and Zn in grains, while conventionally cultivated wheat showed elevated levels of Cd, Cr, Ni, and V, although all remained within regulatory safety limits. The Simeto variety exhibited a greater translocation of Cd to the kernels, highlighting the importance of the genetic factor in element mobility. These findings revealed the relevance of cultivation practices and varietal selection in mitigating the accumulation of harmful elements in edible plant parts. Further research should focus on long-term monitoring of soil contamination and element dynamics under different agricultural systems, including the role of fertilisation and irrigation practices. Additionally, molecular studies on the genetic mechanisms governing element uptake and translocation, particularly those related to toxic elements, mainly Cd, could inform breeding programmes aimed at developing safer wheat varieties.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0014479726100234.
Data availability statement
Not applicable.
Author contributions
EP: conceptualisation, investigation, writing-original draft, writing-review & editing. AB, GP, VP: writing-review & editing. CZ, CM, DP, LG: conceptualisation and validation, supervision, writing-review & editing. All authors read and approved the submitted version.
Funding statement
This paper is funded by METROFOOD-IT project funding from the European Union – NextGenerationEU, PNRR-Mission 4 ‘Education and Research’ Component 2: from research to business, Investment 3.1: Fund for the realisation of an integrated system of research and innovation infrastructures [IR0000033] (D.M. Prot. n.120 del 21/06/2022) and by AGRITECH funding from the European Union-NextGenerationEU, PNRR-Mission 4 ‘Education and Research’ Component 2: from research to business, Investment 1.4: ‘National Research Centre for Agricultural Technologies’ Agritech CUP HUB – B63D21015240004.
This manuscript reflects only the authors’ views and opinions; neither the European Union nor the European Commission can be considered responsible for them. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing interests
The authors declare that they have no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publication
Not applicable.