Introduction
In 2013, nomenclature of the garnet supergroup, in which 32 mineral species were described, was elaborated by Grew et al. and approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (Grew et al., Reference Grew, Locock, Mills, Galuskina, Galuskin and Halenius2013). There are currently ∼40 mineral species in the garnet supergroup. The rarest natural garnet, rubinite, was discovered in refractory inclusions in CV3 chondrites (Vigarano, Allende and Efremovka) in 2015 and has the ideal formula Ca3Ti3+2Si3O12 (Мa et al., Reference Ma, Yoshizaki, Krot, Beckett, Nakamura, Nagashima, Muto, Ivanova and Ulyanov2025). It is considered to be one of the earliest minerals formed in the solar nebula.
Minerals in which titanium has a valence of less than 4+ are considered to be indicators of super-reduced conditions. At terrestrial sites, the discovery of minerals containing trivalent titanium (carmeltazite, magnéliite, sassite, tistarite and others) is related mainly to the study of inclusions in corundum from heavy fractions (separates) that were most likely contaminated by anthropogenic material (Griffin et al., Reference Griffin, Gain, Adams, Huang, Saunders, Toledo, Pearson and O’Reilly2016, Reference Griffin, Gain, Bindi, Toledo, Cámara, Saunders and O’Reilly2018; Litasov et al., Reference Litasov, Kagi and Bekker2019; Balhaus et al., Reference Ballhaus, Helmy, Fonseca, Wirth, Schreiber and Jöns2021; Galuskin and Galuskina, Reference Galuskin and Galuskina2023a, Reference Galuskin and Galuskina2023b, Ma et al., Reference Ma, Cámara, Bindi, Toledo and Griffin2023).
There are rare finds of super-reduced minerals in situ in terrestrial rocks, where there is no doubt as to their natural origin. One example is osbornite, TiN, found in a phosphide-bearing breccia from a small outcrop of pyrometamorphic rocks of the Hatrurim Complex in the Negev Desert, Israel (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022). Rubinite was found in a single sample of the same breccia in association with osbornite. Rubinite completes the list of the garnet supergroup minerals found in the pyrometamorphic rocks of the Hatrurim Complex. Thus, garnets of the andradite–schorlomite–grossular series, often with significant admixture of Cr and V, are most common type found in gehlenite hornfels and wollastonite–rankinite paralavas (Krzątała et al., Reference Krzątała, Krüger, Galuskina, Vapnik and Galuskin2020). The pyrometamorphic rocks of the Hatrurim Complex are the type localities of two new garnets: priscillagrewite-(Y), (Ca2Y)Zr2Al3O12, found in fluorapatite–spurrite marbles, Daba Siwaqa, Jordan (Galuskina et al., Reference Galuskina, Galuskin, Vapnik, Zeliński and Prusik2021) and midbarite, Ca3Mg2(V5+2Si)O12, found in gehlenite paralava, Hatrurim Basin, Israel (Galuskina et al., Reference Galuskina, Galuskin, Kusz, Książek, Vapnik and Marciniak-Maliszewska2024). In addition, garnets of the kerimasite–kimzeyite series, Ca3Zr2Fe3+2SiO12–Ca3Zr2Al2SiO12, an Al-analogue of elbrusite corresponding to the composition series Ca3(Zr1.5U6+0.5)Al3O12–Ca3ZrU5+Al3O12 (Galuskina et al., Reference Galuskina, Galuskin, Vapnik, Murashko, Dzierżanowski and Gazeev2015) and an analogue of midbarite with Mn2+>Mg were found in single cases. Garnets of the grossular–katoite series are widespread in the low-temperature alteration products of the pyrometamorphic rocks of the Hatrurim Complex (Kruszewski et al., Reference Kruszewski, Palchik, Vapnik, Nowak, Banasik and Galuskina2021). All of the above-mentioned garnets are characteristic of oxidised mineral associations of rocks of the Hatrurim Complex, whereas finds of garnets in reduced associations, as in the case of rubinite, are rare. For example, Fe-free Ti–V-bearing uvarovite has been found in wollastonite–gehlenite paralava with large inclusions of native iron up to 1 cm, crystallised under reduced conditions near the iron–wüstite buffer (Futrzyński et al., Reference Futrzyński, Juroszek, Skrzyńska, Vapnik and Galuskin2023).
In this paper, we present the results of a study of the morphology and composition of rubinite and associated minerals, and provide the results of single-crystal structural and Raman studies of rubinite. We also discuss the conditions and growth mechanism of rubinite, which reflect the formation of the mineral under super-reduced conditions at the Earth’s surface.
Methods of investigation
The morphology and composition of rubinite and associated minerals were studied using an Olympus optical microscope, a Quanta 250 scanning electron microscope with UltraDry EDS Detector (Institute of Earth Sciences, Faculty of Natural Sciences, University of Silesia, Sosnowiec, Poland) and an electron probe microanalyser (Cameca SX100, Micro-Area Analysis Laboratory, Polish Geological Institute – National Research Institute, Warsaw, Poland). Chemical analyses were carried out in WDS mode (wavelength-dispersive X-ray spectroscopy, settings: 15 keV, 20 nA and ∼1 μm beam diameter) using the synthetic and natural standards: MgKα – diopside; SiKα and ZrLα – zircon; AlKα and KKα – orthoclase; CaKα – wollastonite; SrLα – celestine; NbLα – metallic Nb; BaLβ – baryte; TiKα – rutile; VKα – metallic V; CrKα – Cr2O3; PKα and FKα – fluorapatite; MnKα – rhodonite; FeKα – pentlandite; and ClKα – tugtupite. Elemental EDS analysis of minerals was performed using a routine procedure with internal standards, ZAF correction and 100% normalisation of totals.
The single-crystal X-ray diffraction study of a rubinite crystal was carried out using a SuperNova diffractometer with a mirror monochromator (СuKα, λ = 1.54184 Å) and an Atlas CCD detector (Agilent Technologies) at the Institute of Physics, University of Silesia, Poland.
The structure of rubinite was refined using the SHELX-2019/2 program (Sheldrick, Reference Sheldrick2015). The crystal structure was refined starting from the atomic coordinates of uvarovite (Futrzyński et al., Reference Futrzyński, Juroszek, Skrzyńska, Vapnik and Galuskin2023). The crystallographic information file has been deposited with the Principal Editor of Mineralogical Magazine and is available as Supplementary material (see below).
The Raman spectrum of rubinite was recorded on a WITec alpha 300R Confocal Raman Microscope (Department of Earth Science, University of Silesia, Poland) equipped with an air-cooled solid laser (488 nm) and a CCD camera operating at –61°C. An air Zeiss LD EC Epiplan-Neofluan DIC-100/0.75NA objective was used. Raman scattered light was focused onto a multi-mode fibre and monochromator with a 1800 mm–1 grating. The power of the laser at the sample position was ∼40 mW. Fifteen scans with an integration time of 3 s and a resolution of ∼3 cm–1 were collected and averaged. The spectrometer monochromator was calibrated using the Raman scattering line of a silicon plate (520.7 cm–1).
Occurrence
Rubinite was found in a single sample of the phosphide-bearing breccia of the Hatrurim Complex, which was discovered in 2019 as a small vertical zone 4–5 metres thick in an exposure created during the construction of the Arad – Dead Sea road (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022, Reference Galuskin and Galuskina2023a). The results of this breccia investigation can be found in our papers describing findings of ‘meteoritic minerals’ such as osbornite, barringerite and schreibersite (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022), andreyivanovite and allabogdanite (Galuskin et al., Reference Galuskin, Galuskina, Vapnik and Zieliński2023a), and caswellsilverite and grokhovskyite (Galuskin et al., Reference Galuskin, Kusz, Galuskina, Książek, Vapnik and Zieliński2023b). In the breccia, xenoliths of altered sedimentary rocks ranging in size from a few centimetres to 0.5 m, cemented by flamite–gehlenite (± rankinite, pseudowollastonite) paralava, usually have a characteristic zonation. The contact facies of black gehlenite–flamite or gehlenite–rankinite amygdaloidal paralava (zone 1) is represented by a light pseudowollastonite–gehlenite zone 2–3 mm thick, intensely replaced by OH-bearing grossular (hydrogrossular) and hydrous silicates (zone 2) (Fig. 1a). This thin zone reflects the effect of contamination of the carbonate xenolith material by the paralava, which is expressed as an increase in the Ca/Si ratio. Contamination led to the formation of pseudowollastonite in the paralava. At the contact with the paralava, the xenolith itself often has a thin zone several millimetres thick, which usually differs in colour (zone 3) from the central parts of the xenolith (zone 4, Fig. 1a). This zone is interpreted by us as a zone of almost complete melting (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022). Rubinite was only found where the zones described are unevenly developed (Fig. 1b).
(a) Typical zonation of breccia: 1 – gehlenite–flamite–rankinite paralava, 2 – pseudowollastonite-bearing paralava contaminated by xenolith material, 3 – altered hematite-bearing xenolith melting zone, 4 – altered hematite-bearing xenolith thermal alteration zone. (b) General view of sample with rubinite, frame outlines a fragments enlarged in (c). 1 – 4 – Zones as in (a). (c) Contact of xenolith with paralava. Fragments magnified in (d) and (e) are indicated by arrows. (d) Typical zonal aggregates of rubinite from zone 4. (e) Paqueite crystal with gehlenite inclusions from zone 3, nearby secondary hematite replaces troilite. (c–e) – BSE images. Abbreviations: Bgr = barringerite; Gh = gehlenite; Hem = hematite; Hgrs = hydrogrossular; HSi = hydrosilicates; Paq =paqueite; Pwo = pseudowollastonite; Rbi = rubinite; Tch = tacharanite.

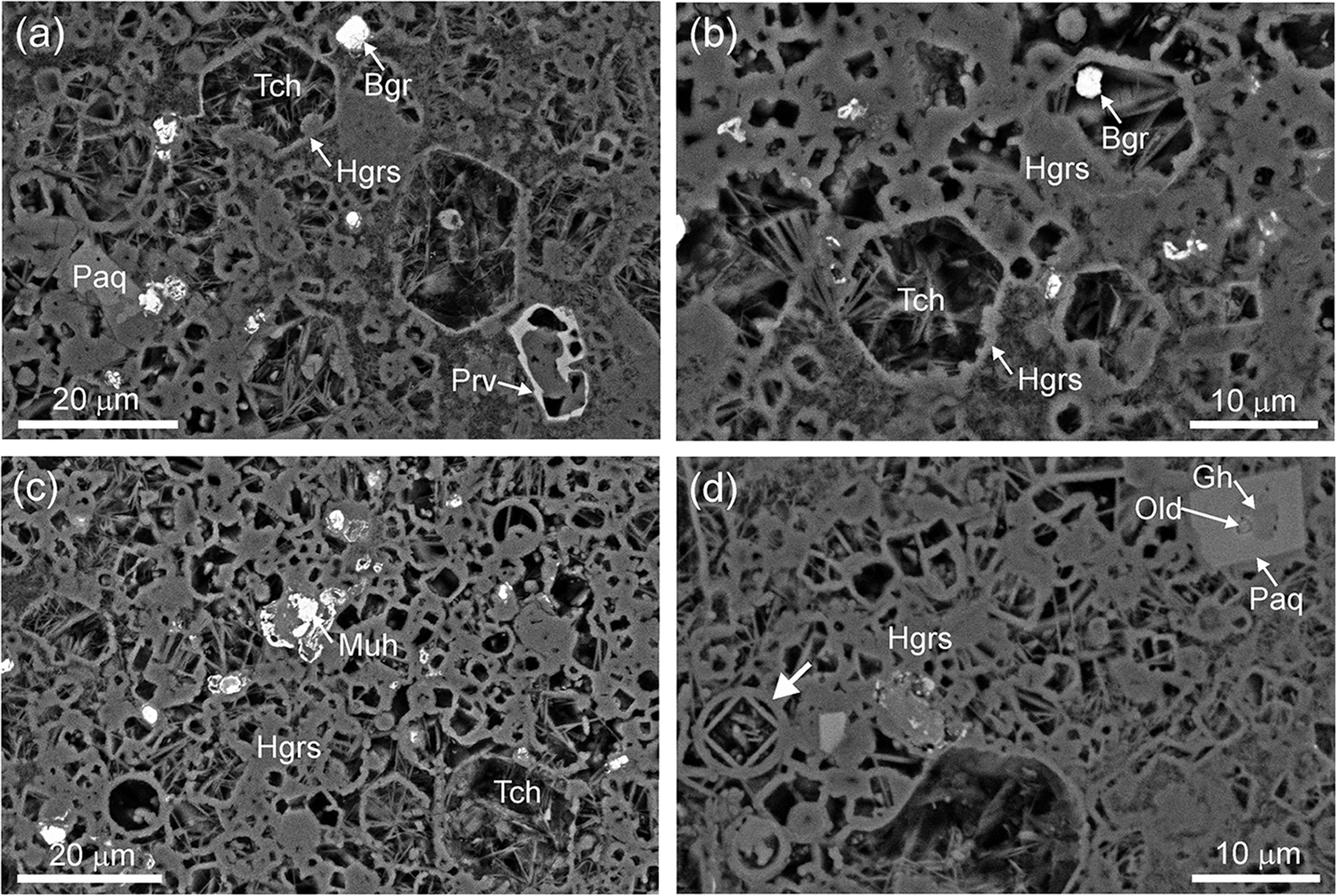
This xenolith, like most xenoliths of phosphide-bearing breccias, consists of hydrogrossular, calcium hydrosilicates (predominantly tacharanite) and calcite; in some fragments the rock is enriched in gypsum, ettringite, whewellite and halite (Fig. 2). Relict high-temperature minerals are represented by small grains of barringerite, murashkoite, perovskite, baghdadite, pseudowollastonite, cuspidine, osbornite, paqueite, fluorapatite, oldhamite and, rarely, gehlenite (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022). In the porous rock of the xenoliths, case-like hydrogarnet pseudomorphs are widespread (Fig. 2a,b), the morphology of which indicates that clasts of clay-carbonate sedimentary rock (Ghareb Formation) were transformed at temperatures >1,200°C into a gehlenite–hatrurite (+ CaO, CaS) clinker-like rock, which was later completely altered by low-temperature processes. The detection of circular forms, in some cases with inscribed rectangles (Fig. 2c,d), indicates the formation of melt droplets during clinkerisation.
BSE images of the xenolith breccia. (a, b) Hexagonal case-like pseudomorphs of hydrogrossular after hatrurite. (c, d) Rounded and rectangular case-like forms of pseudomorphs after hatrurite, gehlenite and melt drops. Abbreviations: Bgr = barringerite; Gh = gehlenite;. Hgrs = hydrogrossular; Muh = murashkoite; Old = oldhamite; Paq = paqueite; Prv = perovskite; Tch = tacharanite.

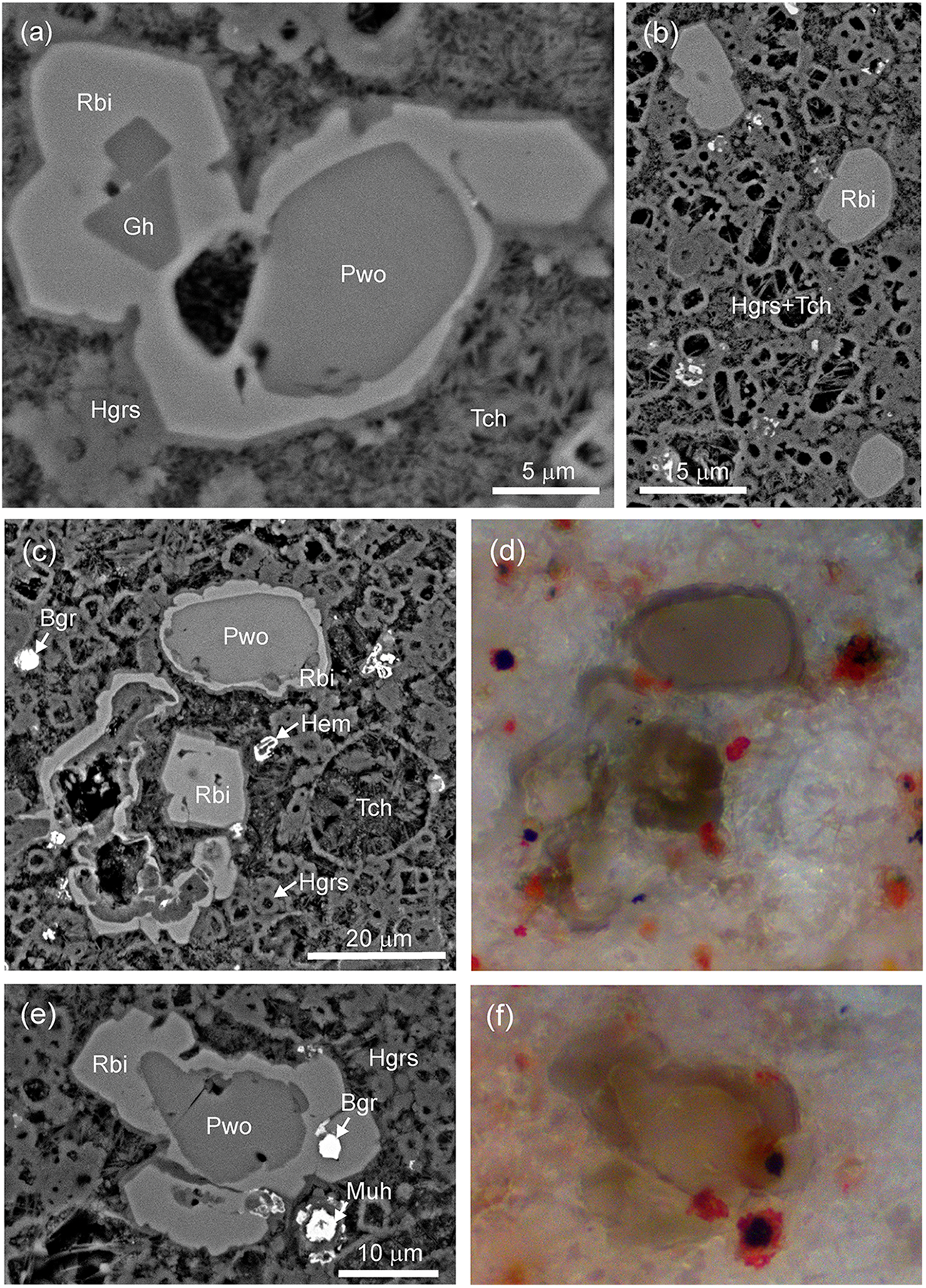
Numerous grains of rubinite, for which the composition is described by the simplified formula Ca3(Ti3+,Al,Ti4+,Mg)2(Si,Al)3O12, occur in zone 4 of the xenolith, whereas paqueite crystals of similar composition – Ca3Ti4+(Al,Ti4+,Si)3Si2O14 – are characteristic of zone 3 (Figs 1c–f, 3a). Rubinite forms aggregates with relics of pseudowollastonite in the central part of the crystals and only rarely forms single crystals (Figs 3b–d, 4). Reaction rims of rubinite on pseudowollastonite up to 10 µm thick appear as an eyed structure (Figs 3c, 4a,c,e), which is particularly visible in optical images where the light core of pseudowollastonite is surrounded by grey–brown rubinite, in some cases with a greenish tinge (Figs 3d, 4d,f).
(a) Central part of a xenolith represented by porous aggregates of hydrogrossular, tacharanite, calcite and gypsum, in some cases enriched with halite. Relict inclusions of high-temperature minerals are represented by rubinite, perovskite, pseudowollastonite, barringerite, murashkoite and osbornite. The white frames show the location of magnified images in (b,c,e). (b) Zonal aggregates, the centre of which is filled by pseudowollastonite and/or products of its alteration, and the rim consists of rubinite. Frame shows the location of magnified image in (d). (c) Aggregates of rubinite crystals with barringerite inclusions and clearly visible {221} and {110} faces. (d) Rubinite rims on pseudowollastonite. (e) Osbornite usually forms rims on barringerite. Images a–c – BSE; d, e – optical images, reflected light; d – XPL; e – PPL. Abbreviations: Bgr = barringerite; Hgrs = hydrogrossular; Hem = hematite; Muh = murashkoite; Obn = osbornite; Pwo = pseudowollastonite; Rbi = rubinite; Tch = tacharanite.

There are rare inclusions of gehlenite and barringerite in the rubinite crystals (Figs 3c, 4a,e) and osbornite associated with the rubinite typically forms as a rim on barringerite (Fig. 3a,e). Some of the rubinite crystals exhibit {211} and {110} faces (Fig. 4a,b,e).
(a) Pseudowollastonite and gehlenite inclusions in rubinite, BSE. (b) Rare single crystals of rubinite with {221} faces. (c–f) Reaction rim of rubinite on pseudowollastonite. Images: a–c, e – BSE; d, f – reflected light, XPL. Abbreviations: Bgr = barringerite; Gh = gehlenite; Hem = hematite; Hgrs = hydrogrossular; Muh = murashkoite; Pwo = pseudowollastonite; Rbi = rubinite; Tch = tacharanite.

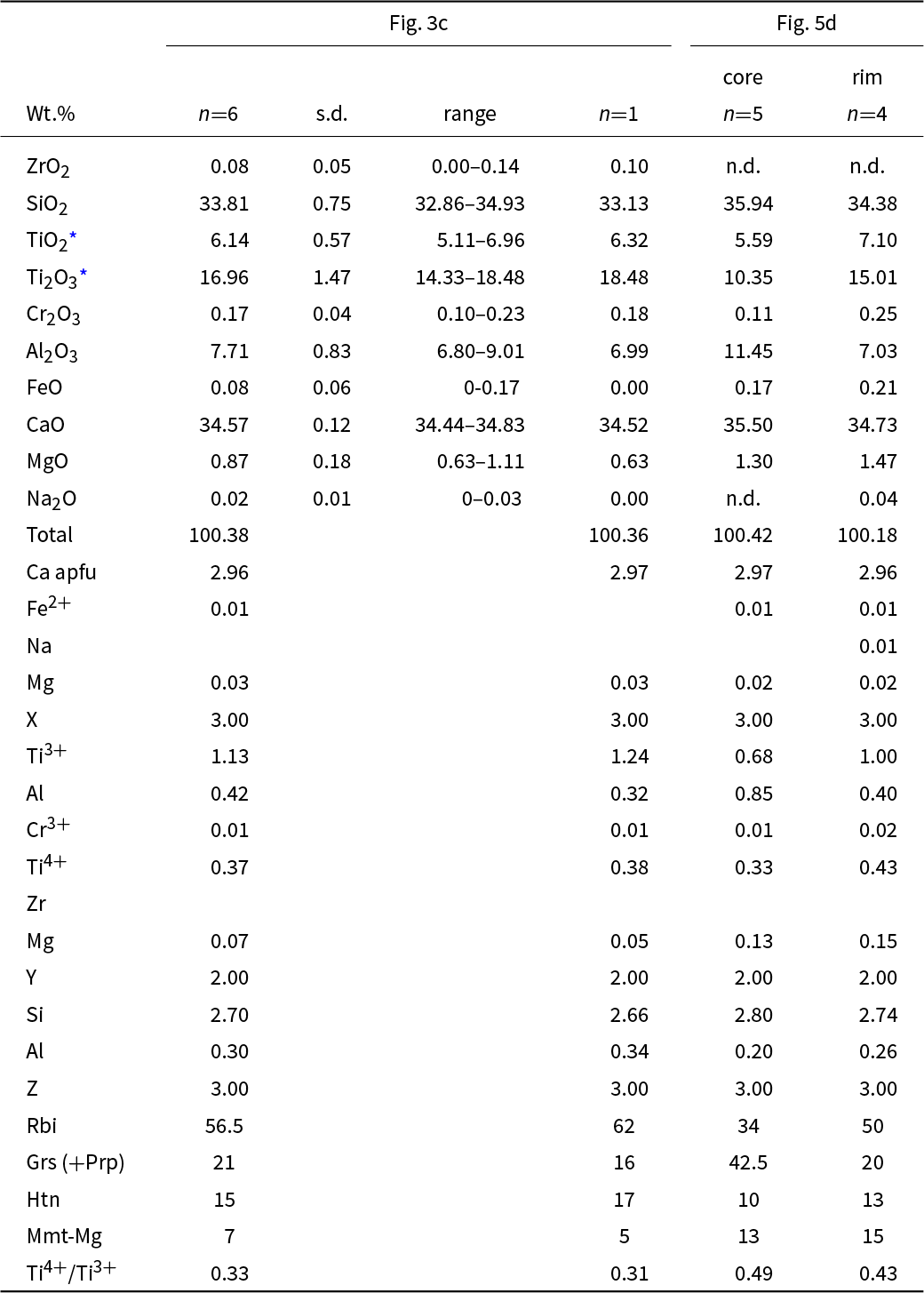
In one case, aggregates of garnet crystals of the grossular–rubinite series were found at the boundary between zones 2 and 3 (Fig. 5a,b). Garnet crystals occur in fragments of phosphide-rich rock and are associated with cuspidine, pseudowollastonite and paqueite (Fig. 5c), rarely with unusual blue Ti3+-bearing perovskite. Rubinite crystals overgrow a core of titanium-bearing grossular (Fig. 5d,e).
(a,b) Rare aggregates of garnet grains of the rubinite–grossular series from the boundary between paralava and xenolith (2 and 3 zones). (c) Position of zoned garnet crystals associated with phosphides and paqueite in the rock. Frame outlines the area magnified in d and e. (d, e) Zoned crystals with a Ti-bearing grossular core and Al-bearing rubinite rim. (e) Bright-red hematite replaces troilite and phosphides. Images: a,c – BSE, b – reflected light, XPL, d – BSE, e – reflected light, XPL. Abbreviations: Bgr = barringerite; Cal = calcite; Csp = cuspidine; Grs = grossular; Fap = fluorapatite; Hgrs = hydrogrossular; Hem = hematite; Muh = murashkoite; Paq = paqueite; Pwo = pseudowollastonite; Rbi = rubinite; Tch = tacharanite.

Chemical composition of rubinite and associated minerals
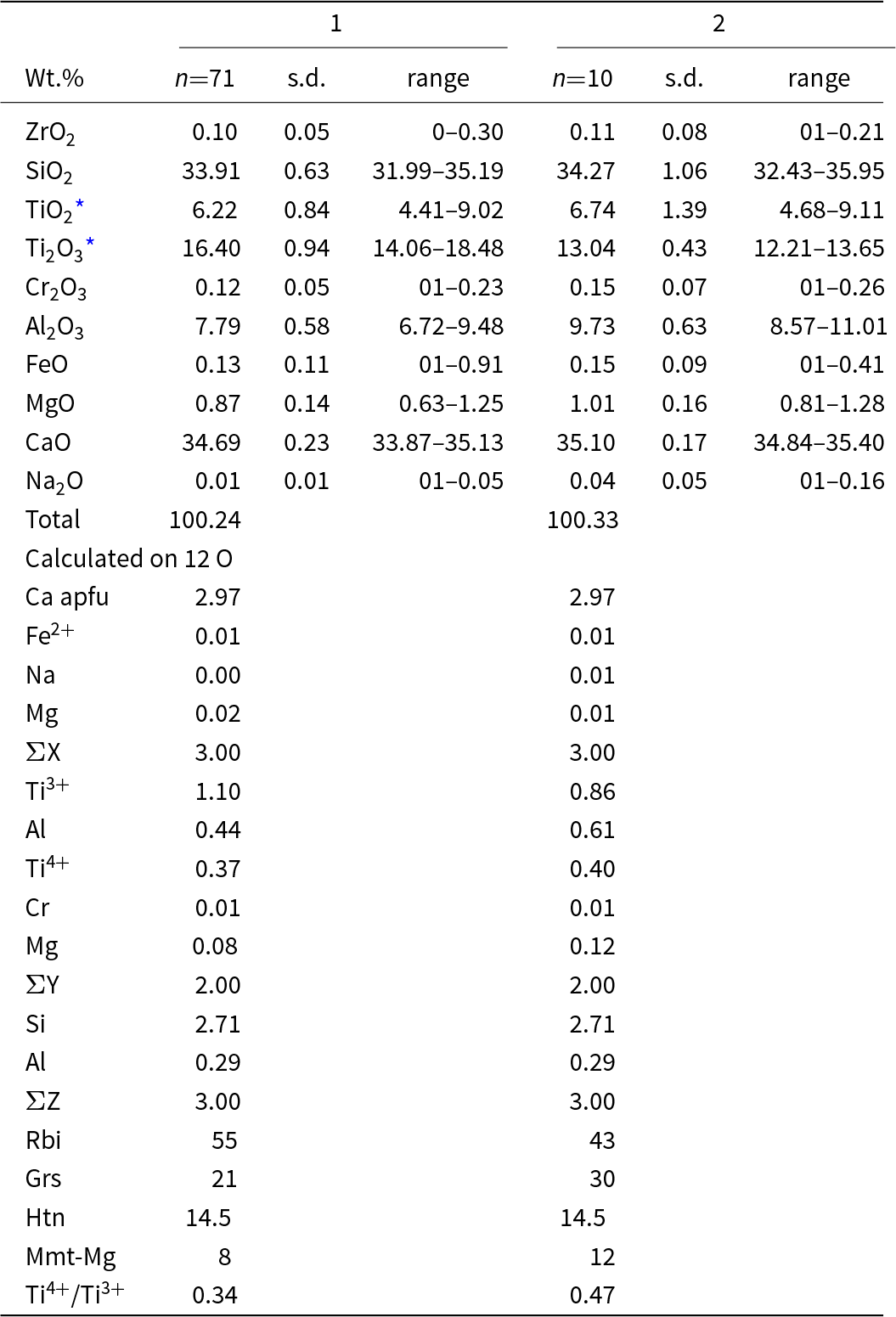
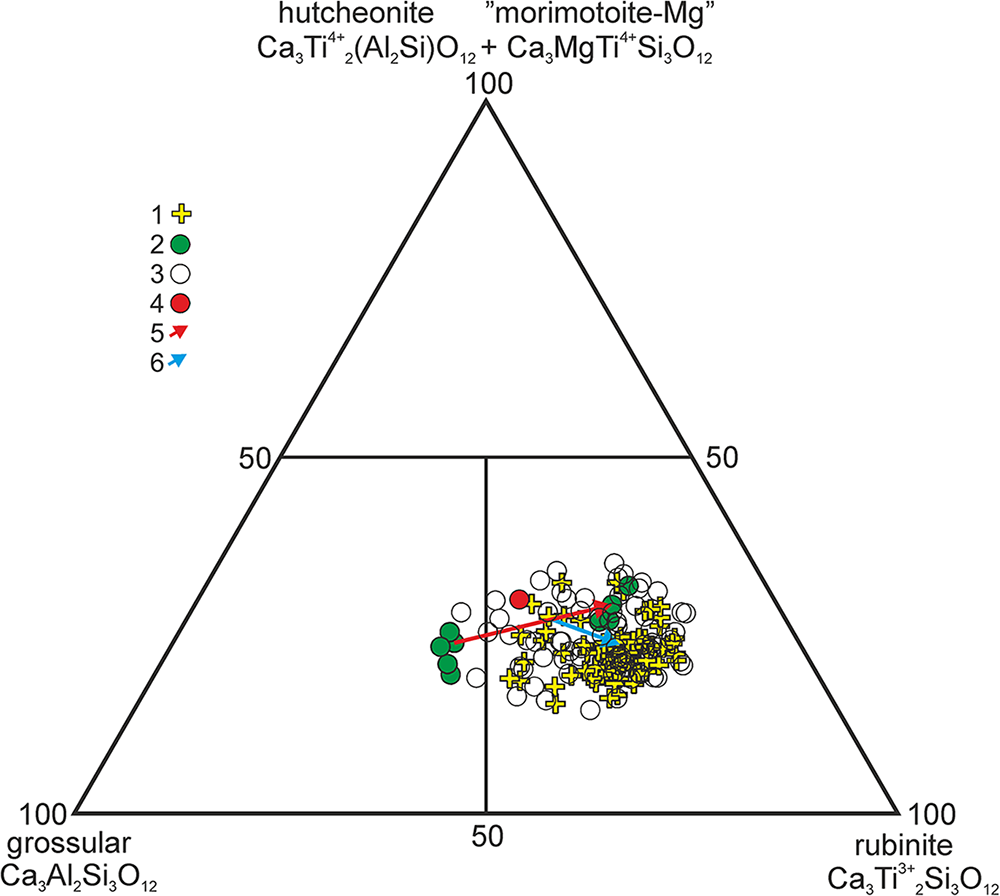
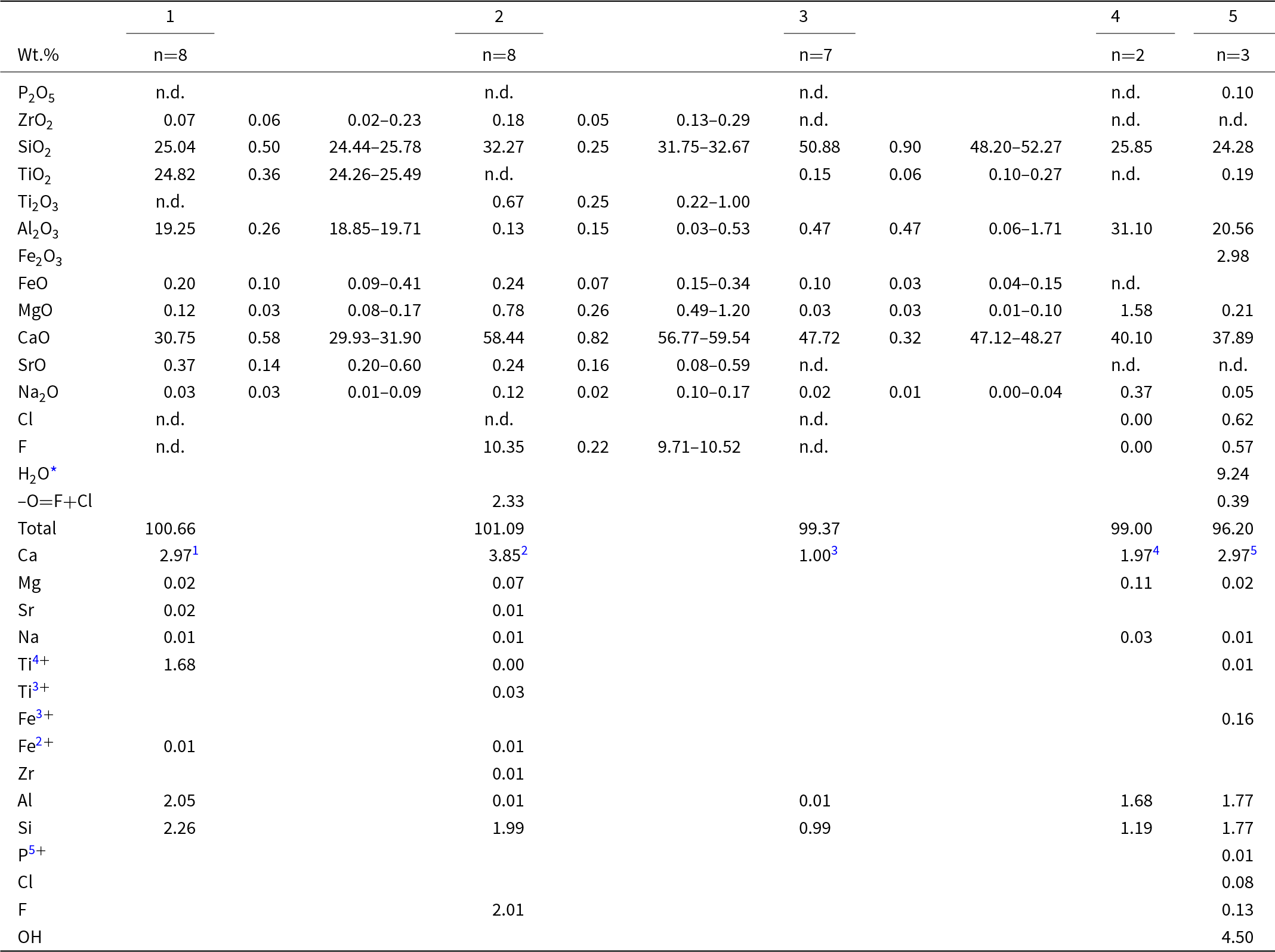
The composition of rubinite forming a reaction rim on pseudowollastonite in zone 4 of the xenolith, as well as its rare single crystals, is described by the average empirical formula (Ca2.97Mg0.02Fe2+0.01)Σ3.00(Ti3+1.10Al0.44Ti4+0.37Mg0.08Cr0.01)Σ2(Si2.71Al0.29)Σ3.00O12 (Table 1, analysis 1), corresponding to 55% rubinite, 21% grossular, 14.5% hutcheonite and 8% Mg-analogue morimotoite end-members. Both in the reaction rims and in the central parts of individual crystals, small Al-enriched spots can be seen, distinguished on back-scattered electron images (BSE) by their darker colour. In these spots, the content of the grossular end-member increases (30%) and the content of the rubinite end-member decreases (43%) (Table 1, analysis 2). The highest content of the rubinite end-member was found in the group of crystals shown in Fig. 3c (Table 2). One of the compositions of rubinite corresponds to the empirical formula (Ca2.97Mg0.03)Σ3.00(Ti3+1.24Ti4+0.38Al0.32Mg0.05Cr0.01)Σ2(Si2.66Al0.34)Σ3.00O12, which is calculated to be 62% rubinite, 16% grossular, 17% hutcheonite and 5% Mg-analogue of morimotoite end-members.
Chemical composition of rubinite in reaction zones on pseudowollastonite and single crystals (1) with dark spots (2) from zone 4 of the xenolith

* Ti3+/Ti4+ ratio was calculated on charge balance
Rbi–Ca3Ti3+2Si3O12, Grs–Ca3Al2Si3O12, Htn–Ca3Ti4+2(Al2Si)O12, Mmt-Mg–Ca3MgTi4+Si3O12
The zoned garnet crystals forming aggregates at the boundary of zones 2 and 3 (Fig. 5c–e) have a core represented by a colourless Ti-bearing grossular with the empirical formula (Ca2.97Mg0.02Fe2+0.01)Σ3.00(Al0.85Ti3+0.68Ti4+0.33Mg0.13Cr0.01)Σ2(Si2.80Al0.20)Σ3.00O12 (grossular 42.5%, rubinite 34%, hutcheonite 10% and Mg-analogue of morimotoite 13%). The marginal zone of the crystals is formed by rubinite of dirty-green colour (Fig. 5d, e) with an average composition described by the empirical formula (Ca2.96Mg0.02Fe2+0.01Na0.01)Σ3.00(Ti3+1.00Ti4+0.43Al0.40Mg0.15Cr0.02)Σ2(Si2.74Al0.26)Σ3.00O12 (rubinite – 50%, grossular – 20%, hutcheonite – 13% and Mg-analogue of morimotoite – 15%).
The diagram ‘rubinite–grossular–hutcheonite+Mg-analogue of morimotoite’ shows the distribution of the garnet compositions, the overwhelming majority of which fall into the rubinite field (Fig. 6). Two trends in garnet composition can be observed: (1) Grs42.5Rbi34Mmt-Mg13Htn10 → Rbi50Grs20Mmt-Mg15Htn13 and (2) Rbi43Grs30Htn14.5Mmt-Mg12 → Rbi55Grs21Htn14.5Mmt-Mg8. Both trends are accompanied by a decrease in the calculated Ti4+/Ti3+ value.
Garnet compositions plotted in the triangular diagram ‘rubinite–grossular–hutcheonite+Mg-analogue of morimotoite’: 1 – rubinite from zone 4; 2 – zoned garnet from the boundary of zones 2 and 3; 3 – EDS analyses; 4 – EDS analysis of crystal used for the structural study; 5, 6 – compositional trends.

The composition of relict high-temperature minerals and hydrogrossular associated with rubinite is given in Table 3. Paqueite from zone 3 of the xenolith (Table 3, analysis 1) is, like rubinite, a silicate of calcium, titanium and aluminium, but unlike the garnet it contains only Ti4+: (Ca2.97Sr0.02Na0.01)Σ3.00(Ti4+0.97Mg0.02 Fe2+0.01)Σ1.00Si2(Al2.05Ti4+0.71Si0.26)Σ3.02O14.
Chemical composition of paqueite (1), cuspidine (2), pseudowollastonite (3), gehlenite inclusions in rubinite (4) and hydrogrossular (5).

* Notes: – calculated; 1– calculated on 14 O; 2– calculated on 9(O+F); 3– calculated on 3 O; 4– calculated on 7 O; 5– calculated on 12(O+OH+F+Cl) and normalised on 3(Ca+Mg+Na). n.d. – not determined
Raman spectra of rubinite
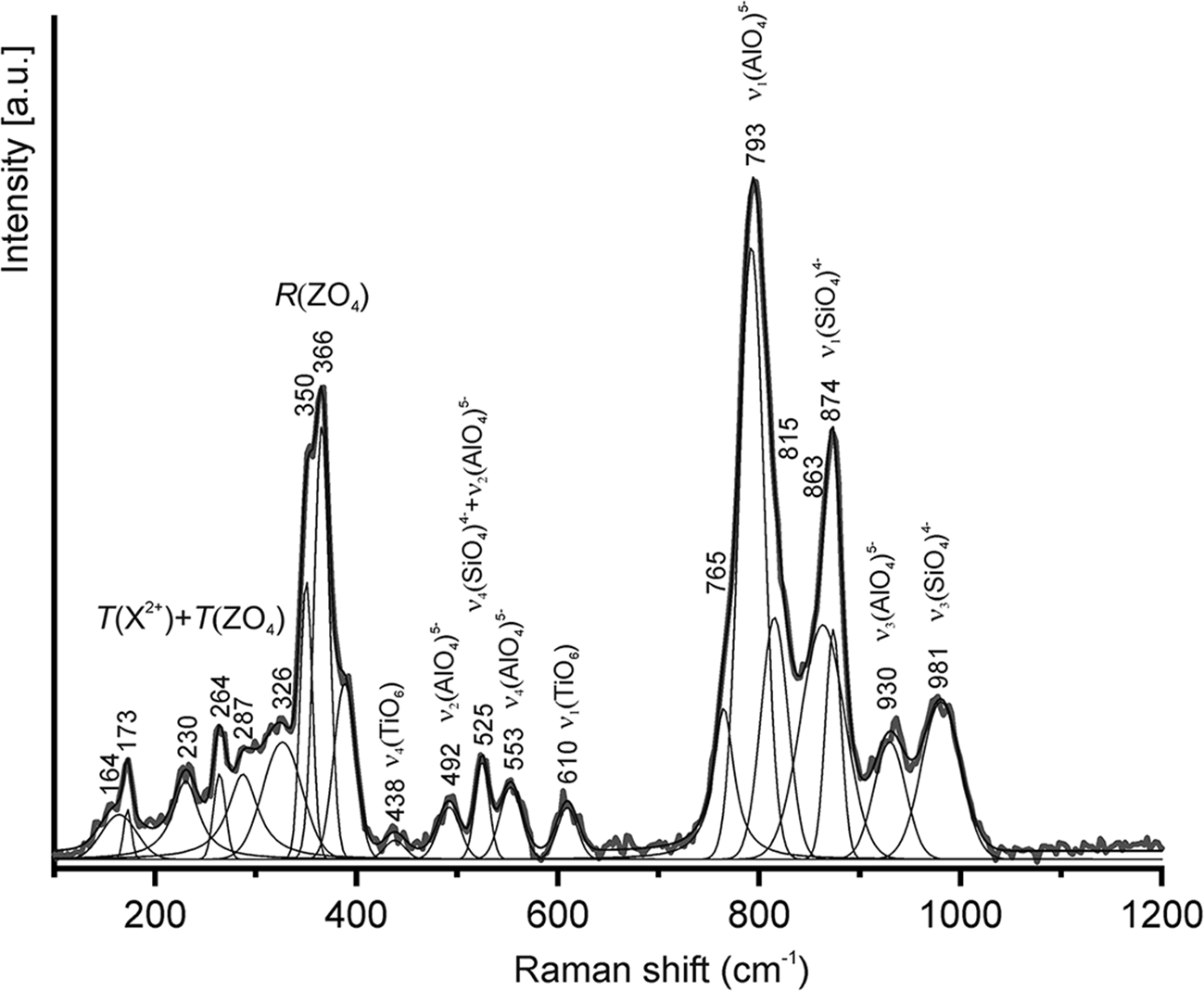
More than 30 Raman spectra have been obtained for rubinite, differing only in minor details. A typical rubinite spectrum is shown in Fig. 7, where there are bands corresponding to Si–O and Al–O vibrations in tetrahedra (TO4) (Hofmeister and Chopelas, Reference Hofmeister and Chopelas1991; Makreski et al., Reference Makreski, Runčevski and Jovanovski2011) and weak bands related to Ti4+–O vibrations in (TiO6)8– octahedra appear: 610 cm–1 ν1(TiO6)8– and 438 cm–1 ν4(TiO6)8–. In paqueite from the phosphide-bearing breccia these bands are at 611 and 437 cm–1, respectively (Galuskina et al., Reference Galuskina, Galuskin and Vapnik2023). A shoulder at 765 cm–1 may correspond to the vibration ν1(TiO4)4–. The Raman spectrum of terrestrial rubinite is similar to that of the holotype rubinite from the meteorite (Ma et al., Reference Ma, Yoshizaki, Krot, Beckett, Nakamura, Nagashima, Muto, Ivanova and Ulyanov2025).
Raman spectrum of rubinite from the Hatrurim Complex.

Results of the study and discussion of the structure of rubinite
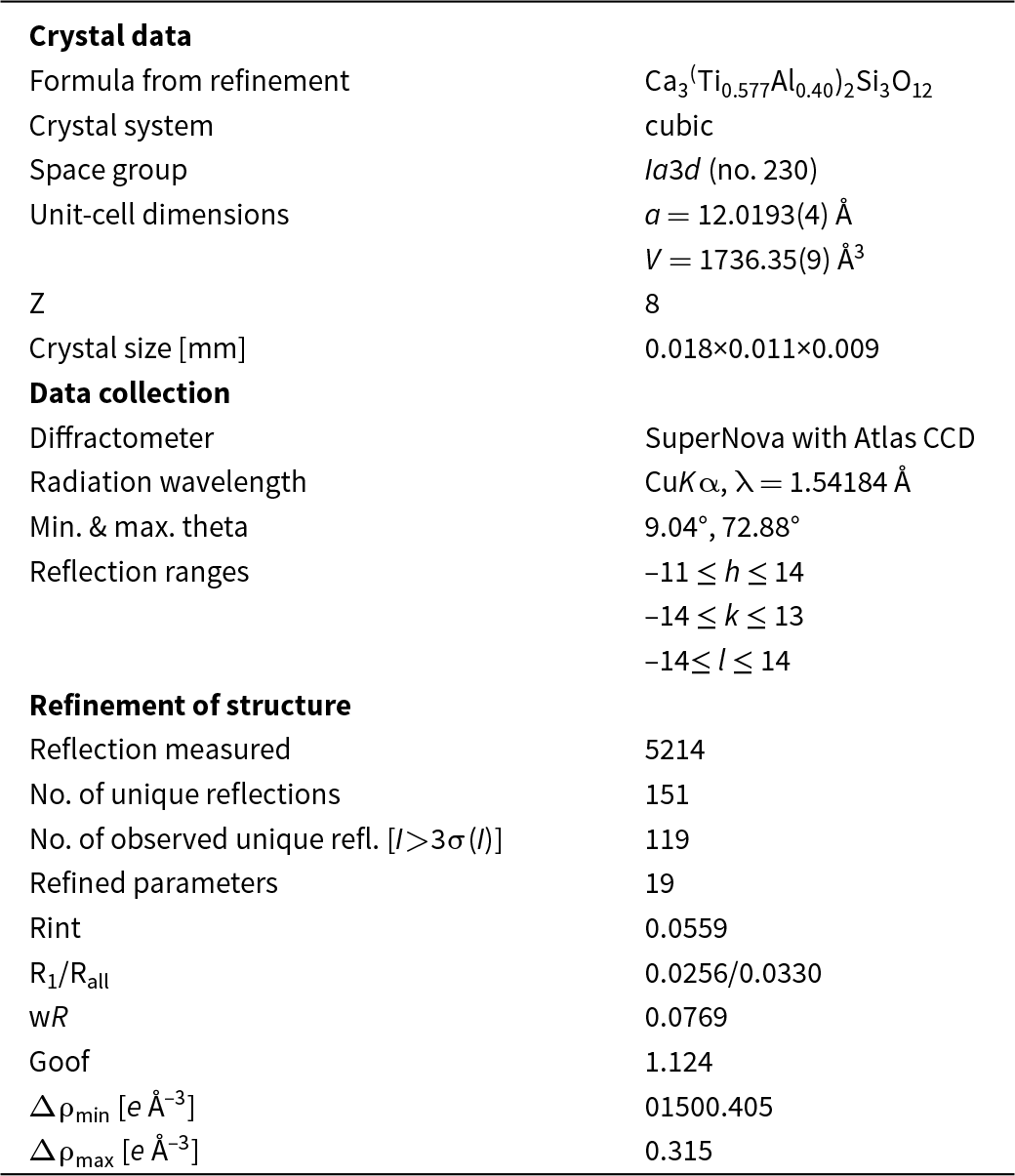
A fragment of the rubinite crystal 18 × 11× 9 µm in size from zone 4 was used for the single-crystal structure study. The average composition of this fragment according to the EDS data (mean of 12 analyses) corresponds to the formula (Ca2.99Mg0.01)Σ3(Ti3+0.78Al0.62Ti4+0.43Mg0.17)Σ2(Si2.74 Al0.26)Σ3O12, and in the ‘rubinite–grossular–hutcheonite+Mg-analogue of morimotoite’ diagram (Fig. 6), its composition plots in the rubinite field. The experimental details and refinement data for rubinite are summarised in Tables 4 and 5.
Crystal data and structure refinement details for rubinite

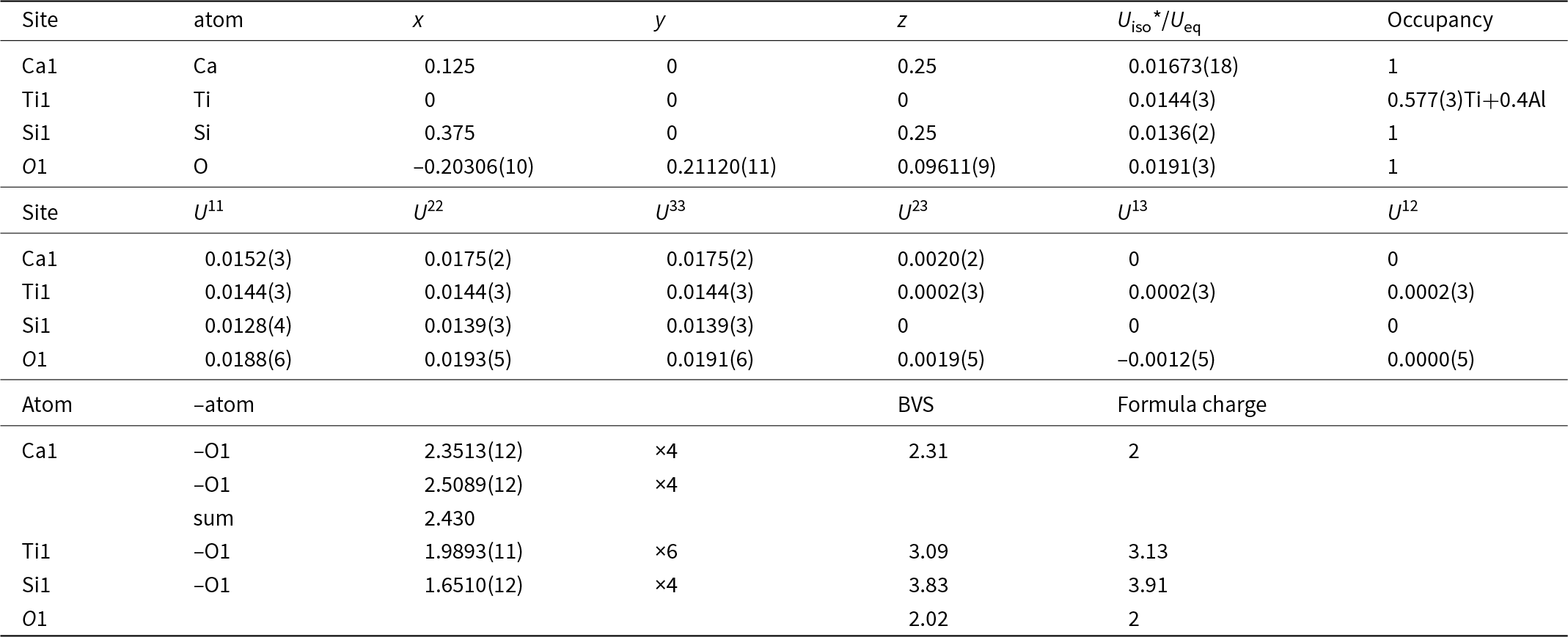
Atomic coordinates and isotropic displacement parameters (Å2), anisotropic displacement parameters (Å2), selected bond lengths (Å) and BVS calculation for rubinite
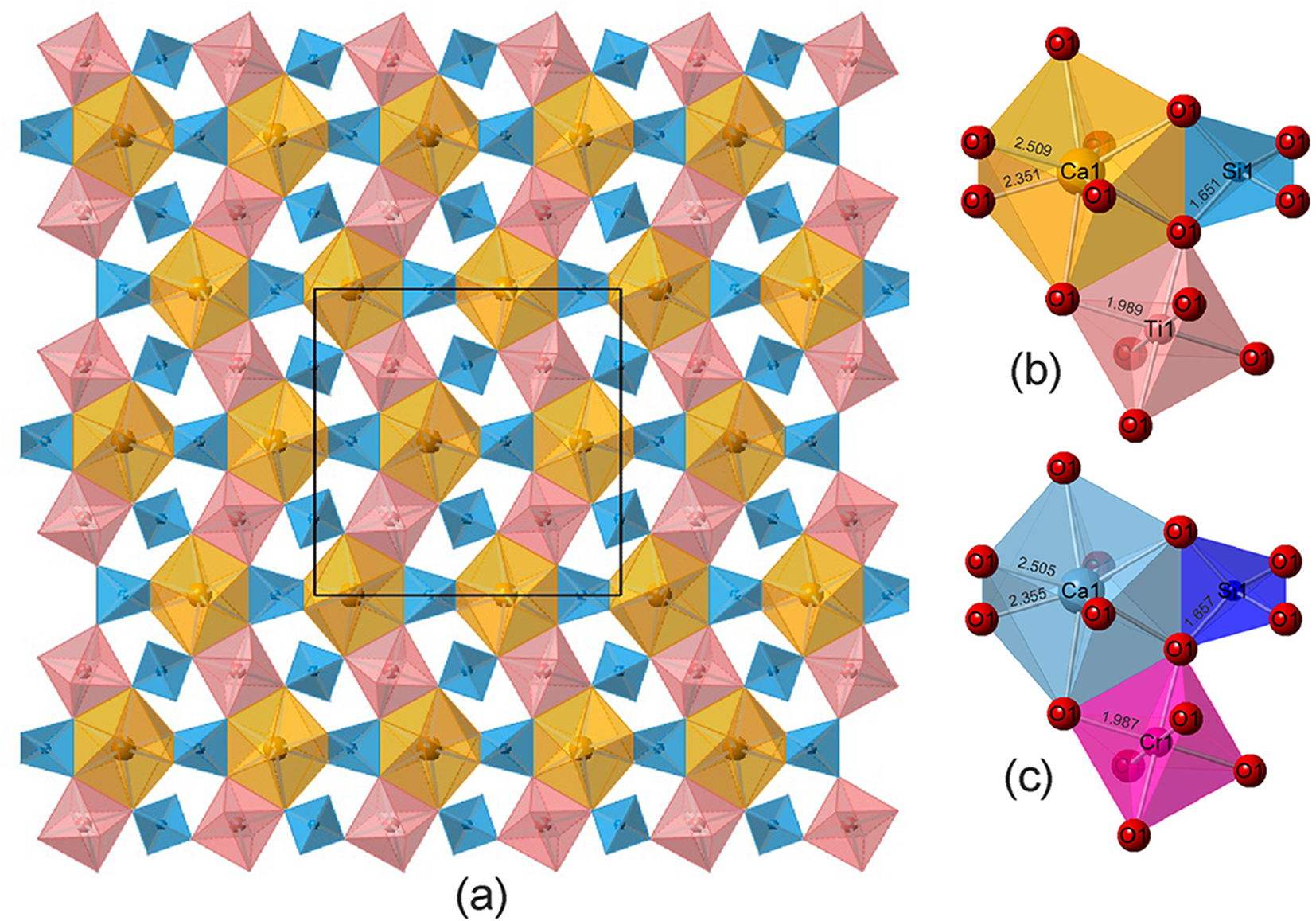
Rubinite belongs to the garnet supergroup. Its structure is formed by zigzag columns consisting of X polyhedra and Y octahedra with Si tetrahedra between them (Grew et al., Reference Grew, Locock, Mills, Galuskina, Galuskin and Halenius2013; Fig. 8). The 8-coordinate X site is occupied by Ca, mean Ca–O distance = 2.43 Å. At the tetrahedral Z site there is Si and a small amount of Al, Z–O distance = 1.651Å. At the octahedral Y site there are four types of cations, Ti3+0.78Al0.62Ti4+0.43Mg0.17, whose ionic radii are 0.670, 0.535, 0.605 and 0.72, respectively (Shannon, Reference Shannon1976). A theoretical calculation shows that a virtual cation with 18.4 e – should be at the Y-site and the Y–O distance is ∼1.98 Å. The results of the structure refinement give the close matches: 17.89 e – and Y–O = 1.989 Å (Table 4).
(a) A layer of the rubinite structure. Between the zigzag columns composed of Ca-polyhedra and Ti-octahedra there are TO4 tetrahedra. The black frame shows the unit cell. (b) and (c) Fragments of rubinite and Fe-free uvarovite structures, respectively, with similar interatomic distances. Images drawn using CrystalMaker 2.7 for Windows.

Terrestrial rubinite has an a unit-cell parameter equal to 12.0193(4) Å. The synthetic analogue, informally named ‘imanite’, whose structure was used as a model during the EBSD study of meteoritic rubinite, has a significantly larger parameter a = 12.1875(2) Å (Valldor et al., Reference Valldor, Uthe and Rückamp2011). A difference in the ionic radii Cr3+ = 0.615 and V3+ = 0.64 is insignificant, corresponding to a slight difference in the parameters of uvarovite Ca3Cr3+2Si3O12, a =11.988(1) Å, and goldmanite, Ca3V3+2Si3O12, a = 12.011(1) Å (Novak and Gibbs, Reference Novak and Gibbs1971). The radius of Ti3+ = 0.67 is slightly larger than the ionic radii of Cr3+ and V3+, but the parameter of the synthetic Ca3Ti3+2Si3O12 is unexpectedly large a = 12.1875 Å. We believe that the parameter a of rubinite from meteorites should be much smaller, as the Y–O distance ≈ 2.01 Å calculated using the composition (Ma et al., Reference Ma, Yoshizaki, Krot, Beckett, Nakamura, Nagashima, Muto, Ivanova and Ulyanov2025) is only slightly larger than the distance of Y–O ≈ 1.98 Å calculated for terrestrial rubinite.
We have studied Fe-free uvarovite from paralava containing native iron from the Hatrurim Complex; its empirical formula is Ca3.00(Cr3+1.24Al0.36Ti4+0.22V3+0.14Fe2+0.02Mg0.02)Σ2.00(Si2.85Al0.15)Σ3O12 and it has an a parameter equal to 12.0253(3) Å, which is close to that of terrestrial rubinite, and similar interatomic distances in polyhedra (Futrzyński et al., Reference Futrzyński, Juroszek, Skrzyńska, Vapnik and Galuskin2023; Fig. 8c,b), therefore the structure of uvarovite was used as a starting model during the structure refinement of rubinite.
It is interesting to note that there is overbonding at the Х site in rubinite (2.31 vu = valence units) (Table 4) and other calcium garnets, such as uvarovite (2.34), grossular (2.52 vu), schorlomite (2.27 vu) and goldmanite (2.37 vu), whereas for almandine (1.83 vu) insignificant underbonding of Fe2+ at the X site is observed (structural data from Novak and Gibbs, Reference Novak and Gibbs1971; Futrzyński et al., Reference Futrzyński, Juroszek, Skrzyńska, Vapnik and Galuskin2023). The geometry of Ca sites is determined by the 3D framework consisting of alternating Z-tetrahedra and Y-octahedra with shared corners.
The genesis of rubinite
The formation of rubinite is induced by high-temperature processes during the interaction of hot paralava generated at temperatures above 1200°C with the thermally altered fragments of carbonate-clay sedimentary rocks of the Ghareb Formation (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022, Reference Galuskin and Galuskina2023a). Geochemical data suggest that the carbonate-clay sedimentary rock was a protolith of the paralava that cemented its clinkerised fragments (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022). Preliminary thermal alteration of the sedimentary rocks caused graphitisation of fish bone remains and replacement of numerous framboids by hematite (Galuskin et al., Reference Galuskin, Galuskina, Vapnik, Kusz, Marciniak-Maliszewska and Zieliński2025). This was a necessary condition for the realisation of carbothermal reduced reactions at the contact of the hot paralava, leading to the mass formation of phosphide and native iron aggregates as well as osbornite (Galuskin et al., Reference Galuskin, Galuskina, Kamenetsky, Vapnik, Kusz and Zieliński2022, Reference Galuskin and Galuskina2023a). Interestingly, osbornite and phosphides were also detected as micrometric inclusions within small xenoliths (up to a few cm in size). Thermal alteration of the xenoliths caused their partial melting and contamination by the paralava, resulting in the formation of a paralava contact facies enriched in pseudowollastonite and phosphides (Galuskin et al., Reference Galuskin, Galuskina, Vapnik and Zieliński2023a). This process was accompanied by the formation of small droplets of melt within the xenolith, from which Ca-silicates (pseudowollastonite and cuspidine) and phosphides formed. The fine-grained porous clinker matrix of the xenolith was mainly represented by gehlenite and hatrurite aggregates, probably with an impurity of oldhamite, lime and minerals of the mayenite group. At the contact with the paralava, two processes related to xenolith melting are observed: contamination of the paralava with xenolith material and formation of a thin ‘melted zone’ of the xenolith itself (Fig. 1a,b). Paqueite, in contrast to rubinite, which is located in the central part of the xenolith, crystallised only in the xenolith melting zone (zone 3) and, in contrast to rubinite and its meteoritic analogue, does not contain any Ti3+ (Figs 1f; 2a,d; 5c; Table 3, analysis 1). Paqueite formation can be represented by the following reaction: CaSiO3 (pseudowollastonite) + Ca2Al2SiO7 (gehlenite) + 2TiO2 (rutile) = Ca3Ti4+(Al2Ti4+)Si2O14 (paqueite). The majority of rubinite grains form a rim on pseudowollastonite, probably at the moment of an abrupt increase in the porosity of the xenolith during the progressive process of clinkerisation. The high porosity of the rocks led to an increase in the activity of reducing gases and the formation of the Ti3+-bearing minerals osbornite and rubinite (Fig. 3d,e). Rubinite was formed by the reaction 3CaSiO3 (pseudowollastonite) + Ti2O3 (tistarite) = Ca3Ti3+2Si3O12 (rubinite). The reduction of Ti was a carbothermic reaction due to the carbon released as a result of the decomposition of the graphitised fish bone remains: 2Ti4+O2 + C/CO = Ti3+2O3 + CO/CO2. Thus, it is possible to follow a decrease in oxygen activity in the direction from the paralava (close to the Fe/FeO buffer, ΔIW ≈ 0) through the contact zone with phosphides (ΔIW ≈ –3 – –4) towards the central parts of the xenolith (ΔIW ≈ –7 – –8). The heterogeneity of the rocks and the involvement of gases would have disrupted this regularity, which is confirmed by the finding of rare aggregates of rubinite crystals with a grossular core formed at the boundary of the contact facies of the paralava contaminated by xenolith materials and the ‘melted zone’ of the xenolith (Fig. 5).
Rubinite found in situ from the phosphide-bearing breccia of the Hatrurim Complex is at present the only authentic silicate with trivalent titanium formed on Earth under super-reduced conditions.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2025.3.
Acknowledgements
The authors thank the three anonymous reviewers, the Technical Editor, the Associate Editor Robert Martin, and the Principal Editor Stuart Mills for remarks and comments that improved an earlier version of the manuscript. This paper is dedicated to Edward Sturgis Grew on the occasion of his 80th birthday.
Financial statement
Investigations were partially supported by the National Science Centre of Poland, Grant No. 2021/41/B/ST10/00130.
Competing interests
The authors declare none.