1. Introduction
The energy crisis and environmental pollution are among the most challenging global issues today (Rahimnejad et al., Reference Rahimnejad, Adhami, Darvari, Zirepour and Oh2015). Rapid population growth, coupled with accelerated industrialization, is expected to exacerbate these issues in the coming decades. Currently, energy consumption is increasing by around 1% annually in developed countries and approximately 5% each year in developing countries (Muneer et al., Reference Muneer, Asif and Munawwar2005). By 2050, global energy demand is projected to rise by an order of magnitude (Stambouli & Traversa, Reference Stambouli and Traversa2002). Developing and utilizing renewable energy sources offer a promising solution to meet rising energy demands and reduce pollution from fossil fuel combustion.
To meet the growing energy demands and address the challenges of global climate change, finding sustainable solutions that achieve carbon neutrality has become one of the most urgent tasks (L. Lu et al., Reference Lu, Guest, Peters, Zhu, Rau and Ren2018). Microbial electrochemical technologies (METs) exploit microorganisms as biocatalysts, coupling microbial metabolism with electrochemical reactions to enable the generation of bioelectricity, biofuels, hydrogen, and other value-added chemicals (K.-R. Kim et al., Reference Kim, Kang and Chae2017). Microbial fuel cells (MFCs) and microbial electrolysis cells (MECs) are representative examples of METs. In MFCs, the anodic biofilm serves as a catalyst to degrade organic pollutants and generate electrons, while the cathode typically undergoes a spontaneous redox reaction. MFCs can directly convert chemical energy into electrical energy, frequently being used to generate electricity from organic matter in wastewater treatment (Jadhav et al., Reference Jadhav, Park, Pandit, Yang, Ali Abdelkareem, Jang and Chae2022; Jiang et al., Reference Jiang, Li, Wang, Jiang, Song, Zhang and Zhu2020). MECs, derived from MFCs technology, utilize externally applied electrical energy to drive electrons from the anode to the cathode, enabling non-spontaneous reduction reactions. These reactions commonly include proton reduction for hydrogen gas production or the reduction of metal ions. The electrons are then involved in reduction reactions to produce value-added products like hydrogen or methane (Deng et al., Reference Deng, Wang, Ngo, Guo, You, Tang, Yu, Tang and Han2023; Song et al., Reference Song, Zhu, Fu, Chen, Liu, Zhang, Lin and Zhu2020). The main difference between MFCs and MECs lies in the need for external energy and the type and products of the cathode reactions. This difference makes MFCs ideal for energy recovery, whereas MECs are more appropriate for the electrochemical synthesis of specific target products (Saravanan et al., Reference Saravanan, Karishma, Kumar, Yaashikaa, Jeevanantham and Gayathri2023). Recently, microbial electrosynthesis systems (MESs) have emerged as promising METs. It uses anaerobic electroactive microorganisms as biocatalysts to electrochemically reduce CO2 into organic compounds, effectively storing excess electrical energy in the form of chemical bonds. Compared to traditional inorganic catalysts, microorganisms offer several advantages, including high product selectivity, long-term stability, low catalytic overpotential, and the capability to produce long-chain organic compounds (Jiang et al., Reference Jiang, Tian, Li, Xia, Song and Zhu2023; Sheldon & Woodley, Reference Sheldon and Woodley2018). Collectively, METs present a versatile and sustainable framework for integrating wastewater treatment with energy recovery and CO2 conversion.
Although METs have demonstrated high performance at the laboratory scale, their application in real-world environments remains limited. Unlike small-scale systems that utilize chemically pure substrates, large-scale MET devices typically rely on cost-effective and readily available wastewater, often supplemented with nutrients such as agarose or sodium acetate. The inherent complexity of these substrates fosters diverse microbial communities and intensifies interspecies competition. For example, in the anode chamber of an MFC, brewery wastewater supports Saccharomyces cerevisiae as the dominant species due to its robustness against environmental fluctuations and high physiological stability (Jatoi et al., Reference Jatoi, Ahmed, Bhutto and Shaikh2024). Similarly, Pseudomonas aeruginosa and Staphylococcus aureus exhibit strong tolerance to harsh conditions and are capable of effectively degrading acenaphthylene, phenanthrene, and chrysene, highlighting their potential for the breakdown of complex and polycyclic hydrocarbons (Khazaal & Ismail, Reference Khazaal and Ismail2023). These shifts in dominant microbial populations can substantially influence system performance, introducing variability that is difficult to manage.
In lab conditions, optimized operating parameters and control strategies allow METs to achieve superior performance in energy output and pollutant degradation (Logan & Rabaey, Reference Logan and Rabaey2012). However, numerous challenges emerge when scaling METs for industrial applications. In theory, large-scale devices face more complex challenges, including intricate electron transfer mechanisms, nonlinear relationships between substrate conversion and energy loss, and issues related to microbial succession during long-term operation, which remain unresolved (Omidi et al., Reference Omidi, Mashkour, Biswas, Garlapati, Singh, Rahimnejad and Pant2024). In practice, numerous challenges persist, including issues related to electrode scalability and stability, mass transfer limitations, fluid dynamics constraints, operational costs and economic viability, as well as environmental adaptability. Consequently, the power output from the anode and chemical production at the cathode remain insufficient for practical large-scale implementation (Butti et al., Reference Butti, Velvizhi, Sulonen, Haavisto, Oguz Koroglu, Yusuf Cetinkaya, Singh, Arya, Annie Modestra, Vamsi Krishna, Verma, Ozkaya, Lakaniemi, Puhakka and Venkata Mohan2016).
In recent years, researchers have undertaken diverse approaches to tackle these challenges. This review highlights recent advancements in scaling up METs, focusing on key challenges for successful implementation, such as reactor design, electrode engineering, and the optimization of operating conditions. Additionally, we present the major challenges and potential strategies for improving industrial-scale applications, offering guidance for efficient waste-to-energy conversion in future MET systems.
2. Scale-up of MFCs
MFCs are a specialized type of fuel cell typically consisting of an anode chamber, a cathode chamber, and a proton exchange membrane (F. Li et al., Reference Li, Sharma, Lei, Li and Zhou2010). The operation of MFCs relies on electrogenic microorganisms in the anode chamber that catalyze the oxidation of organic substrates, like sodium acetate or glucose. The oxidation process generates electrons and protons, with the electrons transferred from the cell to the anode. Simultaneously, protons migrate from the anode to the cathode chamber across the proton exchange membrane. As for the electron acceptors, like oxygen or ferricyanide, react with electrons and protons, converting the chemical energy of substrates into electrical energy (Santoro et al., Reference Santoro, Arbizzani, Erable and Ieropoulos2017).
MFCs provide an effective means for wastewater treatment, resource recovery, and electricity generation. However, the practical application is constrained by low power output and high costs (Tan et al., Reference Tan, Chong, Fang, Pan, Mohamad, Lim, Tiong, Chan, Huang and Yang2021). While recent studies have scaled up reactors to pilot levels, power output has not increased proportionally with the reactor size (Daud et al., Reference Daud, Daud, Bakar, Kim, Somalu, Muchtar, Jahim and Muhammed Ali2020). Recent efforts have aimed to bridge this gap by optimizing reactor designs, electrode materials, and operational conditions, transitioning from laboratory research to practical applications.
2.1. Reactor design
Scaling up MFC reactors is critical to meeting the demands of large-scale wastewater treatment and bioenergy production. Two common strategies for scaling up MFCs are to enlarge a single reactor unit or to stack multiple reactors in a modular system (Ge & He, Reference Ge and He2016). Enlarging a single MFC unit increases the surface area of electrodes and the proton exchange membrane, enabling a larger microbial community and thereby boosting power density and overall output (Logan et al., Reference Logan, Hamelers, Rozendal, Schröder, Keller, Freguia, Aelterman, Verstraete and Rabaey2006). During the scale-up process, however, mass transfer limitations and uneven mixing may lead to variations in pH and dissolved oxygen (DO) levels. Li et al. expanded a dual-chamber MFC to 2 L, with a microbial membrane separating the cathode and anode chambers, using real domestic wastewater as the substrate. The reactor maintained stable DO and pH levels between the chambers, achieving a final cathodic chemical oxygen demand (COD) concentration below 50 mg/L (Li Chao et al., Reference Li Chao, TIAN, Ravi Shanker, Wei-hua and Yu-jie2021). Single-chamber MFCs, in contrast, offer advantages like simpler structure, lower cost, reduced internal resistance, and higher mass transfer efficiency. Ana Carla Sorgato et al. (Reference Sorgato, Jeremias, Lobo and Lapolli2023) scaled up a single-chamber air-cathode MFC to 2 L, operating in continuous flow mode with synthetic wastewater. A scaled-up MFC could serve as a primary wastewater treatment unit, potentially converting wastewater treatment plants into renewable energy producers (Figure 2a, b).
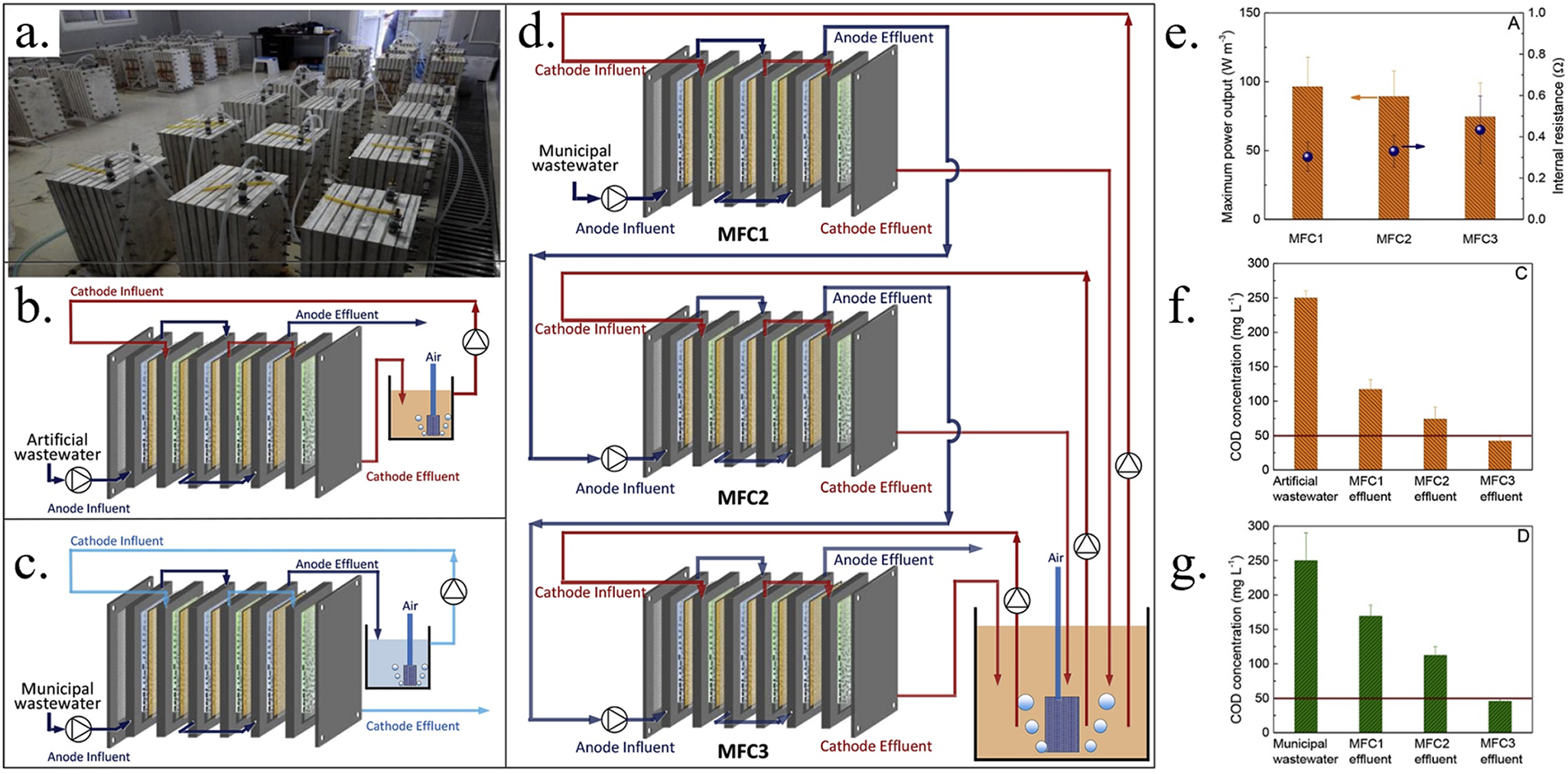
Scaling up MFCs by simply increasing the size of a single reactor is generally not feasible, as the increased distance between the anode and cathode electrodes inevitably increases internal resistance and reduces power generation. Furthermore, the output voltage of a single reactor is constrained by the presence of only one electrode pair, typically yielding less than 0.8 V (Liang et al., Reference Liang, Duan, Jiang, Zhang, Qiu and Huang2018). Consequently, modular MFC reactors are more effective for scaling up than enlarging individual reactors. Similar to chemical fuel cells, stackable MFC reactors connected in series or parallel can achieve the required voltage or current output while also increasing liquid volume capacity. In series connections, the anode of each cell connects to the cathode of the next, increasing the total output voltage. In parallel connections, all anodes and cathodes are interconnected, raising the total output current. Babanova et al. (Reference Babanova, Jones, Phadke, Lu, Angulo, Garcia, Carpenter, Cortese, Chen, Phan and Bretschger2020) developed a 110 L MFC system comprising 12 units connected in series, effectively treating wastewater from a small educational farm. The system ran continuously for over 200 days with a hydraulic retention time (HRT) of 4 hours, achieving up to 65% COD removal and a maximum treatment rate of 5.0 kg/m3 · COD day (Figure 2c, f). Liang et al. (Reference Liang, Duan, Jiang, Zhang, Qiu and Huang2018) scaled up a modular MFC system to 1000 L, containing 50 MFC modules, each with a capacity of 20 L, for treating municipal wastewater. Post-treatment, COD levels in low-COD and high-COD wastewater remained below 50 mg/L, with a removal efficiency of 70–90%, consistently meeting the Class A discharge standard for national wastewater treatment plants (Figure 1). Stacking multiple MFC modules is more practical, providing greater overall capacity while allowing independent operation and maintenance of each reactor. Series connections are suited for high-voltage applications, while parallel connections are ideal for high-current needs (M. Lu et al., Reference Lu, Chen, Babanova, Phadke, Salvacion, Mirhosseini, Chan, Carpenter, Cortese and Bretschger2017).
(a) Photograph of 1000 L modularized MFC system consisted of 50 modules. (b) Water flow connection in a single MFC module for treating artificial wastewater. (c) Water flow connection in a single MFC module for treating municipal wastewaters from Xiao Jiahe MWTP with low COD concentration. (d) Water flow connection for treating the municipal wastewater from Yong Feng MWTP with high COD concentration. Every three MFC modules were assembled to achieve step-wise COD removal. (e) The maximum power output and internal resistance of three MFC modules connected as an assembly to treat artificial wastewater. The COD concentration in the effluent of three MFC modules when treating (f) artificial wastewater, (g) municipal wastewater from Yong Feng MWTP (Liang et al., Reference Liang, Duan, Jiang, Zhang, Qiu and Huang2018).

In recent years, researchers have concentrated on integrating MFC scale-up systems with existing technologies to overcome scale-up challenges. For instance, integrating electronic devices such as DC-DC boosters can enhance current and power output, addressing the relatively low power generation caused by series and parallel configurations (Mukherjee et al., Reference Mukherjee, Patel, Zaveri, Shah and Munshi2022). Furthermore, an appropriate power management system can optimize the collection of generated electricity. Beyond electronic integration, MFCs can also be coupled with modern wastewater treatment technologies. For instance, the anaerobic ultrafiltration membrane biofuel cell incorporates an ultrafiltration membrane in the anode chamber, enhancing wastewater treatment efficiency by reducing the system's footprint and facilitating bioenergy recovery (Bhowmick et al., Reference Bhowmick, Ghangrekar, Zekker, Kibena-Põldsepp, Tammeveski, Wilhelm and Banerjee2022).
2.2. Electrode design
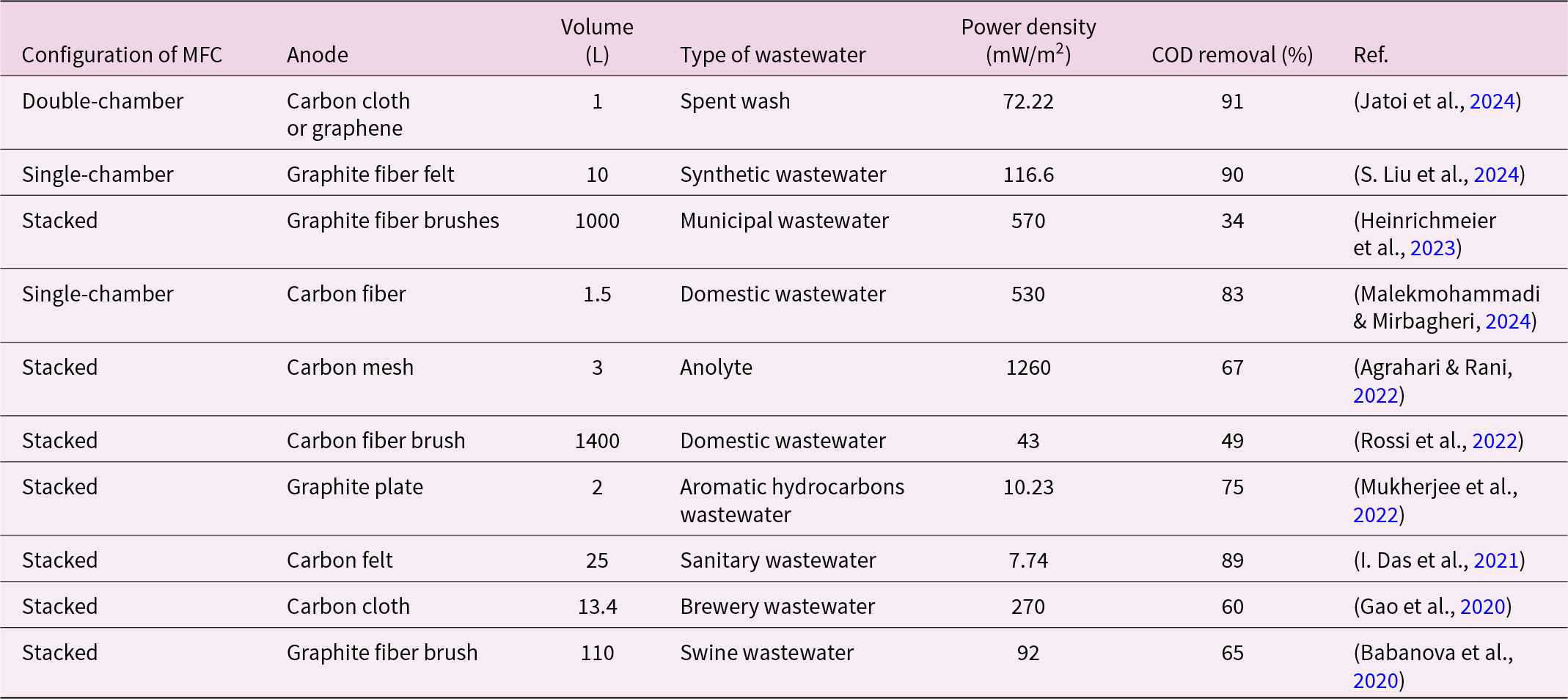
Anodes serve as both attachment sites for electroactive microbial communities and crucial points for electron transfer. High-performance anode materials are defined by excellent conductivity, large surface area, high biocompatibility, and cost-effectiveness (Y. Wang et al., Reference Wang, Hu, Dong and Wang2023). Stainless steel is regarded as a cost-effective anode material for scaling up MFCs due to its low cost. Researchers have improved the biocompatibility and surface area of stainless steel through surface treatments, making it an economical and practical choice for large-scale MFCs (Wu et al., Reference Wu, Tan, Chen, Yang, Hou, Lei and Li2021). Heat treatment of stainless steel wool forms an iron oxide layer on the surface, significantly enhancing electroactive biofilm formation and achieving a current density seven times higher than that of untreated electrodes. Scaling up heat-treated stainless steel wool to 150 cm2 yielded similar current densities (1.5 mA/cm2) on larger electrodes, confirming the material's scalability (Vilajeliu-Pons et al., Reference Vilajeliu-Pons, Puig, Salcedo-Dávila, Balaguer and Colprim2017). However, complex modification processes and long-term corrosion issues limit the use of metal anodes in large-scale MFCs and real wastewater treatment. Carbon-based materials, such as graphite, carbon cloth, carbon paper, carbon nanotubes, and graphene, offer significant advantages over stainless steel, including excellent corrosion resistance, superior conductivity, high specific surface area, chemical stability, versatility, affordability, and environmental sustainability (Zhang et al., Reference Zhang, Liu, Wang and Fu2022). These materials are commonly employed as anodes in MFCs, particularly in larger systems (>1 L). Table 1 summarizes representative MFC systems applied to wastewater treatment and electricity generation, comparing reactor configurations, anode materials, operating volumes, wastewater types, power densities, and COD removal efficiencies reported at different scales.
Wastewater treatment and electricity generation by MFCs

However, the power generation efficiency and economic viability of these anode materials vary considerably. Therefore, the selection of an appropriate anode material must be guided by specific application requirements, taking into account key factors such as material cost, electrochemical stability, and conductivity. Ding et al. conducted a study to determine the optimal and most cost-effective anode material by evaluating carbon brush, carbon cloth, and graphite felt anodes in a 6 L air-cathode MFC. The results showed that the type of anode material did not significantly impact the startup process of compact MFCs. However, regarding electricity generation, MFCs with carbon brush anodes achieved the highest power densities in synthetic wastewater and slaughterhouse wastewater, at (56.3 ± 1.8) W/m3 and (19.5 ± 0.8) W/m3, respectively. MFCs with carbon cloth anodes followed, yielding (46.0 ± 1.7) W/m3 and (16.9 ± 0.6) W/m3, while those with graphite felt anodes showed the lowest performance at (40.8 ± 1.5) W/m3 and (11.9 ± 0.5) W/m3. The COD removal rates remained consistently high, around 90%, across different anodes, regardless of the type of wastewater (synthetic or slaughterhouse). The carbon brush anode MFC demonstrated the highest economic benefits, with (3.44 ± 0.08) mW/unit in sodium acetate and (0.97 ± 0.05) mW/unit in slaughterhouse wastewater, surpassing those of carbon cloth and graphite felt MFCs by 18.6%, 12.8%, 38.7%, and 80%, respectively (Ding et al., Reference Ding, Yu, Chen and Cheng2017).
In pilot-scale MFCs, two main cathode configurations are used for oxygen supply in the oxygen reduction reaction: air cathodes and liquid cathodes. Studies show that air cathode MFCs achieve higher power densities compared to liquid cathode systems (Rossi & Logan, Reference Rossi and Logan2022). The additional liquid chamber in liquid cathode MFCs increases internal resistance, reducing power output, while the aeration process requires significant energy (Rossi et al., Reference Rossi, Jones, Myung, Zikmund, Yang, Gallego, Pant, Evans, Page, Cropek and Logan2019). The limitations of liquid cathodes have hindered their scalability in MFCs, prompting researchers to focus on air cathodes.
Ghadge and Ghangrekar (Reference Ghadge and Ghangrekar2015) reported that in an MFC with a 26 L working volume, the air cathode maintained high current output over 14 months and effectively mitigated cathode fouling. Rossi et al. (Reference Rossi, Hur, Page, Thomas, Butkiewicz, Jones, Baek, Saikaly, Cropek and Logan2022) built the largest air cathode MFC to date, with a total volume of 1400 L, for domestic wastewater treatment. Effluent from this MFC underwent further treatment in a biological filter to reduce organic content. In the combined MFC/biofilter process, up to 91 ± 6% of the COD and 91% of the BOD5 were removed as well as certain bacteria (E. coli, 98.9%; fecal coliforms, 99.1%) (Figure 2d, e).
(a) MFC setup. (b) Average removal of COD at different HRTs (18, 8, and 4 h) (Sorgato et al., Reference Sorgato, Jeremias, Lobo and Lapolli2023). (c) COD concentration of the influent, effluent, and the COD removal for the pilot (Babanova et al., Reference Babanova, Jones, Phadke, Lu, Angulo, Garcia, Carpenter, Cortese, Chen, Phan and Bretschger2020). (d) COD consumption in the MFC and BF over time (weeks 6–19). (e) Concentration of ammonia in the MFC influent, effluent, and post-BF treatment (Rossi et al., Reference Rossi, Hur, Page, Thomas, Butkiewicz, Jones, Baek, Saikaly, Cropek and Logan2022). (f) COD treatment rate over time (Babanova et al., Reference Babanova, Jones, Phadke, Lu, Angulo, Garcia, Carpenter, Cortese, Chen, Phan and Bretschger2020).

2.3. Operating conditions
The selection of inoculum directly influences the dominant microbial species and electrochemical activities in the system. While pure cultures of electrogenic bacteria (EAB) can achieve high bioelectricity generation, mixed microbial consortia are generally more efficient in MFCs (Singh & Kaushik, Reference Singh and Kaushik2021). The effectiveness stems from the stability and syntrophic interactions within the microbial community, resulting in higher power densities than those achieved by the most efficient pure-culture EAB. Various microbial inocula sources, such as sludge, fresh sediments, dye-processing wastewater, marine sediments, manure and compost, have successfully established electroactive microbial communities in MFCs. Soil and sludge are preferred in large-scale MFCs because of their easy accessibility and handling. Logroño et al. (Reference Logroño, Echeverría, Recalde and Graziani2015) utilized undisturbed soil from the Ecuadorian Andes highlands and organic solid waste as inoculum, achieving an average voltage output of 317 mV in a 12 L MFC. Yoshizawa et al. (Reference Yoshizawa, Miyahara, Kouzuma and Watanabe2014) compared the effects of paddy soil and aerobic sludge as inocula on wastewater treatment performance and microbial community composition in a 1 L MFC reactor. The MFC inoculated with soil achieved a COD removal efficiency of 75–80%, a maximum power density of 150–200 mW/m2, and a coulombic efficiency (CE) of 20–30%. These values were comparable to those observed in the MFC inoculated with aerobic sludge. Comparative analysis revealed significant differences between the anode communities enriched from aerobic sludge and those derived from soil, suggesting that distinct community structures can yield similar reactor performance.
HRT significantly affects residual substrate concentrations and DO levels in the reactor, which in turn influences COD removal rates and power output (Zhenglong Li et al., Reference Li, Yao, Kong and Liu2008). In a large-scale MFC with a 720 L working volume, Das et al. (Reference Das, Ghangrekar, Satyakam, Srivastava, Khan and Pandey2020) showed that setting the HRT to 18 hours achieved a COD removal efficiency of 78.45 ± 19.12%, with a final effluent COD of 375 ± 118 mg/L. Increasing the HRT to 36 hours enhanced COD removal efficiency to 87.29 ± 7.28% and reduced the effluent COD to 303 ± 50 mg/L. H. Liu et al. (Reference Liu, Ramnarayanan and Logan2004) studied a single-chamber MFC with eight graphite anodes and one air cathode for domestic wastewater treatment, finding that power output was inversely proportional to HRT. This inverse relationship is due to the increased substrate concentration from reducing the HRT, which elevates bacterial substrate consumption rates and enhances electricity generation. However, continuously reducing the HRT may decrease voltage and power density. This decline results from increased DO concentration in the anode chamber, raising the oxidation-reduction potential and lowering voltage, particularly when the influent is not deoxygenated (Zhenglong Li et al., Reference Li, Yao, Kong and Liu2008). Thus, an optimal HRT is essential to balance COD removal efficiency and power output in MFCs.
3. Scale-up of MECs
MECs and MFCs are closely related in their principles, structure, and applications, both depending on electroactive microorganisms at the anode to oxidize organic matter, releasing electrons and protons (Chaijak et al., Reference Chaijak, Sato, Paucar, Lertworapreecha and Sukkasem2019). In MFCs, however, the electrons generated at the anode are transported to the cathode through an external circuit, typically resulting in the production of electricity and water. In MECs, electrons produced at the anode reduce protons at the cathode under an external power source, generating hydrogen or other valuable chemicals (S. Wang et al., Reference Wang, Gariepy, Adekunle and Raghavan2024). Consequently, MECs are primarily employed for the production of hydrogen or other reduced products.
Hydrogen is considered a promising alternative to fossil fuels because of its clean combustion and high energy content (J. Zhou et al., Reference Zhou, Ji, Cao, He, Fan and Yuan2023). Traditional industrial methods of hydrogen production are energy-intensive and expensive. As a biological approach, MECs offer several advantages for hydrogen production: MECs can use any biodegradable organic material as a substrate and achieve much higher yields, rates, and energy efficiency than other biological methods (Das & Peu, Reference Das and Peu2022). Furthermore, MEC reactors have lower manufacturing costs because they can be assembled from low-cost, easily scalable materials like polyacrylate and polycarbonate. These benefits make MECs an ideal option for biological hydrogen production (Gupta & Parkhey, Reference Gupta and Parkhey2015).
One of the key research focuses in scaling up MECs is minimizing potential (ΔV) losses to achieve high hydrogen production rates with minimal energy input. While theoretically, MECs require an order of magnitude less energy for hydrogen production than water electrolysis under similar conditions, the actual energy required in practice is significantly higher than the theoretical value (Chaijak et al., Reference Chaijak, Sato, Paucar, Lertworapreecha and Sukkasem2019). This discrepancy results from significant impacts of reactor design, electrode overpotentials, and operational conditions on voltage losses in MECs (Singh et al., Reference Singh, Miller, Wang and Liu2021).
3.1. Reactor design
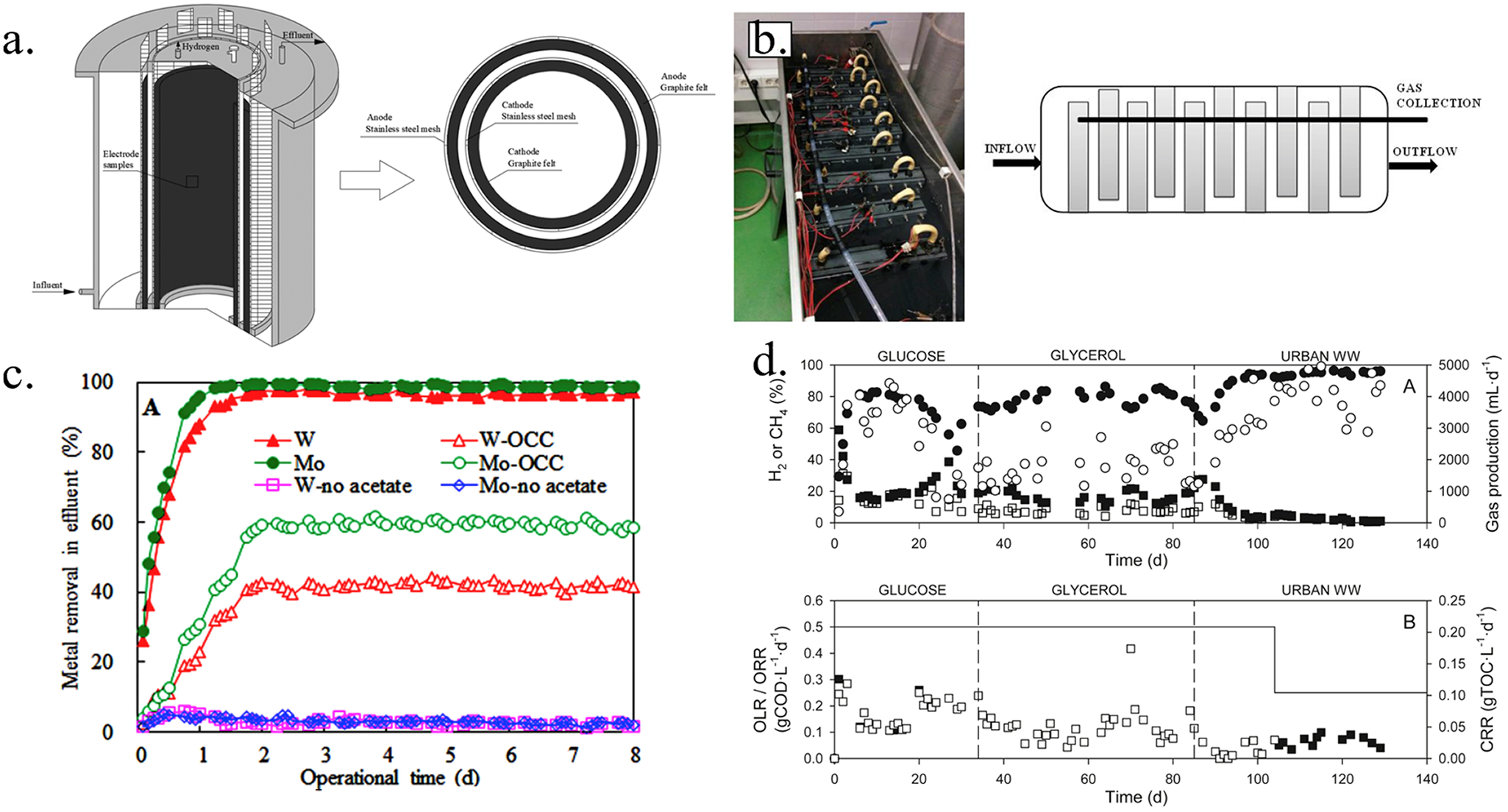
Membrane-less single-chamber designs significantly reduce internal resistance, enhancing electrochemical performance and lowering material and assembly costs, which makes them more economical for large-scale applications (Zeppilli et al., Reference Zeppilli, Cristiani, Fazi, Majone and Villano2022). Single-chamber MECs have been applied to treat straw hydrolysate, achieving high CE (88%) and cathodic hydrogen recovery (58%) (Gupta & Parkhey, Reference Gupta and Parkhey2015). Additionally, large-scale single-chamber MECs have shown effectiveness in removing metal contaminants. For example, Huang et al. (Reference Huang, Tian, Pan, Shan, Shi and Logan2019) utilized a 40 L cylindrical single-chamber MEC, achieving tungsten removal rates of 97–98% and molybdenum removal rates of 98–99% (Figure 4a, c). Despite the multiple advantages of single-chamber MECs, the coexistence of methanogens and hydrogen-producing bacteria in the anode can lead to the conversion of cathodic hydrogen into methane, reducing the proportion of hydrogen (Luo et al., Reference Luo, Jain, Aguilera and He2017). Electrochemical impedance spectroscopy reveals that single-chamber systems exhibit higher diffusion resistance, leading to a significant decline in MECs performance (Escapa et al., Reference Escapa, San-Martín, Mateos and Morán2015).
Most large-scale MEC designs employ a dual-chamber configuration to effectively separate products, improving gas purity and system stability (S. Wang et al., Reference Wang, Gariepy, Adekunle and Raghavan2024). The ion exchange membrane regulates ion transport between the anode and cathode, maintains pH balance, and prevents hydrogen from back-diffusing, where it could react with harmful substances and cause cross-contamination (Butti et al., Reference Butti, Velvizhi, Sulonen, Haavisto, Oguz Koroglu, Yusuf Cetinkaya, Singh, Arya, Annie Modestra, Vamsi Krishna, Verma, Ozkaya, Lakaniemi, Puhakka and Venkata Mohan2016). However, as the volume of traditional dual-chamber reactors increases, mixing efficiency decreases, and the oxidation rate at the anode worsens, requiring a larger volume or higher HRT to achieve the same treatment efficiency. Conventional stirring or circulation pumps can enhance turbulence and alleviate mass transfer limitations but may disrupt the system's energy balance (Fujian Li & Cheng, Reference Li and Cheng2018). Heidrich et al. proposed a box-type reactor design with dual-chamber units alternately positioned on both sides of the reactor, inducing a zigzag flow of wastewater between units (Figure 4b, d) (Baeza et al., Reference Baeza, Martínez-Miró, Guerrero, Ruiz and Guisasola2017). Based on the box design, six identical box units were placed in a 120 L rectangular polypropylene tank to treat domestic wastewater and produce nearly pure hydrogen (100 ± 6.4%) for over 3 months (Heidrich et al., Reference Heidrich, Dolfing, Scott, Edwards, Jones and Curtis2013). Cotterill et al. (Reference Cotterill, Dolfing, Jones, Curtis and Heidrich2017) enhanced this box-type design by incorporating two anodes in each module, which reduced internal resistance. When treating domestic wastewater in a 175 L anode chamber, the average COD removal rate was 63.5%, with an average effluent quality of 124.7 mg/L COD.
In dual-chamber MECs, the membrane is a critical component. Its design and selection are pivotal for product separation, improving gas purity, and enhancing overall system performance. However, membranes in dual-chamber systems face several limitations. Over time, contaminants may accumulate on their surface, diminishing mass transfer capacity and adversely impacting long-term system operation (Radhika et al., Reference Radhika, Shivakumar, Kasai, Koutavarapu and Peera2022). Additionally, reduced membrane selectivity can lead to hydrogen mixing with other gases, such as oxygen and carbon dioxide, adversely affecting gas purity. Therefore, membrane selection must balance gas purity improvement, energy loss minimization, and membrane fouling reduction (Fathima et al., Reference Fathima, Ilankoon, Zhang and Chong2024).
3.2. Electrode design
Researchers have recently developed a range of cathode materials for MECs. Stainless steel is favored for scaled-up MEC systems due to its balanced cost and hydrogen evolution efficiency (Suharto et al., Reference Suharto, Satar, Daud, Somalu and Hong2022). To enhance electrode performance and efficiency, stainless steel has been engineered into mesh (Rader & Logan, Reference Rader and Logan2010), wool (Elizabeth S. Heidrich et al., Reference Heidrich, Edwards, Dolfing, Cotterill and Curtis2014), and velvet forms (E. S. Heidrich et al., Reference Heidrich, Dolfing, Scott, Edwards, Jones and Curtis2013). Platinum (Pt) and nickel (Ni), compared to stainless steel (SS), show superior conductivity and catalytic activity. Pt-coated titanium (Ti) mesh as a cathode significantly reduces reactor internal resistance (Guo et al., Reference Guo, Prévoteau and Rabaey2017). Ni provides a cost-effective alternative to platinum and is widely available, essential for producing stainless steel and batteries (Kadier et al., Reference Kadier, Kalil, Abdeshahian, Chandrasekhar, Mohamed, Azman, Logroño, Simayi and Hamid2016). Guerrero-Sodric et al. (Reference Guerrero-Sodric, Baeza and Guisasola2024) developed a Ni foam cathode that significantly increased hydrogen production and energy efficiency, achieving the highest hydrogen yield reported for a pilot-scale MEC (19.07 ± 0.46 L/(m2 · d)), a threefold increase over the prior stainless steel wool cathode (Figure 3a, b). However, high-performance electrode materials are costly and their performance degrade over time (M. Zhou et al., Reference Zhou, Wang, Hassett and Gu2013). Biocathodes offer a promising alternative to traditional abiotic cathodes by using electroactive bacteria as catalysts for proton reduction to hydrogen (Zeppilli et al., Reference Zeppilli, Cristiani, Fazi, Majone and Villano2022). Biocathodes achieve electrochemical performance comparable to noble metal catalysts while offering long-term stability and simpler manufacturing. L. Wang et al. (Reference Wang, Long, Liang, Xiao and Liu2021) constructed four biocathodes enriched with electroactive microbial films for hydrogen evolution reactions in a 10 L single-chamber MEC, achieving a current density of 16.5 A/m2 and a hydrogen production efficiency of 91% (Figure 3d, e).
(a) Schematic representation of the cathode configurations tested. (b) Average current density (j) and specific hydrogen production over time in each configuration for the continuous operation using diluted industrial wastewater (Guerrero-Sodric et al., Reference Guerrero-Sodric, Baeza and Guisasola2024). (c) Average key performance indices, applied potential, and total gas production at different fixed intensities during the continuous operation with synthetic wastewater (Guerrero-Sodric et al., Reference Guerrero-Sodric, Baeza and Guisasola2023). Schematic of (d) reactor diagram, biocathode assembly, and bioanode assembly and current density increases of (e) biocathodes during the enrichment phase (L. L. Wang et al., Reference Wang, Long, Liang, Xiao and Liu2021). (f) Wastewater temperature (dashed line) and the energy recovery (solid line) trend for the MEC reactor throughout the year of operation, values represent monthly averages (Elizabeth S. Heidrich et al., Reference Heidrich, Edwards, Dolfing, Cotterill and Curtis2014).

(a) The schematic of cylindrical single-chamber MEC reactor (Huang et al., Reference Huang, Tian, Pan, Shan, Shi and Logan2019). (b) Image of the pilot-scale MEC and diagram of the wastewater flow as viewed from above (Baeza et al., Reference Baeza, Martínez-Miró, Guerrero, Ruiz and Guisasola2017). (c) W and Mo removal in the presence of circuit current, or in the controls of open circuit conditions (OCC) or in the absence of either metals or organics (W:Mo:acetate = 0.5:1.0:24 mM, HRT: 2 d) (Huang et al., Reference Huang, Tian, Pan, Shan, Shi and Logan2019). (d) Experimental profiles during the continuous operation of the pilot plant over time. A: hydrogen percentage (●), methane percentage (■), daily hydrogen production (○), daily methane production (□). B: organic loading rate (OLR, solid line), organic removal rate (ORR, □), carbon removal rate (CRR, ■) (Baeza et al., Reference Baeza, Martínez-Miró, Guerrero, Ruiz and Guisasola2017).

Anode materials for scaled-up MECs systems typically include low-cost, high-performance options like carbon or graphite felt, which demonstrate excellent biomass adsorption after heat treatment. Thermal treatment enhances the biomass adsorption capabilities, making them both technically superior and economically viable (Baeza et al., Reference Baeza, Martínez-Miró, Guerrero, Ruiz and Guisasola2017; Guerrero-Sodric et al., Reference Guerrero-Sodric, Baeza and Guisasola2023).
3.3. Operating conditions
High COD concentrations enable higher current densities, making high-organic-content wastewater a promising feedstock for MECs by providing abundant electron donors for hydrogen production (Guerrero-Sodric et al., Reference Guerrero-Sodric, Baeza and Guisasola2024). Guerrero-Sodric et al. (Reference Guerrero-Sodric, Baeza and Guisasola2023) tested three types of wastewater in a 135 L MEC: synthetic wastewater (900 mg/L COD), municipal wastewater (200 mg/L COD), and acetate-amended municipal wastewater (1000 mg/L COD). The system fed with synthetic wastewater achieved the highest current density (1.23 A/m2) and hydrogen production (0.1 m3/(m3 · d)) reported for pilot-scale MECs. Conversely, low-COD municipal wastewater limited system performance (Figure 3c). Baeza et al. (Reference Baeza, Martínez-Miró, Guerrero, Ruiz and Guisasola2017) operated a 130 L MEC with various substrates, including glucose, diluted crude glycerol, and real municipal wastewater. The best results were obtained with real municipal wastewater, yielding over 4 L/d of hydrogen at 95% purity.
Temperature strongly affects microbial metabolism and electrochemical kinetics. Moderate temperature increases enhance electron transfer, current density, and product formation, whereas excessive heating can denature enzymes and inhibit microbial activity (Faraghiparapari & Zengler, Reference Faraghiparapari and Zengler2017). Evaluating long-term MEC operation at ambient temperatures is therefore critical for practical wastewater treatment. Heidrich et al. (Reference Heidrich, Edwards, Dolfing, Cotterill and Curtis2014) reported a 100 L MEC operated for 12 months on raw domestic wastewater at 1–22°C, producing an average of 0.6 L/d H2 with 48.7% energy recovery, demonstrating sustained hydrogen generation under low-temperature conditions (Figure 3f). In contrast, operation near optimal temperatures accelerates microbial enrichment and reaction rates. Cusick et al. constructed a 1000 L MEC designed a 1000 L MEC for treating brewery wastewater and hydrogen production. By increasing the wastewater temperature to 31 ± 1°C, a current density of 7.4 A/m3 and a hydrogen production rate of 0.19 ± 0.04 L/d were achieved (Babanova et al., Reference Babanova, Jones, Phadke, Lu, Angulo, Garcia, Carpenter, Cortese, Chen, Phan and Bretschger2020). Furthermore, the selection of HRT impacts organic removal rates and gas production efficiency in MEC systems. Studies indicate that reducing HRT enhances hydrogen production but may decrease COD removal rates (Gil-Carrera et al., Reference Gil-Carrera, Escapa, Mehta, Santoyo, Guiot, Morán and Tartakovsky2013).
4. Scale-up of MESs
Discovered in 2010, MES is an emerging technology that uses microorganisms as cathodic catalysts to electrochemically reduce CO2 into biofuels and biochemical products (Nevin Kelly et al., Reference Nevin Kelly, Woodard Trevor, Franks Ashley, Summers Zarath and Lovley Derek2010). In a typical two-chamber MES system, the anode decomposes water or organic matter to produce H⁺ and electrons that facilitate the microbial reduction of CO2 in the cathode chamber, forming products like methane and acetate (Tanguay-Rioux et al., Reference Tanguay-Rioux, Nwanebu, Thadani and Tartakovsky2023). MESs are considered one of the most promising technologies for CO2 utilization and resource recovery; however, a significant bottleneck for practical application is relatively low current density, leading to low productivity (Jourdin et al., Reference Jourdin, Sousa, Stralen and Strik2020).
Traditional MES reactors rely on cathodic biofilms, where current density depends on the biomass quantity and activity within the biofilm (Jourdin, Freguia, et al., Reference Jourdin, Freguia, Flexer and Keller2016). While laboratory-scale MES reactors have achieved current densities of 10–100 A/m2, practical applications require much higher densities, exceeding 500–1000 A/m2 (Jourdin et al., Reference Jourdin, Sousa, Stralen and Strik2020). Studies have shown that forming cathodic biofilms is time-consuming, and the resulting current density is relatively low. Therefore, strategies to enhance catalytic activity by overcoming the limitations of dense biofilms are preferable (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022).
H2 mediated extracellular electron transfer is crucial in biofilm-driven MES reactors. When H2 is used as an electron mediator, the reactor can operate without biofilms (Xiu et al., Reference Xiu, Yao, Wu, Huang, Yang, Huang, Lei, Li and Hou2019). Researchers have recently developed MES reactors using suspended microorganisms and in situ electrolytically generated H2 for CO2 conversion. This approach allows higher current densities at the cathode without biofilm constraints (Jourdin, Lu, et al., Reference Jourdin, Lu, Flexer, Keller and Freguia2016). However, the low solubility of H2 necessitates that in situ generated H2 is fully utilized by suspended microorganisms to maintain high CE at elevated current densities (Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023).
4.1. Reactor design
In early MES studies, H-type reactors were widely used, with anode and cathode chambers separated by a cation exchange membrane to facilitate proton transfer from the anode to the cathode, thus maintaining the anaerobic conditions in the anode chamber (M. Kim et al., Reference Kim, Li, Song, Lee and Kim2022; Tanguay-Rioux et al., Reference Tanguay-Rioux, Nwanebu, Thadani and Tartakovsky2023; Zou et al., Reference Zou, Hasanzadeh, Khataee, Yang, Xu, Angelidaki and Zhang2021). However, the H-type reactor design has several limitations that make it unsuitable for scale-up. For example, the large distance between the cathode and anode leads to high internal resistance and affecting input voltage. Furthermore, this reactor type has low electrode and membrane surface area-to-electrolyte ratios and a mixing dead zone near the membrane. Additionally, these designs difficult to assemble and handle, with a high risk of leakage over time (Z. Liu et al., Reference Liu, Xue, Cai, Cui, Patil and Guo2023).
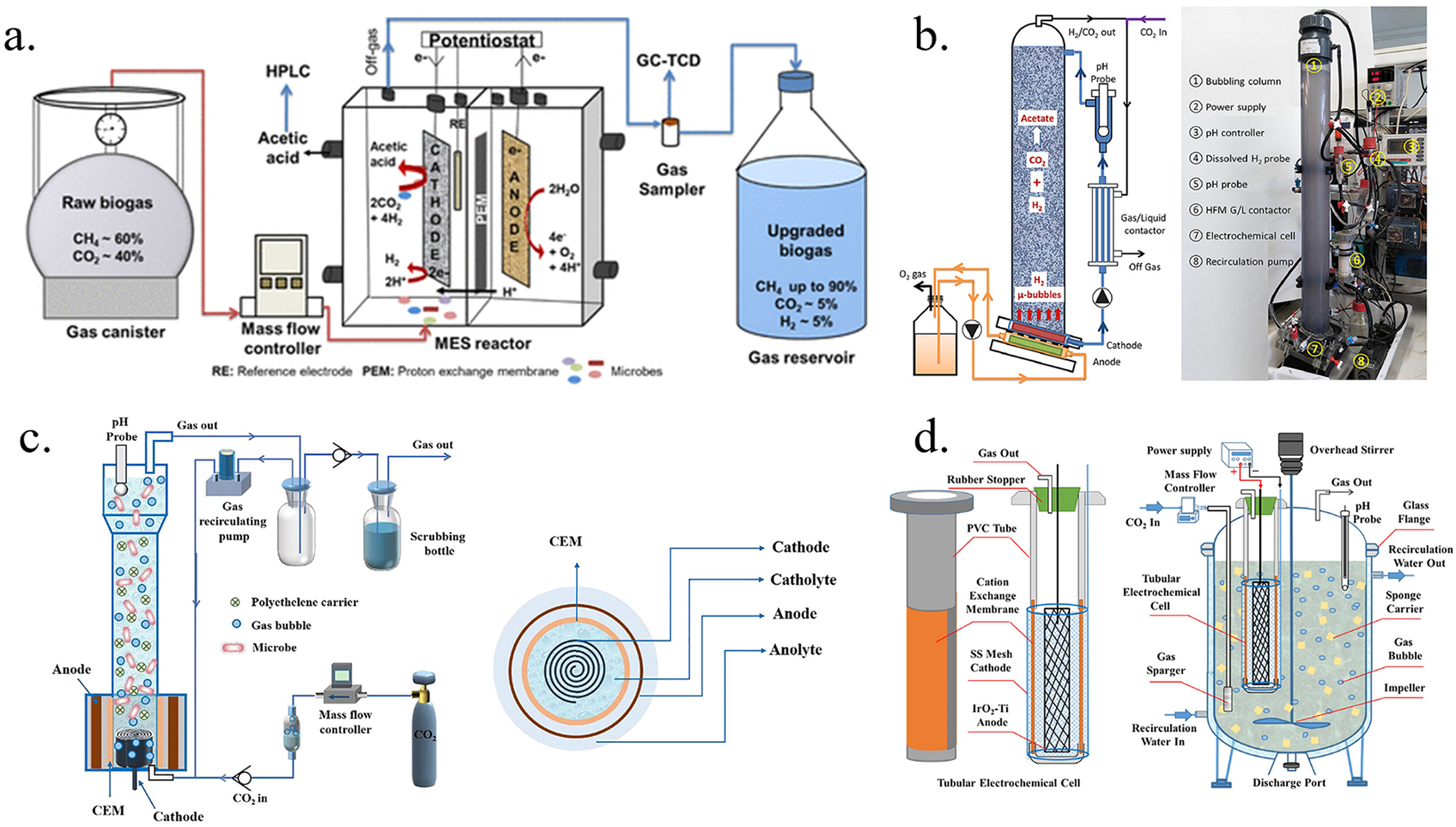
The issues associated with the H-type reactor are largely resolved in parallel plate dual-chamber reactors. In a 4.3 L parallel plate reactor designed by Roy et al., the distance between electrodes was reduced from 110 mm in the H-type reactor to 60 mm (Figure 5a). This design also doubled the electrode volume ratio from 1:25 to 1:12. The parallel plate design is easier to assemble and operate, with a mesh plate added above the tubular distributor to improve biogas distribution and mixing in the cathode chamber. At an E cell of 2.6 V, this setup achieved a CE of 82 ± 16% and an energy efficiency of 36 ± 7%, making it a more scalable and attractive MES reactor design (Roy et al., Reference Roy, Saich and Patil2023).
(a) Schematic representation of the biogas upgradation experimental setup and MES reactor (Roy et al., Reference Roy, Saich and Patil2023). (b) Schematic and photo of the electro-H2 bubble MES reactor (Cui et al., Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023). (c) Schematic diagram of the electro-MBBR reactor used in this study and vertical view of the rector (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022). (d) Schematic of the tubular electrochemical cell and electrochemical CSTR (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022).

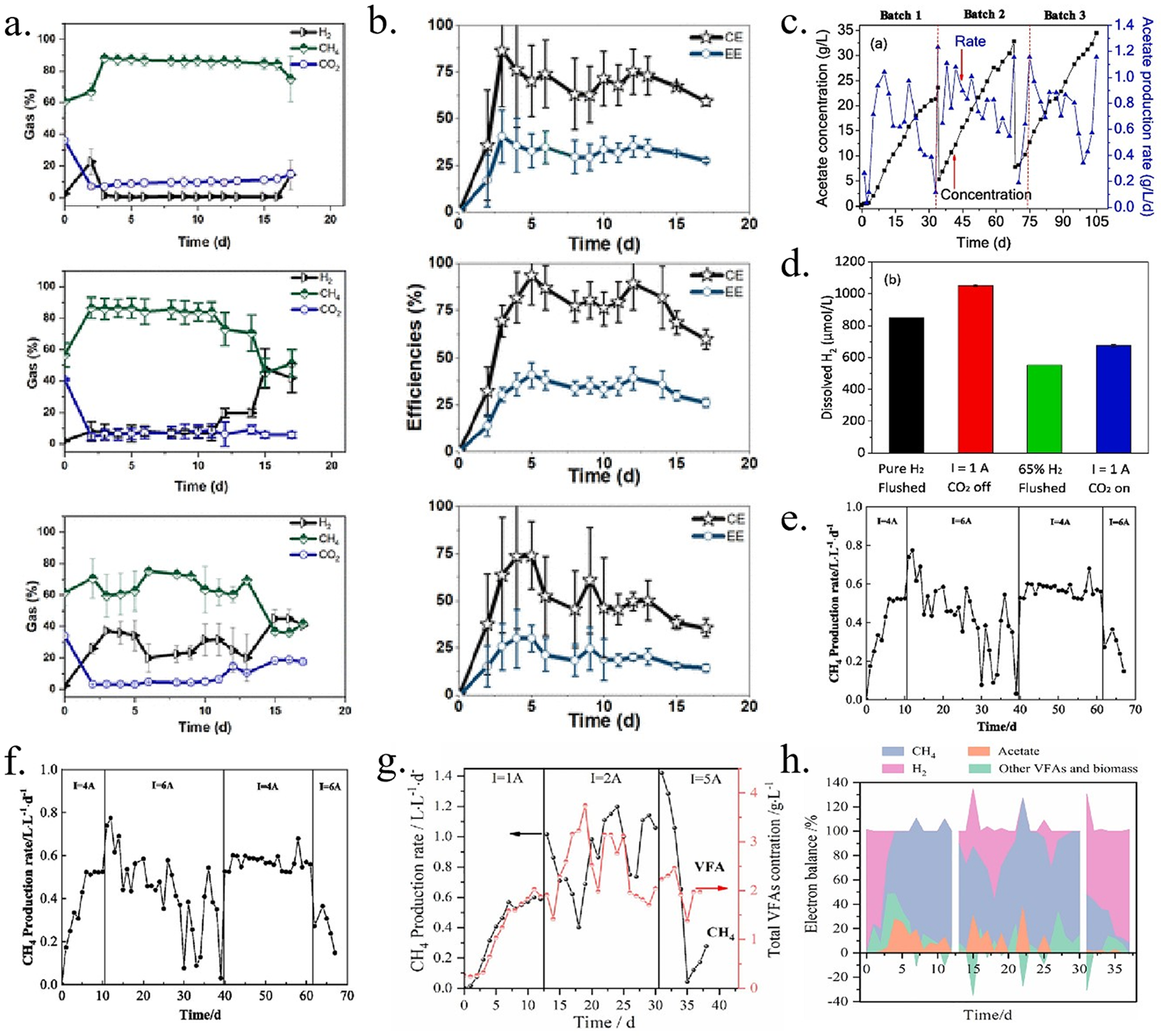
Enhancing H2 mass transfer and retention time in H2-mediated and suspended microorganism-driven MES reactors is key to improving CE, achievable through effective reactor design (Figure 5b–d). Cai et al. (Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022) developed a novel electrolytic hydrogen-supplying moving bed biofilm reactor (MBBR) for methane production, featuring an electrochemical cell at the base and an MBBR tower above. The MBBR tower extended the retention time of generated hydrogen, improving H2 mass transfer (Figure 5c). Consequently, methanogens efficiently converted the hydrogen and supplied CO2 into methane. The 4.5 L reactor achieved a maximum methane production rate of 1.42 L/(L · d) at 5 A, more than twice the highest reported rate for biofilm-driven MES (0.54 L/(L · d)) (Figure 6g). Cui et al. (Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023) introduced a novel electrolytic hydrogen bubble column reactor with an electrolytic cell at the reactor's base to produce fine H2 bubbles. A bubble column at the top of the cathode chamber extended H2 retention time, while a hollow fiber membrane gas–liquid contactor recovered unused gas (Figure 5b). This design allowed acetogens to efficiently convert the produced H2 and CO2 into acetate. With a catholyte volume of 5.5 L, the reactor achieved a high acetate production rate (1.15 g/(Lcatholyte · d)) and a notably high acetate titer (34.5 g/L) (Figure 6c). The continuous stirred-tank reactor (CSTR) is widely used for gas fermentation and has been commercialized at various scales. To evaluate its large-scale performance, a tubular electrochemical cell was integrated into a 20 L CSTR, creating a novel electrochemical CSTR (E-CSTR) reactor. The tubular cell supplied H2 in situ, enabling suspended biomass to utilize electrolytically produced H2 for CO2 fixation (Figure 5d). At a current of 4 A, the reactor achieved an average methane production rate of 317.84 L/(m2 · d), with an average CE of approximately 98% (Figure 6e, f) (Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023).
(a) Off-gas or upgraded biogas profiles of the cathode chamber fed with biogas continuously at 1 L/d in MES reactors operated at different applied current densities: −0.33, −0.5, and −0.66 mA/cm2. (b) Coulombic and energetic efficiencies (CE and EE) of the MES reactors fed with biogas at 1 L/d and operated at differed applied current densities: −0.33, −0.5, and −0.66 mA/cm2 (Roy et al., Reference Roy, Saich and Patil2023). (c) Acetate production and optical density in three consequent batches. (d) Dissolved hydrogen concentrations under different situations in the electro-H2 bubble reactor (Cui et al., Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023). (e) The methane production rate of the E-CSTR reactor. (f) Cell voltage and energy efficiency of E-CSTR reactor of catholyte and anolyte (Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023). (g) Average production rate of CH4 and total concentration of VFAs. (h) The Coulombic efficiency (CE) ended up in identified liquid or gas products (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022).

4.2. Electrode design
The cathode in MES systems supplies electrons that enable microorganisms to reduce CO2 into valuable chemicals (Su et al., Reference Su, Yujing, Yuanfan, Zhaoyuan, Shichao, Bing, Wenlei and Jun-Jie2023). Research has demonstrated that optimizing cathode materials and structures can enhance H2 uptake by bacteria, thereby increasing the efficiency of CO2 reduction to acetate. For example, constructing three-dimensional cathode structures can maximize the surface area for hydrogen evolution. Rolling stainless steel wool into a spiral or folding stainless steel mesh into tubular forms promotes a more uniform distribution of hydrogen bubbles (Cui et al., Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023; Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023; Tian et al., Reference Tian, Lacroix, Yaqoob, Bureau, Midoux, Desmond-Le Quéméner and Bouchez2023). Researchers have shredded carbon felt to fill the cathode, enhancing nutrient transport within the cathode chamber (Knoll et al., Reference Knoll, Jürgensen, Weiler and Gescher2023).
The anode, serving as the electron donor in MES systems, facilitates electron transfer and microbial metabolism. The material and surface properties of the anode directly affect the attachment and growth of electroactive microorganisms, consequently impacting the overall system efficiency (Zengyong Li et al., Reference Li, Lan and Liu2023). 3D porous electrodes outperform planar electrodes in current density and acetate production rate by providing a higher specific surface area for bacterial attachment. IrO2-coated Ti mesh is commonly used as an anode material in large-scale MES systems. IrO2 serves as an electrocatalyst with high conductivity, effectively promoting anodic oxidation reactions, while the Ti mesh offers a greater specific surface area for electroactive bacteria attachment (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022; Cui et al., Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023; Roy et al., Reference Roy, Saich and Patil2023; Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023). However, Ti and Ru are precious metals, rendering their use as electrode materials or catalysts costly and less ideal for large-scale operations. Bioanodes, utilizing microorganisms as catalysts to oxidize organic pollutants in wastewater and facilitate biocathode-driven CO2 conversion, are more suitable for scaling up MES systems due to their self-regulating capacity, environmental sustainability, and cost-effectiveness. Surface cleaning offers a simple yet effective method for addressing bioanode aging, as a clean electrode surface promotes the colonization of Geobacter species, thereby regenerating bioanode activity. Tian et al. designed an anode consisting of two removable parallel plates, enabling one plate to be cleaned while the other remains operational (Tian et al., Reference Tian, Lacroix, Yaqoob, Bureau, Midoux, Desmond-Le Quéméner and Bouchez2023).
4.3. Operating conditions
Current density directly influences microbial growth and metabolic pathways in MES systems. Higher current densities support denser biofilm formation and significantly enhance electron transfer efficiency and product generation rates (Cabau-Peinado et al., Reference Cabau-Peinado, Straathof and Jourdin2021). Cai et al. (Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022) investigated the effect of high current density on CO2 conversion efficiency at three currents (1, 2, and 5 A) and corresponding CO2 flow rates (2, 4.1, and 10.3 mL/min). At a current of 2 A, the maximum CE for CH4 production reached 92.5% (Figure 6g, h). Shang et al. (Reference Shang, Cui, Cai, Hu, Jin and Guo2023) observed that increasing the current from 4 A to 6 A caused performance degradation, as transmembrane O2 altered the microbial community composition (Figure 6e). Roy et al. (Reference Roy, Saich and Patil2023) employed constant current control in a MESs reactor, continuously supplying biogas at 1 L/d with a current density of −0.5 mA/cm2, which increased methane content from 56 ± 2% to 86 ± 6%. At lower current densities, however, the system produced a mixture of CH4 and H2 (i.e., hythane), complicating biogas upgrading (Figure 6a, b). Moreover, current density influences the size distribution of hydrogen bubbles and the concentration of dissolved hydrogen. Applying a current of 1 A (156 A/m2) in an electro-bubble reactor generates fine hydrogen bubbles at the cathode, which are evenly distributed in the bubble column (Figure 6d) (Cui et al., Reference Cui, Guo, Carvajal-Arroyo, Arends and Rabaey2023). Although current density is crucial to system performance, higher current densities do not always result in better performance.
CO2, serving as a carbon source, plays a central role in MES. Its flow rate directly impacts CO2 solubility and mass transfer efficiency, subsequently affecting microbial metabolic activity and the production rate of target products. Studies have shown that excessively high CO2 flow rates can reduce gas–liquid mass transfer efficiency, disrupt reactor fluid dynamics, and hinder microbial growth, ultimately decreasing product generation rates (Rojas et al., Reference Rojas, Zaiat, González, De Wever and Pant2021). Conversely, an optimal CO2 flow rate enhances CO2 solubility, ensuring a sufficient and continuous carbon supply for microbial electrochemical reactions, thereby increasing the system's overall reaction rate. In scaled-up MESs systems, CO2 mass flow controllers are typically used to regulate the flow rate, avoiding the adverse effects of high flow rates while maximizing CO2 fixation rate and acetate production (Cai et al., Reference Cai, Cui, Liu, Jin, Chen, Guo and Wang2022; Shang et al., Reference Shang, Cui, Cai, Hu, Jin and Guo2023).
5. Conclusion
METs have significant potential in treating pollutants from wastewater, recovering sustainable energy, and converting CO2 into valuable resources. However, the large-scale application of METs systems is currently limited by high costs and suboptimal performance. This review delves into the fundamental components, designs, and advancements made over the past decade in scaling up MFCs, MECs, and MESs, with particular emphasis on reactor design, electrode design, and the optimization of operating conditions.
(1) Reactor design: The design of the reactor involves considerations of fluid dynamics and gas–liquid mass transfer efficiency. For MFCs, single-chamber reactors are generally more suitable for scale-up, particularly in applications such as wastewater treatment, due to their low cost, simple structure, and high energy conversion efficiency. For MECs and MESs, dual-chamber reactors are often more ideal as they allow for finer environmental control and more effective product separation. In practical applications, it may be necessary to combine the advantages of both series and parallel connections, connecting multiple unit cells in parallel to increase current output, and then connecting these parallel units in series to achieve the desired voltage and current characteristics.
(2) Electrode design: Electrodes provide the pathway for electron transfer and the reaction interface. In addition to selecting electrode materials with high conductivity and corrosion resistance, performance can be enhanced through surface modification and structural optimization. For instance, using 3D porous structures or coating the electrode surface with nanostructured materials can increase surface area and catalytic activity, thereby improving electron transfer efficiency. Additionally, optimizing the spatial arrangement of the anode and cathode can minimize the distance between electrodes, reduce internal resistance, and enhance current density (Goto & Yoshida, Reference Goto and Yoshida2019).
(3) Operating conditions: The efficiency and stability of the system are influenced by various factors, including inoculum source, HRT, temperature, current density, gas flow rate, and nutrient supply. To maximize the electrochemical reaction rate and overall system stability, it is essential to carefully select appropriate microbial communities, optimize HRT and gas flow rate to balance treatment capacity with reaction efficiency, maintain temperature within the optimal range for microbial activity, and adjust current density and nutrient supply to align with microbial metabolic requirements.
6. Perspective
Based on the review of the literature related to the practical application of METs, it is evident that scale-up studies face several challenges, the most significant of which is that the improvement in reactor performance is limited relative to the increase in cost as the scale expands. To address the challenges, the following future directions are proposed to enhance the scalability of METs (Figure 7):
(1) The biological foundations of METs must be strengthened. The performance of electroactive microorganisms often declines under non-ideal or industrial conditions. Future efforts should prioritize the development of microbial resources with enhanced electrochemical activity, environmental tolerance, and long-term stability. Synthetic biology and metabolic engineering offer powerful tools to improve microbial-electrode interactions and optimize electron transfer efficiency.
(2) Electrode design and material sustainability remain central to successful scale-up. It is crucial to develop low-cost, durable, and high-performance electrode materials that maintain stability over extended operation. Integration of nanostructured surfaces or composite materials may enhance biofilm formation and catalytic activity. Additionally, exploring green and circular strategies for electrode synthesis can reduce environmental and economic costs.
(3) System integration and reactor engineering require further optimization. Develop modular reactors and components suitable for large-scale applications, streamline the system integration process, and optimize internal fluid dynamics, thermal management, and current distribution within the reactor. Address potential challenges related to mass transfer, heat transfer, and uneven current distribution during the scale-up process.
(4) The implementation of advanced monitoring and control technologies is essential to ensure operational stability, energy efficiency, and process adaptability. Introduce advanced sensor technologies and data analysis methods to enable real-time monitoring and optimization of operating conditions in microbial electrochemical reactors, such as temperature, flow rate, and current density. Employ machine learning and artificial intelligence to optimize operational parameters, reduce human error, and improve the overall efficiency of the system. Leverage machine learning and artificial intelligence to optimize operational parameters, minimize human error, and enhance the overall efficiency of the system.
(5) Life cycle assessment (LCA) serves as an essential tool for assessing the environmental impact and sustainability of various technologies. LCA research on MET scale-up systems identifies critical environmental impact hotspots during scaling and offers scientific insights for optimizing reactor design, operational parameters, and resource efficiency. However, research on the LCA of MET scale-up systems remains relatively scarce. Future studies should investigate how LCA can advance the sustainable development of MET scale-up systems by addressing energy losses, reducing greenhouse gas emissions, and enhancing wastewater resource recovery.
Roadmap of scaling microbial electrochemical technologies for climate mitigation and chemical production.

Acknowledgements
We are grateful to all co-participants in our transformative projects.
Author contributions
H. L., Y. J., and Z. W. conceived of the study. H. L. performed the research, analysis, wrote, and revised the manuscript. Y. J., R. Y., and X. L. provided review on drafts of the manuscript. Z. W. contributed to writing and revising. W. Z. supervised the project.
Funding statement
W. Z. would like to acknowledge the support from National Natural Science Foundation of China (22176086), Natural Science Foundation of Jiangsu Province (BK20210189), the Fundamental Research Funds for the Central Universities (021114380214, 021114380222), the Research Funds from Frontiers Science Center for Critical Earth Material Cycling of Nanjing University, State Key laboratory of Pollution Control and Resource Reuse, Research Funds for Jiangsu Distinguished Professor, and Carbon Peaking and Carbon Neutrality Technological Innovation Foundation of Jiangsu Province (BE2022861). Y. J. would like to acknowledge the support from the China Postdoctoral Science Foundation (2024M761388), the Fundamental Research Funds for the Central Universities – Cemac ‘GeoX’ Interdisciplinary Program (021114380217), Frontiers Science Center for Critical Earth Material Cycling of Nanjing University (2024QNXZ07), Postdoctoral Fellowship Program of CPSF (GZC20231105), and the Jiangsu Funding Program for Excellent Postdoctoral Talent (2023ZB226).
Competing interests
The authors declare no conflict of interest.
Research transparency and reproducibility
No unpublished data or methods have been used for the manuscript.