For those who think that success in science must be based on aggressive self-promotion and take-no-prisoners competition, Peter Huttenlocher’s well-spent life teaches how critical insights can gently but inexorably shift the axis of an entire scientific field, and spill out beyond their own confines to influence society at large.
Peering at thousands of neurons under an electron microscope in the 1970s, Peter sought to understand how the structures through which neurons communicate, known as “synapses,” change during development of the human brain. How are these billions of synapses formed and refined? How do these circuits change, and allow us to remember, and to learn? What happens when these connections go awry and what is their relationship to human disease?
The human brain has around 100 billion neurons and each neuron forms multiple branching connections, resulting in roughly a trillion synapses in the adult human brain. In the late 1800s, an improved window into understanding the human brain was enabled by an advance in microscopy and imaging of neurons. The Italian physician Camillo Golgi developed a staining technique using silver nitrate, which enabled a much clearer view of brain cells, known as the “Golgi stain.” It densely stains an entire neuron and its long projections, now known as the dendrites and axons. It was a modification of the Golgi stain that Peter and others used 100 years later to uncover new information about how the human brain develops.
Building on Golgi’s techniques, in the 1890s Santiago Ramón y Cajal discovered the physical structure of the neuron. A Spanish neuroscientist and artist, Ramón y Cajal is known as the father of modern neuroscience. His groundbreaking studies provided the first window into understanding neuronal connectivity, the basis for human memory. As an artist, Ramón y Cajal drew exquisite and detailed images of everything he saw under the microscope. He defined the neuron as the structural unit of the brain and was the first to visualize the beautiful network of connecting extensions and branches that emanate from the body of a neuron. Some projections, the “axons,” tend to be long and relatively unbranched, while others, the “dendrites,” are short and highly branched. Ramón y Cajal also described a gap between the axons of one neuron and the dendrites of a neighboring cell as a site of communication between the two cells, later referred to as the synaptic cleft. Axons and dendrites have since been shown to have distinct functions: generally, the dendrites are post-synaptic, or on the receiving end of signals between neurons, and axons are presynaptic, providing the signals that influence the receiving neuron.
In these early years, Ramón y Cajal postulated that the growth of neurons and their ability to form new connections formed the basis for “memory.” This was based on the observation that neurons form specific connections with other neurons in a kind of “circuit.” He also proposed correctly that information travels in one direction from the axons of one neuron to the dendrites of the neighboring neuron, and that there are specialized functions of neurons. Motor neurons transmit information from the brain to the periphery by, for example, connecting with muscles. Sensory neurons send information from the skin and other peripheral tissue to the brain, to affect brain activity. Other neurons reside in between the motor and sensory neurons and are referred to as “interneurons” that provide communication between the sensory and motor neurons. Ramón y Cajal’s work and theories are credited with providing the origin for the synaptic theory of memory. Together with Golgi, Ramón y Cajal received the 1906 Nobel Prize in Physiology or Medicine for this groundbreaking work. In 1937, when Peter was just a young boy in Germany, Ramón y Cajal eloquently wrote, “I noticed that every outgrowth, dendritic or axonic, in the course of formation, passes through a chaotic period, so to speak, a period of trials, during which they are sent at random experimental conductors, most of which are destined to disappear.” Peter cited this quote in his 2002 book Neural Plasticity, as the first reference to the idea of synaptic elimination [Reference Huttenlocher1].
The gaps between the axons and dendrites of neurons were referred to as a “synapse” by the English physiologist Charles Sherrington. Sherrington received the Nobel Prize in Physiology or Medicine in 1932 for showing that reflexes (think of the human knee reflex) are not a simple arc of connectivity but rather derive from integrated networks of activation between neurons. He demonstrated that some neurons excite other neurons, while others inhibit the activity of neurons, thereby enabling a coordinated and integrated response. As part of this work, Sherrington proposed the idea of “synaptic communication” between neurons.
The idea that neurons communicate, and regulate the activity of other neurons, paved the way for the evolving concept that these connections can change over time based on their input and experience. Neural plasticity or brain plasticity is defined as the ability of the nervous system to adapt by reorganizing its structure, connectivity and function in response to both external and internal stimuli. This concept of plasticity was raised in the fields of both psychology and neuroscience as early as the 1890s. Credit for the specific term “neural plasticity” has been given to the Polish neuroscientist Jerzy Konorski.
When Peter launched his work on synapses in the developing human brain, it was not known how these connections change during human development. However, the idea that synaptic changes underlie brain plasticity was gaining traction during the 1960s and 1970s. The work of David Hubel and Torsten Wiesel elegantly displayed the plasticity of the developing non-adult brain. Using sensory deprivation in kittens, Hubel and Wiesel deprived one eye of visual input from birth by sewing it closed and then recorded the activity of neurons on both sides of the brain. To the surprise of Hubel, Wiesel and their colleagues worldwide, a few months after blocking input to one eye, all neurons on both sides of the “striate cortex” responded to input from the intact eye [Reference Wiesel and Hubel2]. It was expected that the part of the brain that would have responded to the closed eye would have remained quiescent. But instead, some brain areas previously quiescent due to sensory deprivation had regained activity when studied months after the start of the deprivation. The open eye had co-opted some of the synaptic inputs from the closed eye, on the opposite side of the brain, as part of a rewiring of synaptic connectivity. This provided direct evidence that the visual cortical area of the brain is rewired due to the visual deprivation in one eye. It also raised critical questions about how environmental changes lead to a competition between synaptic connections and their remodeling.
The work of Hubel and Wiesel revealed that the plasticity and reorganization of the visual cortex only occurred during a limited developmental window of the kitten, referred to as the “critical period.” If the same experiment was done in older cats, the sensory deprivation did not lead to a change in cortical connectivity and rewiring [Reference Hubel and Wiesel3]. Hubel and Wiesel also performed reverse suturing by opening the previously deprived eye and covering the dominant eye. As long as this was done in the first months of life, during this critical period the newly uncovered eye co-opted the wiring of the ocular dominance pattern, switching the “visual activity” to the uncovered eye that was now receiving the input. These findings suggested that there was something unique about younger animals which allowed for rewiring of neuronal connectivity leading to functional plasticity.
It was precisely these questions about neuronal connectivity that Peter aimed to understand in the early 1970s. In addition to his scientific work, Peter was busy running an active pediatric neurology practice and making substantial medical school teaching and medical resident training contributions at Yale University and then the University of Chicago. It was logical to be drawn primarily to studying brain tissues from human patients with pathologies. Through his work in the first half of the 1970s, Peter discovered that there was stunted development and abnormal shape of synapses from children with significant developmental delay [Reference Huttenlocher4]. This dendritic spine “dysgenesis” provided support for the idea that the shape of synapses, and not just the number of synapses, is a factor to consider when studying the plasticity of the human brain.
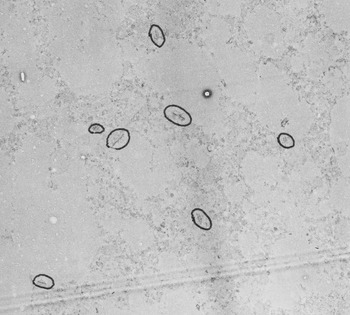
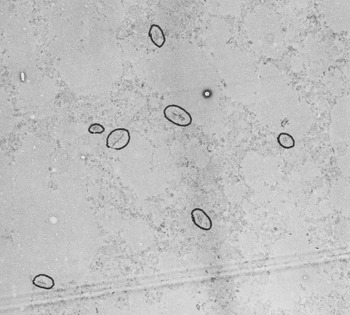
Using specimens obtained from autopsies, Peter studied the brains of children who had intellectual disabilities, comparing them to samples from children with “normal” development who had died for other reasons. He undertook a painstaking set of studies using an electron microscope to acquire hundreds of images from specific brain regions. He then scrutinized photographic prints of the electron microscope images and counted neurons and synaptic junctions by hand (Figure 2.1). Peter found something interesting in the “normal” brain samples. He realized that to understand what went awry with disease he first had to understand what happened during normal human brain development. Peter later said that “the findings in the normal population were more interesting than the abnormal population.” By the 1970s, when Peter was doing this work, much of the work emanating from molecular bioscience laboratories was being done by graduate students, postdoctoral trainees and scientific staff, working under the direction of a lead scientist. Hearkening back to the days of Golgi and Ramón y Cajal, Peter instead did much of this seminal work himself. One of Peter’s former trainees, Dr. Carter Snead (Professor of Pediatrics, Neurology and Pharmacology, University of Toronto), commented that Peter “did it the hard way.” He spent hours at the microscope, analyzing brain samples. It was daunting to collect the samples, and to perform the analysis of synapse numbers in a tiny region of the brain. What did it mean to look at such a small piece of tissue when the human brain was much larger? Could you accurately quantify synapses in these samples? There were many hurdles and more questions than answers as Peter embarked upon this work.
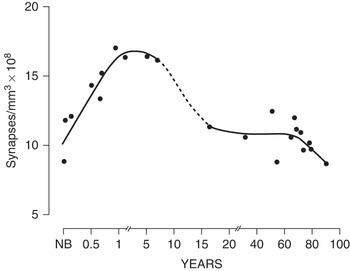
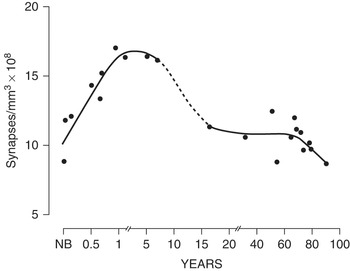
In his landmark single-authored paper published in Brain Research in 1979 [Reference Huttenlocher5], Peter reported on changes in synaptic density in a region of the human cerebral cortex in individuals with no intellectual disabilities. He had found something entirely unexpected. Early results had led him to broaden the study; autopsy specimens were now used from the newborn period to advanced adulthood. He observed that synaptic density in the adults was relatively constant, with some decline at older ages (74–90). At the time of birth, the number of synapses was already high in infants, comparable to the adults, although the morphology of the synapses was different in infants than in adults, suggesting functional differences. During the first months of life, there is robust production of new synapses – to levels 50% greater than the adult brain. The number of synapses peaks during childhood and then regresses. What was most surprising was the elimination of many of these synapses during the subsequent childhood years, a prolonged period of intense learning, up until late adolescence or even adulthood (Figure 2.2). Peter wrote that “the human cerebral cortex is one of a number of neuronal systems in which loss of neurons and synapses appears to occur as a late developmental event.” He showed for the first time that there is elimination or “pruning” of synapses that coincides with key developmental milestones in childhood such as learning to walk or speak. The overall number of synaptic connections goes down, not up, as we learn and remember. Synaptic connections are not just refined. As part of healthy brain development, millions of synapses are pruned away and lost.
Synapse counts from the middle frontal gyrus, showing reduction of synapses with age – the figure that is most frequently shown by other scientists. From: Huttenlocher PR. 1979. Synaptic density in human frontal cortex – developmental changes and effects of aging. Brain Res 163:195–205. doi: 10.1016/0006-8993(79)90349-4.
Since the time of Peter’s discovery of synaptic pruning in childhood, and the work of Hubel and Wiesel, there has been substantial progress in understanding how neuronal activity influences synaptic pruning in mammals. This activity-dependent synaptic pruning occurs in the developing nervous system through the effects of both spontaneous neuronal activity and activity induced by sensory experiences, such as visual or auditory input. Synapses that are stimulated and active are maintained while the quiescent synapses are eliminated during this developmental rewiring of neuronal circuits in the brain. As an example, in work done in the 1980s by Shatz and colleagues, the impact of neural activity on synaptic elimination during development was directly demonstrated [Reference Sretavan and Shatz6]. The retinal cells in the eye extend axons to a structure in the brain known as the lateral geniculate nucleus (LGN). In the LGN, the retinal axons branch and form exuberant synapses during development. Subsequently, these robust branches and synapses in the LGN are pruned and refined to form an organized visual map. The neurons that respond to a particular stimulus are co-localized in specific areas of the LGN. But when neural activity is blocked synaptic pruning fails, leading to a disorganized network of branching axons and synapses in the LGN and, as a result, a tangled visual map. In recent decades, our understanding of the molecular underpinnings of activity-dependent synaptic pruning has exploded and this work has uncovered an elegant multi-tiered system that regulates synaptic pruning during development.
However, at the time of Peter’s discovery of synaptic pruning, widespread recognition by neuroscientists did not quickly follow. While the discoveries fascinated Peter and a cohort of scientists, their reports on the work, like the work of Hubel and Wiesel, were submitted for publication to respected but less widely read specialist journals. Peter gave a number of invited talks internationally about his findings – again, usually to specialist audiences. There were some arranged media interviews orchestrated by the University of Chicago, which were picked up by German newspapers including Die Welt – a pleasing bit of recognition. One of Peter’s sons, who was visiting family in Munich in the summer of 1978, wrote, “Dad: you were on page one of Die Welt in Deutschland, re: work on brain development. Oma H [Peter’s stepmother] suggested maybe you would win a Nobel prize – they were impressed.” However, it took many years and much creative work for an increasing number of neuroscience and psychology experts to absorb, validate, rediscover, expand upon and more widely publicize the core ideas of synaptic pruning.
As neuroscientist Christian Hansel (2021) noted in his book entitled Memory Makes the Brain: The Biological Machinery That Uses Experiences to Shape Individual Brains [Reference Hansel7]:
[Hubel and Wiesel] performed much of their work alone … and published their most important work in a few papers that were published … in journals that focused on science as opposed to flashy headlines … . This was true of Peter Huttenlocher as well. Yet, all three of them were giants in their fields and left a mark on neuroscience like few others.
In an obituary for Peter Huttenlocher published in Nature in 2013 [Reference Walsh. and Huttenlocher8], Professor Chris Walsh of Harvard University said:
Huttenlocher’s discovery that synapses are overproduced and then pruned was 20 years or more ahead of its time. Today, most ideas about human brain development, from the microscopic to the macroscopic to the societal, draw on his work. For instance, researchers are now investigating the mechanisms that control pruning, the possibilities that this synaptic plasticity provides, and how to use an understanding of synaptic development to optimize early educational intervention, language learning or music instruction.
Dr. Jeffrey Lichtman, a developmental neurobiologist at Harvard who uses sophisticated computer imagery to map brain circuitry, said to the New York Times (August 26, 2013):