LEARNING OBJECTIVES
After reading this article you will be able to:
• describe the psychological and physical symptoms of antidepressant withdrawal and how these can overlap with symptoms of depressive or anxiety disorders
• distinguish withdrawal effects of antidepressant discontinuation from relapse of the underlying condition for which antidepressants were initially prescribed
• understand the way in which antidepressant withdrawal may be misdiagnosed as relapse in antidepressant discontinuation studies and how that may influence the perceived relapse prevention properties of antidepressants.
A common circumstance: a patient has stopped taking an antidepressant (either under professional advice or on their own) and reports psychological symptoms such as anxiety, low mood or trouble sleeping (Box 1). Such reports have often led to the automatic conclusion that the patient must be experiencing a relapse of their underlying condition (Haddad Reference Haddad and Anderson2007). However, there is increasing recognition that withdrawal symptoms from antidepressants are common and may be long-lasting, over months or years (Davies Reference Davies and Read2019). Withdrawal symptoms occur in up to half of patients and are probably more common after longer-term use, higher doses and with particular antidepressants, such as serotonin–noradrenaline reuptake inhibitors (SNRIs) and paroxetine. ʻThere should be greater recognition of the potential in some people for severe and long-lasting withdrawal symptoms,' opined a recent position statement from the Royal College of Psychiatrists (RCPsych) (Royal College of Psychiatrists 2019: p. 3), and a corresponding update to the National Institute for Health and Care Excellence (NICE) guidelines highlighted ʻ[antidepressant withdrawal] symptoms lasting much longer (sometimes months or more) and being more severe for some patients' (Iacobucci Reference Iacobucci2019).
BOX 1 Case vignette
A 52-year old woman presents to your clinic complaining of tearfulness, an inability to sleep and extreme anxiety. She has recently tried to cease her sertraline because of weight gain and fatigue. Her general practitioner (GP) recommended that she reduce her sertraline from 100 mg to 50 mg for 2 weeks, then to 25 mg for 2 weeks, and to 25 mg every second day for 2 weeks before stopping. These symptoms occurred when she got down to a dose of 25 mg every second day. Her GP diagnosed her with relapse of an anxious disorder, but agreed to refer her for a second opinion.
Further history reveals that these symptoms came on a few days after reduction to 25 mg of sertraline every second day, and have been gradually getting worse over the past week. She had milder versions of these symptoms on the previous reduction to 25 mg daily. She also reports that she has experienced dizziness, has been feeling unsteady on her feet and had a minor car accident because she did not feel as coordinated as usual. She has also had strange sensations in her head, especially on turning her eyes from side to side, where she feels that her ‘brain has been switched off for a second’.
She has had similar symptoms in the past, although not as severe, when she has gone away for a weekend and forgotten to take her medication. These symptoms went away within a few hours of taking a dose of sertraline.
She has been taking the sertraline at a dose of 100 mg for the past 8 years, following the death of her father. At that time she had a very low mood, was tearful, had stopped eating, slept a lot and had reduced social contact.
She is concerned that she is having a return of the episode she had following the death of her father. You reassure her that she is experiencing antidepressant withdrawal syndrome – that her symptoms are different from what she experienced when she was first prescribed the medication, that they are quite typical of withdrawal symptoms, and include several physical symptoms that help to distinguish them from relapse, including unsteadiness/incoordination and what sounds like electric ‘zaps’. They are typical in timing – coming on a few days after reducing her antidepressant dose. They match her past experience of withdrawal symptoms, which were alleviated quickly by reinstatement. They are made more likely by a rapid rate of reduction and dosing every other day, which causes significant variaition in plasma levels because of the 26 h half-life of sertraline.
You advise her to return to a dose of 50 mg and reassure her that her symptoms are likely to resolve over a few days. Once she is stabilised, she will need to reduce her sertraline in smaller amounts over a longer period, following the Royal College of Psychiatrists’ guidance on this topic to do so (Burn Reference Burn, Horowitz and Roycroft2020). You write a letter to her GP to outline a gradual tapering plan following this guidance and advise that he prescribe her a liquid version of sertraline (available as a ‘Special’) to make the taper more practical, as it will involve going down to doses lower than currently available tablet formulations permit.
The RCPsych's position statement on antidepressants goes on to state: ʻ[withdrawal] effects need to be distinguished from symptoms of relapse or recurrence of the original condition for which they were prescribed to ensure the former is not mistakenly attributed to the latter' (Royal College of Psychiatrists 2019: p. 16). Although antidepressant withdrawal might have previously been considered a controversial issue, it should now be carefully considered by clinicians as an important differential in all cases involving reduction of the dose of antidepressants – especially given that patients commonly report that their antidepressant withdrawal symptoms are misdiagnosed as relapse (White Reference White, Read and Julo2021). Furthermore, it should be a part of informed consent when an antidepressant is being considered (Royal College of Psychiatrists 2019).
In this article we explore the symptoms of antidepressant withdrawal, how they overlap with symptoms of depression or anxiety, and how a clinician might differentiate withdrawal symptoms from relapse of an underlying condition. We also examine discontinuation trials used to demonstrate the relapse prevention properties of antidepressants (their ability to prevent future episodes in a prophylactic manner), and outline evidence that the ascribing of relapse in discontinuation studies of antidepressants may be confounded by withdrawal effects. We conclude with advice for clinicians as well as ways to construct a more robust evidence base for establishing the relapse prevention properties of antidepressants. The studies explored in this article involve adult populations, but similar principles may apply to young people as well, although this area has not been studied.
Withdrawal symptoms from antidepressants
Many medications with central nervous system effects produce both physical (affecting the body) and psychological (affecting the mind) withdrawal symptoms on stopping or reducing the dose, widely attributed to perturbations to neurotransmitter systems affected by these drugs (Lerner Reference Lerner and Klein2019; Cosci Reference Cosci and Chouinard2020). Drugs that produce a withdrawal syndrome include psychotropics such as monoamine oxidase inhibitors (MAOIs), tricyclic antidepressants (TCAs) (Taylor Reference Taylor, Stewart and Connolly2006), benzodiazepines (Sokya Reference Sokya2017; Cosci Reference Cosci and Chouinard2020) and other medications, such as Z-drugs (non-benzodiazepines, e.g. zolpidem, zaleplon and zopiclone), antipsychotics, lithium, mood stabilisers and ketamine (Lerner Reference Lerner and Klein2019; Cosci Reference Cosci and Chouinard2020), as do antihypertensive, antihistaminergic and anticholinergic medications (Howland Reference Howland2010).
Withdrawal symptoms from the modern classes of antidepressant (selective serotonin reuptake inhibitors (SSRIs), SNRIs and similar medications) were first reported in 1991 (Stoukides Reference Stoukides and Stoukides1991), 3 years after the first blockbuster modern antidepressant, the SSRI fluoxetine, was released on to the US market. Following increasing numbers of case reports of withdrawal symptoms from antidepressants (Fava Reference Fava, Gatti and Belaise2015), a Discontinuation-Emergent Signs and Symptoms (DESS) checklist was developed by psychiatrists ʻbased on an evaluation of signs and symptoms reported in the available literature' (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). This checklist contains 43 signs and symptoms of withdrawal in both psychological and physical domains, although the distinction between these categories can be somewhat arbitrary (e.g. fatigue, dizziness and nausea). These myriad effects likely arise from the varied effects of neurotransmitters affected by antidepressants. For example, serotonin has been implicated in the ‘physical domain’ of gut function (Mawe Reference Mawe and Hoffman2013), as well as balance, dizziness through dysmodulation of the serotonin-1A receptors in the raphe nuclei (Coupland Reference Coupland, Bell and Potokar1996) and dysregulation of the somatosensory system resulting in paraesthesia (Olver Reference Olver, Burrows and Norman1999).
The term ‘discontinuation symptoms’ was developed by drug manufacturers to minimise patient concerns regarding their product, and there is now widespread recognition that this term is misleading and the more pharmacologically meaningful term is ‘withdrawal symptoms’, now adopted by RCPsych (Royal College of Psychiatrists 2019; Massabki Reference Massabki and Abi-Jaoude2021). The physical dependence produced by antidepressant use is distinct from addiction, which also requires the presence of compulsion, craving and a narrowing behavioural repertoire, not evident for antidepressants (Lerner Reference Lerner and Klein2019). Conflation of withdrawal effects with addictive properties is misleading (Jauhar Reference Jauhar, Hayes and Goodwin2019a; Lerner Reference Lerner and Klein2019).
Antidepressant withdrawal symptoms are common
A recent systematic review identified 24 studies that evaluate withdrawal symptoms from antidepressants, including randomised controlled trials (RCTs), observational studies and survey data (Davies Reference Davies and Read2019). Given the paucity of data in this area of psychiatry, all studies are of interest but double-blind RCTs are particularly instructive because they include standardised evaluation of symptoms and allow greater causal attribution. One representative and well-conducted RCT published by Rosenbaum and colleagues in 1998 is worth examining in detail because of its high quality and reliability: it uses several measures of withdrawal effects and symptom scales, allowing direct comparison in the same group of patients in a double-blind procedure. Patients (n = 242) with remitted depression who were treated with fluoxetine, sertraline or paroxetine for, on average, 11 months, had their treatment interrupted by either placebo or their usual antidepressant in a double-blind manner for 5–8 days (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). Nocebo effects – when a negative outcome occurs owing to the belief that an intervention will cause harm, the opposite of the placebo effect – were rendered less likely in this study as there were two possible time periods where placebo substitution could occur in a double-blind manner.
In this study, 14% of participants on fluoxetine, 60% of those on sertraline and 66% of those on paroxetine reported four or more symptoms on the DESS (Table 1), the criterion for experiencing a ʻdiscontinuation syndrome' (Rosenbaum Reference Rosenbaum, Fava and Hoog1998), on treatment interruption. As fluoxetine has a long elimination half-life (7–15 days), 5–8 days was probably too short a period to demonstrate withdrawal effects from this medication, leading to an underestimation of their frequency. Participants who did not have their antidepressants disrupted did register small increases in withdrawal symptoms (calculated weighted average: 2.3 symptoms on the DESS), consistent with small nocebo effects. Notably, the numbers of symptoms detected in participants interrupted from sertraline or paroxetine were much greater: 5.7 and 7.8 respectively. Withdrawal symptoms in the sertraline and paroxetine groups resolved after their antidepressants were reinstated (unbeknownst to the participants), consistent with a genuine pharmacological effect, rather than a nocebo effect. Furthermore, the fluoxetine group might approximate a ‘drug continuation’ group because of the drug's long half-life, so 14% might represent the nocebo withdrawal incidence – smaller than that seen for sertraline and paroxetine.
TABLE 1 The 20 most common withdrawal symptoms reported by people stopping antidepressantsa
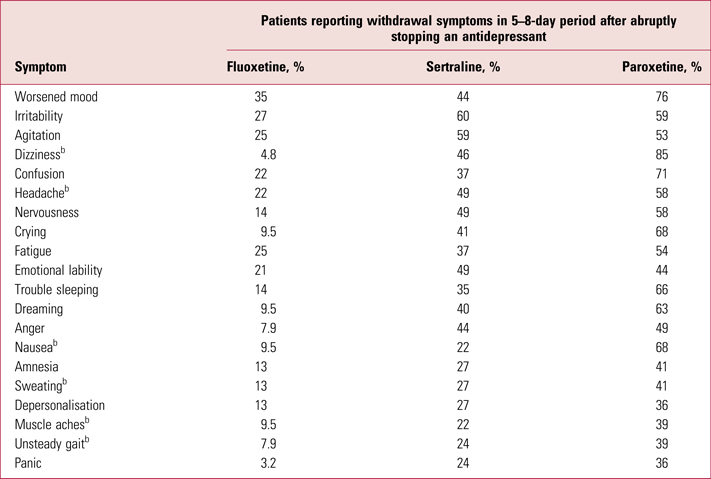
a These data (rounded to two significant figures) are derived from Rosenbaum et al (Reference Rosenbaum, Fava and Hoog1998).
b Physical symptoms.
In a systematic review combining six RCTs with five naturalistic studies and three large surveys, 27–85% of patients were found to have experienced withdrawal symptoms on stopping an antidepressant (mostly SSRIs and SNRIs), with a weighted average of 56% (Davies Reference Davies and Read2019). Restricting the analysis only to double-blind RCTs from this review, the incidence of withdrawal effects is 53.9% (6 RCTs, 731 participants). There was also some variation in incidence rates between different antidepressants in these RCTs, although the number of studies was small for many drugs: paroxetine (58.8%, 3 RCTs), fluoxetine (50%, 3 RCTs), sertraline (59%, 3 RCTs), citalopram (70%, 1 RCT), escitalopram (27%, 1 RCT) and venlafaxine (71%, 1 RCT). Another systematic review reported withdrawal symptoms for 35–78% of people who stopped venlafaxine (Fava Reference Fava, Benasi and Lucente2018). Another study not captured in the review did not detect a withdrawal syndrome for agomelatine following short-term use (Montgomery Reference Montgomery, Kennedy and Burrows2004).
Nocebo effects (that is, withdrawal effects experienced when placebo is discontinued or when an antidepressant is continued) are worth examining in order to compare with the single-arm frequencies provided for antidepressants. In the six RCTs captured by the Davies & Read review (Davies Reference Davies and Read2019) and the five RCTs reviewed by Baldwin (Baldwin Reference Baldwin, Montgomery and Nil2007), six studies included such a ‘nocebo group’. In three studies a placebo was stopped, with incidence rates of withdrawal of 12.2% (n = 123), 1.9% (n = 116) (Baldwin et al (2004b) and Lader et al (2004) respectively, cited in Baldwin et al (Reference Baldwin, Montgomery and Nil2007)) and 13.5% (n = 52) (Oehrberg Reference Oehrberg, Christiansen and Behnke1995). In two studies an antidepressant was continued while patients were masked: in Montgomery et al (Reference Montgomery, Durr-Pal, Loft and Nil2003) (cited in Baldwin et al (Reference Baldwin, Montgomery and Nil2007)) 9.2% of 125 patients met criteria for withdrawal. In the other study (Zajecka Reference Zajecka, Fawcett and Amsterdam1998), 75% of 299 patients continued on fluoxetine reported one or more withdrawal symptom, although this is difficult to compare with other studies which used thresholds of four or more symptoms to detect a withdrawal syndrome. Lastly, stopping fluoxetine, which might be considered a measure of continuation because of its half-life, yielded an incidence of withdrawal symptoms of 14% (Rosenbaum Reference Rosenbaum, Fava and Hoog1998).
Excluding the outlier study for fluoxetine continuation (with an atypical definition of withdrawal syndrome – one symptom) the weighted average of the nocebo effect in these studies is 11.8%. Subtraction of this rate from the overall detected rate in RCTs (53.9% − 11.8%) might therefore give a more reasonable estimate of 42.1%. However, it should be kept in mind that most studies did not measure the severity of withdrawal effects and it is not clear that ‘dizziness’, for example, reported in nocebo conditions is equivalent to that in antidepressant withdrawal (where patients have been referred to emergency departments for investigation of stroke) (Haddad Reference Haddad, Devarajan and Dursun2001). Nevertheless, it is probably the case that a small minority of withdrawal is explained by nocebo effects.
Critical authors recalculated the incidence of withdrawal effects as 44%, when including further studies conducted by a drug company, published as supplements in journals (Jauhar Reference Jauhar and Hayes2019b). The lowest value obtained by that paper, of 31% (for RCTs alone), still represents 2.4 million people at risk of withdrawal in England. A further critique only analysed the five drug company studies, in which patients were treated, on average, for only 12 weeks and in which the authors subtracted out a high estimate for nocebo effects (Jauhar Reference Jauhar, Hayes and Goodwin2019a). The arbitrary study selection, short duration of treatment and idiosyncratic transformation of values in this study make its results difficult to interpret in relation to the general population of people using antidepressants (more than half of whom have been on antidepressants for more than 2 years) (Johnson Reference Johnson, Macdonald and Atkinson2012).
Withdrawal symptoms can be severe and/or long-lasting
A systematic attempt to assess the average severity and duration of withdrawal effects was limited by the lack of studies assessing these dimensions (Davies Reference Davies and Read2019). As regards symptom severity, only data from four large surveys were available. These indicated that about half of patients (46%) who reported withdrawal effects from antidepressants reported that these were ‘severe’, selecting the highest option on a Likert scale used to indicate severity in these studies (Davies Reference Davies and Read2019). These data were not derived from a representative sample and this limits their general applicability: those motivated to answer may have a worse than average experience of withdrawal, although 85% of patients in one included survey felt that antidepressants were helpful to them (Read Reference Read and Williams2018), indicating that negative views towards this class of medication were not widespread. Notably, patients in these surveys did have a duration of use of antidepressants (mode >3 years) similar to the wider population of antidepressant users in England (Johnson Reference Johnson, Macdonald and Atkinson2012).
Ten of the included studies evaluated duration of withdrawal symptoms. These studies are vulnerable to bias themselves – for example, studies derived from patient self-report on online forums – and did not provide enough consistency for a weighted average to be calculated. However, in seven out of the ten studies the average duration of withdrawal symptoms was greater than the 2 weeks previously supposed to be typical of withdrawal effects (Davies Reference Davies and Read2019). Indeed, the mean duration of withdrawal symptoms was 18 months in one study (after, on average, 5 years of use) (Stockmann Reference Stockmann, Odegbaro and Timimi2018) and in another (with 75% of patients using antidepressants for more than 2 years) more than one-third of patients experienced withdrawal symptoms for more than a year (Davies Reference Davies, Pauli-Jones and Montagu2018), suggesting that at least for some people, and especially those with longer-term use, withdrawal symptoms may be long-lasting.
It is commonly thought that protracted withdrawal symptoms, lasting months or years, are pharmacologically implausible; correspondingly, sometimes patients are told that long-lasting symptoms could not be drug-related because it is ‘out of their system’. This misconstrues the pathophysiology of withdrawal symptoms. Repeated use of psychotropic medication causes adaptations in the brain and body; these can be thought of as resetting of the homeostatic set-point for neurotransmitters (Hyman Reference Hyman and Nestler1996; O'Brien Reference O'Brien2011), including downregulation of serotonin receptors observed in positron mission tomography (PET) studies (Meyer Reference Meyer, Kapur and Eisfeld2001; Haahr Reference Haahr, Fisher and Jensen2014). When the drug dose is reduced, the mismatch between what the system ‘expects’ and the input from the drug is experienced as withdrawal symptoms. The elimination half-life of the drug is important: the quicker it is eliminated the sooner withdrawal symptoms will start. However, the most important factor is the length of time taken for adaptations to the presence of the drug to resolve (Reidenberg Reference Reidenberg2011). There has been little research into this area regarding antidepressants but, for example, it has been shown that adaptations to antipsychotics in animals can persist for more than a human-equivalent year (Horowitz Reference Horowitz, Jauhar and Natesan2021). It is plausible that after long-term use of antidepressants these adaptations may persist for many months or longer, consistent with the experience of protracted withdrawal symptoms (Hengartner Reference Hengartner, Schulthess and Sorensen2020a).
Overall, although further research in this neglected area would be welcome it can be concluded that between a third and a half of patients will experience withdrawal symptoms, a portion of these reactions will be severe (with half possibly being an overestimate) and for some individuals withdrawal symptoms will last months or years. The RCPsych position paper, following this evidence, highlighted that withdrawal effects can be ʻsevere and long-lasting' in some people (Royal College of Psychiatrists 2019: p. 3). Further studies will need to survey representative populations of antidepressant users or conduct prospective discontinuation trials that carefully measure incidence, severity and duration of withdrawal effects to help refine our understanding of what characteristics influence risk of withdrawal (e.g. duration and dose of use, type of antidepressant) to help better inform patients of the risks.
Withdrawal symptoms of a psychological nature are common
The psychological symptoms that have been attributed to antidepressant withdrawal are shown in Fig. 1 (Rosenbaum Reference Rosenbaum, Fava and Hoog1998; Fava Reference Fava, Gatti and Belaise2015; Cosci Reference Cosci and Chouinard2020). Psychological withdrawal symptoms are common – for example, in the Rosenbaum study, four out of the five most commonly experienced withdrawal symptoms were psychological: worsened mood, irritability, agitation and confusion. The possibility remains that these symptoms might result from genuine relapse of the patients’ original condition. However, some aspects of the study make this interpretation less likely: the psychological symptoms had onset (and resolved) at the same time as physical symptoms clearly not characteristic of depression (such as dizziness, headache, nausea, sweating, muscle aches and an unsteady gait) and resolved swiftly (within a week following antidepressant recommencement (see in the following)) (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). Patients report that psychological symptoms of withdrawal can be severe and persist for long periods, and can be distinguished from their original condition (ʻI've never felt this before') (Framer Reference Framer2021).
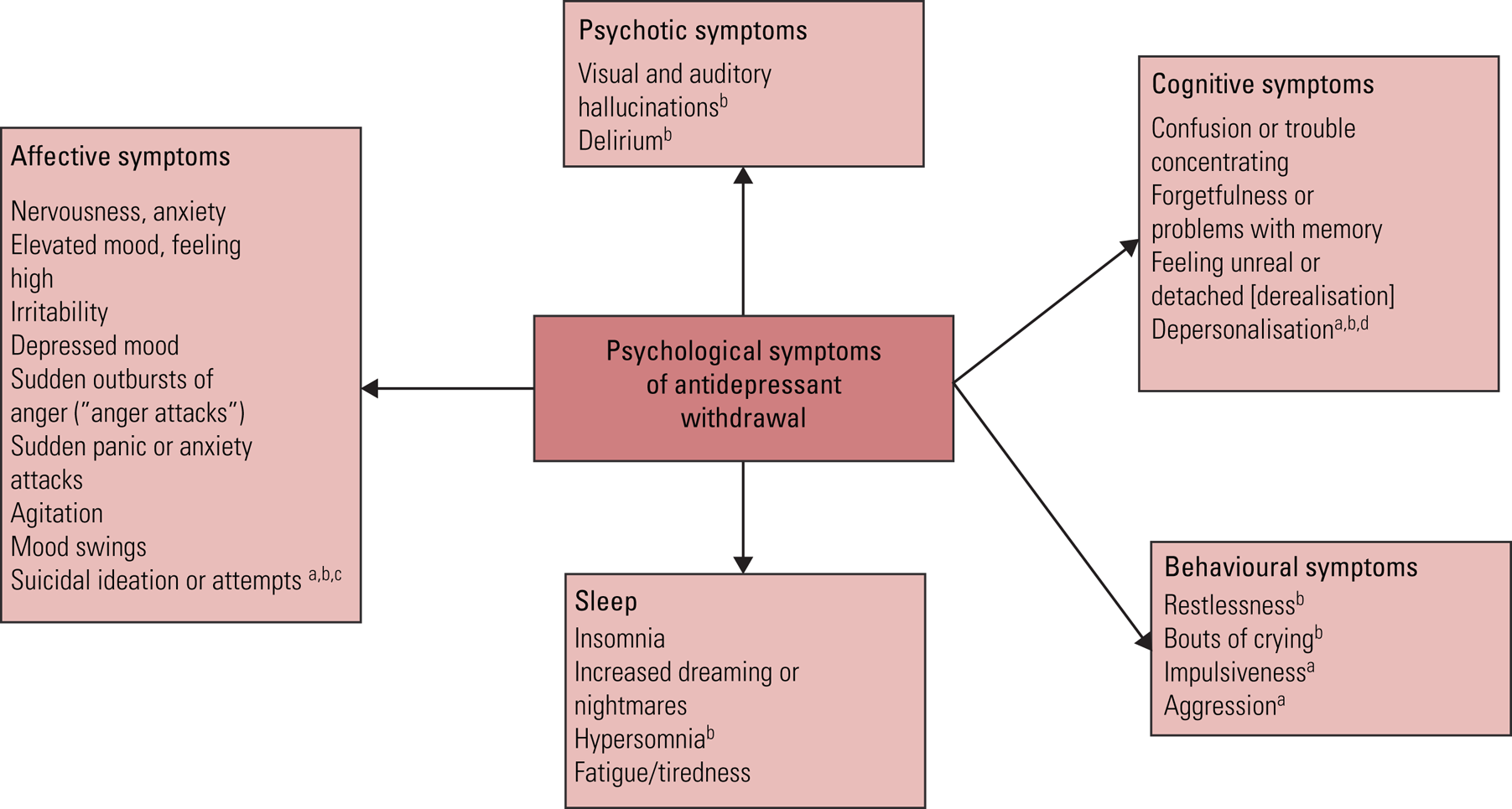
FIG 1 Psychological symptoms of antidepressant withdrawal.
Most of the listed symptoms are derived from the Discontinuation-Emergent Signs and Symptoms checklist (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). Symptoms derived from other sources are referenced individually: a, Fava et al (Reference Fava, Gatti and Belaise2015); b, Cosci & Chouinard (Reference Cosci and Chouinard2020); c, Valuck et al (Reference Valuck, Orton and Libby2009); d, Rusconi et al (Reference Rusconi, Carlone, Muscillo and Piccione2009).
Distinguishing withdrawal from relapse
When antidepressant withdrawal symptoms were widely believed to be mild and brief (as promulgated by most guidance), it was reasonable to conclude that any symptoms reported after stopping or reducing the dose of an antidepressant that were severe and/or long-lasting were likely to be relapse. With an updated understanding that withdrawal symptoms are common and can be severe and/or long-lasting, distinguishing these symptoms from relapse becomes more important.
Several authorities have previously highlighted the possibility of confounding relapse with withdrawal effects: nearly 15 years ago it was identified that antidepressant ʻdiscontinuation symptoms may be diagnosed as a relapse or recurrence of the underlying affective illness for which the antidepressant was originally prescribed' (Haddad Reference Haddad and Anderson2007) and the possibility of mistaking withdrawal for relapse had also been emphasised some years before this (Young Reference Young and Haddad2000). This confounding can have several negative consequences: namely, unnecessary long-term reinstatement of the antidepressant, with ongoing exposure to the adverse effects of the medication, and a more negative prognosis (Haddad Reference Haddad and Anderson2007). These consequences can result in patients internalising the notion that they require antidepressants and that they have a severe condition, and can lead to ʻsignificant social implications' (Haddad Reference Haddad and Anderson2007). There has been no systematic study of how often this misdiagnosis might occur in clinical practice but it is widely reported by patients (Guy Reference Guy, Brown and Lewis2020). Misdiagnosis of withdrawal effects as relapse by their doctors was a common motivation cited by 67 000 people who sought peer advice on social media sites on how to stop antidepressants (White Reference White, Read and Julo2021).
Distinguishing withdrawal from relapse in clinical practice
It has been proposed that there are three ways that withdrawal can be distinguished from relapse: timing, duration and response to medication reinstatement (Haddad Reference Haddad and Anderson2007). Haddad et al outline that withdrawal symptoms usually occur days after reducing or stopping an antidepressant, whereas relapse might be expected to occur weeks, months or years later. There is some variability in this – fluoxetine has a half-life of 7–15 days and so withdrawal symptoms might be delayed for weeks (Zajecka Reference Zajecka, Fawcett and Amsterdam1998). It has also been reported that symptoms of antidepressant withdrawal can be delayed even after short half-life antidepressants are stopped, although the mechanism is not well understood (Hengartner Reference Hengartner, Schulthess and Sorensen2020a). A short duration (1 day to 3 weeks) was previously thought to distinguish withdrawal symptoms from relapse; however, we now recognise that withdrawal symptoms can last for months or even years in some individuals (Davies Reference Davies, Pauli-Jones and Montagu2018; Stockmann Reference Stockmann, Odegbaro and Timimi2018; Hengartner Reference Hengartner, Schulthess and Sorensen2020a) and so this criterion is no longer as useful. Lastly, another means of distinction is the time taken for symptoms to resolve on reinstatement of the antidepressant: for withdrawal symptoms this will be more rapid than for relapse, as demonstrated by resolution within 1 week in one study (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). This also matches clinical experience – when antidepressants are recommenced soon after cessation. However, reinstatement may be less successful when it is delayed for weeks or months after the onset of withdrawal symptoms (Hengartner Reference Hengartner2020b).
There are also distinctive symptoms that mark out withdrawal symptoms from relapse. This is particularly important: physical and psychological symptoms that co-occur are more likely to be withdrawal symptoms. For example, if a patient were to experience a surge of anxiety and lowered mood on stopping an antidepressant, and this was also accompanied by nausea, vomiting or dizziness and electric shock sensations (‘zaps’, a commonly experienced neurological symptom of withdrawal affecting the head, and sometimes the limbs), we would much more readily conclude that these are withdrawal symptoms rather than relapse of a depressive condition. Although a bout of gastroenteritis co-occurring might explain some of these symptoms, it is more likely that one condition will cause several symptoms rather than that several conditions are co-occurring, using Occam's razor (Wildner Reference Wildner1999). Some symptoms are so distinctive – such as the electric ‘zaps’ in the head, often experienced on lateral eye movements, which can persist for months or years (Papp Reference Papp and Onton2018) – that they could be considered pathognomonic of antidepressant withdrawal. Physical symptoms typical of antidepressant withdrawal are shown in Fig. 2.
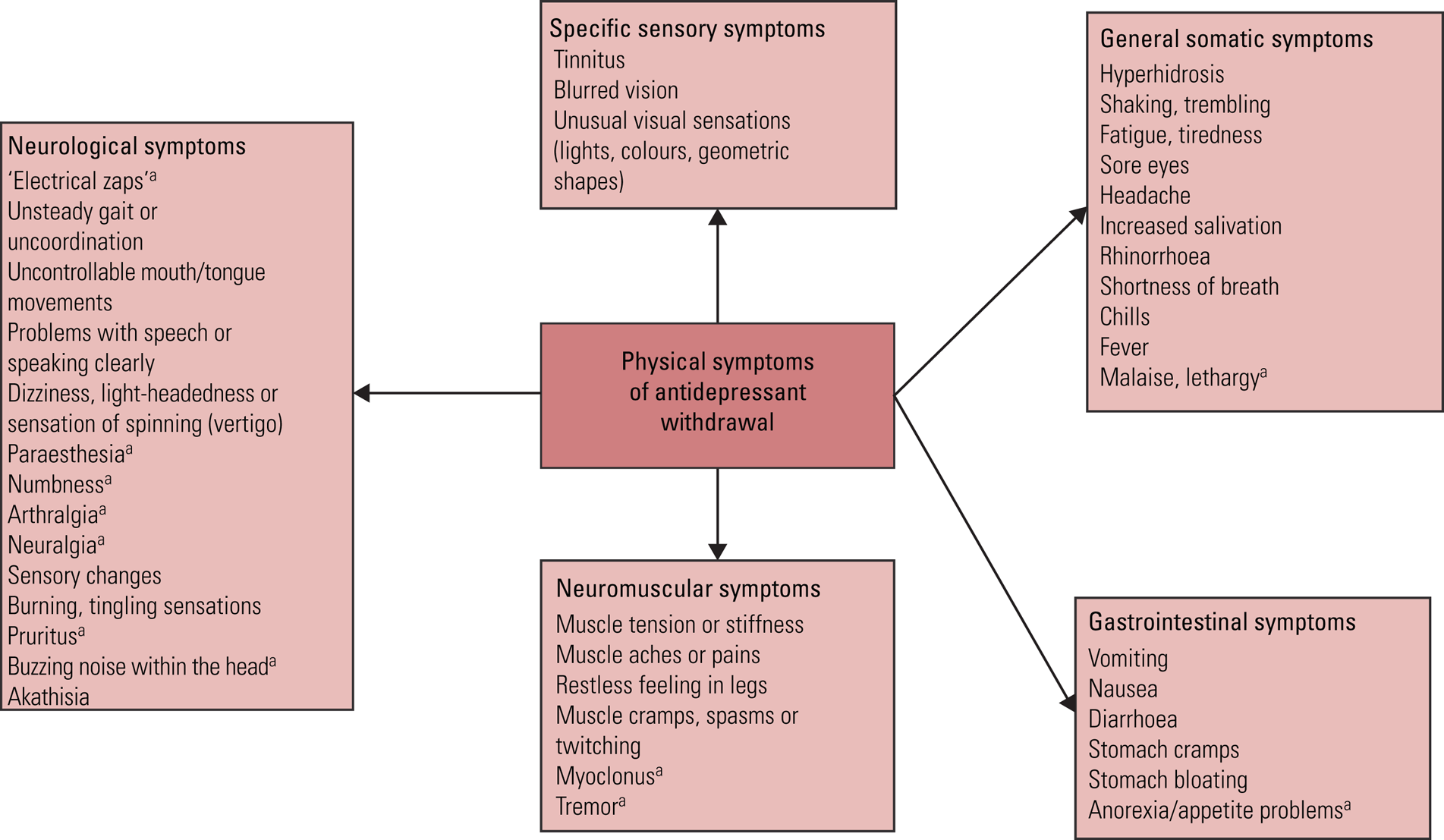
FIG 2 Physical symptoms of antidepressant withdrawal.
Most of the listed symptoms are derived from the Discontinuation-Emergent Signs and Symptoms checklist (Rosenbaum et al, Reference Rosenbaum, Fava and Hoog1998). a, symptoms derived from Cosci & Chouinard (Reference Cosci and Chouinard2020).
The timescale and pattern of withdrawal symptoms can also be distinctive. After a small reduction (e.g. 10% of the most recent dose) of an antidepressant, as currently suggested in RCPsych guidance (Burn Reference Burn, Horowitz and Roycroft2020), withdrawal symptoms normally follow a wave-like pattern similar to withdrawal effects from other drugs: onset a few days after the reduction, worsening, reaching a peak of intensity normally within a couple of weeks, before lessening in intensity and finally resolving usually within a fortnight or month (although with great variability among patients) (Fig. 3). Relapse or recurrence does not follow this clear wave pattern. In practice, careful monitoring of patients’ withdrawal symptoms makes this pattern evident and allows delineation of symptoms from relapse. The major distinguishing features between withdrawal and relapse are summarised in Table 2.
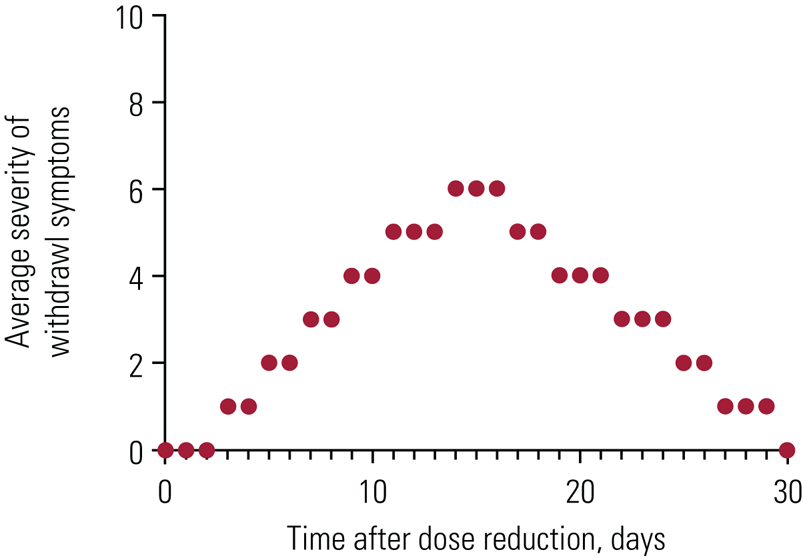
FIG 3 An approximate representation of withdrawal symptoms following a small reduction in antidepressant dose.
Note the wave-like properties of the relationship, whereby symptoms begin a few days after dose reduction and increase in intensity to reach a peak several days later, before lessening in intensity and resolving. A relapse is likely to be much more delayed in onset (although some withdrawal symptoms can also be delayed) and to not have this clear crescendo–decrescendo pattern over time. Note that bigger reductions in dose can lead to withdrawal symptoms that take much longer to resolve (including months or years) in some patients.
TABLE 2 Distinguishing features between antidepressant withdrawal symptoms and relapse of an underlying condition
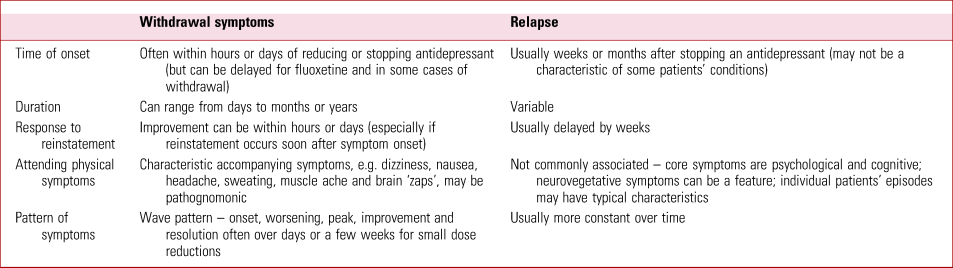
Of course, the physician should continue to be vigilant for genuine relapse of the patient's underlying condition, which may come on weeks or months after treatment is stopped and may have characteristics that are quite typical for the patient.
Clinical discontinuation trials
Withdrawal symptoms are likely mistaken for relapse in clinical trials
We now explore whether misdiagnosis of withdrawal symptoms as relapse can occur in clinical trials aimed at measuring the relapse prevention properties of antidepressants. The NICE guidelines on relapse prevention in depression (National Institute for Health and Care Excellence 2009), and the recent draft update, are primarily based on a meta-analysis of discontinuation trials by Geddes and colleagues (Geddes Reference Geddes, Carney and Davies2003), and other similar trials. In these trials, individuals with depression who have remitted on antidepressants are randomised to either continue taking antidepressants (the maintenance group) or to stop taking antidepressants and be switched to placebo (the discontinuation group). These two groups are then monitored with depression rating scales in order to detect relapse. The difference in relapse rates between the patients maintained on antidepressants and those discontinued is interpreted as the ability of the antidepressants to prevent relapse. The influential meta-analysis of these studies found that relapse rates were 41% in the discontinuation group and 18% in the maintenance group, which has been interpreted as an absolute risk reduction of 23% for antidepressants, or a relative risk reduction of about half (56%), findings similar to other meta-analyses (Glue Reference Glue, Donovan and Kolluri2010; Borges Reference Borges, Chen and Laughren2014). There are, however, several lines of evidence to suggest that some of the relapses diagnosed in these trials are misdiagnoses of withdrawal symptoms.
Rate of tapering employed in these studies
In these 31 studies, the most common method of tapering individuals from their antidepressants (which they had used for months or years) to placebo was abrupt cessation and the weighted mean of taper duration was 5 days. It has been demonstrated that tapering over 2 weeks does not reduce the risk of withdrawal symptoms compared with abruptly stopping (Baldwin Reference Baldwin, Cooper, Huusom and Hindmarch2006; Tint Reference Tint, Haddad and Anderson2008). The latest guidance suggests that for people who have been taking antidepressants for more than a few weeks, tapering should be at a rate that is tolerable to each individual, which can be months or years, likely depending on several factors, including the duration of previous treatment (Horowitz Reference Horowitz and Taylor2019; Burn Reference Burn, Horowitz and Roycroft2020; NICE Reference National Institute2021), so the tapering rate employed in the studies summarised in this meta-analysis are, by modern standards, rapid. The risk of withdrawal symptoms in these discontinuation studies is therefore considerable.
Withdrawal symptoms overlap with all commonly used depression rating scales
Change on depression rating scales is not specific for the detection or measurement of depression, given their measurement of a wide variety of symptoms. Withdrawal effects, both psychological and physical, overlap with the domains measured in depression symptom scores, including the two most commonly used symptom scales, the Hamilton Rating Scale for Depression (the HRSD or HAM-D) and the Montgomery–Åsberg Depression Rating Scale (MADRS) (Table 3). There are few categories in these scales that do not have corresponding antidepressant withdrawal symptoms.
TABLE 3 Symptom domains in the MADRS and HRSD and overlapping withdrawal symptoms derived from the DESS or other authoritative sources
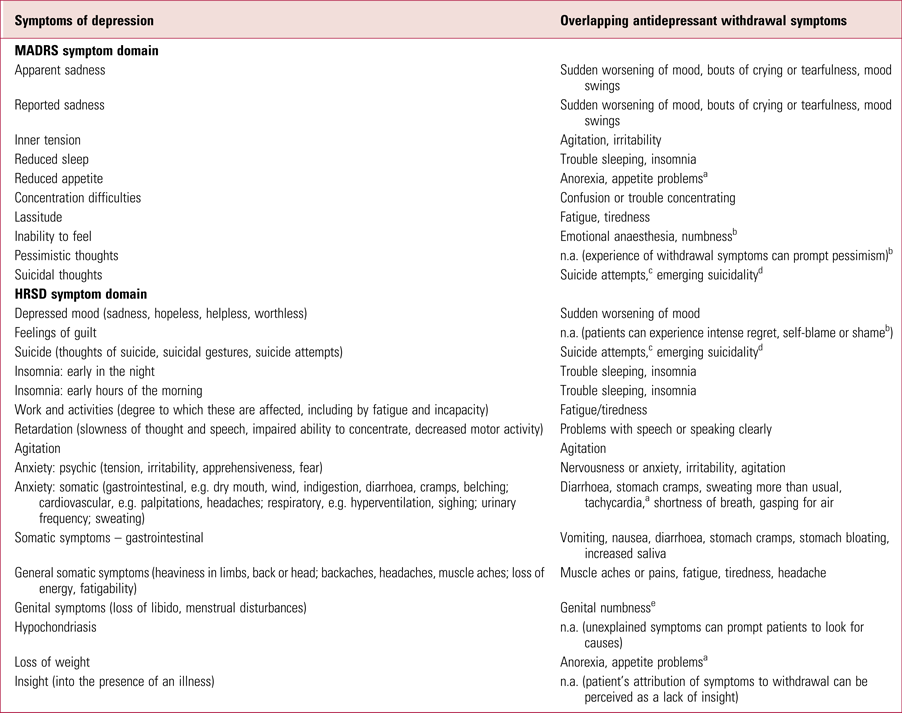
MADRS, Montgomery–Åsberg Depression Rating Scale (Montgomery Reference Montgomery and Åsberg1979); HRSD, Hamilton Rating Scale for Depression (Hamilton Reference Hamilton1960); DESS, Discontinuation-Emergent Signs and Symptoms checklist (Rosenbaum Reference Rosenbaum, Fava and Hoog1998); n.a., not applicable.
a Cosci & Chouinard (Reference Cosci and Chouinard2020).
b Framer (Reference Framer2021).
c Valuck et al (Reference Valuck, Orton and Libby2009).
d Hengartner et al (Reference Hengartner2020b).
e Reisman (Reference Reisman2020).
Furthermore, withdrawal symptoms have been clearly demonstrated to register as significant increases on depression rating scales. For example, in the Rosenbaum et al (Reference Rosenbaum, Fava and Hoog1998) study (Fig. 4) there are larger changes in depression scores for paroxetine (often implicated as the antidepressant associated with the most severe withdrawal syndrome) than sertraline, and minimal changes on all scales for fluoxetine. Both DESS symptoms and depression scores on the MADRS and HRSD reduced rapidly when antidepressants were reinstated (Fig. 4), consistent with withdrawal scores driving the increase in depression scores.
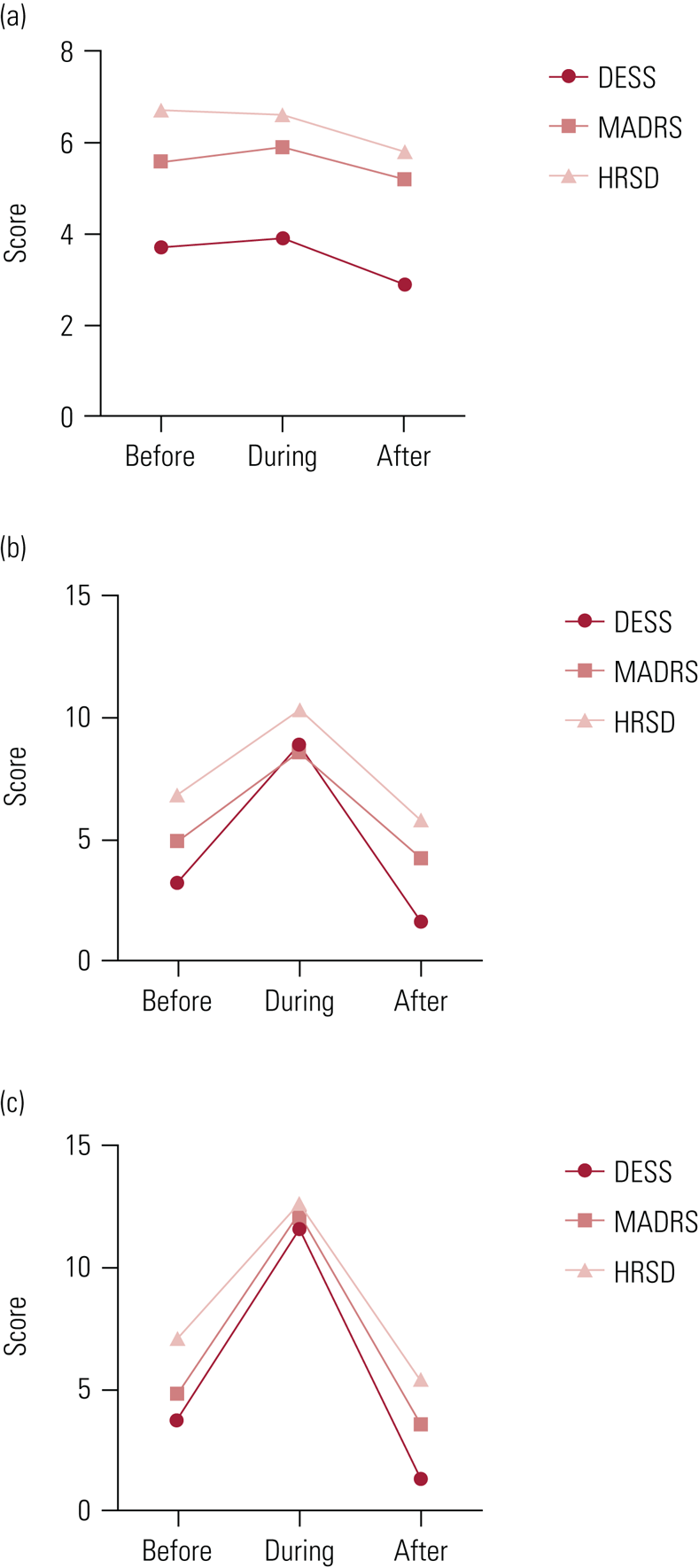
FIG 4 Withdrawal and depression symptom scores from the study by Rosenbaum et al (Reference Rosenbaum, Fava and Hoog1998).
Mean withdrawal symptom and depression scores for patients treated with (a) fluoxetine, (b) sertraline or (c) paroxetine before placebo substitution (‘before’), during placebo substitution (‘during’) and 1 week after reinstatement of the original antidepressant (‘after’). Withdrawal symptom were identified on the Discontinuation-Emergent Signs and Symptoms checklist (DESS) and depression was rated on the Montgomery–Åsberg Depression Rating Scale (MADRS) and Hamilton Rating Scale for Depression (HRSD). Note the close temporal relationship between onset and resolution of withdrawal symptoms and changes in depression scores.
Withdrawal symptoms are severe enough to meet the threshold for depression diagnosis on depression rating scales
It has been shown that withdrawal symptoms increase scores on depression rating scales such as the HRSD (Table 4) to a degree that would meet the criteria for diagnosis of relapse in studies for a portion of patients (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). The average HRSD score at baseline (while on antidepressants) in this study was about 7 points. The usual cut-off for depression relapse in discontinuation studies are HRSD scores >17, so an increase of 8 or more points is likely to meet the definition of relapse for a proportion of individuals; more so for those whose HRSD score increases by 10 or more points. From Table 4 it can be seen that about a third to a half as many patients as met criteria for ‘discontinuation syndrome’ would have met criteria for relapse in a discontinuation trial. We may consider fluoxetine to be a form of control group in this study (as there was not a significant increase in withdrawal symptoms for this drug): subtracting out the proportion of participants who experienced an increase in HRSD score of ≥8 points on the 8-item HRSD or ≥10 points on the 28-item HRSD on discontinuing fluoxetine (due to either natural variation or the nocebo effect) would slightly reduce the proportion of patients on sertraline or paroxetine who met these thresholds for pharmacological reasons. Recorded relapse rates in a discontinuation trial that observes participants for months may have been even greater than this because withdrawal symptoms can increase over time – perhaps especially relevant for the fluoxetine group.
TABLE 4 Patients who met criteria for ‘discontinuation syndrome’a and experienced an increase in score on the Hamilton Rating Scale for Depression for different antidepressants

HRSD-28 and HRSD-8, 28-item and 8-item Hamilton Rating Scale for Depression.
a Discontinuation syndrome: ≥4 symptoms on the Discontinuation-Emergent Signs and Symptoms checklist (DESS).
Source: Rosenbaum et al (Reference Rosenbaum, Fava and Hoog1998).
Discontinuation trials do not specifically measure withdrawal symptoms to distinguish them from relapse
Although there are difficulties in distinguishing withdrawal effects from genuine relapse in antidepressant discontinuation studies, this distinction could be achieved if, alongside depression rating scales used to detect relapse (e.g. the HRSD), withdrawal symptom scales (e.g. the DESS) were also employed. In this case participants who experience significant withdrawal effects could be excluded from assessment of relapse. However, antidepressant discontinuation trials did not usually consider withdrawal symptoms – none of the trials in the Geddes et al (Reference Geddes, Carney and Davies2003) review measured withdrawal symptoms specifically and only ten mentioned the possibility of withdrawal confounding the detection of relapse (mostly dismissing the possibility).
Relapse in the discontinuation arm occurs in the time period when withdrawal effects are most likely
Relapses cluster in the period just after participants are switched to placebo in the discontinuation arm of antidepressant discontinuation studies (Hengartner Reference Hengartner2020b). One analysis found that the entire difference in relapse rate between maintenance and discontinuation arms occurred in the first 6 weeks after randomisation, the period when withdrawal effects are most likely (Récalt Reference Récalt and Cohen2019). Interestingly, this effect was not evident for agomelatine, a medication thought not to be associated with a withdrawal effect. A more recent analysis has found that 70% of the difference between maintenance and discontinuation arms in antidepressant discontinuation trials (average duration 39 weeks) submitted to the US Food and Drug Administration occurred in the first 12 weeks, more than twice the proportion that would be expected if the prophylactic effect of antidepressants were ‘evenly spread’ throughout the follow-up period (Hengartner Reference Hengartner and Plöderl2021).
There are two points of view on whether the prophylactic effects of antidepressants should be ‘evenly spread’ throughout the follow-up period – that is, that antidepressants should be equally protective in the first few weeks as they are later in follow-up. Depression is normally conceptualised as an episodic condition in which antidepressants are thought to prevent relapse. If the antidepressants are removed, and withdrawal effects are not pertinent, then the natural history of the episodic disorder should be exposed: there should be no reason why patients should relapse at the same time if they each have their own pattern of episodic relapse. It follows then that if there is a disproportionate preponderance of relapses soon after stopping antidepressants then it is likely that the consequences of the process of stopping itself (i.e. withdrawal effects) is the plausible cause. This has, for example, been demonstrated for lithium where individuals with bipolar disorder stopping lithium were seven times quicker to relapse than those who had never been treated with lithium (Suppes Reference Suppes, Baldessarini and Faedda1991).
The alternative explanation for early relapses is that patients are prone to early relapse because of the removal of the stabilising effect of the antidepressants. One version of this rationale argues that partially treated patients relapse soon after medication is discontinued, leading to a high-risk period for relapse soon after stopping (Jauhar Reference Jauhar, Hayes and Goodwin2019a). However, participants in these discontinuation studies are selected for having remitted and meta-analyses do not find a difference in relapse rates for participants with longer stabilisation periods before discontinuation (Geddes Reference Geddes, Carney and Davies2003; Glue Reference Glue, Donovan and Kolluri2010; Borges Reference Borges, Chen and Laughren2014), which would be thought to provide more complete treatment. On the other hand, the reduction in relative risk of relapse after antidepressant cessation was similar in magnitude between 12–36 months and 0–12 months in the Geddes review and this is not consistent with withdrawal effect confounding (although it must be noted that the 12–36 month period only included 6 studies, with 35 relapses, so this captures relatively few patients). Notably, agomelatine, which has not been found to be associated with a withdrawal syndrome, fails to demonstrate significant relapse prevention properties when unpublished data are taken into account (Koesters Reference Koesters, Guaiana and Cipriani2013).
Overall, there are two possible reasons for worsening symptoms when stopping medication: either uncovering of the underlying disorder or withdrawal effects (although withdrawal effects might precipitate genuine relapse this should be seen as a withdrawal-related effect). It seems to us that if relapses are clustered soon after stopping then this seems to fit more with a withdrawal effect. This dilemma could be resolved by stopping antidepressants gradually enough (i.e. over months or longer) to prevent withdrawal effects and observing the pattern of relapse.
The degree to which antidepressants prevent relapse is uncertain
Withdrawal symptoms are common: 53.9% in RCTs from the recent systematic review (Davies Reference Davies and Read2019) and 31% in very conservative estimates (Jauhar Reference Jauhar and Hayes2019b). Rates of relapse of depression after cessation of antidepressants are less certain. The Geddes meta-analysis found relapse rates of 41% in the discontinuation arm, 23% greater than in the maintenance arm. However, as outlined above, some proportion of these relapses are likely to be due to withdrawal effects: the original authors themselves concluded that ʻwe cannot exclude the possibility' (Geddes Reference Geddes, Carney and Davies2003). It is hard to estimate what proportion of these relapses might be due to withdrawal confounding but the data presented in Table 4 suggest that perhaps one-third to one-half of the 31–53.9% of patients who experience withdrawal effects (as estimated by different analyses of RCTs) may reach the threshold required for detection of relapse given an 8- or 10-point increase in HRSD-8 or HRSD-28 score (Rosenbaum Reference Rosenbaum, Fava and Hoog1998). This suggests that approximately 10–27 percentage points of relapses, or a substantial proportion of the difference in relapse rates between the maintenance and discontinuation arms in these studies, may be attributable to withdrawal – although there is considerable uncertainty in these estimates.
Therefore, the relapse prevention studies summarised in Geddes et al (Reference Geddes, Carney and Davies2003) may be demonstrating, at least partially, the effect of abrupt (or near-abrupt) cessation, through either withdrawal effects or destabilising participants’ conditions rather than the benefits of continuing treatment (Baldessarini Reference Baldessarini and Tondo2019). Further study is required to confirm the magnitude of the relapse prevention properties of antidepressants by evaluating the degree to which withdrawal confounds relapse detection in these studies. The major lines of evidence suggesting that withdrawal effects confound detection of relapse in antidepressant withdrawal studies are summarised in Box 2.
BOX 2 Lines of evidence suggesting withdrawal symptoms are misdiagnosed as relapse in antidepressant discontinuation trials
• Antidepressants are stopped either abruptly or very rapidly in discontinuation studies, making withdrawal symptoms likely (although not all people will experience withdrawal symptoms)
• Withdrawal effects include psychological and physical symptoms which overlap with almost every domain of the depression scales commonly used to detect relapse (e.g. Hamilton Rating Scale for Depression, HRSD; Montgomery–Åsberg Depression Rating Scale, MADRS)
• Withdrawal effects are not generally measured in discontinuation studies and so no robust attempt is made to distinguish them from relapse
• Relapses tend to occur in the first few weeks following antidepressant discontinuation, a pattern consistent with withdrawal effects (although this may also represent early relapse of partially treated patients)
• When antidepressants are stopped abruptly/rapidly a substantial proportion of patients will experience withdrawal symptoms that meet the threshold for registering as relapse according to scores on the HRSD or MADRS (although it is not clear what proportion this is true for)
Conclusions
Clinical practice
Clinicians should have a high index of suspicion for withdrawal symptoms when patients report anxiety or depression on stopping antidepressants (Box 3). It is not clear whether withdrawal symptoms are more common than genuine relapse. Clinicians should take care to distinguish between relapse and withdrawal symptoms by exploring whether or not reported affective symptoms are accompanied by other symptoms not associated with relapse: for example, dizziness, electric ‘zaps’, nausea, profound insomnia, or any symptoms that were not present in the baseline condition. Some symptoms, such as electric ‘zaps’, may be considered pathognomonic of withdrawal. Clinicians should ask about the timing of symptoms – if they occur within days of reduction or stoppage of an antidepressant with a short half-life (essentially all but fluoxetine) and if the symptoms resolve within a few days of increasing the dose of an antidepressant, then they are likely to be withdrawal symptoms. When advising patients to stop or reduce their dose of antidepressant clinicians should suggest small reductions, monitor for withdrawal symptoms and wait 2–4 weeks for withdrawal symptoms to resolve before making further reductions (Horowitz Reference Horowitz and Taylor2019; Burn Reference Burn, Horowitz and Roycroft2020). This controlled process can help reduce the uncertainty that may arise in distinguishing between withdrawal symptoms and relapse, as withdrawal symptoms are likely to begin, worsen, peak, improve and resolve over a number of days or short number of weeks (if reductions are modestly sized) as distinct from episodes of relapse, which may persist for much longer.
BOX 3 Distinguishing between relapse and withdrawal symptoms following antidepressant cessation or dose reduction
When a patient reports low mood, anxiety or insomnia following dose reduction or stopping an antidepressant the clinician should:
• in addition to considering the possibility of relapse, hold a high index of suspicion for antidepressant withdrawal symptoms, as these are common
• inquire about the symptoms of the original condition: are they different from the symptoms that are currently reported?
• inquire about the presence of symptoms indicative of withdrawal syndrome such as electric shock (‘zap’) sensations in the head, dizziness, nausea, headache (and other symptoms listed in Figs 1 and 2).
• inquire about the timing of these symptoms – did they arise a few days after stopping an antidepressant (or weeks after stopping fluoxetine)?
• inquire about the pattern of these symptoms – have they continued to worsen in the days and weeks after stopping or reducing the antidepressant?
• inquire about past experience of stopping antidepressants – have similar symptoms occurred?
• if the patient has trialled an increase in dose, did this lead to a lessening of these symptoms? How long did this improvement take?
• guided by Table 2, make a diagnosis of antidepressant withdrawal syndrome if it is concluded that a withdrawal syndrome is likely
• following guidance from the RCPsych (Burn Reference Burn, Horowitz and Roycroft2020), suggest increasing the dose back to the last dose at which the patient was stable, allow a period of stabilisation and then suggest reduction in a more gradual manner than previously tried
Discontinuation studies
Current discontinuation studies that are thought to demonstrate relapse prevention properties of antidepressants are likely to be confounded by withdrawal symptoms. These studies often stop antidepressants very quickly and do not measure withdrawal symptoms in order to distinguish them from relapse. As withdrawal symptoms overlap significantly with symptoms commonly used on depression rating scales and can cause an increase in depression scores large enough to meet the threshold for diagnosis of relapse, withdrawal symptoms are likely to increase the detected rates of relapse in the discontinuation arms of these trials, inflating the apparent ability of antidepressants to prevent relapse. This notion is strengthened by the preponderance of relapses detected in the weeks after cessation, the period in which withdrawal symptoms from antidepressants are most common. Overall, existing studies that purport to demonstrate relapse prevention properties of antidepressants should be interpreted with caution, and further evidence is required to definitively demonstrate the magnitude of relapse prevention properties of antidepressants.
Future studies are required to further clarify the incidence, severity and duration of antidepressant withdrawal symptoms for different antidepressants in order for us to be able to better inform patients of risks when starting or stopping an antidepressant. In future, studies that seek to evaluate the relapse prevention properties of antidepressants should explicitly measure withdrawal symptoms and the occurrence of withdrawal symptoms should be considered an exclusion for the diagnosis of relapse (on the premise that one condition producing multiple symptoms is more likely than more than one condition co-occurring). To avoid confounding by withdrawal effects, discontinuation studies should involve slow tapers – which we now understand should be over months or years in some people – so that withdrawal symptoms are excluded as confounders of relapse: this would allow the genuine relapse prevention properties of antidepressants to be evaluated. Another approach would be to conduct trials comparing patients commenced on antidepressants or placebo over long periods of continuation to assess for relapse. As the current NICE guidelines on relapse prevention are based on findings from the meta-analysis of discontinuation studies (Geddes Reference Geddes, Carney and Davies2003) about which there is great uncertainty, the guidelines may need to be reconsidered.
Author contributions
M.A.H. conceived the idea for the article and wrote the manuscript. D.T. made substantial contributions to the analysis, concepts and form of the article.
Funding
This article received no specific grant from any funding agency, commercial or not-for-profit sectors.
Declaration of interest
D.T. reports grants from Janssen, personal fees from Janssen, grants from Recordati, personal fees from Sunovion and personal fees from Otsuka, outside the submitted work.
MCQs
Select the single best option for each question stem
1 Which of the following would suggest that symptoms on stopping an antidepressant are relapse of the underlying condition rather than withdrawal symptoms?
a Symptoms come on 2 days after stopping paroxetine, including anxiety, trouble sleeping, panic attacks and electric ‘zaps’ on moving the eyes
b A patient stops fluoxetine over 12 months, with minimal withdrawal symptoms; 2 years later they report low mood and low energy similar to a depressive episode they have experienced in the past
c A patient stops fluoxetine over 3 months without apparent problems; 6 weeks later they complain of anxiety, trouble sleeping, dizziness and having trouble concentrating at work. Their original condition was depression characterised by low mood and insomnia
d A patient stops sertraline abruptly after deciding they do not need the medication. A week later they present to the emergency department complaining of suicidal thoughts. They also feel nauseous and dizzy.
e A patient halved their dose of venlafaxine 2 weeks ago down to 37.5 mg. They complain of headache, trouble focusing, fatigue, irritability and dizziness.
2 Which of the following is it not consistent with the fact that relapse is likely to be confounded by withdrawal effects in trials of antidepressant discontinuation?
a Withdrawal symptoms overlap with most categories on commonly used depression rating scales
b Most relapses occur in the first few weeks after stopping antidepressants, and reduce in frequency after this
c These trials do not routinely measure withdrawal effects in order to distinguish them from relapse
d Antidepressants are stopped abruptly or very quickly in these trials
e Some patients have had multiple episodes of depression previously.
3 Which of the following is not helpful to ask a patient to help distinguish antidepressant withdrawal from relapse?
a How long after stopping an antidepressant symptoms came on
b Whether they experienced dizziness, nausea, vomiting, electric ‘zaps’
c What were their original symptoms that prompted the prescription of antidepressants
d How long their symptoms have lasted for
e Whether they experienced these symptoms previously on stopping an antidepressant.
4 Which of the following is not usually a symptom of antidepressant withdrawal?
a Anxiety
b Depressed mood
c Dizziness
d Fatigue
e Chest pain.
5 How common are withdrawal symptoms from antidepressants?
a Every patient who stops antidepressants will experience them
b They are very rare
c About a third to half of patients will experience withdrawal symptoms
d It is likely that less than a third of patients will experience them
e It is likely that much more than a half of patients of patients will experience them.
MCQ answers
1 b 2 e 3 d 4 e 5 c












eLetters
No eLetters have been published for this article.