Epidemiological studies indicate that the consumption of nuts is inversely related to risk of CHDReference Fraser, Sabate, Beeson and Strahan1, Reference Hu, Stampfer, Manson, Rimm, Colditz, Rosner, Speizer, Hennekens and Willett2. Subsequent studies have provided a mechanistic basis for these observations and indicate that the regular consumption of nuts lowers plasma concentrations of LDL- and oxidised-LDL cholesterol while preserving HDL-cholesterolReference Spiller, Jenkins, Bosello, Gates, Cragen and Bruce3, Reference Lovejoy, Most, Lefevre, Greenway and Rood4. These data provide a rationale for encouraging nut consumption among the population and are the basis for an FDA (Food and Drug Administration)-approved health claim relating to risk of heart disease.
However, the habitual inclusion of nuts, a high-fat food group, in the diet may promote weight gain which would potentially negate the beneficial effects on blood lipid profiles. Epidemiological studies challenge this concern, revealing no or a negative association between nut consumption and body weightReference Fraser, Sabate, Beeson and Strahan1, Reference Hu, Stampfer, Manson, Rimm, Colditz, Rosner, Speizer, Hennekens and Willett2. These findings are supported by intervention studies demonstrating that the inclusion of nuts in the diet poses limited risk for significant weight gainReference Lovejoy, Most, Lefevre, Greenway and Rood4, Reference Fraser, Bennett, Jaceldo and Sabate5. A mechanistic study exploring how the inclusion of nuts in the diet fails to promote positive energy balance and weight gain has not been conducted.
Three explanations for the lack of expected weight gain noted in nut consumers have been proposed. First, constituents of almonds such as fibre or protein are associated with increased sensations of satietyReference Porrini, Crovetti, Testolin and Silva6–Reference Benini, Castellani and Brighenti8. Therefore, the energy contained in the consumed nuts may be offset by reductions in energy intake from other foods so that overall energy intake remains unchanged. Secondly, previous research has suggested that the regular consumption of nuts may lead to increases in energy expenditureReference Alper and Mattes9. This would, in part, adjust for the energy contributed by nuts. Thirdly, the energy content of nuts that is bioaccessible may be less than predicted as faecal fat excretion increases with consumptionReference Sabate10. One study using almonds demonstrated that this is due to the cell wall's resistance to enzymatic degradation in the gastrointestinal tract which results in the encapsulation of fatReference Ellis, Kendall, Ren, Parker, Pacy, Waldron and Jenkins11. The present study explored these mechanisms by measuring the effect of consuming a 1440 kJ portion of almonds, each day for 10 weeks, on energy intake, diet composition, energy expenditure, faecal energy excretion, body weight and body composition.
Subjects and methods
Eligibility criteria included being of good health, a non-smoker, weight stable (deviation of < 2·5 kg over the previous 3 months), BMI of 23–30 kg/m2, using no medications that would interfere with the outcome measures and obtaining less than 5 % of daily energy from tree or ground nuts. On being accepted into the study, participants were randomised to one of two conditions, almond or control, using a computerised random number generator. The almond group was required to eat a 1440 kJ portion of raw, unsalted almonds each day for 10 weeks whereas the control group followed their usual diet. After the first 10 weeks and a subsequent 3-week washout period, the two groups crossed over so that the almond group became the control group and vice versa. The participants were not given advice about how to incorporate the almonds in the diet and were not instructed to reduce their food intake from other sources. During both conditions, all participants underwent the same measurement procedures. Data were collected between December 2003 and August 2005.
Body weight was measured on a random morning of each week of the study. For this measure, the overnight fasted participant voided and wore a surgical gown. All body weight measurements were made on the same calibrated electronic scale. Fat mass and fat free mass were determined by air displacement plethysmography (Bod-Pod, Life Measurement, Inc., Concord, CA, USA) during weeks 1 and 10 of each study arm. The bod-pod has been validated as a sensitive measure to detect small to moderate body composition changesReference Dempster and Aitkens12.
Resting metabolic rate (RMR) and the thermic effect of food (TEF) were measured by indirect calorimetry using a SensorMedics Vmax 29 n metabolic cart (SensorMedics, Anaheim, CA, USA). The instrument was calibrated before each measurement using known concentrations of gas. RMR measurements were made during weeks 1 and 8 of each study arm. The TEF measure was made during week 8 of each study arm. Participants were required to fast for 12 h and avoid alcohol or heavy exercise in the 24 h prior to measurement. On the test morning, participants were asked to report to the laboratory as soon as possible after waking and by a method of minimal energy expenditure. Upon entering the thermo-neutral laboratory, participants were asked to lie supine and as still as possible on a bed for 20 min. After the rest period, a clear plastic ventilated hood was placed over the participant's head for a period of 45 min. The last 15 min of measurements were used to determine the RMR.
To determine the TEF, an RMR measurement was made before participants consumed a 1672 kJ portion of almonds and 250 ml of water within a 15 min period. TEF was estimated by energy expenditure measurements made for 15 min of each hour over the following 6 h. During this time, the participant was required to remain in a supine position and was asked to remain as stationary as possible.
Physical activity was measured using a three-axis accelerometer (Stayhealthy Inc., Monrovia, CA, USA), and a 3 d activity logReference Bouchard, Tremblay, Leblanc, Lortie, Savard and Theriault13. The accelerometer was worn on the waist from the time of rising in the morning to going to bed at night, and has been validated as a reliable method for measuring physical activityReference Nichols, Morgan, Sarkin, Sallis and Calfas14. The activity log was split into 15 min sections and covered the whole 24 h period. Participants recorded the type and intensity of activities they engaged in for each 15 min period. These measures were made on three random days (two weekdays and a weekend day) during weeks 4 and 8 of each study arm.
Total energy expenditure was measured using doubly labelled water administered during week 8 of each study arm. On the first day of each assessment period, a baseline urine sample was collected from the overnight fasted participant followed by oral dosing with doubly labelled water. The individualised dose was determined by the estimated total body water (TBW) pool size. H218O and D2O were administered at 2·5 g/kg estimated TBW (10 atom % excess) and 0·1 g/kg estimated TBW (99·8 atom % excess), respectively. Participants were then instructed to refrain from eating or drinking for the next 4 h. Following this 4 h period, an additional urine sample was collected, with further urine collections at 24 h, 7 d and 14 d.
Energy intake was measured on three random days of week 4 and 8 with the stipulation that the recording include two weekdays and one weekend day. Participants were interviewed in person or over the telephone to determine the previous day's food intake. The standardised interviews were conducted using NDS multipass software (University of Minnesota, Minneapolis, MN, USA).
The metabolisable energy of the almonds was determined by a controlled feeding study. Participants were required to report to the laboratory, following a 12 h fast, to eat all their meals on four consecutive days during week 10 of each study arm. All meals were provided and they were consumed in the laboratory dining area. The same menu was used during both arms of the study. The baseline diet provided between 10 500 and 12 000 kJ, depending on the body size of the participant, and was comprised of approximately 55 % carbohydrate, 35 % fat and 15 % protein. The nutrient content of the supplied diet was calculated using USDA (United States Department of Agriculture) nutrient tables. During the almond arm, participants also consumed 1440 kJ of almonds each day.
With the first meal of the controlled feeding period, participants ingested a food colour marker (blue). They were then instructed to collect all faeces in separate containers until further notice. With the final meal of day 4, participants were provided with another food colour marker (red) and asked to monitor their collections for its appearance. Faecal composites were made and a sample was freeze-dried before analysis for gross energy by bomb calorimetry (Parr Instruments, Moline, IL, USA). The digestibility coefficient of the diet was calculated as:
Compliance with the protocol was determined in several ways. First, regular contact (weekly) was made with the participants where an informal discussion relating to the almond consumption was conducted. A considerable effort was made to establish a relationship of trust with the participant and it was emphasised that it was vital that non-compliance, for whatever reason, was reported to the investigator. It was stressed that there were no negative consequences of such reports. Secondly, six diet dairies were completed during the study. Thirdly, fasting plasma α-tocopherol concentrations were measured during week 1 and week 10 of each study arm. Plasma α-tocopherol levels were assayed by reverse phase high-performance liquid chromatography. Participants were informed that the blood drawn contained a metabolite that would confirm long-term almond consumption.
All data are reported as means (sd). Mean values were compared using a Student's t test or a repeated measures ANOVA. Statistical significance was set at P < 0·05, two-tailed. This study was approved by the Purdue University Institutional Review Board and all participants signed an informed consent form.
Results
Twenty-four individuals were randomised to treatment groups. Four individuals failed to finish the study. It was not possible to ascertain their reason. All subjects lost to the study withdrew within the first 4 weeks of the commencement of the study. The mean age of the study group was 24 (sd 9) years. Details of the study group's physical characteristics are provided in Table 1.
Table 1 Changes in body composition due to the consumption of 1440 kJ/d of almonds
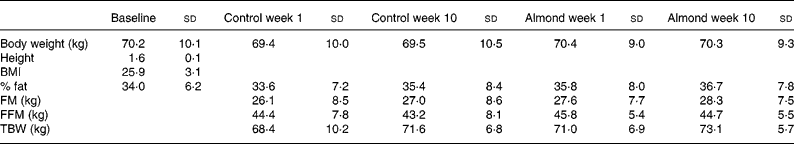
FM, fat mass; FFM, fat free mass; TBW, total body water.
Plasma α-tocopherol concentrations increased significantly following the 10 week almond treatment period by 21·6 % (P < 0·05). As almonds are a rich source of this vitaminReference Jambazian, Haddad, Rajaram, Tanzman and Sabate15, this was interpreted as evidence that the participants were compliant with the requirement to consume the almonds.
Data on body composition over the study are presented in Table 1. There were no significant changes in body weight (F(3,95) 0·66, P>0·05), percent fat (F(3,94) 0·310, P>0·05), fat mass (F(3,94) 0·500, P>0·05), fat free mass (F(3,93) 0·197, P>0·05) or TBW (F(3,93) 0·256, P < 0·05).
The inclusion of 1440 kJ of almonds in the daily diet did not lead to a statistically significant increase in food intake at any time point (F(3,76) 0·311, P>0·05) (Table 2). Fat intake increased significantly during the almond treatment period (F(3,76) 4·648, P < 0·05). No difference in protein intake (F(3,76) 0·206, P>0·05) was detected. The additional energy in the diet derived from fat was accounted for by a non-significant reduction in carbohydrate intake (F(3,76) 0·586, P>0·05) and a non-significant increase in energy intake (F(3,76) 0·311, P>0·05). While not significant, energy intake was greater by 322 kJ during the almond supplementation period. This suggests that dietary compensation for the energy contained in the almonds accounted for 74 % of the energy in the almonds.
Table 2 Energy, macronutrient and micronutrient intake measured during the control period and when 1440 kJ/d of almonds were consumed

* P < 0·05.
Almond ingestion was associated with significant increases in the intake of PUFA (F(3,77) 4·369, P < 0·05) and MUFA (F(3,77) 16·875, P < 0·05) (Table 2). There was no significant change in the intake of saturated fatty acids. There were also significant increases of vitamin E (F(3,77) 35·623, P < 0·05) (Table 2), magnesium (F(3,77) 18·311, P < 0·05) and copper (F(3,77) 9·365, P < 0·05) Table 2).
No significant changes of RMR, TEF (Table 3) or physical activity as measured by the accelerometer or activity diary (P>0·05) were observed. However, there was a significant difference between the two methods for measuring physical activity, with the activity diary indicating a higher value. Total energy expenditure, as measured by doubly labelled water, did not differ between the two study periods.
Table 3 Energy expenditure during the control period or when 1440 kJ/d almonds were being consumed
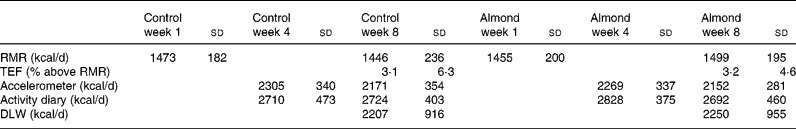
RMR, resting metabolic rate; TEF, thermic effect of food; DLW, doubly labelled water.
Eating almonds led to a significant decrease in the digestibility coefficient of the diet (control = 0·96, almond = 0·95, P < 0·05). This effect accounts for approximately 84 kJ/d of the energy contained in the daily almond portion. It was not possible to ascertain whether this decrease in digestibility was due to increased fat excretion.
Table 4 summarises the routes through which the energy in the 1440 kJ/d almond supplement was offset. Although the changes of energy expenditure were not statistically significant, based upon mean values, 95 % (based on doubly labelled water estimate of energy expenditure) to 98 % (based on the sum of the resting energy expenditure (REE), TEF and physical activity estimates) of the energy from the almond load was compensated.
Table 4 Details regarding the routes through which energy from the almond supplement was dissipated; note these mean increments in energy expenditure were not statistically significant
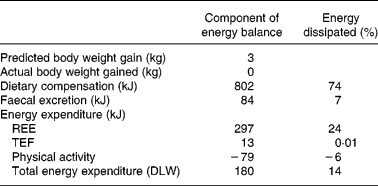
TEF, thermic effect of food; DLW, doubly labelled water.
Discussion
These data indicate that the consumption of a 1440 kJ serving of almonds each day for 10 weeks does not promote weight gain or changes in body composition. The increase in plasma α-tocopherol concentrations suggests that study participants consumed the almonds during the intervention study period.
The predominant mechanism through which the energy contained in the almonds was offset involved a spontaneous reduction of caloric intake from other dietary sources. The compensatory dietary response was 74 % of the energy contributed by the almonds. However, it is likely that the dietary recalls were subject to significant under-reporting as energy intake was significantly below the total energy expenditure as measured by doubly labelled water. Diet recalls are subject to several errors including under-reporting, due to forgotten foods or underestimated portion size, and altered eating behaviour due to participant's knowledge they are being observed. While this present study's use of a within-subjects design minimises these errors, they are still present and the diet recall data should be interpreted cautiously. One method to confirm the degree of dietary compensation would have been to include a laboratory-based test where the participant was given a 1440 kJ almond pre-load followed by an ad libitum buffet meal. However, due to the removal of the normal additional factors that influence food intake, the external validity of such results is uncertain.
Why almonds promote strong dietary compensation is still unclear. Almonds contain a significant amount of protein (21 g per 100 g of almonds), a macronutrient associated with increased satiationReference Porrini, Crovetti, Testolin and Silva6, Reference Weigle, Breen, Matthys, Callahan, Meeuws, Burden and Purnell16. However, studies that demonstrate a satiating effect of protein use a far greater protein load than the 11 g supplied by the almonds in this study. Indeed, due to the compensatory dietary response to almonds, there was only a net 5 g increase in protein intake. Such a small increment in protein intake is unlikely to have a marked effect on appetite. Almonds are a significant source of fibre, a food constituent with documented satiating propertiesReference Slavin17. However, it is also unclear that the 6·5 g of fibre contained in almonds could have a marked effect on appetitive sensations and food intakeReference Burley, Leeds and Blundell18. The crunchy textural property of almonds could also promote satietyReference Sakata, Yoshimatsu, Masaki and Tsuda19. It is possible a synergy between these, and possibly other, properties (e.g. micronutrients, flavour) of almonds account for their high satiety value.
Another possibility is that the participants cognitively adjusted their energy intake. While this cannot be dismissed, there are reasons to believe it was not the case. First, the participants were given the almonds in packets with no indication of their caloric value. Secondly, the study group consisted of predominantly overweight individuals who, by definition, fail to match energy intake to expenditure accurately.
The consumption of a 1440 kJ portion of almonds each day also had some beneficial effects on diet quality. During the almond-supplemented period, intake of MUFA and PUFA increased, which may have a positive effect on blood lipid profilesReference Wahrburg, Martin, Sandkamp, Schulte and Assmann20. Saturated fatty acid concentrations remained unchanged. Other notable changes in diet quality were an increase in vitamin E, magnesium and copper. These changes in diet quality are noteworthy as mean vitamin E intake was only 7 mg/d, substantially lower than the RDA of 15 mg/d. Indeed, only one individual consumed the RDA. During the almond supplementation period, the mean intake of vitamin E was approximately 20 mg/d and every individual consumed more than the RDA. Average intakes of copper and magnesium were also below recommended levels. The addition of almonds to the diet brought the mean group intake above recommended levels.
This study did not confirm previous results in lean and obese individuals, using peanuts, that there is a significant increase in RMR following chronic nut consumptionReference Alper and Mattes9, Reference Coelho, de Sales, Iyer, Bressan, Costa, Lokko and Mattes21. As body weight is the predominant determinant of RMR, it is unclear whether the previous results are anomalous or are specific to peanuts, particularly as another study with almonds also did not observe changes of RMRReference Fraser, Bennett, Jaceldo and Sabate5. Further research is required to clarify the effects of nuts on energy expenditure. In addition, the present study, consistent with othersReference Alper and Mattes9, did not find an effect of habitual almond consumption on TEF. Indeed, the measured TEF following almond consumption was lower than may have been predictedReference Eisenstein, Roberts, Dallal and Saltzman22.
As reported previouslyReference Sabate10, this study provides further evidence that the energy accessible from almonds may be lower than predicted. A decrease in the digestibility coefficient of the diet was noted during the almond ingestion period. This accounted for approximately 84 kJ of the 1440 kJ contained in the daily almond portion. This small amount of energy loss would have only a limited independent effect on body weight.
The 1440 kJ contained in the daily portion of almonds were almost fully offset by changes in energy intake, energy expenditure and faecal fat absorption. These indices accounted for approximately 95–98 % of the daily almond load. Given the measurement errors associated with each of these indices, this accounting must be viewed with some caution. However, it is consistent with the noted stability of body weight over the trial.
In summary, the present study demonstrates that the intake of 1440 kJ of almonds daily for 10 weeks does not promote weight gain. Indeed, other work suggests that almond consumption may aid weight lossReference Wien, Sabate, Ikle, Cole and Kandeel23, probably by increasing compliance with an energy-restricted diet. The effects of slightly lower levels of intake (i.e. 1045 kJ/d), such as proposed in the health claim for nuts, over an extended period of time remain to be evaluated. More broadly, these findings highlight the potential error of questioning the health effects of high-energy dense foods based on this property alone.
Acknowledgements
This work was funded by the Almond Board of California.






