Above the fundamental roles of physical activity and a macronutrient-balanced diet in the maintenance of a healthy body weight, evidence is pointing toward other dietary factors, admittedly of a lower impact range, that could affect energy balance on a long-term basis and therefore represent an avenue worth considering under obesity treatment circumstances. Ca is a good example of this, as low consumption of this micronutrient has been shown to be inversely associated with body weight and adiposity in many studies(Reference Jacqmain, Doucet, Despres, Bouchard and Tremblay1–Reference Zemel, Shi, Greer, Dirienzo and Zemel3). More recently, it was observed in a large cohort of 15 655 individuals that overweight or obese men and women who had a high consumption of multivitamins, vitamin B6, vitamin B12 or Cr had gained less weight than those who had not consumed these supplements over 8–12 years(Reference Nachtigal, Patterson, Stratton, Adams, Shattuck and White4).
How vitamins and minerals could be implicated in the regulation of body energy stores is not known. For instance, one could propose that they influence the control of food intake through their roles in neurotransmitter synthesis and regulation in the central nervous system. Vitamins C and B6 are good examples of this, since they are respectively involved in the hydroxylation and decarboxylation of tryptophan for the synthesis of serotonin(Reference Groff and Gropper5) which is known to control food intake through the melanocortin pathways(Reference Heisler, Cowley and Kishi6). Low status of vitamin B6 and its consequent decrease in brain serotonin synthesis(Reference Anderson and Johnston7) could result in an increased appetite and presumably food intake and favour a positive energy imbalance. On the other side of the equation, the role of micronutrients as enzyme cofactors necessary for energy transformation within cells(Reference Groff and Gropper5) suggests that they could also influence the regulation of energy expenditure. In support to these hypothesis, Singh et al. have shown that vitamins C and E deficiency was a risk factor for higher percentage body fat and central obesity in Indian men(Reference Singh, Beegom, Rastogi, Gaoli and Shoumin8). Moreover, high percentages of vitamin C and B2 deficiency(Reference Harnroongroj, Jintaridhi, Vudhivai, Pongpaew, Tungtrongchitr, Phonrat, Changbumrung and Schelp9), lower bioavailability of vitamin D(Reference Wortsman, Matsuoka, Chen, Lu and Holick10), lower serum vitamins A, C and E(Reference Moor, Wartanowicz and Ziemlanski11) and folic acid concentrations(Reference Tungtrongchitr, Pongpaew, Tongboonchoo, Vudhivai, Changbumrung, Tungtrongchitr, Phonrat, Viroonudomphol, Pooudong and Schelp12) have been found in overweight and obese individuals compared with non-obese subjects. Interestingly, high serum folic acid level was also recently found to be a significant predictor of body-weight loss in morbidly obese patients(Reference Martinez, Ruiz and Candil13). Finally, an effect of vitamin C status(Reference Johnston14) and of Cr supplementation on lipid oxidation and fat-mass (FM) loss(Reference Bahadori, Wallner, Schneider, Wascher and Toplak15, Reference Crawford, Scheckenbach and Preuss16) have been observed in intervention studies.
Taken together, these results suggest that a certain level of availability of vitamins and minerals might influence energy balance regulation and, thus, that an adequate intake could be associated with lower body weight or even determine the success of weight-reducing programmes. In order to investigate this issue, we report data from two separate studies. The objective of study 1 was to determine whether regular consumers (CS) of vitamin and/or dietary supplements were characterised by lower body weight and adiposity values than non-consumers (NCS) of these supplements using data from the Québec Family Study(Reference Bouchard, Bray and Ryan17). The aim of study 2 was to assess in a different group of participants the effect of a weight-loss intervention, combined with either a multivitamin + mineral supplement (MMS) or with a placebo on body-weight and FM loss, energy expenditure and appetite.
Methods
Study 1
Adult men (n 267) and women (n 320) aged 20–65 years with complete data from the dietary questionnaire (explained in the next section) were selected among the subjects who participated in phase 2 (1991–8) of the Québec Family Study(Reference Bouchard, Bray and Ryan17). Families of French descent from the greater Québec City area were recruited to participate in this study which received approval from the Laval University Medical Ethics Committee.
Vitamins and dietary supplements
A questionnaire was used to gather dietary information. One of the questions of interest for this study was as follows: ‘Do you consume vitamin/dietary supplements?’ Since this was the only question asked to record the participants' supplement consumption habits, the dose and the type of vitamins and/or dietary supplements cannot be addressed in this study. The present study therefore pertains to differences between consumers (CS) and NCS of vitamins and/or dietary supplements.
Three-day dietary record and physical activity diary measurement
Dietary habits were determined by a 3 d dietary record which has been previously shown to provide reliable results(Reference Tremblay, Sévigny, Leblanc and Bouchard18). All subjects were requested to record their dietary intake during two week days and one weekend day. Nutritionists explained to each subject how to accurately complete the forms. All subjects were supplied with a balance, measuring cups and spoons to facilitate the measuring procedures. A nutritionist reviewed the records together with subjects during an interview. The Canadian Nutrient File(19) was used to assess macro- as well as micronutrient content of foods. Daily physical activities were evaluated with a 3 d physical activity diary, as previously described(Reference Bouchard, Tremblay, Leblanc, Lortie, Savard and Thériault20). Briefly, subjects recorded the number of 15 min periods during 24 h that they were engaged in activities classified on a 1–9 scale, 1 corresponding to activities of very low energy expenditure such as sleeping and 9 to activities of very high energy expenditure such as running. The 3 d mean of the number of periods in activities 7 to 9 (activity periods) was used as an indication of physical activity practice.
Eating behaviour measurements
A subgroup of participants completed the Three-Factor Eating Questionnaire(Reference Stunkard and Messick21) as validated for the French population(Reference Lluch22). Since this questionnaire was included in the Québec Family Study only at the end of the second phase, data from only 119 men and 174 women were available. The Three-Factor Eating Questionnaire evaluates ‘dietary restraint’, which measures the actual restrictive eating as well as the intention to restrain eating, ‘disinhibition’, which measures how easily external factors, that is, environmental events and emotional reactions, disinhibit the control of eating, and ‘susceptibility to hunger’, which measures the ability to cope with the sensation of hunger(Reference Drapeau, Provencher, Lemieux, Despres, Bouchard and Tremblay23). The Three-Factor Eating Questionnaire has been shown to have acceptable reliability and validity(Reference Stunkard and Messick21, Reference Laessle, Tuschl, Kotthaus and Pirke24).
Anthropometric measurements
Body weight was measured with a standard beam scale. Body density was determined by hydrodensitometry(Reference Behnke and Wilmore25). The closed circuit He-dilution method(Reference Meneely and Kaltreider26) was used to assess the residual lung volume. The Siri formula(Reference Siri27) was used to estimate the percentage of body fat from body density. FM was calculated from the percentage of body fat and total body weight while fat-free mass (FFM) was estimated as the difference between FM and body weight.
Measure of resting energy expenditure
Resting energy expenditure (REE) was measured early in the morning while participants were in a fasted state (12 h overnight fast). Subjects were asked to refrain from physical activity on the day before this measurement. Indirect calorimetry was performed over a 30 min period through an open-circuit system with a ventilated hood. Data from the last 10 min of this measurement period were used for the calculation of respiratory quotient and REE. Gas analyses were assayed with an IR CO2 analyser (Ametek CD-3A; Thermox Instruments Division, Pittsburgh, PA, USA) and an IR O2 analyser (Ametek S-3A; Thermox Instruments Division). Analysers were calibrated before each test. The Weir formula(Reference Weir28) was used to determine the energy equivalent of O2 volume.
Statistical analysis
Statistical analyses were performed using SAS software (version 9.1.2; SAS Institute, Cary, NC, USA). Since most of the participants were from three generations of the same family(Reference Loos, Rankinen, Leon, Skinner, Wilmore, Rao and Bouchard17), analyses were performed to verify whether there were generation × supplement consumed status interaction effects and none were found. All subjects were thus pooled together for further analyses although sexes were analysed separately. A one-way analysis of covariance (CS and NCS) taking into account potential covariates (age, height, daily energy intake, percentage dietary fat and activity periods) which have been shown to affect energy balance and/or body-weight control was performed for body weight, FM, FFM, fat percentage and BMI. A one-way ANOVA (CS and NCS) was performed on all other dependent variables. Differences were considered as significant at P < 0·05 and data are presented as mean values and standard deviations.
Study 2
Screening
Study 2 was approved by the Laval University Ethics Committee and participants were recruited through diverse media sources such as local newspapers, radio stations and email communications. A sample of sixty-three individuals coming from the Québec City metropolitan area met the study inclusion and exclusion criteria which were as follows: aged 25–45 years, BMI 30–40 kg/m2, good health, waist circumference ≥ 90 cm, stable body weight during 6 months before the study, premenopausal, less than 1 h/week of continuous physical activity, normal blood pressure values, normal blood glucose levels, normal thyroid hormone levels, non-smoker, no use of medication that could potentially interfere with the study's objectives and no use of vitamin and mineral supplements 6 months before the beginning of the study. Breast-feeding or pregnant women, verified by a urine-based pregnancy test, were excluded from the study.
Participants came to the laboratory for a screening evaluation in order to confirm their eligibility and for explanation of the study consent form. During this visit, blood samples were collected after a 12 h overnight fast to measure a biochemical profile. Body weight, height and blood pressure were also measured during this visit. Another session was fixed for a medical examination and for explanation of the 3 d dietary record by a nutritionist (as described for study 1). Fasting blood samples were taken and a 75 g oral glucose tolerance test over 3 h was also performed.
Protocol
Following the screening procedures, forty-five obese otherwise healthy men and women were initially enrolled and randomised in a double-blind manner to receive the MMS or the placebo coupled to a 15-week energy restriction. MMS capsules were the Premium Multi-Cap with Ester-C from Quest, Canada (composition of the supplement can be found at www.questvitamins.com), and placebo was lactose monohydrate in gelatine capsules. Dosage was one tablet taken before breakfast. Among the initial participants, five dropped out (three placebo and two MMS); four because of personal reasons and one became pregnant.
The sequence of measurements happened as follow and is described in more detail in the following paragraphs. Measurements were spread over two consecutive days. On day 1, REE and respiratory quotient were measured in the fasting state; appetite ratings were assessed before and after a standardised breakfast test and participants were asked to fill in the Three-Factor Eating Questionnaire. On day 2, 24 h energy expenditure was measured. Day 1 and 2 measurements were realised at baseline (week 0) and were repeated at the end of the 15-week intervention (week 15).
Weight-loss intervention
The subjects followed a non-macronutrient-specific energy-restricted diet for 15 weeks. Prescribed daily energy intake was determined by subtracting 2930 kJ (700 kcal) from the daily energy expenditure calculated by multiplying the REE by an activity factor of 1·4. A 3 d dietary record was also used to estimate energy and macronutrient intake of each subject. Following the baseline visit (week 0), a nutritionist explained to the participants how to reach the targeted daily 2930 kJ (700 kcal) non-macronutrient-specific energy deficit using the food exchange system for diabetics from the Association Diabète Québec(29). During this visit, treatment tablets (MMS or placebo) were given to participants who started the energy restriction and the consumption of the treatment tablets the next morning. The intervention lasted 15 weeks, during which participants met the nutritionist every 2 weeks. In order to assess compliance to the weight-reducing programme, a count of remaining treatment tablets, a 24 h dietary recall assessed with the nutritionist and visual analogue scales evaluating satisfaction with the diet, hunger, and rigor in following the treatment were filled in by the participants during the interview. On the week between these meetings, participants received a phone call from the nutritionist to answer any questions and ensure that participants were following treatment. Each staff member and participants were blinded to treatment assignment for the duration of the study.
Anthropometry
Height, body weight and waist circumference were determined following the procedures recommended at the Airlie Conference(Reference Harrison, Buskirk, Carter, Johnston, Lohman, Pollock, Roche, Wilnor, Lohman, Roche and Martorell30), and BMI was then calculated. FM, FFM and percentage of fat were determined as described in the Study 1 methods section.
Resting and 24 h energy expenditure
REE was measured by indirect calorimetry after a 12 h overnight fast and a 24 h period without alcohol consumption and intense physical activity. Participants had to arrive in the morning at the laboratory and to keep an activity level as low as possible in between their wake-up time and their arrival to the laboratory. A questionnaire was filled in by the subjects upon their arrival to ensure they were meeting these criteria. After resting for 15 min, expired gas collection was performed through a mouthpiece for 15 min while the nose was clipped during the whole measurement. O2 and CO2 concentrations were determined by non-dispersive IR analysis (Uras 10 E; Hartmann & Braun, Frankfurt, Germany) whereas pulmonary ventilation determination was assessed with an S430A measurement system (KL Engineering, Ventura, CA, USA). The Weir formula(Reference Weir28) was used to determine the energy equivalent of O2 volume. The determination of substrate oxidation was assessed through the calculations previously described by Frayn(Reference Frayn31) while assuming that protein oxidation contributes 10 % of total energy expenditure measured under these conditions.
The next day, 24 h total energy expenditure was measured with a whole-body indirect calorimeter, which has been shown to provide highly reproducible data in our laboratory(Reference White, Bouchard, Buemann, Almeras, Despres, Bouchard and Tremblay32). Subjects came back to the laboratory early in the morning and had to have respected the same criteria for fasting, alcohol consumption and physical activity as for the REE measurement. During this stay, subjects were maintained in energy balance by using the REE performed at the initial visit and by extrapolating this value over a 24 h period and then multiplying this value by an activity factor of 1·32. The same energy intake was maintained for the two measurements of 24 h energy expenditure (weeks 0 and 15). Moreover, the nutrient composition of the diet (daily food quotient 0·85), the sedentary lifestyle pattern (watching television, computer use, reading, etc) and the meal pattern, as well as the period of sleep, were also standardised in each session. It was not permitted to eat or drink any other foods than those provided and therefore no foods or beverages containing caffeine were allowed during the metabolic chamber stay. Sleeping metabolic rate was also measured in the chamber and was determined as the mean of the two consecutive hours of the night when the subject had the lowest O2 consumption. This value was then extrapolated over 24 h. Women were tested between days 0 and 8 of their menstrual cycle, because energy metabolism-related factors have been shown to be affected by sex steroid levels(Reference Day, Gozansky, Van Pelt, Schwartz and Kohrt33).
Standardised breakfast and appetite ratings
A standardised breakfast which has previously been described(Reference Drapeau, Blundell, Therrien, Lawton, Richard and Tremblay34) was served to participants after a 12 h overnight fast. The energy content of the breakfast was 3067 kJ (733 kcal) and 2506 kJ (599 kcal) for men and women, respectively, and had a food quotient of 0·85. All participants were instructed to eat within 30 min or less. Before, immediately after and every 10 min for a period of 1 h after the breakfast, subjects were asked to fill in visual analogue scales adapted from Blundell & Hill(Reference Blundell and Hill35). The answer to the following questions were used to determine their ‘desire to eat’, ‘hunger’, ‘fullness’ and ‘prospective food consumption’, respectively: (1) ‘How strong is your desire to eat?’ (very weak – very strong); (2) ‘How hungry do you feel?’ (not hungry at all – as hungry as I have ever felt); (3) ‘How full do you feel?’ (not full at all – very full); (4) ‘How much food do you think you could eat?’ (nothing at all – a large amount), respectively. Appetite ratings measured before breakfast were considered as the fasting measurement, and appetite ratings in response to the breakfast were evaluated by calculating the post-meal area under the curve (AUC) with the trapezoid method as previously described(Reference Doucet, St Pierre, Almeras and Tremblay36). AUC was calculated considering appetite rating responses at 0, 10, 20, 30, 40, 50 and 60 min after the breakfast. Breakfast was consumed and visual analogue scale measurements performed in the same room kept quiet and away from different visual, hearing and olfactory stimuli that might have influenced the measurements. Under these conditions, visual analogue scale measurements in our laboratory have been shown to be highly reliable both before and in response to a meal(Reference Doucet, St Pierre, Almeras and Tremblay36, Reference Arvaniti, Richard and Tremblay37). During this visit, participants were also asked to complete the Three-Factor Eating Questionnaire as described in study 1.
Statistical analysis
Statistical analyses were performed using SAS software (version 9.1.2; SAS Institute, Cary, NC, USA). A two-way ANOVA with repeated measurements on one factor (time) was used to assess the effects of treatment (placebo and MMS) and time, and their interaction on all dependent variables within each sex. In order to adjust the appetite variables for change in body weight, a two-way ANOVA was repeated using residual scores for appetite ratings and the Three-Factor Eating Questionnaire scores derived from linear regression analysis considering body weight as the independent variable. Three-Factor Eating Questionnaire scores that included unanswered questions were corrected for the specific eating behaviour total score. Differences were considered significant at P < 0·05 and data are presented as mean values and standard deviations.
Results
Study 1
Results from study 1 showed that men CS had significantly lower body weight, BMI, FM, percentage body fat and FFM than men NCS after adjustments were made for age, height, daily energy intake, percentage dietary fat and activity periods. Men CS also had a trend toward a greater REE expressed per kg body weight compared with men NCS (P = 0·06). The same trend was found for anthropometric measurements between CS and NCS in women but the differences were less pronounced and statistically non-significant. Variables related to food intake as well as daily physical activity level were all comparable between both groups of men (Table 1). However, the CS group of women had a lower fat intake and tended to be older and to have a lower overall energy intake, greater carbohydrate consumption and to be more physically active (Table 1). Women CS also had a significantly lower disinhibition and hunger scores than NCS women (P < 0·05), while in CS men the slightly lower scores observed for these variables were not statistically different from those in NCS men.
Table 1 Characteristics of regular consumers and non-consumers of supplements in study 1 (Mean values and standard deviations)

* Adjusted for age, height, daily energy intake, percentage dietary fat and activity periods.
† Mean daily number of 15 min periods of activity level 7–9 assessed with a physical activity diary.
Study 2
Baseline daily vitamin and mineral consumption, assessed with the 3 d dietary record, was similar between placebo and MMS groups in both men and women (results not shown). Anthropometric variables, REE and 24 h energy expenditure mean values are presented in Tables 2 and 3 for men and women, respectively. Means for these variables were similar in MMS and placebo groups before intervention. Body weight, BMI, waist circumference, FM and percentage of fat were significantly decreased at week 15 in men and women in both MMS and placebo groups. In women only, a significant decrease in FFM was also observed, with no difference between MMS and placebo groups.
Table 2 Characteristics of multivitamin and mineral supplementation (MMS) and placebo groups of men before (week 0) and after (week 15) the weight-loss intervention in study 2 (Mean values and standard deviations)

Table 3 Characteristics of multivitamin and mineral supplementation (MMS) and placebo groups of women before (week 0) and after (week 15) the weight-loss intervention in study 2 (Mean values and standard deviations)

Appetite ratings measured in the fasting state before breakfast and postprandial appetite rating AUC measured after the breakfast are presented in Table 4. In men, no significant between-group difference was observed for these variables. In women, baseline value for fasting desire to eat was significantly lower in the placebo than in the MMS group. However, a significant time × treatment interaction effect was observed for this variable, revealing a significant decrease in the MMS group and a non-significant increase in the placebo group. Significant time × treatment interaction effects were observed in women for desire to eat, hunger and prospective food consumption AUC, revealing a significant decrease in these variables in the MMS group only, and an AUC value for these three variables that was significantly lower in the MMS group compared with the placebo group at week 15 (Table 4). These differences were observed after statistical adjustments for body-weight change. In both men and women, a significant increase in dietary restraint and a significant decrease in disinhibition and susceptibility to hunger were observed following the programme with no difference between treatment groups (Table 5).
Table 4 Appetite ratings measured before and after a standardised breakfast meal before (week 0) and after (week 15) the weight-loss intervention in study 2 (Mean values and standard deviations)

MMS, multivitamin and mineral supplementation; PFC, prospective food consumption; AUC, area under the curve.
* Mean value was significantly different from that in week 0 (P < 0·05).
† Mean value was significantly different from that in the placebo group (P < 0·05).
‡ Adjusted for body-weight change.
Table 5 Three-Factor Eating Questionnaire mean scores before (week 0) and after (week 15) the weight-loss intervention in men and women in study 2 (Mean values and standard deviations)
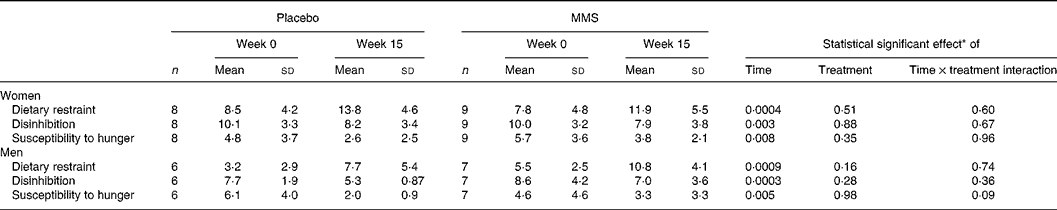
MMS, multivitamin and mineral supplementation.
* Adjusted for body-weight change.
Discussion
Results obtained in study 1 showed that men regularly consuming micronutrients and dietary supplements had a lower body weight and FM than men who did not. This agrees with the recently reported lower 8–12 years body-weight gain in overweight or obese CS of multivitamins, vitamins B6, vitamin B12 and Cr compared with NCS(Reference Nachtigal, Patterson, Stratton, Adams, Shattuck and White4). From a body-weight-regulation standpoint, these observations suggest that a sufficient intake of micronutrients, which have fundamental roles in energy metabolism(Reference Groff and Gropper5), might be needed to achieve energy balance stability. Sufficient intake of micronutrients could thus be important in the context of obesity treatment, where micronutrient intake may be decreased along with macronutrients during reduced-energy diets(Reference Grzybek, Klosiewicz-Latoszek and Targosz38, Reference Vasilaras, Astrup and Raben39). This hypothesis was investigated in study 2, and no difference in the changes in body weight, FM, energy expenditure and lipid oxidation was observed between obese individuals supplemented with a MMS or with a placebo in the context of a weight-reducing programme. However, the supplementation in micronutrients had a beneficial effect on appetite-rating variables in women as shown by the significantly lower AUC for desire to eat, hunger and prospective food consumption in MMS-supplemented women compared with baseline and compared with the placebo group. A significant decrease in fasting desire to eat was also observed in MMS-supplemented women, although this result could be considered less powerful since the baseline value for this variable was significantly higher in this group compared with placebo. These differences remained significant when change in body weight was taken into account, suggesting that an adequate intake in vitamins and minerals might influence satiety.
Appetite is increased in response to body-weight loss(Reference Doucet, St Pierre, Almeras and Tremblay36, Reference Doucet, Imbeault, St Pierre, Almeras, Mauriege, Richard and Tremblay40) due to variations in hormone levels implicated in the regulation of energy balance such as insulin, leptin and cortisol(Reference Doucet, St Pierre, Almeras and Tremblay36, Reference Blundell, Goodson and Halford41, Reference Keim, Stern and Havel42). Since some vitamins and minerals are involved in the synthesis of these and other peptides and neurotransmitters that control food intake, a decreased micronutrient availability could affect peptide hormone levels and thus interfere with the signalling pathways that control food intake, as was observed in Zn-deprived rats(Reference Reeves43). From a more peripheral perspective, it is well known that variations in energy substrate availability can also impact on hunger and energy intake. Studies in animals(Reference Friedman, Tordoff and Ramirez44, Reference Friedman and Tordoff45) and in human subjects(Reference French and Castiglione46, Reference Kahler, Zimmermann and Langhans47) have shown that inhibition of glucose utilisation and fatty acid oxidation resulted in an increased appetite and energy intake, as does a decrease in blood glucose concentrations(Reference Anderson, Catherine, Woodend and Wolever48, Reference Campfield, Smith, Rosenbaum and Hirsch49). Since micronutrients are essential to energy substrate oxidation and must be supplied by the diet(Reference Groff and Gropper5), one can speculate that their intake is also regulated. As such, a lower consumption could result in signalling to feeding centres to increase energy intake in order to satisfy the body's micronutrient needs(Reference Ames50). This hypothesis is supported by the recently described evidence for the existence of an appetite for Ca, Na, Mg and Se(Reference McCaughey and Tordoff51–Reference Zuberbuehler, Messikommer and Wenk54), and is concordant with the lower susceptibility to hunger and disinhibition observed in women CS compared with NCS in study 1. This could also explain the significant decrease in appetite ratings at the end of the programme in MMS women but not in the placebo group in study 2. Interestingly, some studies have shown that postprandial prospective food consumption, which was significantly reduced in MMS women, is the most consistent predictor of energy intake(Reference Doucet, St Pierre, Almeras and Tremblay36, Reference Barkeling, Rossner and Sjoberg55, Reference Flint, Raben, Blundell and Astrup56). Thus, these results strengthen the plausible role for vitamins and minerals in the control of appetite and ultimately energy intake.
In this regard, the absence of any significant effect of the MMS supplement on body weight, energy expenditure and lipid oxidation in study 2 cannot be ignored. It could perhaps be explained by the fact that participants were not selected on the basis of a low micronutrient intake status. Indeed, vitamin and mineral consumption by the participants was in a normal range before intervention, and the appetite for minerals as described above has been reported mainly in circumstances of deprivation in these minerals(Reference Ames50–Reference Zuberbuehler, Messikommer and Wenk54). Moreover, several cross-sectional studies have reported a link between a high body weight and intake of a micronutrient when the latter is inadequate or a deficiency is evident(Reference Jacqmain, Doucet, Despres, Bouchard and Tremblay1–Reference Zemel, Shi, Greer, Dirienzo and Zemel3, Reference Singh, Beegom, Rastogi, Gaoli and Shoumin8, Reference Harnroongroj, Jintaridhi, Vudhivai, Pongpaew, Tungtrongchitr, Phonrat, Changbumrung and Schelp9, Reference Moor, Wartanowicz and Ziemlanski11, Reference Tungtrongchitr, Pongpaew, Tongboonchoo, Vudhivai, Changbumrung, Tungtrongchitr, Phonrat, Viroonudomphol, Pooudong and Schelp12, Reference Carmody, Brunner and St Jeor57). In accordance with this, Volpe et al. found no effect from supplementation of Cr, an essential trace element involved in energy substrate metabolism(Reference Groff and Gropper5), on body composition or REE in obese women whose vitamin and mineral consumption was in a normal range before intervention(Reference Volpe, Huang, Larpadisorn and Lesser58). Overall, this suggests that an appetite-modulating effect of vitamin and mineral supplements could be more significant in individuals characterised by inadequate intake in micronutrients.
It is also possible that the energy restriction in study 2 masked the effect that the vitamin and mineral supplementation could have had on body weight. Indeed, all participants were instructed to reduce their energy intake in proportion to their baseline consumption. This could explain, at least partly, why a decrease in body weight above that attributed to the weight-reducing programme was not seen in supplemented individuals, but that a lower body weight was observed in free-living long-term vitamin and/or dietary supplement CS (study 1 and Nachtigal et al.(Reference Nachtigal, Patterson, Stratton, Adams, Shattuck and White4)) in whom no a priori dietary restriction interfered with the proposed micronutrient-induced appetite-reduction effect. Further research in this issue should focus on large groups of individuals characterised by inadequate vitamin and mineral intake.
It is intriguing that in study 1, the possible influence of vitamin and dietary supplements on body weight was observed only in men (and in women to a lesser extent) while in study 2, significant appetite effects were observed only in women, as for the suggested behavioural effect in study 1. In this last study, this could be due to the fact that other factors affecting energy balance and body weight (see Table 1) were not as closely matched in women as they were in men. Moreover, CS women were older and had a significant lower dietary fat intake than NCS women. With respect to study 2 findings, because appetite control is under the influence of central and peripheral as well as behavioural signals and social factors(Reference Doucet, St Pierre, Almeras and Tremblay36, Reference Blundell, Goodson and Halford41, Reference Keim, Stern and Havel42), dietary restraint typically more present in women than in men(Reference Carmody, Brunner and St Jeor57, Reference Provencher, Drapeau, Tremblay, Despres and Lemieux59) could have influenced appetite ratings. However, in both sexes, changes that occurred during the programme followed the same trend upward for restraint and downward for disinhibition and susceptibility to hunger. Moreover, these changes as well as these variables' scores before and after the programme were similar between treatment groups, suggesting that they probably do not account for the differences in appetite ratings in women and in men. However, it has to be recognised that although we took into consideration the most obvious confounding variables (physical activity, macronutrient composition of the diet), and verified that the education level and total annual familial income was not affecting the results (results not shown), other lifestyle factors we did not measure might have exerted an influence and have partly accounted for the differences between CS and NCS in study 1. This is especially true for the type and composition of the vitamin and/or dietary supplement in study 1 for which we had no detailed information. Although study 2 was based on a much stronger experimental design to control for potential confounders, the number of participants was low. On one hand this indicates that effects were substantial since statistical significance was achieved despite small group sizes but on the other hand, the present results and conclusions need to be treated with caution unless replicated in larger groups of participants.
Conclusion
In conclusion, cross-sectional observations have shown that men regularly consuming vitamin and dietary supplements had a lower body weight and FM and tended to have a higher relative RMR compared with men who did not report consuming such supplements, independently from physical activity level and dietary factors. This trend was also seen in women together with a reduced hunger level. A double-blind randomised controlled study showed no direct evidence that multivitamin and mineral supplementation during a weight-reducing programme in a relatively small number of obese individuals could enhance body-weight and FM loss. However, the multivitamin and mineral supplement was found to significantly decrease appetite ratings in women after weight loss, compared with a placebo group which also lost weight. This therefore suggests that vitamin and mineral supplements could attenuate, in women, the increase in appetite that often accompanies body-weight loss. This issue should be further investigated as should the underlying mechanisms of vitamins and minerals in appetite control.
Acknowledgements
The study was supported by the Canadian Institutes of Health Research. G. C. M. is the recipient of a training fellowship from the Heart and Stroke Foundation of Canada, the Canadian Diabetes Association and the Canadian Institutes of Health Research. A. T. is partly funded by the Canada Research Chair in Physical Activity, Nutrition and Energy Balance. C. B. is partially funded by the George A. Bray Chair in Nutrition. E. D. is a recipient of a CIHR/Merck-Frosst New Investigator Award and CFI/OIT New Opportunities Award. The authors acknowledge Claude Leblanc for his assistance with statistical analyses.







