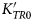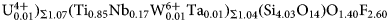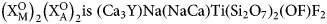Refine listing
Actions for selected content:
Contents
The 51st Hallimond Lecture
Time's arrow, time's cycle: Granulite metamorphism and geodynamics
-
- Published online by Cambridge University Press:
- 12 April 2019, pp. 323-338
-
- Article
-
- You have access
- HTML
- Export citation
Article
Constraints on the Equations of State of stiff anisotropic minerals: rutile, and the implications for rutile elastic barometry
-
- Published online by Cambridge University Press:
- 22 April 2019, pp. 339-347
-
- Article
- Export citation
Magnesioleydetite and straβmannite, two new uranyl sulfate minerals with sheet structures from Red Canyon, Utah
-
- Published online by Cambridge University Press:
- 28 May 2018, pp. 349-360
-
- Article
- Export citation
Gem amphiboles from Mogok, Myanmar: crystal-structure refinement, infrared spectroscopy and short-range order–disorder in gem pargasite and fluoro-pargasite
-
- Published online by Cambridge University Press:
- 14 September 2018, pp. 361-371
-
- Article
- Export citation
Rinkite-(Y), Na2Ca4YTi(Si2O7)2OF3, a seidozerite-supergroup TS-block mineral from the Darai-Pioz alkaline massif, Tien-Shan mountains, Tajikistan: Description and crystal structure
-
- Published online by Cambridge University Press:
- 29 June 2018, pp. 373-380
-
- Article
- Export citation
Subaerial sulfate mineral formation related to acid aerosols at the Zhenzhu Spring, Tengchong, China
-
- Published online by Cambridge University Press:
- 14 January 2019, pp. 381-392
-
- Article
- Export citation
Pampaloite, AuSbTe, a new mineral from Pampalo gold mine, Finland
-
- Published online by Cambridge University Press:
- 04 July 2018, pp. 393-400
-
- Article
- Export citation
Mineralogy of the baotite-bearing Gundrapalli lamproite, Nalgonda district, Telangana, India
-
- Published online by Cambridge University Press:
- 31 January 2019, pp. 401-411
-
- Article
- Export citation
Ferri-fluoro-katophorite from Bear Lake diggings, Bancroft area, Ontario, Canada: a new species of amphibole, ideally Na(NaCa)(Mg4Fe3+)(Si7Al)O22F2
-
- Published online by Cambridge University Press:
- 02 July 2018, pp. 413-417
-
- Article
- Export citation
Discovery of Se-rich canfieldite, Ag8Sn(S,Se)6, from the Shuangjianzishan Ag–Pb–Zn deposit, NE China: A multimethodic chemical and structural study
-
- Published online by Cambridge University Press:
- 08 October 2018, pp. 419-426
-
- Article
- Export citation
Middlebackite, a new Cu oxalate mineral from Iron Monarch, South Australia: Description and crystal structure
-
- Published online by Cambridge University Press:
- 02 July 2018, pp. 427-433
-
- Article
- Export citation
Substitution mechanisms in In-, Au-, and Cu-bearing sphalerites studied by X-ray absorption spectroscopy of synthetic compounds and natural minerals
-
- Published online by Cambridge University Press:
- 04 March 2019, pp. 435-451
-
- Article
- Export citation
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. IX. Arsenatrotitanite, NaTiO(AsO4)
-
- Published online by Cambridge University Press:
- 04 July 2018, pp. 453-458
-
- Article
- Export citation
The crystal structure of Ni-rich gordaite–thérèsemagnanite from Cap Garonne, France
-
- Published online by Cambridge University Press:
- 14 March 2019, pp. 459-463
-
- Article
- Export citation
Potassic-magnesio-arfvedsonite, KNa2(MgFe2+Fe3+)5Si8O22(OH)2: mineral description and crystal chemistry
-
- Published online by Cambridge University Press:
- 02 July 2018, pp. 465-472
-
- Article
- Export citation
Discreditation of the pyroxenoid mineral name ‘marshallsussmanite’ with a reinstatement of the name schizolite, NaCaMnSi3O8(OH)
-
- Published online by Cambridge University Press:
- 22 April 2019, pp. 473-478
-
- Article
- Export citation
NEWSLETTER 49
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC)
New minerals and nomenclature modifications approved in 2019
-
- Published online by Cambridge University Press:
- 24 May 2019, pp. 479-483
-
- Article
-
- You have access
- HTML
- Export citation
Front Cover (OFC, IFC) and matter
MGM volume 83 issue 3 Cover and Front matter
-
- Published online by Cambridge University Press:
- 09 July 2019, pp. f1-f2
-
- Article
-
- You have access
- Export citation
Back Cover (IBC, OBC) and matter
MGM volume 83 issue 3 Cover and Back matter
-
- Published online by Cambridge University Press:
- 09 July 2019, pp. b1-b2
-
- Article
-
- You have access
- Export citation