Refine listing
Actions for selected content:
Volume 78 - Issue 3 - June 2014
Contents
Research Article
Kihlmanite-(Ce), Ce2TiO2[SiO4](HCO3)2(H2O), a new rare-earth mineral from the pegmatites of the Khibiny alkaline massif, Kola Peninsula, Russia
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 483-496
-
- Article
- Export citation
Nestolaite, CaSeO3·H2O, a new mineral from the Little Eva mine, Grand County, Utah, USA
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 497-505
-
- Article
- Export citation
Alpine oxidation of lithium micas in Permian S-type granites (Gemeric unit, Western Carpathians, Slovakia)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 507-533
-
- Article
- Export citation
Hydroniumjarosite, (H3O)+Fe3(SO4)2(OH)6, from Cerros Pintados, Chile: Single-crystal X-ray diffraction and vibrational spectroscopic study
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 535-547
-
- Article
- Export citation
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC) Newsletter 20
New minerals and nomenclature modifications approved in 2014
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 549-558
-
- Article
- Export citation
Research Article
Tangdanite, a new mineral species from the Yunnan Province, China and the discreditation of ‘clinotyrolite’
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 559-569
-
- Article
- Export citation
Vapnikite Ca3UO6 – a new double-perovskite mineral from pyrometamorphic larnite rocks of the Jabel Harmun, Palestinian Autonomy, Israel
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 571-581
-
- Article
- Export citation
Tondiite, Cu3Mg(OH)6Cl2, the Mg-analogue of herbertsmithite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 583-590
-
- Article
- Export citation
Franconite, NaNb2O5(OH)·3H2O: structure determination and the role of H bonding, with comments on the crystal chemistry of franconite-related minerals
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 591-607
-
- Article
- Export citation
Structural, spectroscopic and computational studies on the monoclinic polymorph (form I) of potassium hydrogen disilicate (KHSi2O5)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 609-622
-
- Article
- Export citation
Growth of continental crust: a balance between preservation and recycling
- Part of:
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 623-637
-
- Article
- Export citation
Belakovskiite, Na7(UO2)(SO4)4(SO3OH)(H2O)3, a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 639-649
-
- Article
- Export citation
Lead-antimony sulfosalts from Tuscany (Italy). XV. (Tl-Ag)-bearing rouxelite from Monte Arsiccio mine: occurrence and crystal chemistry
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 651-661
-
- Article
- Export citation
Ekplexite (Nb,Mo)S2·(Mg1−x Alx )(OH)2+x , kaskasite (Mo,Nb)S2·(Mg1−x Alx )(OH)2+x and manganokaskasite (Mo,Nb)S2·(Mn1−x Alx )(OH)2+x , three new valleriite-group mineral species from the Khibiny alkaline complex, Kola peninsula, Russia
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 663-679
-
- Article
- Export citation
First accurate location of two proton sites in tourmaline: A single-crystal neutron diffraction study of oxy-dravite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 681-692
-
- Article
- Export citation
Chemical variability in wakabayashilite: a real feature or an analytical artifact?
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 693-702
-
- Article
- Export citation
Trace element composition of quartz from different types of pegmatites: A case study from the Moldanubian Zone of the Bohemian Massif (Czech Republic)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 703-722
-
- Article
- Export citation
Single-crystal neutron diffraction and Raman spectroscopic study of hydroxylherderite, CaBePO4(OH,F)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 723-737
-
- Article
- Export citation
Kitagohaite, Pt7Cu, a new mineral from the Lubero region, North Kivu, Democratic Republic of the Congo
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 739-745
-
- Article
- Export citation
Torrecillasite, Na(As,Sb)4 3+O6Cl, a new mineral from the Torrecillas mine, Iquique Province, Chile: description and crystal structure
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 747-755
-
- Article
- Export citation
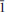 ,
, 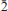 ), 6.912(12)(0
), 6.912(12)(0 ), 3.563(15)(0
), 3.563(15)(0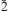 02)(74), 2.9783 (
02)(74), 2.9783 (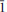 21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to
21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to 
 1 1), 4.333(71) (6 0 2), 5.263(54) (
1 1), 4.333(71) (6 0 2), 5.263(54) (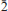 0 2), 3.949(47) (8 0 2) and 2.976(46) (
0 2), 3.949(47) (8 0 2) and 2.976(46) ( 1 1). The unit-cell parameters are
1 1). The unit-cell parameters are 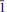
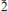 0), 2.764(100)(112 3), 2.266(54)(02
0), 2.764(100)(112 3), 2.266(54)(02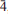 0). Several tondiite crystals have been examined by single-crystal X-ray diffraction and by electron microprobe analysis. The observed Mg content ranges between 0.6 and 0.7 atoms per formula unit. The structural role of Mg is discussed.
0). Several tondiite crystals have been examined by single-crystal X-ray diffraction and by electron microprobe analysis. The observed Mg content ranges between 0.6 and 0.7 atoms per formula unit. The structural role of Mg is discussed.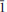 , with
, with  3), 3.057(59)(0
3), 3.057(59)(0
