- BDNF
brain-derived neurotrophic factor
- CREB
cAMP response element-binding protein
- ERK
extracellular signal-regulated protein kinase
- iNOS
inducible NO synthase
- JNK
c-Jun N-terminal kinase
- MAPK
mitogen-activated protein kinase
- PI3K
phosphatidylinositol 3-kinase
- PKB
protein kinase B
Representing one of the most important lifestyle factors, diet can strongly influence the incidence and onset of CVD and neurodegenerative disorders. Various phytochemical constituents of foods and beverages, in particular a class of compounds termed flavonoids, have been avidly investigated in recent years. They have been proposed to exert a multiplicity of neuroprotective actions within the brain, including a potential to protect neurons against injury induced by neurotoxins(Reference Spencer1), an ability to suppress neuroinflammation(Reference Vafeiadou, Vauzour and Spencer2) and the potential to promote memory, learning and cognitive function(Reference Spencer3, Reference Spencer, Vauzour and Rendeiro4). This multiplicity of effects appears to be underpinned by three processes. First, the flavonoids interact with important neuronal signalling cascades in the brain leading to an inhibition of apoptosis triggered by neurotoxic species and to a promotion of neuronal survival and differentiation(Reference Spencer1). These effects include selective actions on a number of protein kinase and lipid kinase signalling cascades, most notably the phosphatidylinositol 3-kinase (PI3K)/Akt and mitogen-activated protein kinase (MAPK) pathways that regulate pro-survival transcription factors and gene expression. Second, the flavonoids induce peripheral and cerebral vascular blood flow in a manner that may lead to the induction of angiogenesis and of new nerve cell growth in the hippocampus(Reference Vafeiadou, Vauzour and Spencer2). Third, the flavonoids may react directly with and scavenge neurotoxic species and pro-inflammatory agents produced in the brain as a result of both normal and abnormal brain ageing(Reference Spencer3). Thus, the consumption of flavonoid-rich fruits, such as berries, throughout life holds a potential to limit the neurodegeneration associated with a variety of neurological disorders and to prevent or reverse normal or abnormal deteriorations in cognitive performance. The present review will highlight the neuroprotective mechanisms of flavonoids through their ability to interact with neuronal signalling pathways, their potential to inhibit neuroinflammation and their impact on the vascular system. It will also attempt to address whether at present there is enough data to support a causal relationship between the consumption of flavonoids and behavioural outcomes such as memory and learning in human subjects. Finally, in light of this current information, potential future areas of research will be highlighted that may help to fully address the impact of flavonoid-rich diets on human cognitive performance.
Flavonoid: sources and structure
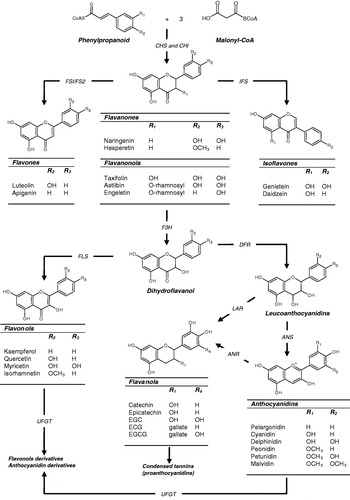
Flavonoids are synthesised in plants from the reaction of a chalcone precursor with three molecules of malonyl-CoA. Under the action of the enzymes chalcone synthase and chalcone flavanone isomerase the chalcone precursor is isomerised into a flavanone(Reference Dixon and Steele5, Reference Winkel-Shirley6). Although they share a similar 2,3-dihydro-2-phenylchromen-4-one skeleton structure, the hydroxylation of the C-3 position of ring C allows differentiation between flavanonols from flavanones (Fig. 1). From these central intermediates the pathway diverges into several side branches, each resulting in a different class of flavonoids. Flavonoids are found ubiquitously in plants and as such are major constituents of a variety of fruit and vegetables, beverages such as tea and wine and seeds such as cocoa beans and grape seeds. All flavonoids share a common structure consisting of two aromatic rings (A and B), which are bound together by three C atoms, forming an oxygenated heterocycle (ring C; Fig. 1). Based on variations in the saturation of the basic flavan ring system, their alkylation and/or glycosylation and the hydroxylation pattern of the molecules flavonoids may be divided into seven subclasses: flavonols; flavones; flavanones; flavanonols; flavanols; anthocyanidins; isoflavones (for review, see Manach et al.(Reference Manach, Scalbert and Morand7)). In addition, flavanols, which are sometimes referred to as flavan-3-ols, exist both as monomers and oligomers also known as condensed tannins or proanthocyanidins. These oligomeric forms differ on the basis of their constitutive units (e.g. catechin and epicatechin), their sequence and the positions of inter-flavanic linkages (C-4–C-6 or C-4–C-8 in the B-type series, with additional C2–O–C7 or C2–O–C5 bonds in A-type structures)(Reference Cheynier8).

Fig. 1. The structures of the main classes of flavonoids and their biosynthesis. The major differences between the individual groups reside in the hydroxylation pattern of the ring structure, the extent of saturation of ring C and the substitution in the C-3 position. All flavonoids are derived from chalcone precursors that are derived from phenylpropanoid and three molecules of malonyl-CoA and biosynthesised by chalcone synthase (CHS). Various enzymes act to bring about the formation of the various flavonoid classes: chalcone isomerase (CHI), flavone synthase (FSI/FS2), isoflavone synthase (IFS), flavanone 3-hydroxylase (F3H), dihydroflavonol reductase (DFR), anthocyanidin synthase (ANS), leucoanthocyanidin reductase (LAR), anthocyanidin reductase (ANR), UDP glucose-flavonoid 3-O-glucosyl transferase (UFGT), flavonol synthase (FLS). EGC, epigallocatechin; ECG, epicatechin gallate; EGCG, epigallocatechin gallate.
The flavanols are found predominantly in green and black teas, red wine, apples and cocoa. Variations in their structure reside in the hydroxylation pattern of ring B and the presence of a gallic acid moiety in C-3 position (Fig. 1). The lack of a double bond at the 2–3 position and the presence of a 3-OH group on ring C creates two centres of asymmetry. Typical dietary flavanols include catechin, epicatechin, epigallocatechin, epicatechin gallate, epigallocatechin gallate and proanthocyanidins, which may be found at high concentration in cocoa, tea, red wine and fruits such as apples, grapes and many berry fruits. The sources of anthocyanins such as pelargonidin, cyanidin and malvidin include red wine and berry fruits such as blueberries (Vaccinium corymbosum), blackberries (Rubus fruticosus), cherries (Prunus avium) and strawberries (Fragaria×ananassa). These compounds exist as glycosides in plants, are water soluble and appear red or blue according to the pH of their environment. Individual anthocyanins arise from the variation in number and arrangement of the hydroxyl and methoxy groups around the three rings (Fig. 1). Flavones, (e.g. apigenin, luteolin) are found in artichoke (Cynara cardunculus), celery (Apium graveolens L.) and parsley (Petroselinum crispum), chives (Allium schoenoprasum) and other herbs. Hydroxylation on position 3 of the flavone structure gives rise to the 3-hydroxyflavones, also termed the flavonols (e.g. kaempferol, quercetin), which are found predominantly in onions (Allium cepa L.), leeks (Allium ampeloprasum var. porrum (L.)) and broccoli (Brassica oleracea; Fig. 1). Dietary flavanones (e.g. naringenin, hesperetin, taxifolin) are found predominantly in citrus fruit and tomatoes (Fig. 1). Finally, isoflavones such as daidzein and genistein are a subclass of the flavonoid family found in soyabean and soya products. They have a large structural variability and >600 isoflavones have been identified to date and are classified according to oxidation level of the central pyran ring (Fig. 1).
Absorption, metabolism and distribution of flavonoids
Although flavonoids have been identified as powerful antioxidants in vitro (Reference Rice-Evans, Miller and Paganga9–Reference Rice-Evans11), their ability to act as antioxidants in vivo is limited by the extensive biotransformation and conjugation that occurs during their absorption from the gastrointestinal tract, in the liver and finally in cells (for review, see Williamson & Manach(Reference Williamson and Manach12), Manach et al.(Reference Manach, Williamson and Morand13), Scalbert & Williamson(Reference Scalbert and Williamson14) and Spencer et al.(Reference Spencer, Abd El and Minihane15)). In the small intestine and liver dietary flavonoids (and other polyphenols) are substrates for phase I (hydrolysing and oxidising) and phase II (conjugating and detoxifying) enzymes, i.e. they are de-glucosylated and metabolised into glucuronides, sulfates and O-methylated derivatives(Reference Scalbert and Williamson14, Reference Spencer, Chowrimootoo and Choudhury16, Reference Spencer17). Further metabolism occurs in the colon, in which the enzymes of the gut microflora induce the breakdown of flavonoids to simple phenolic acids that may then undergo absorption and are further metabolised in the liver(Reference Spencer, Abd El and Minihane15, Reference Scheline18). The extent of metabolism in the large intestine has been largely ignored to date, although there is now intense interest in the generation, absorption and potential bioactivity of these bacterially-derived forms. Moreover, it has recently been suggested that their metabolism by bacteria in the colon may also result in the selective beneficial growth of several bacterial groups and species(Reference Tzounis, Vulevic and Kuhnle19). Post absorption from the gastrointestinal tract flavonoids also undergo at least three types of intracellular metabolism: (1) oxidative metabolism; (2) P450-related metabolism; (3) conjugation with thiols, particularly glutathione(Reference Spencer, Kuhnle and Williams20). Many studies have indicated that although glucuronides, sulfates, O-methyl derivatives and intracellular metabolites such as flavonoid–glutathione adducts may still participate in antioxidant reactions (in particular, scavenging reactive oxygen and nitrogen species) in the circulation, their effectiveness to do so is greatly reduced relative to their parent aglycones (or indeed those found in plants)(Reference Spencer, Schroeter and Crossthwaithe21–Reference da Silva, Piskula and Yamamoto26).
In order for flavonoids to influence brain function directly they must additionally penetrate the blood–brain barrier, which controls entry of xenobiotics into the brain(Reference Abbott27). Flavanones such as hesperetin, naringenin and their in vivo metabolites, along with some dietary anthocyanins, cyanidin-3-rutinoside and pelargonidin-3-glucoside, have been shown to traverse the blood–brain barrier in relevant in vitro and in situ models(Reference Youdim, Qaiser and Begley28). Their extent of blood–brain barrier penetration is dependent on compound lipophilicity(Reference Youdim, Dobbie and Kuhnle29), i.e. less polar O-methylated metabolites may be capable of greater brain uptake than the more polar flavonoid glucuronides. However, evidence exists to suggest that certain drug glucuronides may cross the blood–brain barrier(Reference Aasmundstad, Morland and Paulsen30) and exert pharmacological effects(Reference Sperker, Backman and Kroemer31, Reference Kroemer and Klotz32), suggesting that there may be a specific uptake mechanism for glucuronides in vivo. Their brain entry may also depend on their interactions with specific efflux transporters expressed in the blood–brain barrier such as P-glycoprotein(Reference Lin and Yamazaki33), which appears to be responsible for the differences between naringenin and quercetin flux into the brain in situ (Reference Youdim, Qaiser and Begley28). In animals flavanones have been found to enter the brain following their intravenous administration(Reference Peng, Cheng and Huang34), whilst epigallocatechin gallate(Reference Suganuma, Okabe and Oniyama35), epicatechin(Reference Abd El, Kuhnle and Rechner36) and anthocyanins(Reference Abd El, Marks and Kuhnle37, Reference Talavera, Felgines and Texier38) are found in the brain after their oral administration. Furthermore, several anthocyanins have been identified in different regions of the brains of rats(Reference Passamonti, Vrhovsek and Vanzo39) and blueberry-fed pigs(Reference Kalt, Blumberg and McDonald40), with eleven intact anthocyanins found in the cortex and cerebellum. Although further work is necessary to establish their bioavailability to the brain, particularly in human subjects, these results suggest that they may localise in the brain and are capable of direct neuroprotective and neuromodulatory actions.
Antioxidants or signalling molecules?
Historically, the biological actions of flavonoids, including those on the brain, have been attributed to their ability to exert antioxidant actions(Reference Rice-Evans, Miller and Paganga9), through their ability to scavenge reactive species or through their possible influences on intracellular redox status(Reference Pollard, Kuhnle and Vauzour41). However, it is now thought highly unlikely that this classical H-donating antioxidant activity accounts for the bioactivity of flavonoids in vivo, particularly in the brain where they are found at only very low concentrations(Reference Spencer1). Indeed, it is clear that the concentrations of flavonoids and their metabolite forms accumulated in vivo (Reference Abd El, Kuhnle and Rechner42) are lower (high nm, low μm) than those recorded for small-molecule antioxidant nutrients such as ascorbic acid and α-tocopherol(Reference Halliwell, Zhao and Whiteman43). Consequently, the beneficial effects of flavonoid metabolites in vivo are unlikely to result from their ability to out-compete antioxidants such as ascorbate, which are present at higher concentrations (high μm to mm). However, evidence has accumulated to suggest that the cellular effects of flavonoids may be mediated by their interactions with specific proteins central to intracellular signalling cascades(Reference Schroeter, Boyd and Spencer44), such as the MAPK signalling pathway and the PI3K/Akt signalling cascade (Fig. 2). For example, flavonoids have been shown to be capable of exerting neuroprotective actions (at low concentration) via their interactions with critical neuronal intracellular signalling pathways pivotal in controlling neuronal survival and differentiation, long-term potentiation and memory(Reference Spencer3, Reference Spencer45–Reference Spencer47). The present review will examine such actions and how they may impact on the progression of chronic brain disease.

Fig. 2. Potential points of flavonoid action within mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3K)/Akt signalling cascades in neurons and glia. Activation of extracellular signal-regulated protein kinase (ERK) 1/2/5 and Akt are generally pro-survival, whilst inhibitory actions on c-Jun N-terminal kinase (JNK) and p38 pathways are also likely to be neuroprotective. Through their effects on these pathways they may regulate a wide variety of processes, including cell growth, cell proliferation, differentiation, cell cycle entry, cell migration and apoptosis. MEK, MKK, MAPK kinases; MEKK, MEK kinase; CREB, cAMP response element-binding protein; STAT, signal transducer and activator of transcription; ER, oestrogen receptor; ASK1, apoptosis signal-regulating kinase 1; ATF-2, activating transcription factor 2; MLK3, JNK/stress-activated protein kinase activator mixed lineage kinase 3; TAK, transforming growth factor β-activated kinase 1; PDK, phosphoinositide-dependent kinase-1; PKB, protein kinase B; BAD, Bcl-xL/Bcl-2 associated death promoter; mTOR, mammalian target of rapamycin; eNOS, endothelial nitric oxide synthase.
Direct interactions with signalling pathways
Flavonoids have been shown to exert neuronal effects through their interactions with a number of protein kinase and lipid kinase signalling cascades, such as the PI3K/Akt, tyrosine kinase, protein kinase C and MAPK signalling pathways(Reference Matter, Brown and Vlahos48–Reference Spencer, Rice-Evans and Williams54) (Fig. 2). Inhibitory or stimulatory actions at these pathways are likely to profoundly affect neuronal function by altering the phosphorylation state of target molecules and/or by modulating gene expression. Although selective inhibitory actions at these kinase cascades may be beneficial in cancer, proliferative diseases, inflammation and neurodegeneration, they could be detrimental during development, particularly in the immature nervous system in which protein kinase and lipid kinase signalling regulates survival, synaptogenesis and neurite outgrowth. In the mature brain post-mitotic neurones utilise MAPK and PI3K cascades in the regulation of key functions such as synaptic plasticity and memory formation(Reference Lin, Yeh and Lin55, Reference Sweatt56), thus flavonoid interactions within these pathways could have unpredictable outcomes and will be dependent both on the cell type and disease studied.
MAPK belong to the superfamily of serine/threonine kinases and play a central role in transducing various extracellular signals into intracellular responses(Reference Cobb and Goldsmith57, Reference Goldsmith and Cobb58). The best-characterised MAPK pathways are the mitogenic extracellular signal-regulated protein kinase (ERK) pathway and the stress-activated c-Jun N-terminal kinase (JNK) and p38 cascades (Fig. 2). Once activated ERK, JNK and p38 phosphorylate a number of cytosolic proteins and transcription factors resulting in the enhancement of their transcriptional activities and activation of dependent genes(Reference Karin59). ERK and JNK are generally considered as having opposing actions, in particular in neuronal apoptosis(Reference Xia, Dickens and Raingeaud60). ERK1/2 are usually associated with pro-survival signalling(Reference Anderson and Tolkovsky61–Reference Kaplan and Miller63) through mechanisms that may involve activation of the cAMP response element-binding protein (CREB)(Reference Bonni, Brunet and West62, Reference Crossthwaite, Hasan and Williams64) (Fig. 2), the up-regulation of the anti-apoptotic protein Bcl-2 and non-transcriptional inhibition of Bcl-xL/Bcl-2-associated death promoter(Reference Bonni, Brunet and West62, Reference Kaplan and Miller63). On the other hand, JNK has been strongly linked to transcription-dependent apoptotic signalling(Reference Mielke and Herdegen65, Reference Yuan and Yankner66), possibly through the activation of c-Jun(Reference Behrens, Sibilia and Wagner67) and other activated protein-1 proteins including JunB, JunD and activating transcription factor 2(Reference Davis68). Many investigations have indicated that flavonoids and their metabolites may interact selectively within the MAPK signalling pathways(Reference Kong, Yu and Chen52, Reference Kobuchi, Roy and Sen69). The potential modulation of MAPK signalling by flavonoids is important as ERK1/2 and JNK are involved in growth factor-induced mitogenesis, differentiation, apoptosis and various forms of cellular plasticity(Reference Mielke and Herdegen65, Reference Mielke and Herdegen66, Reference Mielke and Herdegen70–Reference Castagne, Gautschi and Lefevre72).
There is much evidence to support the actions of flavonoids on the ERK pathway(Reference Schroeter, Spencer and Rice-Evans53, Reference Vauzour, Vafeiadou and Rice-Evans73, Reference Llorens, Garcia and Itarte74), which appear to be mediated by interactions with MAPK kinases MEK1 and MEK2 and potentially membrane receptors(Reference Schroeter, Boyd and Spencer44, Reference Kong, Yu and Chen52). Indeed, flavonoids have close structural homology to specific pharmacological modulators of ERK signalling such as PD98059 (2′-amino-3′-methoxyflavone). The flavonol quercetin and to a lesser extent its O-methylated metabolites have been shown to induce neuronal apoptosis via a mechanism involving the inhibition of ERK rather than by induction of pro-apoptotic signalling through JNK(Reference Spencer, Rice-Evans and Williams54). The potent inhibition of ERK activation, and indeed Akt/protein kinase B (PKB) phosphorylation, is also accompanied by downstream activation of Bcl-xL/Bcl-2-associated death promoter and a subsequent strong activation of caspase-3. On the other hand, some flavonoids have been observed to exert a stimulatory effect on ERK1/2. For example, the flavan-3-ol (–)-epicatechin and one of its metabolites, 3′-O-methyl-(–)-epicatechin, have been shown to stimulate phosphorylation of ERK1/2 and the downstream transcription factor CREB at physiologically-relevant concentrations(Reference Schroeter, Bahia and Spencer75). Interestingly, this activation of the ERK pathway is no longer apparent at higher concentrations, suggesting that effects on this pathway are concentration specific. Furthermore, stimulation of ERK1/2 and CREB is not observed with (–)-epicatechin-5-O-β-d-glucuronide, suggesting that effects on the ERK pathway may be dependent on cell or membrane permeability, as has been previously reported(Reference Spencer, Schroeter and Crossthwaithe76). In support of these observations, the protective action of another flavanol, epigallocatechin gallate, against 6-hydroxydopamine toxicity and serum deprivation has been shown to involve the restoration of both protein kinase C and ERK1/2 activities(Reference Levites, Amit and Youdim77, Reference Reznichenko, Amit and Youdim78).
There is strong evidence linking the activation of JNK to neuronal loss in response to a wide array of pro-apoptotic stimuli in both developmental and degenerative death signalling(Reference Mielke and Herdegen65, Reference Davis68, Reference Davis79). The activation of the JNK pathway and the death of specific neuronal populations are crucial events during early brain development(Reference Leppa and Bohmann80). As with the other MAPK, the core signalling unit is composed of an MAPK kinase kinase, typically MAPK kinase kinases MEK1–4, which phosphorylate and activate MAPK kinases MKK4–7, which then phosphorylate and activate the JNK(Reference Davis79, Reference Ichijo81) (Fig. 2). Another MAPK kinase kinase, apoptosis signal-regulating kinase 1, also plays an essential role in stress-induced apoptosis(Reference Ichijo, Nishida and Irie82, Reference Wang, Diener and Jannuzzi83). Apoptosis signal-regulating kinase 1 can be activated in response to a variety of stress-related stimuli and activates MKK4, which in turn activates JNK (Fig. 2) and indeed p38(Reference Matsuzawa and Ichijo84). Overexpression of apoptosis signal-regulating kinase 1 has been shown to induce the activation of both JNK and p38 and lead to apoptosis via signals involving the mitochondrial cell death pathway(Reference Leppa and Bohmann80, Reference Ichijo, Nishida and Irie82). Investigation has indicated that oxidative-induced activation of caspase-3 in neurons is blocked by flavonoids, providing compelling evidence in support of a potent anti-apoptotic action of flavonoids in these cells(Reference Schroeter, Spencer and Rice-Evans53, Reference Vauzour, Vafeiadou and Rice-Evans73, Reference Spencer, Schroeter and Crossthwaithe76, Reference Schroeter, Williams and Matin85). The flavanols epicatechin and 3′-O-methyl-epicatechin have been shown to protect neurons against oxidative damage via a mechanism involving the suppression of JNK and downstream partners c-jun and pro-caspase-3(Reference Schroeter, Spencer and Rice-Evans53, Reference Spencer, Schroeter and Kuhnle86). Similarly, the flavone baicalein has been shown to inhibit 6-hydroxydopamine-induced JNK activation and neuronal cell death and quercetin may suppress JNK activity and apoptosis induced by H2O2(Reference Wang, Matsushita and Araki87, Reference Ishikawa and Kitamura88), 4-hydroxy-2-nonenal(Reference Uchida, Shiraishi and Naito89) and TNFα(Reference Kobuchi, Roy and Sen69).
In addition to the MAPK pathway flavonoids have been shown to modulate signalling through the serine/threonine kinase Akt/PKB, one of the main downstream effectors of PI3K, a pivotal kinase in neuronal survival(Reference Kennedy, Wagner and Conzen90–Reference Crowder and Freeman93) (Fig. 2). Flavonoids have long been known to modulate PI3K, via direct interactions with its ATP-binding site(Reference Gamet-Payrastre, Manenti and Gratacap51). Indeed, a number of studies have demonstrated that the structure of flavonoids determines whether or not they act as potent inhibitors of PI3K(Reference Agullo, Gamet-Payrastre and Manenti50, Reference Ferriola, Cody and Middleton94). One of the most selective PI3K inhibitors available, LY294002, was modelled on the structure of quercetin(Reference Matter, Brown and Vlahos48, Reference Vlahos, Matter and Hui49, Reference Walker, Pacold and Perisic95). Quercetin and some of its in vivo metabolites have been shown to inhibit pro-survival Akt/PKB signalling pathways by a mechanism of action consistent with quercetin and its metabolites acting at and inhibiting PI3K activity(Reference Spencer, Rice-Evans and Williams54). However, other flavonoids such as the citrus flavanone hesperetin induce the activation of Akt/PKB and the inhibition of pro-apoptotic proteins such as apoptosis signal-regulating kinase 1, Bcl-xL/Bcl-2-associated death promoter, caspase-9 and caspase-3 in cortical neurons(Reference Vauzour, Vafeiadou and Rice-Evans73).
Inhibition of neuroinflammation
Neuroinflammatory processes in the brain are believed to play a crucial role in the development of Alzheimer's disease and Parkinson's disease(Reference Hirsch, Hunot and Hartmann96, Reference McGeer and McGeer97) as well as injury associated with stroke(Reference Zheng, Lee and Yenari98). Activated microglia and/or astrocytes release cytokines and other mediators that have been linked to the apoptotic death of neurons. In particular, increases in cytokine production (IL-1β, TNFα), inducible NO synthase (iNOS) and NO and increased NADPH oxidase activation(Reference Kozuka, Itofusa and Kudo99) all contribute to glial-induced neuronal death (Fig. 3). The majority of these events are controlled by upstream MAPK signalling, which mediates both the transcriptional and post-transcriptional regulation of iNOS and cytokines in activated microglia and astrocytes(Reference Marcus, Karackattu and Fleegal100, Reference Bhat, Zhang and Lee101). Evidence suggests that the non-steroidal anti-inflammatory drug ibuprofen may be effective in delaying the onset of neurodegenerative disorders, particularly Parkinson's disease, by reducing inflammatory injury in specific brain regions(Reference Casper, Yaparpalvi and Rempel102). Thus, there is a desire to develop new drugs capable of preventing progressive neuronal loss linked to neuroinflammation(Reference Vafeiadou, Vauzour and Spencer2). Recently, the flavanone naringenin, found at high concentrations in citrus fruits, has been found to be highly effective in reducing lipopolysaccharide- and interferon-γ-induced glial cell activation and resulting neuronal injury(Reference Vafeiadou, Vauzour and Lee103) via an inhibition of p38 and signal transducer and activator of transcription-1 and a reduction in iNOS expression (Fig. 3). The structurally-related flavanone hesperetin and other flavonoids appear to be incapable of inhibiting pathways leading to NO production, although they have been found to partially alleviate neuroinflammation through the inhibition of TNFα production(Reference Vafeiadou, Vauzour and Lee103).

Fig. 3. The cellular mechanisms by which flavonoids and their metabolites protect against neuroinflammation and neuronal injury induced by 5-S-cysteinyl-dopamine (CysDA), dihydrobenzothiazine (DHBT-1) and related reactive oxygen species (ROS). Flavonoids inhibit the p38 pathway glia cells leading to a reduction in inducible nitric oxide synthase (iNOS) expression and nitric oxide release. In neurons they scavenge neurotoxic species and induce pro-survival signalling pathways, such as extracellular signal-regulated protein kinase (ERK) 1/2 and phosphatidylinositol 3-kinase (PI3K)/Akt, leading to an inhibition of neuronal apoptosis. STAT-1, signal transducer and activator of transcription-1; IFN-γ, interferon-γ; RNS, reactive nitrogen species; ASK1, apoptosis signal-regulating kinase 1; JNK, c-Jun N-terminal kinase; BAD, Bcl-xL/Bcl-2 associated death promoter; MEK1/2, mitogen-activated protein kinase kinases; CREB, cAMP response element-binding protein. ![]() , Activation;
, Activation; ![]() , inhibition.
, inhibition.
Flavonoids present in blueberry have also been shown to inhibit NO, IL-1β and TNFα production in activated microglia cells(Reference Lau, Bielinski and Joseph104), whilst the flavonol quercetin(Reference Chen, Ho and Pei-Dawn105), the flavones wogonin and bacalein(Reference Lee, Kim and Kim106), the flavanols catechin and epigallocatechin gallate(Reference Li, Huang and Fang107) and the isoflavone genistein(Reference Afaq, Adhami and Ahmad108) have all been shown to attenuate microglia- and/or astrocyte-mediated neuroinflammation via mechanisms that include inhibition of, in astrocytes and microglia: (1) iNOS and cyclooxygenase 2 expression; (2) NO production; (3) cytokine release; (4) NADPH oxidase activation and subsequent reactive oxygen species generation. All these effects appear to depend on an ability to directly modulate protein kinase and lipid kinase signalling pathways(Reference Williams, Spencer and Rice-Evans45, Reference Spencer46). For example, they may act by inhibiting MAPK signalling cascades, such as p38 or ERK1/2, which regulate both iNOS and TNFα expression in activated glial cells(Reference Bhat, Zhang and Lee101, Reference Vafeiadou, Vauzour and Lee103) (Fig. 3); fisetin inhibits p38 MAPK phosphorylation in lipopolysaccharide-stimulated BV-2 microglial cells(Reference Zheng, Ock and Kwon109) and the flavone luteolin inhibits IL-6 production in activated microglia via inhibition of the JNK signalling pathway(Reference Jang, Kelley and Johnson110). The effects of flavonoids on these kinases may influence downstream pro-inflammatory transcription factors important in iNOS transcription. One of these transcription factors, NF-κB, responds to p38 signalling and is involved in iNOS induction(Reference Bhat, Feinstein and Shen111), suggesting that there is interplay between signalling pathways, transcription factors and cytokine production in determining the neuroinflammatory response in the central nervous system. In support of this notion, some flavonoids have been shown to prevent transcription factor activation, with the flavonol quercetin and the flavanone naringenin able to suppress NF-κB, signal transducer and activator of transcription-1 and activating protein-1 activation in lipopolysaccharide- and interferon-γ-activated microglial cells(Reference Vafeiadou, Vauzour and Lee103, Reference Chen, Ho and Pei-Dawn105).
Inhibition of toxin-induced neuronal injury
The underlying neurodegeneration observed in Parkinson's disease, Alzheimer's disease, and other neurodegenerative diseases is believed to be triggered by multi-factorial processes, including neuroinflammation, glutamatergic excitotoxicity, increases in Fe and/or depletion of endogenous antioxidants(Reference Barzilai and Melamed112–Reference Spires and Hannan114). There is a growing body of evidence to suggest that flavonoids and other polyphenols may be able to counteract this neuronal injury, thereby delaying the progression of these brain pathologies(Reference Spencer1, Reference Spencer46, Reference Spencer115–Reference Joseph, Cole and Head119). For example, a Ginkgo biloba extract has been shown to protect hippocampal neurons against NO- and β-amyloid-induced neurotoxicity(Reference Luo, Smith and Paramasivam120) and studies have demonstrated that the consumption of green tea may have a beneficial effect in reducing the risk of Parkinson's disease(Reference Mandel, Amit and Weinreb121–Reference Mandel, Avramovich-Tirosh and Reznichenko124). In agreement with the latter study, tea extracts and pure (–)-epigallocatechin 3-gallate have been shown to attenuate 6-hydroxydopamine-induced toxicity(Reference Levites, Youdim and Maor125), to protect against hippocampal injury during transient global ischaemia(Reference Lee, Suh and Kim126) and to prevent nigral damage induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine(Reference Levites, Weinreb and Maor127).
The death of nigral neurons in Parkinson's disease is thought to involve the formation of the endogenous neurotoxin 5-S-cysteinyl-dopamine and its oxidation product dihydrobenzothiazine(Reference Spencer, Whiteman and Jenner128, Reference Hastings129) (Fig. 3). 5-S-cysteinyl-catecholamine conjugates possess strong neurotoxicity and initiate a sustained increase in intracellular reactive oxygen species in neurons leading to DNA oxidation, caspase-3 activation and delayed neuronal death(Reference Spencer, Whiteman and Jenner128) (Fig. 3). Such adducts may be generated by reactive species(Reference Vauzour, Ravaioli and Vafeiadou130) and have been observed post mortem to be elevated in the substantia nigra of patients with Parkinson's disease(Reference Spencer, Jenner and Daniel131), suggesting that such species may be potential endogenous nigral toxins. However, 5-S-cysteinyl-dopamine-induced neuronal injury is effectively counteracted by nanomolar concentrations of various flavonoids, including pelargonidin, quercetin, hesperetin, caffeic acid, 4′-O-methyl derivatives of catechin and epicatechin(Reference Vauzour, Ravaioli and Vafeiadou130). Furthermore, in the presence of the flavanol (+)-catechin tyrosinase-induced formation of 5-S-cysteinyldopamine is inhibited by a mechanism linked to the capacity of catechin to undergo tyrosinase-induced oxidation to yield cysteinyl-catechin adducts(Reference Vauzour, Vafeiadou and Spencer132). In contrast, the inhibition afforded by flavanones, such as hesperetin, is not accompanied by the formation of cysteinyl-hesperetin adducts, indicating that it may inhibit via direct interaction with tyrosinase(Reference Vauzour, Vafeiadou and Spencer132).
Reactive oxygen and nitrogen species have also been proposed to play a role in the pathology of many neurodegenerative diseases(Reference Jellinger113) (Fig. 3). There is abundant evidence that flavonoids are effective in blocking this oxidant-induced neuronal injury, although their potential to do so is thought not to rely on direct radical- or oxidant-scavenging activity(Reference Spencer, Schroeter and Crossthwaithe76, Reference Spencer, Schroeter and Kuhnle86). Instead, they are believed to act by modulating a number of protein kinase and lipid kinase signalling cascades, such as the PI3K/Akt, tyrosine kinase, protein kinase C and MAPK signalling pathways(Reference Spencer1, Reference Spencer46). Inhibitory or stimulatory actions at these pathways are likely to profoundly affect neuronal function by altering the phosphorylation state of target molecules, leading to changes in caspase activity, and/or by gene expression. For example, flavonoids have been observed to block oxidative-induced neuronal damage by modulating the activation of both the MAPK(Reference Schroeter, Spencer and Rice-Evans53, Reference Schroeter, Spencer and Rice-Evans87–Reference Uchida, Shiraishi and Naito89) and PI3K/Akt(Reference Vauzour, Vafeiadou and Rice-Evans73) signalling pathways and the activation of caspase-3(Reference Spencer, Schroeter and Crossthwaithe76, Reference Spencer, Schroeter and Kuhnle86), providing evidence in support of their potent anti-apoptotic action, and have been found to protect neurons against a variety of neurotoxic insults.
Modulation of synaptic plasticity and neuro-cognitive performance
There is now much evidence to suggest that fruit- and vegetable-derived phytochemicals, in particular flavonoids, are capable of promoting beneficial effects on memory, learning and cognitive performance(Reference Joseph, Cole and Head119, Reference Williams, El Mohsen and Vauzour133–Reference Shukitt-Hale, Carey and Simon141). It appears that these low-molecular-weight non-nutrient components are able to impact on memory through their ability to exert effects directly on the brain's innate architecture for memory(Reference Spencer3, Reference Spencer47, Reference Spencer142, Reference Stangl and Thuret143). The concentrations of flavonoids and their metabolites that reach the brain are thought to be sufficiently high to exert pharmacological activity at receptors, kinases and transcription factors. Although the precise site of their interaction with signalling pathways remains unresolved, evidence indicates that they are capable of acting in a number of ways: (1) by binding to ATP sites on enzymes and receptors; (2) by modulating the activity of kinases directly, i.e. MAPK kinase kinase, MAPK kinase or MAPK; (3) by affecting the function of important phosphatases, which act in opposition to kinases; (4) by preserving Ca2+ homeostasis, thereby preventing Ca2+-dependent activation of kinases in neurons; (5) by modulating signalling cascades lying downstream of kinases, i.e. transcription factor activation and binding to promoter sequences(Reference Spencer3, Reference Spencer, Vauzour and Rendeiro4). By affecting such pathways they have the potential to induce new protein synthesis in neurons and thus an ability to induce morphological changes that have a direct influence on memory acquisition, consolidation and storage.
Various individual cascades have been linked with this control of de novo protein synthesis in the context of long-term potentiation, synaptic plasticity and memory (Fig. 3): (1) cAMP-dependent protein kinase A; (2) PKB/Akt(Reference Ichijo81); (3) protein kinase C; (4) Ca–calmodulin kinase(Reference Wang, Diener and Jannuzzi83); (5) ERK(Reference Spencer3). All five pathways converge to signal to CREB, a transcription factor that binds to the promoter regions of many genes associated with synapse re-modelling, synaptic plasticity and memory (Fig. 4). Flavonoids are now well known to modulate neuronal signalling pathways crucial in inducing synaptic plasticity(Reference Spencer3), and although each of these pathways are known to be involved in increasing the number of, and strength of, connections between neurons, flavonoids appear to interact primarily with the ERK and PKB/Akt pathways(Reference Spencer46, Reference Spencer, Rice-Evans and Williams54, Reference Schroeter, Bahia and Spencer75). The activation of these pathways by blueberry flavonoids, along with the activation of the transcription factor CREB and production of neurotrophins such as brain-derived neurotrophic factor (BDNF), is known to be required during memory acquisition and consolidation. Agents capable of inducing pathways leading to CREB activation will have the potential to enhance both short-term and long-term memory(Reference Williams, El Mohsen and Vauzour133) by providing a more efficient structure for interpreting afferent nerve or sensory information. One mechanism by which this provision may come about is through flavonoid-induced increases in neuronal spine density and morphology, two factors considered to be vital for learning and memory(Reference Harris and Kater144). Changes in spine density, morphology and motility have been shown to occur with paradigms that induce synaptic as well as altered sensory experience and lead to alterations in synaptic connectivity and strength between neuronal partners, affecting the efficacy of synaptic communication (Fig. 4).

Fig. 4. Flavonoid-induced activation of neuronal signalling and gene expression in the brain. Such processes may lead to changes in synaptic plasticity and neurogenesis in the brain that ultimately influence memory, learning and cognition. PKA, PKB, PKC, protein kinase A, B and C respectively; CaMK, Ca–calmodulin kinase; ERK, extracellular signal-regulated protein kinase; CREB, cAMP response element-binding protein; BDNF, brain-derived neurotrophic factor; Arc, the activity-regulated cytoskeletal-associated protein termed Arc/Arg3.1; NMDA-R, N-methyl d-aspartate receptor; mTOR, mammalian target of rapamycin; VEGF-β, vascular endothelial growth factor β; TGF-β, transforming growth factor β.
Fisetin, a flavonoid found in strawberries, has been shown to improve long-term potentiation and to enhance object recognition in mice by a mechanism dependent on the activation of ERK and CREB(Reference Maher, Akaishi and Abe145, Reference Maher146). Similarly, the flavanol (–)-epicatechin induces both ERK1/2 and CREB activation in cortical neurons and subsequently increases CREB-regulated gene expression(Reference Schroeter, Bahia and Spencer75), whilst nanomolar concentrations of quercetin are effective at enhancing CREB activation(Reference Spencer, Rice-Evans and Williams54). Blueberry-induced improvements in memory have been shown to be mediated by increases in the phosphorylation state of ERK1/2, rather than that of Ca–calmodulin kinase (II and IV) or protein kinase A(Reference Williams, El Mohsen and Vauzour133). Other flavonoids have also been found to influence the ERK pathway, with the citrus flavanone hesperetin capable of activating ERK1/2 signalling in cortical neurons(Reference Vauzour, Vafeiadou and Rice-Evans73) and flavanols such as (–)-epigallocatechin 3-gallate restoring both protein kinase C and ERK1/2 activities in 6-hydroxydopamine-treated and serum-deprived neurons(Reference Levites, Amit and Youdim77, Reference Reznichenko, Amit and Youdim78). Furthermore, this ability to activate the ERK pathway is not restricted to neurons and has also been observed in fibroblasts exposed to low concentrations of epicatechin(Reference Pollard, Whiteman and Spencer147).
CREB activation downstream of ERK appears critical in the induction of long-lasting changes in synaptic plasticity and memory(Reference Pham, Impey and Storm148–Reference Impey, Mark and Villacres150) and disruption of CREB activity specifically blocks the formation of long-term memory(Reference Bourtchuladze, Frenguelli and Blendy151), whereas agents that increase the amount or activity of CREB accelerate the process(Reference Tully, Bourtchouladze and Scott152). CREB is known to be a critical transcription factor linking the actions of neurotrophins such as BDNF to neuronal survival, differentiation and synaptic function(Reference Conkright, Guzman and Flechner153–Reference Finkbeiner, Tavazoie and Maloratsky155). Consequently, the central role of CREB in these processes has led to considerable interest in identifying safe effective agents that may enhance the activity of CREB in specific regions of the brain, as these agents may lead to an improvement in memory(Reference Tully, Bourtchouladze and Scott152). Recent studies have shown that spatial memory performance in rats supplemented with blueberry correlates well with the activation of CREB and with increases in both pro-BDNF and mature BDNF levels in the hippocampus(Reference Williams, El Mohsen and Vauzour133). Regulation of BDNF is interesting as this neurotrophin is intimately linked to the control of synaptic plasticity and long-term memory(Reference Bramham and Messaoudi156) (Fig. 5) and decreases in BDNF and pro-BDNF have been reported in Alzheimer's disease(Reference Peng, Wuu and Mufson157, Reference Michalski and Fahnestock158). Furthermore, a polymorphism that replaces valine for methionine at position 66 of the pro-domain of BDNF is associated with memory defects and abnormal hippocampal function in human subjects(Reference Egan, Kojima and Callicott159).

Fig. 5. Generation of brain-derived neurotrophic factor (BDNF) by flavonoids may occur in the brain (neurons and glial cells) and the periphery (endothelial cells). As BDNF may cross the blood–brain barrier (BBB) both sites of generation have the potential to be relevant to changes in cognition. Additionally, plasma measures of BDNF may reflect brain generation in response to flavonoids and general cognitive performance.
Flavonoid-induced activation of CREB and BDNF expression has also been shown to lead to the activation of the PI3K/Akt signalling pathway(Reference Williams, El Mohsen and Vauzour133), presumably via the binding of BDNF to pre- or post-synaptic tropomyosin receptor kinase B receptor. The activation of Akt by flavonoids in the hippocampus triggers the activation of the mTOR pathway and the increased translation of specific mRNA subpopulations(Reference Schratt, Nigh and Chen160), including the activity-regulated cytoskeletal-associated protein termed Arc/Arg3.1(Reference Williams, El Mohsen and Vauzour133), which is known to be important in long-term potentiation and has been proposed to be under regulatory control of both BDNF(Reference Yin, Edelman and Vanderklish161) and the ERK signalling(Reference Waltereit, Dammermann and Wulff162) (Fig. 4). Increased Arc/Arg3.1 expression may facilitate changes in synaptic strength and the induction of morphological changes such as that observed when small spines are converted into large mushroom-shaped spines through a mechanism dependent on actin polymerisation(Reference Lyford, Yamagata and Kaufmann163). In support of this role, studies have indicated that changes in neuronal morphology occur in response to flavonoid supplementation(Reference van Praag, Lucero and Yeo164) and that certain flavonoids can influence neuronal dendrite outgrowth in vitro (Reference Reznichenko, Amit and Youdim78). Furthermore, nobiletin (a poly-methoxylated flavone found in citrus peel) also induces neurite outgrowth(Reference Nagase, Yamakuni and Matsuzaki165) and synaptic transmission(Reference Matsuzaki, Miyazaki and Sakai166) via its ability to interact directly with MAPK and protein kinase A signalling pathways, whilst its metabolite 4′-demethylnobiletin exerts similar effects via the same pathways(Reference Al Rahim, Nakajima and Saigusa167).
Effects on the peripheral and cerebrovascular system
Recent dietary interventions in human subjects using flavanol-containing foods have substantiated epidemiological data for an inverse relationship between flavanol intake and the risk of CVD, indicating various potential flavanol-mediated bioactivities including the improvement of vasodilatation(Reference Fisher, Hughes and Gerhard-Herman168–Reference Heiss, Dejam and Kleinbongard172), blood pressure(Reference Taubert, Berkels and Roesen173), insulin resistance and glucose tolerance(Reference Grassi, Lippi and Necozione174), the attenuation of platelet reactivity(Reference Holt, Schramm and Keen175) and the improvement of immune responses and antioxidant defence systems(Reference Sies, Schewe and Heiss176, Reference Keen, Holt and Oteiza177) (Fig. 6). The intake of flavanol-rich foods such as cocoa or of pure (–)-epicatechin has been shown to be highly effective in improving peripheral blood flow and surrogate markers of cardiovascular function(Reference Schroeter, Heiss and Balzer178). The intake of flavanols has been shown to result in acute elevations in the level of circulating NO species, an enhanced flow-mediated dilatation response of conduit arteries and an augmented microcirculation(Reference Heiss, Dejam and Kleinbongard172, Reference Schroeter, Heiss and Balzer178, Reference Fisher and Hollenberg179). For example, increases in flow-mediated dilatation and plasma NO species in patients with hypertension, diabetes and coronary artery disease have been observed after consumption of flavanol-rich cocoa or chocolate(Reference Heiss, Dejam and Kleinbongard172, Reference Grassi, Lippi and Necozione174) and flavanol-rich cocoa reverses endothelial dysfunction in smokers(Reference Heiss, Kleinbongard and Dejam180) and hypercholesterolaemia in post-menopausal women(Reference Wang-Polagruto, Villablanca and Polagruto181). Furthermore, increases in flow-mediated dilatation in patients with coronary artery disease have also been observed following consumption of black tea or grape juice(Reference Stein, Keevil and Wiebe182, Reference Duffy, Keaney and Holbrook183). It has also been reported that flavanols might help to lower blood pressure in subjects with hypertension(Reference Taubert, Berkels and Roesen173, Reference Taubert, Roesen and Schomig184, Reference Grassi, Necozione and Lippi185) and in healthy individuals(Reference Grassi, Lippi and Necozione174). It has been proposed that these effects are mediated by an ability of flavonoids to increase circulating NO levels, perhaps through actions directly on endothelial NO synthase(Reference Fisher, Hughes and Gerhard-Herman168, Reference Karim, McCormick and Kappagoda186–Reference Leikert, Rathel and Wohlfart188). Indeed, in vitro experiments have indicated that cocoa flavanols(Reference Karim, McCormick and Kappagoda186) and grape-derived polyphenols(Reference Fitzpatrick, Hirschfield and Coffey189, Reference Cishek, Galloway and Karim190) have the ability to induce endothelium-dependent dilatation via a direct activation of the endothelial NO synthase and NO production. Although, it is unlikely that oligomeric forms of flavanols may exert such actions in vivo (because of their poor absorption), flavanol monomers, specific flavanol metabolites, flavonols and flavones have also been observed to influence NO production and endothelium-dependent relaxation in vivo (Reference Schroeter, Heiss and Balzer178, Reference Steffen, Jung and Klotz191–Reference Ajay, Achike and Mustafa193).

Fig. 6. The peripheral vascular and cardiovascular effects of flavonoid-rich foods. Flavanols in particular have been shown to induce a number of cardiovascular risk factors including blood pressure, vascular function and blood clotting. Such vascular effects are also thought to play a role in determining brain blood flow and changes in cognitive performance. Blueberry, Vaccinium corymbosum.
This ability to affect vascular function may also play a role in determining flavonoid effects in the brain, in particular effects on cognition, which are known to also depend on brain blood flow. There is evidence to suggest that flavonoids are capable of preventing many forms of cerebrovascular disease, including those associated with stroke and dementia(Reference Commenges, Scotet and Renaud194, Reference Dai, Borenstein and Wu195). Flavonoids, in particular flavanols, have been shown to influence cerebrovascular blood flow(Reference Fisher, Sorond and Hollenberg196, Reference Francis, Head and Morris197) and these vascular effects are potentially important as increased cerebrovascular function is known to facilitate adult neurogenesis in the hippocampus(Reference Stangl and Thuret143, Reference Gage198) (Fig. 4). Indeed, new hippocampal cells are clustered near blood vessels, proliferate in response to vascular growth factors and may influence memory(Reference Palmer, Willhoite and Gage199). Furthermore, efficient cerebral blood flow is vital for optimal brain function, with several studies indicating that there is a decrease in cerebral blood flow in patients with dementia(Reference Nagahama, Nabatame and Okina200, Reference Ruitenberg, den Heijer and Bakker201). Brain imaging techniques such as functional MRI and trans-cranial Doppler ultrasound have shown that there is a correlation between cerebral blood flow and cognitive function in human subjects(Reference Ruitenberg, den Heijer and Bakker201). For example, cerebral blood flow velocity is lower in patients with Alzheimer's disease and low cerebral blood flow is also associated with incipient markers of dementia. In contrast, subjects without dementia with higher cerebral blood flow are less likely to develop dementia. As mentioned earlier, flavonoids are capable of inducing increased cerebral blood flow in human subjects 1–2 h post intervention(Reference Fisher, Sorond and Hollenberg196, Reference Francis, Head and Morris197). After consumption of a flavanol-rich cocoa drink the blood oxygenation level-dependent functional MRI shows an increase in blood flow in certain regions of the brain, along with a modification of the flow oxygenation level-dependent response to task switching. Furthermore, arterial spin-labelling sequence MRI also indicates that cocoa flavanols increase cerebral blood flow for ⩽2 h post ingestion(Reference Wang, Fernandez-Seara and Alsop202). In support of these findings, an increase in cerebral blood flow through the middle cerebral artery has been reported after the consumption of flavanol-rich cocoa using trans-cranial Doppler ultrasound(Reference Fisher, Sorond and Hollenberg196).
Present and future perspectives
The actions of dietary flavonoids on cognition appear to involve a number of effects within the brain, including a potential to protect neurons against injury induced by neurotoxins and neuroinflammation, a potential to activate synaptic signalling and an ability to improve cerebrovascular blood flow. These effects appear to be underpinned by an ability to interact with cell signalling cascades in the brain and the periphery, leading to an inhibition of apoptosis triggered by neurotoxic species, the promotion of neuronal survival and differentiation and an enhancement of peripheral and cerebral blood perfusion. Such effects induce beneficial changes in the cellular architecture required for cognition and consequently provide the brain with a more efficient structure for interpreting afferent nerve or sensory information and for the storage, processing and retrieval of memory. Furthermore, such interactions also protect the brain against neuronal losses associated with ageing, which is particularly relevant as this innate brain structure is known to deteriorate with aging, with neuronal populations or synaptic connections lost over time, leaving the system less efficient in the processing and storage of sensory information.
However, although flavonoid consumption may have the potential to limit or even reverse age-dependent deteriorations in brain function, there are a number of questions still to be resolved. Most notably, at present there are no data in support of a causal relationship between the consumption of flavonoids and behavioural outcomes in human subjects. In order to identify such relationships future intervention studies will be required to utilise better-characterised intervention materials, more appropriate controls and more rigorous clinical outcomes. Whilst cognitive behavioural testing in human subjects and animals provides an appropriate way of assessing function, in vivo structural and dynamic quantitative assessments will ultimately be required to provide hard evidence of effects in the brain. For example, it would be highly advantageous to directly link behavioural responses to changes in hippocampal volume and density, changes in neural stem cell and progenitor cells and alterations in brain blood flow using MRI and functional MRI techniques. Functional MRI measures may be used to assess changes in blood flow that underlie improved cognitive functioning as a result of flavonoid supplementation. In addition, such haemodynamic changes may be further compared with changes in grey matter density and to biomarkers of neural stem and progenitor cells using H1-NMR spectroscopy. Such an approach will be essential to provide links between flavonoid intake and brain function in a mechanistic, dynamic and quantitative way. Taking such an approach it may also be possible to assess other factors relating to intake such as what time frame is required to gain maximum beneficial effects, which flavonoids are most effective in inducing these changes and in which doses?
Furthermore, the modulation of neurotrophic factors such as BDNF represent useful targets for the prevention of cognitive decline as they are known to be critical in both the protection and repair of neurons in the central nervous system(Reference Tapia-Arancibia, Rage and Givalois203, Reference Givalois, Arancibia and Alonso204). For example, levels of brain BDNF have been shown to correlate with human learning, memory and cognitive function(Reference Laske, Stransky and Eschweiler205–Reference Laske, Stransky and Leyhe207). Future efforts should focus on whether the ability of flavonoids to induce improvements in cognition is mediated by their ability to induce BDNF and/or other neurotrophin production in either the brain or the periphery (Fig. 5). Although highly-specific behavioural tests exist to determine cognitive performance in human subjects, presently there is a lack of biochemical markers that can be used as surrogates of human cognitive performance. BDNF may be one such functional biomarkers, as it has been shown to cross the blood–brain barrier and thus levels in the plasma may reflect levels in the brain(Reference Shimizu, Hashimoto and Okamura208) (Fig. 5). As flavonoid consumption has been reported to increase BDNF expression in rat brain and this increase is related to an improvement in spatial working memory(Reference Williams, El Mohsen and Vauzour133), there is a possibility that the same may occur in human subjects and that increases in brain BDNF may be detected as a biomarker in plasma (Fig. 5). Thus, future studies should investigate the usefulness of BDNF and other brain-derived components as biomarkers of cognitive changes in human subjects and whether they also respond to intervention with flavonoid-rich foods.
Finally, the potential impact of diet on healthcare costs should not be ignored. Dementia costs in the UK alone have been estimated to be £17 ×109 per year. Development of a treatment that would reduce severe cognitive impairment in older individuals by only 1% per year would cancel out all estimated increases in the long-term care costs for the ageing population(209). Also, there is intense interest in the development of drugs capable of enhancing memory and learning, both in adults and children, and there is a strong possibility that in the future specific nutrients, in particular fruit-derived flavonoids, might act as precursors for the development of a new generation of memory-enhancing drugs.
Acknowledgements
The author is funded by the Biotechnology and Biological Sciences Research Council (BB/F008953/1; BB/E023185/1; BB/G005702/1), the Food Standards Agency (FLAVURS) and the EU (FP7 FLAVIOLA). The author declares no conflict of interest.