- eNOS
endothelial NO synthase
- ET-1
endothelin-1
- HUVEC
human umbilical vein endothelial cells
In the UK there was a dramatic increase in the incidence rates of CVD such as CHD and atherosclerosis during the 20th century up to the end of the 1970s(Reference Allender, Peto, Scarborough, Boxer and Rayner1). Although the numbers of deaths from these diseases are currently decreasing within the UK population, CVD are still the main cause of death in the UK, and the incidence rates of CVD are on the increase worldwide. In 2005 CVD was the main cause of morbidity in the UK, resulting in 208 229 deaths, with CHD accounting for 48·5% of these cases(Reference Allender, Peto, Scarborough, Boxer and Rayner1). Thus, efforts are being made to identify methods for both the prevention and the treatment of CVD to further reduce the rate of incidence of these diseases within both the UK population and worldwide.
CVD refers to a group of diseases affecting the heart and/or blood vessels. These diseases usually result from arterial damage, therefore the symptoms and treatment of these diseases are dependent on the set of arteries affected(Reference Vander, Sherman and Luciano2). The three types of arteries predominantly affected by CVD include coronary arteries (CHD), cerebral arteries (stroke) and peripheral arteries (tissue death and gangrene)(Reference Vander, Sherman and Luciano2). CHD and stroke are the two most important CVD and are responsible for the greatest number of deaths among the UK population(Reference Allender, Peto, Scarborough, Boxer and Rayner1). CHD begins with inflammation of the wall of blood vessels, which then narrow, leading to angina pectoris(Reference Guyton and Hall3). In later stages of this disease blood clots obstruct arteries and severe myocardial ischaemia results in a myocardial infarction (heart attack). In extreme cases of CHD heart failure may occur as a result of deteriorated strength of the heart muscle to pump blood around the body(Reference Guyton and Hall3).
There are also other illnesses that may develop in association with these three main CVD, such as hypertension, cardiomegaly, cardiomyopathy and cardiac arrest(Reference Allender, Peto, Scarborough, Boxer and Rayner1). There are numerous risk factors that predispose individuals to CVD, including obesity, high blood pressure, age, gender, tobacco smoking and diabetes mellitus(Reference De Backer, Ambrosionie and Borch-Johnsen4). Treatments for CVD are dependent on the type of disease, but effective treatments always include prevention of risk factors such as low-fat, low-energy and low-Na diets, reduction in alcohol intake and regular cardiovascular exercise(Reference De Backer, Ambrosionie and Borch-Johnsen4).
The inclusion of plant products such as fruit and vegetables in the diet has long been associated with numerous health benefits, which has led the Department of Health to recommend the intake of five portions of fruit and vegetables daily(5). Early studies addressing the issue of prevention methods for CVD have shown that the consumption of fruit and vegetables plays a role in preventing the onset of CVD such as atherosclerosis, IHD and CHD(Reference Brat, Georgé, Bellamy, Du Chaffaut, Scalbert, Mennen, Arnault and Amiot6, Reference Hertog, Feskens and Kromhout7). Epidemiological studies have also shown an association between the intake of teas, particularly green teas (Camillia sinensis), and a decrease in CHD(8,9). There is also evidence that moderate consumption of other plant-based products such as wine provide cardiovascular protection against disease and offer antiproliferative, anti-inflammatory and antioxidant activity in vitro and in vivo (Reference Manach, Mazur and Scalbert9).
Early assumptions
The beneficial effects of fruit and vegetables on reducing CVD onset and in CVD treatment were originally attributed to the presence of the antioxidant vitamins C and E, which were believed to act as free radical scavengers. However, many intervention studies have failed to find any association between the beneficial effects observed with fruit and vegetable intake and vitamins C and E(Reference Muntwyler, Hennekens, Manson, Buring and Gaziano10). Other studies that have looked at combined supplements of the antioxidant vitamins C and E and β-carotene have also found insufficient correlations between the intake of these vitamins and reduced CVD(11).
Thus, researchers began to look at other compounds present in plants that may be responsible for the beneficial effects of fruit and vegetables on CVD. Numerous compounds such as vitamins, amino acids, polyphenols, alkaloids and lectins present in plant foods have been found to have health-beneficial properties. It was suggested that polyphenols present in plants are the main components responsible for the cardiovascular health benefits conveyed by fruit and vegetables, initially because of their antioxidant properties. Further study has now demonstrated that in addition to their antioxidant properties polyphenols have antiangiogenic and antiproliferative activity, inhibit inflammatory mediators and down regulate the expression of transcription factors and genes involved in hypertension(Reference Hakkinen12).
Polyphenolic compounds
Polyphenols are natural products found in a diverse range of plants that arise through one of two metabolic pathways: the ‘shikimate pathway’, which provides phenylpropanoids, hydroxycinnamic acids and coumarins; the ‘polyketide pathway’ (acetate pathway)(Reference Hakkinen12). They are defined chemically as substances that have an aromatic ring with a hydroxyl substituent, including esters and glycosides (functional derivatives). Most polyphenols possess two or three hydroxyl substituents, and are derived from a common dihydric or trihydric polyphenolic compound(Reference Dey and Harborne13). Thus, because of the diverse range of their chemical structures polyphenols are grouped into different classes, depending on the number of phenol rings they possess and the type and number of structural elements binding the phenolic rings together(Reference Manach, Scalbert, Morand, Remesy and Jimenez14). The classes include simple phenolic acids (e.g. ferulic and gallic acid; Fig. 1) and stilbenes (e.g. resveratrol; Fig. 1) and more complex chalcones (e.g. phloridzin; Fig. 1) and flavonoids, with the latter being split into seven subclasses that include flavonols (e.g. quercetin; Fig. 1), flavanols (e.g. epigallocatechin gallate; Fig. 1), flavones, flavanones, flavanonols, isoflavones and anthocyanins(Reference Hakkinen12).

Fig. 1. Chemical structures of selected polyphenols.
Polyphenols thought to provide health benefits include resveratrol, quercetin, ferulic acid, epicatechin gallate and epigallocatechin gallate, all of which belong to different classes of polyphenol (Fig. 1). Resveratrol is found in the skin of red grapes, wine, apples (Malus domestica), peanuts (Arachis hypogaea), blueberries (Vaccinium spp.) and cranberries (Vaccinium oxycoccus), and has been shown to provide antiangiogenic, anti-inflammatory and antiviral properties, as well as inducing cell death by apoptosis(Reference Signorelli and Ghidoni15). Quercetin is found in apples, tea, onions (Allium cepa), citrus fruit, broccoli (Brassica oleracea) and cherries (Prunus avium and Prunus cerasus), and has been shown to possess anti-inflammatory, antioxidant and antitumour properties(Reference Yance and Sagar16). Quercetin has also been suggested to act as a mast cell inhibitor, which may be useful in the treatment of chronic prostatitis(Reference Shoskes, Zeitlin, Shahed and Rajfer17). Ferulic acid, a hydroxycinnamic acid, is found in cereal grains such as rice, wheat and oats, and in coffee (Coffea canephora and Coffea arabica) beans, apples, artichoke (Cynara scolymus), peanuts, oranges (Citrus sinensis) and pineapples (Ananas comosus). Ferulic acid is reported to display pro-apoptopic effects in cancer cells, along with antitumour activity against breast cancer(Reference Lee18). It is also a powerful antioxidant, and may scavenge oxygen free radicals, provide protection of DNA against oxidative damage and increase intracellular cAMP and cGMP(Reference Sudheer, Muthukumaran, Kalpana, Srinivasan and Menon19). Epigallocatechin gallate is a major component of green tea and other teas, and also cocoa (Theobroma cacao) beans. Epigallocatechin gallate has been shown to interfere with numerous enzyme systems and can protect DNA from UV damage, and provide protection against breast cancer and other cancers(Reference Guo, Yang, Taylor and Sonenshein20). Epigallocatechin gallate may also act as a nutritional chemopreventive agent for the treatment of cancer, atherosclerosis and neurodegenerative diseases. It selectively induces apoptosis in human carcinoma cell lines(Reference Borska, Gebarowska, Wysocka, Drag-Zalesińska and Zabel21) and is thought to be involved in the suppression of vascular endothelial growth factor and angiogenesis(Reference Tosetti, Ferrari, De Flora and Albini22).
The mechanism of action for the beneficial effects of polyphenols on CVD was previously thought to be associated with their antioxidant activity(Reference Zern and Fernandez23). However, many polyphenols are metabolised in the body and these metabolites have much lower antioxidant capacities than their parent compounds, suggesting that antioxidant activity may not be their only mechanism of action(Reference Hakkinen12). Polyphenols present in red wine have been reported to reduce the incidence of CVD through protection against oxidative damage and reduced expression of transcription factors responsible for the activation, induction or control of genes involved in vasoconstriction.
Vascular endothelial cells
Vascular endothelial cells are flat elongated cells that contain a single nucleus and have a width of 1–20 μm and a depth of 0·2–20 μm(Reference Klabunde24). Endothelial cells are the only cell type known to contain Weibel palade bodies, in addition to the other cytoplasmic organelles common to most cells including the golgi and mitochondria(Reference Vickers25). Weibel palade bodies are the site of storage of Von Willebrand factor, a glycoprotein carrier of clotting factor VIII, which is required for the maintenance of haemostasis through the conversion of prothrombin to thrombin(Reference Fox26). The endothelium is composed of a single layer of endothelial cells joined together by one of three types of intercellular junctions (tight, gap and close junctions) determined by the distance between adjacent cells(Reference Vickers25). The endothelium has an internal coating of anionic biopolymers, the glycocalyx, and forms the inner lining of blood vessels(Reference Levick27). The functions of the endothelium include:
1. acting as a semi-permeable barrier for the exchange of macromolecules, fluid and electrolytes between intracellular and extracellular spaces(Reference Klabunde24).
2. site of synthesis of vasoactive substances such as endothelial NO synthase (eNOS), prostacyclin and endothelin-1 (ET-1), which lead to relaxation (eNOS and prostacyclin) or constriction (ET-1) of smooth muscle cells, thereby regulating blood pressure.
3. maintenance of blood coagulation and the inflammatory response, through the transport of clotting factor VIII and the secretion of endothelium-derived hyperpolarising factor, in response to alterations in blood flow-rate. It is also the site of production of adhesion molecules to capture and transport circulating leucocytes to affected tissues(Reference Sherwood28).
4. initiation site for formation of new blood vessels, a process termed ‘angiogenesis’, which is required for normal tissue growth and wound repair; uncontrolled angiogenesis is a major component of tumour growth, with prevention of angiogenesis being an important target for cancer drugs(Reference Klabunde24).
Vascular endothelial genes and cardiovascular health
The expression of specific genes in vascular endothelial cells plays an important role in the functions of the endothelium, and therefore cardiovascular health, through the effects on the formation of new blood vessels during angiogenesis or the regulation of blood pressure(Reference Ambra, Rimbach, de Pascual Teresa, Fuchs, Wenzel, Daniel and Virgili29). For example, vascular endothelial growth factor and pigment epithelium-derived factor are proteins produced by endothelial cells that stimulate and inhibit angiogenesis respectively. Vascular endothelial growth factor is an important signalling protein secreted by the endothelium for the induction of vascular permeability and the initiation of the processes of angiogenesis and vasculogenesis(Reference Oak, Chataigneau, Keravis, Chataigneau, Beretz, Andriantsitohaina, Stoclet, Chang and Schini-Kerth30).
Other genes expressed by vascular endothelial cells are involved in the synthesis of vasoactive substances, including eNOS and ET-1. The membrane-bound enzyme eNOS cleaves NO from l-arginine in response to stress exerted on the endothelium by blood flow and stimulants such as acetylcholine and Ca–calmodulin complexes. NO is a soluble lypophilic gas that is freely diffusible and produced continuously by the endothelium(Reference Fox26). It diffuses into vascular smooth muscle cells to react with the haem group in guanylyl cyclase, leading to the formation of cGMP and relaxation of vascular endothelial cells(Reference Sherwood28). Endothelial-derived NO also inhibits atherogenesis by reducing the expression of genes such as monocyte chemoattractant protein-1, P-selectin and the adhesion molecules CD11+CD18(Reference Wallerath, Li, Godtel-Ambrust, Schwarz and Forstermann31).
ET-1, the main isoform of the peptide endothelin, is synthesised as a preprohormone consisting of 212 amino acids (preproET-1), which is post-translationally cleaved to produce the active vasoconstrictor peptide ET-1 (consisting of twenty-one amino acids). ET-1 predominantly binds to endothelin A and endothelin B receptors on smooth muscle cells resulting in the vasoconstriction of smooth muscle. However, ET-1 can also bind to endothelin B receptors on neighbouring endothelial cells to cause vasodilation by increasing NO production(Reference Fagan, McMurthy and Rodman32).
Bioavailability and effects of polyphenols on gene expression
Polyphenolic compounds have been shown to have a wide range of effects on the expression of numerous genes within the human body, as determined by both in vitro and in vivo experiments(Reference Diebolt, Bucher and Andriantsitohaina33, Reference Noé, Peñuelas, Lamuela-Raventós, Permanyer, Ciudad and Izquierdo-Pulido34). Polyphenols such as resveratrol and quercetin, both found in abundant quantities in red wine, are thought to be responsible for the health-beneficial effects of red wine, also termed ‘the French paradox’(Reference Iijima, Yoshizumi and Ouchi35).
However, studies of the bioavailability and blood concentrations of polyphenols indicate that many of the published in vitro studies showing health benefits have used concentrations (1–100 μm) much higher than those achievable in the body. For example, plasma concentrations of resveratrol are approximately 20 nm and peak plasma levels of resveratrol and its metabolites are approximately 2 μm following consumption by six healthy patients of approximately 25 mg resveratrol in a suspension of simple syrup followed by 250 ml water(Reference Walle, Hsieh, DeLegge, Oatis and Walle36).
Hence, in vitro experiments at Nottingham using cultured human umbilical vein endothelial cells (HUVEC) have studied a more-physiologically-relevant concentration of 0·1 μm, followed by measurement of gene expression by microarray and quantitative RT–PCR. The microarray study used in-house printed 10K human slides to investigate the effects of ferulic acid, quercetin and resveratrol (all at 0·1 μm) on gene expression in HUVEC. The results show that overall these three polyphenolic compounds cause a significant (>2-fold) increase in the expression of 233 genes (2% of the genes; Fig. 2(A)) and a decrease in expression of 363 genes (approximately 4% of the genes; Fig. 2(B)). The majority of genes affected were found to respond to treatment with resveratrol, with fewer genes being affected only by quercetin or ferulic acid. Previous studies have also reported the ability of red wine polyphenols, particularly resveratrol, to alter gene expression in rat aortas(Reference Diebolt, Bucher and Andriantsitohaina33).
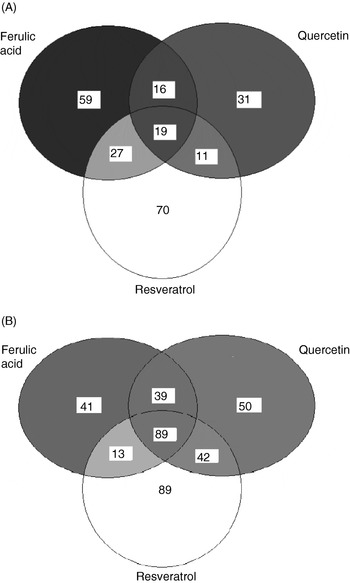
Fig. 2. In human umbilical vein endothelial cells treated with 0·1 μm-polyphenol (ferulic acid, quercetin or resveratrol) for 24 h at 37°C a total of 233 genes were up regulated (A) and 363 genes were down regulated (B).
Bioinformatic analyses of the genes up regulated or down regulated by treatment with the individual polyphenols have indicated that most of the genes are involved in transport, enzyme activity, cell signalling or transcription, accounting for 46% of the 233 genes significantly up regulated and 30% of the 363 genes significantly down regulated (SK Nicholson, GA Tucker and JB Brameld, unpublished results). Encouragingly, because preliminary studies have confirmed that polyphenol treatment at 0·1 μm does not lead to increased cell death, very few of the genes significantly up regulated or down regulated (2% and approximately 4% respectively) are involved in DNA repair or apoptosis. Microarray analysis has shown that resveratrol is the only polyphenol of the three tested that reduces ET-1 gene expression by ≥2-fold. As indicated earlier, ET-1 is a potent vasoconstrictor and is also involved in angiogenesis(Reference Fagan, McMurthy and Rodman32).
Further studies using more quantitative measures of gene expression (real-time PCR, Taqman) have examined the effects of physiological concentrations (0·1 μm) of polyphenols on the expression of three candidate genes involved in the regulation of blood pressure and endothelial function (eNOS, ET-1 and vascular endothelial growth factor). Resveratrol (0·1 μm) was found to significantly increase expression of eNOS in HUVEC following 24 h treatment. However, ferulic acid and quercetin at equimolar concentrations have no effect (Table 1; SK Nicholson, GA Tucker and JB Brameld, unpublished results). A previous study has also found that resveratol at concentrations of 1–33 μm increases the expression of eNOS in HUVEC-derived hy 926 cells, but equimolar concentrations of ferulic acid have no effect(Reference Wallerath, Li, Godtel-Ambrust, Schwarz and Forstermann31). Red wine extracts containing polyphenols have also been reported to increase mRNA and protein expression of eNOS(Reference Wallerath, Poleo, Li and Forstermann37).
Table 1. Effects of treatment with three individual polyphenols at a physiologically-relevant concentration (0·1 μm) on the expression of two candidate genes involved in the regulation of blood pressure in cultured human umbilical vein endothelial cells after treatment for 24 h relative to control
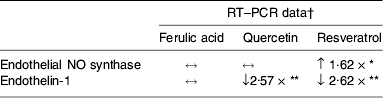
↔, No effect; ↓, decreased expression; ↑, increased expression.
* P<0·05, ** P<0·001 (Dunnett's test).
† All RT–PCR treatments were normalised to glyceraldehyde-3-phosphate dehydrogenase.
Other genes involved in blood pressure regulation and in cardiovascular health such as ET-1 are also affected by polyphenols and polyphenolic-containing foods. Transcription and synthesis of ET-1 are significantly decreased in bovine aorta endothelial cells treated with a red wine extract (6·25–25 μg/ml) containing polyphenols(Reference Corder, Douthwaite, Lees, Khan, Viseu Dos Santos, Wood and Carrier38). The present authors' studies have found that expression of ET-1 mRNA in HUVEC decreases by 2·62-fold and 2·57-fold following 24 h treatment with 0·1 μm resveratrol and quercetin respectively. However, ferulic acid at 0·1 μm has no effect on ET-1 mRNA expression (Table 1; SK Nicholson, GA Tucker and JB Brameld. unpublished results).
Several other polyphenolic compounds have also been tested for their effects on these three genes, including caffeic acid, genistein, phloretin and phloridzin, but none of these compounds were shown to have any significant effect (SK Nicholson, GA Tucker and JB Brameld, unpublished results).
Effects of polyphenols on basal and stress-induced gene expression
Oxidative stress is known to elevate the expression of certain genes in several cell types including endothelial cells (HUVEC) and smooth muscle cells(Reference Liu, Chen, Chan, Cheng and Cheng39). Different methods designed to put cells under oxidative stress or to elevate gene expression by the addition of a stimulating agent include cyclic strain methods(Reference Liu, Chen, Chan, Cheng and Cheng39), treatment with H2O2(Reference Liu, Chen, Chan, Cheng and Cheng39) and the addition of angiotensin II(Reference Chao, Juan, Liu, Yang, Yang, Cheng and Shyu40).
It has been shown that expression of eNOS and ET-1 mRNA in HUVEC increases in a dose-dependent manner following H2O2 (0–50 μm) treatment (SK Nicholson, GA Tucker and JB Brameld, unpublished results). Maximal responses relative to the control are 2·4- and 2·2-fold for eNOS and ET-1 mRNA respectively at 50 μm-H2O2. The addition of 0·1 μm-resveratrol increases eNOS to a similar extent (1·98-fold) but is not additive to the effects of the H2O2 treatment. On the other hand, resveratrol blocks the stimulatory effects of H2O2 on ET-1 mRNA expression (SK Nicholson, GA Tucker and JB Brameld, unpublished results). Previously published studies have found that resveratol (at 100 μm) blocks the stimulatory effect of H2O2 (25 μm) on ET-1 mRNA expression in HUVEC; while concentrations of 1–100 μm-resveratrol reduce ET-1 mRNA expression elevated following cyclic strain(Reference Liu, Chen, Chan, Cheng and Cheng39). Other studies have also shown that resveratrol (1–100 μm) has a similar effect in reducing elevated ET-1 expression in rat aorta smooth muscle cells induced by angiotensin II(Reference Chao, Juan, Liu, Yang, Yang, Cheng and Shyu40). Thus, the ability of resveratrol to reduce elevated gene expression is not specific to one cell type, but importantly has now been demonstrated at a more-physiologically-relevant concentration.
Conclusion
In the present author's studies resveratrol has been found to be the only polyphenol that significantly affects both eNOS and ET-1 expression in endothelial cells at a potentially physiological concentration (0·1 μm). The ability of resveratrol to increase the expression of the gene encoding eNOS (the enzyme synthesising the vasodilator NO) and reduce expression of the gene encoding the vasoconstrictor ET-1 would be expected to result in a potent vasodilation and thereby reduce blood pressure. Of the polyphenols tested to date, only quercetin has any similar effects to resveratrol, in that it appears to reduce ET-1 mRNA expression, but this finding needs to be confirmed. The reasons for the potency of resveratrol and the mechanisms involved in its action remain unclear. Importantly, resveratrol, at a physiological concentration, has been found to have beneficial effects on endothelial gene expression, and these effects are still apparent in cells under oxidative stress. Hence, resveratrol may be an important component of the diet in terms of reducing blood pressure and risk of CVD.





