Higher intake of whole grains has been linked to lower risk of several chronic diseases including obesity, type 2 diabetes and CVD( Reference McKeown, Hruby and Saltzman 1 – Reference Mellen, Walsh and Herrington 4 ). As with other dietary exposures, capturing self-reported whole-grain intake is subject to measurement error( Reference Keogh and White 5 ). For instance, study participants may inaccurately recall or report intake of whole-grain foods, and questionnaires may not be optimized to accurately capture frequently consumed foods containing whole grains. In addition, the inability of individuals to distinguish whole- from refined-grain foods, for example on the basis of colour, may result in unintentional misreporting of whole-grain intake( Reference Mozaffarian, Lee and Kennedy 6 ). The incorporation of independent biomarkers of exposure can be used to confirm diet–disease associations based solely on self-reported intakes( Reference Freedman, Kipnis and Schatzkin 7 ).
Plasma alkyresorcinols (AR), 1,3-dihydroxyl-5-n-alkylbenzene derivatives, have been used as a biomarker for whole-grain intake( Reference Kristensen, Toubro and Jensen 8 – Reference Magnusdottir, Landberg and Gunnarsdottir 10 ). In cereal grains, these compounds are found in the cuticula between the outer testa and inner pericarp layer( Reference Landberg, Kamal-Eldin and Salmenkallio-Marttila 11 ). AR are phenolic lipids abundant in whole wheat and rye grains as homologues with odd-numbered hydrocarbon side chains. They also occur to a lesser extent in barley( Reference Landberg, Kamal-Eldin and Andersson 12 ). During the refining of grains, the outer AR-containing bran layer is discarded; dramatically decreasing AR concentrations( Reference Ross and Kochhar 13 ). AR are rapidly absorbed and metabolized by man( Reference Ross, Kamal-Eldin and Lundin 14 , Reference Landberg, Aman and Friberg 15 ). The half-life of AR in plasma has been estimated at 5 h( Reference Landberg, Linko and Kamal-Eldin 16 ). Although they have a short half-life, single plasma AR measures are thought to reflect long-term whole-grain intake in populations with regular and frequent whole-grain wheat and rye intake( Reference Landberg, Aman and Hallmans 17 ).
To date, few observational studies have used plasma AR as independent biomarkers of whole-grain intake to confirm observed associations between intake and disease outcomes( Reference Olsen, Landberg and Aman 18 – Reference Ma, Ross and Shea 20 ). The main advantage of using an independent biomarker of food intake to determine diet–disease associations in observational studies is that they are independent of subjective errors in reporting dietary intake( Reference Kipnis, Subar and Midthune 21 ). In a sample of postmenopausal women in the Estrogen Replacement and Atherosclerosis trial, we previously reported that progression of coronary atherosclerosis over 3 years was moderately lower in those women who reported consuming higher intakes of cereal fibre and whole-grain foods( Reference Erkkila, Herrington and Mozaffarian 22 ). Using blood samples from this cohort, the objective of the present study was to determine whether a single determination of plasma AR concentrations was associated with progression of coronary atherosclerosis during the same time period. In addition, we examined whether there was an association between plasma AR concentrations and estimates of dietary intake derived from self-reported FFQ.
Experimental methods
Participants
The present study is an ancillary analysis using data from the Estrogen Replacement and Atherosclerosis trial, a randomized, double-blind, placebo-controlled trial conducted in postmenopausal women with coronary stenosis( Reference Herrington, Reboussin and Brosnihan 23 ). Briefly, 309 postmenopausal women were enrolled from six US sites (one in Alabama; one in Connecticut; and four in North Carolina). Women were eligible if they were postmenopausal, were not currently receiving hormone replacement therapy, and had one or more epicardial coronary stenoses ≥30 % of the luminal diameter. Women were randomly assigned to receive conjugated equine oestrogen (0·625 mg), conjugated equine oestrogen (0·625 mg) plus medroxyprogesterone acetate (2·5 mg) or placebo. Neither treatment had a significant effect on the progression of coronary atherosclerosis( Reference Herrington, Reboussin and Brosnihan 23 ).
At baseline (1995–1996), women completed questionnaires about their health status, medical history, physical activity and cardiovascular risk factors, as described elsewhere( Reference Herrington, Reboussin and Brosnihan 23 ). Of the 309 women enrolled in the trial, baseline blood samples for the determination of plasma AR were available for 215 participants. For analyses of plasma AR with progression of coronary artery disease (CAD), we excluded women missing the follow-up angiographic measure (n 33), for a sample size of 182 women. For correlations of plasma AR and dietary intake, we excluded women with missing or invalid baseline dietary data (n 25) for a sample size of 190 women. In order to maximize power, we did not restrict the data set for this analysis to women who also had data on CAD progression, as this would have reduced our sample size to 168 women.
The Institutional Review Board at Tufts Medical Center approved the present ancillary study, which utilized previously collected data from the Estrogen Replacement and Atherosclerosis trial.
Assessment of dietary intake
Habitual dietary and nutrient intake during the previous year was assessed prior to treatment at baseline with an interviewer-administered, 126-item FFQ as described elsewhere( Reference Rimm, Giovannucci and Stampfer 24 ). Dietary information was considered valid if reported energy intake was ≥2·51 MJ/d (600 kcal/d) and <6·74 MJ/d (4000 kcal/d). The FFQ included questions regarding the consumption of whole-grain foods such as cooked and cold breakfast cereals, dark bread, brown rice, popcorn and other grains (e.g. bulgur, kasha and couscous), as well as brand and type of cold breakfast cereal usually eaten. Breakfast cereal intake was subdivided into whole and refined grain as previously reported( Reference Jacobs, Meyer and Kushi 2 , Reference Liu, Stampfer and Hu 25 ). In addition, the contribution of total dietary fibre was calculated for food sources (e.g. fibre from cereal). Prior data using the FFQ indicated that daily intakes of cold and hot breakfast cereals estimated from FFQ and diet records were highly correlated (r≥0·70)( Reference Hu, Rimm and Smith-Warner 26 , Reference Feskanich, Rimm and Giovannucci 27 ). The correlation coefficient for other sources of whole grains ranged between 0·37 for dark bread and 0·79 for popcorn( Reference Feskanich, Rimm and Giovannucci 27 ).
Blood alkylresorcinol measures
The homologues of AR analysed included C17:0, C19:0, C21:0, C23:0 and C25:0; and the sum of these was used to determine the total AR concentrations. Baseline samples obtained prior to treatment were analysed by a GC–MS method in single ion monitoring mode as described previously( Reference Landberg, Man and Kamal-Eldin 28 ). Samples were analysed randomly in ten batches along with four quality control samples per batch. Intra- and inter-assay CV were <15 % for total AR.
Covariate assessment
Potential confounding factors included age (years, continuous), race (white v. other), BMI (kg/m2, continuous), oestrogen treatment arm (placebo v. oestrogen/oestrogen + progesterone), study site (four categories), follow-up time (years, continuous), current smoking status (yes v. no), physical activity (MET/week, continuous; where MET is metabolic-equivalent task-hours), educational level (three categories), lipid-lowering medication (yes v. no), coronary artery bypass grafting (CABG; yes v. no), percutaneous transluminal coronary angioplasty (PTCA; yes v. no) and systolic blood pressure (mmHg, continuous).
Outcome measures
As previously described( Reference Erkkila, Herrington and Mozaffarian 22 , Reference Herrington, Reboussin and Brosnihan 23 , Reference Imamura, Jacques and Herrington 29 ), quantitative coronary angiography was determined using standardized methods to measure the luminal diameters of up to ten proximal epicardial segments (mean: 9·6 segments per women) and the degree of stenosis as a percentage of the reference diameter at baseline and after 3·2 (sd 0·6) years in 248 postmenopausal women. Mean follow-up time in the present sample (n 182) was 3·1 (sd 0·7) years. Review and analyses of the paired films were performed by using a previously validated system of cine projectors (SME-3500; Sony, Park Ridge, NJ, USA) and software (QCAPlus; Sanders Data Systems, Palo Alto, CA, USA), as previously described( Reference Herrington, Reboussin and Brosnihan 23 ). Operators were blinded to the temporal sequence of the films.
Statistical analyses
Normality of continuous variables was checked; to reduce skewness, a natural logarithmic transformation was applied to plasma AR. Age-adjusted means or age-adjusted geometric means were calculated for lifestyle and dietary characteristics; dietary characteristics were also adjusted for total energy intake. The associations of plasma AR with changes in mean minimal coronary artery diameter (MCAD) and mean percentage diameter stenosis (%ST) were tested using mixed-model ANCOVA. The potential confounders controlled for in our basic model included age, oestrogen treatment arm, race, BMI, clinic, follow-up time and the baseline outcome measure. Additional models were further adjusted for current smoking, physical activity, education levels, lipid-lowering medication use, CABG, PTCA and systolic blood pressure. In a sensitivity analysis, we excluded segments of the coronary artery totally occluded at baseline (defined as a baseline MCAD<0·1 mm or baseline stenosis ≥99 %, n 31 segments in twenty-six individuals). To confirm that plasma AR are a biomarker of whole-grain intake in this population, partial Spearman rank-order correlation coefficients were calculated for plasma AR and whole- and refined-grain intakes, as well as various sources of fibre, adjusted for total energy intake. Further adjustment of correlations for age and BMI did not affect the correlations (data not shown).
Values are reported as means with their standard errors, unless otherwise noted. The significance level was set as a two-tailed P value <0·05. Statistical analyses were conducted using the statistical software package SAS version 9·3.
Results
The age-adjusted baseline participant characteristics are presented in Table 1. The mean age was 65·0 (se 0·5) years and mean BMI was 29·6 (se 0·4) kg/m2. In this sample, 85 % were Caucasian, 24 % were taking anti-hypertensive medication, 29 % were classified as having diabetes, and 33 % were taking lipid-lowering medication. Mean baseline whole-grain intake in the subset of women with dietary data (n 190) was 9·6 (se 0·6 servings) per week, in contrast with refined-grain intake of 16·0 (se 0·6) servings per week. Geometric mean plasma AR concentration was 18·0 nmol/l. The distribution of plasma AR was skewed, and the 25th, 50th and 75th percentiles of plasma AR were 10·8, 16·1 and 25·3 nmol/l, respectively.
Table 1 Baseline characteristicsFootnote * of the study participants: postmenopausal women (n 190), Estrogen Replacement and Atherosclerosis trial, USA
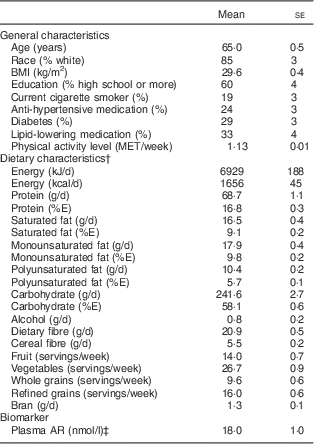
MET, metabolic-equivalent task-hours; %E, percentage of energy intake; AR, alkylresorcinols.
* Data are presented as means or percentages with their standard errors; all characteristics, except age, are age-adjusted.
† Foods and nutrient characteristics, except energy, are also adjusted for energy.
‡ Geometric mean with its standard error.
Estimated progression of coronary atherosclerosis per 50 % increment in total plasma AR concentrations is shown in Table 2. No significant association was observed for change in MCAD or %ST (P=0·61 and P=0·59, respectively) after adjustment for age, oestrogen treatment arm, race, BMI, clinic, follow-up time and the respective baseline outcome measure (model 1). Further adjustments for lifestyle factors (model 2) did not change these observations. In addition, further adjustment for lipid-lowering medication, CABG, PTCA or systolic blood pressure also did not change these observations. Using the median cut point for plasma AR (16·1 nmol/l), no significant difference in change in mean MCAD was observed in those with higher plasma AR (median 25·3 nmol/l) compared with lower plasma AR concentrations (median 10·8 nmol/l; −0·06 (se 0·02) v. −0·08 (se 0·02 mm), P trend=0·43) in any model (data not shown). Using the median cut point, plasma AR concentrations were not significantly associated with mean progression of %ST (2·31 (se 0·82) v. 3·63 (se 0·82) mm in high and low plasma AR categories, respectively, P trend=0·14). Sensitivity analyses excluding occluded segments did not substantively alter any results.
Table 2 Estimated progression of coronary atherosclerosis, as change in MCAD or %ST, per 50 % increment in total plasma AR among postmenopausal women (n 182), Estrogen Replacement and Atherosclerosis trial, USA

MCAD, minimum coronary artery diameter; %ST, percentage diameter stenosis; AR, alkylresorcinols.
* Adjusted for age, race, BMI, treatment group, follow-up years, clinic and the corresponding baseline measure.
† Adjusted as for model 1, plus current smoking, physical activity and education level.
A significant correlation was observed between plasma AR concentrations and weekly servings of whole grains (r=0·35, P<0·001) and cereal fibre (r=0·33, P<0·001). A weaker correlation was observed with total fibre (r=0·22, P=0·003), total bran (r=0·15, P=0·05) and legume fibre (r=0·15, P=0·04; Table 3). In contrast, there was no significant correlation between AR and weekly servings of refined grains, fruit fibre or vegetable fibre. Further adjustment for age and BMI did not substantively alter our results (data not shown).
Table 3 Partial Spearman correlations (r) and P value between energy-adjusted grain and fibre intakes and plasma AR among postmenopausal women (n 190), Estrogen Replacement and Atherosclerosis trial, USA
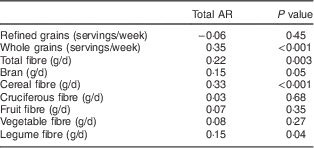
AR, alkylresorcinols.
Discussion
In nutritional epidemiology, a common application of plasma biomarker concentrations is to verify estimation of diet–disease risk associations( Reference Van Dam and Hunter 30 ). In this cohort of postmenopausal women with CAD, plasma AR concentrations, a biomarker of whole-grain intake, were not significantly associated with lesion progression during a 3-year period. These findings are in contrast to prior work that reported higher intakes of whole-grain foods and cereal fibre were moderately associated with less progression of coronary atherosclerosis in the same sample of women( Reference Erkkila, Herrington and Mozaffarian 22 ). Of note, the association with whole grains in the above-mentioned study( Reference Erkkila, Herrington and Mozaffarian 22 ) was slightly attenuated after adjustment for other aspects of diet, suggesting perhaps that dietary patterns associated with whole-grain intake, rather than whole-grain intake per se, were associated with less atherosclerotic progression. In contrast, whole-grain intake was inversely associated with several markers of atherosclerosis progression, independent of a healthy dietary pattern, in a multiethnic cohort of middle-aged adults( Reference Mellen, Liese and Tooze 31 ). The reason for these discrepancies is not clear. We cannot rule out the possibility in the current study that misclassification of exposure attenuated the association between plasma AR concentrations and the progression of coronary atherosclerosis.
Observational studies in free-living adults have only recently begun to include measures of plasma AR concentrations as independent biomarkers for assessing whole-grain exposure( Reference Olsen, Landberg and Aman 18 – Reference Ma, Ross and Shea 20 , Reference Knudsen, Kyro and Olsen 32 ). In a small sample of 241 older community-dwelling adults, plasma AR concentrations were inversely associated with BMI( Reference Ma, Ross and Shea 20 ), confirming a previously observed inverse relationship between whole-grain intake and BMI( Reference McKeown, Yoshida and Shea 33 ). In a nested case–control study( Reference Kyro, Olsen and Landberg 19 ), higher plasma total AR concentrations were negatively associated with incidences of distal colon cancer but not with overall colorectal cancer, proximal colon cancer or rectal cancer( Reference Knudsen, Kyro and Olsen 32 ). In this sample of postmenopausal women, the median plasma AR was low (16 nmol/l), reflective of the low self-reported daily whole-grain intake (average intake ≈1·4 serving/d), and the range was relatively narrow( Reference Erkkila, Herrington and Mozaffarian 22 ). The narrow range may have limited our ability to detect an association between AR and atherosclerotic progression, were there to be one. The data reflect whole-grain intakes in a subgroup of women and document that these intakes are low relative to both recommendations at the time the samples were collected and current recommendations. This median plasma AR concentration observed is similar to that reported for participants in the intervention arm of a metabolic study who were provided with a control diet devoid of whole grains, and that reported for older men and women living in the USA( Reference Ma, Ross and Shea 20 , Reference Ross, Bruce and Blondel-Lubrano 34 ). In comparison to European cohorts, these AR concentrations are low, confirming low intake of whole-grain wheat( Reference Magnusdottir, Landberg and Gunnarsdottir 10 , Reference Landberg, Kamal-Eldin and Andersson 12 , Reference Linko, Juntunen and Mykkanen 35 , Reference Aubertin-Leheudre, Koskela and Marjamaa 36 ).
Although the correlations between the single fasting plasma AR concentrations and self-reported intake of whole grains and cereal fibre were moderate (r=0·33–0·35), the weaker or non-significant associations between plasma AR and the other dietary fibres confirms the specificity of AR as a biomarker of whole grains. The magnitude of this correlation is fairly consistent with other studies that have correlated whole-grain intake estimated from FFQ with plasma AR concentrations( Reference Ross, Bourgeois and Macharia 9 , Reference Ma, Ross and Shea 20 , Reference Landberg, Kamal-Eldin and Aman 37 , Reference Kyro, Olsen and Bueno-de-Mesquita 38 ). Factors that can affect the relationship between whole-grain intake estimated with an FFQ and plasma AR concentrations may include intra-individual variation in AR concentrations inherent to a short-half life, irregular and low whole-grain intake, and inter-individual AR variation in AR metabolism as recently demonstrated by monitoring urinary AR metabolite excretion( Reference Landberg, Townsend and Neelakantan 39 ). Because of these potential limitations, multiple measures of fasting AR may be required to capture an individual’s representative whole-grain intake.
The current work has some limitations. Plasma AR is only reflective of wheat and rye; thus, intakes of popcorn and brown rice, which are also whole grains, particularly in the USA, are not reflected by this biomarker. Further, data on whole-grain intake at follow-up was not available for this analysis and diet may have changed over this time period. While we adjusted for interim procedures such as CABG and PTCA, we did not have information on and, thus, did not adjust for potential changes in usage of medications. In addition, it is possible that sample handling or duration of storage for the blood samples (approx. 20 years) may have influenced the AR concentrations. However, inherent to the molecular structure of AR and on the basis of an ongoing stability study (R Landberg, personal communication), this is unlikely. It is possible that given the short half-life of the intact AR (approx. 5 h), plasma AR concentrations are suitable as a biomarker only in populations with stable and frequent whole-wheat (or whole-rye) intake, where these biomarkers would be useful in distinguishing non-consumers from high consumers( Reference Ross, Bourgeois and Macharia 9 , Reference Landberg, Aman and Friberg 15 ).
In contrast to our a priori hypothesis, plasma AR concentrations were not significantly associated with atherosclerotic lesion progression over a 3-year period in postmenopausal women with CAD. Further studies using this biomarker should be undertaken in populations where there is a wider range of whole-grain intake and in conjunction with traditional dietary assessment methods that capture long-term intake.
Acknowledgements
Acknowledgements: The authors thank Georgia Saylor for data management and Karthik Balekudru Vishwanath for measurements of AR in plasma samples. Financial support: This work was supported by the National Heart, Lung, and Blood Institute (grant number U01 HL-45488). General Mills Bell Institute of Health and Nutrition provided funding to support the analysis of AR in plasma samples. Conflict of interest: N.M.M. is supported, in part, by a grant from the General Mills Bell Institute of Health and Nutrition. The other authors declare no conflicts of interest. Authorship: The authors’ responsibilities were as follows: A.H. conducted the statistical analyses; N.M.M. drafted the manuscript; D.M.H. contributed to the data collection and preparation of the data for analyses; A.H.L. contributed to interpretation of data and review of the manuscript; R.L. supervised the plasma AR analysis and interpretation of data. All authors designed the project, prepared the statistical strategies and discussion materials, and participated in editing the manuscript. Ethics of human subject participation: The Estrogen Replacement and Atherosclerosis study protocol was approved by the Institutional Review Board at participating sites, and the present study by the Institutional Review Board at Tufts Medical Center.






