The metabolic syndrome is a cluster of risk factors – central obesity, dyslipidaemia, hypertension and impaired glucose regulation – associated with increased risk of cardiometabolic diseases and early death( Reference Isomaa, Almgren and Tuomi 1 ). Prevalence of the metabolic syndrome is increasing worldwide( Reference Eckel, Grundy and Zimmet 2 ). To identify factors that can be used as targets for preventing development of the metabolic syndrome is therefore of great relevance. Whereas unhealthy dietary habits in adulthood, such as low intakes of fruits and vegetables( Reference Esmaillzadeh, Kimiagar and Mehrabi 3 ), dairy( Reference Pereira, Jacobs and Van Horn 4 ) and wholegrain( Reference Sahyoun, Jacques and Zhang 5 ) products and high intakes of soft drinks( Reference Dhingra, Sullivan and Jacques 6 ), meat and fried foods( Reference Lutsey, Steffen and Stevens 7 ), have been implicated as a risk factor for the metabolic syndrome, few studies have investigated associations between diet in adolescence and metabolic risk later in life.
Previous studies suggest that regular breakfast consumption improves several metabolic parameters and total diet quality( Reference Ruxton and Kirk 8 ). It has been suggested that eating in the morning is more satiating and may thus reduce total energy intake over the day( Reference de Castro 9 ). Skipping breakfast has been associated with a higher BMI and weight gain in several previous studies( Reference Song, Chun and Obayashi 10 – Reference Timlin, Pereira and Story 12 ). In a randomized crossover trial including ten lean women, total energy intake was higher during the two-week breakfast-skipping phase than in the two weeks with breakfast( Reference Farshchi, Taylor and Macdonald 13 ). The women also had higher fasting total cholesterol and LDL-cholesterol and higher postprandial insulin at the end of the breakfast-skipping phase. Regular breakfast consumption, compared with skipping breakfast or eating breakfast irregularly, has been shown to be prospectively associated with lower risk of CHD( Reference Cahill, Chiuve and Mekary 14 ), type 2 diabetes( Reference Mekary, Giovannucci and Willett 15 , Reference Mekary, Giovannucci and Cahill 16 ) and a spectrum of metabolic conditions in adults( Reference Odegaard, Jacobs and Steffen 17 ).
In the USA and Europe, about 10–30 % of children and adolescents regularly skip breakfast and the practice increases from childhood to adulthood( Reference Rampersaud, Pereira and Girard 18 ). In a recent cross-sectional survey conducted on 13–15-year-olds in northern Sweden, 15 % of the boys and 20 % of the girls reported skipping breakfast at least four days per week( Reference Vaezghasemi, Lindkvist and Ivarsson 19 ).
The definition of a healthy breakfast is not self-evident and different definitions of breakfast consumption have been used in previous studies( Reference Rampersaud, Pereira and Girard 18 ). In an experimental study by Pereira et al. comparing different breakfasts, it was found that a breakfast with high glycaemic index resulted in similar blood glucose levels after 5 h as a breakfast only consisting of water (that is, hypoglycaemia). This was suggested to be an effect of hyperinsulinaemia produced by the high-glycaemic-load condition( Reference Pereira, Erickson and McKee 20 ). Only drinking or eating something sweet for breakfast may thus, from a metabolic health perspective, be considered as poor breakfast habits. Evidently, skipping breakfast may also be considered as poor breakfast habits.
Previous results suggest that skipping breakfast as early as in adolescence may cause enduring health risk into adulthood. In a longitudinal study of Australian 9–15-year-olds followed up concerning cardiometabolic health over 20 years, participants who skipped breakfast in both childhood and adulthood had a larger waist circumference and higher fasting insulin, total cholesterol and LDL-cholesterol than those who reported eating breakfast at both times, after adjustments for age, sex, sociodemographic and lifestyle factors. However, the participation rate was low and the attrition rate was high (67·5 % of the invited children participated at baseline and of these about 33 % participated at follow-up)( Reference Smith, Gall and McNaughton 21 ). To our knowledge there are no other prospective studies on breakfast habits in adolescence and the metabolic syndrome in adulthood and there is a need for such longitudinal studies with low attrition rate.
The objective of our study was to analyse whether poor breakfast habits in adolescence predict the metabolic syndrome and its components in adulthood.
Methods
Selection of participants
Participants were drawn from the Northern Swedish Cohort, a 27-year prospective cohort study, described in detail by Hammarström and Janlert( Reference Hammarström and Janlert 22 ). All school-leavers of the 9th (final) grade (age 16 years) of the Swedish compulsory school in the municipality of Luleå in 1981 (n 1083) were invited to fill out an extensive questionnaire concerning lifestyle, health and socio-economic conditions. The cohort has been found to be representative of the corresponding age cohort of Sweden in demographic comparisons( Reference Hammarström and Janlert 22 ). Follow-up data collections were conducted in 1983 (age 18 years), 1986 (age 21 years), 1995 (age 30 years) and 2008 (age 43 years).
Health examinations were performed at ages 16, 21 and 43 years. At the follow-up at age 43 years a more extensive health examination was performed by trained medical personnel at the participants’ respective health care centre( Reference Gustafsson, Persson and Hammarström 23 ), making it possible to classify the prevalence of the metabolic syndrome. Participation in the follow-up was high; in 2008, 94 % of the baseline individuals still alive participated (n 1003)( Reference Gustafsson, Persson and Hammarström 23 ). The present report was based on data from the age 16 and age 43 years data collections. At age 16 years, questionnaires were completed in school, and at age 43 years, in conjunction with a class reunion. For those not participating in the class reunion, questionnaires were sent and returned by mail. Participants with information on breakfast habits at age 16 years and metabolic syndrome at age 43 years (n 889) were included in the present report.
To examine potential selection bias, those excluded because of incomplete measures on either breakfast habits (n 5) or the metabolic syndrome (n 107) were compared with those included in the study. There were no statistically significant differences in breakfast habits (proportion with poor breakfast habits) or covariates included in the multivariate analyses.
Poor breakfast habits at age 16 years
Breakfast habits were elicited in the questionnaire at age 16 years (‘What did you have for breakfast this morning?’). A number of food items divided into seven different food groups normally eaten for breakfast were suggested (drinks, milk products, egg/meat/fish, fruit/vegetables, porridge/cereals, dark bread, white/sweet bread) and participants were instructed to underline what they had eaten the same morning. The alternative ‘something else’ accounted for food items that could have been missed. Those reporting not eating anything at all for breakfast were defined as breakfast skippers (n 66), and those reporting only drinking something energy-containing (n 12) or only eating something sweet (n 10), such as a bun, cookie or biscuit, were treated as a separate category (‘drink or sweet for breakfast’; n 22). Due to low frequencies in these sub-categories, both representing poor breakfast habits, these categories were collapsed into a larger ‘poor breakfast habits’ category in the main analysis (n 88).
The metabolic syndrome and its components at age 43 years
Waist circumference was measured to the nearest 0·5 cm in indoor clothing. Blood samples were collected and assessed for TAG, HDL-cholesterol and glucose according to the laboratory routines at the Department of Clinical Chemistry, Umeå University. The CV at high/low concentration was 1·7 %/1·5 % for TAG, 2·8 %/2·8 % for HDL-cholesterol and 1·5 %/1·2 % for glucose. External quality control was managed by EQUALIS (External Quality Assurance in Laboratory medicine In Sweden). Blood pressure was measured with a standard sphygmomanometer on the participant's right arm in sitting position after resting for at least 5 min. The mean of two measures was used.
Presence of the metabolic syndrome and its components were operationalized similarly to our previous reports( Reference Gustafsson, Persson and Hammarström 23 , Reference Gustafsson, Persson and Hammarström 24 ). According to the International Diabetes Federation definition( Reference Alberti, Zimmet and Shaw 25 ), for a person to be defined as having the metabolic syndrome they must have central obesity (defined as waist circumference ≥80 cm for women and ≥94 cm for men), plus two or more of the following four components: (i) increased TAG (≥1·7 mmol/l) or specific treatment for that lipid abnormality; (ii) reduced HDL-cholesterol (<1·29 mmol/l for women/<1·03 mmol/l for men) or specific treatment for that lipid abnormality; (iii) increased blood pressure (systolic blood pressure ≥130 mmHg or diastolic blood pressure ≥85 mmHg) or treatment of hypertension; and (iv) increased fasting glucose (≥5·6 mmol/l) or diagnosed type 2 diabetes. Information on medication was self-reported. Current type 2 diabetes was based on self-reported diabetes at age 43 years, excluding those who also reported diabetes at age 30 years, who were regarded as having type 1 diabetes.
Both the metabolic syndrome and its components were used as binary outcomes (1 = presence, 0 = absence) in the analyses.
Assessment of confounders
Family history of diabetes was defined as a parent or sibling with diabetes, and the information was assessed from the questionnaire at age 16 years. Socio-economic status (SES) in adolescence was operationalized as in previous reports( Reference Gustafsson, Persson and Hammarström 23 , Reference Gustafsson and Hammarström 26 ). SES was defined by parents’ occupation when the participant was aged 16 years (both parents as manual workers counted as low SES). Height and weight from school health records were used to calculate BMI (kg/m2). Daily consumption of pure alcohol was estimated from a validated battery of questions (frequency and amount of medium-strength beer, strong beer, wine and spirits). Participants above the 80th percentile (sex-specific) of alcohol consumption were classified as high consumers. Smoking habits were classified as current smoker or not (including ex-smokers). A reliability test–retest of the alcohol and tobacco questions was conducted in 1981, within a 2-month period, showing that similar answers were given by the same pupils to the same questions( Reference Hammarström 27 – Reference Hammarström and Janlert 29 ). Those who reported participation in physical activity more often than once weekly were classified as physically active. Daily consumption of sweets and pastries was defined as high consumption of these food groups.
Statistics
Participants who were classified as having poor breakfast habits were compared with breakfast eaters regarding variables at baseline (age 16 years) and components of the metabolic syndrome at age 43 years, using the χ 2 test or the non-parametric Mann–Whitney U test. Crude and adjusted odds ratios for metabolic syndrome at age 43 years were calculated for those with poor breakfast habits compared with breakfast eaters at age 16 years, using binary logistic regression. Additional analyses were conducted on the sub-categories ‘skipping breakfast’ (n 66) and ‘drink or sweet for breakfast’ (n 22). The crude odds for the metabolic syndrome at age 43 years for those skipping breakfast compared with eating breakfast at age 16 years was OR = 2·18 (95 % CI 1·30, 3·66) and the corresponding odds for those only drinking or eating something sweet for breakfast was OR = 2·47 (95 % CI 1·05, 5·81). The sub-categories were too small to draw conclusions, but the similar point estimates justifies combining these sub-categories to the category ‘poor breakfast habits’.
In the multivariate analysis on the association between poor breakfast at age 16 years and the metabolic syndrome at age 43 years we added sex as covariate in model 1. In model 2 we added family history of diabetes, alcohol consumption, smoking status and physical activity at age 16 years beyond covariates in model 1. In model 3 we added BMI and parents’ SES at age 16 years beyond covariates in model 2. Sex was added because the prevalence of the metabolic syndrome is higher in men than women. The covariates added in model 2 were chosen because they were differing significantly (P < 0·05) between breakfast groups and they also tended to relate to the metabolic syndrome in univariate analyses (P < 0·25). The covariates added in model 3 were chosen to (as far as possible) rule out any effect of pre-existing metabolic or social risk factors. The same models were used in logistic regression analysis on separate components of the metabolic syndrome (Table 3). Our main aim was to analyse if poor breakfast habits at age 16 years predicted the metabolic syndrome and its components at age 43 years. In our predictor concept, a predictor precedes the outcome. A predictor is a variable which foretells the future risk in a population. It is important to recognize that a predictor provides no information about causality.
As an attempt to find out more about possible mediating mechanisms in the association between poor breakfast in adolescence and future risk of the metabolic syndrome, an additional multivariate analysis constituting model 3 plus lifestyle at age 43 years (consumption of fruits and vegetables and physical activity) was performed.
The statistical computations were carried out using the statistical software package IBM SPSS Statistics for Windows version 20.
Missing data
There were a few missing data on covariates at age 16 years; at most six individuals lacked information on alcohol intake (0·7 %). Missing data were not replaced. In the most extensive multivariate model (model 3), seventeen individuals dropped out of the analysis because of missing data on any of the covariates. We compared prevalence of poor breakfast habits and the metabolic syndrome in individuals with any missing data on covariates and individuals with complete data using a χ 2 test. Individuals with any missing data on covariates did not differ significantly from those with complete data regarding breakfast habits at age 16 years (P = 0·232) or prevalence of the metabolic syndrome at age 43 years (P = 0·787).
Ethical approval and informed consent
The study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects were approved by the Regional Ethical Review Board in Umeå. All participants gave their informed consent.
Results
The prevalence of the metabolic syndrome at age 43 years among the 889 participants was 27·0 % (34·1 % in men and 19·3 % in women), according to the International Diabetes Federation definition. Of the 889 participants, eighty-eight individuals (9·9 %) were classified with poor breakfast habits. Of those classified with poor breakfast habits, sixty-six individuals were breakfast skippers and twenty-two individuals reported only drinking or eating something sweet for breakfast. As presented in Table 1, individuals who reported poor breakfast habits at age 16 years had a higher prevalence of family history of diabetes, higher alcohol consumption, a higher prevalence of smoking and a lower physical activity level compared with breakfast eaters. For components of the metabolic syndrome at age 43 years, those with poor breakfast habits at age 16 years had higher prevalence of central obesity, high TAG levels, high blood pressure and high fasting glucose (Table 1).
Table 1 Descriptive statistics for 889 participants from the Northern Swedish Cohort, according to breakfast habits
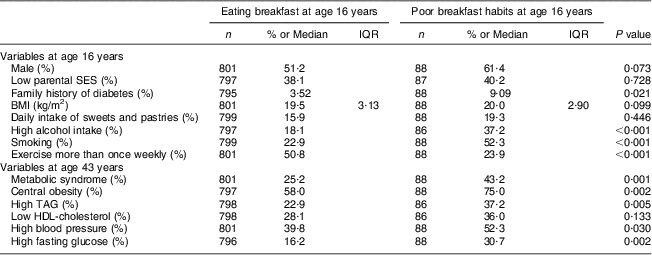
IQR, interquartile range; SES, socio-economic status.
Poor breakfast habits at age 16 years were associated with higher odds of the metabolic syndrome at age 43 years in the unadjusted model (OR = 2·25; 95 % CI 1·43, 3·54; see Table 2). The association was only marginally attenuated by the addition of sex (model 1) and moderately attenuated by the addition of selected covariates (model 2: family history of diabetes, alcohol consumption, smoking status and physical activity at age 16 years). After further adjustments for SES and BMI at age 16 years, there was an insubstantial change of odds ratio, with the odds for poor breakfast habits remaining significant (model 3: OR = 1·68; 95 % CI 1·01, 2·78; Table 2). Covariates significantly related to the metabolic syndrome in model 3 were sex, family history of diabetes, physical activity and BMI at age 16 years (Table 2).
Table 2 Logistic regression analyses of associations between poor breakfast habits at age 16 years and the metabolic syndrome at age 43 years, for 889 participants from the Northern Swedish Cohort
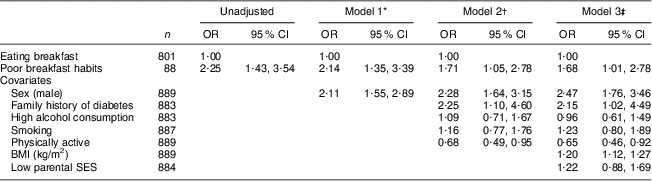
SES, socio-economic status.
*Adjusted for sex.
†Model 1 plus family history of diabetes, high alcohol consumption, smoking and physical activity (variables associated with the metabolic syndrome (P < 0·25) and significantly different between breakfast groups) at age 16 years.
‡Model 2 plus BMI and parents’ SES.
In separate analyses of components of the metabolic syndrome (Table 3), central obesity and high fasting glucose at age 43 years were associated with poor breakfast habits at age 16 years in all models. For TAG at age 43 years, the association with poor breakfast habits at age 16 years was significant in the crude model and model 1 but not in subsequent models. The association between poor breakfast habits at age 16 years and high blood pressure at age 43 years was significant only in the crude model, whereas the association with HDL-cholesterol at age 43 years was not significant in any of the models.
Table 3 Logistic regression analyses of associations between breakfast skipping at age 16 years and components of the metabolic syndrome at age 43 years, for 889 participants from the Northern Swedish Cohort
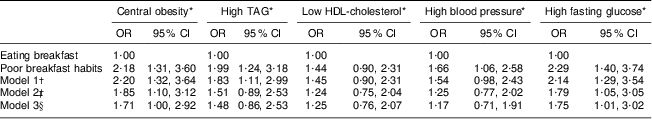
*Definition according to the International Diabetes Federation Guidelines( Reference Alberti, Zimmet and Shaw 25 ).
†Adjusted for sex.
‡Model 1 plus family history of diabetes, high alcohol consumption, smoking and physical activity.
§Model 2 plus BMI and parents’ socio-economic status.
Adding consumption of fruits and vegetables as well as physical activity at age 43 years to the multivariate model attenuated the association between poor breakfast habits at age 16 years and risk of the metabolic syndrome at age 43 years, and it was no longer statistically significant (OR = 1·53; 95 % CI 0·91, 2·57).
Discussion
In the present study with high participation rate and low loss to follow-up, poor breakfast habits in adolescence predicted the metabolic syndrome at age 43 years, also after adjustment for potential confounders (such as lifestyle variables and socio-economic conditions) in youth. Of the components of the metabolic syndrome, central obesity and high fasting glucose remained associated with poor breakfast habits also after multivariate adjustments.
Eating breakfast has been associated with an overall better diet quality and healthy lifestyle in several studies( Reference Ruxton and Kirk 8 , Reference Song, Chun and Obayashi 10 , Reference van der Heijden, Hu and Rimm 11 ). It may therefore be argued that poor breakfast habits predict cardiometabolic risk because it is part of an unhealthy lifestyle. Our finding that those with poor breakfast habits exercise less, drink more alcohol and smoke more than breakfast eaters supports the view that poor breakfast habits is part of an unhealthy lifestyle (Table 1). The finding that poor breakfast habits at age 16 years predicted the metabolic syndrome at age 43 years remained statistically significant after multivariate adjustments with lifestyle variables at age 16 years (smoking, high alcohol consumption, physical activity). This indicates that the association between poor breakfast habits and future metabolic syndrome does not just reflect a covariance between poor breakfast habits and other unhealthy lifestyle habits in adolescence, but that poor breakfast habits have an independent ability to predict metabolic risk. Furthermore, the observation that the association was independent of BMI and SES in adolescence suggests that the association is not simply explained by pre-existing metabolic and social risk factors, which we have previously shown to be related to adult metabolic syndrome( Reference Gustafsson, Persson and Hammarström 23 ).
It has been previously found that eating breakfast is associated with a better total diet quality( Reference Ruxton and Kirk 8 ) and also with physical activity( Reference Schembre, Wen and Davis 30 ). When we added consumption of fruits and vegetables and physical activity at age 43 years to our multivariate model, the association between poor breakfast habits at age 16 years and the metabolic syndrome at age 43 years was somewhat attenuated and no longer statistically significant. This may be interpreted as if the association, at least to some extent, is mediated by future healthy diet and/or high physical activity level. However, because breakfast habits are often established at an early age( Reference Merten, Williams and Shriver 31 ), the association may also be mediated by breakfast habits at age 43 years. We were unable to examine to what degree the reported association relates to stable breakfast habits over the life course, because we did not have information about breakfast habits at age 43 years. The contribution of the present paper is the observation that early-life breakfast habits predict the metabolic syndrome decades later. Further research is needed in order to get a deeper understanding of how adolescent breakfast habits are related to adult metabolic syndrome, including analyses of possible mediating mechanisms.
In our study, central obesity and high fasting glucose at age 43 years were the components of the metabolic syndrome which were most strongly predicted by poor breakfast habits in adolescence. HDL-cholesterol, TAG and high blood pressure were not significantly associated with poor breakfast habits after multivariate adjustments. The stronger associations found for central obesity and high fasting glucose are supported by experimental studies. In the short-term randomized crossover trial by Farshchi et al.( Reference Farshchi, Taylor and Macdonald 13 ), breakfast skipping was associated with high fasting insulin. High fasting insulin may contribute to central obesity( Reference Labayen, Ruiz and Ortega 32 ). An experimental study by Pereira et al. suggests that breakfast skipping, as well as breakfast quality, may have important effects on factors related to appetite control and glycaemic control( Reference Pereira, Erickson and McKee 20 ). Also, findings in recent observational studies of an association between breakfast omitting and increased risk of type 2 diabetes support an effect on weight and insulin resistance( Reference Mekary, Giovannucci and Willett 15 , Reference Mekary, Giovannucci and Cahill 16 ). These previous findings are in line with our finding that central obesity and fasting glucose are the components of the metabolic syndrome with the strongest associations to poor breakfast habits at age 16 years.
In previous research on breakfast habits and cardiometabolic outcomes, skipping breakfast has been the most common way to define the breakfast exposure. However, even the definition of skipping breakfast has not been completely consistent, as some studies included those only drinking as breakfast skippers( Reference Smith, Gall and McNaughton 21 ), while others did not( Reference Deshmukh-Taskar, Nicklas and Radcliffe 33 , Reference Min, Noh and Kang 34 ). When those skipping breakfast and those only drinking or eating something sweet for breakfast at age 16 years were considered in separate categories in our study, the crude odds ratios for the metabolic syndrome at age 43 years were similar for the sub-categories. This justifies combining these into the category ‘poor breakfast habits’. Our study does not have power to allow for conclusions concerning skipping breakfast (with the definition of not eating or drinking anything energy-containing at all for breakfast) and the metabolic syndrome. Skipping breakfast or only drinking or eating something sweet for breakfast are different variants of absence of healthy breakfast habits. As stated by Eilat-Adar et al.( Reference Eilat-Adar, Koren-Morag and Siman-Tov 35 ), there is a need for an exact definition of what a healthy breakfast is. Several previous studies have found an especially beneficial effect of ready-to-eat cereals on cardiometabolic risk factors and total nutrient intake( Reference Deshmukh-Taskar, Nicklas and Radcliffe 33 , Reference Affenito, Thompson and Dorazio 36 ). In our study we did not aim to investigate the influence of breakfast quality, except separating those with distinctively poor breakfast habits. Larger studies are warranted to analyse the relationships between breakfast quality and the metabolic syndrome further.
Method discussion
The prevalence of the metabolic syndrome in the present study (34·1 % in men and 19·3 % in women) was higher compared with a population-based cohort in another part of Sweden( Reference Welin, Adlerberth and Caidahl 37 ). Therefore, the generalizability of our findings to populations with lower prevalence of the metabolic syndrome is unclear.
A limitation of the study is that the definition of breakfast habits at age 16 years was based on one single-day assessment, which would be expected to decrease the measurement precision. However, imprecision of the exposure would lead to dilution and underestimation, rather than overestimation( Reference Carroll 38 ), of the association between poor breakfast habits and the metabolic syndrome.
Some further limitations of the study need to be discussed. Although we were able to adjust for physical activity, alcohol consumption and smoking at age 16 years, we could not adjust thoroughly for adolescent dietary habits. Consumption of sweets and pastries at age 16 years did not differ between the breakfast groups (Table 1), but we did not have information on beneficial dietary habits such as fruit and vegetable consumption at age 16 years. Eating a healthy breakfast may aid in sticking to good dietary habits during the rest of the day, by balancing postprandial blood sugar and keeping a balanced energy intake( Reference de Castro 9 , Reference Farshchi, Taylor and Macdonald 13 ). As previously mentioned, we did not have information on breakfast habits in adulthood. However, even if the association between breakfast eating in adolescence and lower prevalence of the metabolic syndrome in adulthood were completely due to strong correlation between breakfast habits in adolescence and adulthood and/or to assistance in sticking to healthy dietary habits during the rest of the day, these factors should rather be considered as mediators than confounders.
The main strengths of the current study were the prospective design with long follow-up. The high participation rate and low attrition rate resulted in a very good representativeness. The variables were derived from well-known and validated questionnaires( Reference Hammarström and Janlert 22 , Reference Hibell and Jonsson 39 , Reference Kannas and Hietaharju 40 ). In addition, a reliability test of the alcohol and tobacco questions was made in 1981, which showed good 2-month test–retest reliability( Reference Hammarström 27 – Reference Hammarström and Janlert 29 ).
Considering the amount of people skipping breakfast in the USA and Europe( Reference Rampersaud, Pereira and Girard 18 ), it is crucial from a public health point of view to identify predictors and in future research also analyse possible causal mechanisms behind adolescent breakfast habits and future metabolic risk. Recent data from northern Sweden demonstrated that the prevalence of skipping breakfast in adolescence has increased since the early 1980s( Reference Vaezghasemi, Lindkvist and Ivarsson 19 ). Offering breakfast in schools, in so-called School Breakfast Programmes, has been shown to have beneficial effects on academic performance( Reference Hoyland, Dye and Lawton 41 ), but the implications for metabolic health are unclear.
Conclusions
Poor breakfast habits at age 16 years predicted the metabolic syndrome at age 43 years, independently of early confounders (lifestyle, BMI and SES). Of the metabolic syndrome components, poor breakfast habits at age 16 years predicted central obesity and high fasting glucose at age 43 years. Based on our findings, we suggest that School Breakfast Programmes should be evaluated regarding metabolic health consequences. Further research is needed to understand how the relationship between early breakfast habits and adult metabolic syndrome is mediated.
Acknowledgements
Sources of funding: The work was supported through a regional agreement between Umeå University and Västerbotten County Council on cooperation in the field of Medicine, Odontology and Health (ALF). The funding source (ALF) had no role in the design, analysis or writing of this article. Conflicts of interest: The authors have no conflicts of interest to report. Authors’ contributions: A.H. designed the research; A.H. conducted the research (data collection); M.W. analysed the data; M.W., P.E.G., P.W. and A.H. interpreted the data; M.W. wrote the paper; P.E.G., P.W. and A.H. critically revised the paper for important intellectual content; M.W. has primary responsibility for the final content. All authors read and approved the final manuscript.





