Schizophrenia
Schizophrenia is a chronic psychiatric syndrome characterised by disturbances of mental processes including perception, cognition, emotion, thought and behaviour. It is a leading cause of disability, affecting 1 in 222 adults worldwide, 1 with high economic costs. Reference Chong, Teoh, Wu, Kotirum, Chiou and Chaiyakunapruk2,Reference Solmi, Seitidis, Mavridis, Correll, Dragioti and Guimond3 In addition to substantial adverse impacts on social functioning and quality of life, schizophrenia is associated with increased all-cause mortality, Reference Correll, Solmi, Croatto, Schneider, Rohani-Montez and Fairley4,Reference Laursen, Nordentoft and Mortensen5 largely owing to physical comorbidity. The course of schizophrenia is typically characterised by recurrent relapses. Within the five years following recovery from first-episode schizophrenia, the risk of relapse is 81.9%. Reference Robinson, Woerner, Alvir, Bilder, Goldman and Geisler6 The cumulative effects of relapses act to exacerbate symptoms and lead to functional decline and treatment resistance. Reference Almond, Knapp, Francois, Toumi and Brugha7,Reference Takeuchi, Siu, Remington, Fervaha, Zipursky and Foussias8 which emphasises the importance of identifying underlying reasons for relapse to prevent longer-term progression.
Treatment non-adherence
The most consistently documented predictor of relapse in schizophrenia is treatment non-adherence. Reference Guo, Lv, Liu, Kong, Qu and Yue9 Treatment non-adherence is particularly prevalent among patients with schizophrenia compared with patients with other health conditions; over half of patients with schizophrenia are non-adherent to their psychotropic medications. Reference Semahegn, Torpey, Manu, Assefa, Tesfaye and Ankomah10 Treatment non-adherence in schizophrenia results from a combination of illness-related, medication-related and personal factors. Illness-related factors such as lack of insight, negative symptoms, poor cognition and comorbidities result in decreased motivation and perceived need for treatment. Reference Guo, Lv, Liu, Kong, Qu and Yue9,Reference Boyer, Cermolacce, Dassa, Fernandez, Boucekine and Richieri11,Reference Rettenbacher, Hofer, Eder, Hummer, Kemmler and Weiss12 Moreover, the significant side-effect burden and perceived effectiveness of medication further contribute to non-adherence behaviour. Reference Guo, Lv, Liu, Kong, Qu and Yue9,Reference Rivelli, Fitzpatrick, Nelson, Laubmeier, Zeni and Mylavarapu13 A variety of sociodemographic factors including age and ethnicity have also been associated with an increased risk of treatment non-adherence. Reference Rivelli, Fitzpatrick, Nelson, Laubmeier, Zeni and Mylavarapu13 Although non-adherence is a well-recognised risk factor for relapse and hospitalisation in schizophrenia, Reference Abdullah-Koolmees, Nawzad, Egberts, Vuyk, Gardarsdottir and Heerdink14,Reference Higashi, Medic, Littlewood, Diez, Granström and De Hert15 the impact on broader clinical outcomes remains less clear. Existing retrospective studies have the benefit of being larger in scale, but tend to be limited in their range of outcome measures. Reference Eaddy, Grogg and Locklear16–Reference Valenstein, Copeland, Blow, McCarthy, Zeber and Gillon18 Prospective observational studies have additionally demonstrated that treatment non-adherence is associated with violence, Reference Ascher-Svanum, Faries, Zhu, Ernst, Swartz and Swanson19,Reference Morken, Widen and Grawe20 poorer life satisfaction and mental functioning, increased risk of substance abuse Reference Ascher-Svanum, Faries, Zhu, Ernst, Swartz and Swanson19 and suicide attempts. Reference Novick, Haro, Suarez, Perez, Dittmann and Haddad21 Although one small prospective study (N = 50) reported that non-adherence was associated with persistent psychotic symptoms, Reference Morken, Widen and Grawe20 there is limited evidence of how treatment non-adherence impacts specific symptom domains and antipsychotic prescription.
In the present study, using a large, granular database derived from mental health records, we are uniquely able to carry out analyses which are both large-scale and involve a broad range of outcome measures. We investigate the relationships between clinician-reported treatment non-adherence and outcomes including symptom profiles, antipsychotic prescription, hospitalisation and crisis care episodes.
Method
The South London and Maudsley (SLaM) National Health Service (NHS) Trust is one of the largest providers of secondary mental health services in Europe, Reference Bendayan, Kraljevic, Shaari, Das-Munshi, Leipold and Chaturvedi22 covering four inner-city London boroughs (Southwark, Croydon, Lambeth and Lewisham) and a population of around 1.3 m residents. Electronic health records from SLaM NHS Trust have been deidentified to form a large case register which can be accessed via the Clinical Record Interactive Search (CRIS) tool. Reference Perera, Broadbent, Callard, Chang, Downs and Dutta23,Reference Stewart, Soremekun, Perera, Broadbent, Callard and Denis24 This platform was used to collate the data-set for our study.
Our sample included adult patients with a recorded schizophrenia (F20) diagnosis, according to the International Classification of Diseases (ICD-10), who were referred to SLaM services from January 2007 until December 2018. We included patients with a first active referral to SLaM services of minimum 3 months duration, measured between the first attended event and discharge. Baseline variables were calculated using all available data up to and including the first attended event. The follow-up period included the 12 months following the first attended event.
Clinician-recorded non-adherence was ascertained via a natural language processing (NLP) tool. Non-adherence was defined for this purpose as any recorded significant deviation from an agreed treatment recommendation. To minimise ambiguity in the model, statements reflecting partial or fluctuating adherence were labelled as ‘unknown’ due to the broad nature of these terms. To develop the application, a total of 2600 documents were extracted from the CRIS database using the following search terms: ‘Adheren*’, ‘complian*’, ‘non-adheren*’, ‘nonadheren*’, ‘noncomplian*’. The excerpts were annotated as either indicating adherence, non-adherence or unknown by two independent assessors and were subsequently used to train and test a machine learning NLP model. Interrater reliability between the two assessors was tested with a sample of 46 documents, and the agreement rate was found to be 88%. Having optimised performance within the development environment, on an independently extracted test sample of 100 documents, the tool performed with precision (specificity/accuracy) of 83% and recall (sensitivity/coverage) of 86%. We applied this tool to the full CRIS data platform and used the output to ascertain recorded treatment non-adherence during the 12-month follow-up period. We thereby extracted and compared data from two groups on the basis of recorded adherence status: one group with recorded treatment non-adherence and the other being the remainder (i.e. patients with recorded treatment adherence or an unknown adherence status).
Descriptive statistics were extracted, including age at first attended event, gender, ethnicity, neighbourhood deprivation, comorbidities and baseline symptom profile. These were used both to characterise the sample and as control variables for statistical analyses. Relative deprivation was represented by the Index of Multiple Deprivation (IMD) score, a composite measure of 39 indicators of deprivation produced by England’s Ministry of Housing, Communities and Local Government (MHCLG), derived from national census returns and collated at the level of Lower Super Output Area (LSOA), an administrative unit containing around 1500 residents. Comorbidity was extracted using an NLP pipeline, MedCAT, to assess the recording of up to 21 common physical health conditions Reference Bendayan, Kraljevic, Shaari, Das-Munshi, Leipold and Chaturvedi22 which were summed and generated as a score. Baseline symptoms were extracted using multiple NLP tools which ascertain over 60 individual symptoms; the presence or not of all of these were then summed within the following subscales: positive schizophreniform (0 to 16), negative schizophreniform (0 to 12), depressive (0 to 21), manic (0 to 8) and disorganised (0 to 8). Reference Jackson, Patel, Jayatilleke, Kolliakou, Ball and Gorrell25,26
For the 12-month follow-up period, we extracted the number and duration of in-patient admissions and crisis care episodes in the SLaM NHS Trust. We also extracted symptom scores during this 12-month period and subtracted the baseline symptom scores to calculate change relative to baseline. Similarly, we also calculated the change in the number of recorded comorbidities. Finally, recorded antipsychotic use was ascertained via a widely used NLP algorithm, and we used this to derive the number of different antipsychotics recorded over the follow-up period. Reference Kadra, Stewart, Shetty, Jackson, Greenwood and Roberts27
To compare descriptive statistics between the non-adherent group and the remainder, t-tests were used for age, IMD score, comorbidity and symptom scales, and χ 2 tests for gender and ethnicity. Multilinear regression analyses of the change in symptom score relative to baseline as a function of recorded non-adherence were carried out, controlling for age, gender, ethnicity, relative deprivation, comorbidities and baseline symptom score. To facilitate comparison across different symptom domains, variables were then standardised through conversion to z-scores and further multilinear regression analyses carried out to produce standardised beta coefficients. Multilinear regression analyses were also carried out modelling the change in the number of comorbidities relative to baseline, adjusting for control variables. Poisson regression analyses were used to assess the frequency and duration of in-patient admissions and crisis care episodes and number of antipsychotics prescribed as a function of recorded treatment non-adherence and control variables. To further elucidate the relationship between recorded treatment non-adherence and service utilisation, logistic regression analyses were performed to assess the incidence of in-patient and crisis care team admission, coded as binary outcomes, as a function of recorded treatment non-adherence and control variables (see Supplementary Table 1). All data management and statistical analyses were performed using Stata 18 for Windows (StataCorp LLC, College Station, Texas, USA; https://www.stata.com/stata18/).
Results
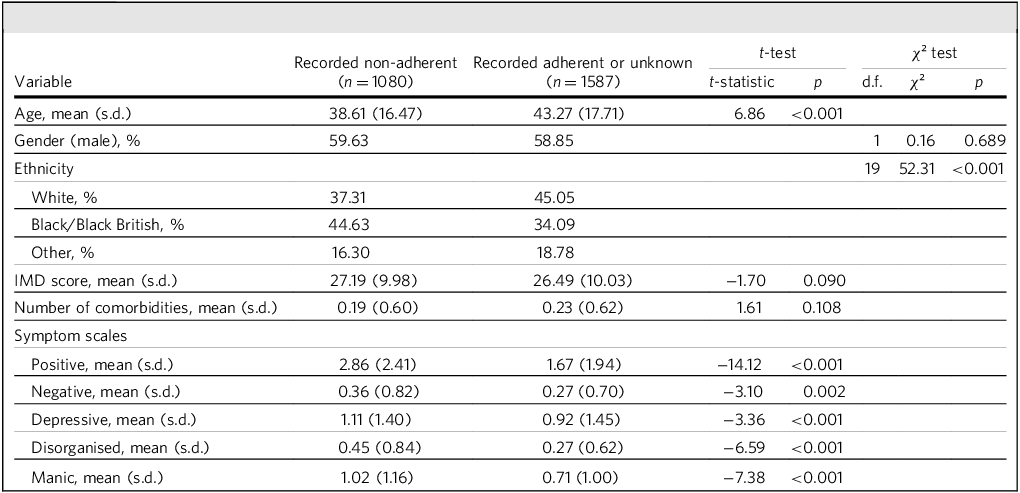
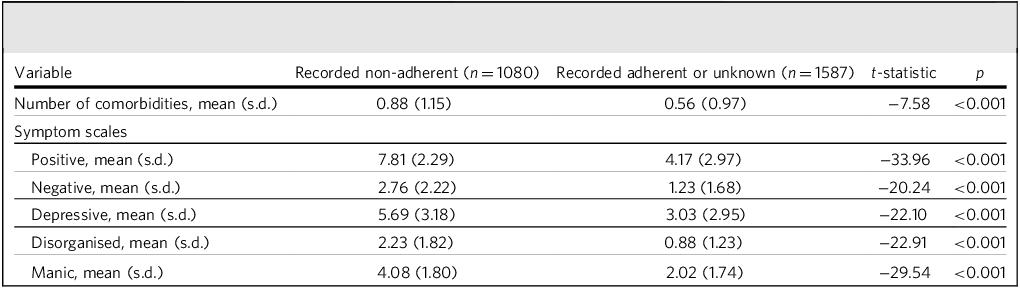
A total of 2667 patients were included. The mean (s.d.) age for the overall sample was 41.4 (17.4) years and 59.17% were male. During the 12-month follow-up period, 1080 (40.5%) were reported to be non-adherent to psychiatric treatment. Baseline demographic and clinical characteristics by adherence status are presented in Table 1. While there were no significant differences in gender, IMD score and comorbidities between groups, patients with recorded non-adherence were significantly younger and had a significantly different distribution of ethnicities compared with the remainder. The non-adherent cohort also had higher symptom scores in all domains at baseline. Table 2 presents comorbidity and symptom scale scores by adherence status during the 12-month follow-up period. Those with recorded non-adherence again had higher symptom scores in all domains, and also more comorbidities.
Baseline characteristics and symptom severity in recorded non-adherent cohort versus the remainder

IMD, Index of Multiple Deprivation.
Comorbidities and symptom severity during 12-month period following F20 diagnosis in recoded non-adherent cohort versus the remainder

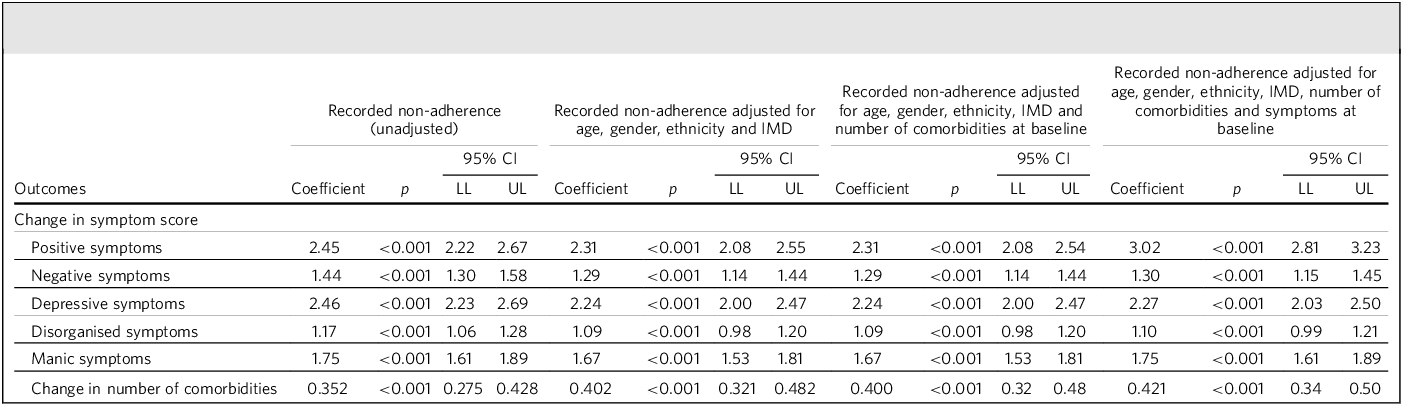
Table 3 presents the results of multilinear regression analyses, comparing the changes in symptom scores relative to baseline in the non-adherent group versus the remainder, adjusting for baseline characteristics including age, gender, ethnicity, IMD, comorbidities and baseline symptom scores. Over the 12-month follow-up period, the non-adherent group exhibited significantly greater increases in positive schizophreniform (β = 3.02, 95% CI [2.81, 3.23], p < 0.001), negative schizophreniform (β = 1.30, 95% CI [1.15, 1.45], p < 0.001), depressive (β = 2.27, 95% CI [2.03, 2.50], p < 0.001), disorganised (β = 1.10, 95% CI [0.99, 1.21], p < 0.001) and manic symptoms (β = 1.75, 95% CI [1.61, 1.89], p < 0.001). Following standardisation of variables to z-scores, further multilinear regression analyses modelling changes in symptom scores while adjusting for baseline characteristics were carried out. The group difference for increases in symptom score was greatest for positive schizophreniform (β = 0.97, 95% CI [0.90, 1.03], p < 0.001), followed by manic symptoms (β = 0.88, 95% CI [0.81, 0.95], p < 0.001), depressive (β = 0.71, 95% CI [0.63, 0.78], p < 0.001) and disorganised (β = 0.71, 95% CI [0.64, 0.79], p < 0.001) symptoms, and weakest for negative schizophreniform symptoms (β = 0.65, 95% CI [0.58, 0.73], p < 0.001), although confidence intervals for many of these overlapped.
Multilinear regression analyses of change in symptom score and change in number of comorbidities relative to baseline as a function of recorded treatment non-adherence and control variables

IMD, Index of Multiple Deprivation; LL, lower limit; UL, upper limit.
Table 3 also presents result of multilinear regression analyses modelling the change in the number of comorbidities relative to baseline, adjusting for control variables. The non-adherent group exhibited significantly greater increases in the number of comorbidities (β = 0.421, 95% CI [0.34, 0.50], p < 0.001) compared with the remainder.
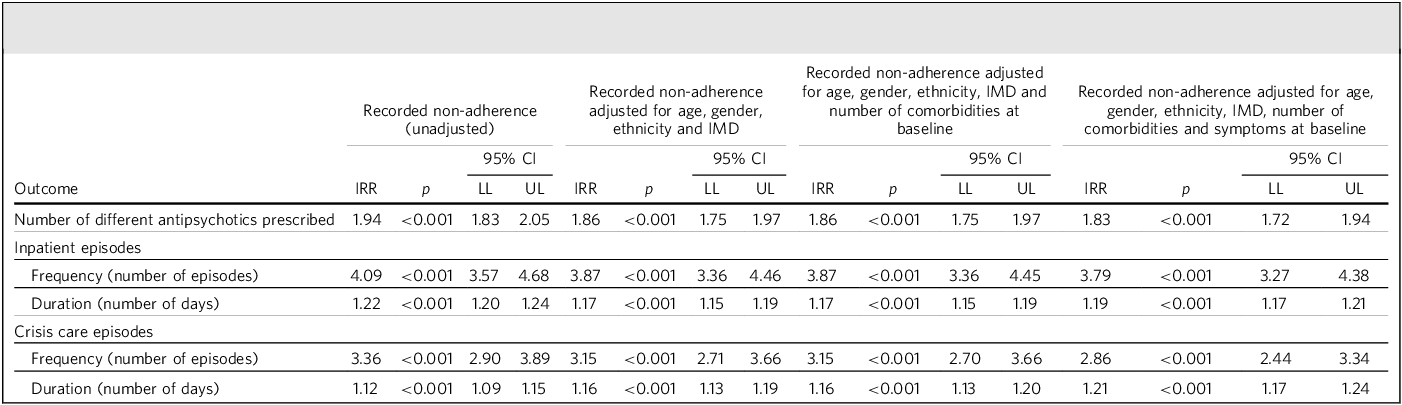
Table 4 presents the results of Poisson regression analyses, modelling the frequency and duration of in-patient admissions and crisis care episodes, and number of antipsychotics received during the 12-month follow-up. The non-adherent group compared with the remainder had a significantly greater frequency of both in-patient admissions (incidence rate ratio (IRR) = 3.79, 95% CI [3.27, 4.38], p < 0.001) and crisis care episodes (IRR = 2.86, 95% CI [2.44, 3.34], p < 0.001). In addition, the total duration of these episodes was significantly longer for the non-adherent group (in-patient days: IRR = 1.19, 95% CI [1.17, 1.21], p < 0.001; crisis care episodes: IRR = 1.21, 95% CI [1.17, 1.24], p < 0.001). The non-adherent group were also prescribed a significantly greater number of different antipsychotics than the remainder (IRR = 1.83, 95% CI [1.72, 1.94], p < 0.001).
Poisson regression analysis of number of different antipsychotics prescribed, and frequency and duration of in-patient and crisis care team admissions and as a function of recorded treatment non-adherence and control variables

IMD, Index of Multiple Deprivation; IRR, incidence rate ratio; LL, lower limit; UL, upper limit.
Discussion
In this study, we investigated recorded non-adherence to psychiatric treatment following a diagnosis of schizophrenia and its association with a range of clinical outcomes over the first 12 months using a large and well-characterised mental healthcare database. We found that recorded non-adherence was associated with a range of worse outcomes including greater use of hospital admission and crisis care, increased symptoms in all domains, higher numbers of different antipsychotic agents used and development of a greater number of recorded comorbidities.
Patients who were recorded as non-adherent with clinical interventions had increased frequency and duration of hospital admissions and crisis care episodes. This corroborates with many previous retrospective and prospective observational studies. Reference Abdullah-Koolmees, Nawzad, Egberts, Vuyk, Gardarsdottir and Heerdink14–Reference Ascher-Svanum, Faries, Zhu, Ernst, Swartz and Swanson19,Reference Novick, Haro, Suarez, Perez, Dittmann and Haddad21,Reference Morken, Widen and Grawe28 Particularly strikingly, our findings show that patients who are non-adherent to their psychiatric treatment have a 3.79 fold greater rate of hospital admission in the 12-month follow-up period relative to the remainder.
As well as crisis care, treatment non-adherence was also associated with greater symptom progression and higher numbers of antipsychotic agents prescribed. Both non-adherent and comparison groups demonstrated an increase in number of symptoms recorded during the 12-month follow-up period relative to baseline. It is expected that patients are followed up particularly closely during this period, so the reported increase in symptoms likely involves clinician recording behaviour as well as patient experiences. Nonetheless, the change in symptom scores in all domains was greater in the non-adherent group, which correlates with previous studies. Reference Morken, Widen and Grawe20 As reflected by regression analyses with standardised variables to compare symptom domains, the greatest group difference was observed in positive schizophreniform and manic symptoms. Symptoms of paranoia, in particular, may be reinforced by unsuccessful treatment engagements, thereby increasing the risk of subsequent coercive interventions. This mechanism may contribute to the increased rate of hospital admission observed among patients who are non-adherent to treatment. It follows that non-adherent patients were trialled on a greater number of antipsychotics firstly as a direct response to symptom progression despite attempted treatment but also possibly to optimise tolerability. Indeed, symptom progression and drug side-effects have been found to be among the strongest predictors of switching antipsychotics in the treatment of schizophrenia. Reference Nyhuis, Faries, Ascher-Svanum, Stauffer and Kinon29 Given the overlap in predictors for non-adherence and antipsychotic switching, alongside our finding that recorded non-adherence is associated with higher numbers of prescribed antipsychotic agents, further research is required to disentangle the relationship between these variables.
Our study also demonstrated that patients with recorded treatment non-adherence experienced a greater increase in the number of recorded physical health comorbidities during the follow-up period. This is particularly pertinent given that physical health conditions are the predominant factor contributing to the elevated mortality rate among individuals with schizophrenia compared with the general population. Reference Correll, Solmi, Croatto, Schneider, Rohani-Montez and Fairley4,Reference Laursen, Nordentoft and Mortensen5 Although an element of this may be attributed to detection bias, the underlying reason is likely multifactorial and reflects the impact of mental illness on a range of cardiometabolic risk factors including diet, exercise and smoking. Reference Osborn30 Disengagement from services may also result in fewer opportunities for health screening and interventions to prevent the onset and progression of physical health comorbidities. Targeted interventions to minimise the rate of treatment non-adherence may be beneficial in reducing morbidity and mortality in this population.
The adverse outcomes associated with treatment non-adherence parallel those associated with a prolonged duration of untreated psychosis (DUP). There is substantial evidence for a relationship between longer DUP and greater symptom severity, lower chance of remission and poorer overall functioning. Reference Howes, Whitehurst, Shatalina, Townsend, Onwordi and Mak31 Given that treatment non-adherence could be effectively conceptualised as an untreated period of illness, the extensive literature on DUP may provide a theoretical framework for examining mechanisms through which treatment non-adherence contributes to poorer clinical outcomes.
Strengths and limitations
Strengths of the study included a large cohort size and relatively detailed characterisation, including potentially important confounders such as symptom profiles and comorbidity. In addition, the sample is more representative of people with first schizophrenia diagnoses in the catchment than a traditional recruited cohort or trial sample.
Some key limitations need consideration when drawing conclusions. Considering the exposure of clinician-reported treatment non-adherence, there may be some circularity in the association, as poorer clinical outcomes may lead clinicians to suspect and record non-adherence. Another limitation is that positive adherence statements were not taken into account. Therefore, it may be that patients whose non-adherence to treatment was undocumented were grouped together with patients with recognised treatment adherence. In view of this heterogeneity, the observed effect sizes may underestimate the true differences between adherent and non-adherent individuals. Furthermore, clinicians have been shown to be poor at predicting adherence behaviour: Stephenson et al showed that 72% of patients with bipolar and/or schizophrenia whose pharmacy records indicated non-adherence were deemed to be adherent by their physician. Reference Stephenson, Tuncelli, Gu, Eisenberg, Panish and Crivera32 On the other hand, subjective measures including clinical assessment can have certain benefits over objective measures. The nature of clinical assessments can capture nuanced patterns of partial adherence which may be undetected by objective measures such as pharmacy records. Furthermore, medication prescription or dispensing (frequently used as adherence measures from routine data) do not necessarily indicate that the medication is being taken. Some studies have used drug plasma levels to estimate adherence, Reference Geretsegger, Pichler, Gimpl, Aichhorn, Stelzig and Grabher-Stoeffler33 but this method is also limited as it is affected by timing and the drug’s pharmacokinetic properties, and only reflects recent ingestion rather than a long-term pattern of behaviour. Overall, given the lack of definitive measures, future studies would benefit from using a combination of subjective and objective methods to more accurately determine the extent of adherence to psychiatric treatment.
Another limitation of this study is the bidirectional relationship between treatment adherence and illness severity. This is reflected by Table 1 in baseline differences between the non-adherent group and the remainder, particularly in symptom scores. While non-adherence is likely to lead to worsening symptoms and relapse, patients with more severe symptoms are also less likely to adhere to treatment. Reference Guo, Lv, Liu, Kong, Qu and Yue9 Although baseline characteristics and symptom profiles were controlled for in the present study, the complex interaction between non-adherence and illness progression makes it difficult to establish causality in either direction. Future studies could usefully aim to disentangle the overlap in predictors and clinical consequences of treatment non-adherence by examining their precise temporal relationship, which is likely to vary substantially between individuals.
Our findings contribute to a greater understanding of the consequences of treatment non-adherence in schizophrenia, which is essential for clinicians and patients to be better informed when considering treatment strategies. From the clinician’s perspective, better understanding would facilitate proactive monitoring and early intervention, which can be psychosocial as well as pharmacological. From the patient’s perspective, understanding of the consequences of treatment non-adherence would facilitate informed decision-making and autonomy. Given that family involvement has been shown to improve treatment adherence, our findings may also inform and encourage those involved in supporting patient care. Reference Pharoah, Mari, Rathbone and Wong34 A shared appreciation of both the value and disadvantages associated with interventions in mental healthcare is essential to forming trusting therapeutic alliances between patients and clinicians.
About the authors
Shayan Fassih and Kerr Hartop are Core Trainees at South London and Maudsley NHS Foundation Trust, London, UK. Jyoti Sanyal is Natural Language Processing Lead in the Centre for Translational Informatics, Institute of Psychiatry, Psychology & Neuroscience, King’s College London, London, UK. Robert Stewart is Professor of Psychiatric Epidemiology and Clinical Informatics at the Institute of Psychiatry, Psychology & Neuroscience, King’s College London, London, UK, and a member of the Mental Health for Older Adults and Dementia Clinical Academic Group at South London and Maudsley NHS Foundation Trust, London, UK.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjb.2026.10223
Data availability
All relevant aggregate data are found within the paper. The data used in this work have been obtained from CRIS, a system that has been developed for use within the National Institute for Health Research (NIHR) Mental Health Biomedical Research Centre (BRC) at South London and Maudsley NHS Foundation Trust (SLaM). It provides authorised researchers with regulated access to anonymised information extracted from SLaM’s electronic clinical records system. Individual-level data are restricted in accordance with the strict patient-led governance established at SLaM, and by NHS Digital for the case of linked data. Data are available for researchers who meet the criteria for access to this restricted data: (a) SLaM employees or (b) those with an honorary contract or letter of access from the trust.
Author contributions
S.F. contributed to the study conception and design, and development of the natural language processing model used for data acquisition, conducted data analysis and drafted the manuscript. K.H. contributed to data acquisition, including the development of the natural language processing model. J.S. contributed to data acquisition, including the development of the natural language processing model and data extraction from the CRIS database. R.S. provided oversight throughout the project and was involved in study conception and design and data acquisition and analysis. All authors critically reviewed the work and provided final approval prior to submission. All authors are in agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
R.S. and J.S. are part-funded by the NIHR Maudsley BRC at SLaM and King’s College London. R.S. is additionally part-funded by: (a) the National Institute for Health Research (NIHR) Applied Research Collaboration South London (NIHR ARC South London) at King’s College Hospital NHS Foundation Trust; (b) UKRI – Medical Research Council through the DATAMIND HDR UK Mental Health Data Hub (MRC reference: MR/W014386); (c) the UK Prevention Research Partnership (Violence, Health and Society: MR-VO49879/1), an initiative funded by UK Research and Innovation Councils, the Department of Health and Social Care (England), the UK devolved administrations and leading health research charities; (d) the NIHR HealthTech Research Centre in Brain Health. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of interest
None.
Ethics statement
The CRIS data platform has received ethics approval as a source of anonymised data for research use (Oxford REC C, reference 23/SC/0257).



eLetters
No eLetters have been published for this article.