The menopausal transition represents a significant phase in a woman’s life, marked by the cessation of ovulatory cycles accompanied by physiological and psychological changes that can strongly impact quality of life(Reference Davis, Pinkerton and Santoro1,Reference Lobo and Gompel2) . Women in the developed world can expect to live one-third of their lives in the postmenopausal phase. About 25 million women experience menopause each year, and a recent article in Lancet Global Health references WHO estimates that ‘by 2030, more than 1·2 billion women globally will be menopausal or post-menopausal’(Reference Delanerolle, Phiri and Elneil3). Yet, the number of women experiencing peri/menopause-like symptoms is likely to be much higher as women in the late-reproductive stage experience multiple symptoms often years before the official menopause transition. It is widely accepted that oestrogen plays a significant role in many disease states and related health outcomes(Reference Baker, Al-Nakkash and Herbst-Kralovetz4). Therefore, alterations in circulating oestrogen can exacerbate a wide range of physiological factors, with clinical implications for brain and gut health, the reproductive tract, and women’s overall well-being. Interestingly, oestrogens exert neuroprotective effects by modulating synaptic plasticity, neurogenesis, neuroinflammation and mitochondrial function(Reference Arevalo-Rodriguez, Smailagic and Roqué-Figuls5,Reference Brinton, Yao and Yin6) . Thus, menopausal hormonal fluctuations influence neurochemical processes within the central nervous system (CNS) leading to symptoms such as mood swings – characterized by unpredictable irritability and anxiety – and cognitive difficulties like memory lapses and reduced attention often referred to as ‘brain fog’(Reference Gareau, Cryan and Clarke7). In addition, vasomotor symptoms, such as hot flashes and night sweats,(Reference Guo, Xiong and Yang8) due to alterations in the hypothalamic center, can be challenging to cope with and may impair sleep, thereby exacerbating stress(9). Oestrogen decline may, over time, contribute to substantial impairments in neuronal integrity, thereby increasing the risk of neurodegenerative disorders, including alzheimer’s dementia(Reference Breeze, Connell and Wileman10). CNS-related symptoms affect more than 85 % of women with 25 % of these describing their symptoms as severe(11). In a survey carried out by The Menopause Hub, 61 % of women said that menopause negatively impacted their relationship with their significant other and 84 % said their work performance was negatively impacted. One third of women had considered giving up work due to symptoms(12).
Gut dysbiosis, which refers to an imbalance within the community of microorganisms, is known to influence functions of the CNS, whilst emotional and psychological stressors perceived by the brain influence the gut microbiome through the gut-brain axis(Reference Anand, Gorantla and Chidambaram13,Reference Mehta, Juneja and Nimmakayala14) . The gut microbiota can produce neurotransmitters as well as microbial-derived neuroactive products such as short-chain fatty acids (SCFAs), which allow microbes to both directly and indirectly impact the nervous system(Reference Kern, Mastandrea and Melekhova15). The gut microbiome is also directly involved in maintaining serum levels of hormones such as oestrogen, progesterone and luteinizing hormone, with a specific community of commensal gut bacteria, known as the oestrobolome, being capable of deconjugating oestrogens and leading to their systemic recycling(Reference Diviccaro, Caputi and Cioffi16). Indeed, the gut microbiome has been noted to be altered in certain female-specific conditions linked with hormonal imbalance such as polycystic ovary syndrome, endometriosis and symptoms of menopause(Reference Siddiqui, Makhlouf and Alharbi17). The interaction between the gut microbiome and female hormones, as well as their potential links to the menopausal transition, offers promising therapeutic opportunities. Recently, the ZOE Predict study identified 32 bacterial species linked to both menopause symptoms and diet quality, possibly suggesting that diet can influence menopause symptoms through effects on the gut microbiome(Reference Bermingham, Linenberg and Hall18). Ongoing research is centred on microbiome-modulating interventions such as dietary changes – particularly increasing fibre and prebiotic foods – that promote microbes associated with anti-inflammatory and neuroprotective properties. Other potential interventions include synbiotics where both prebiotics and probiotics (health-promoting bacteria) are administered together to optimize colonization and efficacy.
This review explores the potential of the microbiome in women’s healthcare. We examine how gut microbiome composition changes during menopause, the influence of gut bacteria on sex hormone levels and their role in modulating neurotransmitter production, stress responses and CNS function throughout the menopausal transition. We aim to highlight the potential of dietary, lifestyle and nutritional supplement interventions in supporting gut and brain health as well as overall well-being during peri- and post-menopause.
Sex differences in the gut microbiome across the lifespan
The gut microbiome is defined as the ecosystem of numerous commensal, symbiotic and pathogenic microorganisms, including bacteria, fungi, protozoa and viruses(Reference Armet, Deehan and O’Sullivan19). This community of microorganisms plays an important role in the development of physiological functions of the host at many levels, including immunological, metabolic and neurological pathways(Reference Zmora, Suez and Elinav20). It has been associated with both health and disease states and through a series of pathways, referred to as the microbiome-gut-brain axis, can impact the CNS including cognitive health and emotional well-being(Reference Mehta, Juneja and Nimmakayala14). Gut microbiota composition varies throughout life, influenced by diet, environment, antibiotics, stress, genetics, leading to significant individual differences(Reference Costello, Lauber and Hamady21). Sex and age are also important factors influencing gut microbiome composition among numerous other environmental and physiological confounder(Reference Falony, Joossens and Vieira-Silva22,Reference Zhang, Zhong and Li23) For this review we will focus on the bacterial element of the microbiome.
Shortly after birth, the gut microbiome undergoes rapid shifts, particularly during the weaning period when a baby transitions from milk to solid food. Breastfeeding is associated with higher levels of Bifidobacterium species (B. breve, B. bifidum), while cessation of breast milk accelerates microbiome maturation, characterized by an increase in the phylum Firmicutes (Reference Stewart, Ajami and O’Brien24). Pre-clinical studies have indicated that the microbiome in childhood influences resilience to psychological stress, with sex-related differences manifesting as early as the first stages of life(Reference Wilmes, Caputi and Bastiaanssen25).
Puberty marks a significant shift in the composition of the gut microbiota. With pubertal development, the faecal microbiota in females (but not in males) becomes more adult-like, with a notable rise in Clostridium bacteria that metabolize oestrogen and a decline in Bacteroidia. These results imply that the gut microbiome may influence the timing of puberty by modifying the levels of sex hormones(Reference Korpela, Kallio and Salonen26,Reference Yuan, Chen and Zhang27) .
Sex differences in the adult gut microbiome are well-documented and are hypothesized to be driven by sex hormones production(Reference Zhang, Zhong and Li23). Large human studies report higher microbial richness with increased Bifidobacterium abundance in women, whereas men more frequently show elevated Prevotella levels(Reference Falony, Joossens and Vieira-Silva22,Reference Zhernakova, Kurilshikov and Bonder28,Reference Özkurt and Hildebrand29) . Direct effects of sex hormones on gut microbes have been demonstrated previously in pre-clinical models. For instance, castration of male mice can lead to a gut microbiome that is more similar to that of females(Reference Yurkovetskiy, Burrows and Khan30). Furthermore, it was noted that faecal microbiota from a single male donor transplanted into gnotobiotic mice produced distinct microbial community structures in male v. female recipients(Reference Wang, Wang and Pang31). Finally, faecal microbiota transfer from adult males to immature females leads to metabolic changes, demonstrating that the microbial composition is closely related to the metabolic profile of the host, particularly in the patterns of hormone regulation and immune defence(Reference Markle, Frank and Mortin-Toth32).
Gut microbiota analysis of postmenopausal women supports the role of sex hormones in shaping these differences. It has been reported that postmenopausal women tended toward lower gut microbiome diversity compared to premenopausal women, while differing less from men(Reference Peters, Lin and Qi33). Three bacterial taxa showed increased abundance in postmenopausal women (Bacteroides sp. strain Ga6A1, Prevotella marshii, Sutterella wadsworthensis), while seven showed decreased abundance (Escherichia coli-Shigella spp., Oscillibacter sp. strain KLE1745, Akkermansia muciniphila, Clostridium lactatifermentans, Escherichia coli, Parabacteroides johnsonii and Veillonella seminalis). Interestingly, these species differed in a similar way between younger men and premenopausal women(Reference Peters, Lin and Qi33). Menopause has been linked to changes in metabolism and previous research has demonstrated that Prevotella and Sutterella are associated with obesity and metabolic dysfunction across various studies. An increase in the abundance of these microbial taxa may, therefore, have detrimental effects on host health(Reference Pinart, Nimptsch and Forslund34). Interestingly, oestrogen supplementation during menopause slows the progression of atherosclerosis and corrects lipid metabolism disorders potentially by regulating the abundance of specific gut microbiota(Reference Meng, Ma and Zhang35). A further study reported that postmenopausal women are marked by a significant intestinal dysbiosis, specifically reflected in the deficiency of Bifidobacterium animalis, Aggregatibacter segnis and Acinetobacter guillouiae. These bacterial species are associated with sex hormone levels, and their imbalance may contribute to the pathophysiology of menopausal syndrome(Reference Liu, Zhou and Mao36). Overall, menopause has been linked to reduced diversity and a shift toward a more ‘male-like’ microbiome, supporting the hypothesis that declining oestrogen levels contribute to these compositional shifts and potentially some of the physiological effects that impact the health of the postmenopausal woman(Reference Peters, Lin and Qi33).
The role of the oestrobolome
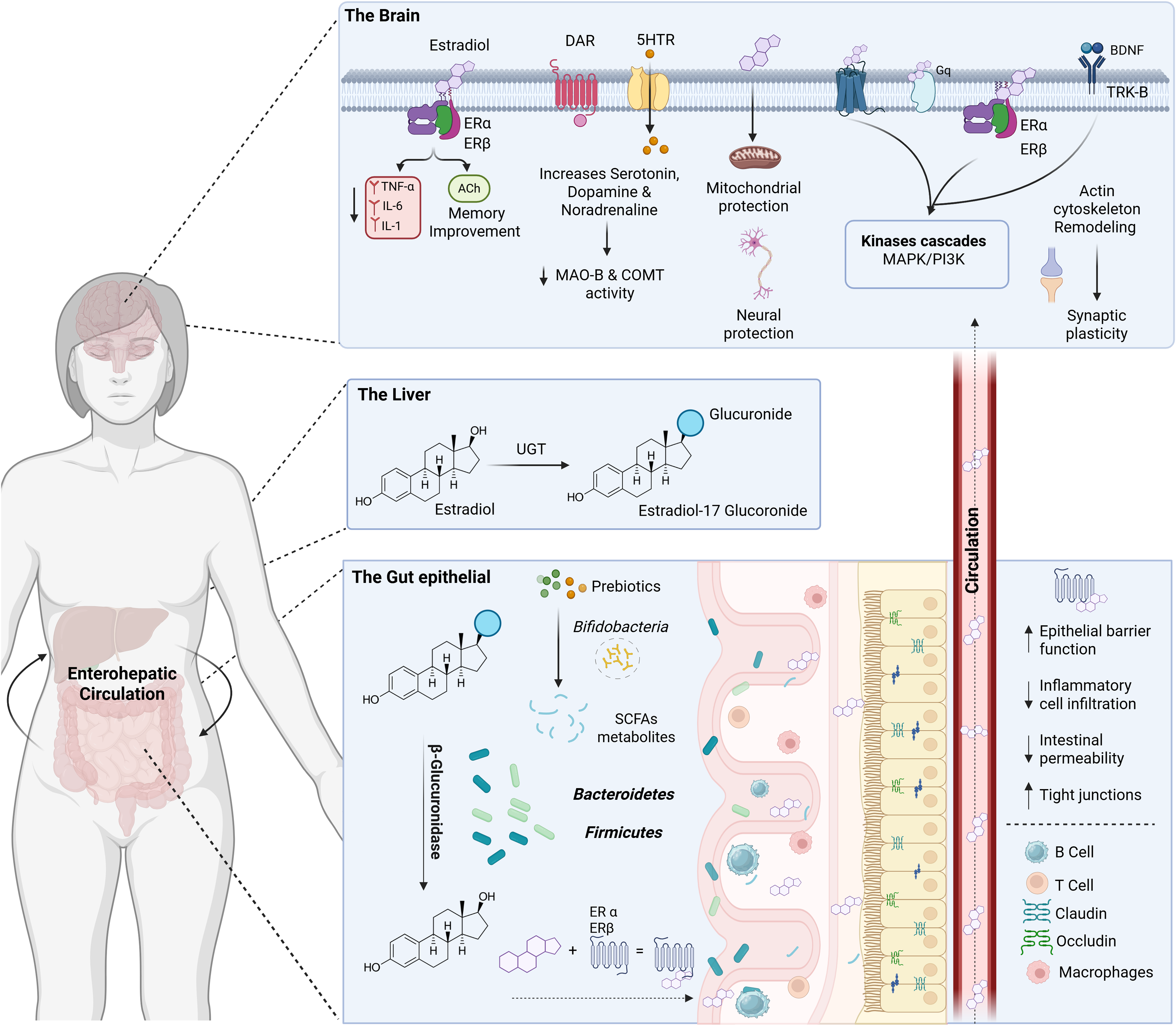
Many studies support a bidirectional relationship between sex hormones and the gut microbiome throughout a woman’s life(Reference Bermingham, Linenberg and Hall18,Reference Peters, Lin and Qi33) . Higher levels of oestrogens promote increased microbial diversity by serving as substrates for microbial metabolism. Recently, researchers isolated oestradiol-degrading gut microbes by culturing with oestradiol as the single carbon source. They also linked this activity to depression in premenopausal women due to reduced serum oestradiol(Reference Li, Sun and Tong37). Moreover, it has been reported that Denitrifying Denitratisoma sp. DHT3 can harvest energy using oestradiol/oestrone as sole substrates anaerobically(Reference Wang, Chen and Wei38). Conversely, gut bacteria may help process and regulate circulating hormone levels. Indeed, oestrogens undergo enterohepatic circulation, being first conjugated in the liver (e.g., as glucuronides and sulphates) by enzymes like UDP-glucuronosyltransferase (UGTs), which increase their hydrophilicity and facilitate biliary excretion. However, a portion of these oestrogens reaches the intestine going through a recycling process. The term ‘estrobolome’ refers to that set of bacteria that encode genes for enzymes capable of deconjugating oestrogens, such as β-glucuronidase (GUS) and sulphatase, preventing their excretion and allowing re-entry of free hormone into the circulation(Reference Diviccaro, Caputi and Cioffi16) (Figure 1).
Gut-brain axis interactions mediated by oestrogen metabolism. The enterohepatic recirculation maintains oestrogens bioavailability through metabolic pathways linking oestrogens, the liver and the gut–brain axis, with implications for cognition, mood regulation, and inflammation. This mechanism is particularly relevant during states of oestrogen decline, such as menopause. Abbreviations: ERα, Oestrogen receptor alpha; ERβ, oestrogen receptor beta; DAR, dopamine receptor; 5HTR, 5-hydroxytryptamine receptor; Gq, Gq alpha subunit of heterotrimeric G protein; TRK-B, tropomyosin receptor kinase B; TNF-α, tumour necrosis factor alpha; IL-6, interleukin 6; IL-1, interleukin 1; ACh, acetylcholine; MAO-B, monoamine oxidase-B; COMT, catechol-O-methyltransferase; MAPK, mitogen-activated protein kinase pathway; PI3K, phosphoinositide 3-kinase pathway; UGT, Uridine 5’-diphospho-glucuronosyltransferase; SCFAs, Short chain fatty acids.

In vitro studies have demonstrated that many microbial taxa, especially within the Firmicutes and Bacteroidetes phyla, hydrolyze the glucuronide or sulphate groups from oestrogen metabolites, converting them back into active forms(Reference Pollet, D’Agostino and Walton39,Reference Ervin, Simpson and Gibbs40) . These microbial enzymatic pathways decline during menopause due to changes in gut microbiota diversity and marked loss of sex hormones oestrogen and progesterone(Reference Flores, Shi and Fuhrman41–Reference Goedert, Jones and Hua43). Peters et al. (2022) showed that abundance of bacterial β-GUS was significantly lower in postmenopausal women compared to premenopausal women and interestingly, also tended to be lower in younger and older men compared to premenopausal women. Akkermansia muciniphila was among the taxa depleted in postmenopausal women. It was involved in retention of sex steroid hormones and thus decreased after menopause due to loss of conjugated sex steroid substrates(Reference Peters, Lin and Qi33). This species is known to express β-GUS and aryl-sulphatase activities in vitro (Reference Ervin, Simpson and Gibbs40,Reference Ervin, Li and Lim44,Reference Tarushi Gupta, Kumar and Bihani45) , and it is positively associated with the abundance of estrobolome-related genes and progestin steroid metabolites, especially in postmenopausal women. Consistent with these observations, A. muciniphila was less abundant in gonadectomized compared to gonadal intact female mice. Moreover, in the presence of β-oestradiol, the growth of this species exhibited exponential increase providing evidence for the identification of A. muciniphila as an oestrogen-responsive microbiota(Reference Sakamuri, Bardhan and Tummala46).
The use of combined hormonal contraceptives in healthy premenopausal women has been associated with a reduced gut microbiome diversity and changes in the relative abundance of several bacterial taxa, which may be a consequence of the hormonal contraceptives-induced decrease in oestradiol and progesterone serum levels(Reference Mihajlovic, Leutner and Hausmann47). For instance, a lower abundance of Eubacterium eligens (Reference Mihajlovic, Leutner and Hausmann47), a well-studied bacterium in the human gut microbiome, known to possess GUS activity(Reference Candeliere, Raimondi and Ranieri48) and linked with healthy dietary habits and favourable cardiometabolic markers in a large human cohort was noted with the use of hormonal contraceptive(Reference Asnicar, Berry and Valdes49). Surprisingly, Akkermansia was inversely correlated with low oestradiol/progesterone levels, while showing significant variations throughout the participants’ menstrual cycles: higher abundance in the luteal phase (high oestradiol and progesterone) v. the follicular phase (low oestradiol and progesterone)(Reference Mihajlovic, Leutner and Hausmann47). These findings imply a rather complex role of hormonal fluctuations in the growth of Akkermansia, which requires further analysis. In women using contraceptives, hormone levels are modified and maintained artificially, which may explain the differences in microbial fluctuations compared to women with a natural menstrual cycle. From this perspective, during menopause, Akkermansia populations might be positively correlated with the physiological and natural progressive decline of oestrogen.
Honda et al. (2024) performed an in vitro screening of GUS activity for 84 bacterial strains from human and fermented food. Lactobacillus brevis KABP052 exhibited the highest deconjugation activity and, formulated within a probiotic blend, maintained serum oestrogen levels in peri- and postmenopausal women(Reference Honda, Tominaga and Espadaler-Mazo50). Further evidence shows a positive correlation between oestrobolome richness and serum levels of progesterone and oestradiol. For example, higher serum oestradiol levels were associated with greater abundances of Firmicutes and Faecalibacterium and decreased abundances of Bacteroidetes in a Chinese study that included 53 women with or without premature ovarian insufficiency(Reference Wu, Zhuo and Liu51). Similarly, among 37 Spanish women, Gammaproteobacteria levels were higher and Prevotellaceae levels were lower in those who were pre- and post-menopausal(Reference Santos-Marcos, Rangel-Zuñiga and Jimenez-Lucena52). These findings highlight the strong impact of hormonal fluctuations on the composition and diversity of the gut microbiota across various populations and menopausal statuses, although there are many potentially confounding factors, including diet(Reference Gentile and Weir53), endocrine factors(Reference Sandhu, Sherwin and Schellekens54) and biological and cultural variables(Reference Dennerstein, Lehert and Koochaki55). Collectively, these investigations provide support for the gut microbiota as a potential therapeutic target for treating oestrogen-dependent disorders in women, including menopausal symptoms and comorbidities. As postmenopausal women lack ovarian hormone production and have low levels of oestrogens and progesterone, enterohepatic recycling by the gut microbiota plays an even more important role in regulating and restoring the balance of these hormones(Reference Peters, Lin and Qi33) Therefore, continued research could lead to novel, microbiome-based strategies for improving the quality of life for women during and after the menopausal transition.
The menopause–brain connection
Menopause is considered a period of neurological transition, which plays a significant role in women’s brain health and emotional well-being(Reference Davis, Pinkerton and Santoro1,Reference Lobo and Gompel2) . Changes in brain structure, connection and metabolism that cannot be entirely explained by chronological aging alone, occur during midlife, coinciding with the reduction and eventual cessation of ovarian hormone production, most notably 17 β-oestradiol(Reference Mosconi, Rahman and Diaz56,Reference Ramli, Yahaya and Mohd Fahami57) . Interestingly, early menopause and surgical ovariectomy have been linked to a decline in cognitive function and an increased risk of dementia(Reference Rocca, Lohse and Smith58). Furthermore, assessments of exposure to hormone therapy indicate that exposure to oestradiol during middle age may influence long-term brain outcomes(Reference Nerattini, Jett and Andy59). According to various epidemiological data, women are disproportionately affected by several serious brain diseases, being more likely to develop Alzheimer’s disease and dementia, compared to men especially in old age(Reference Nerattini, Jett and Andy59–Reference Barth, Galea and Jacobs61). Menopausal hormonal fluctuations can influence neurochemical processes within the CNS. Common symptoms include mood swings – characterized by unpredictable irritability and anxiety – and cognitive difficulties like memory lapses and reduced attention often referred to as ‘brain fog’(Reference Gareau, Cryan and Clarke7). Moreover, multimodal neuroimaging studies across the menopause transition have demonstrated specific structural and functional brain changes, affecting networks that support memory, executive function and mood regulation(Reference Mosconi, Rahman and Diaz56,Reference Kim, Kim and Kim62) . Several studies have shown that oestrogens stimulate neural growth both in vitro (Reference Nathan, Barsukova and Shen63,Reference Pooley, Luong and Hussain64) and in animal models(Reference Phan, Suschkov and Molinaro65–Reference Brill and Linden67). The brain itself produces ‘neuroestrogens’ via local aromatase – documented in human cortex and other regions (notably hippocampus, hypothalamus, striatum and nucleus accumbens) – modulating neurotransmitter systems (dopamine, serotonin, noradrenaline and acetylcholine) and reducing pro-inflammatory cytokine activity (TNF-α, IL-6, IL-1) through nuclear receptors ERα/ERβ and the membrane G-protein–coupled receptor (GPCR)(Reference Arevalo-Rodriguez, Smailagic and Roqué-Figuls5,Reference Brinton, Yao and Yin6,Reference Hara, Waters and McEwen68) (Figure 1). Zhang et al. (2024) described molecular mechanisms by which oestrogens increase brain-derived neurotrophic factor (BDNF) expression in hippocampus, amygdala and cortex via ERα/ERβ and downstream activation of MAPK/PI3K pathways promoting mitochondrial protection, neuronal growth and synaptic plasticity(Reference Zhang, He and Pan69) (Figure 1). Abnormal spontaneous activities in multiple brain regions (frontal, temporal and hippocampal areas) have been detected in perimenopausal women during resting states(Reference Lu, Sun and Gao70,Reference Zhang, Fu and Liu71) . Multimodal imaging shows peri/post-menopausal women have reduced glucose metabolism, reduced grey-matter volumes, white-matter/connectivity changes and increased amyloid-β deposition and structural atrophy than premenopausal women(Reference Mosconi, Rahman and Diaz56,Reference Mosconi, Berti and Dyke72) . Recently, it was also found an association between higher white-matter hyperintensity burden and oestradiol declines/menopausal status(Reference Thurston, Chang and Wu73).
Increasing evidence highlights the critical role of the gut microbiota in health and disease, particularly through the so-called ‘gut-brain axis.’ This crosstalk involves neural, hormonal, immune and metabolic pathways, that not only ensures the proper maintenance of gastrointestinal homeostasis, but also influence CNS influencing brain development, mood and cognitive functions(Reference Fassarella, Blaak and Penders74,Reference Yu, Agirman and Hsiao75) . Gut dysbiosis compromises the physiological functions and contributes to disease pathogenesis. Notably, gut microbiota imbalance is known to influence CNS functions and conversely emotional and psychological stress can influence gut microbiota through the bidirectional gut–brain communication pathways(Reference Möhle, Mattei and Heimesaat76,Reference Vuong, Yano and Fung77) . During menopause, the gut–brain axis is particularly vulnerable due to the negative impact of oestrogen decline on gut microbial ecology. Conversely, a reduction of gut microbiota diversity observed in postmenopause further impacts oestrogen levels(Reference Santos-Marcos, Rangel-Zuñiga and Jimenez-Lucena52), potentially intensifying CNS-related menopausal symptoms. Gut microbial dysbiosis contributes to various neurological disorders via microbial translocation, which occurs when intestinal mucosa becomes more permeable than normal, allowing bacteria and their products to leak into the bloodstream(Reference Brenchley and Douek78). This increased intestinal permeability, commonly referred to as ‘leaky gut’ contributes to systemic inflammation and altered signalling pathways and leads to breakdown of the blood–brain barrier activating the microglia and favouring neuroinflammation(Reference Kelly, Kennedy and Cryan79,Reference Pahwa, Goyal and Jialal80) . The ovarian hormones play a supportive role in the physiology of the gastrointestinal tract, through influencing the mucus and epithelial barriers, as well as affecting resident macrophages(Reference Feng, Tang and Zhang81–Reference Zhang, Zhu and Yu84). In mice, ovariectomy-induced oestrogen deficiency has been shown to induce changes in permeability, tight junction proteins and cytokine gene expression(Reference Collins, Rios-Arce and Atkinson85). Oestradiol also protected the mucus-producing intestinal epithelial cells against oxidant injury, reducing damage, apoptosis and cell permeability in an in vitro model(Reference Diebel, Diebel and Manke86). Emerging evidence underscores how chronic psychological stress can disrupt integrity of the intestinal barrier and adversely affect estrobolome populations in women more significantly than in men(Reference Audet87). A recent study demonstrated that 28 d of stress resulted in significant alterations in the proportions of Erysipelotrichaceae and Lactobacillaceae in both male and female mice, albeit in opposing directions(Reference Kropp, Rainville and Glover88). Moreover, female mice exhibit increased susceptibility to stress-induced dysbiosis indicating that oestrogen-microbiome feedback loops may amplify vulnerability. Female mice are more sensitive to imbalances in the microbiota caused by stress, likely due to the bidirectional system that connects female hormones and gut bacteria, which makes this vulnerability even stronger. Therefore, female subjects may be at higher risk of having health issues related to stress and dysbiosis compared to males(Reference Doney, Dion-Albert and Coulombe-Rozon89). A clinical study demonstrates microglial activation measured with PET-TSPO after systemic administration of LPS, providing direct evidence that endotoxin-induced peripheral stimulation/microbial translocation can produce signals of neuroinflammation detectable by imaging studies(Reference Sandiego, Gallezot and Pittman90). Elevated proinflammatory cytokines, including IL-6 and TNF-α and microbial translocation markers have been observed in perimenopausal women with depressive symptoms(Reference Peters, Lin and Qi33,Reference Shieh, Epeldegui and Karlamangla91,Reference Soares, Sampaio and Ferreira92) . Shieh et al. (2020) also found an increased permeability linked to a reduction in oestradiol and increased FSH, crucial for demonstrating that the endocrine factor can initiate the cascade that then leads to brain changes(Reference Shieh, Epeldegui and Karlamangla91).
These data support the hypothesis that immune activation, resulting from microbial translocation, contributes to the pathophysiology of perimenopausal mood disorders. However, longitudinal investigations that concurrently measure sex hormones, permeability biomarkers, diagnostic imaging and neuropsychiatric evaluations within the same cohort during the menopausal transition would be needed. Overall, gut barrier dysfunction and microbial translocation may be an overlooked mechanistic link between endocrine decline and neuropsychiatric consequences, although few studies have offered direct human evidence. Establishing this connection in larger, prospective human studies could open new therapeutic opportunities targeting the gut barrier and microbiota for the prevention and treatment of peri/post menopausal mood disorders and cognitive decline.
Gut–hormone axis in menopause: microbiota modulation and therapeutic perspectives
The gut microbiota plays a key role in regulating oestrogen metabolism and has emerged as an important area of research for improving women’s mental health during midlife and post-menopause. Psychobiotics, defined as microbes or dietary strategies that modulate gut microbiota, may support mental well-being by promoting microbial balance(Reference Anderson, Cryan and Dinan93). Targeted interventions, including diet, lifestyle modifications and probiotic or prebiotic supplementation, have the potential to restore gut microbial homeostasis, thereby influencing oestrogen regulation, reducing disease risk and supporting overall well-being.
Nutrition is one of the key determinants of the composition of the gut microbiota(Reference Moles and Otaegui94) and one of the most important factors regulating overall health, including both physical and mental well-being(Reference Moore, Hughes and Ward95). Increasing amounts of evidence show the beneficial impact of diet on brain health(Reference Scarmeas, Anastasiou and Yannakoulia96). In discussing the central symptoms of menopause, Cushen and Johnston (2025) propose evidence-based dietary approaches designed for women dealing with cancer treatment-induced menopause(Reference Cushen and Johnston97). The NiMe dietary strategy emphasizes a diet rich in vegetable fibres to support microbial diversity(Reference Armet and Walter98). Indeed, diets that contain high levels of plants, fibre, healthy fats and whole grains, rather than processed foods, can improve the health of the gut microbiome and tend to reduce mental health risks. Researchers examining the relationship between diet and mental health found that a well-balanced diet containing fibre, n-3 fatty acids and essential vitamins is associated with less risk of developing symptoms of depression, anxiety and stress(Reference Taylor and Holscher99) and has been shown to support cognitive function and neurological health(Reference Silva, Oppermann and Reis100). Mediterranean diet, full of fruits, vegetables and wholegrains and lower in red meat, processed grains and fried foods, are also related to a lower risk of depression(Reference Kesse-Guyot, Assmann and Andreeva101) as well as a healthier gut microbiome(Reference Filippis, Pellegrini and Vannini102). Research from a clinical trial found a significant reduction of depressive symptoms among participants after a 12-week regimen of high-quality dietary interventions, in comparison to individuals who did not adhere to dietary guidance(Reference Jacka, O’Neil and Opie103). Some population studies have shown that eating more cruciferous vegetables can prevent age-related cognitive decline in healthy older adults(Reference Nouchi, Hu and Saito104). Furthermore, a plant-based diet supplemented with Raphanus sativus L., belonging to the cruciferous family, may enhance gut health and cognitive performance in perimenopausal women, confirming a bidirectional relationship between the microbiota and the brain(Reference Guo, Xiong and Yang8).
Probiotics are live microorganisms that confer a health benefit when administered in adequate amounts(Reference Hill, Guarner and Reid105). Probiotic supplementation emerges as a promising strategy to restore gut homeostasis, improve neurotransmitter regulation, attenuate neuroinflammation and ultimately alleviate central menopausal symptoms(Reference Hill, Guarner and Reid105). A recent systematic review/meta-analysis of 30 clinical trials on the effects of probiotic supplements on symptoms of depression found positive benefits in alleviating depression and/or anxiety(Reference Merkouris, Mavroudi and Miliotas106). Research into the identification and replacement of bacteria belonging to the oestrobolome is ongoing, with the goal of developing microbiota-based therapies that restore the ‘missing microbes’ of menopause, thereby influencing oestrogen metabolism and availability. A randomized clinical trial demonstrated the oestrogen modulation ability of a probiotic formula containing GUS-positive Lactobacilli species in women. The strains were first tested for GUS activity in vitro, two-week intervention of the GUS-positive species inducing changes to serum oestrogen levels in healthy postmenopausal women compared to a placebo group, setting the stage for future use of probiotics in the postmenopausal population(Reference Honda, Tominaga and Espadaler-Mazo50). Lactobacillus rhamnosus is one of the most widely studied probiotic strains and its beneficial effects on human health have been examined in numerous clinical trials. L. rhamnosus has been shown to effectively reduce postpartum symptoms of depression and anxiety(Reference Slykerman, Hood and Wickens107) and its metabolites were shown to reduce the impact of early-life stress on cortisol(Reference McVey Neufeld, O’Mahony and Hoban108) and CNS gene changes(Reference McVey Neufeld, Strain and Pusceddu109) in pre-clinical studies. L. Rhamnosus is also a member of the oestrobolome bacterial community in the gut and possesses the enzymatic capacity of β-GUS to de-conjugate oestrogen in the gastrointestinal tract to allow it to move back into systemic circulation(Reference Ervin, Li and Lim44). It was found that during the perimenopausal period, the relative abundance of beneficial bacteria such as Lactobacillus and Bifidobacteria is markedly reduced while that of harmful bacteria such as Enterobacter is increased(Reference Wang, Shi and Zheng110). The ZOE predict study, one of the largest research analyses of menopause and nutrition in the world, indicated that postmenopausal women exhibit increased postprandial glucose and insulin responses, compared to premenopausal women. These effects are partially mediated by diet and microbiota composition: it was observed that the loss of fundamental microorganisms – including those involved in oestrogen reactivation in the gut – may amplify the risk of central disorders through effects on blood glucose, inflammation and sleep quality(Reference Bermingham, Linenberg and Hall18). For instance, Bifidobacteria adolescentis showed a significantly lower abundance in postmenopausal women. It is a well-known gamma-aminobutyric acid (GABA) producer which acted as an anxiolytic in both pre-clinical(Reference Duranti, Ruiz and Lugli111) and clinical studies(Reference Murakami, Ko and Ouchi112).
In addition to live microorganisms, postbiotics are bioactive substances generated by bacteria, including peptides, exopolysaccharides and SCFAs. They exert direct effects on the immunological, endocrine and neurological systems of their hosts. Among them, SCFAs like butyrate, propionate and acetate are produced by fibre fermentation and have been demonstrated to control systemic inflammation, strengthen the integrity of the intestinal barrier and interact with receptors and signalling pathways that affect bone metabolism, energy balance and neuroinflammation(Reference Fock and Parnova113–Reference Lucas, Omata and Hofmann116). Research suggests that butyrate also plays a role in regulating β-GUS activity. It has been shown to reduce intestinal leak, markers of systemic inflammation and oxidative stress in postmenopausal women in a randomized controlled trial(Reference Qaisar, Zuhra and Karim117). The decrease of SCFA-producing bacteria has been linked to negative consequences such as metabolic dysfunction and neurocognitive changes in oestrogen-deficient conditions like menopause. Therefore, probiotic or dietary therapies that enrich SCFA-producing taxa, or postbiotic supplements, have the potential to alleviate central and systemic menopausal symptoms through the gut–brain axis.
Prebiotics such as inulin, galacto-oligosaccharide (GOS) or fructo-oligosaccharide (FOS), help feed beneficial bacteria, including members of the Bacteroidetes and some Firmicutes. These bacteria possess specific glycoside hydrolases that allow breakdown of oligosaccharides into monosaccharides, which are then metabolized via glycolysis and fermentation pathways. This fermentation produces SCFAs such as acetate, propionate and butyrate, which lower intestinal pH, nourish colonocytes, strengthen barrier function and modulate immune and neuroendocrine signalling. Through these mechanisms, bacterial metabolism of prebiotics links diet to gut health, systemic physiology and even mental well-being(Reference Gänzle and Follador118,Reference Holscher119) . Prebiotics are usually found in a wide range of vegetables, fruits and grains, including asparagus, onions, leeks, garlic, bananas, Jerusalem artichokes, chicory root, legumes like chickpeas and lentils, barley, oats, rye and other whole grains. GOS are functional oligosaccharides composed of galactose units bound to lactose, mainly produced industrially from milk through the enzymatic action of β-galactosidase, so they are mainly found in enriched milks, infant formulas and supplements. GOS has also been extensively used in infant formula as it has an extremely low risk of adverse effects and is associated with positively impacting the gut microbiome and reducing infections(Reference Miqdady, Al Mistarihi and Azaz120). A double-blind intervention trial provides insights into the potential impact of GOS supplementation on gut microbiota composition, particularly by allowing efficient colonisation of Bifidobacteria adolescentis (Reference Looijesteijn, Schoemaker and van den Belt121). Krumbeck et al. has shown participants who consumed increasing doses of GOS for 9 weeks had an 8-fold enrichment in Bifidobacterium adolescentis strain in their gut during GOS administration indicating a synbiotic relationship between this prebiotic and this missing menopause microbe(Reference Krumbeck, Maldonado-Gomez and Martínez122). Furthermore, Bifidobacteria grown in GOS improved gut barrier function in adults living with obesity(Reference Krumbeck, Rasmussen and Hutkins123) which has been noted to disimprove during menopause with increased systemic inflammation and reduced bone density(Reference Shieh, Epeldegui and Karlamangla91).
Fermented dairy foods, which naturally contain a range of probiotics, show benefits for gut health and menopausal-relevant outcomes in women. For example, there is an increasing interest in the commercial use of kefir since it can be marketed as a natural beverage containing health-promoting bacteria. Peptides, bioactive compounds and strains occurring in kefir, can modulate gut microbiota composition, low-grade inflammation and intestinal permeability, which have all been shown to impact on the gut-brain axis(Reference Peluzio, Dias and Martinez124). Interestingly, kefir exhibited positive effects on sleep disturbances, depression and quality of life in postmenopausal women(Reference Özcan, Oskay and Bodur125). Furthermore, a recent pre-clinical study demonstrated that the consumption of goat’s milk yogurt in young rats increased the beneficial genera Blautia and Fusicatenibacter compared to control mice, contributing to the induction of anxiolytic-like behaviour(Reference Dutra, Aquino and Alves126).
The ability of the gut microbiome to regulate oestrogen levels lies not only in the deconjugation of endogenous oestrogens and their consequent reactivation, but also through the conversion of plant-based compounds, classified as phytoestrogens, into bioactive metabolites which can mimic oestrogens effects in the host. Phytoestrogens, including isoflavones like genistein and daidzein, are present in foods such as fruits (grapes, plum, pear and apple), vegetables (soybeans, kidney beans, cabbage, spinach, hops, garlic and onion), wine and tea; as well as a number of botanical dietary supplements(Reference Bacciottini, Falchetti and Pampaloni127). Bacterial species belonging to the estrobolome have enzymatic pathways capable of hydrolysing glycosidic groups from isoflavones, converting them into free aglycones that can be absorbed and may bind to oestrogen receptors (ERs)(Reference Kumari, Kumari and Dua128). Phytoestrogens can be further processed by specific intestinal bacteria into metabolites such as equol, enterolignans and certain urolithins. These compounds exhibit stronger oestrogen-like activity due to their higher lipophilicity, which leads to a better absorption and a capacity to bind to ERs(Reference Gaya, Medina and Sánchez-Jiménez129,Reference Ruiz de la Bastida, Langa and Curiel130) . A meta-analysis of 543 potentially relevant studies examined the efficacy of phytoestrogens for the relief of menopausal symptoms showing their capability in reducing the frequency of hot flashes in menopausal women(Reference Chen, Liu and Liu131). Bifidobacteria adolescentis, which was shown to be reduced in postmenopausal women, has been identified as being efficient at metabolizing phytoestrogens into beneficial components that can act on ERs(Reference Bermingham, Linenberg and Hall18,Reference Senizza, Rocchetti and Mosele132) . Intake of phytoestrogen-rich foods can in turn influence microbial metabolism, as some in vitro works has shown. If cultured with quercetin, Bifidobacterium adolescentis exerts an enhanced anti-inflammatory activity, through the production of NO suppressor substances, suggesting a potential prebiotic role of isoflavones(Reference Kawabata, Sugiyama and Sakano133). The biosynthesis of these molecules represents a specific function of the gut microbiota in the presence of plant-based compounds highlighting the importance of diet and microbiome interactions.
Synbiotic products (targeted probiotic strains in combination with prebiotics) may have synergistic effects, enhance oestrogen metabolism, reduce inflammation and improve mood. Mustafa et al. found that the addition of inulin to B. longum BB536 and B. breve ATCC15700 significantly increases the production of equol during the fermentation of soy milk, thereby increasing the bioavailability of bioactive compounds and potentially improving nutritional value of soymilk(Reference Mustafa, Mustafa and Ismail134). A large cross-sectional study in the US found that the interaction effects of probiotics and prebiotics could alleviate depression, especially symptoms such as anhedonia, sleep problems, fatigue and appetite changes, although there was no direct comparison showing synbiotics being superior(Reference Ye, Yu and Yang135). However, a broader meta-analysis suggested that synbiotic supplements may offer stronger effects in reducing depressive symptoms via the gut-brain axis, compared to probiotics and prebiotics individually, which showed moderate or NS effect, respectively(Reference Dai, Chen and Chen136).
A limitation of current research on the microbiome in menopause is the lack of diversity in study groups, which limits the standardization of findings. The microbiome is known to be influenced by socioeconomic and cultural factors, such as lifestyle and nutrition that can vary significantly between Western and non-Western populations(Reference Mitchell, Srinivasan and Zhan137–Reference Shen, Wang and Zhou140). For instance, the intake of phytoestrogens through food is varied in different regions of the world due to changes in local diet: in Asian countries where fermented soy products are part of the traditional diet, isoflavone intake levels may aggregate to about 15–50 mg per day, but in the Western countries, the isoflavone intake can be less than 2 mg isoflavones per day(Reference Eisenbrand141). Moreover, the ability to produce equol varies among individuals due to differences in the composition of intestinal microbiota.
Conclusion
Despite living longer, women face unique health challenges as they age. Menopause is an important time of transition, marked by a drop in circulating oestrogens associated with the permanent cessation of ovarian activity alongside numerous physiological and psychological symptoms. Therefore, it is essential to address sex-specific health needs and promote customized strategies to improve the well-being and quality of life of aging women. In recent years, two key biological systems have gained prominence in understanding menopausal health: the estrobolome, a subset of the gut microbiome involved in oestrogen metabolism and the gut–brain axis, the bidirectional communication system linking the gastrointestinal tract with the CNS. Emerging evidence suggests that the interplay between these systems significantly influences menopausal symptomatology and long-term health outcomes. By understanding the role of the oestrobolome and supporting it with psychobiotic supplementations women can take an active role in maintaining hormonal balance and overall wellness. While probiotic supplementation has gained increasing attention as a strategy to alleviate menopausal symptoms via modulation of the gut microbiota, current evidence suggests that changing eating habits might be even more sustainable and effective. The available commercial probiotic formulations typically contain only a limited set of probiotic strains and are often not tested in the target population and although these can exert beneficial effects, they cannot substitute for the complexity of a natural diverse microbiome. Moreover, the variation observed among individuals represents a critical consideration. Each woman shows a unique microbiome shaped by genetics, early-life exposures, lifestyle and dietary patterns. Consequently, the response to any supplementation is very heterogeneous. By fostering a diverse microbial ecosystem through diet, it is possible to achieve synergistic effects to improve mood and responses to the stresses of everyday life through the gut-brain axis. Diets rich in plant-based fibres, polyphenols and fermented foods promote the growth of a wide spectrum of commensals, many of which can produce SCFAs, regulating oestrogen metabolism through the oestrobolome, and modulating neuroinflammation along the gut–brain axis. Overall, understanding the interplay between nutrition and the microbiome is crucial for developing more personalized approaches focused on tailoring dietary interventions to the individual’s microbial profile rather than relying on standardized supplementation.
Author contributions
M.C. and E.G. contributed to the writing, editing and finalization of the manuscript. E.P. contributed to the editing of the manuscript. C.O. realized the figures and contributed to the editing of the manuscript. S.O.M. supervised the project and contributed to the editing of the manuscript. All authors read and approved the final version of the manuscript.
Financial support
Enterprise Ireland Commercialisation Fund - CF-2024-2330-1.
Competing interests
The authors have no conflicts of interest to declare.