Colorectal cancer (CRC) currently stands as the second most significant contributor to cancer-related deaths globally(Reference Ferlay, Ervik and Lam1). During the year 2020, the total number of individuals diagnosed with CRC exceeded 1·9 million, and among them, 935 173 individuals unfortunately succumbed to the disease within the same year(Reference Ferlay, Ervik and Lam1). Apart from the established risk factors of adenomas and CRC development, such as ageing and family history of CRC, several epidemiological studies have also presented that chronic inflammation plays an essential role in initiation, progression and promotion of CRC(Reference Brenner, Kloor and Pox2,Reference Taylor, Burt and Williams3) . Evidence has demonstrated that chronic inflammation operates by promoting cancer cell growth, inhibiting cellular differentiation and decreasing cell survival(Reference Grivennikov4). In this framework, several molecular pathways work together to make this happen(Reference Guina, Biasi and Calfapietra5). The primary manifestation of them is the release of pro-inflammatory cytokines in the blood circulation and tissues, such as IL-1, IL-6, IL-8 and TNF-α(Reference Grivennikov4). The aforementioned pattern further underlines the importance of modifiable risk factors in the development of CRC. In this context, adherence to unhealthy dietary patterns, sedentary lifestyle, smoking and alcohol intake are generally recognised as potential triggers of chronic systemic inflammation, which consequently promotes cancer cell proliferation(Reference Hofseth, Hebert and Chanda6,Reference Gibson, Prochaska and Yu7) . Molecular investigations have also revealed that adherence to unhealthy dietary patterns leads to the development of pro-inflammatory cytokines and other free radicals at the cellular level(Reference Murphy, Moreno and Hughes8,Reference Seiwert, Heylmann and Hasselwander9) .
According to the latest report of the World Cancer Research Fund (WCRF)/American Institute for Cancer Research (AICR) cancer prevention dietary recommendations, greater adherence to dietary components-induced inflammation, including red and processed meat, sugar-sweetened and alcoholic drinks, and lower consumption of anti-inflammatory foods, including fruits, vegetables and beans, were associated with a higher risk of CRC developing(10). Within this particular framework, several inflammatory potential of diets that are based on literature or empirically derived dietary patterns, including the Dietary Inflammatory Index (DII), the Empirical Dietary Inflammatory Pattern (EDIP), the Inflammatory Score of Diet (ISD) and the Dietary Inflammation Score (DIS), were established for assessing an individual’s dietary inflammatory potential(Reference Bahr, Franz and Mähler11). In 2022, a meta-analysis on the influence of DII and CRC was published, indicating that greater adherence to DII was significantly associated with higher risk of CRC by 16 per cent(Reference Syed Soffian, Mohammed Nawi and Hod12). The number of studies included, however, was limited and solely concentrated on investigating the impact of DII in the development of CRC. To the best of our knowledge, no systematic review and meta-analysis evaluated the association between inflammatory potential of diets and the risk of CRC. Therefore, we aimed to perform the current study aiming to find a more precise and quantitative estimate for the association between inflammatory potential of diets and the risk of CRC.
Method
Research registry and standard guidelines
The current study has been officially registered on the PROSPERO database (registration ID: CRD42023495108), and it has been carried out in compliance with the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines(Reference Stroup, Berlin and Morton13). In order to perform a comprehensive systematic review of the existing literature, one researcher (AN) employed the electronic databases of Scopus, PubMed/MEDLINE and Web of Science (WOS). An electronic systematic search was performed until 21 November 2023, with no time limitation, and was limited to publications written in English. The details of strategies employed for conducting electronic systematic database searches are outlined in online Supplementary Table 1. In addition, one researcher (EE) performed a manual reference search of all included studies as well as published relevant systematic reviews and meta-analyses to ensure that no potentially relevant articles were overlooked.
Inclusion criteria
Publications were incorporated into our systematic review and meta-analysis if they satisfied the following criteria: (1) English language studies conducted on adults aged 18 years and older; (2) observational studies encompassing the following design: cohort (retrospective or prospective), case–control and cross-sectional; (3) studies that have considered adherence to priori-defined inflammatory potential of diet scores (according to the definition provided by Bahr LS et al. (Reference Bahr, Franz and Mähler14)) as the exposure of our interest; (4) studies that have considered the total or site-specific CRC (total CRC, colon, proximal, distal and rectal cancer) cases as an outcome of our interest; and (5) studies that provided the adjusted estimates (including OR, hazard ratio or relative risk, with corresponding 95 % CI, as the effect size (ES) for the association between adherence to inflammatory potential of diets and the risk of total or site-specific CRC.
Exclusion criteria
Publications were excluded from our study if there were (1) written in a language other than English, (2) review publications, (3) interventional studies, (4) case reports, case series, editorials, commentaries, notes, letters with insufficient data and conference abstracts, (5) animal studies or in vivo and in vitro experiments, (6) performed on newborns, children, adolescents, pregnant mothers and breast-feeding women, (7) not accessible in full-text format and (8) insufficient data (without relevant ES and 95 % CI for the association between adherence to inflammatory potential of diets and the risk of total or site-specific CRC).
Quality assessment
The present systematic review and meta-analysis utilised the Newcastle–Ottawa Scale (NOS) to assess the quality of included studies(Reference Stang15). The NOS was developed specifically for evaluating the quality of non-randomised studies. According to this scale, the quality of included studies is assessed using the star system, which is determined by three criteria as follows: (1) selection of the study groups (consisting of four items); (2) comparability of the groups (consisting of two items) and (3) assessment of either the exposure or outcome of interest for case–control or cohort studies, respectively (three consisting of three items). Every item can receive a maximum of one star, except for the comparability item, which can receive a maximum of two stars. Publications scoring 7 or higher were designated as high quality and low risk of bias, while those scoring below 7 were classified as low quality and high risk of bias publications.
Data extraction
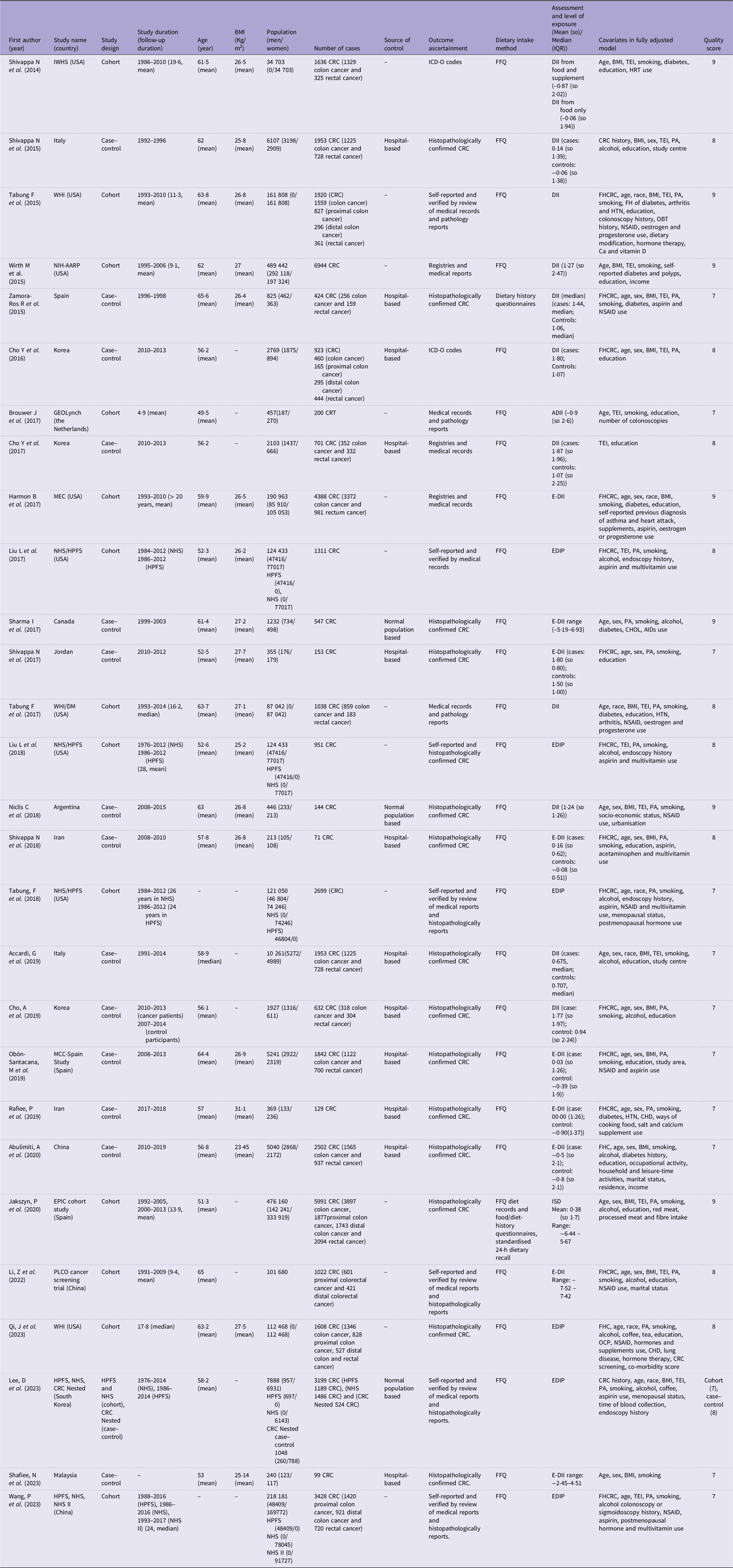
Two researchers (FJ and MS) conducted an extensive evaluation of each eligible study independently. The data extracted from these studies are as follows: first author’s name, year of publication, study name, the country where the study was conducted, study duration, follow-up duration, mean/median/range of age and BMI of participants, number of participants (total and sex-specific), number of cases (total or site-specific CRC), source of control (for case–control studies), outcome ascertainment method, dietary intake method, inflammatory potential of diet scoring method with corresponding mean (sd)/median (IQR) values and variables adjusted in the multivariate analysis. In addition, any inconsistencies were resolved through discussion with a senior author supervising (AN).
Statistical analysis
Descriptive analysis was used to summarise the characteristics of each publication and the demographic details of the participants. In order to initiate the calculation of the pooled ES estimate for the risk of total and site-specific CRC, we assigned relative risk with a corresponding 95 % CI as the ES for the present investigation. The OR obtained from cross-sectional and case–control studies and hazard ratio obtained from cohort studies were considered the equivalent to relative risk(Reference Symons and Moore16). A meta-analysis was conducted when at least two studies provided the ES for the same outcome. The pooled ES of total and site-specific CRC risk were calculated for the highest compared with the lowest adherence as well as per-sd increases in adherence to inflammatory potential of diets using the DerSimonian and Laird random effects model, which accounts for variation within and between studies (heterogeneity)(Reference Jackson, White and Thompson17). The Cochrane Q test (P-heterogeneity)(Reference Cumpston, Li and Page18) and the I2 statistics(Reference Higgins and Thompson19) were used to evaluate the total variation that was attributed to heterogeneity between studies. When heterogeneity was presented, the threshold for determining the significance level of Cochran Q was set at P< 0·10(Reference Cumpston, Li and Page18). Based on the I2 metrics, heterogeneity levels of 25 %, 50 % and 75 % correspond to low, medium and high degrees of heterogeneity, respectively(Reference Higgins, Thompson and Deeks20). In order to identify sources of heterogeneity, we also conducted subgroup and meta-regression analysis by study design (cohort/case–control), region (USA, Europe and Asia), age (< 65 years/> 65 years), sex (men/women), duration of follow-up for cohort studies (< 10 years/> 10 years), number of participants (< 10 000/> 10 000), inflammatory potential of diet calculation (food only/food and supplement), BMI (normal weight/overweight or obese) and smoking status (never/ever). Sensitivity analysis was carried out to assess the robustness of the findings and determine whether single or multiple publications influenced the final pooled ES using the one-study exclusion (leave-one-out) method. When there were at least ten publications included in our analysis, the evaluation of publication bias was performed by visually examining the funnel plot and utilising Egger’s regression test. The statistical analyses were conducted using STATA 14.0 software developed by Stata Corporation in College Station, Texas, USA. A P-value below 0·05 was considered to be statistically significant.
Results
Study selection
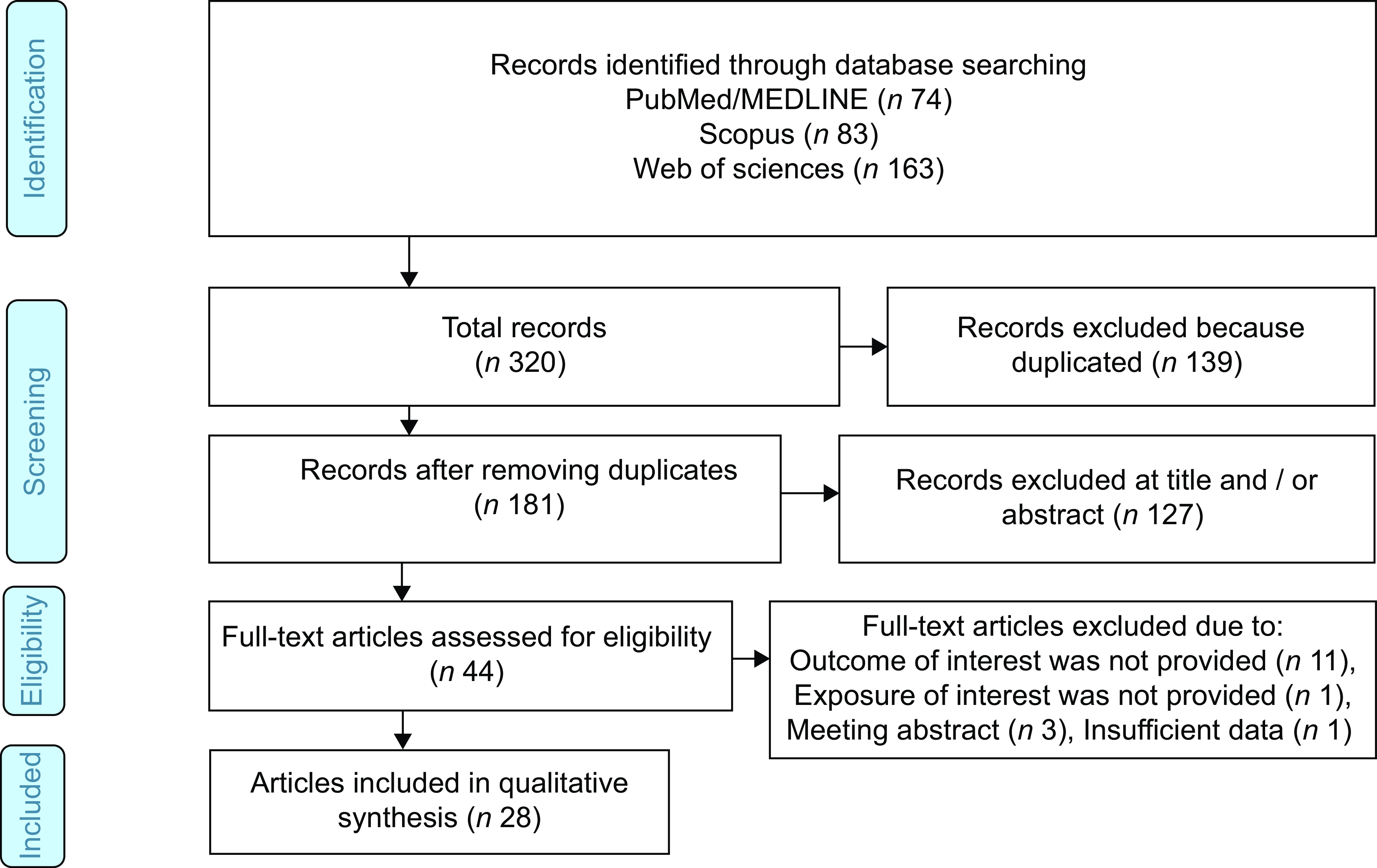
Figure 1 provides the process of literature search and selection. A total of 320 articles from three databases including PubMed/MEDLINE (n 74), Scopus (n 83) and ISI Web of sciences (n 163) were identified. After removal of duplicated publications (n 139), excluding ineligible studies determined by title and abstract screening (n 127), forty-four publications remained for full-text reviewing. In the following for full-text evaluation, eleven publications were excluded since outcome of interest was not provided, one publication for not providing exposure of interest, three publications for being meeting abstract and one publication for insufficient data. Finally, twenty-eight publications were included in the current systematic review and meta-analysis(Reference Shivappa, Prizment and Blair21–Reference Wang, Song and Eliassen45). The characteristics of eligible studies are presented in Table 1. All included publication were published between 2014 and 2023. Eleven studies were performed in Asia including five in Korea(Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Cho, Lee and Oh37,Reference Jin, Shi and Lee43) , three in China(Reference Abulimiti, Zhang and Shivappa40,Reference Li, Wang and Shivappa42,Reference Wang, Song and Eliassen45) , two in Iran(Reference Niclis, Pou and Shivappa34,Reference Rafiee, Shivappa and Hébert39) , one in Jordan(Reference Sharma, Zhu and Woodrow31) and one in Malaysia(Reference Shafiee, Razalli and Shahril44). Eleven studies were conducted in America including nine studies in the USA(Reference Shivappa, Prizment and Blair21,Reference Tabung, Steck and Ma23,Reference Wirth, Shivappa and Steck24,Reference Harmon, Wirth and Boushey29,Reference Liu, Nishihara and Qian30,Reference Tabung, Steck and Ma32,Reference Liu, Tabung and Zhang33,Reference Tabung, Liu and Wang35,Reference Jin, Shi and Lee43) , one in Canada(Reference Sharma, Zhu and Woodrow31) and one in Argentina(Reference Niclis, Pou and Shivappa34). Six studies were implemented in Europe including one multinational(Reference Jakszyn, Cayssials and Buckland41), two in Italy(Reference Shivappa, Zucchetto and Montella22,Reference Accardi, Shivappa and Di Maso36) , two in Spain(Reference Zamora-Ros, Shivappa and Steck25,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38) and one in the Netherlands(Reference Brouwer, Makama and van Woudenbergh27). Of twenty-eight included studies, design of fourteen studies were cohort(Reference Shivappa, Prizment and Blair21,Reference Tabung, Steck and Ma23,Reference Wirth, Shivappa and Steck24,Reference Brouwer, Makama and van Woudenbergh27,Reference Harmon, Wirth and Boushey29,Reference Liu, Nishihara and Qian30,Reference Tabung, Steck and Ma32,Reference Liu, Tabung and Zhang33,Reference Tabung, Liu and Wang35,Reference Jakszyn, Cayssials and Buckland41–Reference Jin, Shi and Lee43,Reference Wang, Song and Eliassen45) and fourteen studies were case–control(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Sharma, Zhu and Woodrow31,Reference Niclis, Pou and Shivappa34,Reference Accardi, Shivappa and Di Maso36–Reference Abulimiti, Zhang and Shivappa40,Reference Shafiee, Razalli and Shahril44) . Study duration of included studies ranged from 1976 to 2019, and follow-up duration ranged from 4·9 to over 20 years. The age and BMI value of the participants were ranged from 49·5(Reference Brouwer, Makama and van Woudenbergh27) to 65·6(Reference Zamora-Ros, Shivappa and Steck25) years old and 23·45(Reference Abulimiti, Zhang and Shivappa40) to 31·1(Reference Rafiee, Shivappa and Hébert39) kg/m2, respectively. These publications enrolled 2 287 836 participants (732 312 men and 1 453 844 women), ranging from 213(Reference Niclis, Pou and Shivappa34) to 489 442(Reference Wirth, Shivappa and Steck24). Among twenty-eight studies, a total of 50 418 CRC, 18 885 colon, 5718 proximal colon, 4268 distal colon and 9523 rectal cancer cases were recorded. Sources for control group among the fourteen case–control studies were hospital-based in twelve studies(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Sharma, Zhu and Woodrow31,Reference Niclis, Pou and Shivappa34,Reference Accardi, Shivappa and Di Maso36–Reference Abulimiti, Zhang and Shivappa40,Reference Shafiee, Razalli and Shahril44) and normal population-based in two studies(Reference Sharma, Zhu and Woodrow31,Reference Niclis, Pou and Shivappa34) . The total or site-specific CRC were ascertained by histopathologically confirmed diagnosis method among the majority of studies. Most of the studies used FFQ as the method for evaluating dietary intake. Inflammatory potential of diet was assessed through DII in eleven studies(Reference Shivappa, Prizment and Blair21–Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Tabung, Steck and Ma32,Reference Niclis, Pou and Shivappa34,Reference Accardi, Shivappa and Di Maso36,Reference Cho, Lee and Oh37) , Energy-adjusted Dietary Inflammatory Index (E-DII) in ten studies(Reference Brouwer, Makama and van Woudenbergh27,Reference Harmon, Wirth and Boushey29,Reference Sharma, Zhu and Woodrow31,Reference Niclis, Pou and Shivappa34,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38–Reference Abulimiti, Zhang and Shivappa40,Reference Li, Wang and Shivappa42,Reference Shafiee, Razalli and Shahril44) , EDIP in six studies(Reference Liu, Nishihara and Qian30,Reference Liu, Tabung and Zhang33,Reference Tabung, Liu and Wang35,Reference Jin, Shi and Lee43,Reference Wang, Song and Eliassen45) and ISD in one study(Reference Jakszyn, Cayssials and Buckland41). Most included studies adjusted for age, BMI, smoking and family history of CRC. Online Supplementary Table 2 represents the quality assessment of the eligible studies. According to the NOS score, all the eligible studies were high in quality.

Figure 1. Flow chart of the included studies.
Table 1. Characteristics of included studies (Mean values and standard deviations; median values and interquartile ranges)
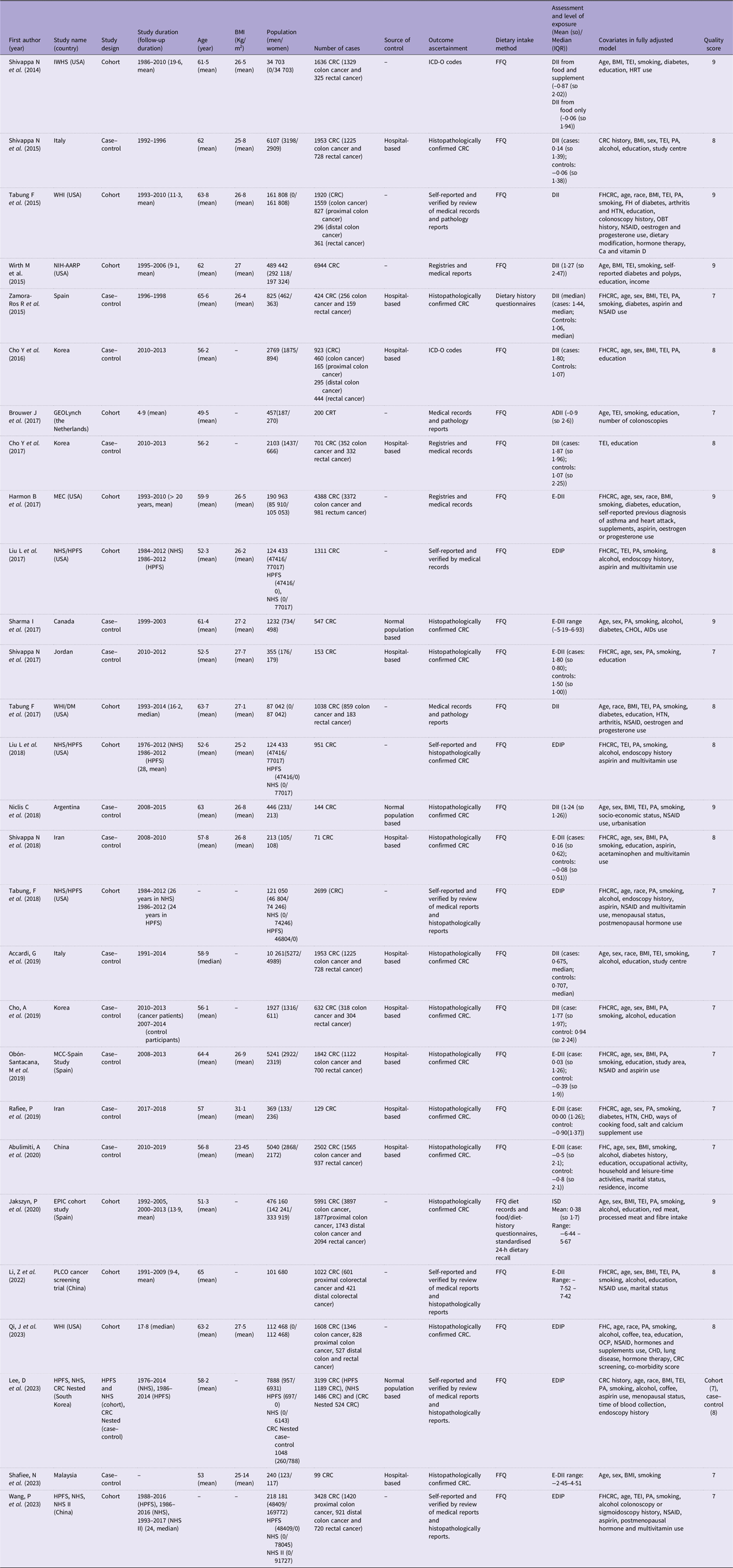
IWHS, Iowa Women Health Study; CRC, colorectal cancer; ICD-O, International Classification of Diseases for Oncology; DII, Dietary Inflammatory Index; TEI, total energy intake; HRT, hormone replacement therapy; PA, physical activity; WHI, Women’s Health Initiative; CHOL, cholesterol; AID, anti-inflammatory drugs; FHCRC: Family History of Colorectal Cancer; HTN, hypertension; OBT, occult blood tests; NSAID, non-steroidal anti-inflammatory drugs; NIH-AARP, National Institutes of Health-American Association of Retired Persons Diet and Health Study; GEOLynch, Genetic, Environmental and Other factors that influence tumour risk in persons with LS study; ADII, Adapted Dietary Inflammatory Index; MEC, multiethnic cohort; E-DII, Energy-adjusted Dietary Inflammatory Index; NHS/HPFS, Nurses’ Health Study/Health Professionals Follow-Up Study; WHI/DM, Women’s Health Initiative/WHI Dietary Modification Trial; FHC, family history of cancer; EPIC cohort study, European Prospective Investigation into Cancer and Nutrition cohort study; ISD, the Inflammatory Score of Diet; PLCO Cancer Screening Trial, Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial; OCP, oral contraceptive; EDIP, Empirical Dietary Inflammatory Pattern; MCC-Spain, Multi Case-Control Study in Spain.
Inflammatory potential of diet and the risk of colorectal cancer
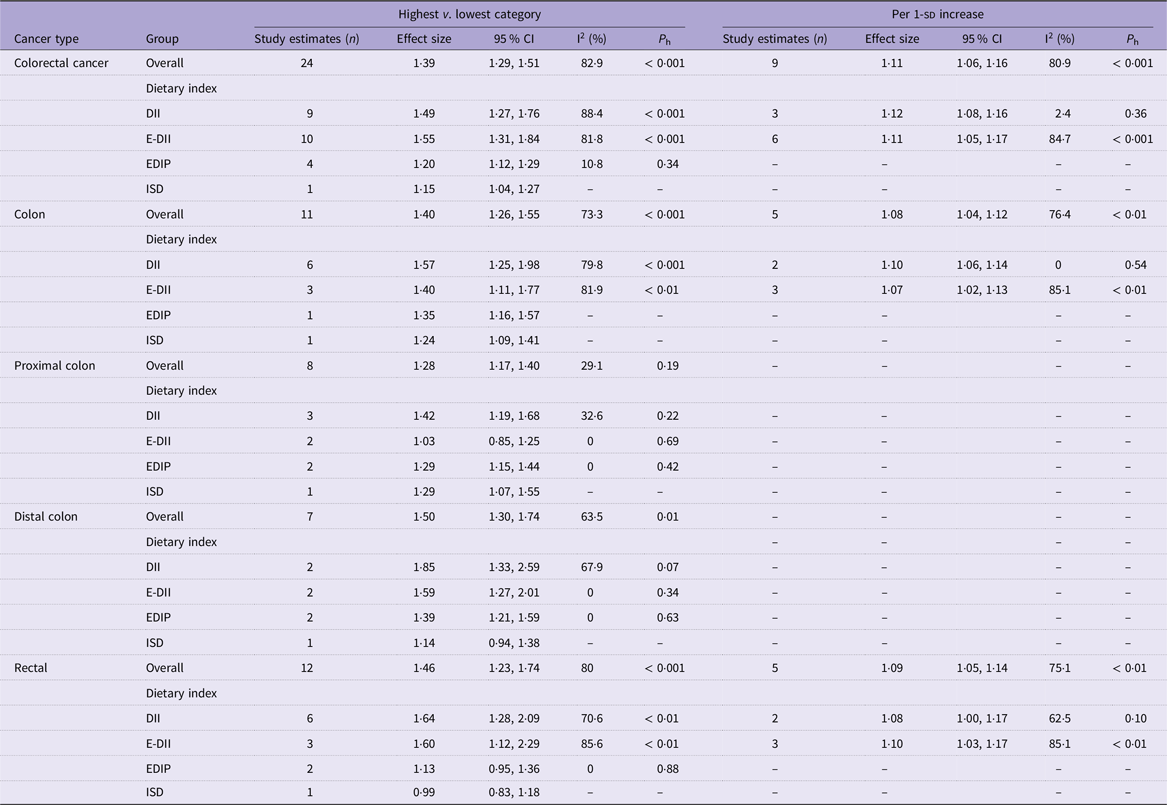
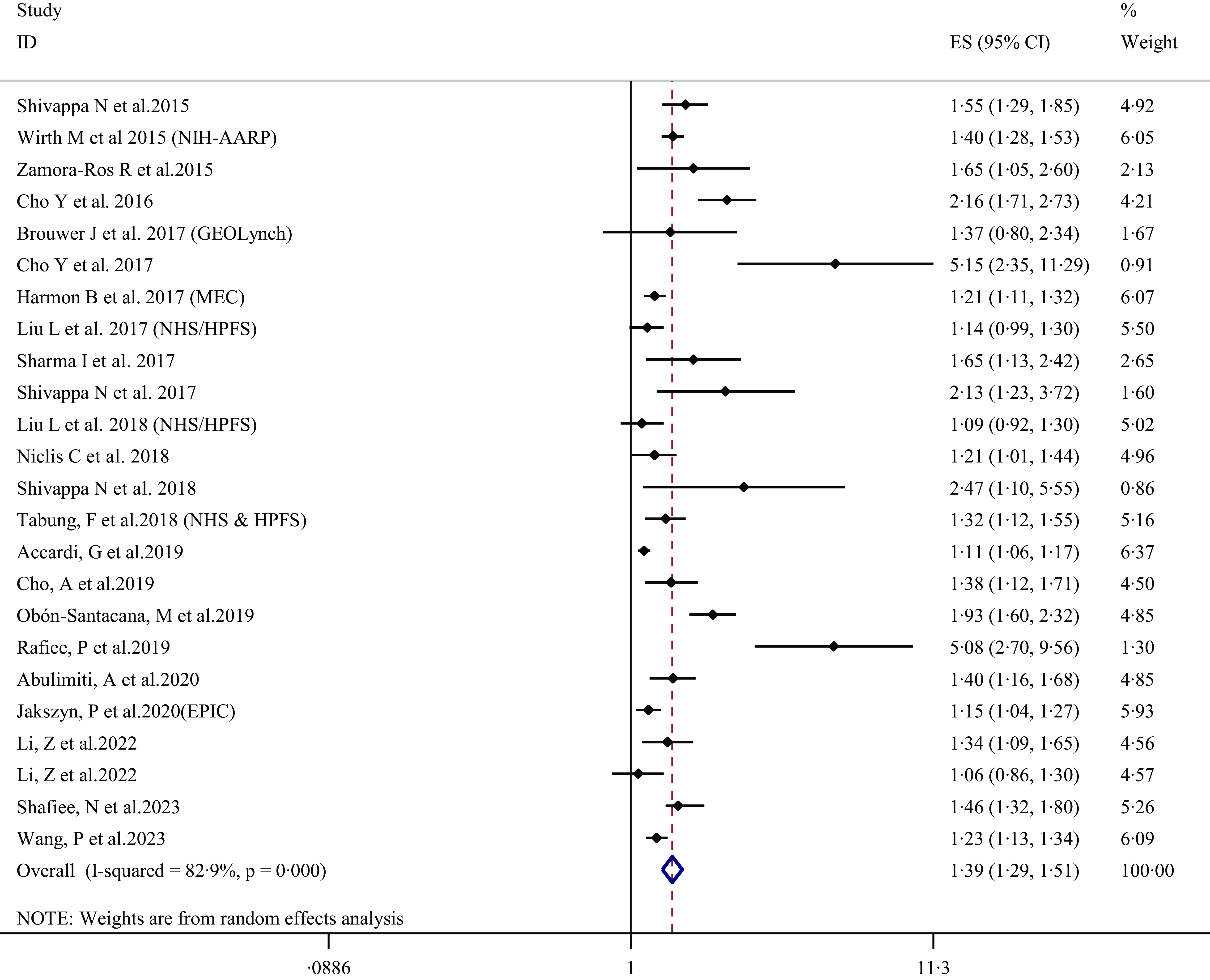
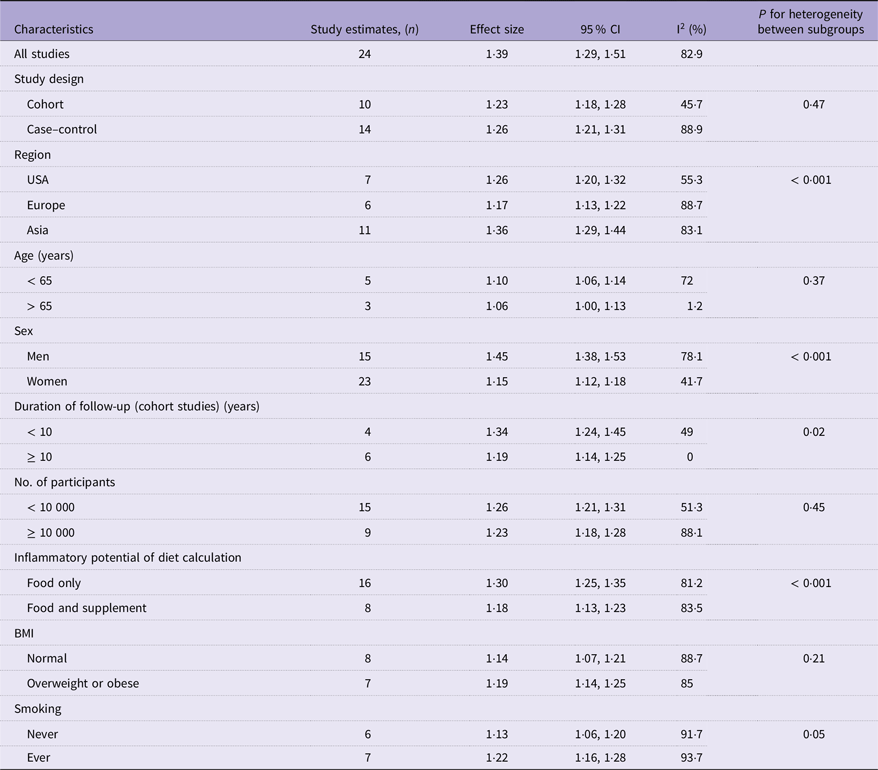
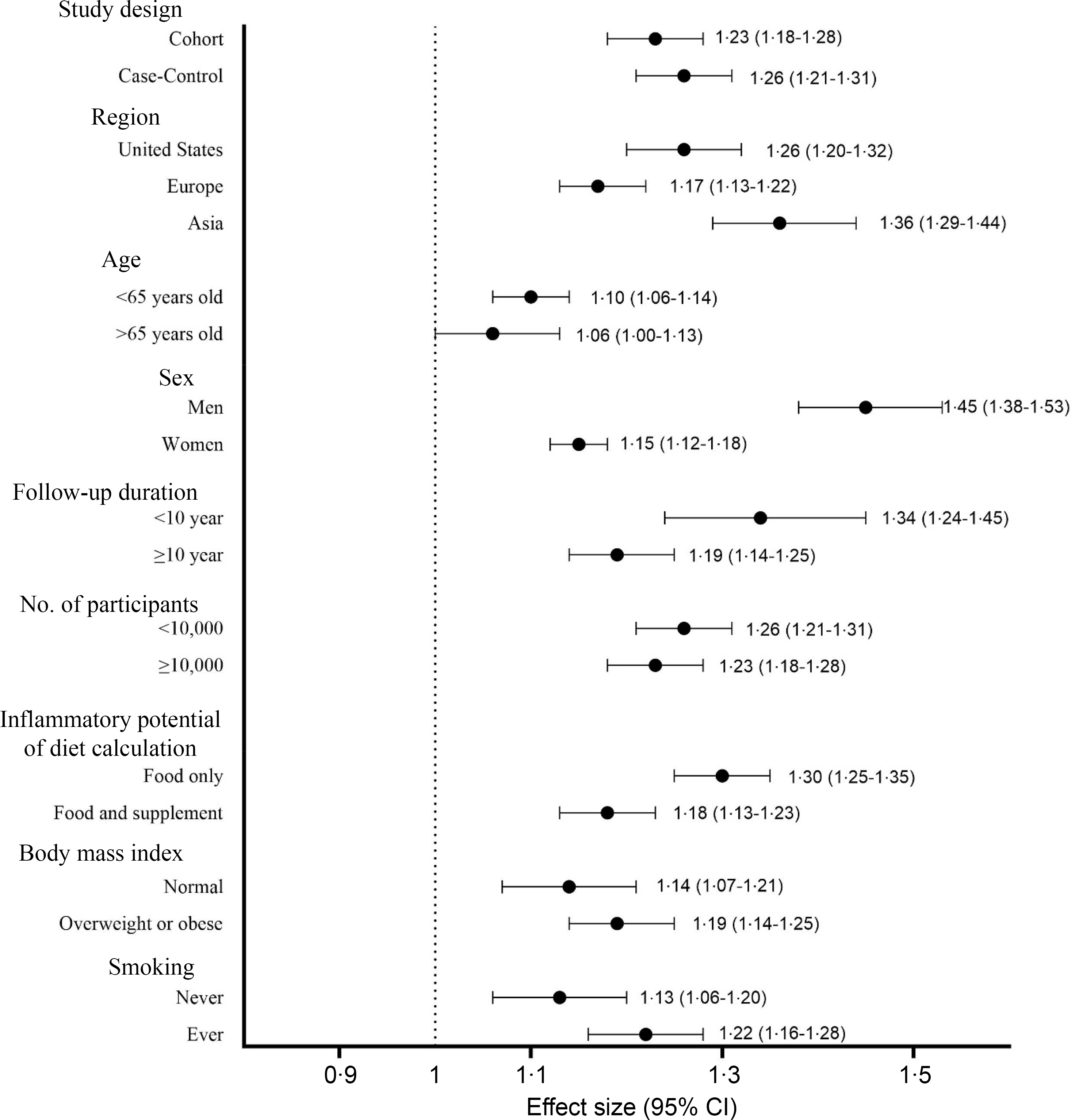
Table 2 presents the pooled multi-variable-adjusted ES from the random effect meta-analysis of the inflammatory potential of diet and the risk of CRC. Twenty-four ES from twenty-three publications(Reference Shivappa, Zucchetto and Montella22,Reference Wirth, Shivappa and Steck24–Reference Sharma, Zhu and Woodrow31,Reference Liu, Tabung and Zhang33–Reference Li, Wang and Shivappa42,Reference Shafiee, Razalli and Shahril44,Reference Wang, Song and Eliassen45) were included in the meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of CRC. The summary ES was 1·39 (95 % CI 1·29, 1·51), with a high degree of heterogeneity (I2 = 82·9 %; P-heterogeneity < 0·001) (Figure 2 and Table 2). Subgroup analysis revealed that region, sex, duration of follow-up (in cohort studies) and inflammatory potential of diet calculation method were all potential sources of heterogeneity (Table 3). As well, increased CRC risk remained significant after stratified by study design (cohort and case–control), region (America, Europe and Asia), age (< 65 years and > 65 years), sex (men and women), duration of follow-up (in cohort studies, < 10 years and ≥ 10 years), number of participants (< 10 000 and ≥ 10 000), inflammatory potential of diet calculation method (food only, food and supplement), BMI (normal, overweight or obese) and smoking (never and ever) (Figure 3 and Table 3). Similar elevated CRC risk was observed when pooling ES from the highest compared with the lowest adherence to different dietary indices, separately for DII (ES: 1·49, 95 % CI 1·27, 1·76; I2 = 88·4 %), for E-DII (ES: 1·55, 95 % CI 1·31, 1·84; I2 = 81·8 %) and for EDIP (ES: 1·20, 95 % CI 1·12, 1·29; I2 = 10·8 %) (Table 2).
Table 2. The association between inflammatory potential of diet and risk of colorectal cancer (95 % confidence intervals)

DII, Dietary Inflammatory Index; E-DII, Energy-adjusted Dietary Inflammatory Index; EDIP, Empirical Dietary Inflammatory Pattern; ISD, the Inflammatory Score of Diet.

Figure 2. Forest plots with overall multi-variable-adjusted effect sizes from the random effects meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of colorectal cancer. ES, effect size; NIH-AARP, National Institutes of Health-American Association of Retired Persons Diet and Health Study; GEOLynch, Genetic, Environmental and Other factors that influence tumour risk in persons with LS study; MEC, multiethnic cohort; NHS/HPFS, Nurses’ Health Study/Health Professionals Follow-Up Study; EPIC, European Prospective Investigation into Cancer and Nutrition.
Table 3. Subgroup analysis of the association between inflammatory potential of diet and risk of colorectal cancer (95 % confidence intervals)

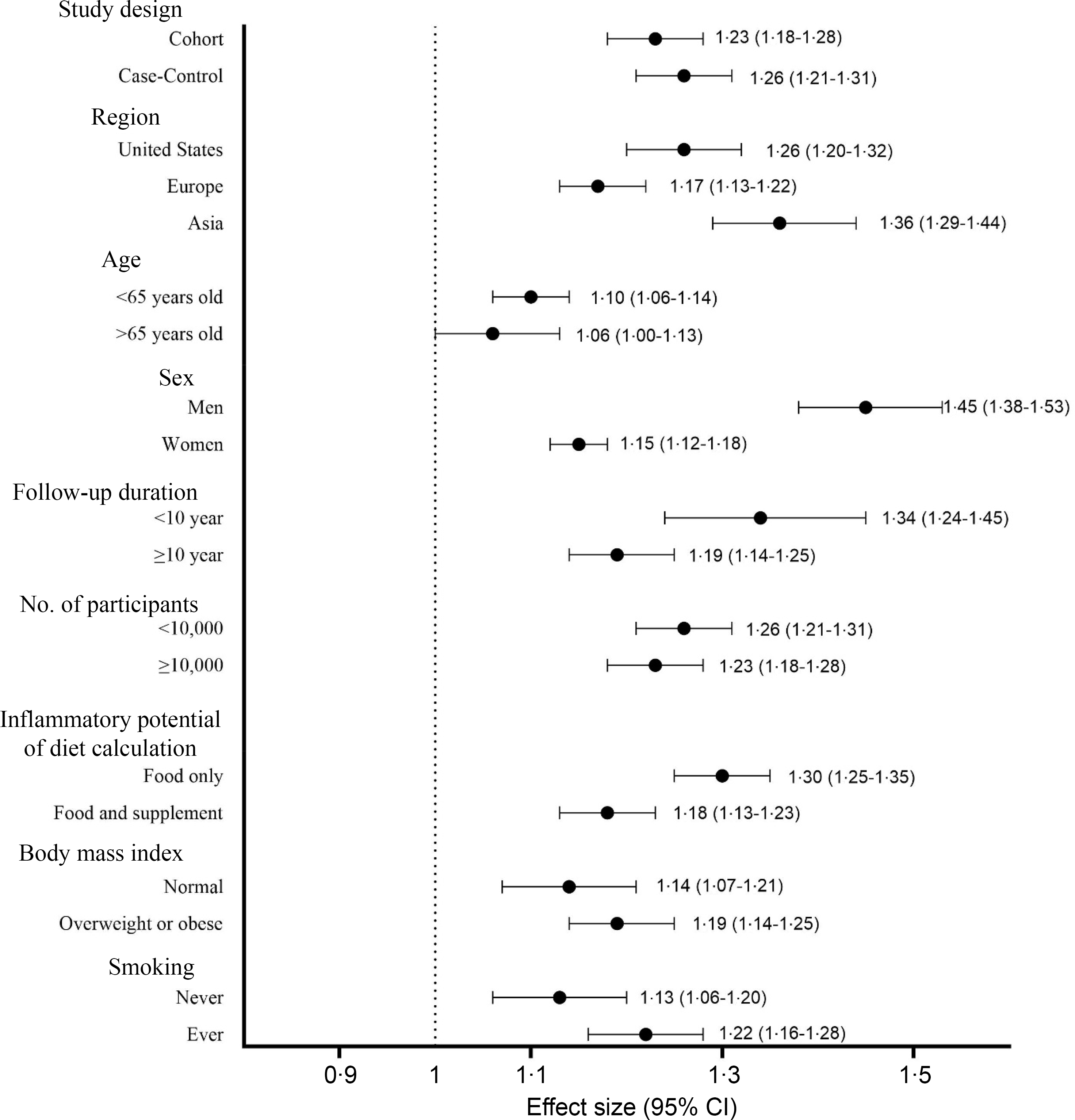
Figure 3. Subgroup analysis of the highest compared to the lowest categories of the inflammatory potential of diet and the risk of colorectal cancer.
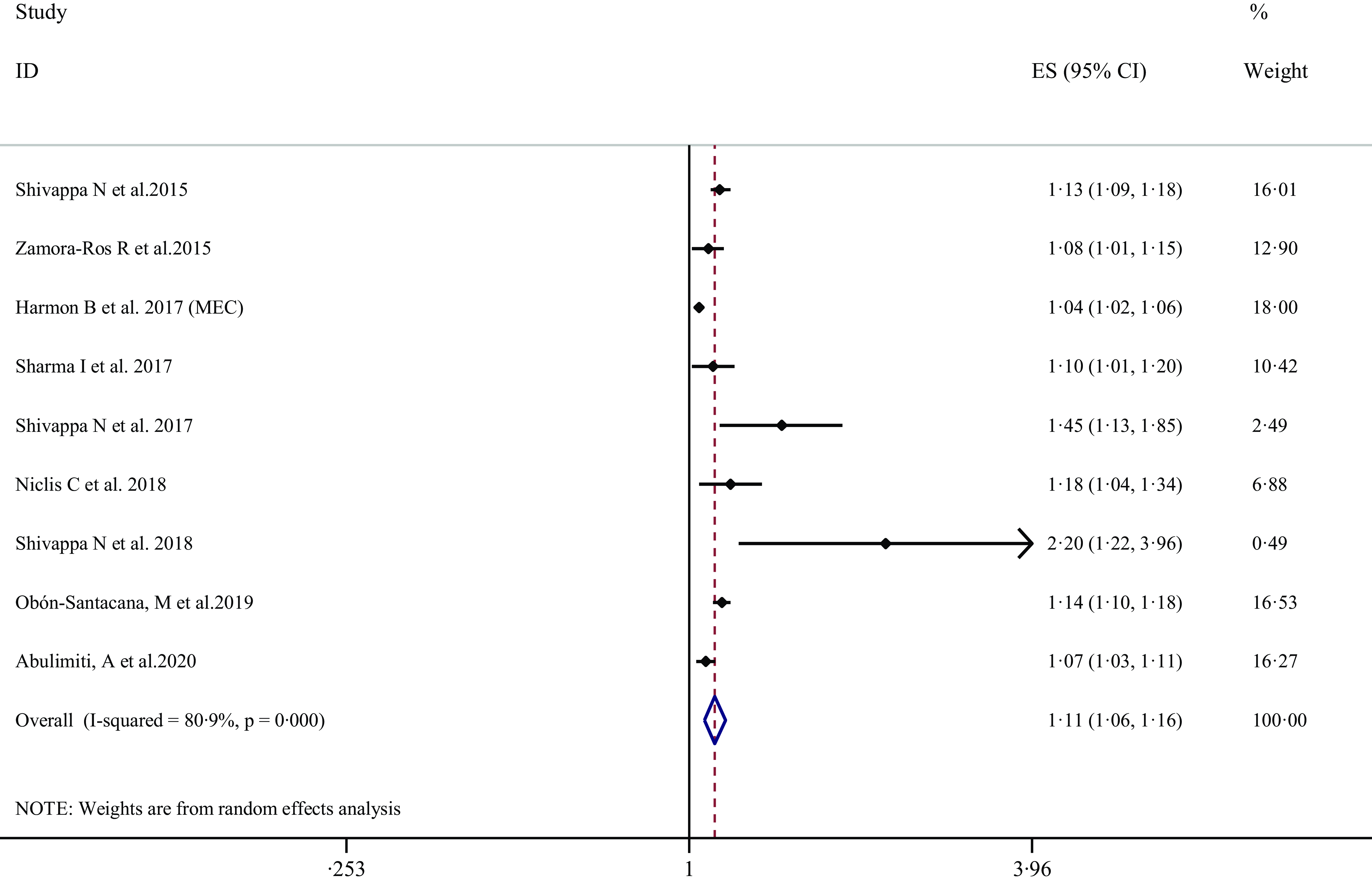
Nine ES from nine publications(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Harmon, Wirth and Boushey29,Reference Sharma, Zhu and Woodrow31,Reference Niclis, Pou and Shivappa34,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40) were included in the meta-analysis of the association between 1-per sd increases in inflammatory potential of diet and the risk of total CRC. The summary ES was 1·11 (95 % CI 1·06, 1·16), with a high degree of heterogeneity (I2 = 80·9 %; P-heterogeneity < 0·001) (Figure 4 and Table 2). Following pooling ES from different dietary indices separately, similar elevated total CRC risk was observed for DII (ES: 1·12, 95 % CI 1·08, 1·16; I2 = 2·4 %) and for E-DII (ES: 1·11, 95 % CI 1·05, 1·17; I2 = 84·7 %) (Table 2).
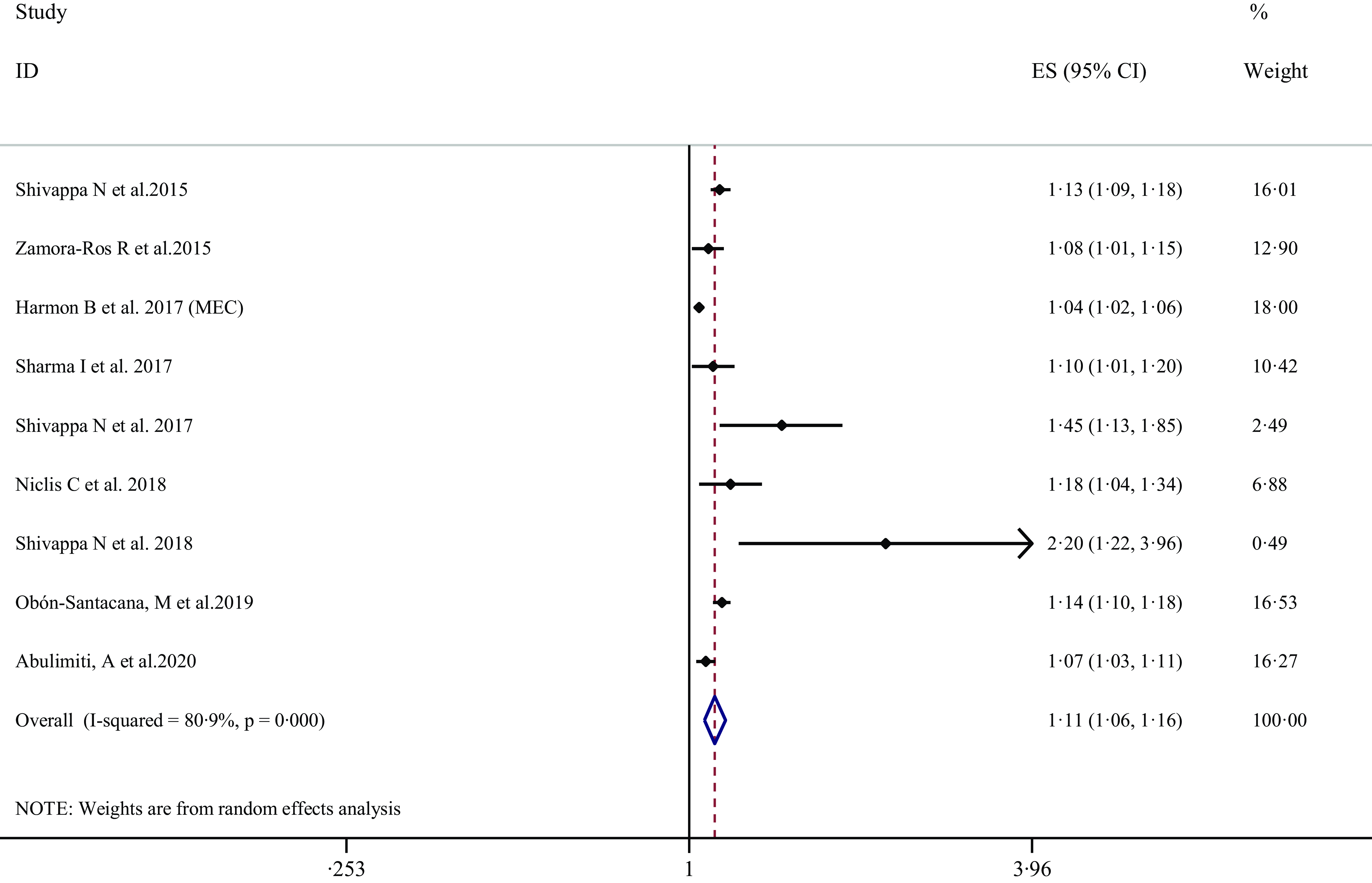
Figure 4. Forest plots with overall multi-variable-adjusted effect sizes from the random-effects meta-analysis of the association between 1-per sd increases in inflammatory potential of diet and the risk of colorectal cancer. ES, effect size; MEC, multiethnic cohort.
Inflammatory potential of diet and the risk of site-specific colorectal cancer
Colon cancer risk
The pooled multi-variable-adjusted ES from the random effect meta-analysis of the inflammatory potential of diet and the risk of site-specific CRC are provided in Table 2. Eleven ES from eleven publications(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Harmon, Wirth and Boushey29,Reference Tabung, Liu and Wang35–Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40,Reference Jakszyn, Cayssials and Buckland41) were included in the meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of colon cancer. The summary ES was 1·40 (95 % CI 1·26, 1·55; I2 = 73·3 %) (Table 2). Similar elevated colon cancer risk was found after polling ES from the highest compared with lowest adherence to DII (ES: 1·57, 95 % CI 1·25, 1·98; I2 = 79·8 %) and E-DII (ES: 1·40, 95 % CI 1·11, 1·77; I2 = 81·9 %) (Table 2).
Five ES from five publications(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Harmon, Wirth and Boushey29,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40) were included in the meta-analysis of the association between 1-per sd increases in inflammatory potential of diet and the risk of colon cancer (Table 2). The summary ES was 1·08 (95 % CI 1·04, 1·12; I2 = 76·4 %). Following pooling ES from different dietary indices separately, similar elevated colon cancer risk was observed for DII (ES: 1·10, 95 % CI 1·06, 1·14; I2 = 0 %) and for E-DII (ES: 1·07, 95 % CI 1·02, 1·13; I2 = 85·1 %) (Table 2).
Proximal colon cancer risk
Eight ES from six publications(Reference Wirth, Shivappa and Steck24,Reference Cho, Lee and Oh26,Reference Tabung, Liu and Wang35,Reference Jakszyn, Cayssials and Buckland41,Reference Li, Wang and Shivappa42,Reference Wang, Song and Eliassen45) were included in the meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of proximal colon cancer. The summary ES was 1·28 (95 % CI 1·17, 1·40; I2 = 29·1 %) (Table 2). Similar increased proximal colon cancer risk was found after pooling ES from the highest compared with the lowest adherence to DII (ES: 1·42, 95 % CI 1·19, 1·68; I2 = 32·6 %) and EDIP (ES: 1·29, 95 % CI 1·15, 1·44; I2 = 0 %), while no significant association was found for E-DII (ES: 1·03, 95 % CI 0·85, 1·25; I2 = 0 %) (Table 2).
Distal colon cancer risk
Seven ES from six publications(Reference Wirth, Shivappa and Steck24,Reference Cho, Lee and Oh26,Reference Tabung, Liu and Wang35,Reference Jakszyn, Cayssials and Buckland41,Reference Li, Wang and Shivappa42,Reference Wang, Song and Eliassen45) were included in the meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of distal colon cancer. The summary ES was 1·50 (95 % CI 1·30, 1·74; I2 = 63·5 %) (Table 2). Similar increased distal colon cancer risk was found when pooling ES from the highest compared with the lowest adherence to different dietary potential of diet indices, separately for DII (ES: 1·85, 95 % CI 1·33, 2·59; I2 = 67·9 %), for E-DII (ES: 1·59, 95 % CI 1·27, 2·01; I2 = 0 %) and for EDIP (ES: 1·39, 95 % CI 1·21, 1·59; I2 = 0 %) (Table 2).
Rectal cancer risk
Twelve ES from twelve publications(Reference Shivappa, Zucchetto and Montella22,Reference Wirth, Shivappa and Steck24–Reference Cho, Lee and Oh26,Reference Cho, Lee and Oh28,Reference Harmon, Wirth and Boushey29,Reference Tabung, Liu and Wang35,Reference Cho, Lee and Oh37,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40,Reference Jakszyn, Cayssials and Buckland41,Reference Wang, Song and Eliassen45) were included in the meta-analysis of the highest compared with the lowest categories of inflammatory potential of diet and the risk of rectal cancer. The summary ES was 1·46 (95 % CI 1·23, 1·74; I2 = 80 %) (Table 2). Similar increased rectal cancer risk was found after pooling ES from the highest compared with the lowest adherence to DII (ES: 1·64, 95 % CI 1·28, 2·09; I2 = 70·6 %) and E-DII (ES: 1·60, 95 % CI 1·12, 2·29; I2 = 85·6 %), although no significant association was found for EDIP (ES: 1·13, 95 % CI 0·95, 1·36; I2 = 0 %) (Table 2).
Five ES from five publications(Reference Shivappa, Zucchetto and Montella22,Reference Zamora-Ros, Shivappa and Steck25,Reference Harmon, Wirth and Boushey29,Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40) were included in the meta-analysis of the association between 1-per sd increases in inflammatory potential of diet and the risk of total rectal cancer. The summary ES was 1·09 (95 % CI 1·05, 1·14; I2 = 75·1 %) (Table 2). Following pooling ES from different dietary indices separately, similar elevated colon cancer risk was observed for DII (ES: 1·08, 95 % CI 1·00, 1·17; I2 = 62·5 %) and for E-DII (ES: 1·10, 95 % CI 1·03, 1·17; I2 = 85·1 %) (Table 2).
Findings from sensitivity analysis, publication bias and meta-regression
According to sensitivity analysis utilising the random effect model, the overall ES regarding the association between greater adherence to inflammatory potential of diet and the risk CRC did not rely on a single study (C 1·26–1·54) (online Supplementary Figure 1). The meta-regression association between greater adherence to inflammatory potential of diet and the risk CRC according to age and BMI is provided in online Supplementary Figures 2 and 3, respectively. According to these findings, age and BMI did not appear to be significant sources of heterogeneity in these associations (all P-values > 0·05). The assessment of publication bias is shown in online Supplementary Figure 4 demonstrating the funnel plots of ES for inflammatory potential of diet and the risk of CRC without asymmetry and Egger’s and Begg’s tests. Based on inspecting the funnel plot visually, no evidence of publication bias was found, also confirmed by Egger’s and Begg’s tests (all P-values > 0·05).
Discussion
This meta-analysis was conducted to determine the association between inflammatory properties of diet and risk of CRC which inflammation plays an important role in its aetiology(Reference Borowczak, Szczerbowski and Maniewski46). Our results showed that the adherence to diets with high inflammatory scores reflected by DII, E-DII, EDIP and ISD were associated with greater risk of CRC. The certainly of the evidence for most included studies were high. Additionally, when cancer cite was included in analyses, positive association between the indices of inflammatory potential of diet and colon cancer, proximal, distal and rectal cancer were seen. However, the number of studies conducted on the association between dietary inflammatory scores and site-specific CRC are limited. After subgroup analysis, the positive relationship between inflammatory potential of diet and CRC risk remained unchanged except for individuals over than 65 years old. Also, it seems that sex, region, follow-up duration of cohort studies and approaches of inflammatory potential of diet score calculation (diet or supplements included) are the main sources of heterogeneity across studies. Additionally, our results indicated that the adherence to a pro-inflammatory diet regardless of type of inflammatory potential of diet score can induce the risk of CRC. Despite the methodological differences between EDIP and DII including derivation approaches, predictors, number of components and statistical methods, both have ability to predict circulating levels of inflammatory biomarkers, with higher correlation between EDIP and concentrations of plasma inflammatory markers due to its derivation from circulating inflammatory markers(Reference Tabung, Smith-Warner and Chavarro47). Moreover, our results revealed that both categorical and continuous forms of DII score demonstrated significant increased risk for CRC. Previous meta-analysis on the association between DII and CRC was limited to studies utilised FFQ for dietary assessment and those used DII as tool to measure inflammatory potential of diet which led to small number of studies. Also, the relation between inflammatory potential of diet and site-specific cancer was not examined(Reference Syed Soffian, Mohammed Nawi and Hod12). In order to fill the gap in existing literature, this comprehensive review was designed to cover larger population, variety of cultural dietary patterns and different measurements for evaluating inflammatory potential of diet.
Recently, dietary indices such as healthy eating index, plant-based dietary index and Mediterranean dietary index have been used to measure the adherence to healthy dietary style. Most of these dietary patterns recommend consuming plant-based foods, including the fruits, vegetables, nuts, legumes and wholegrain products, combined with regular intake of seafood and low intake of red and processed meat(Reference Nikparast, Etesami and Rahmani48–Reference Hejazi, Ghanavati and Hejazi51). However, some studies have indicated that despite the high intake of plant-based foods or low intake of red meat in some populations, the incidence rate of CRC is still high, indicating that other factors may contribute to CRC progression(Reference Wong, Ding and Wang52,Reference Hur, Jo and Yoon53) . A pro-inflammatory diet which is characterised by a high intake of SFA, refined grains, soft drinks, processed, organ and red meats has been proposed to increase CRC risk due to its effects on metabolic impairment(Reference Shivappa, Prizment and Blair21,Reference Wirth, Shivappa and Steck24,Reference Tabung, Steck and Ma32) . The positive association between pro-inflammatory diet and development of metabolic abnormalities such as obesity, hyperinsulinemia and glucose intolerance was seen previously(Reference Li, Zhan and Huang54). Hyperinsulinemia directly and indirectly through IGF-1 stimulation plays important roles in colon cancer progression(Reference Kasprzak55). Also, these significant results for the inflammatory potential of diet and risk of CRC could be explained by the influence of pro-inflammatory diet on circulating levels of inflammatory markers such IL-6, TNF-α and C-reactive protein which are significant contributors to neoplasia through changing immune responses(Reference Borowczak, Szczerbowski and Maniewski46). In this regard, pooled results of two cohort studies revealed that the association between inflammatory potential of diet and CRC risk was dependent mainly on degree of peritumoral lymphocytic reaction so that higher diet inflammatory scores were associated with elevated risk of CRC with an absent or low peritumoral lymphocytic reaction(Reference Liu, Nishihara and Qian30). A recent comprehensive analysis on association between dietary patterns and CRC revealed that among different dietary styles, Dietary Approaches to Stop Hypertension score (DASH), Western dietary pattern, Empirical Dietary Index for Hyperinsulinemia (EDIH) and EDIP were the most effective dietary approaches on CRC incidence(Reference Wang, Song and Eliassen45). Consistent with our study, these results show that consuming dietary components with anti-inflammatory and antioxidant properties such as fruits, vegetables especially yellow and leafy, nuts, legumes, nutraceuticals which are rich in n-3 fatty acids, vitamin A, vitamin B group, vitamin C, vitamin E, β-carotene, and minerals like Zn, Mg, and Se can reduce the risk of CRC. However, dietary inflammatory indices focus mostly on macronutrients, micronutrients and nutraceuticals, not food groups only representing a balance between multitude of dietary factors(Reference Bahr, Franz and Mähler14). Regardless of being cohort or case–control, all studies included in our meta-analysis showed positive significant association between dietary inflammatory indices and risk of CRC, except for two studies(Reference Brouwer, Makama and van Woudenbergh27,Reference Li, Wang and Shivappa42) in which the importance of inflammatory potential of diet in CRC development may be relatively smaller than the influence of genetic predisposition or regular supplements consumption. Moreover, non-significant results on the association between inflammatory potential of diet and risk of CRC among individual over than 65 years old may attribute to sedentary life and more body fat in elderlies despite healthier dietary intake they usually consume. Although the positive relationship between inflammatory potential of diet and CRC risk remained unchanged across subgroup analysis based on region, sex, follow-up duration and approaches of inflammatory potential of diet score calculation, this association was stronger among Asian population, men, studies with duration of follow-up less than 10 years and those studies used only food to calculate DII. Evidence indicates a correlation between a higher DII and an increased risk of CRC particularly among Asian population(Reference Obón-Santacana, Romaguera and Gracia-Lavedan38,Reference Abulimiti, Zhang and Shivappa40) . Several factors may contribute to this evaluated risk, including: the tradition dietary habits in some Asian populations characterised by higher processed foods and red meats can result in higher dietary inflammatory score(Reference Wada, Oba and Tsuji56), Asians are recognised as predisposing CRC and diet with higher inflammatory score may increase their susceptibility(Reference Wong, Ding and Wang52) and lifestyle behavioural changes in Asian population such as lower physical activity, obesity, tobacco use and alcohol consumption are shown to increase cancer risk(Reference Pardamean, Sudigyo and Budiarto57). Our results showed a stronger relationship between inflammatory potential of diet and risk of CRC among men. Several factors contribute to this association; the lower levels of oestrogen in men compared with women, which have been shown to have anti-inflammatory properties, may result in the higher cancer risk among men(Reference Das, Saha and Pillai58). Also differences in lifestyle factors, dietary habits, body composition and genetic and metabolic are attributed to sex inequality in cancer susceptibility(Reference Sharman, Harris and Ernst59). The stronger association between higher DII and CRC in studies with shorter follow-up can be due to early cancer development or population sensitivity. Participants in shorter studies may be in early stage of cancer development or pre-existing conditions at the beginning of the study. Shorter follow-up may capture these early developments, representing a stronger link between dietary DII and initial cancer risk. Regarding approaches of inflammatory potential of diet score calculation, it is obvious that studies did not consider supplement use in their methodology revealed stronger relationship between DII and risk of CRC due to the higher dietary inflammation. Findings of our study add to existing evidence by highlighting the importance of antioxidant and anti-inflammatory potential of diet in addition to healthy dietary components in the prevention of CRC.
Our study has several strengths and limitations; to the best of our knowledge, this study is the most comprehensive review assessing the association between variety of dietary inflammatory indices and site-specific CRC. We conducted subgroup analysis based on different cofounders to identify the source of heterogeneity among included studies. Another strength of our study is the number of included studies which cover the largest population, different countries and study designs. Also, factors which may confound the association between inflammatory potential of diet and risk of CRC were controlled among most of the included studies. However, certain limitations should be mentioned; first, the number of studies for some dietary inflammatory indices such as ISD and EDIP was limited. Second, most included studies in this review utilised a FFQ to assess dietary intake, which can lead to measurement errors and misclassifications.
Conclusion
In conclusion, we found that high inflammatory potential of diet scores reflecting pro-inflammatory diets was associated with CRC risk. Results indicated that the positive association between inflammatory potential of diet scores and CRC remained significant when cancer site or different approaches of inflammatory potential of diet scores calculation were included in analyses. Due to the highly heterogeneous studies, lack of statistical power of subgroup analyses (lack of included studies), and the fact that the Asian region has the highest burden of CRC worldwide(Reference Wong, Ding and Wang52) and that dietary intake is culturally specific, future well-designed studies with the largest sample size among the Asian region are needed to confirm our findings. Furthermore, since the standardised calculation method of DII across all studies can increase the comparability, future studies should consider this into their methodology.
Supplementary material
Supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114525000121
Acknowledgements
This study was supported in part by a grant no. 43011387-3-1 from the Shahid Beheshti University of Medical Sciences. Shahid Beheshti University of Medical Sciences had no role in the design or conduct of the study, the collection, analysis, or interpretation of the data, or the preparation, review, or approval of the manuscript.
M. G. was administrator and supervisor of current project. E. E., M. S., F. J-T. and A. M. contributed to data curation and formal analysis. A. N. contributed to methodology and validation. M. G. and A. N. contributed to the study conceptualisation and writing original draft. All authors have read and approved the final manuscript.
The authors declare no conflict of interest.
The research protocol has been performed based on PRISMA guideline.
Availability of supporting data: Data sharing is applicable.