The impact of processed foods (particularly ‘ultra’-processed foods (UPF)) on health, and potential policy action, remains a conundrum for national organisations making population dietary recommendations and guidelines. While several countries refer to food processing per se in their national dietary guidelines – including Belgium, Brazil, Malta and New Zealand – many, including the USA, Canada and most European countries, have concluded that the evidence base is currently too weak(1,2) . The Nordic Guidelines are perhaps unique in finding some compromise. While stating that ‘the categorization of foods as ultra-processed foods does not add to the already existing food classifications’, they also recommend ‘minimal intake of…processed foods containing high amounts of added fats, salt and sugar’(3). Colombia was the first country to introduce fiscal policies stated as being based on UPF and drink products(Reference Daniels4). All foods containing non-sugar sweeteners (NSS) are classified as UPF under the NOVA definition(Reference Monteiro, Cannon and Levy5). The WHO published a guideline in May 2023 suggesting ‘non-sugar sweeteners not be used as a means of achieving weight control or reducing the risk of noncommunicable diseases’(6).
In the UK, government dietary recommendations and subsequent policy are based on independent advice from the Scientific Advisory Committee on Nutrition (SACN) and its predecessor, the Committee on Medical Aspects of Food and Nutrition Policy (COMA)(7). SACN does not have a policy-making role but undertakes robust risk assessments using systematic methods to advise the four UK governments.
SACN has published a position statement and subsequent rapid evidence update on processed foods and health(1,2) . SACN has also reviewed the WHO guideline on NSS and associated evidence(8). This paper provides an overview and commentary on these SACN assessments.
Background
The role of food processing – for example, to improve convenience, palatability or shelf life – is well documented(1). A number of classification systems have been developed which group foods by their level of processing(Reference Sadler, Grassby and Hart9). The most frequently cited is the NOVA (Portuguese for new) classification(Reference Monteiro, Cannon and Levy5) which identifies ‘ultra-processed foods’ based on the inclusion of ingredients that are ‘never or rarely used in kitchens’(Reference Monteiro, Cannon and Levy10). Published commentaries(Reference Drewnowski, Gupta and Darmon11–Reference Valicente, Peng and Pacheco19) have hypothesised potential mechanisms for observed associations between (ultra-) processed foods and adverse health outcomes. These include higher palatability, energy density, promotion of faster eating rate due to their soft texture or changes to the food structure or matrix and/or widespread marketing(1).
Existing UK dietary advice, based on SACN recommendations, emphasises eating plenty of fruit and vegetables and wholegrain or higher-fibre foods. It recommends consuming foods and drinks high in saturated fat, salt or free sugars less often and/or in small amounts. These foods are clearly shown outside of the national food model, the Eatwell Guide(20). UK dietary advice currently includes some specific recommendations on processed foods, including advising against high intakes of red and processed meat and minimising sugar-sweetened drinks.
Foods that contain additives such as sweeteners or emulsifiers are defined as processed under most classifications and as UPF under the NOVA definition. The WHO guideline defines NSS as ‘all synthetic and naturally occurring or modified non-nutritive sweeteners that are not classified as sugars’. All NSS used in the UK have undergone a rigorous safety assessment by the European Food Safety Authority or the UK Food Standards Agency (FSA). Manufacturers are obliged to list NSS in the ingredients of pre-packaged food and drink, but not the amount of NSS. In the UK, policies to reduce sugar intakes, such as the Soft Drinks Industry Levy, may have resulted in an increase in NSS intakes. Due to the expanding evidence base and increasing concerns in relation to UPF and publication of the WHO guideline on NSS, SACN undertook risk assessments on both processed foods and sweeteners.
Methods
The assessments were undertaken in line with the SACN ‘Framework for the evaluation of evidence’(21).
Processed foods
The remit of SACN’s assessments on processed foods was to review:
-
1) existing classifications of processed foods (considered against pre-defined screening criteria to identify systems that were practical and applicable for the UK);
-
2) available evidence that applies NOVA to the UK National Diet and Nutrition Survey (NDNS);
-
3) available evidence on the associations between different levels of food processing and health outcomes.
Full details of methods, including search terms, search dates and databases and PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses), are included in the SACN assessments(1,2) .
The assessment of published evidence focused on systematic reviews (SR) with or without meta-analyses examining the relationship between two or more levels of food processing and health outcomes among predominantly healthy population groups. SACN primarily considers evidence provided by SR and meta-analyses of randomised controlled trials (RCT) and prospective cohort studies (PCS)(21). However, given the nature of the evidence base, SACN’s 2023 statement included SR if RCT and/or PCS formed equal to or more than 70 % of the total participant weighting. Following a UK expert round table(22), SACN’s 2025 rapid evidence update focused on higher quality evidence (largely excluding cross-sectional designs) and included a specific focus on individual PCS with subgroup or substitution analysis.
Sweeteners
In relation to the WHO guideline on NSS, SACN reviewed the associated SR with meta-analysis by Rios-Leyvraz and Montez (2022)(Reference Rios-Leyvraz and Montez23). SACN focused on the evidence considered in relation to 47 RCT and 79 PCS on body fatness, type 2 diabetes, CVD and dental caries. PCS were included that compared NSS consumption with no or lower NSS consumption. Most included RCT comparing sugar-containing drinks with drinks containing an NSS. Most of the RCT, the majority of which lasted less than 3 months, were conducted in adults. Most PCS considered adult exposure to NSS as part of a drink. Follow-up in adults ranged from 2 years to more than 30 years. Included studies were assessed for risk of bias and certainty of the evidence using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach(Reference Ignacio Neumann, Meerpohl and Davoli24).
Results
Review of existing processed food classification systems
In its 2023 position statement, SACN identified eight classification systems which aimed to group foods by their level of processing(1). Of the eight classification systems identified, NOVA was the only one that met the initial screening criteria based on potential applicability for use in the UK. While NOVA met the criterion on a clear, usable definition and the criterion on the availability of data on inter-assessor agreement, assessment beyond the initial screen identified less certainty on the clarity, reliability and feasibility of the system. The literature was dominated by NOVA, raising the risk that any limitations or biases present within the NOVA classification system may be replicated throughout the research literature.
NOVA started with three categories in 2010(Reference Monteiro, Levy and Claro25) and was later adjusted and redefined to include four categories in 2016(Reference Monteiro, Cannon and Levy5). They are given as:
-
1) unprocessed/minimally processed foods – such as fruits, vegetables, legumes, grains, nuts, unprocessed meat and fish, eggs and milk;
-
2) processed culinary ingredients – such as salt, sugar, vegetable oils, butter and other fats;
-
3) processed foods – such as bottled vegetables or meat in salt solution, fruits in syrup, bread, cheese, purees or pastes;
-
4) UPF – such as infant formulas, ready meals, ice creams, soft drinks, hamburgers, confectionary.
UPF are ‘formulations of ingredients, mostly of exclusive industrial use, typically created by series of industrial techniques and processes’(Reference Monteiro, Cannon and Levy5). UPF are classified solely by their level of processing, not by their energy or nutrient content. However, many products that are energy-dense and high in saturated fat, salt or free sugars, and/or low in fruit and vegetables and fibre, tend to be categorised as UPF(Reference Drewnowski, Gupta and Darmon11,Reference Gupta, Hawk and Aggarwal15,Reference Rolls, Cunningham and Diktas17,Reference Dicken, Batterham and Brown26) . The UPF category can also capture products that other approaches to dietary assessment may classify as ‘healthier’, such as fortified foods, low-fat yogurts, vegetable sauces and higher-fibre breakfast cereals.
Review of available evidence that uses the UK National Diet and Nutrition Survey dataset to apply the NOVA food processing classification
SACN identified twelve studies that applied NOVA to dietary data collected through the UK NDNS datasets, 2008–2019(Reference Aceves-Martins, Bates and Craig27–Reference Souza, Andrade and Rauber38). Differences were identified with regard to how studies treated both ‘homemade’ and ‘manufactured’ food groups within the nutrient databank.
Ten of the twelve studies(Reference Aceves-Martins, Bates and Craig27,Reference Madruga, Martínez Steele and Reynolds30–Reference Souza, Andrade and Rauber38) estimated the contribution of UPF to total dietary energy intake. This ranged from 51 % for adults to 68 % for children aged 12–18 years. In a comparison with National Health and Nutrition Examination Survey (NHANES) data from the USA, similar estimated average UPF contribution (58·2 % ± 0·9 %) was attributed to total dietary energy intake(Reference Steele, O’Connor and Juul39). The NutriNet-Santé data from France estimated average UPF consumption to be 35·9 % of total dietary energy intake(Reference Beslay, Srour and Méjean40,Reference Julia, Martinez and Allès41) . In 2025, SACN identified one further study which applied NOVA to NDNS intake data(Reference Dicken, Batterham and Brown26). The study reported partial overlap between the UPF classification and food and drinks high in saturated fat, salt or sugars. UPF tended to have an ‘unhealthier’ nutritional profile than ‘minimally processed’ foods. The authors noted that by their definitions, not all UPF were considered ‘unhealthy’.
Several limitations were identified in applying NOVA to UK dietary survey data. At the time of publication, the NDNS did not capture all the detail required by NOVA (such as the source of production or additive content of foods). Therefore, researchers may under- or over-estimate UPF consumption because of oversimplified interpretation of the NDNS food groupings.
Review of available evidence on the association between processed food consumption and health outcomes
In its position statement in 2023, SACN identified twenty SR that evaluated associations between processed food consumption and health outcomes, ten of which were extracted as they included data from predominantly RCT and/or PCS. Health outcomes considered were given as:
-
• overweight and obesity in two SR(Reference Jardim, Costa and Pessoa42,Reference Pagliai, Dinu and Madarena43) ;
-
• chronic non-communicable diseases (including type 2 diabetes, hypertension and CVD, cerebrovascular disease and gastrointestinal tract disease) in six SR(Reference Jardim, Costa and Pessoa42–Reference Wang, Du and Huang47);
-
• depression in two SR(Reference Pagliai, Dinu and Madarena43,Reference Lane, Davis and Beattie48) ;
-
• mortality risk (including all-cause mortality, CVD-cause mortality, heart-cause mortality and cancer-cause mortality) in four SR(Reference Pagliai, Dinu and Madarena43,Reference Suksatan, Moradi and Naeini46,Reference Lane, Davis and Beattie48,Reference Taneri, Wehrli and Roa-Diaz49) ;
-
• maternal and child health outcomes in two SR(Reference de Oliveira, de Sousa and Assunção50,Reference Paula, Patriota and Goncalves51) .
Most included studies used the NOVA classification. SR consistently showed positive associations between UPF consumption and adverse health outcomes. Most SR reported that increased consumption of processed food (specifically UPF) was associated with an increased risk of the adverse health outcomes considered. However, the available evidence was almost exclusively observational, and several of the SR identified did not disaggregate prospective and cross-sectional data. Furthermore, there was inconsistent adjustment for covariables.
In 2025 SACN identified a further five umbrella reviews(Reference Barbaresko, Bröder and Conrad52–Reference Wang, Lu and Wang56), eight SR including PCS only(Reference Claudino, Bueno and Piloneto57–Reference Yuan, Hu and Li64) and eleven SR of observational studies with a separate analysis by PCS(Reference Babaei, Pourmotabbed and Talebi65–Reference Tian, Deng and Xie75). The majority of umbrella reviews and SR reported that increased consumption of UPF was associated with increased risk of adverse health outcomes including obesity, type 2 diabetes, CVD, cancer and mental health conditions.
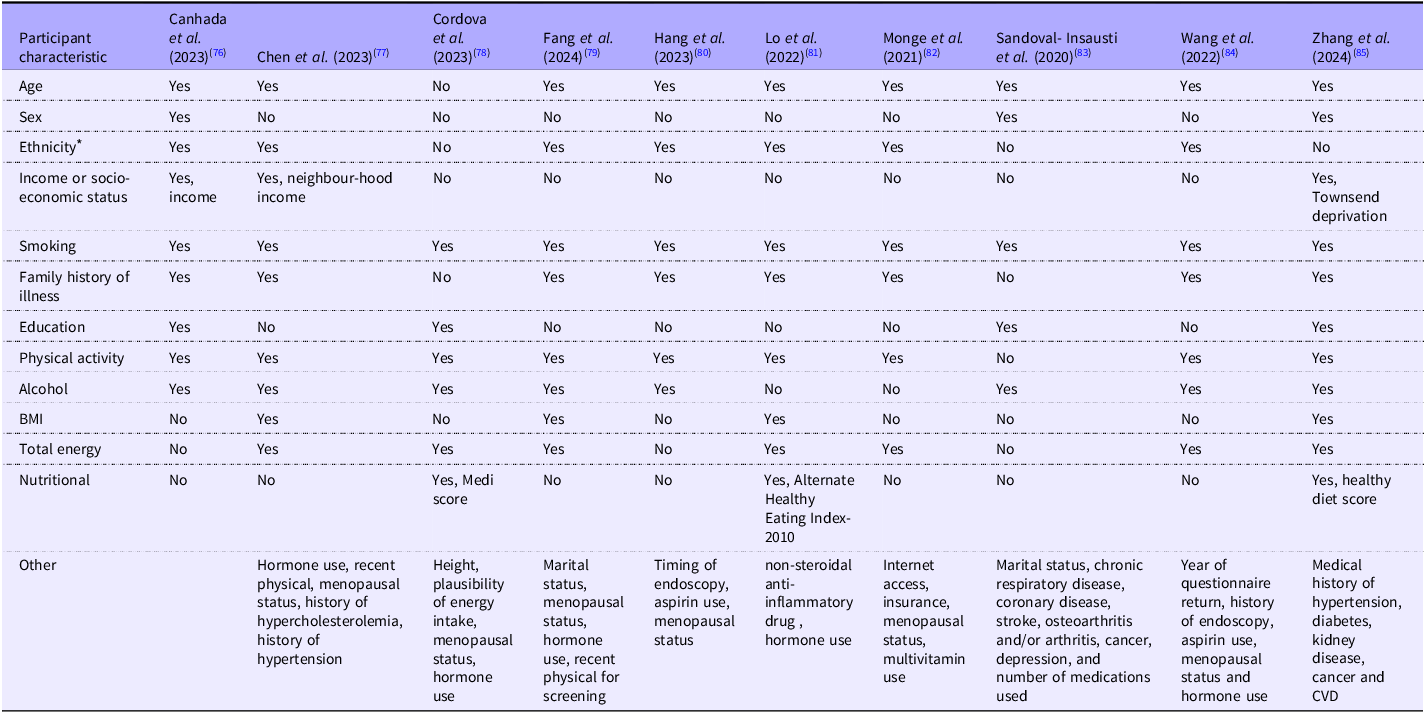
SACN further identified ten PCS that carried out a subgroup analysis by food type(Reference Canhada, Vigo and Levy76–Reference Zhang, Xu and Chang85). The range of food groups considered made it difficult to compare the findings from the different studies. However, analyses suggest that some UPF categories, including meat and animal products and sweetened drinks, tended to be associated with an increased risk of adverse health outcomes. In contrast, UPF categories including vegetarian alternatives were not associated with adverse health outcomes. Mixed results were found for UPF categories including bread and cereals, savoury snacks, dairy products, sweets and desserts, sauces and spreads, fats, ready-to-eat foods and alcoholic drinks. Observed associations were inconsistently adjusted for covariates (see Table 1). Therefore, although adverse health associations were consistently reported, it was unclear whether these associations were due to, or independent of, the poor nutritional profiles that are typical of many UPF.
Summary table of adjustments made by prospective cohort studies included in SACN’s rapid evidence update on processed foods and health(2)

SACN, Scientific Advisory Committee on Nutrition.
* Ethnicity is often stated in studies as ‘race’ and ‘white/non-white’.
Limited information was available on the impact on population subgroups. This impacts both the potential generalisability of study findings to the UK population and limits our understanding of potential differential effects of UPF consumption on health. Dietary data within observational studies were mostly based on dietary collection methods (such as FFQ) that were unlikely to have been designed or validated for assessing the level of food processing.
Few RCT have been undertaken. In its position statement in 2023, SACN identified one small (n 20) crossover RCT by Hall et al., 2019(Reference Hall, Ayuketah and Brychta86) that assessed the effect of UPF on energy intake and body weight. Participants received either a UPF diet or an unprocessed diet for 2 weeks, followed immediately by the alternate diet for 2 weeks. Authors reported greater energy intake and faster meal eating rate during the ultra-processed diet, and both body weight and body fat mass increased during the ultra-processed diet and decreased during the unprocessed diet. Several limitations were identified, including that the diets in the study groups were not isoenergetic, with the UPF diets higher in energy. In 2025, SACN identified two further crossover RCT. The first, a small RCT (n 9) by Hamano et al., 2024(Reference Hamano, Sawada and Aihara87), found that participants living with overweight or obesity gained more weight (1·1 kg) on a UPF diet compared with a non-UPF diet. Participants consumed 813·5 kcal more per d (342·4–1284·7; P = 0·004) compared with during the non-UPF period. The second by Dicken et al., 2025(Reference Dicken, Jassil and Brown88) (n 55) found that participants lost weight on both minimally processed food and UPF diets but to a greater extent on the minimally processed food diet –2·06 %, v. –1·05 % on the UPF diet. Authors reported that energy intake was –503·7 kcal d–1 (se, 130·2; P < 0·001) and –289·6 kcal d–1 (se, 102·8; P = 0·007) lower during the minimally processed food and UPF diet compared with baseline, respectively.
Scientific Advisory Committee on Nutrition assessment of WHO guideline and associated review of sweeteners
The SR with meta-analysis by Rios-Leyvraz and Montez (2022) reported conflicting evidence(Reference Rios-Leyvraz and Montez23). The RCT data indicated that use of NSS may result in a small reduction in body weight based on low-certainty evidence. But PCS evidence indicated that higher NSS intake was associated with higher measures of body fatness in adults and children based on low- to very low-certainty evidence. The PCS data also indicated that NSS may be associated with a broad range of adverse health outcomes (including type 2 diabetes, CVD and mortality in adults) based on low- to very low-certainty evidence. There was limited evidence on dental health outcomes in children and adults. SACN noted that it may have come to different conclusions to the SR on the application of GRADE and the certainty of the evidence.
WHO prioritised PCS evidence over RCT evidence in reaching conclusions. SACN noted that randomisation is particularly important when looking at weight outcomes given that NSS use is potentially confounded by weight and associations with diet. In this case, SACN would prioritise evidence from RCT, which showed that NSS use in place of sugars resulted in a small reduction in body weight and BMI scores in adults. However, SACN also noted that the effect size was small and limited by the length of the trials (most were 3 months or less; shorter-term weight loss outcomes would commonly be assessed over 3–6 months and longer term over 12 months)(89).
WHO considered that the shorter-term RCT were not long enough to be able to determine the impact on weight loss. SACN notes that there may be a health benefit from short-term weight loss even if there is subsequent weight gain. SACN noted that one NSS RCT lasted longer than 2 years and reported a greater reduction in body weight (approximately 5 kg compared with less than 1 kg in shorter-term RCT). The association remained when studies of less than 8 weeks were excluded.
Observational studies such as PCS are at high risk of ‘reverse causality’ and confounding, which are likely to have contributed to the differences between observational and trial evidence. For example, some people may have chosen to consume NSS-sweetened foods and drinks because they were trying to manage their weight, rather than consumption of NSS causing weight gain. Also, consuming NSS foods and drinks may be linked to other factors known to affect health such as socio-economic status, smoking or alcohol. SACN welcomed the detailed list of confounders adjusted for in the PCS. However, in SACN’s opinion, the authors of the SR did not fully acknowledge the possibility of confounding that could not be accounted for.
SACN noted that WHO issued a conditional guideline that NSS should not be used as a means of achieving weight control or reducing risk of non-communicable diseases. This was due to uncertainties of the evidence.
SACN concluded that evidence of risk to health from consuming NSS is inconsistent, particularly in relation to measures of body fatness. However, SACN shared WHO’s concerns regarding the risks of non-communicable diseases associated with NSS intake given the range of studies observing such associations. The evidence on the impact of consuming NSS on dental health is poor, and more high-quality research of adequate duration (greater than 2 years) is needed to determine long-term impact.
Discussion
There have been consistent reports from SR that higher consumption of UPF is associated with higher risks of adverse health outcomes. However, there continue to be uncertainties around the quality of current evidence. SACN found that the available evidence was almost exclusively observational and there may not have been adequate accounting for confounding factors or key variables such as energy intake, BMI, smoking and socio-economic status. SACN noted that diets high in UPF were often energy-dense, high in saturated fat, salt or free sugars, high in processed meat and/or low in fruit and vegetables and fibre. SACN has concluded in previous robust risk assessments that consumption of excess energy, saturated fat, salt, free sugars and processed meats is linked to poor health outcomes, and that higher consumption of fruit and vegetables, wholegrain foods and fibre reduces health risks.
SACN has noted that the UK’s national food model, the Eatwell Guide(20), which is based on SACN recommendations, already indicates that many foods classified as ultra-processed such as crisps, biscuits, cakes, confectionery and ice cream are not part of a healthy, balanced diet. It also emphasises consumption of a diet based on fruit, vegetables and wholegrain or higher-fibre starchy carbohydrates, with less red and processed meat and less foods high in saturated fat, salt and free sugars.
SACN made a range of research recommendations in its 2023 position statement on processed foods and health. Following its publication, the UK Government Office for Science (GOS) published summaries of the discussions from two expert roundtables on this topic held in November 2023(22). Of particular relevance were comments that working with the existing NOVA system and developing subcategories within category 4, based on nutritional composition in addition to processing, was considered a better alternative when compared with developing a new classification system at present. It also noted that critical evidence gaps included a lack of controlled feeding studies and trials to capture ‘real-world’ food behaviour.
WHO are developing a guideline on UPF, which will include consideration of the definition and application to policy(90). The UK National Institute for Health and Care Research (NIHR) and UK Research and Innovation (UKRI) have subsequently commissioned research in this area. In addition, the new contract for the NDNS rolling programme provided an opportunity to review the dietary data collection tool, Intake24, to achieve better estimates of exposure to processed foods, and considerations are ongoing regarding options to better monitor population intakes of NSS. SACN continues to keep the topic of UPF under annual review and will consider this topic again in 2026.
SACN has made recommendations to the government in relation to improving the data publicly available on the NSS content of foods and consumption trends (particularly among high-consuming and vulnerable groups). Priority research gaps include addressing concerns relating to confounding between NSS and health outcomes, consideration of the impact of NSS on vulnerable groups (including high consumers, young children and pregnant or lactating women) and exploration of innovative ways to reliably monitor exposure to NSS.
Conclusions
UK dietary recommendations are based on robust independent risk assessments by the SACN. SACN has concluded that the observed associations between higher consumption of UPF and adverse health outcomes are concerning. SACN recommended that, on balance, most people are likely to benefit from reducing their consumption of processed foods high in energy, saturated fat, salt and free sugars and low in fibre.
In a separate report on feeding young children aged 1–5 years, SACN has also recommended that commercially manufactured foods and drinks marketed specifically for infants and young children are not needed to meet nutrition requirements(91).
In relation to sweeteners, SACN made a precautionary recommendation that intake of NSS should be minimised. SACN also reiterated its recommendation that average population intake of free sugars should not exceed 5 % of energy(92). For younger children, SACN recommended not offering drinks sweetened with sugar or NSS but unsweetened food (not sweetened with either sugar or NSS). For older children and adults, SACN recommended that swapping free sugars for NSS may help reduce free sugar intake from foods and drinks (and so reduce energy intake), at least in the short term – the long-term goal is to limit both sugar and NSS intake.
These issues remain on SACN’s watching brief.
Acknowledgements
The processed food assessments and review of the WHO guideline on sweeteners were developed, reviewed and approved by SACN. A list of SACN members is available here: https://www.gov.uk/government/groups/scientific-advisory-committee-on-nutrition. SACN is a committee of the UK Department of Health and Social Care.
Support from wider members of the Nutrition, Evidence, Surveys and Translation team at the Department of Health and Social Care (DHSC) – Tazeem Bhatia, Sian Magee and Gillian Swan.
The report was funded by the UK Department of Health and Social Care.
R. A., A. C. and C. S.-G. prepared the first draft of the manuscript, which was revised by J. L. and I. Y. All authors commented on the draft manuscript and approved the final manuscript.
The authors declare that there is no conflict of interest.
Details of declarations of interest for SACN members are available here: https://www.gov.uk/government/groups/scientific-advisory-committee-on-nutrition#register-of-interests