Traumatic brain injury (TBI) is a significant global health concern, contributing to high mortality and morbidity rates, with most cases involving intracranial bleeding(Reference Lawati, Sharif and Maqbali1). The global incidence of TBI is approximately 69 million cases per year, primarily caused by road traffic injuries and falls(Reference James, Theadom and Ellenbogen2,Reference Dewan, Rattani and Gupta3) . In lower- and middle-income regions, TBI are notably more frequent, significantly impacting the youth population(Reference Dewan, Rattani and Gupta3,Reference Harrison, Berry and Jamieson4) . TBI development encompasses both primary and secondary brain injuries(Reference Ferreira, Rodrigues and Della-Pace5). The primary injury often results from direct mechanical impact, causing damage such as lacerations, contusions, intracranial haemorrhages and diffuse axonal injuries(Reference Ferreira, Rodrigues and Della-Pace5). The secondary injury is characterised by a delayed and intricate response leading to long-term neuropathological alterations, including metabolic imbalances, neurovascular disruptions, neuroinflammation, oxidative stress, axonal damage and potentially neurodegeneration, depending on the initial trauma’s severity(Reference Cornelius, Crupi and Calabrese6). The pro-inflammatory response of the cerebrovascular unit, marked by increased expression of adhesion molecules like intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1) on the endothelial surface, leads to the recruitment of circulating leukocytes to the injury site, thereby amplifying neuroinflammation(Reference Dell’Aquila, Maiese and De Matteis7). Inflammation and oxidative stress play a central role in the pathogenesis, progression and severity of TBI(Reference Djordjevic, Golam Sabbir and C Albensi8,Reference Lehnardt9) . Because secondary brain injury might be reversible, it provides a critical window for therapeutic intervention aimed at preventing long-term damage(Reference Abdul-Muneer, Chandra and Haorah10).
Recently, dietary supplements with anti-inflammatory effects, such as probiotics, have been suggested as a strategy to improve recovery and reduce morbidity, mortality and long-term consequences in TBI patients(Reference Tan, Zhu and Du11,Reference Wan, Wang and Zhang12) . A 2020 study by Wan et al. (Reference Wan, Wang and Zhang12) revealed that 15 d of probiotic administration (Bifidobacterium longum, Lactobacillus bulgaricus and Enterococcus faecalis ≥1·0 × 107 colony-forming units (CFU)) led to a significant reduction in serum markers, including IL-6, IL-10, C-reactive protein and TNF-α in patients with severe TBI. Furthermore, a prospective randomised clinical trial by Ebrahimi-Mameghani in 2013 demonstrated that supplementation with the probiotic VSL#3—comprising four strains of Lactobacillus (L. casei, L. plantarum, L. acidophilus and L. delbrueckii subsp. bulgaricus), three strains of Bifidobacterium (B. longum, B. breve and B. infantis) and Streptococcus salivarius subsp. thermophilus—led to a significant reduction in C-reactive protein levels, although it did not impact total antioxidant capacity (TAC) or malondialdehyde (MDA) levels in critically ill patients(Reference Ebrahimi-Mameghani, Sanaie and Mahmoodpoor13). A meta-analysis conducted by Rezazadeh et al. in 2023, which reviewed six randomised clinical trials, revealed that probiotic treatment might positively impact VCAM-1 in adults, with no significant influence on ICAM-1(Reference Rezazadeh, Pourmoradian and Tutunchi14).
Overall, prior research has predominantly focussed on specific inflammatory and oxidative stress markers in critically ill patients, yielding various inconclusive results. In terms of TBI patients, it is unclear whether the administration of probiotics could improve cell adhesion molecules (CAMs) such as ICAM-1 and VCAM-1, dietary intakes, oxidative stress and antioxidant biomarkers. This randomised parallel clinical trial was therefore designed to examine the effect of probiotic supplementation on dietary intakes, oxidative stress biomarkers and CAMs in TBI patients.
Methods
Study design and participants
In this double-blind placebo-controlled randomised clinical trial, patients with TBI admitted to the neurosurgery intensive care unit (ICU) of Imam Reza Hospital and Shohada Hospital of Tabriz University of Medical Sciences, Tabriz, Iran were enrolled between December 2020 and November 2021 (Figure 1). The trial protocol was approved by the Ethics Committee of Tabriz University of Medical Sciences (Ethics code: TBZMED.REC.1401·355) and was registered at the Iranian Registry of Clinical Trials (IRCT20100209003320N18). In this publication, we report secondary study outcomes, while primary study outcomes, including immune function variables, anthropometric measures, disease severity and markers of inflammation, have been reported previously(Reference Abbaszadeh, Yousefi and Arefhosseini15).
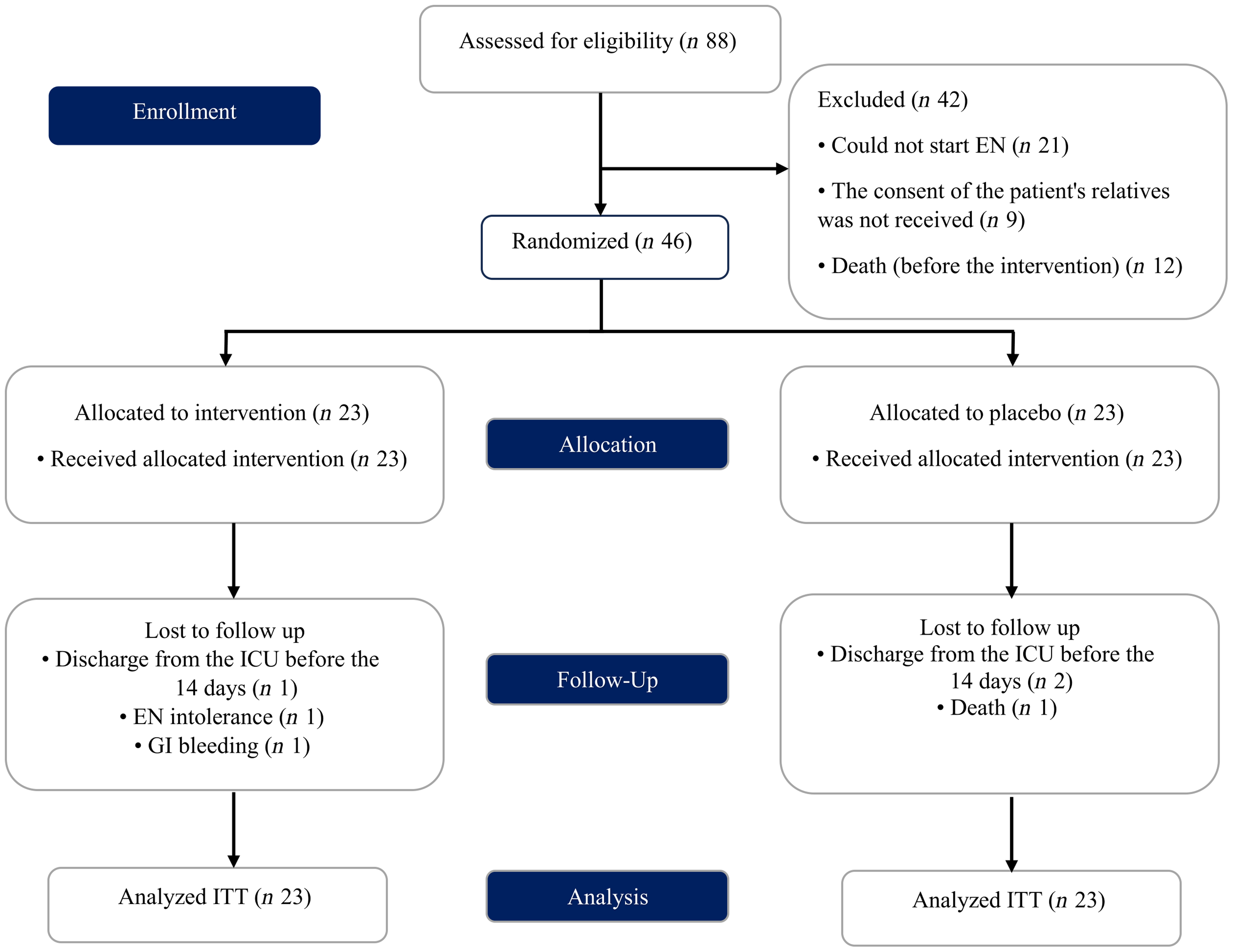
Study flow diagram (EN, enteral nutrition; ICU, intensive care unit; GI, gastrointestinal bleeding ITT, intention-to-treat).

After a full explanation of the study aims and protocol, informed written consent was obtained from the patients’ first-degree relatives. The inclusion criteria were as follows: patients with TBI, aged 18–60 years of both genders, with a low level of consciousness (Glasgow Coma Scale < 8), receiving gastrointestinal feeding through a nasal tube, with an APACHE II score of 15–30, and staying at ICU for at least 14 d. Exclusion criteria included (1) lactating or pregnant; (2) unstable haemodynamics; (3) intestinal obstruction; (4) intestinal ischaemia; (5) short bowel syndrome; (6) pancreatitis and receiving total parenteral nutrition for more than 2 d and (7) treatment with immunosuppressive medications.
Sample size
Sample size was calculated based on the reported change in plasma MDA levels by Ebrahimi-Mameghani et al after probiotic preparation (VSL# 3) in critically ill patients(Reference Ebrahimi-Mameghani, Sanaie and Mahmoodpoor13). By considering α = 0·05 and β = 0·20 (power = 80 %), as well as assuming a 20 % drop-out rate, the sample size was estimated to be twenty-three subjects in each arm using the following formula(Reference Cleophas, Zwinderman and Cleophas16):
 $$n = {{{[{{\left( {{{\rm{Z}}_{1 - {{\alpha } \over {2}}}} + {{\rm{Z}}_{1 - \beta }}} \right)}^2} \times {\sigma ^2}]{\rm{\;}}}} \over {{{{({\rm{\mu }}1 - {\rm{\mu }}2)}^2}}}}$$
$$n = {{{[{{\left( {{{\rm{Z}}_{1 - {{\alpha } \over {2}}}} + {{\rm{Z}}_{1 - \beta }}} \right)}^2} \times {\sigma ^2}]{\rm{\;}}}} \over {{{{({\rm{\mu }}1 - {\rm{\mu }}2)}^2}}}}$$
Randomisation, blinding and intervention
Patients were randomly allocated to the ‘probiotic’ or ‘placebo’ group with a 1:1 distribution. The randomisation list was computer-generated by the permutated block method. Randomisation was conducted by a study personnel member who was not involved in patient recruitment, assessment or data analysis. The investigators, patients, those responsible for patient enrollment, administration of the intervention and assessors (nurses) were blinded.
Demographic and personal details, including laboratory data, medical history and vital signs, were recorded for each patient at baseline. Enteral nutrition feeding was initiated at 25 ml/h and then increased by 25 ml/h every 4 h until the target rate was achieved. If the gastric residual volumes exceeded 200 ml, prokinetic agents were initiated, and feeding was advanced until the target rate was reached. According to the physician’s prescription, patients received routine therapy (e.g. antibiotics). Per ICU standard protocol, all patients were fed by a nasogastric tube (size 16F) for enteral feeding. Patients received enteral feeding by a feeding pump consisting of a 2-hour infusion, followed by an hour of rest. Given the gut resting time from 02.00 to 06.00, patients received enteral nutrition 7 times/day. In case of enteral nutrition intolerance, if parenteral nutrition was initiated, the patient was excluded from the study.
Forty-six patients with TBI were randomly assigned to two groups. Those in the placebo group (n 23) received standard treatment plus placebo capsules containing 500 mg of sterile maltodextrin. In contrast, the patients in the probiotic group (n 23) received standard treatment plus probiotic capsules containing 109 bacteria, consisting of four strains of Lactobacillus (L. casei, L. bulgaricus, L. rhamnosus, L. acidophilus), two strains of Bifidobacterium (B. longum, B. breve) and Streptococcus thermophilus every 6 h for 14 d (four capsules daily). The probiotic and placebo capsules, produced by the same company (Lactocare, ZIST TAKHMIR, Tehran, Iran), were identical in size, shape and colour. At the end of the gavage formula, the probiotic or placebo capsules were mixed with sterile distilled water at room temperature and injected through a nasogastric feeding tube using a sterile syringe to wash the tube.
Anthropometric and nutritional assessments
Targeting calorie intake and protein needs at 25–30 kcal/kg at 1·2–1·5 gr/kg of body weight. Energy intake was started by 500–600 kcal/day based on patients’ tolerance. Energy intake increased gradually periodically through the formula. Food intake through enteral feeding was recorded daily. The daily dietary intakes of energy, macronutrients, fibre and antioxidant micronutrients (including vitamins A, E, C and Zn, Cu and Se) were obtained using Nutritionist IV software (N Squared Computing, San Bruno, CA) on days 1 and 14 of the study.
Moreover, all anthropometric assessments and measurements were conducted on days 1 and 14 of the study. To estimate height, the length of the ulna (cm) was first measured at the midpoint between the elbow and the prominent bone of the wrist (preferably on the left side), and height (m) was determined using an available chart(17). Mid-upper arm circumference was measured at the midpoint between the tips of the shoulder and the elbow (olecranon and acromion) on the right arm using a non-stretchable tape to the nearest 0·1 cm. Weight was then estimated using height and mid-upper arm circumference(Reference Darnis, Fareau and Corallo18). Skinfold thickness was also measured according to standard protocol(Reference Mahan19) using a calibrated plastic caliper (Slim Guide Skinfold Caliper, Zhejiang, China) to the nearest 1 mm.
Biochemical analysis
At baseline and after 14 d, in an overnight fasting state, 10 ml of blood was taken from each patient and stored for 20 min at room temperature. The samples were then centrifuged at 3000 g for 5 min, and the serum was stored at −80°C until analysis. Serum levels of ICAM-1, VCAM-1, pro-oxidant–antioxidant balance (PAB), TAC, MDA, nitric oxide (NO) and arylesterase (ARE) activity were determined on the first day the patient was admitted to the hospital following a TBI. ICAM-1 and VCAM-1 levels were assessed using enzyme-linked immunosorbent assay kits from ZellBio GmbH, Ulm, Germany, while NO concentration was determined using enzyme-linked immunosorbent assay commercial kits from Navand Salamat Company, Iran. Additionally, serum levels of TAC, MDA and ARE activity were assessed using spectrophotometric methods. PAB was assessed using a method previously developed by Faraji-Rad et al. (Reference Faraji-Rad, Khajavi and Arjmand20).
Statistical analysis
Statistical analyses were performed using SPSS (IBM SPSS Statistics) version 23. Data analysis was conducted according to the intention-to-treat principle. The Shapiro–Wilk test, skewness, Q–Q diagram and Kolmogorov–Smirnov test were used to examine whether the continuous variables were normally distributed. For variables that were negatively skewed, a square transformation was applied, whereas a natural logarithm transformation was used for those that were positively skewed. Summary data are presented as numbers (percentages) for categorical variables and as means (standard deviation, sd) or medians (25th, 75th) for normally and not normally distributed continuous variables, respectively. An independent-samples t test was applied to compare quantitative variables between the two groups, while within-group comparisons were made using a paired-sample t test. Furthermore, Chi-square test was applied to examine differences in categorical variables between the two groups. To estimate the intervention effect for all outcomes, an ANCOVA was used, adjusting for potential differences between the groups at baseline. Confounders were identified based on a literature review and were considered prognostic(Reference Karimi, Naeini and Niazkar21–Reference Keshani, Alikiaii and Babaei23).
For log-transformed variables, the ratio of geometric means with corresponding 95 % CI was used. A two-tailed P value of 0·05 was considered statistically significant.
Results
The CONSORT flowchart of the trial is provided in Figure 1. This figure has been previously published(Reference Abbaszadeh, Yousefi and Arefhosseini15). Of the forty-six patients enrolled in the trial, forty completed the study. In each group, three patients were lost to follow-up due to discharge from the ICU before 14 d (placebo group: n 1, supplement group: n 2), enteral nutrition intolerance (placebo group: n 1), gastrointestinal bleeding (placebo group: n 1) or death (supplement group: n 1). Additionally, no side effects were reported by the nurses.
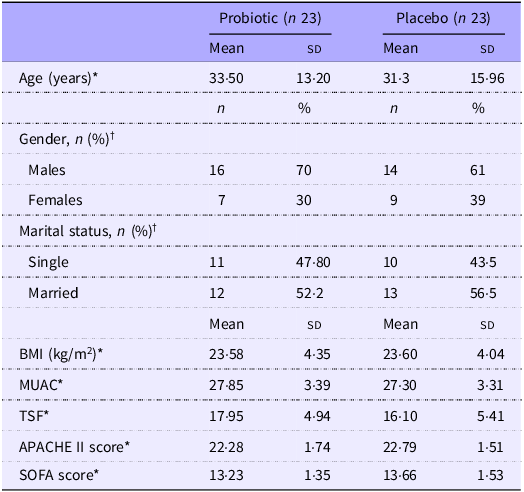
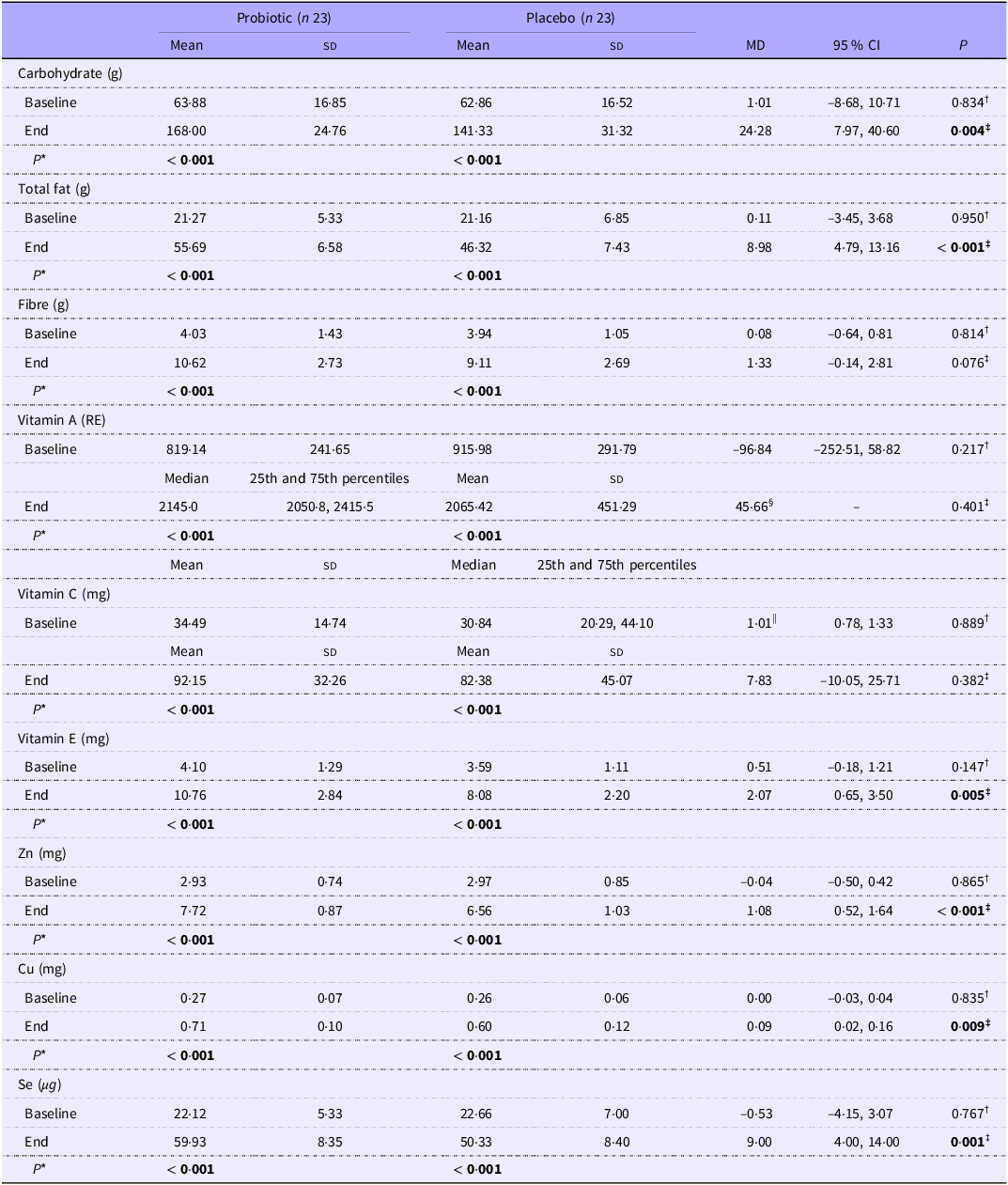
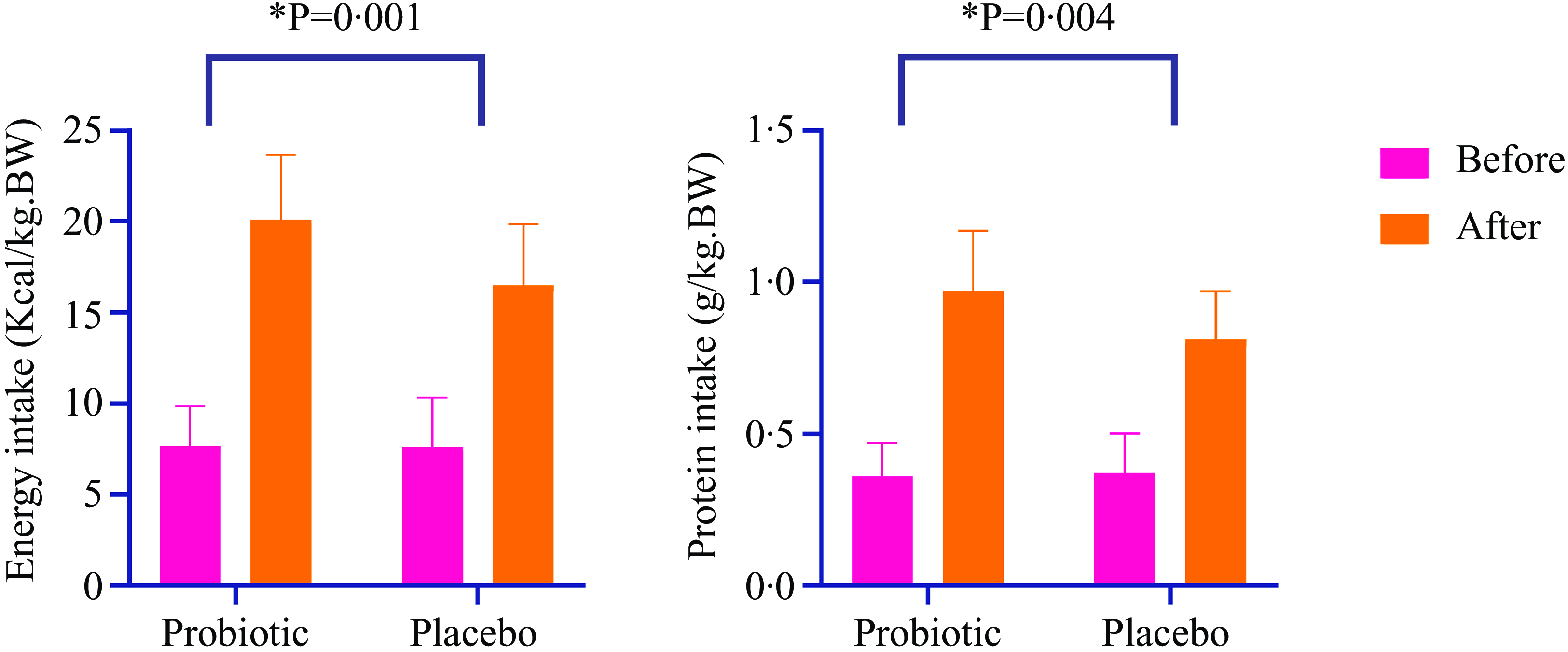
The baseline characteristics of the patients were similar between the two groups (Table 1). This table has been previously published(Reference Abbaszadeh, Yousefi and Arefhosseini15). As shown in Table 2 and Figure 2, there were no significant differences in dietary intakes between the study groups at baseline. The intake of energy, macronutrients, fibre and micronutrients (vitamins E, C and A, Zn, Cu and Se) significantly increased over the intervention in both groups ( P< 0·001). Moreover, significant between-group changes were observed for dietary intakes of total energy (P= 0·001), carbohydrate (P= 0·004), protein (P= 0·004) and total fat (P< 0·001), vitamin E (P= 0·005), Zn (P< 0·001), Cu (P= 0·009) and Se (P< 0·001) at the end of the study.
Baseline characteristics of the study participants (Numbers and percentages; mean values and standard deviations)

Abbreviations: MUAC, mid-upper arm circumference, TSF, Triceps skinfold thickness. *Values are expressed as means (standard deviation) based on independent sample t test. †Values are expressed as numbers (percent) based on chi-square.
Comparison of energy and daily actual nutrient intakes among study groups (Mean values and standard deviations; median values and 25th and 75th percentiles; 95 % CI)

Abbreviations: Mean (standard deviation) is presented for normally distributed data. Median (25th and 75th percentiles) is presented for data not normally distributed. *P-value based on paired-sample t test. † P-value based on independent sample t test. ‡ P-value based on ANCOVA adjusted for age, sex and baseline values. §Due to negatively skewed data, the data are expressed as a median difference (squared data cannot be transformed back to the original scale because of the presence of negative values and the difficulty of interpreting the result). ||Due to positively skewed data, the data are expressed as a ratio of geometric means (95 % CI).
Bold values indicate statistically significant (P < 0.05).
The effect of the intervention on energy and protein intake in two study groups. Values are mean (standard deviation). *Data analysis was done using ANCOVA test (adjusted for age, sex and baseline values).

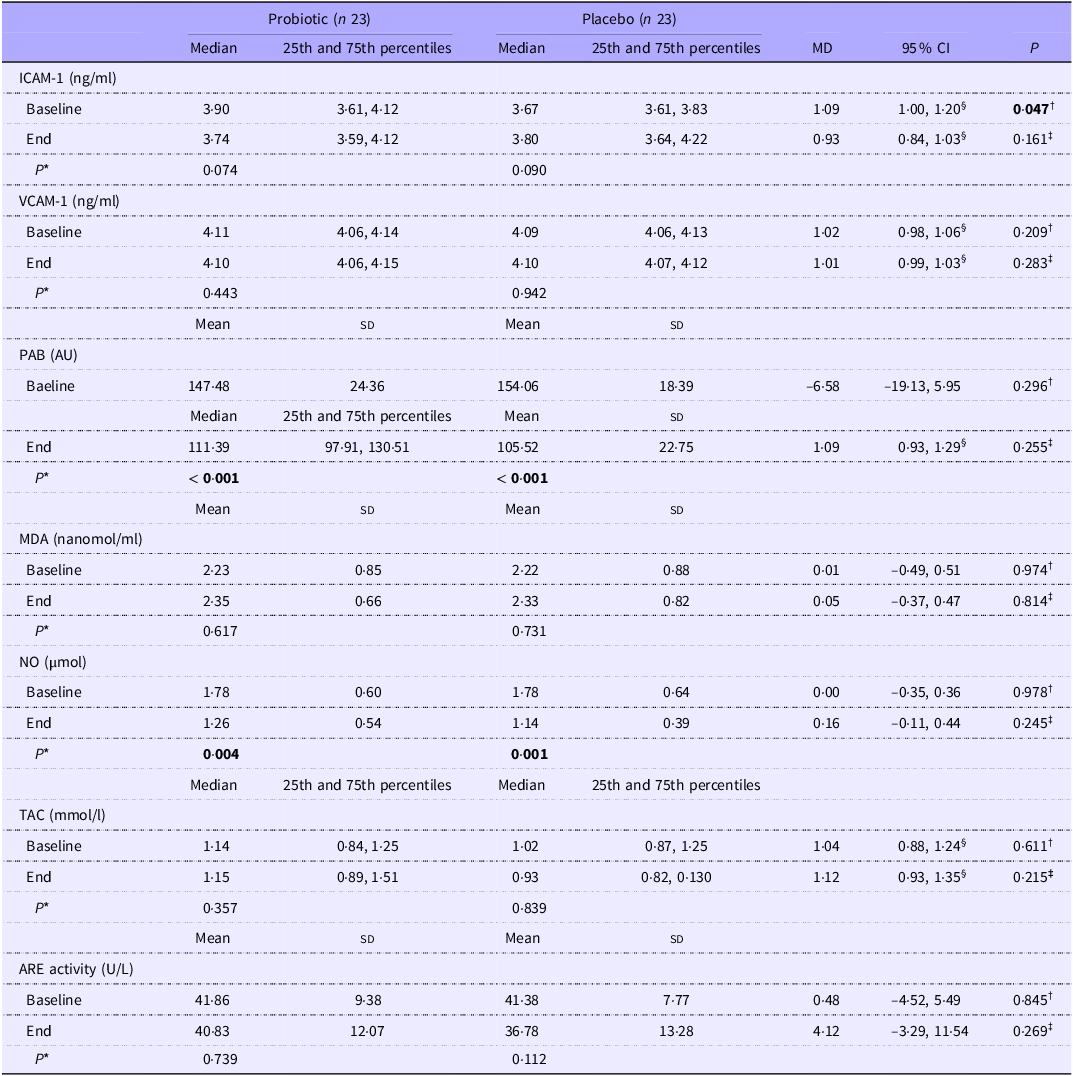
Changes in the biochemical parameters of the study patients are presented in Table 3. Apart from serum ICAM-1 levels (P= 0·047), there were no significant differences in biochemical parameters between the groups at baseline. However, within-group comparisons revealed significant reductions in serum concentrations of PAB (probiotic group, P< 0·001; placebo group, P< 0·001) and NO (probiotic group, P= 0·004; placebo group, P= 0·001). At the end of the study, no significant between-group differences were observed in ICAM-1 (P= 0·161), VCAM-1 (P= 0·283), PAB (P= 0·255), MDA (P= 0·814), NO (P= 0·245), TAC (P= 0·215) or ARE levels (P= 0·269).
Cell adhesion molecules and oxidative stress/antioxidant parameters of the patients throughout the study (Mean values and SDs; median values and 25th and 75th percentiles; 95 % CI)

Abbreviations: ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular cell adhesion molecule 1; PAB, pro-oxidant–antioxidant balance; MDA, malondialdehyde; NO, nitric oxide; TAC, total antioxidant capacity; ARE, arylesterase; MD, mean/median of difference. Mean (standard deviation) is presented for normally distributed data. Median (25th and 75th percentiles) is presented for data not normally distributed. *P-value based on paired-sample t test. † P-value based on independent sample t test. ‡ P-value based on ANCOVA adjusted for age, sex and baseline values. §Due to skewed data, the data are expressed as a ratio of geometric means (95 % CI).
Bold values indicate statistically significant (P < 0.05).
Discussion
This randomised clinical trial investigated the effect of probiotic supplementation in TBI patients, demonstrating a significant improvement in total energy and nutrient intake after 14 d. However, there was no significant effect on serum ICAM-1, VCAM-1, PAB, MDA, NO, TAC or ARE levels.
Current guidelines consider nutritional interventions a critical component in the clinical management of ICU patients(Reference Mehta, Sunavala and Zirpe24). Among available dietary supplements, probiotics have appeared as a biologically plausible approach to treating or preventing a wide range of infectious, inflammatory and autoimmune conditions(Reference Dwivedi, Amaresan and Sankaranaryanan25). Therefore, it seems that probiotic administration could result in an improvement in clinical outcomes in TBI patients. To the best of our knowledge, this study appears to be the first randomised, double-blind, placebo-controlled trial to investigate the effects of probiotic supplementation on serum pro-oxidant and antioxidant biomarkers, ICAM-1 and VCAM-1 in TBI patients.
In this study, we did not find any significant effect of probiotic supplementation on serum levels of ICAM-1 and VCAM-1 compared with the placebo group (Table 3). Most previous research on probiotics in TBI patients has primarily focussed on inflammatory factors, with less attention given to CAMs(Reference Tan, Zhu and Du11–Reference Ebrahimi-Mameghani, Sanaie and Mahmoodpoor13). Although less straightforward, our findings are comparable to studies examining the effect of probiotics on inflammation. A prospective randomised clinical trial investigating the effect of probiotic supplementation on inflammatory factors in patients with mild TBI reported no significant effects on serum levels of C-reactive protein, interferon-γ (IFNγ), TNF-α and various interleukins over eight weeks(Reference Brenner, Forster and Stearns-Yoder26), similar to our findings. Nevertheless, the results of Wan et al. (Reference Wan, Wang and Zhang12) showed that probiotic supplementation for 15 d in patients with severe TBI significantly reduced serum levels of IL-6, IL-10, TNF-α and C-reactive protein. In the study by Tan et al. (Reference Tan, Zhu and Du11), TBI patients were supplemented with probiotics consisting of 0·5 × 108 Bifidobacterium longum, 0·5 × 107 Lactobacillus bulgaricus and 0·5 × 107 Streptococcus thermophilus for 21 d, and the findings showed significant increases in serum levels of IL-12p70 and IFNγ, as well as significant reductions in serum concentrations of IL-4 and IL-10. Differences in study populations, TBI severity, ancillary therapies such as corticosteroids and probiotic strains may have contributed to these varying findings. More studies are needed to clarify the effect of probiotics on CAMs.
Although we found no significant effect of probiotic supplementation on CAMs, the literature suggests several potential mechanisms. One proposed mechanism involves the role of adhesion molecules like ICAM-1 and VCAM-1, which are critical for leukocyte adhesion to inflamed tissues(Reference Hua27). The expression of these molecules is induced by inflammatory cytokines like IL-1, IL-6 and TNF-α (Reference Hua27). Alterations in the gut microbiota, characterised by an imbalance between harmful and beneficial bacteria, have been associated with systemic inflammation and metabolic endotoxemia(Reference Anhê, Barra and Cavallari28–Reference van Lier, Geven and Leijte30). Elevated levels of circulating lipopolysaccharides, derived from an abnormal gastrointestinal microbiome, can trigger a systemic inflammatory response and increase pro-inflammatory cytokine levels(Reference Rezazadeh, Alipour and Jafarabadi31–Reference Naito, Yoshida and Makino33). Available evidence suggests that probiotics may alleviate endothelial dysfunction and vascular inflammation by reducing endotoxemia and inflammatory responses(Reference Rezazadeh, Pourmoradian and Tutunchi14,Reference Grylls, Seidler and Neil34,Reference Bernini, Simão and Alfieri35) .
Another mechanism involves the metabolites of probiotics, particularly SCFA like propionate, acetate and butyrate, produced by beneficial bacteria such as bifidobacteria and lactobacilli, which exert anti-inflammatory effects by interacting with specific receptors on intestinal epithelial cells to inhibit the NF-κB pathway, suppress regulatory T cells and reduce pro-inflammatory cytokine production by neutrophils and macrophages, thereby preventing inflammation and promoting an anti-inflammatory state(Reference Cristofori, Dargenio and Dargenio36,Reference Vinolo, Rodrigues and Hatanaka37) .
In this clinical trial, no significant changes were found in serum levels of PAB, MDA, TAC, NO and ARE activity compared to the control group. A relatively extensive literature has examined the effects of probiotics on antioxidant biomarkers(Reference Mohammadi, Jazayeri and Khosravi-Darani38–Reference Vaghef-Mehrabany, Homayouni-Rad and Alipour40). However, no human studies have investigated the effects of probiotics on oxidative stress indicators in head trauma patients. Therefore, we compared our findings with studies conducted in patients with different health conditions and animal studies. In human studies, supplementation with probiotics (containing L. acidophilus, L. casei and B. bifidum) in diabetic haemodialysis patients resulted in significant decreases and increases in MDA and TAC serum levels, respectively. However, no significant effect was seen on NO and glutathione levels(Reference Soleimani, Mojarrad and Bahmani41). Findings from another study with a similar design documented significant reductions and increases in MDA and TAC serum levels, respectively, in diabetic haemodialysis patients, with no significant effect on NO levels(Reference Soleimani, Motamedzadeh and Zarrati Mojarrad42). Following 8 weeks of supplementation with probiotics (L. casei at a dose of 108 CFU/day) in patients with rheumatoid arthritis, a significant increase in serum superoxide dismutase levels was observed. However, there were no significant changes in catalase, MDA and TAC levels compared with the control group(Reference Vaghef-Mehrabany, Homayouni-Rad and Alipour40). Moreover, a prospective randomised trial reported that probiotic supplementation (VSL#3) did not have any significant effect on serum concentrations of MDA and TAC in critically ill patients(Reference Ebrahimi-Mameghani, Sanaie and Mahmoodpoor13). Synbiotic supplementation (Synbiotic 2000Forte) in critically ill trauma patients has been shown to significantly reduce MDA levels compared with the placebo group(Reference Kotzampassi, Giamarellos-Bourboulis and Voudouris43). Collectively, our findings should be interpreted in light of the following points. First, it is possible that probiotic supplementation in the current study had a small protective role, which was not detected. Second, the lack of significant effect of probiotics on antioxidant biomarkers could be due to the severe TBI, where the inflammatory response leads to irreversible damage. Third, studies are performed on specialised populations with different health conditions, and the effect of probiotics may vary among diseases. Fourth, conflicting results of studies may be related to different doses, genera, species or strains of probiotics.
In terms of animal studies, Sun et al. (Reference Sun, Ling and Wang44) showed that the administration of Clostridium butyricum significantly decreased MDA content and increased superoxide dismutase activity in mice with cerebral ischaemia/reperfusion injury. Similar results were also observed in the study by Wanchao et al. (Reference Wanchao, Chen and Zhiguo45), in which administering inactivated Lactobacillus in rats with cerebral ischaemia–reperfusion injury led to a significant reduction in MDA levels and a significant increase in superoxide dismutase activity. Moreover, a study documented that treatment with probiotics for 2 weeks in mice with ischaemic brain tissue effectively reduced serum levels of MDA(Reference Akhoundzadeh, Vakili and Shadnoush46). Supplementation with glutamine and probiotics in a rat with burn injury model significantly increased superoxide dismutase and reduced reactive oxygen species (ROS) and NO content(Reference Gong, Yuan and Dong47). Yilmaz et al. (Reference Yılmaz and Erdem48) reported that L. rhamnosus administration in mice with sepsis significantly decreased endothelial nitric oxide synthase and MDA and increased glutathione peroxidase and paraoxonase 1 (PON1). Moreover, Erel et al. (49) have found that Bacillus clausii (B.clasuii) did not have any protective effect on serum levels of MDA, glutathione peroxidase, endothelial nitric oxide synthase and PON1 in mice with sepsis. Similarly, Rahmati et al. (Reference Rahmati, Momenabadi and Vafaei50) failed to find any significant effect of probiotic supplementation (pretreatment) on MDA levels in a mouse model with cerebral hypoperfusion over three weeks. Overall, animal and human studies on the effects of probiotics in TBI patients have provided conflicting findings. More studies are needed to draw definitive conclusions about the effects of probiotics on oxidative stress factors in TBI patients.
While we found no significant effect of probiotic supplementation on oxidative stress biomarkers, the following mechanisms have been suggested for the beneficial effects of probiotics on oxidative stress biomarkers. One of the suggested underlying mechanisms is related to the nuclear factor-erythroid 2-related factor 2, kelch-like ECH-associated protein 1 and antioxidant response element (Nrf2-Keap1-ARE) pathway(Reference Wang, Wu and Wang51). Nrf2 activation regulates genes involved in ROS detoxification to resist oxidants(Reference Jones, Desai and Darby52). One of the Nrf2 molecular switches is Keap1, and at low levels of ROS, Nrf2 binds to its cytoplasmic inhibitor Keap1. When cells are assaulted by free radicals or nucleophiles, redox-sensitive cysteine residues of Keap1 react and change the Keap1 functional conformation, thereby destroying Nrf2 inactivation(Reference Ulasov, Rosenkranz and Georgiev53). Nrf2 is then transported to the nucleus and binds to antioxidant response element sequences, boosting the transcription of ARE-driven genes such as detoxifying protein and antioxidant enzyme genes(Reference Maher and Yamamoto54). ROS can also activate the redox-sensitive transcription factor NF-kB, leading to the production of inflammatory cytokines during inflammation(Reference Srivastava, Yadav and Reddy55). Moreover, extracellular polysaccharides from some probiotic strains inhibit NF-kB and ROS generation, preventing lipopolysaccharide-induced inflammation in RAW 264·7 macrophages(Reference Diao, Xin and Zhou56).
In this study, supplementation with probiotics led to a significant increase in the intake of energy, macronutrients and micronutrients compared with the placebo group. Probiotics contribute to maintaining homeostasis by modulating the immune system through the regulation of immunoglobulins and cytokines, stimulating macrophages and enhancing the response to food antigens. They also strengthen the intestinal epithelial barrier, promote nutrient absorption, foster the proliferation of beneficial microorganisms, compete with pathogens for nutrients and adhesion to the intestinal epithelium, modulate the host immune system and inhibit pathogenic bacteria(Reference Dwivedi, Amaresan and Sankaranaryanan25,Reference Bermudez-Brito, Plaza-Díaz and Muñoz-Quezada57) . Taken together, it seems that supplementing with probiotics could improve dietary intake by increasing patient tolerance.
This trial has several limitations. First, a fixed dosage of probiotics was used, and higher doses or longer administration times may have produced different results. Second, enrollment was limited to TBI patients, and including patients from other ICU settings may have yielded different results. Third, the relatively small sample size limited our ability to perform subgroup analyses based on disease severity and other demographic characteristics. Fourth, some patients were lost to follow-up, but we used intention-to-treat analysis to address this issue. Fifth, the duration of the intervention might not have been long enough to affect oxidative stress and antioxidant parameters. Additionally, our study did not consider signalling pathways such as Nrf2, which would allow investigation of the molecular effects of probiotics.
Conclusion
Among TBI patients, probiotic supplementation, compared with placebo, improved total energy and nutrient intakes. However, it did not significantly affect CAMs, pro-oxidant and antioxidant biomarkers. Further clinical trials with larger sample sizes are needed to clarify the efficacy of probiotics in patients with TBI.
Acknowledgements
We sincerely thank the patients who participated in this study. We would like to appreciate of the cooperation of Clinical Research Development Unit, Imam Reza General Hospital, Tabriz, Iran in conducting of this research.
This research was funded by Tabriz University of Medical Sciences [grant number: 65 716].
The authors’ responsibilities were as follows: N. N., M. E-M., S. N., S. S. S., H. T. and S. R. A. wrote the original paper; N. N. and S. S. S. helped with data collection; A. M. and S. S. contributed to patient selection; M. E-M., S. N. and N. N. performed statistical analysis; M. E-M., H. T., S. N., S. S., S. R. A. and A. M. contributed to the conception of the article as well as to the final revision of the manuscript. All authors read and approved the final version of the manuscript.
The authors declare that they have no competing interests.
The datasets used and/or analysed during this study are available from the corresponding author upon reasonable request.
Ethics Approval and Consent to Participate
All procedures performed in this study were in accordance with the ethical standards of the Ethics Committee of Tabriz University of Medical Sciences. The first-degree relatives of the patients signed the consent form, and the study protocol was approved by the Ethics Committee of Tabriz University of Medical Sciences (Ethics code: TBZMED. REC.1401·355).