Introduction
Early-onset dementia (EOD), defined as dementia with onset prior to the age of 65, is estimated to have affected 3.9 million people worldwide in 1990, rising to 7.8 million in 2021 (He et al., Reference He, Wang, Zhang, Zhu, Li and Liu2025). The increase is largely attributed to improvements in the diagnosis of EOD; however, it continues to be underdiagnosed (He et al., Reference He, Wang, Zhang, Zhu, Li and Liu2025; Leroy et al., Reference Leroy, Bertoux, Skrobala, Fouchard, Didierjean, de Flores and Habert2021). EOD can have devastating consequences for families and create a high burden for caregivers, the healthcare system, and the overall economy (Antonioni et al., Reference Antonioni, Raho, Lopriore, Pace, Latino, Assogna, Mancuso, Gragnaniello, Granieri, Pugliatti, Di Lorenzo and Koch2023; Hendriks et al., Reference Hendriks, Peetoom, Bakker, van der Flier, Papma, Koopmans, Verhey, de Vugt and Köhler2021). Within the spectrum of EOD, frontotemporal dementia (FTD) is the third most common form of dementia after Alzheimer’s disease (AD) and dementia with Lewy Bodies (DLB) (Hendriks et al., Reference Hendriks, Peetoom, Bakker, van der Flier, Papma, Koopmans, Verhey, de Vugt and Köhler2021). Typically manifesting between ages 45 and 65, and in some cases as early as the 20s, FTD usually spans a disease course of 8–10 years, although durations can range from 2 to 20 years (Antonioni et al., Reference Antonioni, Raho, Lopriore, Pace, Latino, Assogna, Mancuso, Gragnaniello, Granieri, Pugliatti, Di Lorenzo and Koch2023; Snowden et al., Reference Snowden, Neary and Mann1996).
Timely diagnosis of FTD is critical yet challenging. Due to its involvement of the frontal lobes, FTD is generally characterized by changes in a person’s personality, behaviour, and executive function as originally described in the Lund-Manchester Research Criteria (Neary, Reference Neary1999; Snowden et al., Reference Snowden, Neary and Mann1996). Contemporary frameworks, however, conceptualize FTD as a clinically heterogeneous spectrum with profiles that can include changes in language, social skills, motor skills, emotion processing, and other areas of cognitive impairment (e.g., semantic memory) (Bang et al., Reference Bang, Spina and Miller2015). Differentiating FTD from other conditions, such as depression, AD, or other psychiatric disorders, continues to be difficult (Hendriks et al., Reference Hendriks, Peetoom, Bakker, van der Flier, Papma, Koopmans, Verhey, de Vugt and Köhler2021; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007). Rosness et al. (Reference Rosness, Haugen, Passant and Engedal2008) found that the interval from symptom onset to diagnosis in FTD ranged from 49.5 to 59.2 months on average, and Leroy et al. (Reference Leroy, Bertoux, Skrobala, Fouchard, Didierjean, de Flores and Habert2021) found the interval to be 37.8 months on average, with both studies reporting it was significantly longer than for AD. International research under the Genetic Frontotemporal Dementia Initiative (GENFI) has demonstrated that structural and cognitive changes can be detected up to 10 years before diagnosis (Rohrer et al., Reference Rohrer, Nicholas, Cash, van Swieten, Dopper, Jiskoot, van Minkelen, Rombouts, Cardoso, Clegg and Binetti2015). GENFI studies also found that gene-specific cognitive profiles exist (Poos et al., Reference Poos, MacDougall, van den Berg, Jiskoot, Papma, van der Ende, Seelaar, Russell, Peakman and Convery2022) and that cognitive measures are sensitive indicators for tracking disease progression (Poos et al., Reference Poos, Jiskoot, Papma, van den Berg, van der Ende, Meeter and Pijnenburg2015). A more comprehensive understanding and tracking of the cognitive profile of early-onset FTD (EO-FTD), a major cause of EOD, has the potential to advance clinical practice by facilitating earlier diagnosis and intervention to ultimately enhance quality of life for people with EO-FTD.
FTD has been historically described as comprising three primary subtypes or variants encompassing FTD, each with distinct cognitive profiles. In the behavioural variant of FTD (bvFTD), cognitive impairments predominantly involve executive function, attention, abstraction, set shifting, response inhibition, planning and sequencing, verbal fluency, and decision-making. Semantic dementia (svFTD), or the semantic variant of primary progressive aphasia, primarily affects semantic memory, leading to difficulties with word comprehension, object recognition, face recognition, and sensory processing (e.g., tastes, smells, and tactile stimuli). Progressive non-fluent aphasia, or the non-fluent variant of primary progressive aphasia, is chiefly a language disorder characterized by speech apraxia with syntactic errors or word substitutions, though early deficits in calculation and verbal memory may also be evident, with executive dysfunction emerging later (Antonioni et al., Reference Antonioni, Raho, Lopriore, Pace, Latino, Assogna, Mancuso, Gragnaniello, Granieri, Pugliatti, Di Lorenzo and Koch2023; Snowden et al., Reference Snowden, Neary and Mann1996).
Discussion persists in the categorization of FTD. For example, Snowden et al. (Reference Snowden, Neary and Mann1996) noted the difficulty in including loss of insight as a criterion for bvFTD, although present in 85% of people with bvFTD, because of its evaluation reliability issues. They also noted the challenges with the interchangeable use of svFTD with the semantic variant of primary progressive aphasia, given that it is more of a semantic memory disorder than a primary aphasia. Similarly, Shelley et al. (Reference Shelley, Al-Khabouri, Hussein and Raniga2007) refer to bvFTD as the frontal variant of FTD (fvFTD) and further delineate fvFTD into apathetic and disinhibited types. More recently, additional less common subtypes have sometimes been included under the umbrella of FTD such as right temporal variant (rtvFTD), FTD associated with motor neuron disease (FTD-MND), corticobasal syndrome (CBS), and progressive supranuclear palsy because of their frontal lobe involvement (Antonioni et al., Reference Antonioni, Raho, Lopriore, Pace, Latino, Assogna, Mancuso, Gragnaniello, Granieri, Pugliatti, Di Lorenzo and Koch2023). Within these expanded classifications, further clinical distinctions have emerged, with symptoms such as face recognition deficits now being more closely associated with rtvFTD rather than svFTD (Antonioni et al., Reference Antonioni, Raho, Granieri and Koch2025; Ulugut et al., Reference Ulugut, Bertoux, Younes, Montembeault, Fumagalli, Samanci, Illán-Gala, Kuchcinski and Leroy2024), highlighting the evolving nature of diagnostic frameworks. Such heterogeneity among the FTD variants further complicates the differentiation of FTD from other dementias or non-dementia-related aetiologies, hindering timely diagnosis and early intervention.
Cognitive symptoms remain a critical yet under-characterized component of EO-FTD. Although EO-FTD is increasingly recognized as involving a constellation of biomarkers and clinical symptoms (e.g., behavioural, motor) that extend beyond cognition, cognitive changes are often the first domain to be systematically assessed in clinical settings, are central to referral pathways for specialist evaluation, and are incorporated into most diagnostic testing at the time of presentation. Cognitive screening tests frequently used in dementia research and clinical practice, however, often do not adequately assess the executive dysfunction characteristic of FTD (Cavedoni et al., Reference Cavedoni, Chirico, Pedroli, Cipresso and Riva2020). As a result, early signs of FTD may be overlooked, especially in the absence of more comprehensive neuropsychological testing and detailed reports from patients and companions (family or friends). As cognitive symptoms are amenable to standardized measurement and longitudinal tracking, improved characterization of early cognitive presentation offers an opportunity to enhance early identification and inform the development of more sensitive assessment approaches within the broader multidimensional prodromal landscape of EO-FTD.
Objective and research questions
To clarify how cognition is currently characterized in EO-FTD, a scoping review was conducted to systematically map the presenting cognitive symptoms of EO-FTD prior to diagnosis. This review synthesizes findings from studies reporting cognitive symptoms as described by patients, their companions, health professionals, or derived from cognitive testing at the time of or prior to diagnosis, with the aim to identify gaps in current characterization that may limit early recognition, as well as key areas for further research and potential improvements in early diagnostic practices.
The original research questions were: (1) Which cognitive symptoms do adults, without other neurological conditions, experience prior to receiving a diagnosis of EOD? and (2) How are these cognitive symptoms identified – whether by the patient, companions (family or friends), a health professional, or via cognitive testing? Following title and abstract screening, due to the high volume of studies eligible for full-text review for all types of EOD, the study team refined the population in the first research question to focus on EO-FTD exclusively.
Methods
Design and eligibility criteria
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis extension for scoping reviews (Tricco et al., Reference Tricco, Lillie, Zarin, O’Brien, Colquhoun, Levac, Moher, Peters, Horsley, Weeks, Hempel, Akl, Chang, McGowan, Stewart, Hartling, Aldcroft, Wilson, Garritty and Straus2018). The review protocol was not registered.
Cognitive symptoms were defined as ‘observable or reported impairment in mental processes, such as memory, attention, language, executive function, and visuospatial abilities, which may indicate underlying neuropsychological dysfunction’ (Lezak et al., Reference Lezak, Howieson, Bigler and Tranel2012). For the purposes of this scoping review, symptoms that are not inherently cognitive were excluded. Examples of these excluded symptoms include mental health or mood-related symptoms (e.g., anxiety, mood swings, sadness), specific language difficulties (e.g., aphasia), behavioural changes, psychiatric symptoms, and functional impairments (e.g., social dysfunction or difficulty with activities of daily living).
Studies were included in this scoping review if they met the following criteria: (1) reported specific cognitive symptoms experienced by individuals prior to, or at the time of, an EO-FTD diagnosis. Studies that only provided overall cognitive test scores or described cognitive symptoms solely after diagnosis or during disease progression were excluded; (2) were prospective studies reporting cognitive symptoms at initial presentation (prior to EO-FTD diagnosis) or retrospective studies detailing presenting cognitive symptoms at the time of EO-FTD diagnosis; (3) had a quantitative or qualitative study design. Case studies involving single patients, kindreds, or pedigrees were excluded; (4) identified cognitive symptoms using self-reports from patients, informant reports from companions (e.g., family or friends), cognitive test findings, or clinical observations; (5) were peer-reviewed journal articles or thesis dissertations written in English that involved adult human participants without other neurological conditions (e.g., comorbid brain injury, stroke, Down’s Syndrome or Parkinson’s, etc.). Articles focusing exclusively on brain imaging results, genetics, or non-cognitive symptoms (e.g., mood or neuropsychiatric features) of EOD were excluded.
Information sources and search strategy
Potentially relevant papers were identified through searches of the following online bibliographic databases: Medline, Embase, CINAHL, PsycInfo, Scopus, and Proquest Dissertations and Theses. The literature search was executed on November 28, 2024. The search strategies, drafted by an experienced research librarian (LD) and further refined through study team discussion, combined EOD and assessment/diagnosis with either cognition terms or qualitative research terms to be as sensitive as possible. No date, language, or study type limits were used. The full search strategy is included in the Supplementary Materials. The final search results were exported into Covidence (Veritas Health Innovation, n.d.). Duplicates were removed automatically by Covidence or manually by the reviewers.
Screening and eligibility
Two reviewers (TM and FA) independently applied the predefined inclusion and exclusion criteria to an initial set of 40 abstracts in parallel to calibrate their understanding and ensure consistent application of the eligibility criteria. The reviewers then proceeded with independent title and abstract evaluations of all remaining records using the same a priori criteria. Publications were advanced to full-text review if they met the refined inclusion criteria – specifically, focusing on EO-FTD and excluding case studies involving single patients, kindreds, or pedigrees. Both reviewers then assessed the full texts to confirm eligibility for data extraction, and data were extracted by both reviewers. Any disagreements during study selection or data extraction were resolved through consensus and, when necessary, discussion with the broader study team.
Data-charting process
A data-charting form was collaboratively developed by the two reviewers in consultation with the study team to systematically extract information on cognitive symptoms reported prior to EO-FTD diagnosis, as well as the sources from which these symptoms were identified.
For each publication, the form also captured the study characteristics such as year of publication, study objective, study type and design, method of analysis, study setting, country, number of EO-FTD participants, as well as EO-FTD participant demographic characteristics including sex, education, ethnicity (when reported), and age at onset or first clinical presentation. The two reviewers independently and iteratively reviewed the texts, charted the data, and subsequently updated the form based on their findings, ensuring consistency and accuracy throughout the process.
Data analysis and synthesis
Comprehensive descriptive summaries were generated for each included study to support synthesis and interpretation. A formal appraisal of the methodological quality or risk of bias of included studies was not conducted given the objective of this scoping review to map and summarize the existing body of knowledge regarding the presenting cognitive symptoms of EO-FTD. This follows established methodological guidance for scoping reviews on clinical topics (Tricco et al., Reference Tricco, Lillie, Zarin, O’Brien, Colquhoun, Kastner, Levac, Ng, Sharpe, Wilson, Kenny, Warren, Wilson, Stelfox, Rios, Moher, Godfrey, MacDonald, Langlois and Straus2016).
Cognitive symptoms experienced by adults prior to an EO-FTD diagnosis were extracted from the included studies. All cognitive symptoms are reported as originally named in each of the studies. The frequency with which each cognitive symptom was measured, along with the frequency of each source type. In addition, a breakdown of the studies by country is provided.
Results
Selection of sources of evidence
Six databases (Scopus, Embase, MEDLINE, PsycInfo, CINAHL, and Proquest Dissertations and Theses) were searched originally for EOD (all types) and yielded 2,197 studies after duplicate removal. Following title and abstract screening by the two reviewers, 313 articles were identified as eligible for full-text review across all EOD types, including case studies. Given the study team’s decision to exclusively focus on EO-FTD, these 313 studies were imported into a separate Covidence instance. Title and abstract screening were then repeated with the additional inclusion criteria of FTD and the exclusion criteria of case studies applied. Ultimately, 34 studies proceeded to full-text review, of which 16 studies met the eligibility criteria for inclusion. Studies were excluded at full-text review for the following reasons: not written in English (n = 2), failure to identify cognitive symptoms (n = 13), inability to distinguish EO-FTD symptoms from late-onset FTD symptoms (n = 2), or failure to differentiate the presenting cognitive symptoms for EO-FTD from other dementia types (n = 1). Figure 1 presents the PRISMA flow diagram outlining the study selection process.
PRISMA flow diagram of study selection process.

Study characteristics
Characteristics of each study were identified, including publication date, study objective, study design, study setting and country, and method of analysis (Table 1). The following is a synthesis of these findings.
Overview of included studies

Publication dates ranged from 2007 to 2024, with half of the studies (n = 8) published between 2007 and 2012 (Chow et al., Reference Chow, Pio and Rockwood2011; Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Panegyres & Frencham, Reference Panegyres and Frencham2007; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011). Research settings were internationally distributed (Figure 2).
Distribution of study settings by country.

Clinical profiles of dementia were examined in the majority of studies, with 11 of the 16 studies explicit in this aim (Edahiro et al., Reference Edahiro, Okamura, Arai, Ikeuchi, Ikeda, Utsumi, Ota, Kakuma, Kawakatsu, Konagaya, Suzuki, Tanimukai, Miyanaga and Awata2023; Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Panegyres, Reference Panegyres2021; Panegyres & Frencham, Reference Panegyres and Frencham2007; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023). Six of these specifically stated an intent to identify cognitive or neuropsychological symptoms as part of the clinical profile (Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Panegyres, Reference Panegyres2021; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008).
Study designs were observational, descriptive cross-sectional, and qualitative in nature. The majority of studies employed observational designs (n = 13) (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Panegyres, Reference Panegyres2021; Panegyres & Frencham, Reference Panegyres and Frencham2007; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023), including prospective (n = 6) (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Panegyres, Reference Panegyres2021; Panegyres & Frencham, Reference Panegyres and Frencham2007; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023), retrospective (n = 4) (Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Rivas Nieto, Reference Rivas Nieto2014; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011), prospective and retrospective (n = 2) (Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014), and one case control study (Panegyres et al., Reference Panegyres, Graves and Frencham2007). Two studies used descriptive, cross-sectional survey designs (Chow et al., Reference Chow, Pio and Rockwood2011; Edahiro et al., Reference Edahiro, Okamura, Arai, Ikeuchi, Ikeda, Utsumi, Ota, Kakuma, Kawakatsu, Konagaya, Suzuki, Tanimukai, Miyanaga and Awata2023) and one study employed a qualitative interview approach (Johannessen et al., Reference Johannessen, Helvik, Engedal and Thorsen2017). Nearly, all studies (n = 15) reported analyses of cognitive symptoms using descriptive statistics, apart from the qualitative study by Johannessen et al. (Reference Johannessen, Helvik, Engedal and Thorsen2017).
Cognitive symptoms preceding EO-FTD diagnosis
A total of 35 uniquely named cognitive symptoms were identified as presenting symptoms of EO-FTD. The most frequently reported cognitive impairments were in memory (all studies), followed by attention (n = 10) (Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Johannessen et al., Reference Johannessen, Helvik, Engedal and Thorsen2017; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023) and then executive function (n = 7) (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023). All other cognitive symptoms were mentioned in only one to three studies.
The cognitive symptoms identified in each study and their reported sources are summarized in Table 2, along with the EO-FTD participant demographics (number, sex, and education, when reported). Ethnicity was not included in Table 2 as it was only reported in one study (van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023) with 77% of the participants being Caucasian. Across the 16 included studies, there were 663 EO-FTD participants, with half of the studies (n = 8) (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Johannessen et al., Reference Johannessen, Helvik, Engedal and Thorsen2017; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008) comprising 25 or fewer participants.
Cognitive symptoms preceding EO-FTD diagnosis and source of symptoms

Sources of presenting cognitive symptoms
Cognitive symptom data were variably derived. Ten studies (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024; Panegyres & Frencham, Reference Panegyres and Frencham2007; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023) obtained data from cognitive tests. Three additional studies (Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Panegyres, Reference Panegyres2021; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008) performed cognitive testing but reported overall scores only, with no detail on the specific cognitive domains affected.
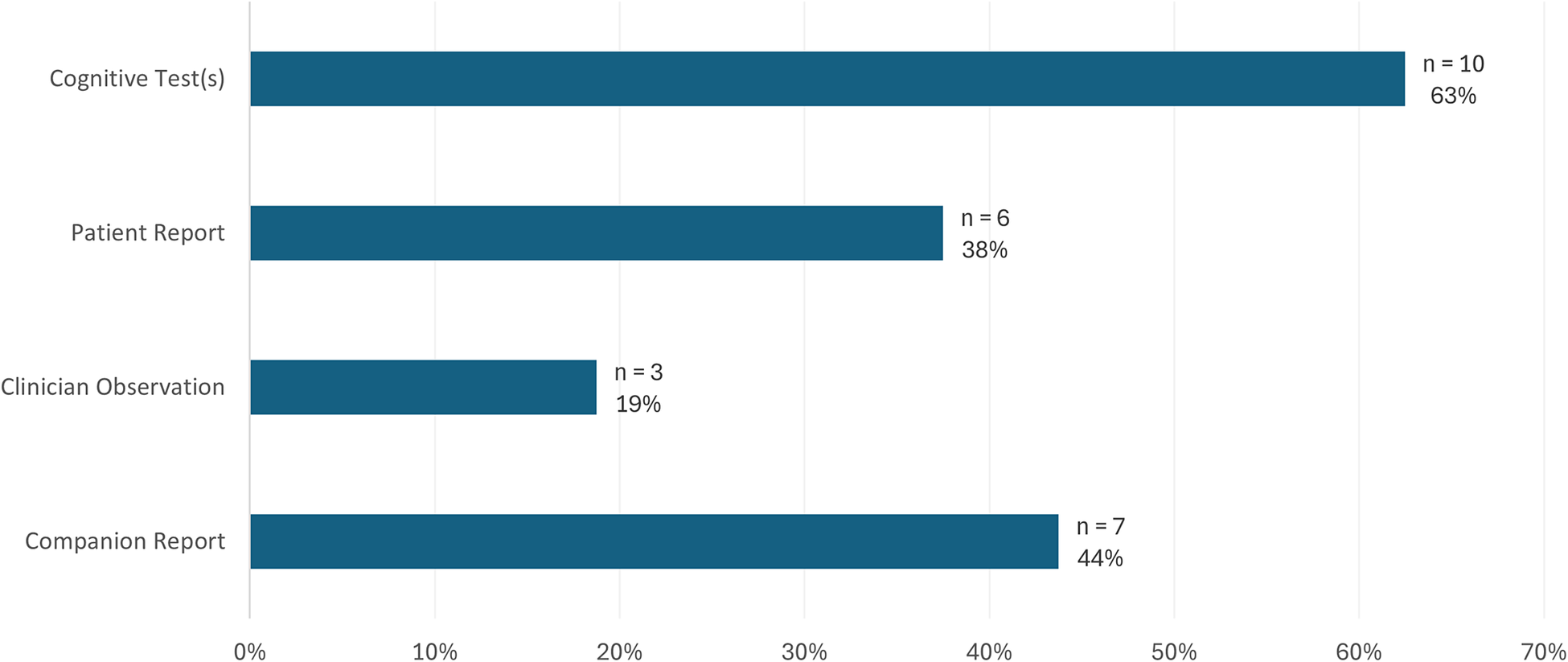
Cognitive symptom identification relied on various sources across studies. Figure 3 illustrates the frequency and proportion of studies that utilized each source. Notably, nine of the 16 studies (Chow et al., Reference Chow, Pio and Rockwood2011; Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Johannessen et al., Reference Johannessen, Helvik, Engedal and Thorsen2017; Panegyres & Frencham, Reference Panegyres and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008) relied on a single source for symptom identification – six of these studies (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Panegyres & Frencham, Reference Panegyres and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Serrano et al., Reference Serrano, Dillon, Heisecke, Castro, Perez Leguizamon, Allegri and Taragano2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008) exclusively reported cognitive test results and three studies relied solely on companion reports (Chow et al., Reference Chow, Pio and Rockwood2011; Hornberger et al., Reference Hornberger, Shelley, Kipps, Piguet and Hodges2009; Johannessen et al., Reference Johannessen, Helvik, Engedal and Thorsen2017). Patient report was included in only six of the 16 studies (Edahiro et al., Reference Edahiro, Okamura, Arai, Ikeuchi, Ikeda, Utsumi, Ota, Kakuma, Kawakatsu, Konagaya, Suzuki, Tanimukai, Miyanaga and Awata2023; Panegyres, Reference Panegyres2021; Panegyres et al., Reference Panegyres, Graves and Frencham2007; Rosness et al., Reference Rosness, Haugen, Passant and Engedal2008; Snowden et al., Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023).
Frequency and proportion of studies utilizing each source of cognitive symptom data (n = 16).

Dementia screening instruments were used in many studies. Among the 10 studies reporting cognitive symptoms from cognitive tests, six studies (Falgas et al., Reference Falgas, Tort-Merino, Balasa, Borrego-Ecija, Castellvi, Olives, Bosch, Fernandez-Villullas, Antonell, Auge, Lomena, Perissinotti, Bargallo, Sanchez-Valle and Llado2019; Panegyres & Frencham, Reference Panegyres and Frencham2007; Rivas Nieto, Reference Rivas Nieto2014; Shelley et al., Reference Shelley, Al-Khabouri, Hussein and Raniga2007; Shinagawa et al., Reference Shinagawa, Ikeda, Toyota, Matsumoto, Fukuhara, Hokoishi, Mori and Tanabe2008; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023) utilized traditional dementia screening instruments (e.g., MMSE, MoCA, or Clock Drawing Test), with two of these studies (Rivas Nieto, Reference Rivas Nieto2014; van Gils et al., Reference van Gils, Rhodius-Meester, Leeuwis, Handgraaf, Bakker, Peetoom, Bouwman, Pijnenburg, Papma, Hoogendoorn, Schoonenboom, van Strien, Verwey, Kohler, de Vugt and van der Flier2023) depending solely on dementia screening tests.
Summary
Sixteen internationally distributed studies were included in this scoping review when mapping the literature on the presenting cognitive symptoms of EO-FTD, spanning publication dates from 2007 to 2024. Few studies had the specific aim to assess the cognitive symptoms of EO-FTD. In response to this scoping review’s first research question, 35 uniquely named cognitive symptoms were found to be experienced by adults, without other neurological conditions, prior to receiving a diagnosis of EO-FTD. Memory, attention, and executive function were the most frequently reported. In response to the second research question on how cognitive symptoms were identified, it was found that the majority of studies relied on a single source of data, with patient reports notably underrepresented. Cognitive tests provided symptom data in the majority of studies, and dementia cognitive screening tests were often utilized.
Discussion
There is a notable paucity of studies that report the specific cognitive symptoms preceding a diagnosis of EO-FTD with only 16 studies included in this scoping review. Studies were internationally dispersed enhancing generalizability but highlighted a need for more in-depth, country-specific research that explicitly aims to identify the cognitive symptoms preceding EO-FTD.
Nomenclature to describe presenting cognitive symptoms was inconsistent across studies. The wording of the 35 cognitive symptoms that were found to present prior to EO-FTD diagnosis frequently mirrored the terms used in the cognitive tests employed, resulting in a variety of labels for what appear to be similar cognitive constructs (e.g., verbal fluency with word generation; reasoning with verbal reasoning; attention with concentration). This inconsistency underscores the need for harmonization of cognitive symptom terminology across research, particularly for cross-study comparisons and clinical translation.
Broad cognitive domains such as ‘memory’ or ‘attention’ were reported as impaired in most studies rather than delineating more precise symptoms (e.g., verbal delayed memory, working memory, or prospective memory under memory; sustained attention, selective attention, or divided attention under attention). This lack of specificity is clinically important, as these more specific cognitive impairments are likely to affect daily functioning in different ways. The ambiguity that is inherent in the reporting of broad cognitive domains is also particularly problematic when reported by patients or companions because these terms can encompass a wide range of distinct cognitive functions (Rasmussen et al., Reference Rasmussen, Hellzen, Stordal and Enmarker2019). Patients and companions typically lack clinical vocabulary and rely on general language to describe changes they observe. They are unlikely to use specific terms like ‘working memory’ or ‘selective attention’, which creates a communication gap between lived experience and clinical interpretation. This mismatch in language complicates a clinician’s ability to accurately interpret reported symptoms, especially when trying to distinguish between neurodegenerative conditions with overlapping presentations (Rasmussen et al., Reference Rasmussen, Hellzen, Stordal and Enmarker2019).
These communication challenges are supported in previous research. Rasmussen et al. (Reference Rasmussen, Hellzen, Stordal and Enmarker2019) found through interviews with family caregivers of individuals with FTD that they often described early symptoms in vague or non-clinical terms, contributing to diagnostic delays. Similarly, Forbush et al. (Reference Forbush, Gundlapalli, Palmer, Shen, South, Divita, Carter, Redd, Butler and Samore2013) demonstrated how patients’ own words are frequently transformed or lost when translated into clinical terminology in documentation. Both studies underscore the need to develop a shared framework for symptom reporting that can bridge clinical and lay language. Such a framework might link functional examples of cognitive changes to the specific cognitive symptom (e.g., ‘difficulty following multi-step directions’ to illustrate working memory issues or ‘trouble focusing in conversations’ to represent sustained attention difficulties) fostering a shared understanding between patients, companions, and clinicians.
Variability in the cognitive assessments employed across studies was found in this scoping review, even when targeting the same cognitive domain. For instance, one study may assess verbal delayed memory through a word list recall (e.g., Rey Auditory Learning Test in Panegyres et al., Reference Panegyres, Graves and Frencham2007), while another uses story recall (e.g., The Wechsler Memory scale as part of neuropsychological testing in Heikkinen et al., Reference Heikkinen, Paajanen, Hanninen, Tikkanen, Hublin, Koivisto, Remes and Kruger2024); both target memory but in functionally different ways. In addition, the use of cognitive screening tests in studies, while quick to administer, inherently restricts the number of cognitive symptoms that can be identified. This can contribute to potentially missing presenting cognitive symptoms of EO-FTD that are either not assessed or inadequately assessed by these screening tests (e.g., executive function, a common symptom of EO-FTD) (Cavedoni et al., Reference Cavedoni, Chirico, Pedroli, Cipresso and Riva2020). Even in studies where more detailed neuropsychological testing was reported to have been performed, the specific tests administered were not consistently reported. In addition, while overall scores were often reported in studies as they facilitated group comparisons, aggregate scores lack the granularity needed for a more nuanced clinical understanding of presenting cognitive symptoms. In contrast, the Snowden et al. (Reference Snowden, Thompson, Stopford, Richardson, Gerhard, Neary and Mann2011) study which employed the Manchester Neuropsychological Profile, a bespoke assessment instrument designed to characterize different forms of dementia, offered the most detailed list of initial cognitive symptoms, underscoring the potential value in employing comprehensive cognitive assessment tools in research and clinical practice.
The source of cognitive symptoms within a study also often lacked diversity with the majority of studies relying on a single source of cognitive symptoms data, often cognitive tests. The lack of inclusion of first-person perspectives and the lack of more qualitative studies can limit the understanding of how cognitive symptoms are experienced and described outside of clinical settings – an important consideration for both clinical diagnosis and the ability to develop sensitive AI-based detection tools from clinical documentation review that could augment the clinical assessment process in future.
The lack of consistency in cognitive nomenclature, test selection, and sources across studies limits comparability and weakens the foundation for synthesizing symptom profiles that could aid in early detection or differential diagnosis of EO-FTD. Collectively, these issues expose key gaps in the current approach to characterizing cognitive symptoms that precede EO-FTD diagnosis. They highlight the need for greater standardization in both the terminology used to describe cognitive symptoms and the assessment tools and sources employed across studies.
Limitations
Several potential limitations were identified. First, only articles published in English were included in the full-text review, resulting in the exclusion of two potentially relevant studies published in French and Spanish (Croisile et al., Reference Croisile, Tedesco, Bernard, Gavant, Minssieux-Catrix and Mollion2012; Vilalta-Franch et al., Reference Vilalta-Franch, Garre-Olmo, Lopez-Pousa, Turon-Estrada and Pericot-Nierga2008). Consequently, potentially relevant research could have been omitted, introducing a language bias that may have influenced the comprehensiveness of our findings.
Second, sample sizes varied considerably across the included studies, with half reporting fewer than 25 participants. The limited sample sizes reduce the generalizability of findings and can increase the risk of over-representing or missing certain cognitive symptoms due to small study populations. Furthermore, some of the included studies were not primarily designed to characterize the presenting cognitive symptoms of EO-FTD with FTD often a subset of a broader study including more dementia types, highlighting a gap in the literature regarding the description of cognitive symptoms that precedes diagnosis of EO-FTD on an international level.
Third, the lack of differentiation in the studies by the FTD variants when reporting the presenting cognitive symptoms is another limitation. Given bvFTD, svPPA, and nfvPPA exhibit distinct cognitive profiles, the absence of subtype-specific symptom reporting may obscure meaningful clinical distinctions.
Future directions and recommendations
More standardized, granular investigations into the presenting cognitive symptoms in EO-FTD are identified as needed from this scoping review. Future studies should aim for increased sample sizes, greater language inclusivity, and clearer differentiation by FTD subtype. More detailed and transparent reporting of cognitive testing results is needed to support synthesis across studies and advance early identification and differential diagnosis of EO-FTD.
Incorporating diverse sources of symptom information such as patient report, companion report, and clinician observations in addition to cognitive tests will enhance the accuracy and richness of symptom characterization. Within studies, cognitive symptoms need to be reported more precisely than broad cognitive domains such as ‘memory’ or ‘attention’. Standardizing the use of both cognitive symptom terminology and assessment tools across research and clinical practice would further enhance and support earlier and more reliable identification of EO-FTD.
Future scoping reviews by the study team will address the presenting cognitive symptoms of other types of EOD and include broader comparisons across dementia types. Case studies will also be reviewed as their exclusion may have limited the identification of rich, detailed cognitive symptom descriptions often captured in individual case reports, particularly those based on direct observations or firsthand accounts from patients and their companions.
For clinical practice, the development of standardized and accessible cognitive profiles tailored to FTD subtypes as well as different types of dementia to aid in differential diagnosis should be prioritized. These profiles should incorporate shared nomenclature that aligns with clinical frameworks and can be used across patients, companions, and clinicians to promote clearer communication and improve early recognition. The development of supportive tools, such as visual aids, and patient-friendly symptom lists with corresponding functional activity impacts can help patients and companions more precisely articulate cognitive changes. Clinical guidelines should recommend the use of specific standardized cognitive tests appropriate to the suspected dementia types and subtypes.
Future early detection strategies will likely require integration of cognitive symptom profiles with scalable, non-invasive diagnostic tools given the current non-specificity of early cognitive symptoms in EO-FTD. Emerging approaches such as blood-based biomarkers (Antonioni et al., Reference Antonioni, Raho, Granieri and Koch2025), brain stimulation (Benussi et al., Reference Benussi, Grassi, Palluzzi, Cantoni, Cotelli, Premi, Di Lorenzo, Pellicciari, Ranieri, Musumeci, Marra, Manganotti, Nardone, Di Lazzaro, Koch and Borroni2021), and other minimally invasive techniques that have shown promise for population-level screening may help refine referral pathways when combined with sensitive cognitive characterization. Moreover, this underscores the importance of developing well-defined cognitive phenotypes that can be meaningfully integrated into multimodal diagnostic frameworks.
Conclusions
This scoping review reveals several critical insights and persistent gaps in the current literature on the presenting cognitive symptoms of EO-FTD. Although 35 distinct cognitive symptoms were identified, the terminology used to describe these symptoms varied across studies often resulting in symptoms that appeared to overlap in cognitive constructs. Most studies did not precisely delineate the impaired cognitive domains, and symptom data were most often from a single source, typically cognitive tests, both limiting the depth and contextual understanding of EO-FTD. Overall, these insights point to the need for the development of a unified, standardized framework for cognitive symptom nomenclature to foster a shared understanding among patients, companions, and clinicians. Greater standardization and consistency in cognitive test selection are needed, as well as the integration of more comprehensive, methodologically diverse approaches, including qualitative methods. Addressing these gaps to better capture and standardize the complex presentation of the cognitive symptoms that precede the diagnosis of EO-FTD can assist to improve timely and accurate diagnosis.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0714980826100634.
Funding
No funding was received for this scoping review.