Uterine infection during pregnancy predisposes to pre-term birth and may be associated with an increased overall neonatal morbidity. It remains unclear, however, how prenatal bacterial stimulation (systemic or luminal) affects the sensitivity to inflammatory responses and necrotising enterocolitis (NEC) in the intestine of pre-term neonates(Reference Aziz, Cheng and Caughey1–Reference Alexander, Gilstrap and Cox4). The immature intestine may show an increased expression of the toll-like receptor (TLR)-4 to Gram-negative lipopolysaccharide (LPS), leading to an exaggerated pro-inflammatory response via the NF-κB pathway(Reference Lotz, Gutle and Walther5, Reference Claud, Zhang and Petrof6). In the immediate postnatal period of term neonates, this innate immune response to LPS is down-regulated, facilitating mucosal tolerance towards Gram-negative bacteria and general bacterial colonisation(Reference Lotz, Gutle and Walther5, Reference Claud, Zhang and Petrof6). Insufficient developmental down-regulation of the TLR-4-mediated response to Gram-negative bacterial stimuli may at least in part explain the exaggerated inflammatory response leading to NEC in pre-term neonates.
An early exposure to bacterial stimuli may have the potential to enhance epithelial immune development, as suggested by improved postnatal lung maturation and the reduced respiratory distress syndrome following uterine infection in human pregnancy(Reference Goldenberg, Andrews and Faye-Petersen7–Reference Fung, Bawden and Chow9). Correspondingly, intra-amniotic LPS (LPSa) or IL-1 injection of fetal lambs, mimicking a uterine infection, is associated with an increased number of inflammatory cells in the amniotic and lung fluid, increased surfactant production and increased compliance and lung gas volume(Reference Kallapur, Moss and Ikegami10–Reference Newnham, Shub and Jobe12). The effect was a result of direct epithelial stimulation when the fetus inhaled and swallowed the amniotic fluid with bacterial and inflammatory compounds(Reference Newnham, Moss and Kramer11). A corresponding epithelial stimulation and increased structural, functional and immunological development could occur in the gastrointestinal tract and affect the NEC susceptibility of the immature intestine. A systemic stimulation with relatively high doses of LPS was associated with acidosis and death in fetal lambs(Reference Newnham, Moss and Kramer11) and with NEC in pigs and rats(Reference Ewer, Al-Salti and Coney13, Reference Giannone, Nankervis and Richter14), indicating that the route of bacterial exposure (luminal or systemic) may influence the postnatal response. Such pro-inflammatory responses may be modified by diet just after birth as shown in pre-term pigs, where the mother's colostrum with its high concentrations of trophic and immunomodulatory compounds stimulates gut function and suppresses pro-inflammatory mediators and NEC development relative to formula(Reference Bjornvad, Thymann and Deutz15–Reference Van Haver, Sangild and Oste18). A pre-term neonate subjected to prenatal systemic infection may thus benefit from colostrum feeding to counteract the pro-inflammatory bacterial insults.
Given this background, the present study was designed to test whether LPS administration to fetal piglets affects gut development and immunological responses just after birth, and how different diets (formula or colostrum) may influence this response. Based on the effects reported previously for neonatal lung function in lambs, we hypothesised that prenatal LPSa stimulation of fetal pigs would improve gut development, modulate the pro-inflammatory responses and improve bacterial tolerance, and thereby reduce the diet-dependent NEC sensitivity of the immature intestine. Conversely, we hypothesised that intramuscular LPS injections might increase NEC sensitivity. In the present initial study, we tested relatively low doses of intramuscular and LPSa, considering the high risk of premature labour and fetal death following intra-fetal surgery and LPS administration in late gestation. Structural, functional and immunological indices of the gut tissue were measured to identify the effects of intra-fetal LPS administration as well as of the postnatal diet.
Methods and materials
Prenatal surgery and lipopolysaccharide injections
All procedures in the animal protocol mentioned below were approved by the National Committee on Animal Experimentation, Denmark. At 85–87 % gestation, five pregnant sows (Large White × Danish Landrace) underwent laparatomy after sedation (azaperone), anaesthesia (propofol and isoflurane after intubation) and systemic (fentanyl) and local (lidocaine) analgesia treatment. The uterus was exposed, and in total seventy-two live fetuses were injected with saline or LPS from Escherichia coli serotype K-235 (Sigma-Aldrich, St Louis, MO, USA) through the uterine wall (Fig. 1). Fetuses (estimated body weights 1·0 kg) were injected intramuscularly in the hind leg with either 0·5 ml saline (n 27) or 0·5 ml low-dose LPS (0·014 mg; n 23), or they were injected into the amniotic fluid via the oral cavity with 1 ml high-dose LPS (total 0·4 mg, n 22). Each pig was chipped for postnatal recognition, and the uterus and fetuses were put back into the abdominal cavity and the flank was closed by sutures. The sows were returned to the stables and treated daily with broad-spectrum antibiotics (amoxycillin intramuscularly) and progesterone (50 mg) to avoid infections and spontaneous birth before caesarean delivery.
Schematic overview of the fetal interventions and the groups of postnatal pre-term pigs. Pig fetuses received saline or lipopolysaccharide (LPS) injections (intra-amniotic or intramuscular, (A)) 3–5 d before pre-term delivery by caesarean section (B) and fed colostrum (C) or formula (F) for 2 d postnatally (C). For the tissue analyses, data from the intramuscular saline and LPS groups were pooled according to the diet (control-C and control-F) and compared with formula-fed pigs receiving intra-amniotic LPS (LPSa-F).

Caesarean section and postnatal feeding protocol
Caesarean section was planned to take place no later than 7 d after the fetal surgery, unless signs of pre-term labour developed beforehand, leading to sedation, anaesthesia and immediate caesarean delivery as described earlier(Reference Bjornvad, Thymann and Deutz15, Reference Sangild, Siggers and Schmidt16). A total of sixty-one pigs were delivered and immediately transferred to temperature- and oxygen-regulated incubators (Air-Shields, Hatboro, PA, USA). Of the sixty-one pigs, one died soon after birth from complications not related to NEC, leaving sixty pigs to be included in the final analyses. Blood samples were drawn from the umbilical cord at caesarean section and at 6 h after birth, and from the heart at euthanasia to measure blood gases, and plasma haptoglobin and cortisol, as described previously(Reference Bjornvad, Thymann and Deutz15, Reference Sangild, Siggers and Schmidt16).
Following our standard rearing and feeding protocol(Reference Bjornvad, Thymann and Deutz15, Reference Sangild, Siggers and Schmidt16), the pigs were fitted with orogastric catheters (6F; Pharmaplast, Roskilde, Denmark) and fed full enteral nutrition (15 ml/kg per 3 h) for 2 d until euthanasia (pentobarbitone: 60 mg/kg, intravenously) and tissue collection. Pigs injected intramuscularly were fed either cows' colostrum (n 6 for saline and n 5 for LPS injection, respectively) or infant formula (n 18 for saline and n 14 for LPS injection, respectively) (Fig. 1). All pigs given LPSa were fed formula after birth (n 17). The formula was made of three commercial products (per litre of water: 80 g Pepdite 2-0, 70 g Maxipro and 75 ml Liquigen-MCT, kindly donated by SHS International, Liverpool, Merseyside, UK). The macronutrient content was designed to match the composition of sows' milk and contained per litre of formula: energy, 4140 kJ; protein (mainly whey protein), 64 g; carbohydrate (mainly glucose), 45 g; fat, 61 g (saturated, 44 g; monounsaturated 10 g; polyunsaturated, 4 g); Na, 0·30 g; K, 0·64 g; Ca, 0·59 g; P, 0·42 g(Reference Bjornvad, Thymann and Deutz15). Bovine colostrum was collected as the first milking after parturition from dairy cows (Holstein Friesian, Gjordslev Gods, Denmark) and diluted with 30 % water to achieve an energy content similar to that in the formula.
Clinical evaluation and tissue collection
Pigs were checked every 3 h for NEC symptoms (bloody diarrhoea, lethargy, respiratory distress and abdominal distension). When these symptoms were evident, pigs were euthanised and tissue was collected. All remaining pigs were euthanised on day 3 (41–57 h postpartum). We evaluated five gastrointestinal regions (stomach, proximal, mid and distal small intestine and colon) for NEC lesions and scored 1–6 according to our standard macroscopic NEC pathology evaluation: (1) absence of lesions; (2) local hyperaemia, inflammation and oedema; (3) hyperaemia, extensive oedema and local haemorrhage; (4) extensive haemorrhage; (5) local necrosis and pneumatosis intestinalis; (6) extensive necrosis and pneumatosis intestinalis(Reference Bjornvad, Thymann and Deutz15). NEC was defined as a score of minimum 3 in minimum of one region. Organs (stomach, small intestine, colon, liver, spleen, kidneys, lungs and heart) were weighed, and the tissues from three small-intestinal regions and from the liver were sampled and snap-frozen for functional and immunological analyses. Samples from the distal small intestine were formaldehyde-fixed and paraffin-embedded to investigate mucosal structures. Degree of lung expansion was determined as lung density (weight/volume), with volume recorded by water displacement.
Mucosal permeability, villus structure and digestive function
Intestinal permeability was tested, as described previously(Reference Bjornvad, Thymann and Deutz15), as the post-mortem urine ratio of lactulose and mannitol after oral administration of the two sugars (0·5 and 0·3 g/kg, respectively) 4 h before euthanasia. Dimensions of villi, crypts and non-mucosal tissue (submucosa, lamina muscularis and serosa) in the distal small intestine were measured (SoftWoRx Explorer version 1.1; Applied Precision, Issaquah, WA, USA) on scanning pictures obtained with an ArrayWoRxe microarray scanner (Applied Precision). The activity of mucosal disaccharidases and peptidases was analysed as described previously(Reference Sangild, Siggers and Schmidt16).
Systemic and mucosal immune parameters
Plasma levels of the acute-phase protein, haptoglobin, at birth, 6 h and at euthanasia were determined by a sandwich ELISA using an in-house mouse anti-porcine haptoglobin monoclonal catching antibody and a commercial rabbit anti-human haptoglobin detection antibody (DAKO A0030; DAKO, Glostrup, Denmark), as described previously(Reference Sorensen, Tegtmeier and Andresen19).
Gene expression in the distal small intestine and liver was investigated by an in-house immune-focused mRNA Micro Array POM4 chip with 373 oligonucleotides, representing more than 250 different genes (platform accession no. GPL7576) following the procedures described previously(Reference Skovgaard, Mortensen and Boye20). In short, total RNA was extracted (RNeasy Midi Kit with TRIzol reagent; Qiagen, Ballerup, Denmark) from the tissues from saline-injected formula-fed pigs (n 9), saline-injected colostrum-fed pigs (n 5) and LPSa-injected formula-fed pigs (n 8). RNA quality and quantity were determined on a Nano Chip 6000, Agilent 2100 bioanalyser (Agilent Technologies, Nærum, Denmark) and on a Nanodrop ND-1000 spectrophotometer (Saveen and Werner AB, Limhamn, Sweden), respectively, before cDNA synthesis (QuantiTect Reverse Transcription Kit; Qiagen). Samples and a reference pool of all samples were labelled (Oyster 550 and 650, respectively) using 3DNA Array 900 expression array detection kits (Genisphere, Hatfield, PA, USA). Hybridisation, washing and scanning were performed as described previously(Reference Skovgaard, Mortensen and Boye20). Images were processed in GenePixPro 6.0 (Molecular Devices Corporation, Sunnyvale, CA, USA) and analysed using Acuity 4·0 (Molecular Devices Corporation), revealing pairwise differences in gene expression among treatment groups and between sick and healthy pigs.
Tissue levels of TLR-4, IL-1 receptor-associated kinase-1 and IκB were investigated by immunoblotting analysis as described previously(Reference Burrin, Stoll and Guan21) by separation on denatured SDS-PAGE gels (7 % for TLR-4 and IL-1 receptor-associated kinase-1 (IRAK1), and 15 % for IκB) and overnight incubation with a primary antibody (Table 1) on nitrocellulose membranes followed by 1 h incubation with a secondary antibody (goat anti-rabbit IgG–horseradish peroxidase or goat anti-mouse IgG–horseradish peroxidase, 1:5000; Santa Cruz Biotechnology, Inc.). Blots were visualised with the horseradish peroxidase substrate (ECL-plus; Amersham Biosciences, Piscataway, NJ, USA), scanned and quantified by ImageQuant 5.0 software (Molecular Dynamics; Amersham Biosciences, Sunnyvale, CA, USA).
List of antibodies used for Western blotting
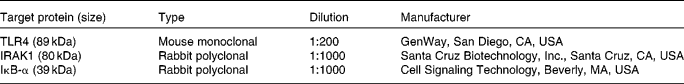
TLR, toll-like receptor; IRAK1, IL-1 receptor-associated kinase-1.
Activity of NF-κB was determined on nuclear extracts from the distal small intestine using the TransAM p65 NF-κB Transcriptions Factor Assay Kit (Active Motif, Carlsbad, CA, USA). Following the manufacturer's protocol, nuclear extracts were obtained, and duplicates of 2–20 μg protein were added into the ninety-six-well plate together with different dilutions of the manufacturer's positive control. After incubation with the NF-κB p65 antibody, detection with a secondary antibody and development, following the manufacturer's protocol, absorbance was read on a SpectraMax 340 microplate spectrophotometer (Molecular Devices Corporation) at 450 and 655 nm.
Calculations and statistical analyses
All results are presented as means with their standard errors. Group differences in NEC incidences were tested using Fisher's exact test (SAS/STAT version 9.1; SAS Institute, Cary, NC, USA). The PROC MIXED procedure of a two-way ANOVA of SAS, with treatment, diet and sex as fixed variables, and pigs and sows as random variables, was used to test the mean differences among treatment groups and between pigs with and without NEC for all remaining parameters. After the initial analyses showing no effect of intramuscular LPS injections, we used for the additional analyses a reduced model incorporating only diet (colostrum and formula), sex and LPS (with or without amniotic LPS administration) as the fixed effects. mRNA microarray data were analysed in Acuity 4.0 (Molecular Devices Corporation) as described earlier. Differences in the number of differentially regulated genes after Acuity treatment comparisons were tested using Fisher's exact test (SAS). For all analyses, P = 0·05 was used as the critical level of significance.
Results
Clinical outcomes
All five sows showed signs of labour 3–5 d after the fetal surgery, leading to immediate caesarean delivery. Survival of the injected pigs was high for four of the sows with only one or two stillborns, while eight of the fifteen pigs from one sow were stillborn. A total of sixty viable pigs were included in the further study (Fig. 1). Gestational age at delivery was 101–106 d among the five litters, and the mean birth weight was 0·9–1·3 kg across the litters. Cortisol levels at birth did not differ among litters or treatment groups (73·2 (sem 2·4) ng/ml). The mean postnatal lifetime was 46·1 (sem 1·2) h.
The clinical, structural, functional and immunological parameters shown in Table 2 did not differ among the groups of pigs that received intramuscular injections with saline or LPS for any of the diet groups (colostrum and formula). Hence, the intramuscular LPS dose appeared to be too low to elicit any significant effects, and for each diet, we pooled pigs given intramuscular LPS or saline in utero to form two larger control groups according to diet (control-colostrum (control-C), n 11 and control-formula (control-F), n 32; Fig. 1). The pigs given the higher LPSa dose, LPSa-formula (LPSa-F, n 17), remained a separate group, as these pigs differed in several clinical and functional parameters from the control-F and control-C groups. At the time of tissue collection, none of the control-C pigs had any NEC lesions in the gastrointestinal organs. NEC incidence in the LPSa-F group was 18 % (three out of seventeen) v. 38 % (twelve out of thirty-two) in the control-F group (P = 0·2), while the NEC lesion score (Fig. 2(A)) was significantly reduced in the LPSa-F v. control-F group (P < 0·05). The control-C pigs lost less weight after birth (32 (sem 19) g) than the control-F (115 (sem 13) g) and LPSa-F pigs (97 (sem 12) g, both P < 0·05) and had lower relative liver weight (25·7 (sem 0·6) v. 30·6 (sem 1·1) g/kg (LPSa-F) and 30·8 (sem 0·8) g/kg (control-F), both P < 0·05). Otherwise, relative organ weights and relative length of the small intestine did not differ among the three groups. Lung density was significantly higher in the control-F pigs than in the control-C and LPSa-F pigs, which did not differ (0·83 (sem 0·02) v. 0·77 (sem 0·02) and 0·78 (sem 0·02) g/ml, respectively; Fig. 2(D)).
(Mean values with their standard errors)

LPS-im-F, formula-fed pigs given intramuscular LPS injections; saline-im-F, formula-fed pigs given intramuscular saline injections; LPS-im-C, colostrum-fed pigs given intramuscular LPS injections; Saline-im-C, colostrum-fed pigs given intramuscular saline injections; NEC, necrotising enterocolitis; SI, small intestine; ApN, aminopeptidase N; ApA, aminopeptidase A; DPP IV, dipeptidyl peptidase IV.
* Mean values were compared between the two groups of formula-fed pigs and between the two groups of colostrum-fed pigs, and none were found to be significant (ANOVA).
† Loss of body weight from birth to killing.
‡ Mean lesion score across five gastrointestinal regions.
§ Organ weight relative to body weight at killing.
∥ Mean values for the proximal, mid and distal small intestine. U = μmol hydrolysed substrate per min at 37 °C.
¶ Villus height in the distal small intestine.
** Values in the whole blood at euthanasia.
(A) Necrotising enterocolitis lesion score, (B) villus height in the distal small intestine, (C) intestinal permeability as indicated by urinary lactulose:mannitol ratio, (D) lung density and (E) plasma haptoglobin levels at birth, 6 h after birth and at the time of tissue collection. Values are means, with their standard errors represented by vertical bars. a,b Mean values with unlike letters were significantly different between groups (P < 0·05). ![]() , Control-formula;
, Control-formula; ![]() , intra-amniotic lipopolysaccharide; □, control-colostrum.
, intra-amniotic lipopolysaccharide; □, control-colostrum.

Blood gas values at birth and at 6 h were not different among the three treatment groups, but at euthanasia, pigs with NEC, compared with healthy pigs, showed marked acidosis with low pH (7·01 (sem 0·12) v. 7·34 (sem 0·02)), low HCO3 levels (9·0 (sem 2·0) v. 25·2 (sem 1·2) mmol/l), and high lactate (9·2 (sem 2·3) v. 1·6 (sem 0·2) mmol/l) and K levels (6·3 (sem 1·1) v. 3·9 (sem 0·1) mmol/l). At birth and 6 h, only low haptoglobin levels could be detected, but the levels increased thereafter and were significantly higher in formula-fed v. colostrum-fed pigs at 2 d (Fig. 2(E)).
Mucosal integrity, structure and digestive function
Intestinal permeability was threefold higher in the control-F pigs, relative to the control-C pigs (P = 0·08), with intermediate values for the LPSa-F group (Fig. 2(C)). Villus height in the distal small intestine showed the opposite trend with highest values in the control-C pigs, lowest values in the control-F pigs (P < 0·05 relative to control-C) and intermediate values in the LPSa-F pigs (Fig. 2(B)). Likewise, the villus height:crypt depth ratio was significantly higher in colostrum-fed pigs (control-C: 5·1 (sem 0·4)) than in formula-fed pigs (3·9 (sem 0·4) in the control-F and 4·3 (sem 0·3) in the LPSa-F groups, both P < 0·01). Compared with healthy formula-fed pigs, formula-fed pigs with NEC had shortened villi (200 (sem 49) v. 531 (sem 31) μm), lowered crypts (65 (sem 10) v. 109 (sem 5)) and a thinner layer of non-mucosal tissue (88 (sem 6) v. 105 (sem 3) μm, all P < 0·01).
Except for sucrase activity, which was highest in the LPSa-F pigs (0·22 (sem 0·03) v. 0·15 (sem 0·02) U/g in the proximal region and 0·36 (sem 0·09) v. 0·23 (sem 0·03) U/g in the middle region, both P < 0·01) (where 1 U = 1 μmol hydrolysed substrate per min at 37 °C), activity of disaccharidases and peptidases across all three regions of the small intestine was highest in colostrum-fed pigs (Fig. 3). LPSa increased the mean lactase, aminopeptidase A and N activities of formula-fed pigs, but the effects reached statistical significance only for lactase activity in the proximal and middle intestine (14·2 (sem 1·9) for LPSa-F pigs v. 11·2 (sem 1·8) U/g for control-F pigs, P < 0·05).
Brush-border enzyme activities across three regions of the small intestine. Values are means, with their standard errors represented by vertical bars. a,b Mean values with unlike letters were significantly different between groups (P < 0·05). ApN, aminopeptidase N; ApA, aminopeptidase A; DPP IV, dipeptidyl peptidase IV. ![]() , Control-formula;
, Control-formula; ![]() , intra-amniotic lipopolysaccharide; □, control-colostrum. 1 U = 1 μmol hydrolysed substrate per min at 37 °C.
, intra-amniotic lipopolysaccharide; □, control-colostrum. 1 U = 1 μmol hydrolysed substrate per min at 37 °C.

Mucosal immune parameters
Based on mRNA microarray analysis of the small intestine, treatment group comparisons revealed pairwise differences in gene expression due to prenatal treatments, postnatal diet regimens and disease states. Due to relatively high tissue complexity and small differences in expression levels, we judged that the microarray analysis was too insensitive to detect quantitatively the up- or down-regulation of individual genes. We therefore used the total number of differentially expressed genes in the intestinal and hepatic tissue as the main outcome measure for specific group comparisons. These analyses showed that type of diet (formula or colostrum) had a greater influence on the number of differentially expressed genes compared with the presence or absence of LPSa treatment, as six intestinal genes were differentially expressed between the LPSa-F and control-F pigs, while thirty-three genes were differentially regulated between the control-C and control-F groups (Fig. 4(A), P < 0·05). Mainly genes involved in apoptosis, inflammation and acute-phase responses in the small intestine were affected by formula-feeding. LPSa administration appeared to compensate for the adverse effects of formula, since only seventeen intestinal genes were differentially regulated between the LPSa-F and control-C pigs (Fig. 4(A), P < 0·05). The fifty-five genes differentially expressed between pigs with and without NEC showed that the presence of intestinal lesions influenced immune gene regulation even more than diets (Fig. 4(A)). Mainly genes involved in apoptosis, immune response and metabolism accounted for this difference. The following genes, or groups of genes, were affected when NEC-affected intestines were compared with healthy formula-fed intestines: angiopoietin 2, Bcl2-xL, fass-associating death domain, components in TLR pathways, IL, transforming growth factor b3 and its receptor, proteasome activator, endothelial NO synthase, complement components, haptoglobin, plasminogen activating factor, transferrin, apo 1, sugar transporters (GLUT and sodium-glucose-linked transporter 1), aminopeptidase A and N, lactate dehydrogenase-c and fatty acid-binding protein. Treatment group differences in the number of differentially regulated genes in the liver were fewer than for the intestine, and acute-phase proteins dominated (Fig. 4(B)). Only twelve liver genes were differentially expressed between healthy and NEC formula-fed pigs, indicating minimal effects of NEC on hepatic immune genes. None of the included acute-phase proteins was differentially regulated between the LPSa-F and control-C pigs, neither in the intestine nor in the liver.
Different functional groups and total number of genes that were differentially expressed in the (A) small intestine and (B) liver tissue between groups of pre-term pigs. The pairwise comparisons of gene expression between prenatal and postnatal treatments (control-formula (control-F), control-colostrum (control-C) and intra-amniotic lipopolysaccharide (LPSa)-F), and between pigs with and without necrotising enterocolitis (NEC), indicate the extent of tissue immune and metabolic responses to the various treatments. The number of differentially regulated genes between pigs exposed to intra-amniotic LPS (LPSa-F), and each of the other two groups, were significantly lower than between these two groups (control-C v. control-F, thirty-three differentially expressed genes, P < 0·05 for both comparisons) and much lower than the number of differentially regulated genes between healthy pigs and pigs with NEC (fifty-five differentially regulated genes, P < 0·001). The numbers of differentially regulated genes in the liver between the groups were lower and not significantly different between different comparisons. a,b,c,d Mean values with unlike letters were significantly different between comparisons (P < 0·05). ![]() , Apoptosis;
, Apoptosis; ![]() , immune response;
, immune response; ![]() , inflammation;
, inflammation; ![]() , acute-phase response;
, acute-phase response; ![]() , metabolism;
, metabolism; ![]() , signal transduction; □, other.
, signal transduction; □, other.

Immunoblots revealed a significantly higher expression of TLR-4 in the control-C pigs compared with the two groups of formula-fed pigs, which did not differ (Fig. 5(A)). However, protein abundance of IκB, the inhibiting protein of the pro-inflammatory transcription factor, NF-κB, was higher in the control-C (14·7 (sem 1·1) units) and the LPSa-F (16·4 (sem 0·8) units) pigs. IL-1 receptor-associated kinase-1 (IRAK1) and levels of activated NF-κB in nuclear extracts did not differ among the three groups (Fig. 5(B)).
Intestinal protein abundance for (A) toll-like receptor (TLR)-4, IL-1 receptor-associated kinase-1 (IRAK1) and IκB in response to treatment or (B) the presence of necrotising enterocolitis (NEC) as determined by Western blot analysis of intestinal extracts. Values are means, with their standard errors represented by vertical bars. Relative amount of (C) active NF-κB among treatment groups and (D) between pigs with and without NEC from nuclear extracts, as determined by ELISA. a,b Mean values with unlike letters were significantly different between treatment groups (P < 0·05). * Mean values were significantly different between pigs with and without NEC (P < 0·05). ![]() , Control-formula;
, Control-formula; ![]() , LPSa, intra-amniotic lipopolysaccharide-formula; □, control-colostrum;
, LPSa, intra-amniotic lipopolysaccharide-formula; □, control-colostrum; ![]() , healthy;
, healthy; ![]() , NEC.
, NEC.

Discussion
Uterine infections during pregnancy have been associated with increased risk of pre-term birth, infection and overall neonatal morbidity(Reference Aziz, Cheng and Caughey1, Reference Goepfert, Andrews and Carlo22, Reference Satar, Turhan and Yapicioglu23). Some studies have reported elevated NEC incidence following prenatal infection, but results have been inconsistent(Reference Goldenberg, Andrews and Faye-Petersen7–Reference Fung, Bawden and Chow9). The difficulties in reaching conclusions regarding the effects of bacterial exposure before birth may be explained by the different location, type and length of exposure in utero, and the multitude of differences in clinical variables ex utero, such as feeding and antibiotic regimens. After birth, stimulation of the TLR-4 receptor by Gram-negative bacteria stimulates NEC development during the suckling period of rodents(Reference Leaphart, Cavallo and Gribar24–Reference Nanthakumar, Fusunyan and Sanderson28), while in neonatal mice, Gram-negative exposure may provide enhanced immunological tolerance(Reference Lotz, Gutle and Walther5). Thus, prenatal LPS stimulation due to intra-uterine infections may affect postnatal NEC resistance. Against this background, we hypothesised that prenatal exposure to LPS would enhance postnatal gut development and NEC resistance in pre-term pigs by providing better tolerance to gut colonisation. Consistent with our hypothesis, we found that LPSa exposure for 3–5 d before pre-term delivery modestly reduced the severity of NEC lesions, villus atrophy, intestinal permeability and damage to brush-border enzyme activities following formula-feeding. The finding that the effects were most pronounced in the proximal intestine indicates that this region was more exposed to swallowed LPS than the more distal regions. Nevertheless, our analyses of the number of differentially expressed intestinal immune-related genes show that formula-fed pigs exposed prenatally to luminal LPS were indeed affected also in the distal intestine. Hence, the number of differentially regulated genes for this group was intermediate between the numbers for control formula- and colostrum-fed pre-term pigs. The distal part is usually the region mostly affected by NEC lesions(Reference Sangild, Siggers and Schmidt16–Reference Van Haver, Sangild and Oste18), possibly due to the high density of immune cells.
Our hypothesis that an intra-amniotic stimulation with Gram-negative LPS would induce immunological development of the immature gut epithelium was partly based on the clinical observations of decreased respiratory distress following prenatal infections(Reference Goldenberg, Andrews and Faye-Petersen7–Reference Fung, Bawden and Chow9) and on the observed beneficial effects of LPSa injections on lung maturation and surfactant production in newborn lambs(Reference Kallapur, Moss and Ikegami10, Reference Newnham, Shub and Jobe12, Reference Kramer, Moss and Willet29). Intra-amniotic IL-1 administration also improved lung maturation and surfactant production in fetal rabbits(Reference Bry, Lappalainen and Hallman30). In fetal lambs, the LPS effects on the fetal lung were independent of a rise in fetal cortisol levels(Reference Jobe, Newnham and Willet31), and also in the present study, circulating levels of cortisol did not differ among groups. In lambs, LPSa induced both systemic and local inflammation with increased inflammatory cells and cytokines in the cord blood, amniotic fluid and alveolar fluid(Reference Newnham, Shub and Jobe12). This occurred as a direct result of luminal epithelial stimulation with bacterial compounds since lung maturation occurred only after LPS injection into the amniotic cavity, trachea or stomach, while systemic intra-peritoneal LPS injections caused severe acidosis or death(Reference Newnham, Moss and Kramer11). The responses in fetal lambs were dose-dependent, and doses below 2 mg/kg resulted in inflammation without tissue or organ maturation(Reference Kramer, Moss and Willet29), while fetal lambs survived after as much as 40 mg/kg into the amniotic cavity(Reference Jobe, Newnham and Willet31). In the present study, the LPSa dose of 0·4 mg/kg was probably not sufficient to elicit a full inflammatory, tolerogenic and maturational response. This, combined with the variable ingestion of the amniotic fluid by each fetus, may explain that the luminal LPS administration had a limited and somewhat variable effect in the present study. On the other hand, the significant effects on lung density values (reflecting increased alveolar expansion) and intestinal structure and function, particularly in the proximal intestine (e.g. sucrase, lactase and villous integrity), support that LPSa had physiological effects. Better control of the dose, length and site of fetal LPS exposure will be important in future experiments to clarify how LPSa administration mimics the uterine, fetal and neonatal responses to Gram-negative chorioamnionitis.
Systemic LPS injections are commonly used to induce NEC in experimental NEC animal models(Reference Ewer, Al-Salti and Coney13, Reference Giannone, Nankervis and Richter14, Reference Chan, Wong and Luk32). As our fetal pigs injected intramuscularly with LPS did not differ from the saline control pigs, the chosen dose of 0·014 mg/kg LPS appeared insufficient to induce systemic inflammation and altered postnatal development. Nevertheless, acidosis, increased TNF-α and brain injury were the result of low LPS doses (0·005–0·015 mg, intravenously) to fetal lambs(Reference Duncan, Cock and Suzuki33), while 1 mg/kg caused severe acidosis and death(Reference Newnham, Moss and Kramer11). In postnatal pigs, 0·2 mg/kg induced mild acidosis(Reference Groner34), while higher doses to newborn pigs (2 mg/kg, intravenously) resulted in decreased numbers of leucocytes and lesions similar to infant NEC(Reference Ewer, Al-Salti and Coney13). We chose relatively low doses of LPS for both administration routes, considering that injection of multiple fetuses late in gestation is associated with a high risk of spontaneous abortion. Spontaneous signs of labour occurred for all sows within 3–5 d of surgery, indicating that the modest LPS stimulation, combined with fetal surgery, was sufficient to advance parturition. Maternal anaesthesia and fetal surgery alone do not usually initiate parturition, as shown in our many earlier studies on fetal pigs at similar gestational ages(Reference Bjornvad, Schmidt and Petersen17, Reference Petersen, Burrin and Sangild35, Reference Sangild, Schmidt and Elnif36).
The present study confirms earlier observations that cows' colostrum stimulates gut development and NEC resistance(Reference Bjornvad, Thymann and Deutz15, Reference Sangild, Siggers and Schmidt16), probably via immunoregulatory, antibiotic or trophic compounds(Reference Playford, MacDonald and Johnson37). The effects of colostrum-feeding, relative to formula, were generally greater than those of prenatal LPS treatment. Beneficial effects of colostrum were also observed in newborn rats where the mother's milk rapidly reduced the high intestinal NF-κB activity(Reference De Plaen, Liu and Tian38). On the other hand, formula-feeding stimulated NEC and maintained high TLR-4 and NF-κB activity with low levels of the inhibitory protein IκB(Reference De Plaen, Liu and Tian38). In pre-term pigs, we have not found a consistent formula- and NEC-induced up-regulation of intestinal TLR-4 expression, and in the present study, TLR-4 protein was elevated in the colostrum group. Likewise, maternal milk enhanced, rather than reduced, the TLR-4-mediated responses in adult and fetal enterocytes and leucocytes in vitro (Reference LeBouder, Rey-Nores and Rushmere39). Immunoblotting revealed that protein abundance of IκB, an inhibitory protein of the TLR-4-dependent NF-κB pathway, was elevated in the LPS-exposed and colostrum-fed pigs. Hence, the exact role of TLR-4 protein and mRNA expression, and TLR-4-mediated inflammatory pathways in association with bacterial stimuli, pro-inflammatory diets and NEC remains controversial.
The results of the microarray analyses of gut and liver tissues indicated that diet exerted a main influence on the inflammatory processes, although LPSa injections also appeared to modify the number of differentially expressed genes. In general, gene regulation was less affected by diet and LPS exposure, than by the presence of NEC, as assessed by the number of differentially expressed genes. The semi-quantitative nature of the microarray analysis precluded any detailed study of single genes. Intestinal genes related to both immune function and metabolic/digestive activities were affected, and the changes in intestinal gene expression were associated with only a few changes in expression for the same genes in the liver. Nevertheless, a systemic acute-phase response was indicated by the increased plasma haptoglobin levels in formula-fed v. colostrum-fed pigs.
The pig represents an elegant animal model to investigate how the immature intestine is affected by systemic or luminal bacterial stimuli in utero. Extensive fetal manipulations are possible in the late gestation, and NEC develops spontaneously after pre-term birth and formula-feeding. We have shown that LPSa may enhance intestinal maturation, immunity and NEC resistance, although the effects at the present dose were modest compared with those of a NEC-protective diet such as cows' colostrum. More studies using different bacterial stimuli and varying doses and sites of bacterial exposure are required to better understand how prenatal bacterial products may affect the pre-term neonate, and thus influence the clinical interventions for infants being born prematurely following uterine and fetal infections.
Acknowledgements
The present study was supported by the Danish Strategic Research Council under the program of FØSU. We wish to thank Bente Synnetsvedt, Axel Kornerup Hansen, Malene Birck and Elin Skytte for their assistance with the animal procedures. We also thank Kristina Møller, Sophia Rasmussen, Joanna Amenuvor and Thomas Boye Pihl for their technical assistance with the RNA analyses. The authors have no conflicts of interest.








