LEARNING OBJECTIVES
After reading this article, you will be able to:
recognise clinical red flags that suggest a neurological aetiology in patients presenting with psychotic symptoms, and understand the risk factors and complications associated with neuropsychiatric disorders in the context of neurological diseases
identify key clinical features and psychotic symptoms in people with neurological diseases such as epilepsy, autoimmune conditions, traumatic brain injury and neurodegenerative diseases
apply the correct differential diagnostic approaches using neuroimaging and clinical evaluations in people with psychosis, particularly in the context of neurological disorders.
Psychosis is a syndrome characterised by delusions, hallucinations and thought disorders. Without proper treatment, individuals experiencing psychotic states often exhibit significant impairments in reality testing and interaction with their environment. This, in turn, leads to reduced social functioning and may be associated with high-risk behaviours – although not all individuals with psychosis pose a danger to themselves or others.
This article offers guidelines for identifying neurological diseases potentially associated with the onset of psychosis, emphasising the need for both clinical and paraclinical evaluation using technological resources such as electrophysiology, brain imaging and cerebrospinal fluid analysis via lumbar puncture. It is presented as a narrative review, supported by illustrative clinical vignettes from our experience in a tertiary care neuropsychiatry unit. Although it does not follow a systematic review methodology, the aim is to synthesise current clinical knowledge and highlight key diagnostic and therapeutic considerations based on both the literature and expert experience.
Definition of psychosis
In his Taxonomic Dictionary of Psychiatry, Jean Garrabé recounts that the term ‘psychosis’ was used by Ersnt von Feuchtersleben in 1845 ‘to designate the psychic manifestations of mental illness. The original meaning thus encompassed, within the field of psychopathology, all states characterised by the presence of psychic disorders, that is, almost all of them’ [our translation] (Garrabé Reference Garrabé1993, p. 232). Over time, the concept of psychosis was used in a narrower sense to refer to cases of mental disorder with a significant impairment in reality perception. ICD-11 (World Health Organization 2022) provides a helpful current definition: psychotic disorders are characterised by significant impairments in reality testing and alterations in behaviour as manifested in symptoms such as delusions, hallucinations, formal thought disorder (typically manifested as disorganised speech) and disorganised behaviour. In Box 1 we offer an operational definition of the term ‘psychosis’ for the purposes of differential diagnosis.
Operational definition of the term ‘psychosis’
Psychosis is a syndrome (a recognisable pattern of signs and symptoms that may have various aetiologies) defined by the demonstration that the patient experiences a loss or severe alteration of reality judgement. This can be expressed through one or more of the following cardinal symptoms:
delusions: false beliefs that are out of keeping with the individual’s cultural or subcultural norms, firmly held by the patient and resistant to criticism or evidence;
sensory hallucinations: false perceptions in any sensory modality that do not originate from real external stimuli; if the patient recognises them as false, they are referred to as ‘hallucinosis’; however, when hallucinations are interpreted with delusional conviction – meaning their content is taken as real and projected onto the external environment – they indicate a psychotic state;
conceptual disorganisation: a disturbance in the thought process due to a severe failure in logical association of ideas, manifesting as incoherent or incomprehensible speech.
This clinical pattern requires medical attention if it is intrinsically associated with:
severe subjective distress, i.e. suffering expressed by the patient, and/or
clinically significant psychosocial dysfunction, expressed as an inability to interact socially (at work, school, interpersonally, etc.), considering family and cultural standards but primarily based on the individual’s own standards, and/or
risk to self or others.
Regarding temporal course, psychosis can be acute (<1 month), subacute (1–6 months) or chronic (>6 months).
Regarding clinical course, psychosis may be self-limiting, fully or partially reversible, episodic (with or without inter-episodic recovery), permanent, static or progressive.
Differential diagnosis
Most cases of psychosis occur in the context of so-called primary psychotic disorders. These are psychopathological processes in which a secondary cause for psychosis has been reasonably ruled out. This category includes schizophrenia spectrum disorders (such as schizophrenia, schizoaffective disorder and schizophreniform disorder) and affective psychoses (such as bipolar I disorder and psychotic depression). Among the most important causes that must be ruled out in people with psychosis, the first to consider is the use of psychoactive substances such as serotonin 5-HT2A receptor agonists (lysergic acid diethylamide (LSD), mescaline, psilocybin), N-methyl-d-aspartate (NMDA) glutamate receptor antagonists (ketamine, phencyclidine) and cannabinoid receptor agonists (cannabis). Second, some general medical conditions should be considered. As seen in Table 1, the psychotic syndrome has multiple aetiologies relevant to general medicine and neurology.
Secondary psychosis due to general medical conditions by age group

Sources: Ramirez-Bermudez & Sosa Ortiz (Reference Ramirez-Bermudez and Sosa Ortiz2018); Ramirez-Bermudez (Reference Ramirez-Bermudez2022); Sachdev (Reference Sachdev, Sachdev and Keshavan2011).
Conventional computed tomography, magnetic resonance imaging (MRI) and electroencephalogram (EEG) studies in individuals with primary psychosis are expected to yield normal results. For this reason, these studies are valuable when a secondary aetiology is suspected and confirmation is needed. How do we determine which cases require neuroimaging evaluation? When studies are conducted indiscriminately, the likelihood of finding clinically significant abnormalities is very low. This suggests the need for prioritisation. A pragmatic strategy relies on red flags, which help identify priority cases where the probability of detecting clinically significant abnormalities is higher (Blackman Reference Blackman, Neri and Al-Doori2023; Reilly Reference Reilly, Butler and Abdelsamie2025). These red flags are illustrated in Box 2.
Red flags for psychosis due to general medical conditions
Atypical psychiatric pattern: requires clinical experience to detect unusual phenomenology in the pattern and morphology of symptoms, their combinations or age at onset (e.g. late-onset psychosis).
Psychiatric syndromes suggesting neurological conditions: some psychotic syndromes increase the likelihood of an underlying neurological cause, such as Capgras syndrome, reduplicative paramnesia, Cotard’s delusion/syndrome, Ekbom syndrome (delusional parasitosis) and catatonia.
Abnormal neurological examination with sensory or motor focalisation: a comprehensive neurological examination is imperative for first-episode psychosis or poorly characterised chronic psychoses with poor treatment response.
Focal neuropsychological syndrome: identification of cognitive syndromes (amnesia, aphasia, apraxia, alexia, Balint syndrome, Gerstmann syndrome, etc.) is valuable in detecting neurological pathology.
Severe or disproportionate cognitive impairment: even if patients are difficult to assess, attention, orientation and expressive language should be evaluated. Severe cognitive impairment may indicate delirium/encephalopathy or dementia.
Sudden onset of symptoms.
History of head trauma.
Poor treatment response: this criterion is valuable if patients show significant neurological side-effects to psychotropic drugs or fail to respond despite adequate trial duration and dosage.
History of seizures.
(Sources: Pollak et al (Reference Pollak, Lennox and Müller2020); Ramirez-Bermudez & Sosa-Ortiz (Reference Ramirez-Bermudez and Sosa Ortiz2018); Ramirez-Bermudez (Reference Ramirez-Bermudez2022).)
Psychosis in patients with neurological disease
In this section, we provide a summary of the main neurological conditions that cause psychotic states, as well as clinical examples from the Neuropsychiatry Clinic at the National Institute of Neurology and Neurosurgery, Mexico. Written informed consent was obtained from all patients, or their relatives (in the cases in which the patients themselves were not able to provide it) and anonymity has been preserved in all cases.
Epilepsy
The International League Against Epilepsy defines epilepsy as either two or more unprovoked seizures occurring more than 24 h apart, or a single unprovoked seizure with a recurrence risk greater than 60%. The estimated prevalence of epilepsy in high-income countries is 1%, and psychosis develops in 5–7% of individuals with epilepsy (Maguire Reference Maguire, Singh and Marson2018; de Toffol Reference de Toffol2024). People with epilepsy are twice as likely to develop schizophrenia compared with the general population, and this risk increases with age (Maguire Reference Maguire, Singh and Marson2018; de Toffol Reference de Toffol2024). Temporal lobe epilepsy is more strongly associated with psychosis than other forms of epilepsy, with a prevalence ranging from 10 to 15%. The risk of psychosis is also higher in individuals with specific risk factors: poorly controlled seizures, a left temporal epileptogenic focus, hippocampal sclerosis, neurodevelopmental disorders, early onset of epilepsy, a history of status epilepticus and a family history of psychosis or affective disorders (Maguire Reference Maguire, Singh and Marson2018).
Psychotic disorders in epilepsy are categorised based on their relationship to seizures (Maguire Reference Maguire, Singh and Marson2018; de Toffol Reference de Toffol2024):
ictal psychosis: the psychotic symptoms are a direct expression of the epileptic discharge;
postictal psychosis: psychotic symptoms occur following seizures, after a lucid interval of 24–72 h during which the individual returns to baseline functioning; this is one of the most common presentations of epilepsy-related psychosis (Trimble Reference Trimble2001);
interictal psychosis: psychotic symptoms are related to the presence of epilepsy (whether active or in remission) but are not chronologically linked to the seizures;
psychosis associated with complete seizure control (a phenomenon known as forced normalisation).
The psychotic episodes often include visual or auditory hallucinations, they are typically brief and they are more common after a seizure. Compared with primary psychoses such as schizophrenia, epilepsy-related psychosis tends to involve less disorganised thought, shorter duration and better preservation of insight. Delusions are frequently less systematised and more transient. This distinction is helpful for clinical management, as misdiagnosis may lead to unnecessary long-term antipsychotic use or overlook the need for optimised seizure control (Trimble Reference Trimble2001).
Administration of psychotropic medications, particularly in agitated and aggressive patients, may reduce the duration of psychotic episodes. In cooperative patients, antipsychotic treatment can be administered orally, such as olanzapine 2.5–20 mg/day or risperidone 0.5–6 mg/day. For non-cooperative patients with severe agitation, intramuscular treatment for rapid tranquillisation may be necessary, such as haloperidol 2–5 mg combined with lorazepam 1–2 mg. These doses are not intended for maintenance therapy but rather for the acute management of extreme behavioural disturbance. In some jurisdictions, the administration of such interventions to non-consenting individuals posing a risk to themselves or others is regulated under legal frameworks such as the Mental Health Act 1983 in England and Wales (Maguire Reference Maguire, Singh and Marson2018; de Toffol Reference de Toffol2024).
Autoimmune encephalitis
It is well-known that neuroimmunological disorders such as systemic lupus erythematosus and multiple sclerosis may cause psychotic episodes. The discovery of anti-neuronal antibodies associated with neuropsychiatric disturbances led to the scientific conceptualisation of autoimmune encephalitis (Dalmau Reference Dalmau and Graus2018). This condition has been suggested to account for about 6.5% of first episodes of psychosis (Zandi Reference Zandi, Irani and Lang2011). An international consensus established in 2019 recommends specific approaches to facilitate early recognition and treatment of psychosis of autoimmune origin (Pollak Reference Pollak, Lennox and Müller2020). Red flags, EEG and structural magnetic resonance imaging are useful for identifying patients who should undergo cerebrospinal fluid analysis, leading to the diagnosis of autoimmune encephalitis.
Clinical features that suggest autoimmune psychosis include severe cognitive disturbances (e.g. disorientation, memory loss), catatonia and atypical patterns of hallucination, especially visual or tactile. Additionally, a history of systemic or infectious symptoms, along with abnormal movements, seizures and autonomic instability, are key indicators (Najjar Reference Najjar, Steiner and Najjar2018; Pollak Reference Pollak, Lennox and Müller2020). A preliminary classification of autoimmune psychosis includes psychosis associated with neuronal autoantibodies (Table 2), psychosis in inflammatory diseases like lupus, and ‘seronegative’ cases where the clinical presentation suggests autoimmunity despite negative antibody tests (Najjar Reference Najjar, Steiner and Najjar2018). The most common form is anti-NMDA receptor encephalitis, which causes psychotic symptoms in around 80% of the cases, along with catatonia, severe cognitive impairment, seizures and dyskinesia (Espinola-Nadurille Reference Espinola-Nadurille, Restrepo-Martinez and Bayliss2022; Ramirez-Bermudez Reference Ramirez-Bermudez, Espinola-Nadurille and Restrepo-Martinez2025). However, other autoantibodies, such as anti-LGI1 (leucine-rich glioma-inactivated 1), anti-CASPR2 (contactin-associated protein 2) and anti-GABAA (gamma-aminobutyric acid A) have also been associated with autoimmune encephalitis, often with psychiatric features at onset. Recognition of this spectrum is essential for timely diagnosis and immunotherapy. Some individuals present with psychiatric symptoms without overt neurological signs, which may cause diagnostic delays and therapeutic errors in general hospitals or psychiatric wards.
Main neuronal antibodies related to autoimmune encephalitis, causing psychotic symptoms and neurological alterations

NMDA, N-methyl-d-aspartate; LGI1, leucine-rich glioma-inactivated 1; HLA, human leukocyte antigen; CASPR2, contactin-associated protein-like 2; AMPAR, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor; GABA, gamma-aminobutyric acid; CRMP5, collapsin response-mediator protein-5; D2R, dopamine D2 receptor; DPPX, dipeptidyl-peptidase-like protein-6; MGlur5, metabotropic glutamate receptor 5; IgLON5, immunoglobulin-like cell adhesion molecule 5; REM, rapid eye movement.
Sources: Dalmau & Graus (Reference Dalmau and Graus2018); Pollak et al (Reference Pollak, Lennox and Müller2020).
Targeted treatment affects the course of the illness, improving functional outcomes and reducing mortality (Titulaer Reference Titulaer, McCracken and Gabilondo2013).
Case vignette: autoimmune encephalitis
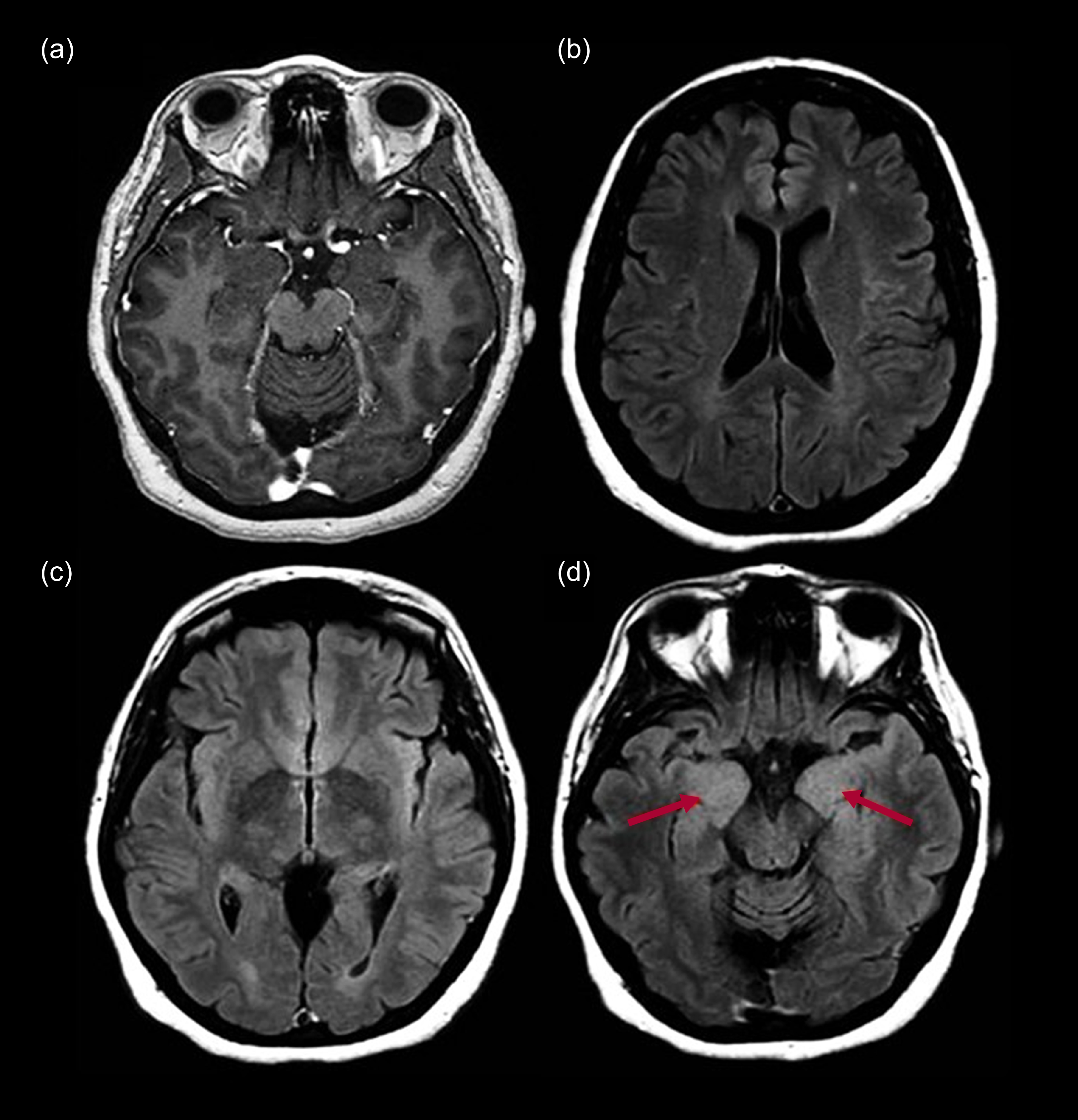
Figure 1 shows MRI scans of the brain (axial post-contrast T1-weighted, axial T2-weighted fluid-attenuated inversion recovery (FLAIR)) of a 50-year-old female patient with a confirmed diagnosis of anti-LGI1 autoimmune encephalitis admitted with psychosis. In the months prior to admission, she presented with bilateral tonic–clonic seizures, faciobrachial seizures and psychotic symptoms. These included auditory hallucinations with pejorative content and poorly structured delusional ideas of harm, as well as cognitive symptoms such as inattention and disorientation. She received treatment with methylprednisolone pulses and plasmapheresis (PLEX), along with antiseizure and antipsychotic medications. Remission of the symptoms and recovery of functional capacities was observed after several weeks. In the T1 post-contrast sequence (Fig. 1(a) and (b)), no abnormalities were observed. However, in the T2 FLAIR sequence, bilateral hyperintensity was seen at the level of the anterior cingulate cortex, bilateral insular cortex and the medial aspect of both hippocampi (Fig. 1(c) and (d)).
Magnetic resonance imaging of the brain of a 50-year-old female with anti-LGI1 autoimmune encephalitis: (a) axial post-contrast T1-weighted; (b), (c), (d) axial T2-weighted fluid-attenuated inversion recovery (FLAIR). The arrows indicate bilateral medial temporal hyperintensity.

Multiple sclerosis
This demyelinating disease predominantly affects young adults and occurs more frequently in women, with a female:male ratio ranging from 2:1 to 4:1 globally (Browne Reference Browne, Chandraratna and Angood2014). Lesions can appear at any level of the central nervous system, with a predilection for white matter tracts. The clinical course of multiple sclerosis is predominantly relapsing–remitting. Some cases can evolve into a secondarily progressive form. In addition to motor and sensory disturbances, the disease is associated with various neuropsychiatric symptoms. The most common are anxiety, depression, irritability, disinhibition and cognitive impairment. Although psychosis is uncommon in multiple sclerosis, its prevalence is two to three times higher than in the general population (Sachdev Reference Sachdev, Sachdev and Keshavan2011). Furthermore, pharmacological treatment for multiple sclerosis can induce or exacerbate psychotic symptoms, underscoring the importance of considering this manifestation in the clinical evaluation of patients (Gilberthorpe Reference Gilberthorpe, O’Connell and Carolan2017).
Case vignette: multiple sclerosis
Figure 2 shows MRI scans of the brain (axial post-contrast T1-weighted, axial T2-weighted FLAIR, sagittal T2-weighted FLAIR) of a 26-year-old female with a history of relapsing–remitting multiple sclerosis. She experienced episodes at ages 13, 14, 18, 19 and 23 years, with secondary epilepsy, undergoing treatment with prednisone pulses, alemtuzumab and antiseizure medications. At age 22, she had presented with persistent psychotic symptoms (complex visual and auditory hallucinations of a demon, as well as delusional ideas about being harmed by others and delusions of reference involving family members). As seen in Fig. 2(c) and (d), the MRI at age 26 revealed corpus callosum and white matter lesions taking the form of the classic ‘Dawson’s fingers’ finding.
Magnetic resonance imaging of the brain of a 26-year-old female with a history of relapsing–remitting multiple sclerosis: (a) axial post-contrast T1-weighted; (b) axial T2-weighted fluid-attenuated inversion recovery (FLAIR); (c), (d) sagittal T2-weighted FLAIR. The arrows indicate hyperintense lesions on the corpus callosum and white matter.

Neurodegenerative dementia
Psychosis is highly prevalent in neurodegenerative diseases. The prevalence of psychotic symptoms varies by dementia subtype. In Alzheimer’s disease, recent studies report rates of 22–54%, with a median around 41% (Morrow Reference Morrow and Pontone2024). In dementia with Lewy bodies (DLB), psychosis occurs in roughly 75% of cases – often early in the course (Atchison Reference Atchison, Wu and Watt2024). In Parkinson’s disease dementia (PDD), psychotic features are observed in about 50–60% of patients (Weintraub Reference Weintraub, Aarsland and Chaudhuri2022). In frontotemporal dementia (FTD) delusional symptoms have been reported in 38% (Pessoa Reference Pessoa, Maximiano-Barreto and Lambert2023). The specific clinical features and time at onset of psychosis are helpful in predicting the potential implicated neuropathology. Onset can be classified as occurring before, early or late in relation to the onset of the neurodegenerative disease (Naasan Reference Naasan, Shdo and Rodriguez2021). The prevalence and clinical presentation of psychosis vary significantly across subtypes, informing early diagnosis and tailored management.
Dementia with Lewy bodies
As mentioned above, psychosis occurs in approximately 75% of people who have DLB, often early in the disease course (Atchison Reference Atchison, Wu and Watt2024). Visual hallucinations are especially prominent, with delusions – frequently paranoid – fluctuating in severity. These features are exacerbated by both dopaminergic therapy and the heightened sensitivity to antipsychotics seen in DLB (Cressot Reference Cressot, Vrillon and Lilamand2024; Gibson Reference Gibson, Mueller and Stewart2024). Early psychosis manifestations, particularly visual hallucinations, have been included as a core clinical criterion in the research diagnostic criteria for prodromal DLB (McKeith Reference McKeith, Ferman and Thomas2020).
Alzheimer’s disease
In Alzheimer’s disease, psychotic symptoms – especially visual hallucinations and delusions – are observed in 22–54% of cases over the disease course (Morrow Reference Morrow and Pontone2024). These symptoms typically emerge in the middle to late stages and are linked to accelerated cognitive decline and increased caregiver burden (Cressot Reference Cressot, Vrillon and Lilamand2024; Gibson Reference Gibson, Mueller and Stewart2024). Psychosis is typically a symptom of disease progression, being more frequent as dementia severity increases (Ballard Reference Ballard, Kales and Lyketsos2020). Interestingly, the syndrome seen in behavioural-variant Alzheimer’s disease is similar to that in behavioural-variant FTD (see below) principally affects behaviour. Delusions and hallucinations are more prevalent than in behavioural-variant FTD or typical amnestic Alzheimer’s disease and these symptoms are included as a supportive clinical feature of behavioural-variant Alzheimer’s disease in the current research diagnostic criteria (Ossenkoppele Reference Ossenkoppele, Singleton and Groot2022).
Another important topic in the evaluation of Alzheimer’s disease is the assessment of complex visual hallucinations, particularly in the early stages of the disease. Their presence is usually associated with mixed Alzheimer’s disease/DLB pathology (Chiu Reference Chiu, Hsu and Wang2017). Visual hallucinations are not common in ‘pure’ Alzheimer’s disease.
The recently described criteria for limbic-predominant amnestic neurodegenerative syndrome refer to a clinical profile similar to typical Alzheimer’s disease but occurring in older adults (>75 years). These patients show a slower clinical decline and greater atrophy restricted to limbic regions. The diagnostic criteria for this syndrome do not include hallucinations or psychotic symptoms (Corriveau-Lecavalier Reference Corriveau-Lecavalier, Botha and Graff-Radford2024), but a post-mortem study of patients with ante-mortem amnestic syndrome and psychotic symptoms found that TDP-43 pathology (see next section), which is present in limbic-predominant amnestic syndrome, was the second most common underlying pathology, following mixed DLB and Alzheimer’s disease (Weintraub Reference Weintraub, Aarsland and Chaudhuri2022).
Behavioural-variant frontotemporal dementia
Overall, psychosis occurs in 10–42% of people with behavioural-variant FTD, although the rate is higher in genetic forms of the disease (Pessoa Reference Pessoa, Maximiano-Barreto and Lambert2023). Psychotic symptoms can occur at different stages of the disease, depending on the underlying pathology and genetic background. In cases associated with TDP-43 pathology – particularly those with C9orf72 hexanucleotide repeat expansion – schizophrenia-like paranoid delusions may appear as prodromal symptoms, even before prominent behavioural or cognitive changes (Ducharme Reference Ducharme, Pijnenburg and Rohrer2024).
TDP-43 pathology involves the cytoplasmic mislocalisation and aggregation of transactive response (TAR) DNA-binding protein 43 in neurons and glial cells. It is a neuropathological hallmark frequently found in genetic behavioural-variant FTD, especially in individuals with C9orf72 expansion, which is also associated with amyotrophic lateral sclerosis (ALS). In this context, early-onset psychosis – manifesting as hallucinations, delusions or behavioural disorganisation – should prompt suspicion of a genetic aetiology (Ducharme Reference Ducharme, Pijnenburg and Rohrer2024). However, in other cases of familial behavioural-variant FTD, psychosis may emerge later in the disease course, mimicking late-onset primary psychotic disorders or dementia-related psychosis. A family history of dementia or psychosis with late onset may also raise clinical suspicion of an underlying genetic form of behavioural-variant FTD (Kertesz Reference Kertesz, Ang and Jesso2013; Ducharme Reference Ducharme, Pijnenburg and Rohrer2024). This underscores the importance of considering neurogenetic evaluation of patients with either early- or atypically late-onset psychosis (Ducharme Reference Ducharme, Pijnenburg and Rohrer2024).
Parkinson’s disease
In Parkinson’s disease, psychotic symptoms may appear gradually, starting with visual illusions typically at earlier stages, progressing to visual hallucinations and finally delusions as disease progresses. An early characterisation of these visual illusions/hallucinations is important as they are a risk factor for development of dementia in Parkinson’s disease (Pagonabarraga Reference Pagonabarraga, Bejr-Kasem and Martinez-Horta2024).
The prevalence of psychosis in Parkinson’s disease is estimated to range from 16 to 23%, and up to 75% if ‘minor forms’ of hallucinations are included (Abdul-Rahman Reference Abdul-Rahman, Herrera-Calderón and Aderinto2024). Many people with Parkinson’s disease psychosis (PDP) retain insight into their hallucinations, recognising them as unreal. This preserved insight has led some authors to question whether these experiences qualify as true psychotic symptoms in the traditional sense, and whether the current diagnostic criteria for PDP adequately reflect this distinction. There has been a call for a new definition of psychosis in Parkinson’s disease (Friedman Reference Friedman2024). The presence of psychosis appears to correlate with advanced disease stages, the progression of cognitive decline, loss of function and death due to the underlying neurodegenerative processes (Davydow Reference Davydow, Okun and Pontone2025). Also, these symptoms are exacerbated by dopaminergic treatment regimes (Gibson Reference Gibson, Mueller and Stewart2024). Due to these factors, psychotic symptoms are more common in patients with Parkinson’s disease dementia. As mentioned above, they can be present in 50–60% of cases, with visual hallucinations being the most common (Weintraub Reference Weintraub, Aarsland and Chaudhuri2022).
Clozapine, pimavanserin and quetiapine are the antipsychotic drugs with the greatest evidence of efficacy for the treatment of PDP, with a lower likelihood of increasing motor symptoms. However, the USA’s Food and Drug Administration has established a warning box regarding the risk of death with the use of antipsychotics in neurodegenerative diseases (Longardner Reference Longardner, Wright and Alakkas2023). Taking this into account, we believe that neuropsychiatric assessment and therapeutic decision-making in PDP should follow some recommendations:
establish the operational definition used to define the diagnosis;
take into account the patient’s ability to retain insight;
consider the severity of the psychosis and its negative impact on daily functioning, including risky and harmful behaviour;
assess the level of distress it causes to the patient and caregivers;
consider what other neuropsychiatric symptoms it triggers, e.g. insomnia, anxiety and/or agitation;
correlate it with the patient’s cognitive status, if possible.
Huntington’s disease
Huntington’s disease is a trinucleotide repeat disorder caused by an abnormal expansion of cytosine–adenine–guanine (CAG) repeats in HTT (the gene encoding the huntingtin protein), located on the short arm of chromosome 4. It follows an autosomal dominant inheritance pattern. Typically, ≤26 CAG repeats are considered normal and not associated with the disease, whereas ≥36 are pathogenic, leading to the production of the mutant huntingtin protein.
Huntington’s disease is primarily characterised by a combination of neuropsychiatric symptoms, progressive motor disturbances and dementia (Stoker Reference Stoker, Mason and Greenland2022). Longitudinal studies have shown that signs and symptoms of Huntington’s disease can begin many years before motor symptoms are clearly diagnosed. In fact, brain changes related to the disease can be detected as early as 10–15 years prior to the onset of motor symptoms, and these changes gradually progress over time. A wide range of neuropsychiatric symptoms have been described in Huntington’s disease, including depression, anxiety, irritability, apathy, perseveration and psychosis. These symptoms often cause significant distress to patients and their families, and they have been shown to have a greater impact on functional disability and quality of life than the motor or cognitive symptoms (Thompson Reference Thompson, Harris and Sollom2012).
In an international multicentre study involving 1690 people with Huntington’s disease, four main behavioural symptom groups were identified: depression; difficulties with executive functioning and impulsivity; irritability/aggression; and a specific group for psychosis, which included symptoms such as hallucinations and delusions. Hallucinations and delusions were relatively rare, occurring in only 3% and 10% of patients respectively (Rickards Reference Rickards, De Souza and Van Walsem2011). A significant association between cognitive decline and psychosis has been observed in Huntington’s disease, which mirrors the cognitive deficits seen in schizophrenia (McAllister Reference McAllister, Gusella and Landwehrmeyer2021).
Although psychotic symptoms are less frequent than mood or motor disturbances in Huntington’s disease, they pose significant clinical challenges. Atypical antipsychotics (e.g. risperidone, olanzapine, aripiprazole) are commonly used as first-line treatments for behavioural symptoms, including psychosis and aggression. Clinical trials with olanzapine have not demonstrated significant improvements in core motor or cognitive functions; however, some reductions in behavioural disturbances have been reported. Nonetheless, owing to clinical necessity and expert consensus, antipsychotics remain a pragmatic choice in managing severe neuropsychiatric symptoms in Huntington’s disease, despite limited evidence from randomised controlled trials (Andriessen Reference Andriessen, Oosterloo and Molema2025). The following clinical vignettes illustrate two clinical scenarios of this disease.
Case vignette: newly diagnosed Huntington’s disease
A 36-year-old woman with a family history of Huntington’s disease presented with a 2-year history of 15 kg weight loss and motor symptoms such as choreiform movements in all four limbs. At consultation, she exhibited disorganised behaviour, persecutory delusions, irritability and aggression that started 18 months after the onset of motor symptoms. MRI findings revealed bilateral atrophy of the caudate nuclei and putamen, reduced cortical and subcortical volume and non-specific gliosis in the frontoparietal regions. These radiological findings correlate with the clinical diagnosis of Huntington’s disease that was confirmed by genetic testing (CAG repeats 19/51).
Case vignette: Huntington’s disease in a 65-year-old
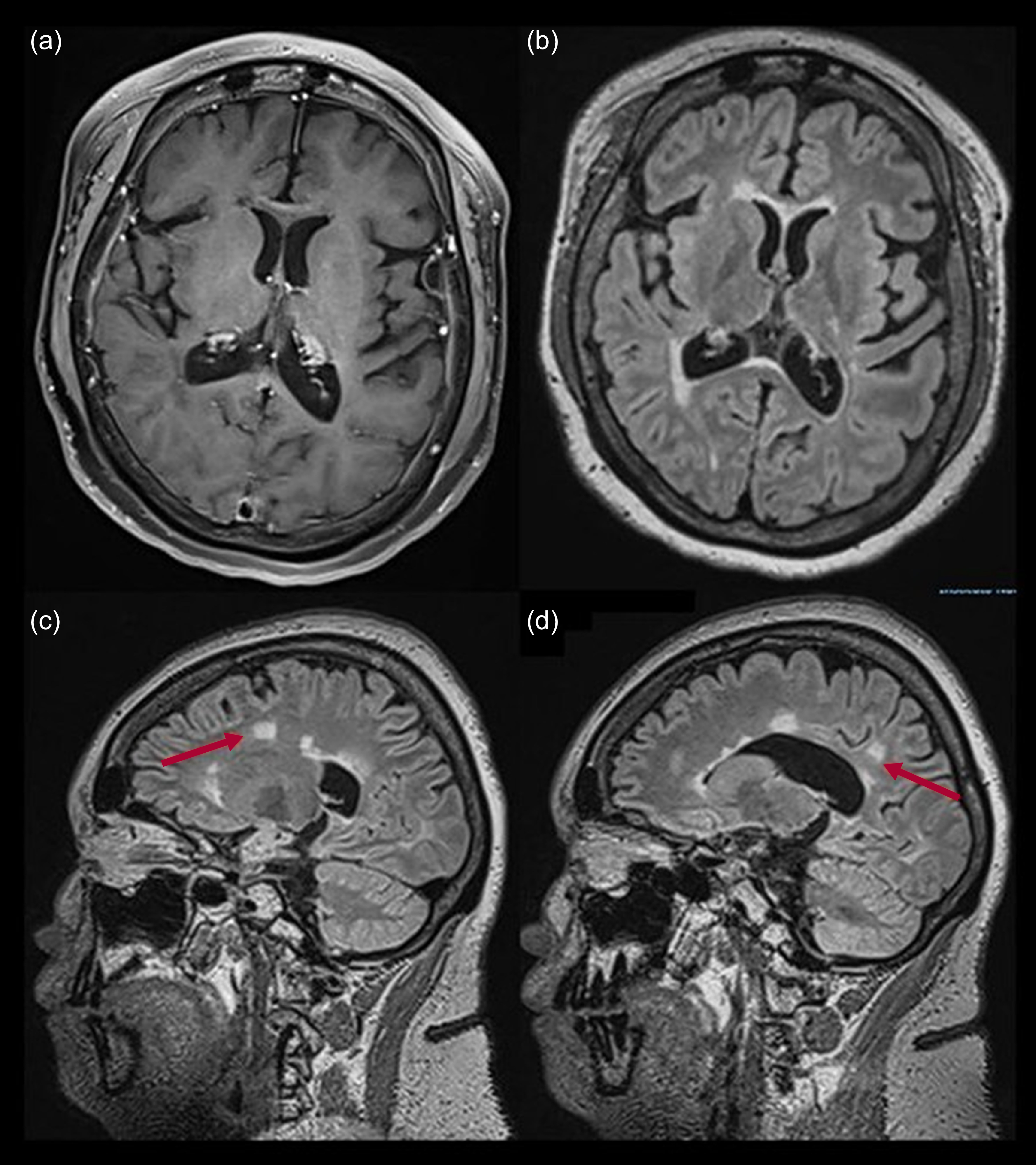
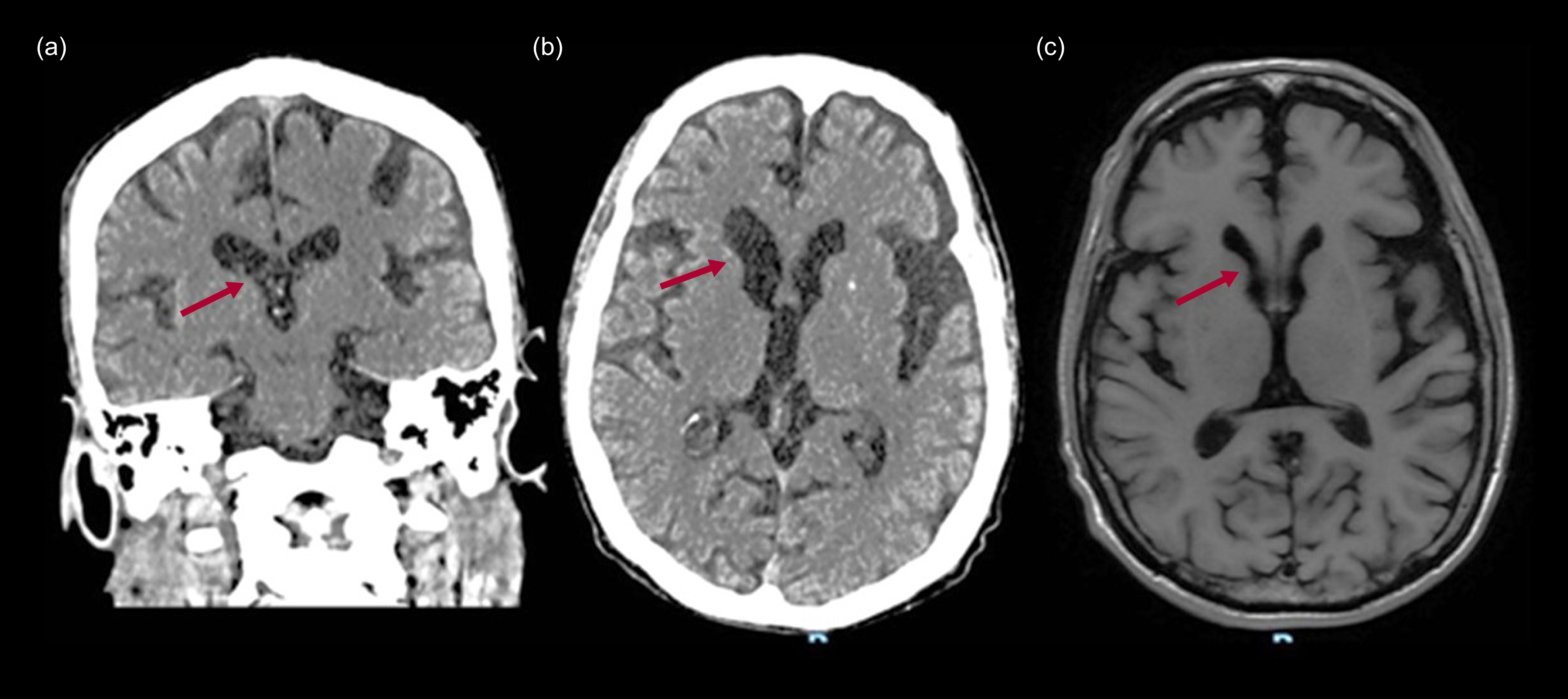
A 65-year-old male with Huntington’s disease (CAG repeats 17/41) presented with cognitive decline, psychiatric symptoms and motor dysfunction. At age 49 he had started with anhedonia and suicidal ideation. Over the years he had exhibited cognitive decline and choreiform movements that were clearly noted at age 56. Nine years later he developed persecutory delusions and increased agitation, leading to antipsychotic treatment. Computed tomography (Fig. 3(a) and (b)) revealed atrophy in the caudate nuclei and putamen, indicative of subcortical involvement, and MRI (axial T2-weighted FLAIR: Fig. 3(c)) showed atrophy in the caudate nuclei.
Computed tomography scans and magnetic resonance imaging of the brain of a 65-year-old male with Huntington’s disease: (a) coronal, (b) axial CT scans; (c) axial T1 weighted. The arrows indicate changes on the right caudate nucleus.

Creutzfeldt–Jakob disease
Creutzfeldt–Jakob disease is an irreversible and fatal cause of rapidly progressing dementia. Its prevalence is similar in both genders, with an incidence of approximately one case per million population per year and an average age at onset of 60 years. In 85% of cases, it occurs sporadically. Clinically, it is characterised by rapidly progressing cognitive decline and motor impairments, leading to severe disability and total dependence (Huang Reference Huang, Shafiian and Masi2024).
Diagnosis begins with clinical suspicion and is supported by ancillary tests such as MRI, EEG and cerebrospinal fluid (CSF) biomarkers. Among these, the real-time quaking-induced conversion (RT-QuIC) assay in CSF has become a crucial diagnostic tool owing to its high sensitivity and specificity. Neuropsychiatric symptoms such as depression, mania, and psychosis may be initial manifestations of the disease. Up to 80% of patients experience these symptoms within the first 100 days, which can delay diagnosis. Therefore, Creutzfeldt–Jakob disease should be considered in patients with MRI abnormalities, poor response to treatment and rapidly progressing cognitive impairment (Huang Reference Huang, Shafiian and Masi2024).
Case vignette: probable Creutzfeldt–Jakob disease
Figure 4 shows MRI scans of the brain (axial slices in apparent diffusion coefficient (ADC), diffusion and T2-weighted FLAIR sequences) of a 34-year-old female with probable sporadic Creutzfeldt–Jakob disease. The patient presented with a 7-month history of cognitive decline, with memory failures, language alterations consistent with anomia and phonemic paraphasias, and executive dysfunction, leading to complete dependence on family members. She also experienced psychotic symptoms consisting of auditory hallucinations of voices and the sound of a helicopter, along with ideas of harm directed at third parties ‘who were coming to hurt her’. During disease progression, generalised myoclonus and akinetic mutism developed. MRI (Fig. 4) showed diffusion restriction in the frontal, temporal and parietal cortices (b), as well as an increased signal intensity in the T2 FLAIR sequence in the same areas (c), compatible with the ‘cortical ribbon’ sign.
Magnetic resonance imaging of the brain of a 34-year-old female patient with a diagnosis of probable sporadic Creutzfeldt–Jakob disease: (a) axial T2-weighted; (b) axial diffusion-weighted imaging (DWI); (c) axial fluid-attenuated inversion recovery (FLAIR). The arrows signal changes in intensity on caudate nuclei and cerebral cortex.

Cerebrovascular disease
Cerebrovascular risk factors such as hypertension and diabetes are well-known contributors to the likelihood of dementia. White matter rarefaction or hyperintensities are a common finding in neuroimaging studies. Overall, vascular cognitive impairment may also develop neuropsychiatric symptoms by itself, the highest prevalence being for depression, apathy, irritability and anxiety. Moreover, there is an increased risk of psychotic symptoms, such as delusions, hallucinations and agitation, in people with lacunar infarcts, subcortical or both cortical and subcortical vascular dementia compared with those with vascular cognitive impairment without dementia (Tiel Reference Tiel, Sudo and Alves2015). The most commonly used evaluation tool for this clinical spectrum is the Neuropsychiatric Inventory (NPI) (Cummings Reference Cummings, Mega and Gray1994).
A systematic review of post-stroke psychosis found that it was more common in males and that in both genders onset may be delayed after the stroke. The most common type of psychosis was delusional disorder, followed in frequency by schizophrenia-like psychosis and mood disorder with psychotic features. Symptom remission was frequent after antipsychotics, even though poor functional outcomes and elevated mortality were found (Stangeland Reference Stangeland, Orgeta and Bell2018). Early detection and intervention may diminish caregiver burden and prevent complications.
Case vignette: cerebrovascular disease
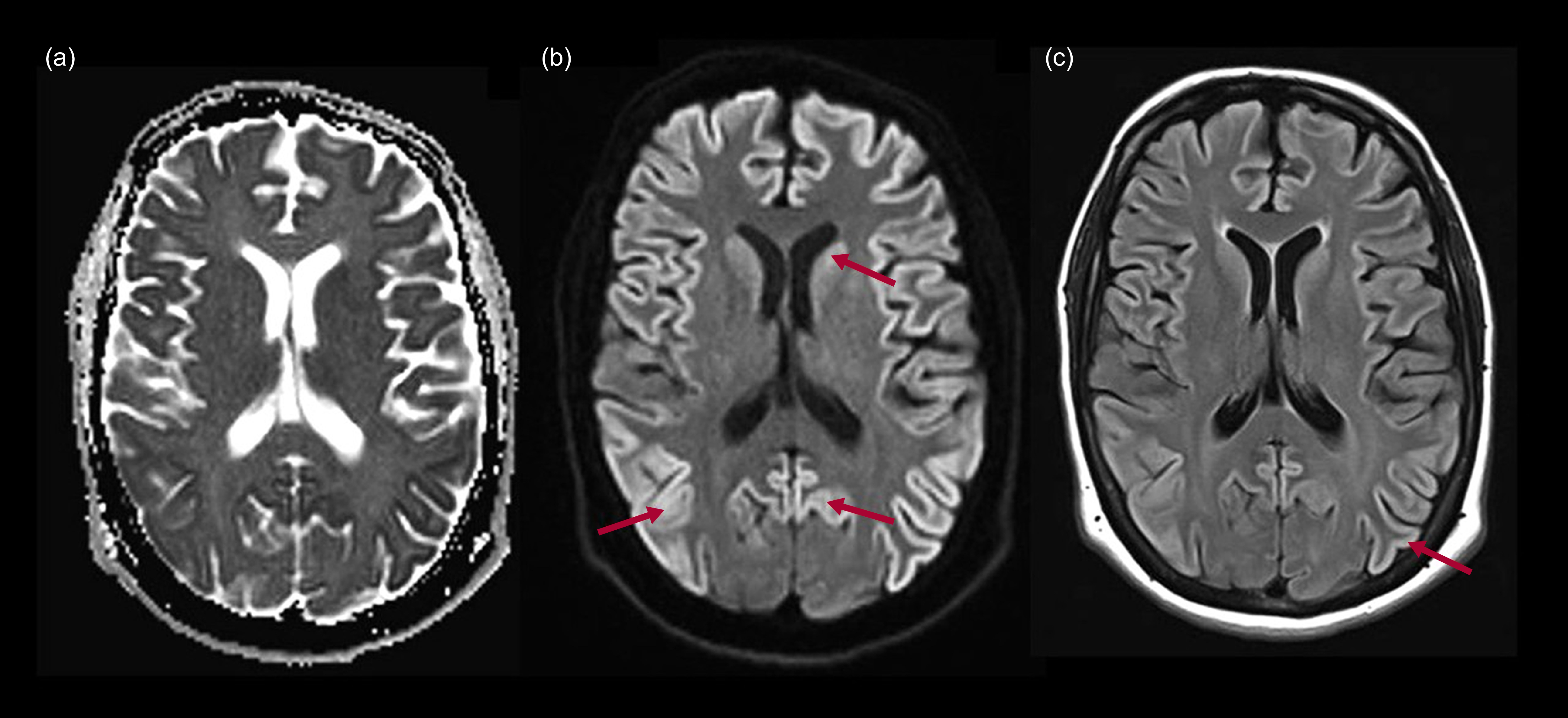
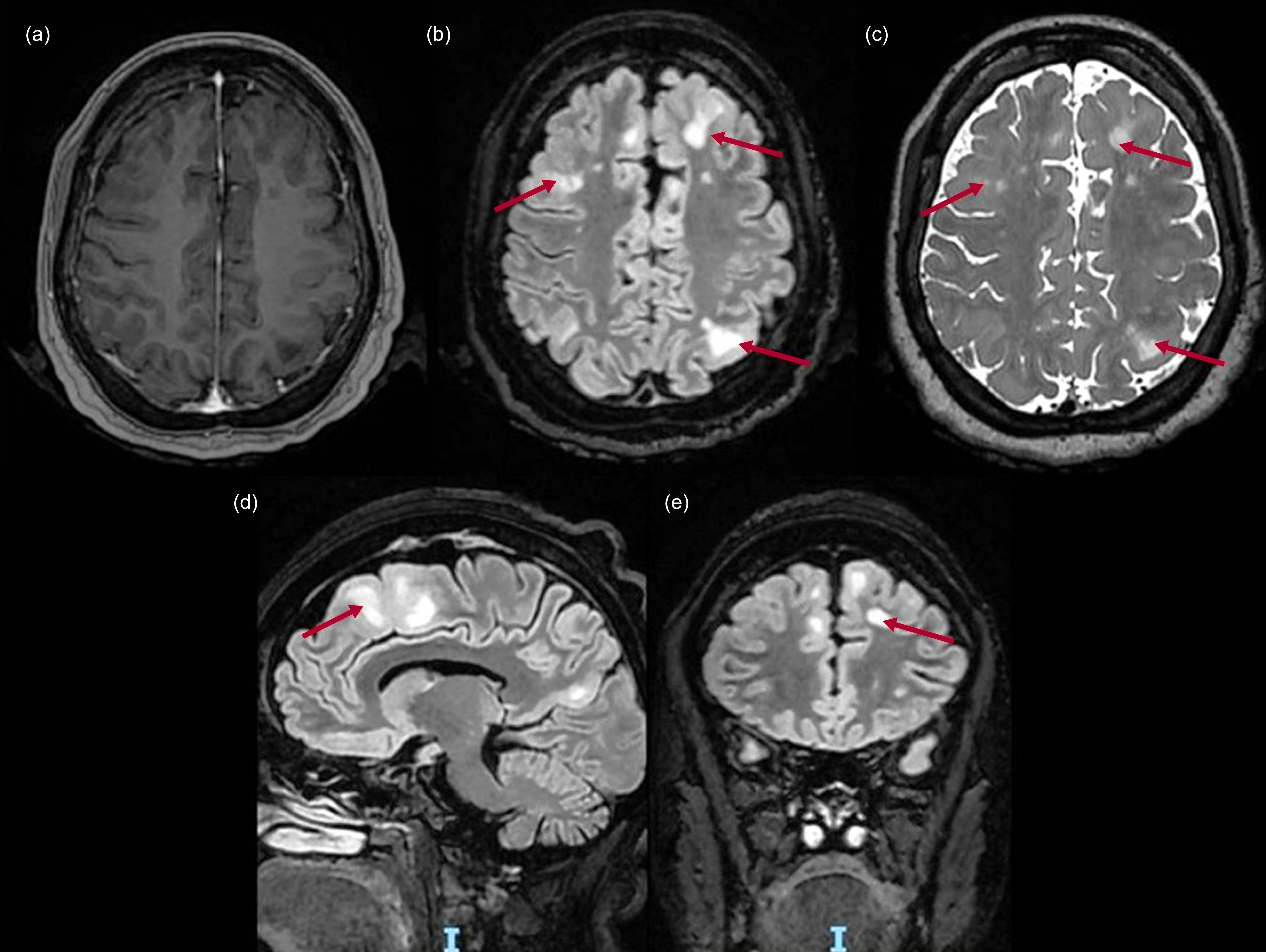
Figure 5 shows MRI of the brain of a 48-year-old woman diagnosed with anti-neutrophil cytoplasm antibody (ANCA)-positive vasculitis affecting the central nervous system. The patient presented with an acute state of altered consciousness, with psychomotor agitation and hallucinatory behaviour, as well as signs of neurological focalisation, including motor and language dysfunction. Changes in intensity secondary to vascular lesions located in the temporal and parietal lobes may be seen in T1-weighted with contrast (a, c), and axial T2-weighted FLAIR (b, d). These images are consistent with areas of encephalomalacia in the bilateral parietal, insular and temporal regions, as well as a lacunar infarct in the rostral portion of the right putamen.
Magnetic resonance imaging of the brain with contrast of a 48-year-old female patient diagnosed with anti-neutrophil cytoplasm antibody (ANCA)-positive vasculitis affecting the central nervous system and multiple vascular lesions. The arrows indicate changes in intensity secondary to vascular lesions located in the temporal and parietal lobes: (a), (c) T1-weighted with contrast; (b), (d) axial T2-weighted fluid-attenuated inversion recovery (FLAIR).

Traumatic brain injury
Traumatic brain injury (TBI) is a significant public health concern, with an estimated annual incidence of 200 cases per 100 000 inhabitants in the USA (Zhang Reference Zhang and Sachdev2003). Severe TBI often has a devastating impact on patients, but even mild TBI can result in serious and disabling neuropsychiatric disorders. These conditions range from cognitive deficits and personality changes to severe and chronic psychosis (Zhang Reference Zhang and Sachdev2003). The estimated prevalence of post-TBI psychosis ranges from 1 to 10%. Its onset follows a bimodal distribution, occurring either in the acute phase or several years after the injury. The prevalence of psychosis is two to three times higher in individuals with a history of TBI than in the general population, suggesting that TBI is an independent risk factor for psychotic symptoms (Fujii Reference Fujii and Ahmed2014; Li Reference Li, Carson and Dams-O’Connor2023).
Schizophrenia-like psychosis of late onset secondary to TBI is typically characterised by hallucinations and delusions. Hallucinations occur in 97% of cases, with auditory hallucinations being the most frequent (88%), followed by visual hallucinations (22%). Delusions are present in 85% of cases, with persecutory delusions being the most common type (65%). Bizarre delusions, ideas of reference and grandiose delusions are less frequent, occurring in 20%, 18% and 16% of cases respectively. Negative symptoms are observed in 43% of people with TBI-related psychosis, with the most common features including affective blunting (70%), social withdrawal (49%) and poor personal hygiene (40%) (Fujii Reference Fujii and Ahmed2014; Li Reference Li, Carson and Dams-O’Connor2023).
The management of psychosis following TBI is primarily guided by clinical experience derived from treating psychosis in individuals without a history of TBI. To date, no interventional studies have specifically investigated post-TBI psychosis. Atypical antipsychotics are most commonly used, with olanzapine being preferred, particularly in acute cases, followed by quetiapine and risperidone (Fujii Reference Fujii and Ahmed2014; Li Reference Li, Carson and Dams-O’Connor2023).
Case vignette: traumatic brain injury
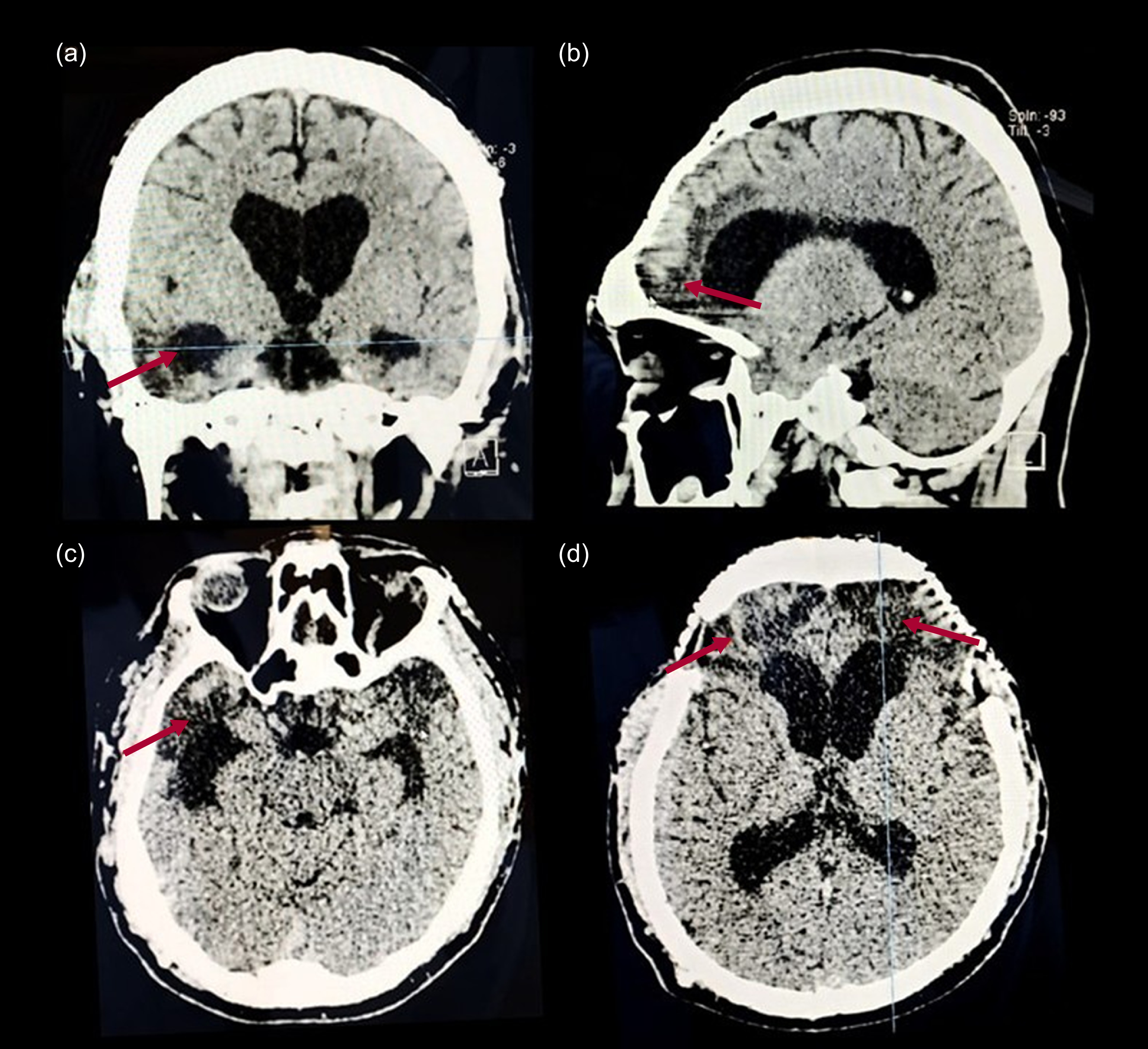
A 41-year-old male presented with a history of traumatic brain injury (TBI), leading to epilepsy and hydrocephalus requiring a ventriculoperitoneal shunt. Several weeks after the acute phase of the TBI, he developed aggressive behaviour and psychotic symptoms, including auditory and complex visual hallucinations, persecutory delusions and mystical/religious delusions of divine salvation. Computed tomography of the brain (Fig. 6) demonstrated a hypodense lesion consistent with encephalomalacia involving the temporal poles (a, c) as well as the medial (b) and the orbitofrontal cortex bilaterally (d). There is evidence of bifrontal craniectomy. The ventricular system shows compensatory enlargement, with dilation of the lateral ventricles, particularly in the frontal and temporal horns.
Non-contrast brain computed tomography scan of a 41-year-old male with a history of traumatic brain injury: (a) coronal, (b) sagital, (c), (d) axial CT scan. The arrows indicate changes characterised by hypodensities in the frontal and temporal lobe.

Brain neoplasms
Central nervous system tumours are a significant cause of morbidity and mortality. These neoplasms have a significant impact on patients’ quality of life and impose an economic burden both individually and socially. In the brain, gliomas are the most common primary tumours, accounting for 40–55% of all lesions (Win Reference Win, Krishnappa and Deol2021; Ramirez-Bermudez Reference Ramirez-Bermudez2022).
Neuropsychiatric manifestations in people with brain tumours are diverse. Psychiatric symptoms and behavioural changes – such as apathy, anxiety, depression, psychosis and cognitive impairments – have been reported in 50–78% of cases, with a higher prevalence in neoplasms affecting the frontal and limbic regions (Win Reference Win, Krishnappa and Deol2021; Romero-Luna Reference Romero-Luna, Mejía-Pérez and Ramírez-Cruz2022; Villanueva-Castro Reference Villanueva-Castro, Hernández-Reséndiz and Munuzuri-Camacho2025). The most common symptoms involve mood disturbances; psychotic symptoms have been reported in up to 22% of patients. Temporal and limbic tumours have been more frequently associated with psychosis (Win Reference Win, Krishnappa and Deol2021; Romero-Luna Reference Romero-Luna, Mejía-Pérez and Ramírez-Cruz2022). One study found that 20% of temporal lobe neoplasms presented with psychotic symptoms, and other research has reported a higher incidence of psychosis in slow-growing, low-grade tumours, even in the absence of evident neurological symptoms (Win Reference Win, Krishnappa and Deol2021). Given the impact of neuropsychiatric manifestations in these patients, their evaluation should be an integral part of the clinical approach.
Genetic diseases affecting the development of the nervous system
Several neurogenetic diseases cause cognitive and behavioural disturbances, including psychosis (Feinstein Reference Feinstein and Chahal2009). Psychosis may be present in diseases leading to intellectual disability, such as Down syndrome, fragile-X syndrome, Prader–Willi syndrome and 22q11.2 deletion syndrome (Feinstein Reference Feinstein and Chahal2009).
22q11.2 deletion syndrome, also known as velocardiofacial syndrome or DiGeorge syndrome, is a microdeletion syndrome. A minority of the cases are inherited, with an autosomal dominant pattern, but most cases are sporadic and related to de novo mutations in one parent’s germ cells. 22q11.2 deletion syndrome is strongly associated with schizophrenia (Feinstein Reference Feinstein and Chahal2009).
Another condition of interest is metachromatic leukodystrophy. This is a rare demyelinating genetic disorder, caused by an autosomal recessive mutation on chromosome 22q, leading to a near-complete absence of the lysosomal enzyme arylsulfatase A. Prominent psychotic features are common, sometimes resembling the schizophrenia phenotype (Hyde Reference Hyde, Ziegler and Weinberger1992).
Tuberous sclerosis is also important (Gupta Reference Gupta, Spoorthy and Raikar2024). This is an autosomal dominant genetic disorder that affects multiple organs, including the kidneys, skin and central nervous system. Its incidence is estimated at approximately 1 in 6000 to 10 000 births. In addition to neurological manifestations such as epileptic seizures, tuberous sclerosis has a high prevalence of neuropsychiatric symptoms, collectively referred to as TAND (tuberous sclerosis-associated neuropsychiatric disorders). The most common manifestations are mood disturbances, cognitive impairments and behavioural disorders. From early childhood, affected individuals often present with comorbidities such as autism spectrum disorder, attention-deficit hyperactivity disorder and intellectual disability. Psychotic symptoms can emerge in childhood or adulthood, with paranoid delusions and hallucinations being the most frequently reported. Given its broad spectrum of manifestations, tuberous sclerosis requires a multidisciplinary approach for comprehensive diagnosis and management (Gupta Reference Gupta, Spoorthy and Raikar2024).
Case vignette: tuberous sclerosis
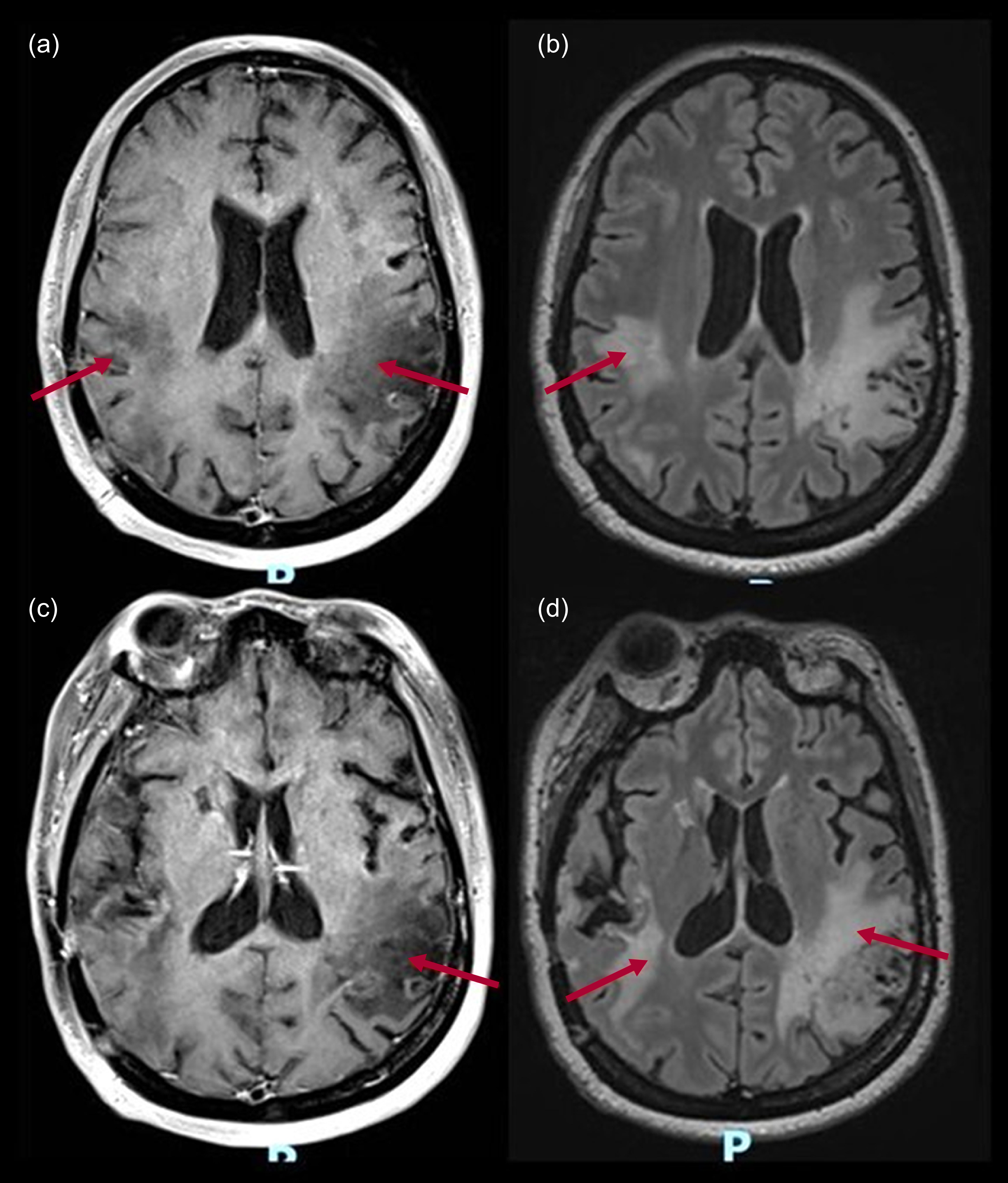
Figure 7 shows MRI of the brain of a 40-year-old female with a diagnosis of tuberous sclerosis and secondary epilepsy since childhood. At the age of 37, she had developed psychotic symptoms, including persecutory delusions and auditory hallucinations of voices mocking her, along with verbal and physical aggression towards neighbours and family members. During the same episode, she exhibited misidentification of her mother, claiming that she was not her mother but a ‘nanny’ who had come to take care of her. The MRI findings showed multiple hypointense lesions in the axial T1-weighted sequence, and hyperintense lesions (tuberculomas) in the frontal and left occipital regions, as seen in the axial (b), sagital (d) and coronal (e) T2-weighted FLAIR.
Magnetic resonance imaging of the brain of a 40-year-old female with a diagnosis of tuberous sclerosis and secondary epilepsy: (a) axial T1-weighted with contrast; (b) axial, (d) sagital, (e) coronal T2-weighted fluid-attenuated inversion recovery (FLAIR); (c) T2-weighted. The arrows indicate hyperintense lesions (tuberculomas) in the frontal and left occipital regions.

Conclusion
Accurate diagnosis of psychosis in neurological conditions requires a multidisciplinary approach, integrating the clinical recognition of the psychiatric pattern, the identification of red flags, as well as neuroimaging, electrophysiological studies and biomarker analysis to distinguish primary psychiatric disorders from secondary neurological causes.
Treatment strategies vary based on aetiology, incorporating antipsychotic medications, immunotherapy and neuromodulation techniques. A careful balance is required to mitigate potential adverse effects, particularly in neurological diseases where dopamine blockade can exacerbate motor symptoms. We emphasise the need for early recognition and targeted interventions to improve patient outcomes.
Future research should focus on refining diagnostic criteria, developing neurobiological biomarkers and exploring novel therapeutics tailored to specific neurological substrates. Enhanced collaboration between neurologists and psychiatrists is essential to bridge the gap in understanding and to effectively manage psychosis in patients with neurological conditions.
MCQs
Select the single best option for each question stem
-
1 What is the prevalence of psychotic symptoms such as hallucinations and delusions in patients with Huntington’s disease?
-
a 3% for hallucinations, 10% for delusions
-
b 10% for hallucinations, 3% for delusions
-
c 20% for hallucinations, 5% for delusions
-
d 50% for hallucinations, 25% for delusions
-
e 1% for hallucinations, 3% for delusions.
-
-
2 How do psychotic symptoms typically progress in patients with Parkinson’s disease?
-
a Visual hallucinations → delusions → agitation
-
b Visual illusions → visual hallucinations → delusions
-
c Auditory hallucinations → delusions → aggression
-
d Visual hallucinations → auditory hallucinations → delusions
-
e Visual hallucinations → agitation → aggression.
-
-
3 In vascular disease, which is an important factor that increases the risk of psychotic symptoms?
-
a Hypertension
-
b Lacunar infarcts
-
c Diabetes
-
d Hyperlipidaemia
-
e Insomnia.
-
-
4 Which type of neuroimaging study is particularly useful for identifying patients with psychosis who require cerebrospinal fluid analysis to rule out an autoimmune pathology?
-
a Computed tomography (CT)
-
b Structural magnetic resonance imaging (MRI)
-
c Single-photon emission computed tomography (SPECT)
-
d Positron emission tomography (PET)
-
e X-ray.
-
-
5 In patients with suspected autoimmune psychosis, which of the following is considered a red flag suggesting an autoimmune aetiology rather than a primary psychotic disorder?
-
a Predominantly auditory hallucinations with systematised delusions
-
b Severe cognitive disturbances, catatonia and atypical hallucinations (visual or tactile)
-
c Gradual onset of persecutory delusions without neurological symptoms
-
d Positive family history of schizophrenia or affective disorders
-
e Onset in late adulthood with progressive cognitive decline.
-
MCQ answers
-
1 a
-
2 b
-
3 b
-
4 b
-
5 b
Data availability
Data availability is not applicable to this article as no new data were created or analysed in this study.
Author contributions
V.M.-A. contributed to study design, literature search, drafting and critical review; A.J.-J. assisted coordination, literature search and manuscript revision; J.C.L.-H. helped with the study design, intellectual input, manuscript writing and revision; R.R.-G. assisted in study design, literature synthesis and manuscript revision; F.M.-C. participated in study conception, methodology and critical review; A.R.-C. worked on data acquisition, literature analysis and manuscript refinement; M.R.-V. contributed to literature review, writing and intellectual input; D.C.-M. focused on methodology, drafting, data organisation and manuscript revision; M.E.-N. contributed to data interpretation, drafting and intellectual input; C.A.-C. assisted in study conceptualisation, literature review and manuscript review; C.E.S.-G. engaged in literature synthesis, discussion and manuscript editing. J.R.-B. led study conceptualisation, literature analysis and manuscript drafting, ensuring methodological rigour and intellectual coherence. All authors approved the final version and ensured the integrity of the work.
Funding
This research received no specific grant from any funding agency, commercial or not-for-profit sector.
Declaration of interest
None.



eLetters
No eLetters have been published for this article.