Introduction
Life expectancy is increasing, with the global population aged 60 years and older projected to rise from 1 billion in 2020 to 2.1 billion by 2050(1), presenting challenges for countries worldwide. Increasing life expectancy does not necessarily equate to a healthier life span, as ageing is a complex biological process associated with many physiological changes and functional decline, increasing susceptibility to infections and chronic disease(Reference Tenchov, Sasso and Wang2). As ageing weakens biological systems, this can lead to dysregulation of the gut-brain axis which is widely recognised as important for maintaining host homeostasis. Furthermore, interest has emerged in understanding how this axis is mediated by the gut microbiome and the closely interconnected immune system(Reference Warren, Nyavor and Zarabian3). Age-related changes in the microbiome composition and its metabolic functions, and in the immune system, results in chronic inflammation which may contribute to the pathogenesis of numerous diseases, including cognitive dysfunction and dementia(Reference Borrego-Ruiz and Borrego4). To facilitate healthy ageing and reduce burdens on healthcare systems, economies and society worldwide, strategies which can help to maintain healthier ageing are urgently required.
Improving nutritional status across the lifespan offers a potential strategy to support better health and well-being into very old age. In particular, certain dietary patterns such as the Mediterranean diet, probiotics and prebiotics and specific nutrients have been linked with better brain health. Dietary interventions also have the potential to modulate the gut microbial and metabolite signatures, with benefits for cognitive health(Reference Berding, Vlckova and Marx5). Folate and related B vitamins, essential for one-carbon metabolism, are associated with preserving cognitive health and reducing the risk of dementia in older adults(Reference Gordon, Hoey and McNulty6). Although the mechanisms remain elusive, the potential role of B vitamins in restoring the gut-immune-brain axis through beneficially altering the microbiota composition and influencing immune function could contribute towards their role in protecting against dementia(Reference Park, Kang and Kim7,Reference Zhang, Thakur and Zhang8) . This review aims to explore the complex interrelationships between the gut microbiota, immune function and inflammation in relation to the ageing brain. In particular, the evidence linking B vitamins with cognitive function and their potential role in modulating the gut microbiota and inflammation will be evaluated, with consideration for proposed biological mechanisms.
The ageing brain
The human brain is remarkably sensitive to the effects of ageing showing declines in cognitive, motor, emotional and tactile functions(Reference Tenchov, Sasso and Wang2). Cognitive function gradually declines in normal ageing, with memory being the most affected domain as individuals age. Mild cognitive impairment (MCI) is a recognised clinical condition where noticeable changes in thinking and memory are observed, but the criteria for the diagnosis of dementia is not met(Reference Tarailis, Lory and Unschuld9). Dementia is a multifactorial syndrome associated with declining cognitive functions, where judgement, memory and thinking are impacted to such an extent that the ability to perform daily life activities is impacted. Alzheimer’s disease (AD) is the most common form of dementia, accounting for 60–70% of cases, followed by vascular dementia (10–20%), dementia with Lewy bodies (10%) and frontotemporal dementia (4%)(10). Currently, more than 55 million people, of whom 60% live in low-middle-income countries, suffer from this condition and this number is expected to increase to 139 million by 2050(10,11) . The higher proportion of dementia cases in low-middle-income countries is likely attributable to factors including more rapid growth in population ageing, a higher burden of modifiable risk factors such as lower educational attainment and diabetes and limited availability and access to preventative healthcare and early diagnosis in these settings(Reference Allegri12). In addition, differences in dietary patterns, sanitation, early-life microbial exposures and antibiotic use between low-and high-income countries may shape the gut microbiota composition, with emerging evidence linking microbiome-related immune and inflammatory dysregulation to AD risk(Reference Yatsunenko, Rey and Manary13,Reference Cryan, O’Riordan and Cowan14) . Given that dementia is a global problem which is projected to grow over time, with associated health and social care costs set to reach £90 billion by 2040 in the UK alone(15), preventing or delaying the onset of dementia should be an urgent public health priority.
A number of mechanisms or hallmarks of ageing have been identified as causes of cellular and molecular damage. These include epigenetic modifications, genomic instability, dysregulated protein turnover, cellular senescence, impaired autophagy, mitochondrial dysfunction, chronic inflammation and gut dysbiosis(Reference Li, Li and Zhang16). Specifically, within the brain, these hallmarks can result in structural and functional changes including loss of white and grey matter, loss of neurons and disrupted synaptic plasticity. They can also lead to alterations in production of neurotransmitters such as dopamine and in protein synthesis and turnover that foster aggregation of toxic β-amyloid and tau proteins(Reference Azam, Haque and Balakrishnan17–Reference Zia, Pourbagher-Shahri and Farkhondeh19). Additionally, mitochondrial dysfunction, that promotes the generation of reactive oxygen species and oxidative stress, along with chronic inflammation and reduced blood-brain barrier function, can lead to neuroinflammation and the progression of dementia(Reference Blinkouskaya, Caçoilo and Gollamudi18,Reference Abadin, de Dios and Zubillaga20,Reference Conway and Duggal21) .
Inflammation, gut dysbiosis and the ageing brain
Inflammageing
Ageing adversely affects functioning of both the innate and adaptive immune systems, referred to as immunosenescence(Reference Calder, Bosco and Bourdet-Sicard22). As part of this altered immune response, a chronic low-grade inflammatory state arises, commonly known as inflammageing. This is characterised by low serum concentrations of interleukin (IL)-10 (an anti-inflammatory cytokine) and increased concentrations of pro-inflammatory cytokines such as tumour necrosis factor-alpha (TNF-α), C-reactive protein (CRP), IL-1ß and IL-6(Reference Santoro, Martucci and Conte23). Elevated pro-inflammatory cytokines in the systemic circulation are known to be associated with age-related diseases(Reference Li, Li and Zhang16), including dementia and other neurodegenerative diseases(Reference Cai, Zhao and Pang24). In a prospective 3-year study involving people diagnosed with MCI (n = 105, aged ≥ 65 years), individuals who progressed to mixed dementia exhibited higher IL-4 compared to those who converted to AD, while participants who remained stable had significantly elevated concentrations of IL-10(Reference Romero-Sevilla, López-Espuela and Fuentes25). The Conselice Study of Brain Ageing showed that the combination of high CRP and IL-6 was associated with greater risk of vascular dementia, whereas no inflammatory markers were associated with AD after 4 years of follow-up in a dementia-free Italian cohort of older adults (n = 804, aged ≥ 65 years)(Reference Ravaglia, Forti and Maioli26). One recent systematic review and meta-analysis, including 79 cross-sectional and longitudinal studies, found that people with AD or MCI had increased concentrations of IL-1β, IL-6, TNF-α and CRP compared to controls. In longitudinal studies, higher IL-6 was associated with increased risk of cognitive decline(Reference Leonardo and Fregni27). The underlying causes of inflammageing are unknown but are considered to be multifactorial and involve senescent cells and their inflammatory secretions, disturbances in the gut microbiota (referred to as gut dysbiosis) and a dysfunctional intestinal barrier(Reference Ling, Liu and Cheng28).
Intestinal permeability
The intestinal barrier is composed of the mucus layer, the luminal microbiota and a physical layer formed by immune and epithelial cells(Reference Untersmayr, Brandt and Koidl29). Tight junction proteins such as occludin and zonulin are interlinked with epithelial cells to regulate the transportation of ions and nutrients and protect against the entry of toxins and pathogens into the epithelium(Reference Chelakkot, Ghim and Ryu30). Intestinal permeability increases with age as tight junction proteins decrease, leading to a decline in barrier function and a potential driving factor of inflammageing. One study in aged mice showed thinning of the protective mucus layer lining in the gastrointestinal tract, decreased T-cell-specific transcripts and signalling pathways and impaired immune responses(Reference Sovran, Hugenholtz and Elderman31). Additionally, increased endothelial gaps and age-related microbial dysbiosis can enhance intestinal permeability (‘leaky gut’) and release of outer membrane vesicles, promoting the translocation of antigens and microbial products into the bloodstream(Reference Wilson, Wells and Davis32). This in turn can trigger inflammatory pathways and contribute to systemic inflammation(Reference Jiang, Yabluchanskiy and Deng33). Evidence from human studies is limited; however, increased gut permeability, systemic inflammation and alterations in both beta diversity (which shows diversity between samples) and taxonomic composition of the gut microbiota were reported in a cohort of people living with dementia(Reference Stadlbauer, Engertsberger and Komarova34).
The gut microbiome
The term ‘microbiome’ refers to a (normally) heterogeneous community of microorganisms, their genetic material and by-products and the environmental conditions they encounter in their habitat. Unique microbiomes can be found in various anatomical compartments of the human body, including the gastrointestinal tract, epidermis, oral and nasal cavities. They regulate various physiological processes and are considered essential for the host’s health. The gut microbiota refers to a diverse group of microorganisms, including bacteria, viruses, fungi, protists, archaea and other eukaryotes that make up the microbial element of the gut microbiome(Reference Maheshwari, Musyuni and Moulick35). Its composition is affected by many factors including age, diet, hormones, lifestyle and medications such as antibiotics(Reference Conway, Rees, Duggal and Marotta36). To date, there is no consensus in the literature on how to define a ‘healthy’ gut microbiota, but generally it is characterised by a diverse and balanced community of microorganisms that perform vital functions for the host(Reference Van Hul, Cani and Petitfils37).
The overwhelming research focus to date has been on the bacterial component of the microbiota. Microbial data are commonly reported as relative abundance, representing the proportion of each taxon within the total community, whereas absolute abundance quantifies the number of microbial cells and therefore reflects actual microbial load(Reference Aroniadis and Grinspan38,Reference Props, Kerckhof and Rubbens39) . While bacteria certainly are the most numerous microorganisms in the gut and they play essential roles in the health of their hosts, information on other types of microorganisms has been more difficult to collect, primarily for technical reasons. Such challenges include low abundance of non-bacterial microbes, difficulty in cultivating many species, biases in PCR amplification and sequencing methods and limited reference databases for accurate taxonomic classification(Reference Lloyd-Price, Arze and Ananthakrishnan40,Reference Quince, Walker and Simpson41) . Even bacterial profiling has limitations, as most studies rely on stool samples that may not fully capture microbial diversity in other gut regions. Furthermore, variation in sample collection (for example timing and oxygen exposure), processing (DNA extraction methods) and storage conditions can substantially influence microbial profiles, while sequencing-based approaches introduce compositional bias and may under-represent low-abundance taxa(Reference Gorzelak, Gill and Tasnim42,Reference Kim, Hofstaedter and Zhao43) . With most studies of the microbiome being almost exclusively bacterial focussed, it is likely that our understanding of the microbiome will change as technology provides broader microorganism detection capability in the future(Reference Filardo, Di Pietro and Sessa44).
In human adults, the bacterial component of the gut microbiota is generally reported to consist of 8 phyla, 18 families, 23 classes, 38 orders, 59 genera and 109 species. The majority of the bacterial species belong to Firmicutes (Faecalibacterium, Ruminococcus, Lactobacilli, Clostridium), Bacteroidetes (Prevotella, Bacteroides), Actinobacteria (Bifidobacterium), Proteobacteria (Helicobacter, Escherichia, Shigella) and Verrucomicrobia (Akkermansia) phyla(Reference King, Desai and Sylvetsky45,Reference Qin, Li and Raes46) . The gut microbiota can exert both pro-inflammatory and anti-inflammatory effects, therefore, changes in its composition may influence systemic immune responses. For instance, a higher abundance of Bifidobacterium, Faecalibacterium, Ruminococcus and Prevotella has been associated with lower concentrations of low-grade inflammatory markers(Reference van den Munckhof, Kurilshikov and ter Horst47). Conversely, a higher abundance of Escherichia coli and Shigella has been associated with increased gut inflammation(Reference Donaldson, Fyfe and Martin48). These effects are partly mediated by bacterial metabolites, particularly short-chain fatty acids (SCFAs), whose type and concentration can either promote or protect against inflammation and metabolic disorders(Reference Hartstra, Bouter and Bäckhed49). Among these, butyrate is a key anti-inflammatory metabolite known to inhibit signalling pathways that drive the production of pro-inflammatory cytokines(Reference Lührs, Gerke and Schauber50). Butyrate has also been shown to reduce lipopolysaccharide (LPS) translocation across the intestinal barrier, thereby limiting LPS-induced immune activation(Reference Bakshi and Mishra51). Faecalibacterium prausnitzii, a major butyrate-producing bacterium, has been inversely associated with several pro-inflammatory markers, including high-sensitivity CRP and IL-6(Reference van den Munckhof, Kurilshikov and ter Horst47). Overall, SCFAs contribute to gut health by supporting intestinal barrier integrity and modulating gut immunity, ultimately offering protection against inflammation(Reference Silva, Bernardi and Frozza52).
Ageing is often accompanied by microbial alterations including a reduction in key commensal groups important for regulating gut health, including Bifidobacterium, Bacteroides, Lactobacilli and Verrucomicrobia, and an increased abundance of potentially pro-inflammatory taxa such as Proteobacteria (Escherichia) and opportunistic microbes including Parabacteroides, Fusobacterium and Clostridioides difficile (Reference Ragonnaud and Biragyn53–Reference Tavella, Rampelli and Guidarelli55). One cross-sectional study conducted in Japanese individuals (n = 367; aged 0–104 years) explored the progressive changes in gut bacteria composition across the lifespan. The researchers categorised participants into three age groups (infant, adult and elderly) and identified distinct microbial patterns and transitional phases associated with each cluster. The shift from infancy to old age was marked by the dominance of a unique co-abundance group comprising Bacteroides, Eubacterium and members of the Clostridiaceae family. Notably, the elderly group exhibited a significantly higher relative abundance of Bacteroidetes, Betaproteobacteria and Deltaproteobacteria(Reference Odamaki, Kato and Sugahara56). A further study in a cohort of Italian adults, that included 24 extremely elderly adults (aged >105 years), showed that the relative abundance of Coprococcus, Faecalibacterium and Roseburia declined with increasing age. In contrast, Oscillospira and two less dominant members from the Bacteroidales order, Odoribacter and Butyricimonas, showed a positive association with age(Reference Biagi, Franceschi and Rampelli57). A systematic review of 27 human studies found that Akkermansia was most consistently reported to be relatively more abundant with ageing, whereas Bacteroidaceae, Faecalibacterium and Lachnospiraceae were relatively low in older age(Reference Badal, Vaccariello and Murray58). Discrepant findings between studies in the microbial changes that occur with age at the various taxonomic levels may be attributable to numerous factors. These include differences in geographical location, dietary habits, cultural and other lifestyle factors(Reference Yatsunenko, Rey and Manary13). Additionally, heterogeneity in study populations regarding age and health status, as well as variations in methodologies used to characterise microbes, may contribute to the inconsistencies observed between studies(Reference Fontana, Panebianco and Picchianti-Diamanti59,Reference Pollock, Glendinning and Wisedchanwet60) . In line with this, findings are also inconsistent regarding alpha diversity, which shows the diversity within a single sample. Thus, some studies observed an increase in alpha diversity with ageing and in long-living groups(Reference Odamaki, Kato and Sugahara56,Reference Kong, Hua and Zeng61,Reference Tuikhar, Keisam and Labala62) , whereas other studies found no differences when compared with younger adults(Reference Kim, Choi and Shin63,Reference Wu, Zeng and Zinellu64) . Noteworthy, despite these inconsistencies, the reported age-related shifts in bacterial composition and in the production of key metabolites, including SCFAs and secondary bile acids, may impact intestinal permeability and compromise barrier integrity(Reference Escalante, Artaiz and Diwakarla65). This in turn can drive systemic inflammation and heighten the risk of morbidity and mortality in older adults(Reference Ragonnaud and Biragyn53).
The gut-brain axis
It is widely acknowledged that there is bidirectional communication between the gut and the brain, referred to as the gut–brain axis. Communication between these two organs occurs via several systems including the autonomic nervous system, the enteric nervous system, the central nervous system and the immune system. The vagus nerve is the most direct pathway through which signals are sent but other pathways include neural, immune, endocrine and metabolic, including the production of SCFAs and neurotransmitters such as serotonin and gamma-aminobutyric acid(Reference Stopińska, Radziwoń-Zaleska and Domitrz66,Reference Gupta, Dinesh and Sharma67) , all of which play critical roles in maintaining host homeostasis(Reference Loh, Mak and Tan68,Reference Mhanna, Martini and Hmaydoosh69) . Dysregulation of the gut-brain axis, caused by various factors including age-related changes in the gut microbiota and concomitant inflammation, has been linked with dementia.
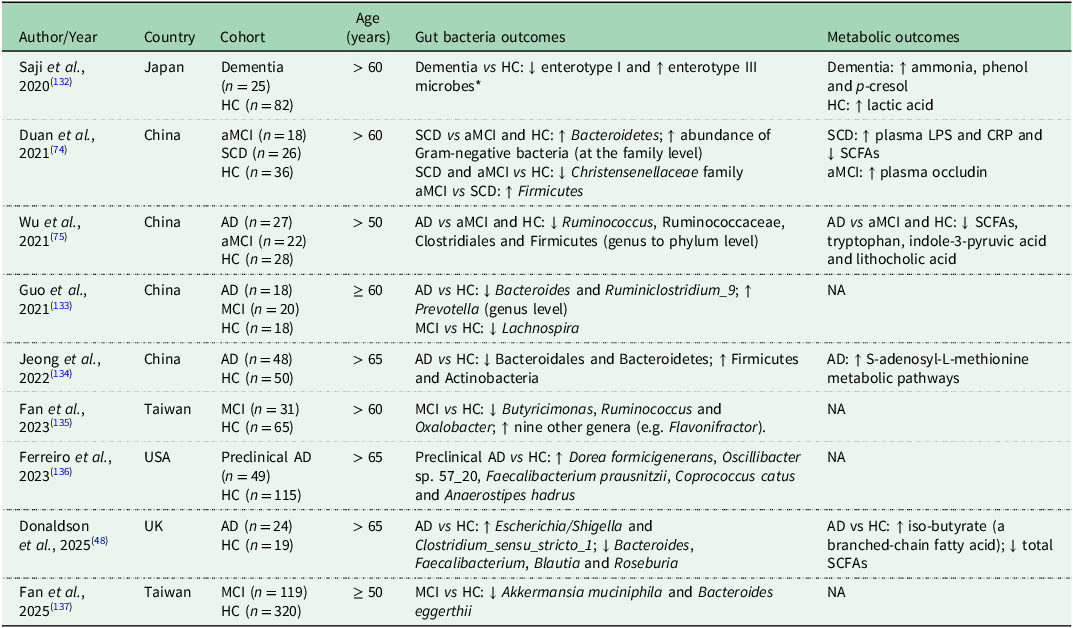
Animal and human studies report reduced bacterial diversity and distinct microbial patterns in those with dementia compared to healthy controls(Reference Bello-Medina, Hernández-Quiroz and Pérez-Morales70–Reference Duan, Liu and Fu74). A systematic review and meta-analysis of 11 observational studies, including 378 healthy controls and 427 individuals across the AD spectrum (from MCI to AD), reported a significant reduction in gut bacterial diversity among people living with AD, but not among those with MCI. Additionally, people across the AD spectrum exhibited decreased abundance of Rikenellaceae, Clostridiaceae, Lachnospiraceae and Firmicutes, alongside increased abundance of Phascolarctobacterium, Proteobacteria and Bifidobacterium, compared to healthy controls. Moreover, the relative abundance of Proteobacteria and Phascolarctobacterium progressively increased from healthy individuals to those with AD, whereas Clostridiaceae abundance declined progressively across the same trajectory(Reference Hung, Chang and Huang73). Table 1 summarises studies investigating gut bacteria and their metabolites in older adults with cognitive disorders compared to healthy controls. Common findings include reduced levels of SCFA-producing bacteria (e.g. Faecalibacterium, Roseburia) and increased pro-inflammatory taxa (e.g. Escherichia/Shigella)(Reference Props, Kerckhof and Rubbens39). Several studies also report lower SCFA concentrations and elevated inflammatory markers in cognitively impaired individuals(Reference Duan, Liu and Fu74,Reference Wu, Han and Zheng75) . However, the findings are not entirely consistent across studies, reflecting differences in study design, geographical location, population characteristics and microbiome profiling methodologies.
Summary of studies investigating gut bacteria and their metabolites in older adults with cognitive disorders compared to healthy controls

Abbreviations: AD, Alzheimer’s disease; MCI, mild cognitive impairment; aMCI, amnestic mild cognitive impairment; SCD subjective cognitive decline; HC, healthy control; LPS, lipopolysaccharide; CRP, C-reactive protein; SCFAs, short-chain fatty acids; NA, not analysed.
↑ indicates increased or higher levels; ↓ indicates decreased or lower levels.
* The human gut microbiome can be broadly classified into three enterotypes: Enterotype 1 (Bacteroides-dominated), Enterotype 2 (Prevotella-dominated) and Enterotype 3 (Ruminococcus-dominated)(Reference Arumugam, Raes and Pelletier138).
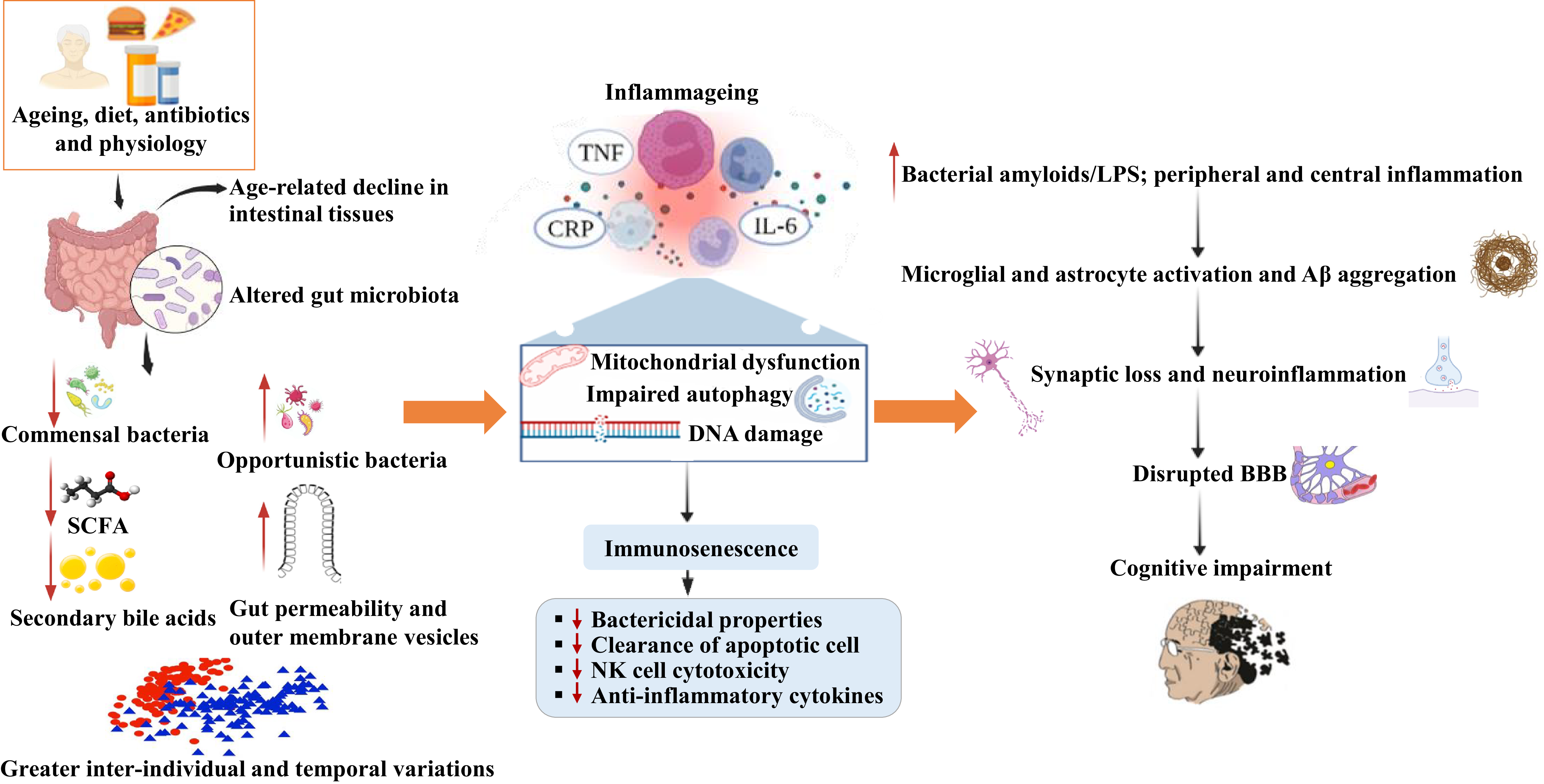
Disturbances in the production of microbial metabolites such as bacterial amyloids and LPS may trigger immune responses that enhance neuronal β-amyloid production and aggregation in the brain, thereby contributing to neuroinflammation and AD pathology(Reference Das, Blasco-Conesa and Korf76). People with amyloid-positive status have been shown to exhibit decreased concentrations of IL-10 and higher concentrations of pro-inflammatory cytokines including IL-6, IL8, IL-1ß, CXC Motif Chemokine Ligand 2 and NLR family pyrin domain containing 3 (NLRP3). These inflammatory changes are associated with taxonomic alterations in the gut bacteria(Reference Cattaneo, Cattane and Galluzzi77). Collectively, these findings support the potential of targeting the gut bacteria to preserve blood-brain-barrier integrity, reduce neuroinflammation and support healthy brain ageing. Figure 1 summarises potential mechanisms linking the gut microbiota with immune function and brain (the gut-immune-brain axis).
Gut-immune-brain axis.
Abbreviations: NK, natural killer; SCFAs, short-chain fatty acids; TNF, tumor necrosis factor; IL, interleukin; CRP, C-reactive protein; Aβ, beta-amyloid; LPS, lipopolysaccharides; BBB, blood-brain barrier.
Several factors including age, diet, physiological changes and antibiotic use can lead to microbial dysbiosis that is associated with increases in opportunistic bacteria and decreases in commensal bacteria, resulting in reduced bioavailability of SCFAs and secondary bile acids, increased gut permeability and greater inter-individual and temporal variations. These changes may fuel inflammageing (increase in pro-inflammatory markers due to ageing), mitochondrial dysfunction, impaired autophagy and DNA damage leading to immunosenescence via the gut-immune axis. Additionally, dysbiosis can increase the production of amyloids and lipopolysaccharides via the gut-brain axis which can activate microglia and astrocytes along with Aβ aggregation. This can result in synaptic loss and neuroinflammation which contributes to cognitive impairment and neurodegenerative diseases.

Emerging roles of B vitamins in healthy ageing
Reduced dietary intake and decline in physiological functions, such as impaired bioavailability and/or altered metabolism of nutrients with ageing, can adversely impact nutritional status contributing to poorer health and well-being(Reference Calder, Carding and Christopher78). Maintaining optimal nutritional status is therefore a key factor for promoting healthy ageing as it may help to delay or prevent the onset of chronic diseases(Reference Bruins, Van Dael and Eggersdorfer79). The protective roles of folate and the metabolically related B vitamins (B12, B6 and riboflavin) and/or the potential adverse effects of elevated homocysteine in a number of age-related conditions including dementia have gained attention over the past couple of decades(Reference Porter, Hoey and Hughes80).
Metabolic role of B vitamins in one-carbon metabolism
One-carbon metabolism, mediated by folate, vitamins B12, B6 and riboflavin, supports multiple physiological processes(Reference Ducker and Rabinowitz81,Reference Bailey, Stover and McNulty82) . It is a network of interrelated biochemical reactions involving the transfer and utilisation of one-carbon units required for DNA synthesis and repair, amino acid metabolism and numerous methylation processes(Reference Bailey, Stover and McNulty82,Reference Dang, Jain and Dhanda83) . Reduced folates enter the folate cycle as tetrahydrofolate (THF), which acquires a carbon unit from serine in a vitamin B6 (pyridoxal 5’-phosphate; PLP) dependent reaction. This subsequently forms 5,10-methyleneTHF which is required for the synthesis of thymidine and purines or converted to 5-methylTHF. The reduction of 5,10-methyleneTHF to 5-methylTHF is catalysed by methylenetetrahydrofolate reductase (MTHFR), with riboflavin (flavin adenine dinucleotide, FAD) as a co-factor. 5-methylTHF subsequently acts as the methyl donor in the remethylation of homocysteine to methionine through the vitamin B12-dependent enzyme methionine synthase. Methionine is a precursor for S-adenosyl-methionine (SAM), the universal methyl donor for the methylation of proteins, DNA, membrane phospholipids and neurotransmitters such as serotonin and catecholamine(Reference Bailey, Stover and McNulty82,Reference Guéant, Guéant-Rodriguez and Kosgei84) . After donation of its methyl group, SAM is converted to S-adenosylhomocysteine and then homocysteine which is either metabolised in the transsulphuration pathway to form cysteine, in a vitamin B6-dependent process or remethylated back to methionine(Reference Bailey, Stover and McNulty82). Given the integral role that these B vitamins play in one-carbon metabolism, sub-optimal status of any one B vitamin could disrupt this network of pathways, leading to reduced methylation status within the relevant tissues and hyperhomocysteinemia, which in turn may trigger both oxidative stress and inflammation. Furthermore, increased uracil misincorporation into DNA may occur from impaired thymidylate synthesis due to low 5,10-methylene-THF concentrations, causing uracil to be inserted into DNA instead of thymidine. These changes can contribute to adverse health outcomes associated with ageing(Reference Araújo, Martel and Borges85,Reference Brachet, Chanson and Demigné86) .
Evidence linking B vitamins and the gut microbiota
In addition to dietary sources of these essential B vitamins, the gut bacteria is considered to be a complementary source of these nutrients by synthesising B vitamins which can be absorbed in the colon. However, as B vitamins are used for growth and metabolic activity by the microbes, cross-feeding between producers and non-producers may limit the bioavailability of B vitamins to the host. Thus, it is unknown whether microbial synthesis can contribute towards dietary requirements and nutrient status in humans(Reference Zheng, Xia and Liu87).
A study evaluating the genomes of 256 common human gut bacteria using the PubSEED platform predicted that most genomes were able to produce riboflavin (65%), followed by vitamin B6 (50%), folate (43%) and vitamin B12 (42%). Biosynthesis pathways for riboflavin, folate and B6 were observed in all or nearly all Bacteroidetes genomes and for vitamin B12, in nearly half the genomes. All or nearly all Fusobacteria had the potential to produce riboflavin, vitamin B12 and folate, whereas nearly all Proteobacteria could produce folate and vitamin B6. All Actinobacteria genomes could potentially synthesis vitamin B6, but biosynthesis pathways were found to be rare for the other B vitamins(Reference Magnúsdóttir, Ravcheev and de Crécy-Lagard88). Furthermore, the authors predicted that microbial synthesis could potentially provide 86%, 37%, 31% and 2.8% of dietary recommendations for vitamin B6, folate, B12 and riboflavin respectively. However, evidence to support this prediction is greatly lacking. In an observational study of adults on a healthy dietary pattern compared to a typical western diet, Bacteroidetes and Proteobacteria were identified as having the potential to produce folate ex vivo but no associations with higher folate or lower homocysteine concentrations were observed in the study participants(Reference Malinowska, Schmidt and Kok89). This finding suggests that food sources (both natural and fortified foods) and dietary supplements remain essential sources of folate to ensure that dietary requirements are met.
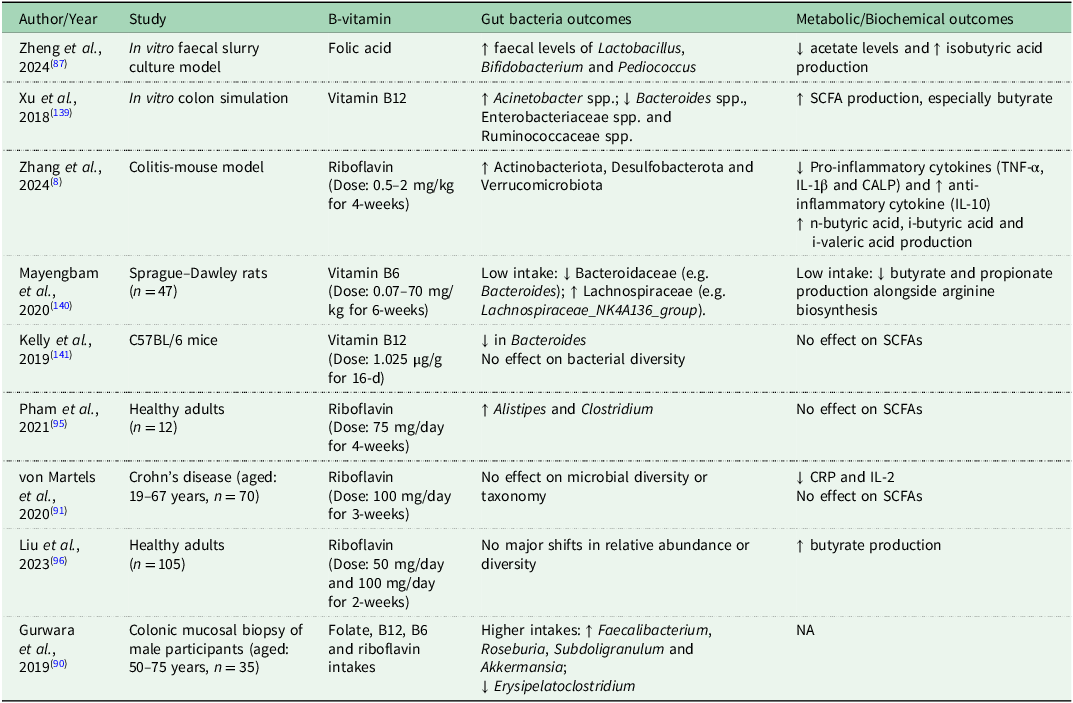
In addition to microbes synthesising B vitamins, interest has emerged in understanding how B vitamins can modulate the composition and metabolic functions of gut bacteria. Evidence from in vitro (Reference Zheng, Xia and Liu87), animal(Reference Zhang, Thakur and Zhang8) and cross-sectional human studies(Reference Gurwara, Ajami and Jang90,Reference von Martels, Bourgonje and Klaassen91) suggests that B vitamins may increase bacterial diversity, enhance beneficial taxa and suppress potentially harmful ones, while also promoting anti-inflammatory and metabolically favourable changes including increased butyrate production (Table 2). Of the B vitamins, vitamin B12 has been the most thoroughly investigated in relation to gut microbes. In a systematic review of 22 studies (11 human observational, 8 animal and 3 in vitro), 19 studies claimed that vitamin B12 intake, supplementation or status was linked to gut microbe outcomes, including α-diversity and β-diversity, bacterial abundance and functional capacity including SCFA synthesis. In vitro work has consistently reported that vitamin B12 could increase diversity and the relative abundance of bacteria, whereas the evidence from animal and human studies showed greater heterogeneity. Furthermore, the impact on bacterial composition and metabolism differed according to the form of B12 used in the studies(Reference Guetterman, Huey and Knight92). To date, the limited studies available in healthy humans have mostly been conducted in infants, children or women of childbearing age, with a lack of studies in older adults. In addition, the existing evidence is overwhelmingly derived from high-income countries, despite the higher burden of AD in low-and middle-income settings. This geographical imbalance limits the generalisability of current findings, particularly given regional differences in diet, infection burden, antibiotic exposure and healthcare access(Reference Jaya and Brito93,Reference Sun, Wang and Tsilimigras94) , all of which may influence both B-vitamin status and the gut microbiota composition. Furthermore, intervention trials tended to focus on riboflavin. One prospective intervention study in people with Crohn’s disease reported that supplementation with 100mg/d riboflavin for 3 weeks had no effect on diversity, taxonomy or metabolic pathways of the faecal bacteria(Reference von Martels, Bourgonje and Klaassen91). A pilot randomised controlled trial (RCT) in healthy adults which involved several vitamins but included a riboflavin arm, found that 75mg/d delivered to the colon increased Alistipes and Clostridium but had no effect on SCFA production(Reference Pham, Fehlbaum and Seifert95). A further larger RCT in healthy adults (n = 105) concluded that although oral riboflavin supplementation for 2 weeks promoted butyrate production, no major shifts in relative abundance or diversity were observed(Reference Liu, Sadaghian Sadabad and Gabarrini96). Given the emerging evidence, further research is warranted across all stages of the lifecycle and in diverse geographical locations to understand whether B vitamins, which are mostly absorbed in the proximal small intestine, can reach the distal colon and modulate composition of the gut bacteria(Reference Cunningham, Azcarate-Peril and Barnard97,Reference Pham, Dold and Rehman98) . Such work should also be cognisant of the challenges and limitations of using faecal samples as a proxy of the entire gut bacterial composition and follow best practice for sample collection, processing and storage to minimise technical bias and improve reproducibility(Reference Kim, Hofstaedter and Zhao43,Reference Tang, Jin and Wang99) .
Studies investigating B vitamins in relation to gut bacteria and associated metabolic and biochemical measures

Abbreviations: CRP, C-reactive protein; IL, interleukin; CALP, calprotectin; TNF-α, tumor necrosis factor-α; spp., multiple species; SCFAs, short-chain fatty acids; NA, not analysed.
↑ indicates increased or higher levels; ↓ indicates decreased or lower levels.
Evidence linking B vitamins and inflammation
A bidirectional relationship exists between nutritional factors and immunity, whereby intake of B vitamins and other dietary components affects immune competence while inflammation in turn alters nutrient metabolism and requirements, thus potentially leading to deficient status(Reference Tourkochristou, Triantos and Mouzaki100,Reference Venter, Eyerich and Sarin101) . Numerous studies, both population-based and in patient groups, show that PLP concentrations are inversely associated with inflammatory markers(Reference Ueland, McCann and Midttun102). Furthermore, B6 supplementation can improve these markers and increase immune responses in critically ill people(Reference Cheng, Chang and Lee103) and in inflammatory disease states(Reference Huang, Wei and Wu104). A possible mechanism underlying these observations in inflammatory states is the mobilisation of vitamin B6 to sites of inflammation, where it may serve as a co-factor in pathways producing metabolites with immunomodulating effects. In this regard, the kynurenine pathway, dependent on PLP and FAD as cofactors, provides a crucial connection between B-vitamin status and immune modulation. This pathway metabolises tryptophan into downstream products that regulate immune cell proliferation and oxidative balance. Inflammatory states often upregulate kynurenine activity, increasing the kynurenine-to-tryptophan ratio, which has been associated with accelerated cognitive decline and depression(Reference Favennec, Hennart and Caiazzo105,Reference Ogyu, Kubo and Noda106) . PLP and FAD are required cofactors in kynurenine metabolism, but human evidence that vitamin B6 status modifies kynurenine flux is limited and largely observational. Longitudinal and interventional studies are therefore needed(Reference Wilson, Umans and Swarovski107,Reference Dugué, Hodge and Ulvik108) to determine whether a more optimal B6 status may help to normalise this pathway, promoting immune homeostasis and mitigating inflammation-related neurodegeneration.
Regarding folate, a systematic review and dose–response meta-analysis of 12 RCTs reported that high-dose folic acid supplementation reduced serum concentrations of CRP but had no effect on IL-6 and TNF-α, although the analysis included a small number of participants(Reference Asbaghi, Ashtary-Larky and Bagheri109). Potential mechanisms underlying the influence of folic acid supplementation may include its homocysteine-lowering effect(Reference Brönstrup, Hages and Prinz-Langenohl110). Elevated homocysteine is suggested to trigger the expression of inflammatory cytokines by increasing poly adenosine diphosphate ribose polymerase activation and prompting nuclear factor kappa B (NF-kB) activation(Reference Wang, Siow and O111). Supplementation with folic acid may inhibit the activation of NF-kB, a master regulator of inflammatory gene expression(Reference Asbaghi, Ashtary-Larky and Bagheri109).
In the case of riboflavin, a recent mouse study found that administration of higher doses improved physiological indicators (changes in body weight, stool consistency and rectal bleeding) and restored intestinal damage by modulating oxidative homeostasis and immune signalling. This was achieved by inhibiting the pro-inflammatory cytokines IL-1β and TNF-α and enhancing the anti-inflammatory cytokine IL-10(Reference Zhang, Thakur and Zhang8). The potential mechanism explaining this finding is that riboflavin inhibits activation of p38, a transcription factor that activates some mitogen-activated kinases, which play a significant role in the inflammatory process(Reference Cobianchi, Fornoni and Pileggi112,Reference Cuadrado and Nebreda113) . Human studies are however needed to confirm the clinical relevance of such findings. Regarding vitamin B12, one cross-sectional study among people with type 2 diabetes (aged 20–84 years) reported an inverse association between serum B12 and IL-6 concentrations(Reference Lee, Wang and Lin114). The anti-inflammatory properties of vitamin B12 may be due to its ability to suppress NF-kB, inhibit nitric oxide synthase and enhance mitochondrial oxidative phosphorylation(Reference Lee, Wang and Lin114,Reference Wheatley115) . Taken together, there is emerging evidence to support the role of one-carbon-related B vitamins in reducing inflammation and modulating immune function. Optimising B-vitamin status, which is commonly found to be compromised in older adults(Reference Porter, Hoey and Hughes80), may therefore be particularly beneficial during ageing or in chronic inflammatory states.
Evidence linking B vitamins with cognitive function
A large body of observational evidence suggests that suboptimal status of folate and related B vitamins and/or elevated concentrations of homocysteine are associated with cognitive dysfunction(Reference Tucker, Qiao and Scott116), greater rates of cognitive decline(Reference Clarke, Birks and Nexo117) and increased risk of dementia(Reference Haan, Miller and Aiello118). Of the B vitamins, the focus has predominately been on folate and vitamin B12, with far fewer studies considering the others, particularly riboflavin. In one longitudinal study from this centre, lower dietary intake of vitamin B6 (0.9–1.4 mg/d) and biomarker status (PLP concentrations ranging from 15.4 to 42.9 nmol/L) were each associated with a higher risk of cognitive decline in older adults over 4 years of follow-up, whereas no associations were observed for folate, B12 or riboflavin with cognitive decline(Reference Hughes, Ward and Tracey119). Notably, intakes of vitamin B6 were below currently set reference intakes for older European adults (1.6–1.7 mg/d depending on sex), and consequently many participants had PLP concentrations <30 nmol/L, the cut-off value commonly used to define vitamin B6 deficiency(Reference Hughes, Ward and Tracey119,120) . In a study drawing on US population-based data (NHANES), riboflavin intake was found to be positively associated with cognitive performance in older adults(Reference Ji, Sun and Li121). Moreover, Hoffmann and colleagues (2021) observed that people with AD had lower serum concentrations of folate (median 6.6 ng/mL vs 8.7 ng/mL in controls), B12 (median 248 ng/L vs 415 ng/L in controls) and B6 (median 6.4 mg/dL vs 7.5 mg/dL in controls), whereas people with Lewy body dementia had reduced vitamin B12 concentrations (median 224 ng/L vs 415 ng/L in controls). Vitamin B6 was also lower in vascular dementia (median 4.5 mg/dL vs 7.5 mg/dL in controls), and elevated concentrations of homocysteine were observed in people living with AD (median 23.0 µmol/L vs 16.5 µmol/L in controls) and MCI (median 25.2 µmol/L vs 16.5 µmol/L in controls)(Reference Hoffmann, Busse and von Hoff122). However, direct comparison between studies is challenging due to differences in selected biomarkers, reported units and cut-off values used to define deficiency. Furthermore, the body of evidence which suggests a link between low B-vitamin status and dementia is largely observational, and therefore residual confounding and reverse causality cannot be excluded.
The VITACOG RCT provides the most robust evidence to date to support the role of B vitamins in maintaining better brain health. Specifically, supplementation for 2 years with folic acid (0.8 mg), B12 (0.5 mg) and B6 (20 mg) to adults aged ≥70 years with MCI resulted in reducing the rate of cognitive decline(Reference De Jager, Oulhaj and Jacoby123). Additionally, magnetic resonance imaging (MRI) scans showed 30% less brain atrophy in participants on B vitamins for 2-years compared to the placebo group. Moreover, participants with the highest baseline homocysteine concentrations exhibited a 53% reduction in the rate of brain atrophy with intervention(Reference Smith, Smith and De Jager124). However, this and other RCTs have used B vitamin doses greatly in excess of current dietary recommendations (i.e. folate: 330 µg dietary folate equivalents (DFE)/d; vitamin B6: 1.6mg/d; vitamin B12: 4 µg/d)(120,125,126) ; such doses, typically within the pharmacological range, can be problematic in terms of translating and interpreting the findings for public health and nutrition policy. Furthermore, despite mounting evidence suggesting beneficial effects of folate-related B vitamins in cognitive health, not all studies report benefits of these B vitamins, either alone or in combination, with some studies even reporting adverse effects. One large-scale UK Biobank study, involving over 466,000 participants (median age 58 years), found that supplementation with folic acid alone was associated with an increased risk of AD and vascular dementia, as well as adverse brain structural changes(Reference Ling, Yuan and Huang127). Interestingly, this risk was not observed in participants who took folic acid in combination with other B vitamins, suggesting that balanced supplementation may be key to avoiding potential adverse effects. It should be noted, however, that this Biobank study was limited by the absence of data on supplement formulation, dose and duration and by the lack of biomarker measures of B-vitamin status. Further investigation is therefore required, particularly in the form of well-designed RCTs of appropriate duration, conducted in populations with inadequate dietary intakes and/or status (who are most likely to benefit from intervention) and using doses aligned with nutritional rather than pharmacological levels to enhance the generalisability of the findings.
B vitamins and the gut-immune-brain axis
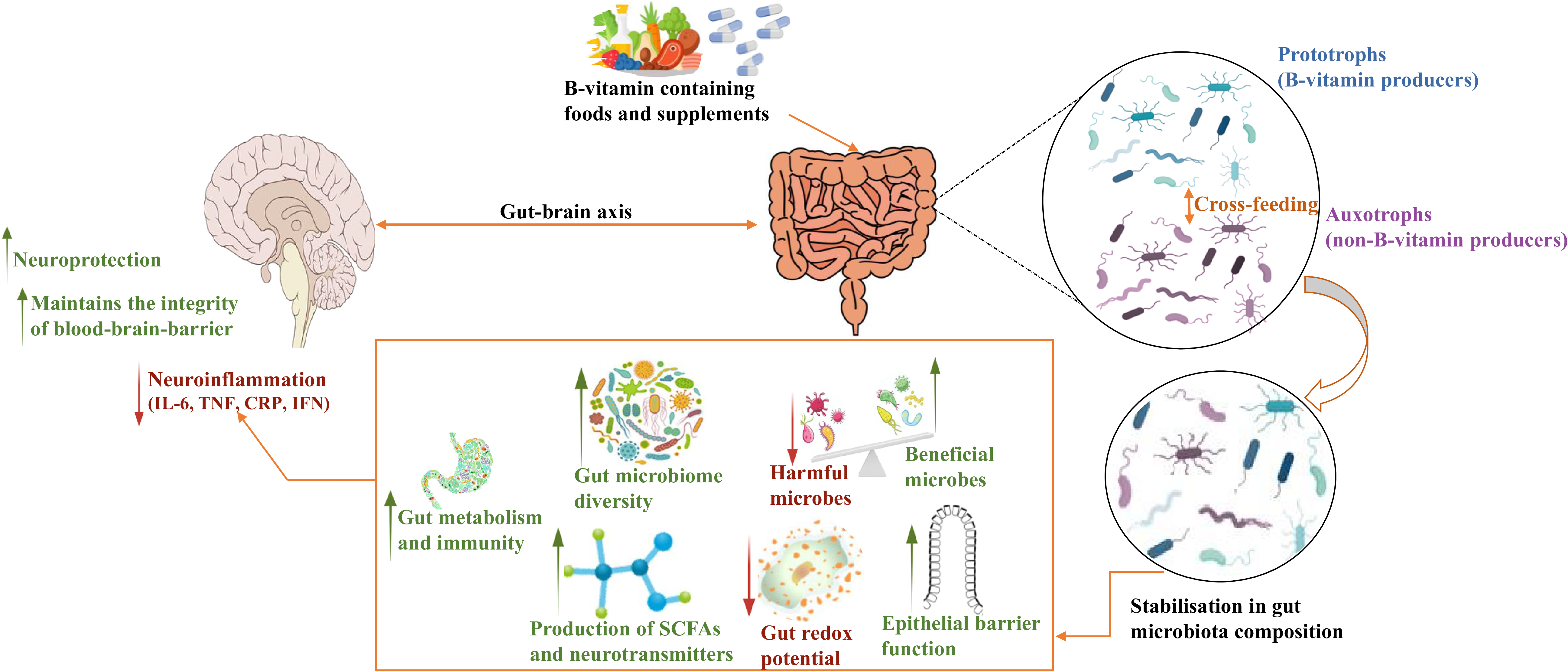
As demonstrated in the above-mentioned studies, B vitamins appear to have a bidirectional relationship with gut bacteria, which may influence the gut-immune-brain interface. Interactions between B vitamins and the gut microbiota and potential mechanisms involved in neuroinflammation are illustrated in Figure 2. Current evidence on how B vitamins influence gut bacteria and inflammation is limited and remains to be fully established. One study in a colitis mouse model showed that the administration of riboflavin enhanced intestinal barrier integrity, reduced gut inflammation and increased relative abundance of bacterial phyla Actinobacteriota, Desulfobacterota and Verrucomicrobiota(Reference Zhang, Thakur and Zhang8). In a human study, riboflavin supplementation significantly decreased serum concentrations of inflammatory markers, although no changes in bacterial composition were observed, thus providing no evidence to suggest that the inflammatory benefits were mediated by gut bacteria(Reference von Martels, Bourgonje and Klaassen91). To the best of our knowledge, only one study has examined associations between B-vitamin status, gut bacteria and cognitive health(Reference Park, Kang and Kim7). It reported that deficiency of folate and B12 in amyloid-β-infused rats significantly altered the bacterial composition and impaired memory. Given the emerging evidence, it is possible that manipulating the gut bacteria and thereby modulating the immune response through B-vitamin supplementation could offer a promising strategy to delay or prevent the onset of neurodegenerative diseases. However, it is important to note that evidence from human studies, particularly in older adult populations, remains very limited, and further research is required to clarify these mechanistic links and their clinical implications.
Potential mechanisms linking B vitamins with the gut microbiome and brain.
Abbreviations: IL, interleukin; TNF, tumor necrosis factor; CRP, C-reactive protein; IFN, interferon; SCFAs, short-chain fatty acids.
Intake of B vitamins could influence the stability of the gut microbiota via cross-feeding of prototrophs (B-vitamin producing bacteria) and auxotrophs (non-B-vitamin producing bacteria). This stability in the microbial composition may result in increased diversity along with a higher proportion of beneficial bacteria and increased production of SCFAs and neurotransmitters, enhancing epithelial barrier function, gut metabolism and immunity. It could also reduce the proportion of pathogens and gut redox potential. This in turn could reduce neuroinflammation by decreasing the concentrations of IL-6, TNF, CRP and IFN and maintaining the blood-brain-barrier, thus offering neuroprotection via the microbiota-gut-brain axis.

Research limitations and future perspectives
Despite the emergence of numerous studies profiling the gut microbiome, the definition of a ‘healthy’ gut remains to be elucidated, and the specific beneficial functions of the constituent bacteria, viruses and eukaryotes are still to be revealed. Advancements in next-generation sequencing, including metagenomics and metatranscriptomics, will allow for a more complete characterisation of the gut microbiome by allowing both metabolic and immunological functions to be explored. This will lead to better understanding of the mechanisms linking the microbiome with healthy ageing(Reference Hemmati, Monemi and Asli128). Studying interactions between the microbiome and health-related outcomes in human participants also poses methodological challenges, particularly regarding the collection of faecal samples, which may lead to participant non-compliance, embarrassment and contamination risks(Reference Tang, Jin and Wang99). Although some studies have attempted to characterise microbial signatures in the circulation as a proxy for the gut microbiome(Reference Craven, McGuinness and Buchanan129), the existence of a blood microbiome remains disputed. Observed species may reflect bacterial translocation from other body sites or contamination rather than a stable microbial community; thus, such approaches require cautious interpretation(Reference Tan, Ko and Chen130).
To date, most microbiome-focussed studies involve population groups other than older adults and address dietary components other than B vitamins(Reference Chen and Boyle131). While B vitamins have independently shown promise in modulating the composition of gut bacteria and immune responses, current evidence is inconsistent and very limited, predominantly derived from animal studies or small human trials. A limitation of many observational studies with large participant numbers is the lack of comprehensive data on the dose, formulation or duration of B-vitamin supplementation as such information is typically obtained through self-reported questionnaires(Reference Smith, Smith and De Jager124). This limits the reliability and reproducibility of findings. Therefore, longitudinal studies are needed in older populations with differing baseline B-vitamin status to generate comprehensive datasets which can be subjected to novel methodologies such as machine learning algorithms and artificial intelligence. This will help to better understand the complex network of interactions amongst the B vitamins, the gut microbiome and host immune function in relation to brain health and disease. Well-powered RCTs are also needed to evaluate the effects of intervention with folate and related B vitamins, administered alone and in combination, on the human gut microbiome. Furthermore, the inclusion of brain imaging techniques would allow for objective assessment of structural and functional responses to B-vitamin intervention. Additional studies are also required to determine whether bacterial synthesis can contribute towards dietary requirements and nutrient status in humans.
Conclusion
Folate and related B vitamins are linked with cognitive health, a relationship which may be mediated by the gut microbiome and inflammation. Emerging evidence suggests that improving B-vitamin status can modulate the composition and metabolic activity of gut bacteria, which in turn may regulate the gut barrier and reduce inflammation, with benefits for health including cognitive health in ageing. However, these interrelationships remain to be robustly demonstrated, and current evidence is inconsistent with limited data from human studies, particularly in older populations. Therefore, well-designed, large-scale RCTs and integrative mechanistic studies, potentially utilising metagenomics, metabolomics and artificial intelligence-driven analytics, are crucial to better understand the complex interactions involved. As the evidence accumulates, there may be a role for personalised, evidence-based nutritional strategies aimed at promoting better brain health in ageing.
Financial support
This work was conducted as part of a PhD programme funded by the Department for the Economy (DfE) through the Ulster University. The funder had no role in the design, interpretation or writing of this review.
Author contributions
US, HM and LH conceptualised the article. US produced the first draft, with significant additions and revisions from LH, HM, CH, MW and JD. All authors approved the final version of the article for submission.
Competing interests
We have no conflicts of interest to report.
Data Sharing Statement
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.