Introduction
Johnsongrass [Sorghum halepense (L.) Pers.] is a C4 perennial graminoid in the Poaceae family, native to Asia and northern Africa (Holm et al. Reference Holm, Plucknett, Pancho and Herberger1977; Howard Reference Howard2004). Its aggressive growth, ecological flexibility, and dual reproductive capacity have enabled it to establish across Africa, Europe, South America, Australia, the United States, and Asia, making it one of the world’s 10 most problematic weeds (Holm et al. Reference Holm, Plucknett, Pancho and Herberger1977; Holm et al. Reference Holm, Doll, Holm, Pancho and Herberger1997; Valverde and Gressel Reference Valverde and Gressel2006). Globally, S. halepense has been reported in 124 countries and infests more than 30 different crops (Valverde and Gressel Reference Valverde and Gressel2006; Yang et al. Reference Yang, Zhao, Xian, Qi, Li, Guo, Chen and Liu2023). In Australia, S. halepense was originally introduced as a forage in the late 1800s, but has become a major weed in temperate and tropical cropping systems (Parsons and Cuthbertson Reference Parsons and Cuthbertson2001; Parsons and Cuthbertson Reference Parsons and Cuthbertson1992; Monaghan Reference Monaghan1978; Velez-Gavilan Reference Velez-Gavilan2024). Prolific seed production, an extensive rhizome network, and broad environmental adaptability underpin its persistence (Navie Reference Navie2004; Parsons and Cuthbertson Reference Parsons and Cuthbertson2001; Sharp and Simon Reference Sharp and Simon2002), particularly in eastern Australian cropping systems characterized by subtropical climates, summer-dominant rainfall, and mild winters (Grose et al. Reference Grose, Bhend, Argüeso, Ekstrom, Dowdy, Hoffman, Evans and Timbal2015; Hemming et al. Reference Hemming, Roughan, Malan and Schaeffer2023; Nguyen et al. Reference Nguyen, Wheeler, Hendon, Lim and Otkin2021). Double cropping, short fallows, and tillage practices that fragment and redistribute rhizomes have further exacerbated local proliferation (Dang et al. Reference Dang, Balzer and Crawford2018; Haque et al. Reference Haque, Gurusinghe, Brown and Weston2022; Travlos et al. Reference Travlos, Montull and Kukorelli2019). Heavy reliance on glyphosate has created strong selection pressure, leading to the first confirmed case of glyphosate-resistant S. halepense in Queensland (Heap Reference Heap2024).
The success of S. halepense stems from its dual reproductive strategy, which allows for both dispersal and local persistence (Liu et al. Reference Liu, Scursoni and Moreno2019; Warwick and Black Reference Warwick and Black1983). Individual plants produce 28,000 to 80,000 seeds annually, many of which have physical dormancy controlled primarily by seed coat impermeability, which restricts water uptake and delays germination (Mehdi et al. Reference Mehdi, Mojtaba and Karimian2018; Schantz Reference Schantz2025; Şin et al. Reference Şin, Kadıoğlu and Yıldırım2024; Warwick and Black Reference Warwick and Black1983). Soil and management factors, such as burial depth, tillage intensity, and surface residue cover, modulate dormancy release and seedling emergence by altering the physical environment, thereby producing heterogeneous recruitment patterns (Loddo et al. Reference Loddo, Vasileiadis, Masin, Zuin and Zanin2016; Prostko et al. Reference Prostko, Wu and Chandler1998). Rhizomes complement seed-based persistence by serving as both regenerative organs and carbohydrate reserves, allowing for rapid regrowth following mechanical disturbance, herbicide exposure, or climatic stress (Atwater et al. Reference Atwater, Kim, Tekiela and Barney2017; Mitskas et al. Reference Mitskas, Tsolis, Eleftherohorinos and Damalas2003). Together, seedbank and rhizomes form a resilient survival system, often allowing populations to reestablish even after intensive control efforts (Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014). Additional traits such as nutrient-use plasticity, allelopathy, and associations with nitrogen-fixing organisms further strengthen competitiveness across diverse soil and climatic conditions (Ghosh et al. Reference Ghosh, Rajan, Phuyal, Subramanian and Bagavathiannan2024; Rout and Chrzanowski Reference Rout and Chrzanowski2009; Ştef et al. Reference Ştef, Cărăbeţ, Grozea, Radulov, Manea and Berbecea2015; Vasilakoglou et al. Reference Vasilakoglou, Dhima and Eleftherohorinos2005). The ecological and agronomic consequences of S. halepense infestations are substantial. It reduces crop yields (Williams and Hayes, Reference Williams and Hayes1984), serves as an alternative host for pests and diseases, and can be toxic to livestock (Ahn et al. Reference Ahn, Prom and Magill2021; Bridges and Chandler Reference Bridges and Chandler1987; Karkanis et al. Reference Karkanis, Athanasiadou, Giannoulis, Karanasou, Zografos, Souipas, Bartzialis and Danalatos2020). These impacts, together with the emergence of herbicide-resistant populations, underscore the critical need for evidence-based, region-specific management strategies.
Extensive research from other regions has shown that dormancy release and seedling recruitment of S. halepense are strongly influenced by burial depth, soil moisture, temperature regimes, residue cover, and salinity (Benvenuti et al. Reference Benvenuti, Macchia and Miele2001; Loddo et al. Reference Loddo, Vasileiadis, Masin, Zuin and Zanin2016; Mehdi et al. Reference Mehdi, Mojtaba and Karimian2018; Prostko et al. Reference Prostko, Wu and Chandler1998). Burial depth alters light availability, temperature fluctuations, and mechanical resistance. Shallow placement promotes emergence, whereas deep burial restricts or prevents emergence due to mechanical resistance, reduced oxygen, and energy limitations (Benvenuti et al. Reference Benvenuti, Macchia and Miele2001; Prostko et al. Reference Prostko, Wu and Chandler1998). Temperature and moisture interact to regulate dormancy loss and germination timing (Krenchinski et al. Reference Krenchinski, Albrecht, Albrecht, Villetti, Orso, Barroso and Victoria Filho2015; Şin et al. Reference Şin, Kadıoğlu and Yıldırım2024), while salinity and osmotic stress impose further constraints, with population-level differences reflecting variation in stress tolerance (Mehdi et al. Reference Mehdi, Mojtaba and Karimian2018; Sinha et al. Reference Sinha, Gupta and Rana1986).
Despite these ecological drivers being studied extensively in other regions, their relevance to eastern Australia remains uncertain. Australian cropping systems differ in rainfall distribution, temperature regimes, soil types, salinity risks, irrigation practices, cropping practices, and the presence of herbicide-resistant populations. Without region-specific germination data, predictions of emergence windows and timing of integrated weed management tactics may be inaccurate, as emergence timing determines the effectiveness of stale seedbeds, knockdown herbicides, early-season crop competition, and strategic tillage. Understanding how seeds from Australian populations of S. halepense respond to temperature, light, salinity, osmotic stress, and burial depth is essential for developing effective, regionally relevant management strategies.
Although S. halepense is one of Australia’s most persistent perennial weeds, its germination ecology under eastern Australian environmental conditions remains poorly understood, limiting accurate prediction of emergence and the optimization of management timing. This study addresses this gap by (1) identifying the primary dormancy mechanisms in Australian populations; (2) testing the effectiveness of sodium hypochlorite immersion and mechanical scarification for breaking dormancy; and (3) quantifying germination and emergence responses to temperature, light, salinity, osmotic stress, and burial depth under controlled conditions. By generating a comprehensive dataset on the germination ecology of S. halepense, this research provides information critical for predicting recruitment windows and improving the design of integrated weed management strategies.
Materials and Methods
Seed source and preparation
Seeds of two populations of S. halepense (SH 2/24 and SH3/24) were collected from the Emerald region of central Queensland. Population SH3/24 (24.198270°S, 148.435859°E) was collected from a cultivated sorghum [Sorghum bicolor (L.) Moench] field, and population SH2/24 (24.290291°S, 148.358460°E) was collected from fallows in July 2024. Seeds from more than 100 mature S. halepense plants were collected, cleaned to remove debris, oven-dried at 40 C for 2 d, stored in plastic containers at room temperature in the laboratory, and used in the experiments.
All experiments were conducted from 2024 to 2025 at the Queensland Alliance for Agriculture and Food Innovation (QAAFI) Weed Science Laboratory, University of Queensland, Gatton Campus, Australia. Unless otherwise stated, experiments followed a completely randomized design with four replications and were repeated once for confirmation.
Preliminary Assessment of Seed Dormancy
To assess whether freshly collected seeds possessed primary dormancy, we first evaluated their germination capacity under conditions known to be optimal for S. halepense. Seeds from both populations were germinated in two experimental runs in a completely randomized design with four replications. Twenty-five seeds were placed in 9-cm-diameter petri dishes lined with a double layer of Whatman No. 1 filter paper, moistened with 5 ml of distilled water, and incubated at alternating temperatures of 30/20 C with a light/dark photoperiod of 12 h for 15 d. This regime was selected based on previous studies showing that alternating temperatures within this range are optimal for germination of summer annual weeds, including S. halepense (Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014; Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). Germination under these conditions remained below 30% for both populations. Low germination in a favorable environment is a standard diagnostic indicator of seed dormancy. The observed reduction in germination therefore provides evidence for seed dormancy. The nature of dormancy was inferred from published work, which showed that S. halepense exhibits physiological dormancy, imposed by mechanical or biochemical inhibition within the seed coat and pericarp (Al Sakran et al. Reference Al Sakran, Almhemed, Dal and Ustuner2020; Nosrati et al. Reference Nosrati, Mathiassen and Alizade2012; Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). Based on these findings, we proceeded with two dormancy-breaking experiments designed to alleviate seed coat–imposed physiological dormancy.
Breaking Seed Dormancy using Sodium Hypochlorite (NaOCl)
Seed dormancy was tested using NaOCl. Seeds of SH2/24 and SH3/24 were immersed in 100 ml of a commercial NaOCl solution (42 g L−1, containing 40 g L⁻¹ Chlorine and 9 g L−1 NaOH) for 0, 1, 2, 4, and 8 h. After treatment, seeds were rinsed thoroughly under running tap water for 30 min and incubated as described in the general germination protocol described later. Germination was assessed every 3 d for 15 d. The experiment commenced in September 2024 and was repeated once at the end of the first experiment.
Breaking Seed Dormancy with Mechanical Scarification
Mechanical scarification with sandpaper was evaluated as a dormancy-breaking mechanism. Seeds of both populations were subjected to 0, 2, 6, and 8 rubbings with sandpaper treatment. Fifty seeds were placed on a sheet of P800 sandpaper and rubbed with another sheet according to treatment intensity. One complete back-and-forth movement constituted one pass. After treatment, 25 seeds per replication were incubated as described in the general protocol. Germination counts were recorded every 3 d for 15 d. The experiment commenced in September 2024 and was repeated once for validation
General Protocol for Seed Germination
Mechanical scarification was more effective than NaOCl treatments and was therefore adopted for all subsequent experiments. Seeds were scarified six times with P800 sandpaper before incubation. Twenty-five uniform, undamaged seeds were evenly placed in 9-cm-diameter petri dishes lined with two layers of Whatman No. 1 filter paper, moistened with 5 mL of water or appropriate treatment solution. Petri dishes were sealed in transparent, airtight ziplock bags to minimize evaporation and incubated at alternating temperatures of 30/20 C under a 12-h photoperiod unless otherwise specified. A fluorescent lamp providing a light intensity of 85 mol m−2 s−2 was used. For complete darkness treatments, petri dishes were wrapped in three layers of aluminum foil and opened only after 15 d. Seeds were considered germinated when the radicle reached 2 mm in length. Germination was recorded every 3 d for 15 d, after which no additional germination occurred. All subsequent experiments followed a factorial completely randomized block design with four replications, except for the burial depth experiment, which had three replications.
Interactive Effect of Light and Alternating Temperature on Germination
Seeds from both populations were incubated at alternating temperature regimes of 15/5, 20/10, 25/15, 30/20, and 35/25 C under light/dark and complete dark conditions to determine optimal temperature and light conditions for germination. These temperature scenarios simulated typical seasonal conditions in Queensland, corresponding to winter (June to August), spring (September to November), summer (December to February), and autumn (March to May) (Australian Bureau of Meteorology, 2025).
Effect of Salt Stress on Germination
To evaluate salinity tolerance, seeds were placed in petri dishes moistened with 5 ml of NaCl solutions of 0, 25, 50, 100, 150, 200, and 250 mM prepared from analytical-grade NaCl (Sigma-Aldrich, St Louis, MO, USA). These concentrations approximate salinity levels found in Australian cropping soils (Rengasamy Reference Rengasamy2006; 2010). All other conditions followed the general germination procedure.
Effect of Osmotic Potential on Germination
Osmotic stress was imposed using polyethylene glycol 8000 (PEG; Sigma-Aldrich) to create water potentials of 0, −0.1, −0.2, −0.4, −0.6, −0.8, and −1.0 MPa following Michel and Radcliffe (Reference Michel and Radcliffe1995). Seeds were germinated in petri dishes moistened with 5 ml of each PEG solution and incubated at 30/20 C.
Effect of Burial Depth on Seedling Emergence
To examine the emergence of both populations, 25 seeds per replication were sown at depths of 0, 1, 2, 4, 8, and 10 cm in pots (17.5-cm diameter by 18.5-cm height) filled with field soil consisting of 46% silt, 33% sand, 21% clay, 2.6% organic matter, and pH 7.5. The soil was collected from Gatton Research Station of the University of Queensland and sieved using a 3-mm metal sieve. Pots were placed in nonporous plastic trays and subirrigated to maintain field capacity in a screen house. Emergence (appearance of the shoot on the soil surface) was recorded every 3 d for 15 d. The experiment commenced in November 2024 and was repeated once.
Statistical Analyses
All experiments followed a completely randomized design with four replications, except for the burial depth experiment, which had three replications. Each experiment was repeated once. Data from the runs were combined when no significant (P > 0.05) run or run–treatment interaction effect was detected. Germination and emergence data (percentages) were checked for normality and homogeneity of variance using Shapiro-Wilk and Levene’s tests, and means were presented. ANOVA was performed on germination and emergence using R statistical software (R Core Team 2022) to test main and interaction effects at P ≤ 0.05 (detailed ANOVA tables are provided in the Supplementary Material).
For salt stress, osmotic potential, and burial depth experiments, germination or emergence responses were further analyzed using a three-parameter log-logistic model fit in SigmaPlot v. 15.0 (Systat Software, San Jose, CA, USA):

where by G/E is germination or emergence percentage at osmotic potential, salt concentration, or burial depth x; a is maximum germination/emergence %; x 50 is the NaCl concentration (mM), osmotic potential (MPa), or burial depth (cm) required to achieve 50% of the maximum germination/emergence response; and b is the slope of the curve. For salinity, the NaCl concentration that reduced maximum germination by 50% relative to the control (IC50) was derived from the fitted curve. t 50 (time to 50% of treatment-specific maximum germination) was extracted from the time-course curves. Model adequacy was evaluated by residual analysis and the coefficient of determination (R2), and graphs of fitted curves were generated in SigmaPlot v. 15.0.
Results and Discussion
Breaking Seed Dormancy Using NaOCl
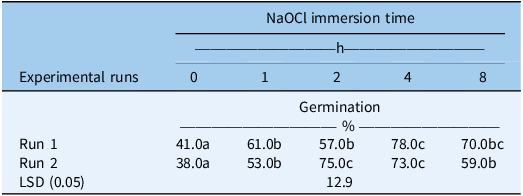
Seed germination of S. halepense was significantly influenced by NaOCl immersion time and experimental runs (Supplementary Table S1). Across both runs, the untreated seeds (0 h NaOCl) showed the lowest germination (38% to 41%). All NaOCl treatments significantly increased germination compared with the control, indicating the effectiveness of NaOCl in overcoming seed dormancy in S. halepense (Table 1). Germination increased progressively with immersion duration, reaching a maximum at 4 h of NaOCl immersion (78% in Run 1; 73% in Run 2). Although the overall response pattern was consistent between the two runs, the magnitude of the effect varied slightly at intermediate durations. This study suggests that NaOCl immersion substantially increased germination, confirming that physiological dormancy limits germination under favorable conditions, most likely by partially corroding or thinning the seed coat, which enhances oxygen diffusion and activates oxidative enzymes without fully removing the husk (Hsiao and Huang Reference Hsiao and Huang1988; Mohammadi et al. Reference Mohammadi, Noroozi and Nosratti2013; Nosrati et al. Reference Nosrati, Mathiassen and Alizade2012). The decline in germination at 8-h immersion, although still above that of the control, suggests a threshold beyond which prolonged exposure leads to oxidative injury or membrane disruption, consistent with previous reports (Balicevic et al. Reference Balicevic, Ravlic and Balic2016). This identifies 4-h immersion as a practical optimum, long enough to overcome dormancy and short enough to avoid damage. The significant NaOCl and population interaction indicates that the magnitude of response varied between populations.
Effect of sodium hypochlorite (NaOCl) immersion on the germination of Sorghum halepense incubated under alternating light/dark for 15 d at 30/20 C across two experimental runsa.

a Values are means ± SE (pooled across populations). Means followed by different letters are significantly different at P ≤ 0.05.
The greater variation between experimental runs at the 2-h and 8-h NaOCl immersion treatments is most likely attributable to the inherent sensitivity of S. halepense seed coats to chemical scarification. NaOCl can produce inconsistent levels of seed coat weakening, particularly when small differences in seed age, inherent seed lot variability, or NaOCl activity (which declines with storage time, temperature, and exposure to air) occur between runs. Because NaOCl immersion produced variable germination outcomes between runs, particularly at the 2-h and 8-h exposures, this method was not preferred for dormancy breaking; therefore, mechanical scarification was used as the standard approach for subsequent experiments, because it produced consistent results.
Breaking Dormancy Using Mechanical Scarification (Sandpaper Rubbing)
Sandpaper scarification had a significant effect on germination of S. halepense seeds incubated at 30/20 C under alternating light/dark conditions for 15 d (Table S2, Table 2). Seeds without scarification exhibited the lowest germination (43%), whereas four passes of scrabbing increased germination to 82%. Maximum germination was achieved at six and eight passes (89.0%), with no further improvement beyond six passes, indicating a clear threshold in the abrasion required to alleviate seed coat–imposed dormancy.
Effect of mechanical scarification (sandpaper rubbing) on the germination of Sorghum halepense incubated under alternating light/dark for 15 d at 30/20 C (averaged over experimental runs and populations).

a Means followed by different letters are significantly different at P ≤ 0.05.
Mechanical scarification with sandpaper increased germination across both populations, suggesting that seed coat dormancy plays a major role in regulating S. halepense germination (Balicevic et al. Reference Balicevic, Ravlic and Balic2016). The seed coat acts as a hard, partially impermeable barrier inhibiting water uptake and gas exchange, consistent with earlier reports on seed coat–imposed dormancy in this species (Al Sakran et al. Reference Al Sakran, Almhemed, Dal and Ustuner2020; Nosrati et al. Reference Nosrati, Mathiassen and Alizade2012; Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). Scarification removes or weakens physical barriers, enhancing water uptake and gas exchange, thereby enhancing embryo activation and rapid germination (Huang and Hsiao Reference Huang and Hsiao1987; Mohammadi et al. Reference Mohammadi, Noroozi and Nosratti2013). This strong positive response is consistent with reports from other authors, in which mechanical abrasion, such as rubbing seeds with sandpaper or nicking the seed coat with a knife, alleviates seed coat–imposed or combined physiological dormancy (Krenchinski et al. Reference Krenchinski, Albrecht, Albrecht, Villetti, Orso, Barroso and Victoria Filho2015; Nosrati et al. Reference Nosrati, Mathiassen and Alizade2012; Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014; Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). The limited increase in germination observed between six and eight rubbings indicates that the abrasion threshold required for germination, beyond which additional abrasion did not produce further improvement.
Critically, these findings are relevant for S. halepense behavior in eastern Australian farming systems, especially under conservation agriculture and short-fallow rotations. No-till and minimum-till systems common in Queensland and northern New South Wales maintain high surface-residue loads, microbial-mediated degradation, and repeated wet–dry cycles. This environmental pressure can mimic the abrasion effects observed here, gradually weakening the pericarp and hull and facilitating dormancy break over time (Chauhan and Manalil Reference Chauhan and Manalil2022; Thomas et al. Reference Thomas, Titmarsh, Freebairn and Radford2007; Thompson et al. Reference Thompson, Mahajan and Chauhan2021). Rainfall variability in eastern Australia is sporadic, with intense rainfall followed by extended dry periods that intensify these cycles, leading to irregular but strong germination flushes under low moisture conditions (Waha et al. Reference Waha, Clarke and Dayal2022). Disturbances caused by residue management, livestock movement, excessive tillage, and shallow mechanical operations can also wear down seed coats, inadvertently trigger germination, and increase the risk of early-season establishment (Krenchinski et al. Reference Krenchinski, Albrecht, Albrecht, Villetti, Orso, Barroso and Victoria Filho2015; Walsh et al. Reference Walsh, Broster, Chauhan, Rebetzke and Pratley2019). Conversely, the predictable germination flush following such disturbances could be used to optimize the timing of knockdown herbicide applications. These insights highlight the importance of integrated strategies that combine timing of disturbance, surface residue dynamics, predictions related to moisture availability, and chemical fallow management (Chauhan et al. Reference Chauhan, Gill and Preston2006a; Reference Chauhan, Gill and Preston2006b; Reference Chauhan, Singh and Mahajan2012; GRDC 2022). An improved understanding of mechanical dormancy release should support more accurate prediction of emergence windows, informing the design of effective integrated weed management strategies under eastern Australian cropping systems.
Interactive Effect of Light and Temperature Regimes on Germination
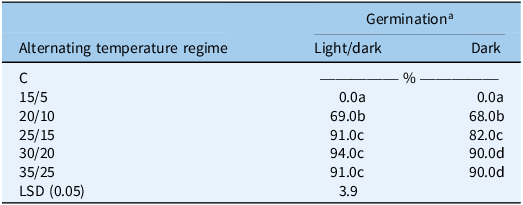
The interaction between light and alternating temperature regimes influenced the germination of S. halepense seeds (Table S3, Table 3). No germination occurred under light/dark and complete darkness regimes at the lowest temperature level (15/5 C), indicating that this winter temperature regime is below the basal thermal threshold required for S. halepense germination. This helps explain why seeds persist through winter and contribute to a multiseasonal seedbank (Benech-Arnold et al., Reference Benech-Arnold, Ghersa, Sanchez and Insausti1990a; Reference Benech-Arnold, Ghersa, Sánchez and Insausti1990b). Such a response is consistent with previous reports of optimal germination of S. halepense between 25 C and 35 C, with strong suppression occurring below 10 C and above 40 C (Ayse and İlhan Reference Ayse and İlhan2015; Krenchinski et al. Reference Krenchinski, Albrecht, Albrecht, Villetti, Orso, Barroso and Victoria Filho2015; Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). In the present study, germination increased progressively with temperature, consistent with the positive thermal response widely reported for this species (Egley and Chandler Reference Egley and Chandler1978; Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014). At 20/10 C, germination increased sharply to 69% under light/dark conditions and 67% in complete darkness, suggesting that 20/10 C lies close to the lower thermal threshold for germination. Although still relatively cool, this temperature is sufficient to support early-season emergence when adequate moisture is available.
Interactive effect of light and temperature regimes (15/5 to 35/25 C) on the germination of Sorghum halepense under light/dark (12-h photoperiod) and complete darkness after 15 d of incubation (averaged over experimental runs and populations).

a Means followed by different letters are significantly different at P ≤ 0.05.
High germination (∼90%) occurred at 30/20 C and 35/25 C under both light/dark and complete darkness, closely matching findings from earlier studies (Egley and Chandler Reference Egley and Chandler1978; McWhorter and Jordan Reference McWhorter and Jordan1976; Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014; Vargas Reference Vargas1995). These temperature regimes provide both adequate daytime warmth for metabolic activation and sufficiently cool nights to enhance thermal fluctuation cues, which are known to promote germination in S. halepense (Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014). At lower temperatures (25/15 C), germination was higher under light/dark (91%) than in complete darkness (82%), indicating that while light slightly enhanced germination, temperature is the dominant regulator.
This broad temperature tolerance (20/10 to 35/25 C) and high dark-condition germination reflect physiological flexibility that enables emergence under residue cover, shaded microsites, and at shallow soil depth, which are conditions common in eastern Australian conservation agriculture systems characterized by high residue retention and minimal disturbance (Anderson Reference Anderson2015; Anderson et al. Reference Anderson, Stephens, Siddique and Farooq2017; Walsh et al. Reference Walsh, Broster, Chauhan, Rebetzke and Pratley2019). This also indicates that a large proportion of the seedbank can emerge rapidly across much of the growing season. Light/dark and dark interactions further underscore that darkness suppresses germination at intermediate temperatures, while at higher temperatures, thermal energy predominates. This condition frequently occurs during spring and early summer in Queensland and New South Wales, where sporadic rainfall events rapidly warm soil profiles and trigger emergence flushes (GRDC 2022).
In eastern Australian cropping systems, crop rotations often occur under conservation agriculture, short fallows, and residue-dense surfaces, creating complex temperature–light interactions. Residue shading alone is insufficient to suppress emergence during warm periods, because S. halepense retains high germination capacity in complete darkness at optimal temperatures (Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). This allows emergence in high crop residue, under cover crops, or in unburnt stubble, increasing early-season competition risks. The absence of population-level differences in this study mirrors findings from other regions (Benvenuti et al. Reference Benvenuti, Macchia and Miele2001; Travlos et al., Reference Travlos, Gazoulis, Kanatas, Tsekoura and Zannopoulos2020, indicating that eastern Australian biotypes have a consistent germination ecology, which simplifies region-wide management recommendations.
This study suggests that climate change may exacerbate this issue. Higher average temperatures and frequent heat waves are likely to extend the germination window and expand the geographic distribution of S. halepense (Beaumont et al. Reference Beaumont, Hartenthaler, Keatley and Chambers2015; Navie Reference Navie2004; Parsons and Cuthbertson Reference Parsons and Cuthbertson2001; Queensland Government 2021; Sharp and Simon Reference Sharp and Simon2002). In warmer regions of Queensland, where high temperatures coincide with sporadic rainfall events, more frequent emergence flushes are expected, increasing the risk of seedbank replenishment and rhizome expansion (Šapčanin-Tabaković et al. Reference Šapčanin-Tabaković, Đikić, Gadžo, Grahić and Gavrić2015). The ability to germinate efficiently in darkness indicates that even deeply buried seeds can emerge once thermal thresholds are met, challenging management in reduced-tillage systems.
To manage S. halepense infestations effectively, strategies should prioritize disrupting or delaying germination by minimizing seedbank exposure to favorable temperatures. Practices such as the stale seedbed technique (allowing weed flushes before planting and then controlling them), use of surface mulches (to moderate soil temperature and light penetration), and targeted tillage (to either bury or expose seeds unfavorably) can help reduce emergence. In systems with early spring planting, timely preemergence herbicide applications may be critical. This study also highlights the importance of thermal-based models for forecasting germination flushes, enabling more timely and effective weed control interventions in cropping systems prone to S. halepense infestations.
Effect of Salt Stress on Germination
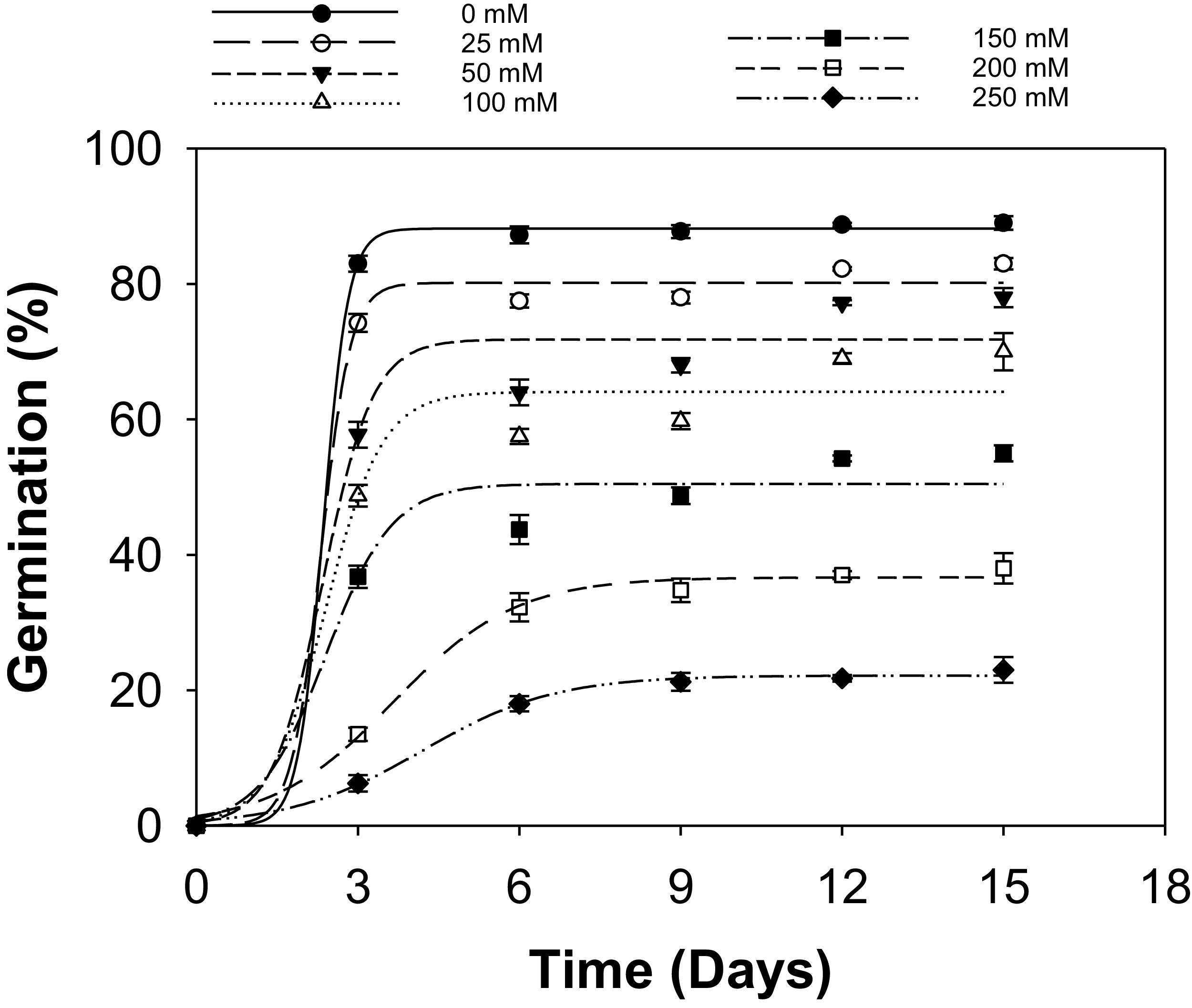
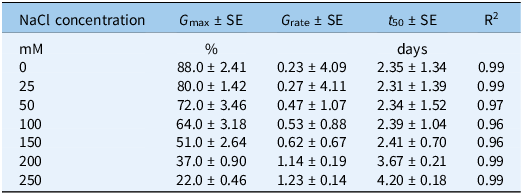
Sorghum halepense populations had similar behavior in their germination responses to different concentrations of NaCl (Table S4). Thus, data across the populations were averaged and regressed (Figure 1; Table 4). Germination dynamics of S. halepense were significantly influenced by salinity, reducing maximum germination (G max) and increasing the time to achieve 50% germination (t 50). Increasing NaCl concentration inhibited seed germination. Maximum germination (88%) was observed at 0 mM NaCl, with a t 50 of 2.4 d, showing high and uniform germination under non-stressful conditions.
Influence of sodium chloride (NaCl) concentration (mM) on seed germination of two Sorghum halepense (SH2/24 and SH3/24) populations (pooled data over populations) incubated at 30/20 C in light/dark conditions. The lines represent the sigmoid model fit to data with estimated parameters given in Table 4. Vertical bars represent the ± standard errors of the mean (n = 8).

Parameter estimates G max (maximum germination), t 50 (time to reach 50% of maximum germination), and G rate (slope of a three-parameter sigmoid model fit to the seed germination data in Figure 1).

There was a decrease in G max from 80% to 72%, at low salinity (25 to 50 mM NaCl), with the t 50 (2.3 d) remaining constant. The stability in t 50 suggests that moderate salinity reduces the proportion of seeds able to germinate, but does not substantially slow the germination of the tolerant fraction (Bewley et al. Reference Bewley, Bradford, Hilhorst and Nonogaki2013). Increasing NaCl concentrations from 0 to 150 mM resulted in a gradual decrease in G max from 88% to 51%. However, the t 50 value remained constant (2.4 d), reflecting a compressed and rapid phase of germination that may lead to more synchronized germination among salt-tolerant fractions of the seed populations once minimum conditions are met (Korres Reference Korres2016). This is a stress-avoidance mechanism that enables seeds to germinate quickly and uniformly as a response to moderate abiotic stresses (Almansouri et al. Reference Almansouri, Kinet and Lutts2001). The residual seeds that do not germinate due to stress can contribute to seedbank persistence (Chauhan et al. Reference Chauhan, Gill and Preston2006b; Korres Reference Korres2016). Together, these responses suggest that salinity does not eliminate germination potential but instead restricts emergence to a smaller, more salt-tolerant fraction of the seed population, while some seeds remain ungerminated. These ungerminated seeds are likely to persist in the soil and contribute to seedbank persistence under fluctuating salinity conditions typical of eastern Australian cropping systems, where salinity is largely environmentally driven. A distinct inflection in response occurred between 100 and 150 mM NaCl, where G max dropped from 64% to 50%, marking the transition from moderate to severe salinity stress. From the fitted nonlinear model, the NaCl concentration required to reduce G max by 50% relative to control (IC50) was approximately 173 mM, providing a quantitative threshold for salinity tolerance in these populations.
At high salinity levels (200 and 250 mM NaCl), G max decreased sharply from 38% to 22%, respectively, and t 50 increased from 3.7 to 4.2 d respectively. This shift indicates that high ionic and osmotic stress progressively impair both the capacity and speed of germination (Farooq et al. Reference Farooq, Bibi, Sarwar, Ihsan, Sarwar, Khaliq, Mubeen, Ali, Ramzan and Iqbal2022). This shift may influence the timing of the seasonal emergence of weeds, competitive behavior, and seedbank persistence in saline regions (Dinari et al. Reference Dinari, Meighani and Farzami2013). This type of trait is noteworthy in Australia, where salinity has already affected nearly a third of the soils, with an additional 16% impacted by saline groundwater (Rengasamy Reference Rengasamy2006). Resilience under saline conditions might allow S. halepense to become a significant threat in salt-prone regions. However, seed germination can be successful, but seedling establishment may fail later due to osmotic or ionic stress, necessitating studies to assess seedling growth and survival to evaluate performance under salt-stress conditions. If seeds germinate and establish under these conditions, this weed could provide competition to crops, such as grain sorghum, Columbus grass (Sorghum almum Parodi), and Sudangrass [Sorghum bicolor (L.) Moench ssp. drummondii (Nees ex Steud.) de Wet & Harlan; syn. Sorghum × drummondii (Steud.) Millsp. & Chase] and pose a serious management challenge (Yang et al. Reference Yang, Newton and Miller1990).
It is also possible that the genetic trait responsible for salt tolerance in S. halepense could offer the opportunity for crop improvement if the seedlings can establish under saline conditions. Incorporating salt-tolerant traits from S. halepense into the sorghum genome could enhance the salinity tolerance of grain sorghum, making it suitable for cultivation in marginal and salt-affected areas. There is a need to investigate the recruitment, competitiveness, phenology, and physiological mechanisms of S. halepense under salinity stress, particularly in comparison to crops like sorghum. Such insights are vital for developing effective integrated management strategies in changing climatic and edaphic conditions.
Effect of Osmotic Stress on Germination
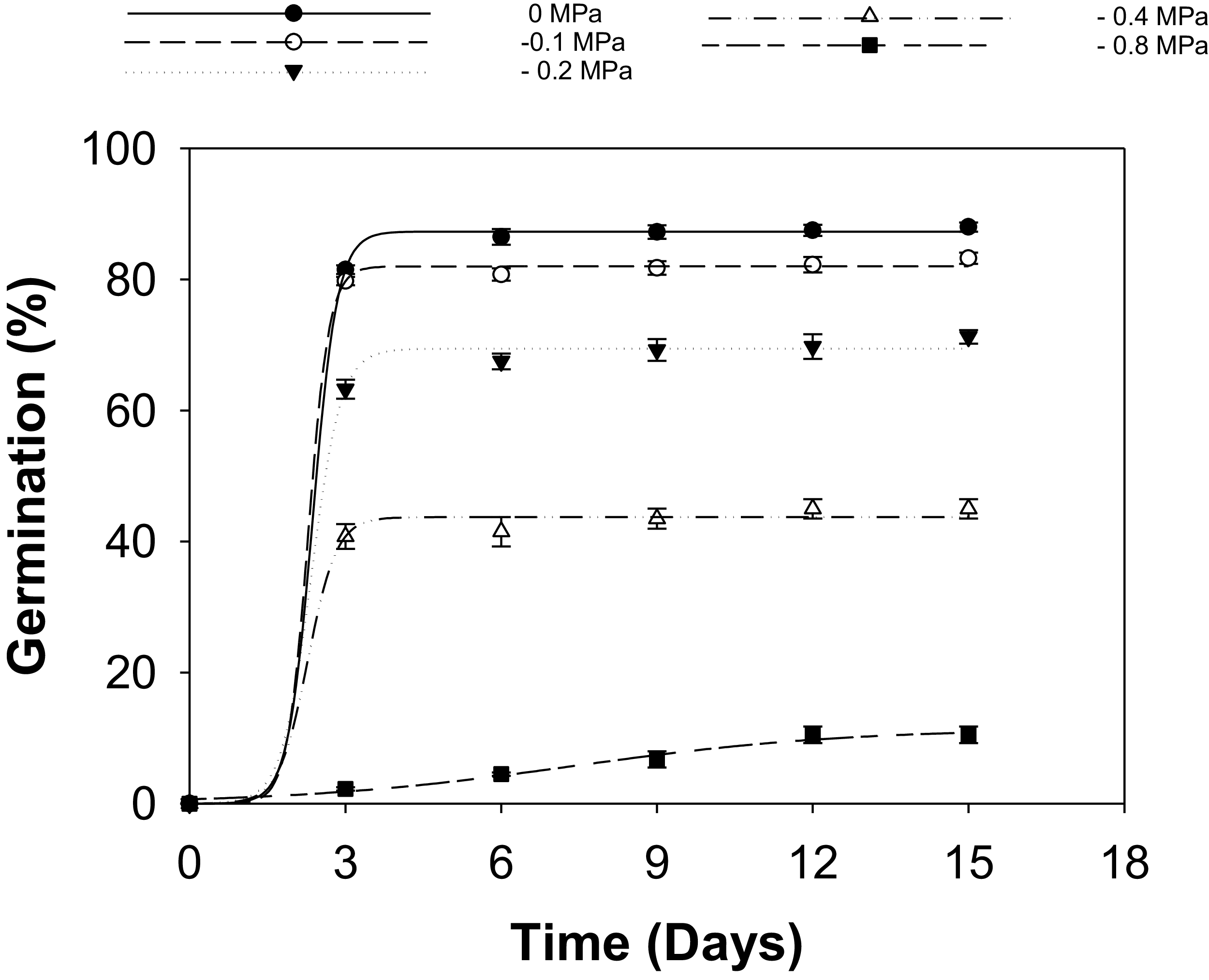
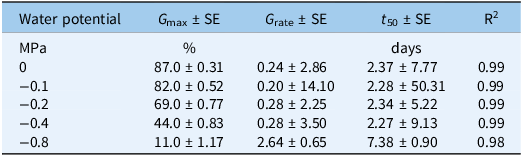
There was no significant interaction between populations and osmotic potential levels (Table S5); thus, data were pooled across populations and fit to a regression model. A sigmoid pattern was observed in S. halepense seed germination across varying osmotic potentials (Figure 2; Table 5). Germination declined progressively with decreasing osmotic potential, and this reduction was accompanied by slower germination under severe stress. At 0 MPa, seeds achieved a maximum germination percentage (G max) of 87%, with a maximum germination time (t 50) of 2.4 d. Moderate osmotic stress at −0.2 to −0.4 MPa decreased G max to 69% and 44%, respectively, while t 50 estimates remained near 2.3 d. This pattern suggests that a fraction of the stress-tolerant seeds germinates rapidly once moisture thresholds are met, while the remainder delays or fails to germinate. In contrast, severe osmotic stress at −0.8 MPa sharply reduced G max to 11% and increased t 50 to 7.4 d, marking a clear physiological failure threshold.
Influence of osmotic potential (MPa) on seed germination of Sorghum halepense (pooled data over populations) incubated at 30/20 C in light/dark conditions, modeled with the use of equation G% = G max/(1 + e [−(x − t 50)/G rate]) with estimated parameters given in Table 5. Vertical bars represent the standard errors of the mean (n = 8).

Parameter estimates G max (maximum germination), T 50 (time to reach 50% of maximum germination), and G rate (slope of a three-parameter sigmoid model fit to the seed germination data in Figure 2).

These findings align with patterns reported for other weed species, where only physiologically resilient weeds germinate under moderate water stress. Wild oats (Avena fatua L.) exhibit selective germination of tolerant individuals under similar osmotic conditions (Dinari et al. Reference Dinari, Meighani and Farzami2013), and bermudagrass [Cynodon dactylon (L.) Pers.] seeds show a comparable stress-filtering response, thus providing a potential competitive advantage in fluctuating climatic conditions (Rouhi et al. Reference Rouhi, Aboutalebian and Sharif-Zadeh2011). Furthermore, the osmotic potential threshold observed for S. halepense in this study was higher than that reported for other sorghum species. For instance, Columbus grass exhibited a sharp decline in germination at −0.9 MPa (Eberlein Reference Eberlein1987). Sorghum-Sudan grass hybrids (Sorghum bicolor(L.) Moench× Sorghum bicolor var. sudanense(Piper) Stapf) maintained 80% germination at −1.0 MPa (Hoveland and Buchanan Reference Hoveland and Buchanan1973), and grain sorghum germination was 79% at −1.0 MPa and 61%, even at −1.5 MPa (Evans and Stickler Reference Evans and Stickler1961). These comparisons suggest that S. halepense is less tolerant of moisture stress than some cultivated sorghums; however, its tolerance remains sufficient to allow establishment under the variable moisture regimes typical of eastern Australian cropping systems.
High germination under non-stress conditions indicates that S. halepense infestations may increase rapidly under favorable soil moisture conditions. In regions such as eastern Australia, where rainfall is more evenly distributed or crops are irrigated, S. halepense can exhibit enhanced seedling emergence, extended germination periods, and faster seedbank depletion (Australian Bureau of Meteorology 2025; Holm et al. Reference Holm, Plucknett, Pancho and Herberger1977; Warwick and Black Reference Warwick and Black1983). These dynamics can lead to increased early-season competition with crops, necessitating early and repeated weed control interventions.
The ability of S. halepense to germinate at −0.4 MPa (67%) can result in a significant ecological advantage in dryland agricultural regions of Australia, complicating the timing and effectiveness of control measures (Radosevich et al. Reference Radosevich, Holt and Ghersa1997). This ability enables the weed to germinate under fallow conditions or establish before the emergence of crops, particularly under intermittent rainfall or suboptimal soil conditions. Conversely, germination at −0.5 MPa implies that prolonged dry periods can limit emergence but simultaneously prolong seedbank persistence, complicating long-term management (Fletcher et al. Reference Fletcher, Varnon and Barney2020; Holm et al. Reference Holm, Plucknett, Pancho and Herberger1991; Mohler et al. Reference Mohler, Teasdale and DiTommaso2021; Shirdel et al. Reference Shirdel, Siahmarguee and Yones-Abadi2016). Overall, these water potential thresholds identify moisture conditions under which S. halepense is most likely to emerge in eastern Australian agroecosystems. Consideration of these windows should improve the timing of integrated management strategies, including fallow weed control, crop planting dates, and early-season interventions aimed at preventing establishment and reducing long-term seedbank inputs.
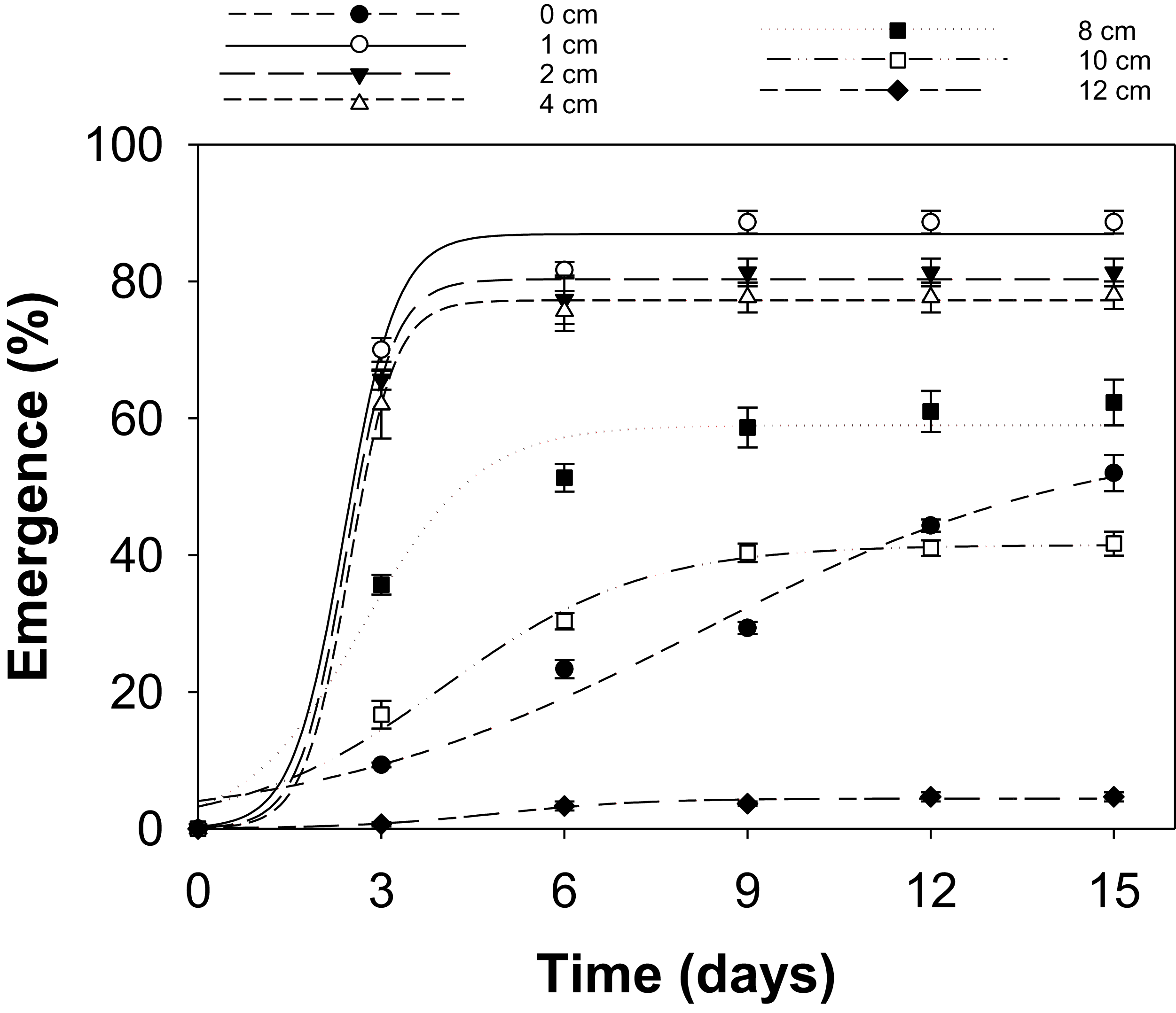
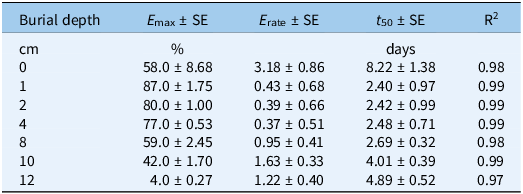
Effect of Burial Depth on Emergence
Seed burial depth significantly affected seedling emergence (P < 0.0001) (Table S6), influencing both the total emergence (E max) and time to 50% emergence (E 50). Population, runs, and all interactions were not significant (P > 0.05); data were pooled (Figure 3; Table 6). At 0 cm (soil surface), total emergence (E max) was 58%, while the time for 50% emergence (E 50) was markedly prolonged at 8.2 d. This delay may be attributed to suboptimal surface conditions, including poor seed–soil contact, greater diurnal temperature fluctuation, and increased exposure to microbial activity. At burial depths of 1, 2, and 4 cm, emergence was high, with E max values of 87%, 80%, and 77%, respectively. t 50 was similar across burial depths, with values of 2.40 d (1 cm) to 2.42 d (2 cm) and 2.48 d (4 cm), indicating the optimal burial depths for synchronous germination and seedling emergence, potentially allowing for improved weed management timings(Buhler Reference Buhler1991).
Influence of burial depth (cm) on seedling emergence of Sorghum halepense (pooled data over populations) incubated at 30/20 C in light/dark conditions, modeled with the use of equation E% = E max/(1 + e[−(x − t 50)/E rate]) with estimated parameters given in Table 6. Vertical bars represent the standard errors of the mean (n = 6).

Parameter estimates E max (maximum germination), t 50 (time to reach 50% of maximum germination), and E rate (slope of a three-parameter sigmoid model fit to the seed germination data in Figure 3).

There was a sharp decrease at 10 cm, with E max falling to 42% and t 50 increasing to 4 d. This is consistent with reported findings that S. halepense seedlings rarely emerge from seeds buried at depths greater than 10 cm (Benvenuti et al. Reference Benvenuti, Macchia and Miele2001; Leguizamon Reference Leguizamon1986). Emergence from 12 cm dropped sharply to an E max of 4% and t 50 of 4.9 d. Similarly, it has been reported that only 5% of S. halepense seedlings emerged from a 12-cm soil depth (Howard Reference Howard2004). Low emergence from deeper burial may have been caused by seedling mortality due to energy depletion before emergence, reduced oxygen availability, and increased mechanical resistance to seedling emergence (Benvenuti et al. Reference Benvenuti, Macchia and Miele2001). Across depths, a clear response threshold was evident. Emergence remained consistently high between 1 and 4 cm, but declined sharply beyond 4 cm, with a near-linear reduction from 8 to 12 cm. This depth threshold marks the transition from favorable to strongly restrictive emergence conditions. These findings indicate that shallow seed burial enhances rapid and uniform germination of S. halepense, while deep burial promotes long-term persistence by delaying seedling emergence and hindering germination, in agreement with earlier studies that reported higher emergence of S. halepense from shallow burial depths of 1 cm to 4 cm (Podrug et al. Reference Podrug, Gadžo, Grahić, Srebrović and Đikić2014; Shouhui et al. Reference Shouhui, Chaoxian and Chunhua2008; Tóth and Lehoczky Reference Tóth and Lehoczky2006).
Shallow tillage in S. halepense–infested fields may exacerbate infestation, especially under favorable climatic conditions, by bringing seeds to depths conducive to germination. Burying seeds deeper than 8 cm can be a cultural control approach to prevent the emergence of seedlings, enhancing the depletion of seedbanks through decay or death of seeds before germination (Macchia and Miele Reference Macchia and Miele2001; Warwick and Black Reference Warwick and Black1983). This soil inversion can bring seeds from deeper layers to the surface, where they are again exposed to conditions that promote germination. Therefore, it is important to know the seedbank longevity of S. halepense and the timing of previous deep-tillage operations to avoid bringing viable seeds to the surface. Previously buried seeds remain dormant, potentially serving as a survival strategy that contributes to the persistence of a latent seedbank capable of producing seedlings upon subsequent tillage or soil disturbance (Baskin and Baskin Reference Baskin and Baskin2014; Benvenuti et al. Reference Benvenuti, Macchia and Miele2001). Furthermore, germination delays from deeper burial can lead to asynchronous emergence, complicating the timing of control methods, such as cultivation or herbicide application, and reducing the effectiveness of control methods (Leguizamon Reference Leguizamon1986). Thus, there is a need for integrated management strategies that consider both temporal and spatial aspects of seed germination.
Sorghum halepense exhibited a consistent ecological strategy across both populations, combining strong primary dormancy, broad thermal tolerance, the ability to germinate under moderate moisture or salt stress, and a strong dependence on shallow burial for successful emergence. These traits help explain its persistence across diverse environments in eastern Australian cropping systems, indicating the periods and conditions under which recruitment is most likely. Although similar patterns have been described in North America and the Mediterranean (Travlos et al. Reference Travlos, Montull and Kukorelli2019), this study provides region-specific thresholds for Australian biotypes, particularly regarding dormancy, temperature, moisture stress, and emergence depth. The general consistency across populations suggests that recruitment behavior is predictable within the region, with germination favored by warm post-rain conditions and emergence largely restricted to shallow soil layers. Practices that retain seeds near the surface, such as reduced tillage or residue retention, are likely to increase recruitment risk, whereas deep burial limits emergence and extends seedbank persistence. The sensitivity of germination to moisture availability supports timely fallow control and the use of stale seedbeds to target early flushes. Integrating these ecological thresholds into tillage practices, herbicide timing, and fallow management can improve the effectiveness of control programs.
While these insights offer practical guidance for regional management, field-based studies across a broad range of populations are needed to refine emergence thresholds under varied field conditions, including soil types, microbial interactions, temperature, and moisture dynamics, and seedbank heterogeneity. Such validation will strengthen the application of these findings within integrated weed management programs in eastern Australia.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/wsc.2026.10099
Acknowledgments
LKK is grateful to the University of Queensland and the Grains Research and Development Corporation (GRDC) for the scholarship to undertake this study. An AI-based tool (ChatGPT, OpenAI) was used in a limited capacity to improve the clarity of wording in some sections.
Funding statement
This research was funded by the University of Queensland and GRDC.
Competing interests
The authors declare no conflicts of interest.