Introduction
The global incidence of cardiac arrest is over 55 per 100,000 person-years. Reference Yan, Gan and Jiang1 Case fatality rates are as high as 90%, and survivors and families remain at risk for significant morbidity and caregiver stresses. This is in addition to large health care and economic impacts. Reference Girotra, van Diepen and Nallamothu2–4
For patients in whom there is return of spontaneous circulation (ROSC), secondary reperfusion brain injury can occur. Reference Sandroni, Cronberg and Sekhon5 Approximately 80% of patients admitted to the intensive care unit (ICU) following cardiac arrest remain comatose, most often as a result of hypoxic ischemic brain injury (HIBI). Reference Nolan, Sandroni and Böttiger6 The initial focus of care is on minimizing secondary brain injury by optimizing cerebral perfusion and oxygenation, attenuating cerebral edema, providing temperature control, maintaining metabolic homeostasis, and treating seizures, which occur in up to 30% of patients. Reference Sandroni, Cronberg and Sekhon5 Postarrest myoclonus is also common and can complicate patient management by causing family and healthcare worker distress, ventilator asynchrony and the potential need for high doses of sedatives that confound neuroprognostication.
Despite supportive care, approximately 60% of postarrest patients die following withdrawal of life sustaining measures (WLSM) because of a predicted poor neurological prognosis. Reference Nolan, Sandroni and Böttiger6 As such, consistent, objective and evidence-based neuroprognostication is crucial to avoid premature WLSM. Accurate neuroprognostication can also circumvent prolonged, invasive, potentially harmful and costly therapies that could perpetuate patient and family suffering when there is no realistic chance of a favorable recovery. Several guidelines Reference Nolan, Sandroni and Böttiger6–Reference Rajajee, Muehlschlegel and Wartenberg9 outline an approach to neuroprognostication of postarrest patients that ensure confounders are excluded and sufficient time has passed, while also emphasizing multimodal assessment, including repeated neurological examinations, neuroimaging, electrophysiological testing and biomarker levels.
Electroencephalography (EEG) is one electrophysiological test that can be used to aid in the process of neuroprognostication but can also be used to support the diagnosis and management of postarrest seizures, myoclonus and potentially other contributors to secondary brain injury. There are several considerations regarding appropriate use of EEG that merit attention. This review explores these considerations, including EEG basics, postarrest indications and the logistics of performance and interpretation within the ICU.
EEG basics
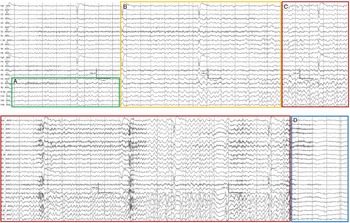
Electroencephalography displays brain activity as the electrical potential difference (voltage) between two cerebral locations plotted over time. Signals originate from neuronal inhibitory and excitatory post synaptic potentials generated from the underlying cerebral cortex. In clinical practice, surface electrodes are placed in accordance with the international 10-20 system on the head. Electrodes are labeled with letters (Fp – Frontopolar, F-frontal, T-temporal, P-parietal, O-occipital, C-central) and numbers (odd-left hemisphere, even-right hemisphere) corresponding to specific cerebral locations. Electrodes detect the cerebral signal that undergoes volume conduction through the meninges, skull and scalp. While other technologies are emerging, such as subdural strips/grids and intraparenchymal electrodes as part of multimodal brain monitoring for patients with various types of acute brain injury, they are not routinely used in postarrest patients. Once detected, the signal undergoes complex processing including differential amplification and filtering to attenuate signals that are not cerebral in origin (e.g., artifacts) and is then displayed as waveforms for interpretation (Figure 1A).
Electroencephalography (EEG) acquisition and interpretation. A - EEG acquisition. Signals generated in cerebral cortex are transmitted via volume conduction through the meninges, skull and scalp and detected by electrodes placed in accordance with the international 10–20 system. (Fp: frontal polar / F: frontal / T: temporal / P: parietal / O: occipital / C: central. Odd numbers for left side of head, even for right side of head, and z represents midline). The signal undergoes processing via amplifiers and filters (Hz: frequency) to attenuate signals that are not cerebral in origin and amplify those that are cerebral in origin. Channels display the voltage between two electrodes over time as waveforms. Channels are often grouped into anatomical chains to facilitate localization of signals. B - EEG interpretation *using American Clinical Neurophysiology Society Critical Care EEG Terminology descriptors divides EEG into background activity and superimposed abnormalities.

Interpretation of raw ICU EEG has been standardized by the American Clinical Neurophysiology Society’s Standardized (ACNS) Critical Care EEG Terminology. Reference Hirsch, Fong and Leitinger10 EEG background waveforms are described based on location, amplitude/voltage, frequency, morphology, continuity and reactivity. Reference Hirsch, Fong and Leitinger10 Superimposed abnormalities may include sporadic epileptiform discharges, rhythmic and periodic patterns (RPPs) and seizures, and brief potentially ictal rhythmic discharges (BIRDs). Reference Hirsch, Fong and Leitinger10 (Figure 1B)
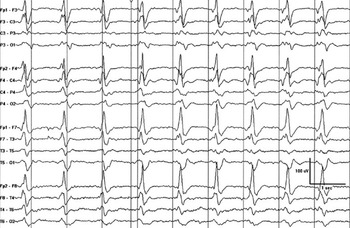
Electrographic seizures (ESz) defined by the Salzburg criteria Reference Hirsch, Fong and Leitinger10 include epileptiform discharges averaging > 2.5 Hz or discharges/rhythmic activity and associated spatiotemporal evolution (of morphology, location, or frequency) lasting ≥ 10 s (Figure 2). Electroclinical seizures (ECSz) include discharges or rhythmic activity with associated time-locked ictal clinical correlates; or both EEG and clinical improvement with administration of a parenteral antiseizure medication (ASM). Reference Hirsch, Fong and Leitinger10 Electrographic status epilepticus is ≥ 10 continuous minutes Reference Hirsch, Fong and Leitinger10 or over 20% seizure burden (e.g. 12 minutes of ESz over 1 hour) as it has been shown that over 20% seizure burden is associated with increased mortality and morbidity Reference Hirsch, Fong and Leitinger10,Reference Payne, Zhao and Frndova11 . Electroclinical status epilepticus can be defined similarly, or by greater than 5 or 10 minutes of generalized or focal clinical seizure activity respectively. Reference Hirsch, Fong and Leitinger10 BIRDs are defined as focal or generalized rhythmic activity that is either sharply contoured or similar in morphology and location as previously recorded epileptiform discharges or seizures in the patient, and last < 10 s. Reference Hirsch, Fong and Leitinger10
Electrographic seizure. Example of an electrographic seizure consisting of (A) Focal rhythmic sharp discharges in the right temporal region that (B) Evolve in frequency, morphology and location into (C) Generalized, maximal right temporal 3–4 Hz polyspike & wave activity. Once the seizure ends the electroencephalography demonstrates (D) Generalized suppression and slowing.

RPPs are defined in terms of the type of pattern (PDs-periodic discharges; RDA- rhythmic delta activity; SW-spike-and-wave or sharp-and-wave) and localization (G-generalized; L-lateralized; BI-bilateral independent; UI-unilateral independent; MF-multifocal). Reference Hirsch, Fong and Leitinger10 These patterns often lie within the ictal-interictal continuum (IIC). Reference Cormier, Maciel and Gilmore12,Reference Kalamangalam and Pohlmann-Eden13
Post-cardiac arrest EEG indications
Seizures and ictal-interictal continuum
Seizures have been reported in 3%–44% of postarrest patients. Reference Amorim, Rittenberger, Baldwin, Callaway and Popescu14–Reference Vespa, Martin and Nenov24 Rhythmic and periodic patterns classified as IIC have been reported in 10%–35% of comatose postarrest patients. Reference Rossetti, Oddo, Liaudet and Kaplan18,Reference Rossetti, Logroscino and Liaudet25–Reference Backman, Westhall and Dragancea29 These wide ranges likely reflect differences in classification of EEG patterns, timing and duration of EEG monitoring. EEG findings classified as IIC suggest an increased risk of seizures. Reference Cormier, Maciel and Gilmore12 Seizures and IIC have been associated with secondary brain injury, increased mortality and worsened recovery in critically ill patients. Reference Hirsch, Fong and Leitinger10,Reference Cormier, Maciel and Gilmore12,Reference Kalamangalam and Pohlmann-Eden13,Reference Claassen, Mayer, Kowalski, Emerson and Hirsch30–Reference Vespa, Tubi and Claassen40 Most seizures in critically ill patients are non-convulsive. Clinical manifestations may be confounded by sedation and neuromuscular blockade required for systemic concerns. Reference Amorim, Rittenberger, Baldwin, Callaway and Popescu14–Reference Dragancea, Backman, Westhall, Rundgren, Friberg and Cronberg22 As such, EEG is an important modality for diagnosis and management of seizures and status epilepticus (SE) in postarrest patients.
A routine or intermittent EEG (commonly referred to as a “spot” EEG) which is often performed for 20-30 minutes may miss more than 50% of electrographic seizures. Reference Claassen, Mayer, Kowalski, Emerson and Hirsch30 In addition, studies suggest that RPP and IIC may develop at various times post ROSC. Reference Beretta, Coppo and Bianchi23,Reference Ruijter, Keijzer and Tjepkema-Cloostermans41 In critically ill adults with impaired consciousness due to a variety of conditions including HIBI, continuous EEG leads to increased seizure detection and modification of antiseizure treatment, although with no effect on patient outcomes shown to date. Reference Rossetti, Schindler and Sutter42 While preferred for both diagnosis and to guide management, continuous EEG has several logistical considerations and may not always be possible.
When detected, uncertainty exists regarding whether treating IIC, seizures and SE is beneficial in comatose postarrest patients. Surveys demonstrate that there is variability of treatment approaches Reference Abend, Dlugos, Hahn, Hirsch and Herman43 and other literature suggests the effects of antiseizure medications (ASM) depend on the specific EEG pattern being treated. Reference Rossetti, Oddo, Liaudet and Kaplan18,Reference Backman, Westhall and Dragancea29
The recent TELSTAR trial Reference Ruijter, Keijzer and Tjepkema-Cloostermans44 investigated whether the treatment of RPP in comatose postarrest patients improved outcomes. This was a multicenter, randomized, open-label trial involving 172 patients, with blinded assessment of primary and secondary outcomes. Eligible patients were adult (≥18 years old), comatose (GCS ≤ 8) postarrest patients with RPP identified on continuous EEG. Patients in the intervention group received stepwise treatment with ASM based on international guidelines for the treatment of status epilepticus Reference Ruijter, Keijzer and Tjepkema-Cloostermans44–Reference Glauser, Shinnar and Gloss46 to suppress RPP for at least 48 hours (defined as more than 90% activity suppressed) in addition to standard postarrest care. Patients in the control group received standard care alone, which was left to the discretion of the treating physicians and followed European Resuscitation Council (ERC) guidelines. Reference Nolan, Sandroni and Böttiger6 In the control group the treating physician could utilize sedative medications for management of mechanical ventilation and treatment of myoclonus regardless of EEG findings but the additional use of ASM was discouraged. Decisions regarding WLSM during the trial were also made based on ERC guidelines. Reference Nolan, Sandroni and Böttiger6,Reference Ruijter, Keijzer and Tjepkema-Cloostermans44
At 3 months, 90% of the intervention group and 92% of control group had a poor outcome defined as a Cerebral Performance Category (CPC) of 3-5 (CPC 1: no/mild disability; CPC 2: moderate disability; CPC 3: severe disability; CPC 4: coma; CPC 5: death). Mortality was 80% and 82, respectively. Serious adverse events were similar between groups.
While informative, the TELSTAR trial has several important considerations that preclude generalization to all postarrest patients. Perhaps most important in the era of precision medicine is the fact nearly 80% of patients had generalized periodic discharges (GPD) (and < 10% actually had electrographic seizures), more than 50% had myoclonus and over 30% of those who underwent somatosensory evoked potentials (SSEPs) had bilaterally absent N20 responses. As such, it is possible that many of the RPP patterns that were being treated were simply epiphonema of established very severe HIBI. Beretta et.al. showed in their observational study that 44% patients with refractory status epilepticus and no other unfavorable prognostic indicators can obtain a good outcome (CPC 1-2) with aggressive and prolonged treatment of seizures. Reference Beretta, Coppo and Bianchi47 This was in stark contrast to those patients with GPD, of whom none survived with a good functional outcome. Although only hypothesis generating, the post hoc analysis in TELSTAR of GPD vs non-GPD patterns found that aggressive treatment of non-GPD patterns with continuous, non-suppressed background activity may be of benefit. However, a small sample size and large confidence interval limit this analysis.
Additional limitations include that only 56% of patients in the treatment arm obtained the goal of 48 hours of complete RPP suppression as this often requires aggressive dosing of sedatives when trying to suppress GPDs. While outcome assessors were blinded, physicians caring for patients were not, which may have introduced cognitive biases and influenced choices regarding medical therapies and WLSM. Post- arrest mortality was mostly related to WLSM due to a perceived poor prognosis. Concerningly, 16% of patients died of WLSM within 24 hours of randomization.
Taken together, we suggest that postarrest patients should undergo individualized precision-based management of seizures and RPP based on three pillars of (1) avoidance of secondary brain injury that can be caused by seizures as has been shown in several studies, Reference Claassen, Perotte and Albers33,Reference Vespa, Tubi and Claassen40,Reference Giovannini and Meletti48 (2) avoidance of the “self-fulfilling prophecy,” and (3) multimodal neuroprognostication. If seizures or non-GPD RPP are identified on EEG postarrest, particularly when they occur upon a continuous, non-suppressed background, it is reasonable to consider suppressing these patterns with anesthetic and/or ASM with the goal of attenuating secondary brain injury. One must be mindful to avoid a self-fulling prophecy and seek out other objective prognosticating indicators. If no indicators of a poor prognosis exist, then ongoing aggressive treatment of these EEG patterns should be considered with the context of other medically relevant information (e.g. patient age, comorbidities, other active medical issues, etc). If other poorly prognosticating indicators exist, it is possible that the RPP pattern is an epiphenomenon of an injured brain. Expert consultation can be helpful.
Postarrest patients may experience movements due to a variety of reasons other than electroclinical seizures including posturing, paroxysmal sympathetic hyperactivity, and myoclonus. A spot or prolonged EEG can be used to exclude electroclinical seizures in patients with atypical movements so long as they are captured during the test. This may allow for sedation to be weaned, avoidance of unnecessary ASM and initiation of other therapies targeted at the underlying issue.
Myoclonus
Myoclonus consists of sudden, involuntary, brief, irregular shock-like muscle movements. Myoclonus is common postarrest and occurs in 16%–37% of patients. It is often viewed as a single entity and historically as an indicator of poor prognosis. Reference Lybeck, Friberg and Aneman34,Reference Kramer49–Reference Sandroni, Cariou and Cavallaro54 However, studies have reported high false positive rates when using myoclonus in isolation to predict a poor prognosis. Reference Elmer, Rittenberger and Faro51,Reference Seder, Sunde and Rubertsson52,Reference Mikhaeil-Demo, Gavvala and Bellinski55–Reference Sivaraju, Gilmore and Wira57
Myoclonus may present with considerable clinical and electrophysiologic variability. It can occur shortly after ROSC or in a delayed manner. Clinical detection may be masked by sedation or neuromuscular blocking agents. Movements may be generalized, multifocal, or focal; can involve various anatomical distributions including eyelids, face, trunk and proximal/ and/or distal limbs; may be spontaneous, action or stimulus induced; and stereotyped (similar characteristics from one twitch to the next) or variable. Reference Kramer49
Historically, Lance-Adams syndrome, a form of action or stimulus induced myoclonus in patients who regained consciousness postarrest was thought to appear days to weeks post ROSC. However, it is now recognized that patients who evolve into this syndrome may have myoclonus immediately post ROSC. Reference Lee and Lee58,Reference Malhotra and Mohinder59 Clinical features of myoclonus in postarrest patients who have a good outcome have been described as asynchronous, multifocal, nonstereotyped and involving distal limbs. Status myoclonus has been variably defined in the literature, but is usually defined as continuous, spontaneous and generalized myoclonus that is unrelenting and lasting for 30 minutes or more. Reference Nolan, Sandroni and Böttiger6,Reference Kramer49,Reference Sandroni, Cariou and Cavallaro54 Patients with status myoclonus more often have a respiratory cause for their arrest, a non-shockable rhythm and features of postarrest syndrome (post-cardiac arrest global brain injury, myocardial dysfunction, systemic ischemia and reperfusion response and persistent precipitating pathology). Reference Neumar, Nolan and Adrie60 However, all these clinical features are imperfect when prognosticating patients, as studies demonstrate variable specificities ranging between 89%–100%. Reference Kramer49
EEG can assist in better characterizing postarrest myoclonus. Both cortical and subcortical forms of postarrest myoclonus exist. Specialized EEG techniques called jerk-locked EEG back averaging may help distinguish these two localizations. Reference Kramer49,Reference Reynolds, Rohaut and Holmes61 However, distinguishing between the two, does not necessarily aid in prognosticating patients, as one study reported that 12% and 16% of patients with cortical and subcortical myoclonus had good outcomes, respectively. Reference Reynolds, Rohaut and Holmes61
Important prognostic findings on EEG are background characteristics and the presence of highly malignant patterns. Patients with myoclonus, but also continuous, non-suppressed and reactive EEG backgrounds have greater potential for clinical improvement and favorable functional outcomes. It is important to note that patients with Lance-Adams syndrome may have epileptic discharges on their EEG. These discharges commonly occur in the parasagittal chains or central regions and can be quite prevalent, but do not indicate a poor prognosis (Figure 3). Reference Kramer49 Patients with status myoclonus, often have malignant EEG patterns, such as burst suppression or GPDs on a suppressed background. Reference Kramer49 They may also have absent N20 potentials on somatosensory evoked potential testing and concerning neuroimaging. Based on a multimodal approach as recommended in guidelines, Reference Nolan, Sandroni and Böttiger6–Reference Fordyce, Kramer and Ainsworth8 status myoclonus coupled with these other indicators is suggestive of a poor prognosis. However, particular caution is needed when myoclonus is not associated with other indicators of a poor prognosis.
Electroencephalography (EEG) from a patient with Lance-Adams Syndrome. EEG of a patient day 4 postarrest showing abundant spike/polyspike and wave mainly occurring in the parasagittal regions. Importantly these occur upon a background that is comprised of continuous, normal voltage mild slowing with reactivity. Clinically the patient had status myoclonus with generalized myoclonus persisting for greater than 30 minutes and was unresponsive. Valproic acid and clobazam were used to suppress the clinical myoclonus. Day 6 postarrest the patient became responsive, with ongoing action induced myoclonus in keeping with Lance-Adams Syndrome.

Neuroprognostication
Evidence supporting the use of EEG to aid in neuroprognostication has increased substantially. While older literature was limited by the use of several different EEG classification schemes, development of the ACNS Critical Care EEG Terminology Reference Hirsch, LaRoche and Gaspard62 has been an important step toward standardizing EEG assessments and research. Several studies have suggested that EEG can be used as one component of a multimodal approach to help predict prognosis. Unlike other prognosticating modalities, EEG can be used not only to help predict a poor prognosis but also shows promise in predicting a favorable prognosis.
Predicting a poor neurological prognosis
As the majority of postarrest patients die following WLSM, consistent, objective and evidence-based neuroprognostication is of the utmost importance. There are several EEG patterns that are considered highly malignant and raise concern for a poor prognosis, including burst suppression, suppressed background and generalized period discharges superimposed upon a suppressed background. The timing of these patterns in relation to a patient’s arrest is also an important factor to consider. Reference Nolan, Sandroni and Böttiger6,Reference Panchal, Bartos and Cabañas7,Reference Perera, Khan and Singh35
Burst suppression is a pattern consisting of 50-99% suppression alternating with bursts of higher voltage activity. Reference Nolan, Sandroni and Böttiger6,Reference Hirsch, Fong and Leitinger10 ACNS categorizes burst suppression patterns as those with or without highly epileptiform bursts and/or those with or without identical bursts. Reference Hirsch, Fong and Leitinger10 One study that separated burst suppression into synchronous (those with highly epileptiform and/or identical bursts) and heterogenous/non-synchronous (those without highly epileptiform and/or identical bursts) categories showed that the predictive value of these patterns in prognostication varied. Synchronous burst suppression patterns detected from 6-96 hours post ROSC predicted a poor neurological outcome at 6 months with a false positive rate (FPR) of 0% and high precision. Heterogenous burst suppression was predictive of a poor neurological outcome at 6 months with a FPR 0%–1.4% and high precision, but only if detected beyond 24 hours post ROSC (Figure 4). Reference Ruijter, Tjepkema-Cloostermans and Tromp63
Burst suppression patterns. Examples of (A) Highly epileptiform and identical burst suppression (aka synchronous burst suppression) vs (B) Heterogenous burst suppression (aka burst suppression that is not identical and does not contain highly epileptiform bursts). Synchronous burst suppression patterns detected up to 72 hours post return of spontaneous circulation (ROSC) predicted a poor neurological outcome at 6 months with false positive rate (FPR) 0% and high precision in a single study. Reference Ruijter, Tjepkema-Cloostermans and Tromp63 Heterogenous burst suppression in this same study was predictive of a poor neurological outcome at 6 months with a FPR 0%–1.4% and high precision only if detected beyond 16 hours post ROSC. Other studies assessing burst suppression that do not specify whether synchronous or heterogenous have shown that when present on an electroencephalography within 24 hours and beyond 24 hours from ROSC have a FPR of 2% (95% CI 1, 8) and 1% (95% CI 0, 2), respectively, for predicting a poor neurological outcome from hospital discharge to 6 months.

Many EEG recordings will show burst suppression in patients immediately post ROSC but will return to a normal voltage in less than 24 hours in patients destined for favorable neurologic outcomes. Reference Nolan, Sandroni and Böttiger6,Reference Cloostermans, van Meulen, Eertman, Hom and van Putten36,Reference Rundgren, Westhall, Cronberg, Rosén and Friberg37 Sedation required for systemic reasons, in addition to profound metabolic derangements, can also cause non-synchronous burst suppression in some patients who may have favorable outcomes. Reference Nolan, Sandroni and Böttiger6,Reference Amorim, Rittenberger and Zheng19,Reference Backman, Cronberg and Friberg21,Reference Perera, Khan and Singh35,Reference Cloostermans, van Meulen, Eertman, Hom and van Putten36,Reference Zhou, Maciel, Ormseth, Beekman, Gilmore and Greer38,Reference Westhall, Rossetti and van Rootselaar39,Reference Benarous, Gavaret and Soda Diop64–Reference Duez, Johnsen and Ebbesen66
A systematic review and meta-analysis of EEG patterns and outcomes postarrest found that timing of EEG recordings overlapped with sedation in 17/22 (77%) of studies reporting burst suppression and reported significant heterogeneity for burst suppression alone predicting a poor prognosis. Reference Perera, Khan and Singh35 This likely partially explains why other studies assessing burst suppression that do not specify whether synchronous or heterogenous have shown that when present on an EEG within 24 hours and beyond 24 hours from ROSC have a FPR of 2% (95% CI 1, 8) and 1% (95% CI 0, 2) respectively for predicting a poor neurological outcome from hospital discharge to 6 months. Reference Amorim, Rittenberger and Zheng19,Reference Backman, Cronberg and Friberg21,Reference Sivaraju, Gilmore and Wira57,Reference Ruijter, Tjepkema-Cloostermans and Tromp63,Reference Leão, Ávila, Cavaco, Germano and Bento65,Reference Scarpino, Carrai and Lolli67–Reference Rundgren, Rosén and Friberg77 (Figure 4) Guidelines recommend various time points when burst suppression can be used to predict outcomes. Regardless, it is important that physicians ensure no confounders (e.g., ongoing infusions or delayed clearance of propofol, barbiturates and midazolam), are present that could cause burst suppression, particularly when non-synchronous burst suppression is present.
A suppressed background is defined as voltages less than 10uV while an isoelectric background is less than 2uV. An isoelectric background, when combined with specific electrographic criteria (full set of electrodes, impedance 100-100,000 ohms, and interelectrode distance > 10 cm) is consistent with electrocerebral silence. Reference Hirsch, Fong and Leitinger78 Ideally, suppression should be measured and reported objectively using quantitative software such as amplitude integrate EEG due to challenges in visually interpreting the recording due to contamination from electrical noise at required voltage sensitivities. An isoelectric background detected within 72 hours of ROSC has been shown to predict a poor neurological outcome at 6 to 12 months with FPR 0-1.4% and high precision. Reference Amorim, Rittenberger and Zheng19,Reference Scarpino, Carrai and Lolli67,Reference Azabou, Navarro and Kubis79 (Figure 5A)
Isoelectric and suppressed electroencephalography (EEG) backgrounds. Examples of (A) Isoelectric (< 2uV) EEG background and (B) Suppressed (< 10uV) EEG background. An isoelectric background detected within 72 hours of return of spontaneous circulation has been shown to predict a poor neurological outcome at 6 to 12 months with false positive rate (FPR) 0%–1.4% and high precision. Suppressed backgrounds, especially when detected beyond 72 hours are also predictive of a poor neurological outcome with a FPR of 0% and high precision, however other studies assessing suppression at earlier time points have shown mixed results.

Suppressed backgrounds, especially when detected beyond 72 hours are also predictive of a poor neurological outcome with a FPR of 0% and high precision. Reference Backman, Cronberg and Friberg21,Reference Ruijter, Tjepkema-Cloostermans and Tromp63,Reference Scarpino, Carrai and Lolli67 Other studies assessing suppression at earlier time points have shown mixed results (Figure 5B). Reference Backman, Cronberg and Friberg21,Reference Ruijter, Tjepkema-Cloostermans and Tromp63,Reference Benarous, Gavaret and Soda Diop64,Reference Scarpino, Carrai and Lolli67,Reference Lamartine Monteiro, Taccone and Depondt69 The high FPRs and wide CIs within this time frame (especially < 24 hours postarrest) may be partially explained by the effect of sedation and metabolic derangements that are not well tracked in many studies reporting on the prognostic utility of EEG. Guidelines recommend various time points (beyond 24 and 72 hours) when suppression may be used as part of a multimodal approach to predict poor prognosis. It is crucial for providers to consider confounders that may cause suppression when using this as a marker for poor prognosis.
RPPs as previously outlined, comprise a heterogenous group of EEG patterns. Many studies combine these patterns together and have shown that when present on an EEG from ROSC up to 5 days postarrest, their FPR in predicting a poor neurological outcome at 1 to 6 months ranges from 0-33%. Reference Westhall, Rossetti and van Rootselaar39,Reference Benarous, Gavaret and Soda Diop64,Reference Lamartine Monteiro, Taccone and Depondt69,Reference Scarpino, Lolli and Lanzo80–Reference Rossetti, Tovar Quiroga and Juan82 The combination of multiple patterns into one category, makes it impossible to draw conclusions about each individual pattern. Studies specifically assessing generalized periodic discharges during the initial 72 hours postarrest Reference Amorim, Rittenberger and Zheng19 , particularly when superimposed on a suppressed (<10uV) background Reference Backman, Cronberg and Friberg21,Reference Ruijter, Tjepkema-Cloostermans and Tromp63 have consistently found them to predict a poor neurologic outcome with a very low FPR (Figure 6).
Generalized periodic discharges on a suppressed background. Generalized periodic discharges occurring upon a suppressed background (< 10uV) are predictive of a poor prognosis with false positive rate of 0%.

Studies assessing the association of electrographic seizures and status epilepticus with outcome have defined seizures in a variety of ways. Such studies have shown mixed results, regardless of the timing of seizure detection postarrest. Reference Amorim, Rittenberger and Zheng19,Reference Backman, Cronberg and Friberg21,Reference Dragancea, Backman, Westhall, Rundgren, Friberg and Cronberg22,Reference Zhou, Maciel, Ormseth, Beekman, Gilmore and Greer38,Reference Ruijter, Tjepkema-Cloostermans and Tromp63–Reference Leão, Ávila, Cavaco, Germano and Bento65,Reference Ihara, Sakurai, Kinoshita, Yamaguchi and Sugita68–Reference Oh, Park and Shon70 Although seizures and status epilepticus are often associated with poor neurological outcomes, this association was not consistent across all studies. Late appearing seizures (after TTM) Reference Backman, Westhall and Dragancea29 or those occurring on a reactive or continuous background, Reference Dragancea, Backman, Westhall, Rundgren, Friberg and Cronberg22 may suggest greater potential for recovery. Patients warrant an individualized multimodal approach to treatment and prognostication when electrographic seizures are identified.
Predicting a favorable neurological prognosis
While predictors of a poor neurologic outcome are more extensively assessed in the literature, there are increasing studies assessing electrographic predictors of good outcome. Reference Sandroni, D’Arrigo and Cacciola83 The presence of favorable prognostic findings can be helpful in counterbalancing pessimism that may exist in patients who have factors often associated with poor outcomes, such as prolonged arrest duration or a non-shockable rhythm. Reference Sandroni, D’Arrigo and Cacciola83
Several EEG background features have shown promise in predicting a good functional recovery. Continuous or nearly continuous activity, backgrounds comprised of relatively faster frequencies (alpha [8–12 HZ] and theta [4–7 HZ] rather than delta [1–3 HZ]), normal voltages, tracings demonstrating organization (e.g. an anterior-posterior gradient), EEG reactivity (change in EEG voltage or frequency with stimulation and eye opening) and that absence of superimposed discharges recorded from 12–72 hours from arrest generally have specificity varying between 50-100% for predicting a good functional outcome at 6 months. Reference Sandroni, D’Arrigo and Cacciola83 Another feature suggesting good outcome is the time to recovery of these favorable EEG background characteristics. Post ROSC, the EEG background is often suppressed or burst suppressed and gradually returns to continuous with normal voltages. While this transition is not specific for a good outcome, its timing is, as patients with outcomes typically exhibit recovery within 24 hours. Reference Backman, Cronberg and Friberg21,Reference Oh, Park and Shon70,Reference Sandroni, D’Arrigo and Cacciola83
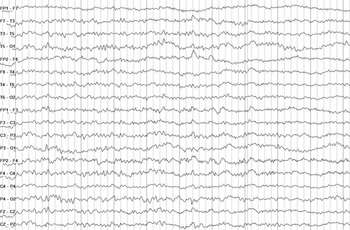
Available literature does not definitively determine which combination of favorable features most strongly predict a good outcome. However, the combination of a more continuous background, with fewer discharges, appearing early after ROSC is promising in numerous studies (Figure 7). When EEG reactivity is included in assessments, specificity for predicting a good outcome improves. However, the assessment of EEG reactivity remains non-standardized, with only fair interrater reliability. Further research is needed to define objective stimuli thresholds of frequency and voltage changes that indicate “reactivity”. Reference Azabou, Navarro and Kubis79
Continuous, normal voltage, mildly slow electroencephalography (EEG) background. Example of a continuous, normal voltage, mildly slow EEG background with no superimposed discharge recorded day 2 postarrest predictive of a good prognosis.

Evolving evidence for other EEG indications
Several quantitative and advanced EEG techniques with relevance to postarrest care and prognostication remain under investigation. Evolving literature suggests that advanced EEG techniques can aid in the detection of cerebral ischemia, cortical spreading depolarizations and intracranial hypertension. Reference Alkhachroum, Appavu and Egawa84 While much of this work has been conducted in patients suffering from other forms of acute brain injury such as traumatic brain injury and subarachnoid hemorrhage, the foundational principles of detecting and preventing secondary brain injury can also be applied to postarrest patients.
EEG has the potential to augment the accuracy of existing prognostication methods. Bedside clinical exams are a crucial component of prognostication and predicting recovery of consciousness. However, clinical examination of comatose patients is relatively crude, may be limited by confounders, and may vary due to inter-examiner differences, and potential cognitive biases. Reference Alkhachroum, Appavu and Egawa84 Standardized neurological exams may inappropriately classify patients as comatose and unresponsive despite covert consciousness Reference Alkhachroum, Appavu and Egawa84 which may influence WLSM in patients with HIBI postarrest. Reference Alkhachroum, Appavu and Egawa84,Reference Sitt, King and El Karoui85 Several EEG techniques have emerged as promising for augmenting the accuracy of prognostication models and may be beneficial for identifying covert consciousness in ICU patients and improving long-term outcomes. Reference Alkhachroum, Appavu and Egawa84,Reference Giacino and Edlow86 These include the use of various quantitative techniques and machine learning algorithms. Reference Alkhachroum, Appavu and Egawa84,Reference Claassen, Doyle and Matory87 EEG signals can be analyzed during resting state, but also with passive and active perturbations. Passive perturbation tasks evaluate the patient’s ability to detect stimuli (e.g., somatosensory, auditory), while active perturbation tasks evaluate the patient’s ability to interact (e.g., respond to a motor command).
These techniques require specialized equipment, and some are invasive (e.g., depth electrodes and subdural grids for detection of cortical spreading depolarizations). Specialized processing, machine learning and the expertise required for interpretation currently limit the accessibility and feasibility of these approaches.
Logistical considerations of ICU EEG
Advantages of EEG include that it is a relatively accessible, portable, noninvasive and safe means of monitoring brain activity. However, ICU EEG acquisition and interpretation require substantial human, technological and financial resources. In addition, several important logistical considerations need to be navigated to ensure ICU EEGs are performed and interpreted properly. There are several guidelines Reference Brophy, Bell and Claassen45,Reference Herman, Abend and Bleck88,Reference Claassen, Taccone, Horn, Holtkamp, Stocchetti and Oddo89 pertaining to EEG in ICU settings, but these provide limited guidance on the logistical challenges faced by many institutions.
Confounders
When performing and interpreting any EEG, one must be mindful of the effect of potential confounders at the time of the EEG. Ongoing sedative infusions, the lingering effects of drugs/medications taken in excess and contributing to a patient’s cardiac arrest, or profound metabolic disturbances seen in the context of the postarrest syndrome can influence the EEG. When EEGs are performed with the intent of excluding seizures, these confounders may result in false negatives and electrographic/non-convulsive seizures may develop as these confounders dissipate. When EEG is used for prognosticating purposes with or without associated myoclonus, these confounders can contribute to some of the previously described malignant EEG patterns such as suppression and or non-synchronous/heterogenous burst suppression. While it is common practice, if safe, to discontinue sedatives when patients are undergoing spot EEGs, it important to be mindful of the context sensitive half-life of sedatives in addition to the influence of temperature control and multi-organ dysfunction.
Continuous vs intermittent EEG
Controversy remains regarding the optimal duration of EEG depending on the indication. Studies have suggested that intermittent EEG may miss a large proportion of non-convulsive seizures. The recent pragmatic multicentre CERTA trial Reference Rossetti, Schindler and Sutter42 compared continuous (lasting 30–48 hours) to intermittent (2, 20–30 minute recordings) EEG in adult (>18 years) patients admitted with altered level of consciousness (GCS ≤ 11 or FOUR score ≤ 12) of any etiology (30% of whom had HIBI). Patients with prior seizures or status epilepticus were excluded from enrollment and if any patient was diagnosed with seizures or status epilepticus the protocol was stopped and they were subsequently treated with best practices including conversion to continuous EEG if required as it was considered unethical to withhold continuous EEG from these patients. The intermittent EEG group had a lower burden of comorbidities, while the continuous EEG group had more patients with HIBI. Continuous EEG did lead to increased seizure detection and more frequent modification of ASM, but no significant difference in patient mortality. Reference Rossetti, Schindler and Sutter42 It’s difficult to make definitive conclusions from this study because of bias that may have been introduced by ASM adjustments that were not protocolized and it is unclear whether ASM adjustments were made for EEG features on the IIC or solely for definitive electrographic seizures/status epilepticus. Considering the limitations of the CERTA trial, many of the previously discussed considerations regarding the TELSTAR trial remain important when deciding which postarrest patients may warrant continuous EEG for detection and/or management of non-convulsive seizures. A scoring system (2HELPs2B score) Reference Struck, Tabaeizadeh and Schmitt90 using both clinical and EEG features has been developed and validated for identifying higher-risk patients who may need longer EEG recordings for diagnosis of non-convulsive seizures, or conversely, when it is safe to perform only a routine EEG. When treating non-convulsive seizures in postarrest patient, continuous EEG may support goal directed management for those who have access to it. Other studies have assessed the yield of continuous vs repeat intermittent EEGs on postarrest prognostication and showed no difference. Reference Fatuzzo, Beuchat, Alvarez, Novy, Oddo and Rossetti81,Reference Alvarez, Sierra-Marcos, Oddo and Rossetti91 While serial EEGs have been suggested as an alternative to continuous, these too can be resource intensive and particularly more challenging for EEG technologists.
Acquisition considerations
Guidelines for qualifications of neurodiagnostic technologists exist in various countries. Reference López, Ahn-Ewing and Emerson92,Reference Dash, Dash and Primrose93 Experience can vary, and responsibilities should be tailored accordingly regarding the performance of EEGs, reactivity assessments and maintaining technical quality of continuous EEG recordings.
Standards exist for the equipment recommended for formal ICU EEG. Reference López, Ahn-Ewing and Emerson92,Reference Dash, Dash and Primrose93 While seizures are often non-convulsive in critically ill patients, synchronized video and audio recordings are recommended as they are very helpful in assessing for associations of EEG findings with changes in clinical behavior, and potential causes for artifacts.
In centers where continuous EEG is available, pending tests requiring patient transport (e.g., neuroimaging) ideally should be completed prior to connecting the patient to EEG. While specialized electrodes made with conductive plastic and non-ferrous metals that are compatible with both CT and MR imaging exist, Reference Abend, Dlugos, Zhu and Schwartz94,Reference Mirsattari, Lee, Jones, Bihari and Ives95 many centers may not have access, thus requiring patients to be disconnected and reconnected to EEG repeatedly to facilitate tests which is resource intensive, costly and demanding on technologist and physician time. Even when specialized electrodes are utilized, the risk of dislodging them from the patient is high upon transport. Patients undergoing continuous EEG require daily inspection for skin breakdown and infection of the scalp. Electrode impedance and artifacts necessitate lead maintenance and should be assessed regularly.
At times, during both continuous or intermittent EEG recordings, ICU bedside personnel may be required to administer medications for benzodiazepine/antiseizure medication trials when attempting to diagnose seizures. Often myoclonus or other patient movements that may have necessitated an EEG can cause profound muscle and movement artifacts and neuromuscular blocking agents may be required (when deemed safe and appropriate) to suppress these artifacts and allow visualization of the EEG signal underneath. ICU bedside personnel should be familiar with how to annotate EEG, as annotation of relevant information such as doses and titration of sedation/neuromuscular blocking agents, administration of ASM, and clinical signs that may not be evidence on the video recording is helpful for interpretation.
Interpretation considerations
Standards also exist regarding training and qualifications required for EEG interpretation Reference López, Ahn-Ewing and Emerson92,Reference Dash, Dash and Primrose93 and reporting. Reference Dash, Dash and Primrose93,Reference Tatum, Olga and Ochoa96 For ICU EEG, all reporting should use ACNS Critical Care EEG Terminology. Individuals must be mindful of the impact wording of EEG reports may have and clinical decision-making regarding seizure or myoclonus management and neuroprognostication. The interpretation of EEG results and incorporation into clinical decision-making should ideally be guided by individuals with content expertise.
Continuous EEG recordings produce a large amount of data that requires regular interpretation by neurophysiologists to ensure treatments are delivered in a timely manner. Raw EEG can undergo computational analysis via various mathematical techniques to produce quantitative EEG. There are several commercially available software packages that can produce several different graphs (also known as trends) that display various EEG features (frequency, voltage/amplitude, rhythmicity, presence of discharges, etc.) on Y and Z axes and time on the X axis. Commonly used trends include color and/or density spectral array (CSA, CDSA, DSA), rhythmicity spectrograms, relative asymmetry spectrograms, amplitude integrated EEG, suppression ratios, alpha/delta ratio, relative alpha variability. These can be augmented by seizure and RPP detection software. Quantitative EEG facilitates screening of long periods of EEG, detection of gradual changes over time, and identification of clinically relevant events that require further review of raw EEG. Relevant to postarrest care, quantitative EEG can be used to detect and quantify seizures and intracranial hypertension from cerebral edema, thus facilitating timely management. From a prognostication perspective it can assist with detection of EEG reactivity, continuous measurement of background suppression (versus a single spot measurement) and appreciating gradual background changes over time.
Resource limitations and bridging the gap
Resource limitations may impede timely EEG recording or interpretation, and thus delay necessary escalations or de-escalations of care.
Several technologies exist including peel and stick electrodes, electrode caps and bands, abbreviated montages and user-friendly EEG machines. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97 Studies suggest that training bedside personnel to utilize these technologies to perform EEGs (vs EEG technologists) results in a statistically significant reduction in the time delay to EEG by 86–667 minutes, no difference in the quality of the EEG, and most importantly changes in the diagnostic work-up and treatment of patients 49% and 40%–42% of the time respectively. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97 At institutions where EEG technologists may not be available around the clock, these devices, may help facilitate timely performance of EEGs by bedside ICU care providers. Alternatively at institutions that lack access to formal EEG (especially continuous EEG), these devices may assist in identifying patients who require transfer to other centers that can facilitate EEG monitoring. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97
At most centers, continuous EEG tracings are only reviewed remotely by neurophysiologists a few times per day. Bedside care providers can be trained to screen for non-convulsive seizures using raw, and sonified EEG. Various quantitative EEG trends have been used, resulting in 40%–93% sensitivity and 38%–95% specificity for seizure detection. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97 Sensitivity and specificity for seizure detection using raw EEG varied between 3 quantitative 8%–100% and 65%–100% respectively. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97 Bedside personnel screening sonified EEG for non-convulsive seizures has produced some of the most promising results with sensitivity 95%–100% and specificity 65%–85%. Reference Kromm, Fiest, Alkhachroum, Josephson, Kramer and Jette97 Detection of possible non-convulsive seizures by bedside care providers can help facilitate notification of neurophysiologists and timely therapies when appropriate. No studies to date have assessed non-EEG expert accuracy of interpreting other aspects of EEG that may be helpful for neuroprognostication of postarrest patients.
Conclusion
Electroencephalography is a relatively accessible, portable, noninvasive and safe means of evaluating a patient’s brain activity. It can aid in diagnosis and management decisions for postarrest patients with seizures, myoclonus and other non-epileptic movements. It also may aid in a multimodal approach to neuroprognostication predicting both poor and favorable outcomes. Individuals ordering, performing and interpreting these tests, regardless of the indication, must understand the supporting evidence, logistical considerations, limitations and impact the results may have on postarrest patients and their families.
Author contributions
-
CB contributed to the design of the review paper, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
CJ contributed to the acquisition, statistical analysis and interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
AS contributed to the statistical analysis and interpretation of data, draft and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
CA contributed to the interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
MS contributed to the interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
SVD contributed to the interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
AK contributed to the conception and design of the review paper, interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
-
JK contributed to the conception and design of the review paper, acquisition and interpretation of data, drafting and critically revising the manuscript for intellectual content and approved the final version of the manuscript.
Funding statement
This work was not funded.
Competing interests
-
CB has no disclosures.
-
CJ reports unrestricted educational grants from Eisai Inc. and UCB Canada for work unrelated to this study. He has received a grant from Epilepsy Canada for work unrelated to this study. He has received honoraria for speaking engagements from Eisai Inc., UCB Canada Inc. and Jazz Pharma Inc. for work unrelated to this study.
-
AS has no disclosures.
-
CA has no disclosures.
-
MS has no disclosures.
-
SVD has no disclosures.
-
AK has no disclosures.
-
JK was the electrophysiology lead for Neuroprognostication in Post Cardiac Arrest Patient: A Canadian Cardiovascular Society Position Statement.