Introduction
The helminth fauna of small mammals found in the British Isles and on the European mainland is well known (Lewis Reference Lewis1987; Feliu et al. Reference Feliu, Renaud, Catzeflis, Hugot, Durand and Morand1997; Sáez-Durán et al. Reference Sáez-Durán, Debenedetti, Sainz-Elipe, Sabater-Tena, Galán-Puchades and Fuentes2021; Lewis et al. Reference Lewis, Morley and Behnke2023 and references cited therein). The life cycles of the dominant rodent-infecting nematode genera Heligmosomoides, Heligmosomum, Syphacia and Aspiculuris have all been thoroughly described and are well documented in the literature (Durette-Desset Reference Durette-Desset1968; Lewis Reference Lewis1968a, Reference Lewis1987; Bryant Reference Bryant1973; Lewis and D’Silva Reference Lewis and D’Silva1986). The species in these genera are all directly transmitted, either following a brief spell as free-living organisms (Heligmosomoides spp., Heligmosomum spp.) or without a free-living stage, via rapidly embryonating eggs passed from one infected host to another (Syphacia spp.) or via eggs with temporary residence in the environment (Aspiculuris spp., Trichuris spp.). In contrast, much less is known about the life cycles of helminths that employ intermediate hosts (e.g. Cestoda, Digenea and even some Nematoda). It is perhaps surprising that, given more than a century and a half of research on the helminths of wood mice and bank voles, the full life cycle of one of the species that is often dominant in wild rodent populations is still so poorly known.
The digenean species Corrigia vitta belongs to the family Dicrocoeliidae and is often reported to show high prevalence in wood mouse populations (Tables 1 and 3; Langley and Fairley (Reference Langley and Fairley1982) recorded a prevalence > 90% in June). It inhabits an unusual location in its host and has a life cycle that is still incompletely documented. Nevertheless, the development of more model species, facilitating laboratory exploration as well as field studies, is urgently required in order to deepen our understanding of the Trematoda (Poulin Reference Poulin2025), and this widespread European species has potential in this regard. Here, we trace the history of changes in the nomenclature of this species and highlight studies that have reported components of its life cycle. We also review the epidemiology of C. vitta, searching for commonalities between studies that have assessed the effects of intrinsic and extrinsic variables on both prevalence and abundance of the species in wood mice, and we evaluate the influence of extrinsic and intrinsic factors on these measures of infection. Finally, we identify key questions that merit research attention.
Historical background of nomenclature changes
In his magnum opus, Dujardin (Reference Dujardin1845) first reported this species as Distoma vitta, providing an incomplete description because he only examined the posterior portion of 1 worm extracted from the small intestine of a wood mouse in France. He dealt with the species in section 4 of his monograph, concerned with the Brachylaimus-like digenea (p 418 of Dujardin Reference Dujardin1845). In his reference list of trematode parasites of British mammals, in a section entitled ‘Unclassified species’, Nicoll (Reference Nicoll1927) also referred to this species as D. vitta.
The genus Lyperosomum (Dicrocoeliidae, Odher, 1910) was erected half a century later by Looss (Reference Looss1899). Fragmented sections of the worms, collected by Elton from wood mice trapped near Oxford (reported later in Elton et al. Reference Elton, Ford, Baker and Gardiner1931), were examined by Baylis (Reference Baylis1927), who moved the species to Lyperosomum, and the worm then became L. vitta, but not for long. Following Travassos (Reference Travassos1944), the species was moved next to the newly created genus Orthorchis, as O. vitta. This change in genus was not recognized by some subsequent authors for over a decade (e.g. Thomas Reference Thomas1953; Dawes Reference Dawes1968, both referred to L. vitta). However, issues of priority were raised by Dollfuss (Reference Dollfuss1954), who drew attention to the work of Strom (Reference Strom1940). The latter author had revised the genus Lyperosomum and had erected Corrigia for species with the specific characters attributed by Travassos to Orthorchis. The parasite is thus currently known as Corrigia vitta (Dujardin Reference Dujardin1845) Strom Reference Strom1940.
Descriptions of Corrigia vitta
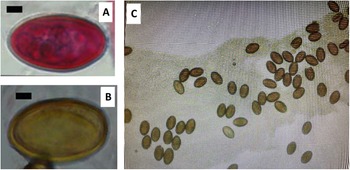
Baer (Reference Baer1932) provided the first detailed drawing of the anatomical arrangements of C. vitta, and a good drawing of the worm is also provided by Harvey and Channon (Reference Harvey and Channon1956). We have included this drawing in Figure 1C, as well as a photograph of a stained worm extracted from an infected wood mouse, caught in Surrey. Both show clearly the 2 testes located anteriorly, and the ovary located more posteriorly. The asymmetrically developed vitelline glands lying laterally on both sides of the worms, with one extending further in a posterior direction than the other, are also evident, as is the uterus containing eggs. Harvey and Channon (Reference Harvey and Channon1956) tabulated a list of dimensions of key morphometric characters of C. vitta and compared their values to those provided by some earlier workers (Table 1 in Harvey and Channon Reference Harvey and Channon1956).
Corrigia vitta, adult worms. A. Complete worm extracted from the bile duct of a wood mouse from Surrey (labels are as follows: OS, oral or anterior sucker; VS, ventral sucker; T1, anterior testis; T2, posterior testis; Ov, ovary; Vit, vitelline glands; Ut + eggs, uterus containing eggs). B. Transverse section through a pancreatic duct showing 5 worms lying alongside one another. C. Whole mount drawn by camera lucida under 42 mm. Objective and ×10 ocular lenses. Image copied from Harvey and Channon (Reference Harvey and Channon1956). A and B are images taken from microscope slides in the collection of the late Prof John W. Lewis, held at the Department of Biological Sciences, Royal Holloway, University of London (with permission of his family). A was stained with borax carmine stain and B with haematoxylin and eosin stain. Scale bars in A and B, 0·5 mm, and in C, 2·0 mm.

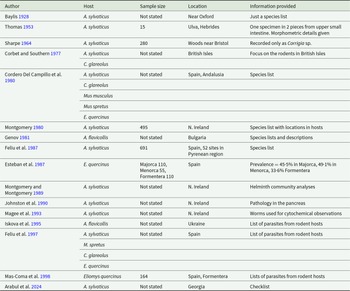
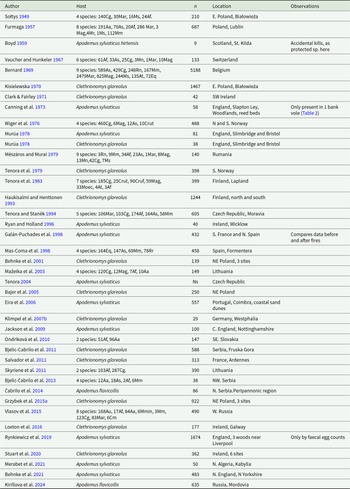
Selected studies of the occurrence of Corrigia vitta in wood mouse (Apodemus sylvaticus), yellow-necked mouse (A. flavicollis) and Ural mouse (A. uralensis) populations

a Apodemus sylvaticus
b Yellow-necked mice, Apodemus flavicollis.
c Ural field mouse, Apodemus uralensis.
d Monthly values, from June to May, for the number of parasites per mouse are provided in the paper.
Ng = not given in the paper.
In particular, eggs are small, typically brown in colour, with a smooth surface. They are operculated at 1 pole and when discharged from the adult parasite are embryonated, i.e. contain a fully developed miracidium (Figure 2; Baer Reference Baer1932; Ashour Reference Ashour1995). Recorded egg size can vary between different sampling locations (Baylis Reference Baylis1927; Baer Reference Baer1932; Harvey and Channon Reference Harvey and Channon1956), although it has been suggested that a ‘normal’ size may be approximately 37–38 μm length and 18–23 μm width (Harvey and Channon Reference Harvey and Channon1956). Such potential variation in size makes diagnosis using egg morphology unreliable, and consequently, infections are typically determined through dissection.
Eggs of Corrigia vitta. A. Egg stained by haematoxylin and eosin stain. B. Unstained egg. C. Compressed posterior section of an adult worm, with released eggs. A and B are images taken from microscope slides in the collection of the late Prof John W. Lewis, held at the Department of Biological Sciences, Royal Holloway, University of London (with permission of his family). Scale bars in A and B are 5 μm.

More recently, the morphology and cytochemistry of the adult parasite have been studied at the ultrastructural level (Robinson and Halton Reference Robinson and Halton1982, Reference Robinson and Halton1983, Reference Robinson and Halton1984; Magee et al. Reference Magee, Cahir, Halton, Johnston and Shaw1993; Ashour Reference Ashour1995). In particular, tegument structure has been found to be highly specialized. The distribution and forms of mitochondria in the tegument suggest that the ventral surface of the parasite, which is in close approximation with the host pancreatic duct brush border, may be functionally specialized for absorption at the relatively high local oxygen tension that may occur in this microanatomical context during host feeding cycles. On the other hand, the dorsal tegument may be adapted for energy metabolism during prevailing periods of anaerobiosis (Robinson and Halton Reference Robinson and Halton1983, Reference Robinson and Halton1984).
Taxonomic identity and related species
Based on Dawes (Reference Dawes1968), Corrigia vitta is currently assigned to the family Dicrocoeliidae Odher, 1910 (subfamily Dicrocoeliinae, Looss Reference Looss1899). The Dicrocoeliidae comprise over 400 described species, which are hepatopancreatic specialists living predominantly in the liver, gall bladder, pancreas and associated ducts. Most parasitize birds and mammals, including domestic animals (Otranto and Traversa Reference Otranto and Traversa2002; Manga-González et al. Reference Manga-González, Ferreras and Kafle2024), but some species also have reptiles and marsupials as definitive hosts. One characteristic of this family of trematodes is that their life cycles involve 2 intermediate hosts and can be completed in terrestrial environments without access to water (Pojmańska Reference Pojmańska, Bray, Gibson and Jones2008). The family contains many genera, including species that parasitize rodents in Africa (e.g. Paraconcinnum leirsi, Ribas et al. Reference Ribas, Makundi and Goüy de Bellocq2012) and S. America (e.g. Platynosomoides lunaschiae, Martins et al. Reference Martins, Panisse, Robles, Diaz and Navone2022).
Other Corrigia spp. have also been described, and mostly from birds, including Phasianidae (C. petrowi Sultanov Reference Sultanov1961, in the rock partridge, Alectoris graeca; C. corrigia in black grouse, Lyrurus tetrix; Tizzani et al. Reference Tizzani, Fanelli and Belleau2021), Anatidae (C. obscura, Daniels and Freeman Reference Daniels and Freeman1976 in the N. American black duck, Anas rubripes) and Numididae (C. vulturini, Lori and Balbo Reference Lori and Balbo1990 in vulturine guineafowl, Acryllium vulturinum).
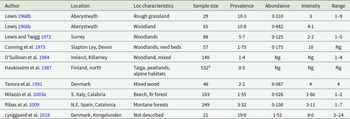
The only molecular sequence data of reliable provenance that we are aware of for Corrigia is a single 18S rDNA sequence obtained from C. vitta (Ribas et al. Reference Ribas, Makundi and Goüy de Bellocq2012; GenBank ref, JN831599, 1770bp). Based on 18S rDNA sequences, Ribas et al. (Reference Ribas, Makundi and Goüy de Bellocq2012) identified 2 genetic clades in the Dicrocoeliidae, with C. vitta clustering with Concinnum, Eurytrema, Lyperosomum and Parasconcinnum spp. The second clade incorporated Dicrocoelium and Brachylecithum spp. However, a similar analysis (Figure 3), to which more recent 18S rDNA sequences for other dicrocoeliid species have been added, returns C. vitta as a singular lineage within the Dicrocoeliidae not falling within any significant subfamilial cluster. This may be a function of the limited amount of information in the currently available 18S rDNA sequences to estimate increasingly complex trees, and is indicative that multi-locus analyses may be required in future to resolve the genus-level relationships of Corrigia within the family.
Updated phylogenetic analysis of dicrocoeliid trematodes based on 18S rDNA, showing the position of Corrigia vitta, employing longer sequences (>1400 bp) currently available in GenBank (see Ribas et al. Reference Ribas, Makundi and Goüy de Bellocq2012, for previous analysis). Where more than 1 long sequence was available for a nominal species, a single arbitrarily selected sequence was included per species. Sequences for members of other plagiorchiid families are included as outgroups. Analysis was carried out via the Maximum Likelihood method (following Ribas et al. Reference Ribas, Makundi and Goüy de Bellocq2012) in MEGA11 (Tamura et al. Reference Tamura, Stecher and Kumar2021), employing a general time reversible model (Nei and Kumar Reference Nei and Kumar2000) with invariant sites. Initial trees were obtained via the BioNJ method applied to pairwise distances estimated by Maximum Composite Likelihood (MCL). The inferred tree (log likelihood −3286) is shown with evolutionary distances indicated via the scale (substitutions per site). Bootstrap support (Felsenstein Reference Felsenstein1985) for clusters is indicated where this is greater than 70% (based on 1000 replicates). The analysis was based on 1434 base positions, excluding all positions with gaps or missing data.

Life cycle
The full life cycle of C. vitta has not been determined, although much speculation exists in the literature. Some authors have promoted the Dicrocoelium dendriticum life cycle as a suitable template for C. vitta, going so far as to suggest that ants may also act as second intermediate hosts for this species (Canning et al. Reference Canning, Cox, Croll and Lyons1973; McCarthy et al. Reference McCarthy, Litten-Brown and Collins2023). However, there is no evidence to support this analogy.
The C. vitta life cycle has not been satisfactorily resolved experimentally, with only the studies of Schmidt (Reference Schmidt1967, Reference Schmidt1969) providing tentative, if not conclusive, evidence of the hosts involved. The Dicrocoeliidae family have 3 or 4 hosts in their life cycles. It has proved possible within this family to differentiate amongst known life cycles a number of types related to taxa, subfamilies or genera (Niewiadomska and Pojmańska Reference Niewiadomska and Pojmańska2011). The genus Corrigia is considered to be related to Eurytrema (see Figure 3), with both having daughter sporocysts that are highly specialized and secondarily altered (Galaktionov and Dobrovolskij Reference Galaktionov and Dobrovolskij2003). Experimental studies of a number of species within these 2 genera have allowed the determination of a ‘Type-Eurytrema’ which encompasses a molluscan first intermediate host, an arthropod second intermediate host and a terrestrial mammal definitive host. This type possesses xiphidiocercariae, with only a rudimentary tail, that do not emerge from the sporocyst (Pojmańska Reference Pojmańska, Bray, Gibson and Jones2008; Niewiadomska and Pojmańska Reference Niewiadomska and Pojmańska2011).
Within the genus Corrigia, these life cycle patterns have been most extensively studied for Corrigia corrigia (Panin and Romanenko Reference Panin, Romanenko and Gvozdev1978; Žďárská Reference Žďárská1986). The adults of this species reside in the pancreas of gallinaceous birds. Eggs are released into the environment within faeces and consumed by a terrestrial snail intermediate host. Cercariae develop in daughter sporocysts, which lack a birth pore. Once the cercariae are fully developed, the sporocyst migrates through the snail’s tissues until it reaches the mantle cavity, where it is expelled into the environment. A well-developed endocyst surrounds the sporocyst, protecting it from climatic conditions. An isopod second intermediate host ingests the cyst (Hemilepistus fetschenkoi and Orthometopon planum in Panin and Romanenko Reference Panin, Romanenko and Gvozdev1978), and once within the body of the host, the cercariae penetrate the tissues and develop into metacercariae. Transmission to the definitive host occurs through predation on isopods as part of their normal diet (Panin and Romanenko Reference Panin, Romanenko and Gvozdev1978). A detailed account of the structural and histochemical characteristics of the transmission stages from the snails to the isopod second intermediate host is given by Žďárská (Reference Žďárská1986).
Schmidt (Reference Schmidt1967, Reference Schmidt1969) studied the trematode parasites of terrestrial snails over a number of years in a habitat in which wood mice (A. sylvaticus) were commonly infected with C. vitta. The snail Clausila bidentata was frequently infected with Dicrocoeliidae sporocysts containing xiphidiocercariae with rudimentary tails. A second mollusc, Cochlodina laminata, was also infected less frequently by a morphologically similar parasite. Both of these snail species have also been recorded as hosts of C. vitta in the Ukraine (Iskova et al. Reference Iskova, Sharpilo, Sharpilo and Tkach1995; Gibson et al. Reference Gibson, Bray and Harris2005). Although Schmidt (Reference Schmidt1967, Reference Schmidt1969) failed to find naturally infected isopods among 523 that he examined, when he experimentally fed infected C. bidentata and C. laminatas to woodlice (Philoscia muscorum, Porcellio scaber), Dicrocoeliidae metacercariae were recovered from 30% of fed isopods. Two wild A. sylvaticus caught from a different habitat, where no natural C. vitta infections had been recorded, were fed on metacercarial cysts and specimens of C. vitta were subsequently found in the pancreas of these mice. However, Schmidt (Reference Schmidt1967, Reference Schmidt1969) was unable to completely eliminate the possibility of a pre-existing infection in these wild-caught mice, raising questions about the reliability of his experiments, and so far, no follow-up experiments have been undertaken. Nevertheless, the studies of Schmidt (Reference Schmidt1967, Reference Schmidt1969) follow the life history pattern of the ‘Type-Eurytrema’, as would be expected, and consequently woodlice remain a strong possibility as the most likely and commonest second intermediate host of C. vitta. However, it may be that in different geographical regions, C. vitta exploits other arthropods as second intermediate hosts, particularly in habitats where woodlice may be absent or rare. In N. Ireland, where the prevalence of C. vitta varied between deciduous and coniferous woodland sites, isopods were never recorded in the diet of wood mice from these habitats, based on their stomach contents (Montgomery Reference Montgomery1982; Montgomery and Montgomery Reference Montgomery and Montgomery1990a). However, increased consumption of arthropods in their food is consistent with C. vitta being more common among the mice from coniferous woodland, which fed more on animal food, compared with those from a deciduous site where seeds dominated the diet (Montgomery and Montgomery Reference Montgomery and Montgomery1988, Reference Montgomery and Montgomery1990a)
Mammalian definitive hosts of C. vitta
In Europe, a common host of C. vitta is the wood mouse, A. sylvaticus, but it has also been recorded from the yellow-necked mouse (A. flavicollis; Debenedetti et al. (Reference Debenedetti, Sainz-Elipe, Sáez-Durán, Galicia, Imaz, Galán-Puchades and Fuentes2016), Ural mouse (A. uralensis; Kirillova et al. Reference Kirillova, Kirillov, Ruchin and Alpeev2024; Makarikov et al. Reference Makarikov, Stakheev and Orlov2017) and Black Sea field mouse (A. ponticus), Makarikov et al., Reference Makarikov, Stakheev and Orlov2017; (Table 1).
Bank voles (Clethrionomys glareolus) are susceptible to infection with C. vitta, but generally, prevalence and abundance are low in this species (Table 2), and in some sites, C. vitta has been recorded as absent from bank voles, while prevalent in sympatric wood mouse populations (Clark and Fairley, Reference Clark and Fairley1971; Tables 1 vs 4). Lewis (Reference Lewis1968b) recovered low-intensity infections from bank voles living in rough grassland sites and in the woodlands near Aberystwyth, Wales, but not on Skomer Island (Table 1). O’Sullivan et al. (Reference O’Sullivan, Smal and Fairley1984) recorded infections in bank voles in Ireland but concluded that C. vitta occurred only rarely in bank voles, which concurs with Canning et al. (Reference Canning, Cox, Croll and Lyons1973), working in Devon, SW England, who recorded only 1 infected bank vole in their sample of 57 animals. Milazzo et al. (Reference Milazzo, Casanova, Aloise, Ribas and Cagnin2003a) recovered C. vitta from just 3 bank voles from their sample of 193 wild rodents from S Italy (Table 2), but prevalence and abundance in wood mice were also very low in Calabria (Tables 1 and 2), indicating that this region was not a good transmission zone for C. vitta. There are also records of C. vitta from Microtus agrestis in England (Lewis and Twigg Reference Lewis and Twigg1972; Canning et al. Reference Canning, Cox, Croll and Lyons1973), but other studies of the helminth fauna of Microtus spp. have not recorded C. vitta as a parasite of voles of this genus (e.g. see Jackson et al. Reference Jackson, Hall, Friberg, Ralli, Lowe, Zawadzka, Turner, Stewart, Birtles, Paterson, Bradley and Begon2014 for M. agrestis in the UK).
Selected studies of the occurrence of Corrigia vitta in bank and grey-sided vole populations

a Clethrionomys rufocanus, grey-sided vole.
Ng = not given in the paper.
Mus species may also harbour C. vitta, but reports are very few. In the Navarra region of N Spain, Sainz-Elipe et al. (Reference Sainz-Elipe, Galicia, Imaz, Galan-Puchades and Fuentes2004) recorded C. vitta in Mus spretus, the Mediterranean mouse, although both prevalence and abundance were very low (1% and 0·11, respectively). We found 1 report of C. vitta from house mice and black rats on Corsica (Milazzo et al. Reference Milazzo, de Bellocq, Cagnin, Casanova, de Bella, Feliu, Fons, Morand and Santalla2003b, citing unpublished theses [Esteban, 1983 and Piqueras 1992] from the University of Valencia). However, C. vitta has not been reported from R. rattus, R. norvegicus and M. musculus in other studies of these commensal rodents from neighbouring regions of Europe and further afield (Tattersall et al. Reference Tattersall, Nowell and Smith1994; Alfonso-Roque Reference Alfonso-Roque1995; Casanova et al. Reference Casanova, Miquel, Fons, Molina, Feliu, Mathias, Torres, Libois, Santos-Reis, Collares-Pereira and Marchand1996; Kataranovski et al. Reference Kataranovski, Vukićević-Radić, Kataranovski, Radović and Mirkov2008; Feliu et al. Reference Feliu, López, Gómez, Torres, Sánchez, Miquel, Abreu-Acosta, Segovia, Martín-Alonso, Montoliu, Villa, Fernández-Álvarez, Bakhoum, Valladares, Orós and Foronda2012; Stojcevic et al. Reference Stojcevic, Mihaljevic and Marinculic2004; Juhász et al. Reference Juhász, Tóth, Eldridge and Majoros2024; see also Table 4).
Relatively high prevalence rates of C. vitta have been recorded in garden dormice (Eliomys quercinus) on the islands of Majorca, Menorca and Formentera, in the Balearic Archipelago in the Mediterranean (Table 3; Esteban et al. Reference Esteban, Bargues, Valero, Mas-Coma, Sans-Coma, Mas-Coma and Gosálbez1987). In a subsequent survey of 502 small mammals on Formentera, the garden dormouse was the only host species infected with C. vitta (Mas-Coma et al. Reference Mas-Coma, Esteban, Fuentes, Bargues, Valero and Galan-Puchades1998). Algerian hedgehogs (Atelerix algirus), wood mice, house mice and black rats were also examined, but none of these carried C. vitta (Tables 3 and 4), perhaps suggesting that this may have been a different species of Corrigia, or a dormouse–infective genetic variant.
Records of C. vitta in wood mice, bank voles and other hosts or in which few additional details were given

Selected studies of helminths in rodent populations, with no record of C. vitta

Ns not stated.
Abbreviations used.
Aa (Apodemus agrarius), Af (Apodemus flavicollis), As (Apodemus syvaticus), At (Arvicola terrestris), Au (Apodemus uralensis), Cg (Clethrionomys glareolus), Cm (Cricetulus migratorius), Cruf (Clethrionomys rufocanus), Crut (Clethrionomys rutilus), Eq (Eliomys quercinus), Mag (Microtus agrestis), Mar (Microtus arvalis), Mm (Mus musculus), Mmin (Micromys minutus), Mn (Microtus nivalis), Moec (M. oeconomus), Mr (Microtus raticeps), Ms (Microtus subterraneus), Rn (Rattus norvegicus), Rr (Rattus rattus).
Note that Microtus raticeps is considered to be a synonym of M. oeconomus, and both are currently known as Alexandromys oeconomus.
Microtus subterraneus = Pitymus subterraneus.
There are also reports of C. vitta from water voles (Arvicola amphibious) (Gibson et al. Reference Gibson, Bray and Harris2005; McCarthy et al. Reference McCarthy, Litten-Brown and Collins2023) in the British Isles and Spain, as well as from muskrats (Ondatra zibethica) from the USSR (Serkowa Reference Serkowa1948; Gibson et al. Reference Gibson, Bray and Harris2005), although more recent studies in Lithuania and Germany failed to find the species in muskrats (Mažeika et al. Reference Mažeika, Kontenyte and Paulauskas2009; Schuster et al. Reference Schuster, Specht and Rieger2021).
The preferred site of C. vitta in hosts and pathology
As in other Dicrocoeliidae, C. vitta is a parasite that is adapted for life in the hepatopancreatic system of its mammalian host. Adult worms live in the interlobary ducts of the pancreas of wood mice and bank voles, as described for the first time by Baer (Reference Baer1932; see also Harvey and Channon, Reference Harvey and Channon1956). Usually, they can be seen easily through the duct walls when the pancreas is spread out during dissection of the alimentary system, as dark lines running along the length of the ducts, reflecting the dark colour of the eggs in the uteri of the worms (Behnke, pers. obs.). Occasionally, worms may be detected in the duodenum or small intestine, perhaps individuals that had been displaced by intense competition in the narrow interlobary ducts or by host resistance, or through senility.
Harvey and Channon (Reference Harvey and Channon1956) and Schmidt (Reference Schmidt1969) observed no obvious pathology of the interlobary ducts, other than distention and some thickening of the duct walls when more than 1 worm lay alongside each other (see also Figure 1B). However, both may have been dealing with low-intensity infections (maximum burden was 8 worms). Johnston et al. (Reference Johnston, Cahir, Halton, Shaw, Fairweather and Buchanan1990), on the other hand, observed severe consequences of infection with C. vitta, including gross dilation of pancreatic interlobary ducts, epithelial cell hyperplasia, fibrous tissue, lymphatic infiltration and signs of inflammation. They also observed disorganization of the exocrine acini (clusters of cells, forming the functional units that secrete enzymes) in the pancreas, loss of zymogen granules and atrophy, and concluded that C. vitta induced partial chronic pancreatic ductal occlusion in wood mice.
There is also strong evidence of an association between C. vitta infection and the richness of intestinal microbiota. Marsh et al. (Reference Marsh, Raulo, Webster and Knowles2024) found that microbial species diversity in the ileum and caecum of wood mice was positively associated with the abundance of C. vitta. It was speculated that in heavy infections of C. vitta, the flow of anti-microbial pancreatic fluids into the gastrointestinal tract may be disrupted, allowing a greater diversity of microbiota to develop (Marsh et al. Reference Marsh, Raulo, Webster and Knowles2024).
Epidemiology of C. vitta
Definition of terms
Following Margolis et al. (Reference Margolis, Esch, Holmes, Kuris and Schad1982) and Bush et al. (Reference Bush, Lafferty, Lotz and Shostak1997), we refer below to the mean intensity of worm burdens as the mean worm burden of mice that were infected with C. vitta, and to abundance as the mean worm burden of all the animals in the subset referred to, including uninfected animals. Where authors did not provide data on mean abundance (e.g. Lewis Reference Lewis1968a) but gave the intensity and the number of infected animals, we calculated abundance as ([mean intensity × no of infected mice]/all mice in the data subset). If mean intensity was not provided, we calculated mean intensity as ([mean abundance × total rodents sampled]/no of infected mice in the data subset). Prevalence is given as the percentage of animals harbouring at least 1 worm or shedding at least 1 egg/gram of faeces.
Methodology of worm removal
It is very difficult to extract whole worms from the interlobary ducts, as they are very fragile and easily tear when grasped with forceps in an attempt to extract them from a duct. Great care and delicate, patient work are required. The difficulties of extracting whole worms were experienced by the early workers on this species (see Baer Reference Baer1932, who initially located the remainders in the interlobary ducts of the pancreas). Prior to Baer (Reference Baer1932), the pancreas was probably mostly neglected. Elton et al.’s (Reference Elton, Ford, Baker and Gardiner1931) study reported an extremely low prevalence of worms found only in the upper part of the small intestine, probably because the pancreas was not examined. Our own data reveal that C. vitta can have a high prevalence in wood mice from woods from the same region of Oxfordshire (JMB personal observations; and see Marsh et al. Reference Marsh, Raulo, Webster and Knowles2024).
Extrinsic factors
Geographical distribution of C. vitta
Corrigia vitta appears to have a patchy distribution across the British Isles (Montgomery and Montgomery Reference Montgomery and Montgomery1990b) with most records concentrated in Southern England (Baylis Reference Baylis1928; Elton et al. Reference Elton, Ford, Baker and Gardiner1931; Harvey and Channon Reference Harvey and Channon1956; Sharpe Reference Sharpe1964 [but see by Murúa Reference Murúa1978]; Lewis and Twigg Reference Lewis and Twigg1972; Behnke et al. Reference Behnke, Lewis, Mohd Zain and Gilbert1999; Abu-Madi et al. Reference Abu-Madi, Behnke, Lewis and Gilbert2000). In more northerly areas of England our own local surveys in Nottinghamshire have never revealed C. vitta here in wood mice or bank voles (Jackson et al. Reference Jackson, Friberg, Bolch, Lowe, Ralli, Harris, Behnke and Bradley2009) and studies in Yorkshire and Merseyside have also failed to report the species (Table 4).
However, C. vitta has been well documented in Wales, in Ceredigion (Lewis Reference Lewis1968a, Reference Lewis1968b) and in N. Ireland (Montgomery and Montgomery Reference Montgomery and Montgomery1988) as well as in the Republic of Ireland (Langley and Fairley Reference Langley and Fairley1982; O’Sullivan et al. Reference O’Sullivan, Smal and Fairley1984; Loxton et al. Reference Loxton, Lawton, Stafford and Holland2017; Stuart et al. Reference Stuart, Paredis, Henttonen, Lawton, Ochoa Torres and Holland2020). There is only 1 record of C. vitta from Scotland, and that was on Ulva in the Inner Hebrides (Thomas Reference Thomas1953).
On the European mainland, there are records from Denmark (Tenora et al. Reference Tenora, Andreassen, Hindsbo and Lodal1991), Germany (Klimpel et al. Reference Klimpel, Förster and Schmahl2007a), Finland (Haukisalmi et al. Reference Haukisalmi, Henttonen and Tenora1987), Spain (Debenedetti et al. Reference Debenedetti, Sainz-Elipe, Sáez-Durán, Galicia, Imaz, Galán-Puchades and Fuentes2015, Reference Debenedetti, Sainz-Elipe, Sáez-Durán, Galicia, Imaz, Galán-Puchades and Fuentes2016), Italy (Milazzo et al. Reference Milazzo, Casanova, Aloise, Ribas and Cagnin2003a, Reference Milazzo, Aloise, Cagnin, Di Bella, Geraci, Feliu and Casanova2005, Reference Milazzo, Di Bella, Casanova, Ribas and Cagnin2010), Georgia (Arabul et al. Reference Arabul, Murvanidze, Faltynkova and Mumladze2024) and Russia (Kirillova et al. Reference Kirillova, Kirillov, Ruchin and Alpeev2024). Equally fascinating are studies that have failed to find C. vitta, some of which were located in regions where others have found the parasite (Compare records in Tables 1–3 with those in Table 4).
It has been suggested that this patchy distribution is associated with the geographical distribution of molluscan intermediate hosts (Schmidt Reference Schmidt1969; Mas-Coma and Montoliu Reference Mas-Coma and Montoliu1978). Certainly, the distribution in the UK of C. bidentata, the potential snail host proposed by Schmidt (Reference Schmidt1967, Reference Schmidt1969), is not uniform, preferring woodland habitats and calcareous environments. Although common in the southeast of England, its occurrence becomes more restricted towards the north of the country, particularly in Scotland (Kerney Reference Kerney1999).
Site-dependent variation in infections
Some authors have studied infections in rodents sampled from different sites within a local region. O’Sullivan et al. (Reference O’Sullivan, Smal and Fairley1984) utilized 20 trap sites, clustered in 5 ecologically distinct habitat types and found that there was a significant difference in the prevalence of C. vitta between these in wood mice. In woodlands with light and heavy ground cover prevalence was 55% and 31%, respectively, and in sites with dense grass, bracken and brambles 26%, while no infected mice were recovered from sites with rhododendron dominating, and without ground cover.
Perhaps the largest study in this field was by Montgomery and Montgomery (Reference Montgomery and Montgomery1990b), who reported on helminth burdens from wood mice trapped at 2 sites where long-term monitoring was implemented and also at 15 additional sites across N. Ireland. Corrigia vitta showed a patchy occurrence, with high prevalence and abundance in some sites, but was totally absent from others. Abu-Madi et al. (Reference Abu-Madi, Behnke, Lewis and Gilbert2000) found that 28·1% of wood mice from the Isle of Wight carried C. vitta, but only 2·3% at the Egham site and none were infected at Dungeness. Abundance was also significantly higher in mice from the Isle of Wight (2·1 worms/host) compared to the other 2 sites (0·2 and 0 at Egham and Dungeness, respectively.
In an interesting variant on this theme, Loxton et al. (Reference Loxton, Lawton, Stafford and Holland2017) studied infections in wood mice in regions of Ireland that were free of bank voles and those that had been invaded by bank voles and found that the abundance of C. vitta was significantly higher in the absence of bank voles. While the prevalence was overall 20% in 3 sites, the 1 exception was Coole Nature Reserve in Galway, which is located in a bank vole invaded region where no worms all of this species were recorded. However, in the other bank vole invaded site, Merlin Park, C. vitta was prevalent and with an abundance similar to those in the 2 bank vole non-invaded sites. Stuart et al. (Reference Stuart, Paredis, Henttonen, Lawton, Ochoa Torres and Holland2020) found a highly significant site effect for C. vitta, for wood mice sampled from 9 sites across Ireland, representing the original introduction core sites for bank voles (3 sites), the invasion front (3 sites) and sites that were still free of bank voles (3 sites). Interestingly, the parasite was totally absent from wood mice sampled at locations in Limerick, in the core sites close to Foynes Port, which is believed to be where bank voles were introduced into Ireland from Germany in the 1920s (Stuart et al. Reference Stuart, Mirimin, Cross, Sleeman, Buckley, Telfer, Birtles, Kotlík and Searle2007). Abundance in wood mice was significantly higher at the expansion front than at the uninvaded sites.
Several explanations can be offered to explain site effects. For the life cycle of C. vitta to be completed and to persist, it is likely that at least 4 taxonomically unrelated organisms must be available locally, and their distributions overlap (the parasite, a molluscan first intermediate host, an arthropod second intermediate host and a mammalian definitive host). In the case of a total absence of a parasite species from the definitive hosts in a site, therefore, the most obvious explanation is that at least one of these taxa is missing locally. The parasite may not have been introduced into the site when colonized by rodents, and/or susceptible intermediate hosts of C. vitta have not colonized the site. It may be that in some sites the intermediate molluscan and arthropod intermediate hosts are present, but in such low densities (because the environment is not entirely suitable for their optimal development and survival) that transmission cannot be maintained consistently. In addition to the total absence of C. vitta from the Dungeness site (Abu-Madi et al. Reference Abu-Madi, Behnke, Lewis and Gilbert2000), infections with Heligmosomoides polygyrus also showed very low prevalence and abundance (Abu-Madi et al. Reference Abu-Madi, Behnke, Lewis and Gilbert1998). It was suggested that acidification of the soil surface at Dungeness (Ferry and Waters Reference Ferry and Waters1988) may have been detrimental to the survival of H. polygyrus. High soil acidity may also have been detrimental to the survival of the intermediate hosts of C. vitta, although Abu-Madi et al. (Reference Abu-Madi, Behnke, Lewis and Gilbert1998, Reference Abu-Madi, Behnke, Lewis and Gilbert2000) did not monitor the diversity or densities of molluscan or arthropod species in the site (but see Morris and Parsons Reference Morris and Parsons1991). Another possibility in the case of wood mice in Ireland is that the absence of C. vitta from the core site reflects a dilution effect arising from the introduction and subsequent local proliferation of bank voles (Stuart et al. Reference Stuart, Paredis, Henttonen, Lawton, Ochoa Torres and Holland2020), although neither Stuart et al. (Reference Stuart, Paredis, Henttonen, Lawton, Ochoa Torres and Holland2020) nor Loxton et al. (Reference Loxton, Lawton, Stafford and Holland2016) found the parasite in bank voles in Ireland. However, like wood mice, the bank voles would have fed on wood lice, but the introduced genotypes may have been more resistant to infection with C. vitta than those in other locations where bank voles were found to harbour mature worms (Table 2).
Annual variation in infections
Few studies have monitored C. vitta infections in rodent populations for more than a year at a time. The earliest study lasting for more than a single year was by Elton et al. (Reference Elton, Ford, Baker and Gardiner1931), who trapped rodents monthly for almost 4 years in Oxfordshire (from September 1925 until April 1928). However, the prevalence of C. vitta in this population was too low to allow a meaningful temporal analysis. Although Montgomery and Montgomery (Reference Montgomery and Montgomery1988) sampled wood mice monthly from the same sites for almost 3 years (33 months), their analysis of data focused on seasonal changes (see below). Behnke et al. (Reference Behnke, Lewis, Mohd Zain and Gilbert1999) reported on sampling for 4 years, but only in September of each year, and found that the prevalence of C. vitta differed markedly between years. In their study, 1996 was a year with the lowest prevalence and 1997 with the highest, but there was no long-term trend, and it was suggested that the between-year variation in prevalence probably reflected differences in the availability of the arthropod intermediate hosts, possibly influenced by climatic differences between years and availability of predators. As for prevalence, mean abundance also varied significantly between years, from a low in 1994 to a peak in 1997. Climatic conditions can also influence the availability of parasitic infections within molluscan hosts (Morley and Lewis Reference Morley and Lewis2008), which may impact annual occurrence in mammalian hosts.
Seasonal variation in infections
In W. Ireland in Galway prevalence of infection with C. vitta increased steadily from a low in August (≈30%) to a peak in March (≈80%), followed by a dip in April–May, and an even higher peak in June with almost 95% of mice carrying the parasite (Langley and Fairley Reference Langley and Fairley1982). Mean worm burdens were steady throughout most of the year, averaging about 8–10/mouse, but peaked in June at ≈ 25 worms/mouse. The fall in prevalence after June was attributed to an influx of uninfected juveniles following the breeding period. Prevalence rose subsequently as gradually more mice became infected, but the lack of a rise in mean worm burdens was attributed to a lack of heavily infected animals at this time of the year compared to June, when 35% of mice had more than 30 adult worms each.
Marked seasonal cycles, indicating cyclic occurrence of C. vitta, were recorded by Montgomery and Montgomery (Reference Montgomery and Montgomery1988) among wood mice trapped in Tollymore and Clandeboye in N. Ireland. Peak worm burdens were recorded in the winter period in Tollymore (February–March) in each of the 3 years, but only in February 1980 at Clandeboye. Although no statistical models were provided for C. vitta from bank voles in Catalonia, NE Spain, tabulated data show higher values for prevalence in summer months (7·9%, compared with 3·6% in autumn, 3·5% in winter and 1·7% in spring [Ribas et al. Reference Ribas, Torre, Feliu, Arrizabalaga and Casanova2009]). The mean intensity of infection was highest in winter (7·5 worms/infected host).
Three studies, involving monthly or seasonal sampling of wood mouse populations, failed to detect any major or significant seasonal variation in infections with C. vitta. In County Kerry, in Ireland, C. vitta was present in wood mice throughout the year, with prevalence varying from about 10% in April-May to over 40% in September (O’Sullivan et al. Reference O’Sullivan, Smal and Fairley1984), but not showing any marked season-dependent peak. Abu-Madi et al. (Reference Abu-Madi, Behnke, Lewis and Gilbert2000) also failed to find a significant difference in the prevalence or abundance between seasons, but both prevalence and abundance in these surveys were very low, with a total absence of C. vitta from one of the sites (Dungeness). More recently, Stuart et al. (Reference Stuart, Paredis, Henttonen, Lawton, Ochoa Torres and Holland2020) sampled at 9 sites located across Ireland and also did not report seasonal differences of infection parameters as significant, nor any statistical interaction between season and the other factors included in their statistical models.
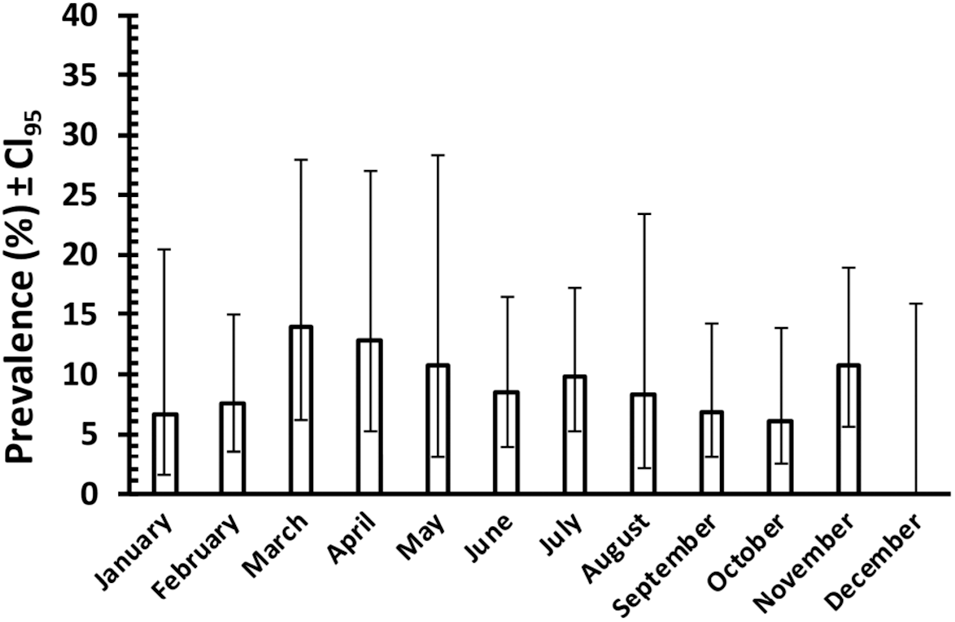
Figure 4 shows the prevalence of C. vitta, by month, based on faecal egg counts recorded from 561 samplings of adult wood mice in Surrey in the period from August 1975 until February 1978 (J. W. Lewis unpublished data). Although these data cannot be analysed statistically because the mice were not individually tagged and therefore probably include repeated measures, the difference in prevalence between months varied only from 0 % in December to 13·9% in March, and given the wide 95% confidence limits, this suggests that seasonal variation in prevalence as assessed by monthly values was minimal in this population.
Prevalence of C. vitta in adult wood mice in Surrey in the period from August 1975 until February 1978 (inclusive, and both sexes combined). Prevalence is based on faecal C. vitta egg counts of mice trapped in the months shown, and released after inspection. The mice were not marked individually; therefore, the data probably include repeated assessments of certain individuals. The number of animals sampled in each month from January to December was as follows: 45, 53, 36, 39, 28, 59, 51, 48, 59, 66; 56, 21. (Unpublished data of J. W. Lewis).

Where dips in prevalence and abundance were found in late summer months (Langley and Fairley Reference Langley and Fairley1982; Montgomery and Montgomery Reference Montgomery and Montgomery1988), this is most likely attributable to an influx of juvenile, uninfected mice into the population. The longevity of C. vitta in its definitive hosts is not known, but the marked age-dependent increase in infection parameters (see below) suggests that the species is long-lived. In Langley and Fairley’s study, transmission probably occurred throughout the year, but this cannot have been the case in Montgomery and Montgomery’s study, in which worm burdens declined to zero in between the peaks they recorded. Despite the wood mouse population size fluctuating with a drop in late spring months, the number of sampled animals throughout this period was still sufficient to allow a realistic estimate of prevalence and abundance. This may indicate that the longevity of C. vitta is limited and that after acquisition in late summer, parasite burdens begin to decline in spring, in situations where continued exposure to infection is interrupted. Either way, it is clearly of relevance and reflects the longevity of C. vitta (see below).
Unusual, rare weather conditions can also affect the prevalence of C. vitta in relevant years. A prolonged period of hot, dry weather in the British Isles in the summer months of 1976 resulted in drought, particularly in the south of England, and was followed by an atypically wet period. Morley and Lewis (Reference Morley and Lewis2014) found that while wood mice in S. England were free of infections with C. vitta in the summer period during the drought, there was a surge of infections in both juvenile and adult wood mice in the autumn following the post-drought heavy rainfall. This was attributed to increased availability of infected molluscan and arthropod intermediate hosts in the recovering ecosystem, benefitting from the prolonged post-drought heavy rainfall.
Intrinsic factors
Age-dependent variation in infections
Helminths are generally regarded as long-lived parasites in their hosts because of their strategies for evading host responses (Behnke Reference Behnke1987; Maizels et al. Reference Maizels, Balic, Gomez-Escobar, Nair, Taylor and Allen2004, Reference Maizels, Hewitson, Murray, Harcus, Dayer, Filbey, Grainger, McSorley, Reynolds and Smith2012, Reference Maizels, Smits and McSorley2018). Not surprisingly, therefore, helminth species richness tends to increase with host age (Montgomery and Montgomery Reference SSJ and Montgomery1989) as does the abundance of long-lived species (Behnke et al. Reference Behnke, Lewis, Mohd Zain and Gilbert1999), because hosts accumulate worms that they cannot eliminate throughout their lives.
Although the exact longevity of C. vitta is not known, if it were a long-lived species, we would expect both prevalence and abundance to increase with host age. Surveys of wood mice in which there was some focus on age have confirmed this prediction. The first study to quantify differences in the prevalence of C. vitta in juvenile and adult wood mice was that of Lewis (Reference Lewis1968a) in Wales. Prevalence of infection in adult wood mice was 34·6% in the rough grassland site and 53·1% in woodlands, whilst in juvenile mice the values were 2·5% and 29%, respectively. Langley and Fairley (Reference Langley and Fairley1982), working in Ireland, also noted a significant difference in prevalence between juvenile (28%) mice and adults (79%), when the borderline between age classes was based on mouse weight (<17·0 g and >16·5 g). Montgomery (Reference Montgomery1982) found that both intensity and prevalence were positively associated with mean eye lens weight (a correlate of age) among wood mice from Tollymore, where both infection parameters were high. Behnke et al. (Reference Behnke, Lewis, Mohd Zain and Gilbert1999) found that both prevalence and abundance differed between age classes. Mice in the youngest age class were not infected, but those in the intermediate and oldest age classes showed similar prevalence of C. vitta of about 20%. Despite the overall low prevalence of C. vitta in the study by Abu-Madi et al. (Reference Abu-Madi, Behnke, Lewis and Gilbert2000), 18 of the 20 mice with C. vitta were adult mice. An age effect was also recorded in wood mice in S. Italy by Milazzo et al. (Reference Milazzo, Aloise, Cagnin, Di Bella, Geraci, Feliu and Casanova2005), with prevalence in adult wood mice being 5 times that in juveniles (2·1 vs 0·4%, respectively), although no statistical support was provided. Loxton et al. (Reference Loxton, Lawton, Stafford and Holland2017) found a highly significant difference in abundance between 3 age classes of wood mice in Ireland, with abundance lowest in the youngest age cohort and highest in the oldest, and this relationship was evident among wood mice trapped in bank vole-invaded sites and those without bank voles, in both years of the study.
The data in Table 5 also show a marked and significant difference in prevalence between juvenile and adult wood mice, and this was consistent in both sexes (Unpublished data of J. W. Lewis). However, these data were based on faecal egg counts, and the low values, particularly in juvenile mice, may be partially attributable to the presence of immature worms, which would not have been detected by faecal egg counts.
Prevalence of Corrigia vitta in wood mice from Surrey in the period from August 1975 until February 1978, inclusive, based on faecal egg counts (Unpublished data of J. W. Lewis)

a Not significantly different: χ 21 = 0·13, P = 0·7.
b Significantly different: χ 21 = 7·87, P = 0·005.
Taken together, these studies concur and show that an age effect on the prevalence and abundance of C. vitta in wood mice is a highly predictable phenomenon. With increasing host age, worms accumulate in wood mice, with the consequence that the values of both parameters are reliably higher in older mice compared to those derived from juveniles. Surveys across the geographic range in which C. vitta infections have been observed show that this is a universal aspect of the epidemiology of this parasite in wood mice.
Host sex-dependent variation in infections
Many different studies have shown that in mammals, prevalence and abundance of infection with helminths may differ between the sexes (Alexander and Stimson Reference Alexander and Stimson1988; Zuk and McKean Reference Zuk and McKean1996). In most cases, males are reported to be more likely to be infected and to harbour more and larger parasites than females (Poulin Reference Poulin1996a, Reference Poulin1996b), and some studies have attributed male-bias in parasite burdens to host body mass which in sexually dimorphic mammalian species, is often greater in males compared to females (Moore and Wilson Reference Moore and Wilson2002; Harrison et al. Reference Harrison, Scantlebury and Montgomery2010; Zduniak et al. Reference Zduniak, Serafini, Wróbel and Zwolak2023). Others have attributed male bias to host behaviour, hormonal differences between the sexes influencing immunocompetence and a range of other factors (Schalk and Forbes Reference Schalk and Forbes1997; Ferrari et al. Reference Ferrari, Cattadori, Nespereira, Rizzoli and Hudson2004; Skorping and Jensen Reference Skorping and Jensen2004; Duneau and Ebert Reference Duneau and Ebert2012). However, there are also studies showing that in certain situations the bias may be in favour of females harbouring more intense infections (Sanchez et al. Reference Sanchez, Devevey and Gize2011; Grzybek et al. Reference Grzybek, Bajer, Behnke-Borowczyk, Alsarraf and Behnke2015b; Bourgoin et al. Reference Bourgoin, Portanier, Poirel, Itty, Duhayer, Benabed, Cockenpot, Callait-Cardinal and Garel2021).
Lewis (Reference Lewis1968a) wrote that there was very little difference in the prevalence of C. vitta between the sexes. In his rough grassland site, 16% of males and 12·3% of females harboured C. vitta, and in the woodland site, 43·4% of males and 20·9% of females were infected. Intensity of infection for male and female mice was 14·1 and 9·0/infected mouse, respectively, in the rough grassland site and 8·1 and 9·6/infected mouse, respectively, in the woodlands. Although Lewis (Reference Lewis1968a) did not employ statistical analysis to test these data, numerically, 3 of the 4 values indicate a bias in favour of male mice, and overall, he concluded that male mice were more heavily infected with C. vitta. A male bias was also recorded by Loxton et al. (Reference Loxton, Lawton, Stafford and Holland2016, 17·2% prevalence in males and 8·5% in females), but without statistical support. However, intensity was in the opposite direction (7·81 worms/infected male and 14·8 for females).
Three studies failed to find a sex bias in infections with C. vitta. No difference between sexes in C. vitta infections was found by O’Sullivan et al. (Reference O’Sullivan, Smal and Fairley1984, 33% prevalence in males and 30% in females), Behnke et al. (Reference Behnke, Lewis, Mohd Zain and Gilbert1999, 19·8% prevalence in male and 14·0% in female mice, and for abundance 1·0 and 1·3, respectively), nor by Abu-Madi et al. (Reference Abu-Madi, Behnke, Lewis and Gilbert2000; 5·7% prevalence in male and 4·8% in female mice, and for abundance 0·37 and 0·33, respectively) in data that were thoroughly analysed by statistical models. The data in Table 5 also show little difference in prevalence between the sexes.
Other studies have found a female bias in prevalence and/or abundance. A sex bias in the prevalence of C. vitta in favour of female mice (67% in females and 58% in males) was noted by Langley and Fairley (Reference Langley and Fairley1982) in Ireland, but was not significant, although the authors explained this bias as female mice requiring more food in the breeding season because of the metabolic demands in pregnancy and lactation. Loxton et al. (Reference Loxton, Lawton, Stafford and Holland2017), who also fitted appropriate statistical models to their data, reported a significant female sex bias, with the abundance of C. vitta higher in female mice in both years, although the difference between sexes was greater in 2012 compared with 2011, when the overall abundance of C. vitta was higher. Milazzo et al. (Reference Milazzo, Casanova, Aloise, Ribas and Cagnin2003a) found C. vitta only in female bank voles (prevalence = 3·7%. The males were not infected.
Studies of helminth infections in wood mice have generally found that sex-bias is context dependent (Lewis et al. Reference Lewis, Morley and Behnke2023), and taken together, those studies considered above in which prevalence and abundance of C. vitta were quantified for each sex, and analysed statistically with other factors taken into account, support this view. Where a difference between the sexes has been detected, it was relatively small and swayed one way or the other depending on how prevalent the parasite was in the population in particular seasons, years and/or sites. This conclusion is consistent with studies in other mammalian hosts subject to parasitism by helminths and arthropod ectoparasites, which have also found significant sex-bias in parasitism to be highly context dependent (Krasnov et al. Reference Krasnov, Morand, Hawlena, Khokhlova and Shenbrot2005, Reference Krasnov, Bordes, Khokhlova and Morand2012; Junker et al. Reference Junker, Boomker, Horak and Krasnov2022).
Conclusions and recommendations
Given that Corrigia vitta has been known about for so long, it is surprising that the details of its life cycle have still not been fully clarified. Although there are some hints as to the likely molluscan first intermediate hosts, there is still no reliable evidence for their identity. Evidence from work on D. dendriticum shows that through dedicated and painstakingly thorough scrutiny of all available molluscs in a vicinity where transmission takes place, it is possible to establish the likely identity of the molluscan host (Manga-González et al. Reference Manga-González, Ginzalez-Lanza, Cabanas and Campo2001). However, even for that species, experimental evidence is required to complete the work. A similar approach could be adopted for C. vitta.
The second intermediate host is also not known with any degree of certainty. As for the molluscan host, therefore, species of arthropods, particularly isopods, living in endemic regions need to be surveyed for the presence of metacercariae (see Panin and Romanenko Reference Panin, Romanenko and Gvozdev1978 for C. corrigia). Although there have been indications that woodlice may carry metacercariae, other invertebrates living in transmission zones also need to be carefully scrutinized. Such studies, if based on traditional techniques such as dissection or tissue digestion, require resilience and patience by skilled workers, since undoubtedly the proportion of infected hosts is likely to be extremely low, given the range of possible invertebrate hosts and their population densities. Moreover, all will undoubtedly also harbour other species of helminths. Nevertheless, from a practical point of view, it will often be quicker and cheaper to examine large numbers of arthropod intermediate hosts for Dicrocoeliid metacercariae using the traditional techniques of squashing between 2 glass slides and examining under a dissecting microscope than applying molecular methods. Metacercarial cysts are fairly easy to spot this way, and it is possible to process a lot of material in a few hours. Molecular techniques might then be applied to confirm the identity of metacercariae in the positive samples. Additionally, and perhaps more ambitiously, modern high-throughput molecular methods and automated sample processing could be exploited. For example, homogenization of the whole (relatively small) candidate intermediate hosts could be followed by nucleic acid extraction and PCR, with 1 or more of these steps automated to allow very large sample sizes to be handled efficiently. A preliminary to this would be more genomic information for C. vitta (such as a whole genome or mitogenome) to allow greater scope for the design of efficient and specific primers.
Experimental completion of the life cycle could be achieved by first selecting naturally infected wood mice with high egg loads in their faeces. The eggs of C. vitta are easily recognizable, and measures of eggs/gm of faeces can be obtained by the McMaster method (M.A.F.F. 1977), if necessary, by replacing saturated salt solution with higher density solutions such as sucrose or 35–40% zinc sulphate. Eggs could then be concentrated by collecting from the surface of the solution and syphoning off the top ml or so into another test tube. By adding water, the density would be reduced, and the eggs can then be concentrated by centrifugation. Locally collected snails could then be fed on appropriate food contaminated with the eggs, and then, a few weeks later, sporocysts should be evident on the dissection of infected snails. Snail species that produce slime balls or extrude sporocysts containing cercariae would then need to be exposed to a range of locally abundant invertebrates, including ants and wood lice, to identify those species that are susceptible to infection by C. vitta. Metacercarial cysts should be evident on dissection a few weeks after exposure.
The longevity of C. vitta in its definitive hosts is still unknown. Although the age-dependent accumulation of adult worms suggests it is a long-lived species, the seasonal collapse in prevalence and abundance reported by Montgomery and Montgomery (Reference Montgomery and Montgomery1988) perhaps indicates that it may be a matter of a few months rather than a year or more. Longevity could be determined by housing naturally infected wood mice under animal house conditions and monitoring faecal egg output. Alternatively, uninfected, lab-raised wood mice or captured wild wood mice treated with a suitable anthelmintic could be infected with metacercariae, but this would depend on the life cycle having been established in the laboratory or infected second intermediate hosts being available in the field for the harvest of metacercariae.
Since Sainz-Elipe et al. (Reference Sainz-Elipe, Galicia, Imaz, Galan-Puchades and Fuentes2004) recovered worms from M. spretus, the Mediterranean mouse, it is possible that other Mus species, including laboratory-maintained M. musculus, may be susceptible to infection with C. vitta. Although we are unaware of any reliable evidence that house mice may be susceptible to C. vitta (Milazzo et al. Reference Milazzo, de Bellocq, Cagnin, Casanova, de Bella, Feliu, Fons, Morand and Santalla2003b; Feliu et al. Reference Feliu, López, Gómez, Torres, Sánchez, Miquel, Abreu-Acosta, Segovia, Martín-Alonso, Montoliu, Villa, Fernández-Álvarez, Bakhoum, Valladares, Orós and Foronda2012), there is a report of Corrigia sp. occurring in both house mice and black rats (R. rattus) on Corsica (Milazzo et al. Reference Milazzo, de Bellocq, Cagnin, Casanova, de Bella, Feliu, Fons, Morand and Santalla2003b). However, C. vitta was not found in R. rattus and M. musculus from the nearby islands of Eivissa (Mas-Coma et al. Reference Mas-Coma, Esteban, Fuentes, Bargues, Valero and Galan-Puchades2000) and Sicily (Milazzo et al. Reference Milazzo, de Bellocq, Cagnin, Casanova, de Bella, Feliu, Fons, Morand and Santalla2003b), nor in other studies of these commensal rodents (Feliu et al. Reference Feliu, López, Gómez, Torres, Sánchez, Miquel, Abreu-Acosta, Segovia, Martín-Alonso, Montoliu, Villa, Fernández-Álvarez, Bakhoum, Valladares, Orós and Foronda2012). If house mice and rats (R. norvegicus) are found to be susceptible to infection with C. vitta, then experimental work on the host/parasite relationship of the adult worms will be possible. Parasites that live in the hepatopancreatic system of their host must be equipped with survival strategies that allow them to persist despite the locally secreted enzymes and products of the immune system, including high concentrations of IgA. Since epidemiological data indicate that worms are likely to be long-lived to some extent, this suggests that the worms employ countermeasures for avoiding the consequences of such host secretions and the effectors of host resistance (immunomodulatory factors; Shepherd et al. Reference Shepherd, Navarro, Wangchuk, Wilson, Daly and Loukas2015; Okakpu and Dillman Reference Okakpu and Dillman2022; Yeshi et al. Reference Yeshi, Ruscher, Loukas and Wangchuk2022). These may offer possibilities for developing novel medicines for the treatment of human diseases associated with the liver and pancreas, e.g. autoimmune diabetes (Hernández-Bello et al. Reference Hernández-Bello, Escobedo, Guzmán, Ibarra-Coronado, López-Griego and Morales-Montor2010; Wang et al. Reference Wang, Wu, Weng, Zheng, Wu and Lv2017).
In summary, C. vitta is undoubtedly a fascinating parasite, but one for which many gaps in knowledge remain to be filled. Here we have reviewed studies that have contributed to the current understanding of its biology, identified questions that warrant research attention and indicated that potentially it may have much to offer for human and veterinary medicine as a study model. We hope thereby to have encouraged others to rise to the research challenges and grasp the research opportunities presented by this enigmatic organism.
Acknowledgements
We thank Andrew McCarthy for advice on sources of information, Sally Montgomery, Ian Montgomery and Celia Holland for checking, and advising on, earlier versions of this manuscript, Kirsty J. Marsh for provisions of data from her study (Marsh et al. Reference Marsh, Raulo, Webster and Knowles2024) and staff at the University of Nottingham library, who helped us to trace papers that were not otherwise easily available for scrutiny. We are grateful to the family of the late Professor John Lewis for permission to use some of his unpublished data.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article. We have also included unpublished data extracted from the laboratory books of Prof John W. Lewis, who passed away in 2022. Following convention, we have obtained the approval of John’s family for the inclusion of these data in this paper.
Authors’ contributions
The paper was written jointly by the authors.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
None.