1.1 Basic Anatomy and Histology of the Prostate
The adult prostate surrounds the urethra and is located posterior to the inferior symphysis pubis, superior to the urogenital diaphragm, and anterior to the rectum. It measures 5 cm x 4 cm x 3 cm and weighs 20 gr through ages 20 to 50 years, then there is an increase to 30 gr from ages of 60 to 80 years. Hyperplasia frequently occurs at later ages with larger prostates >30 gr. Table 1.1
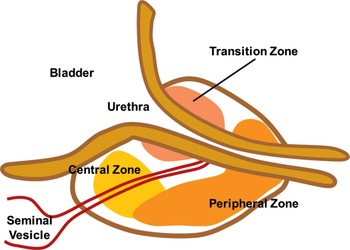
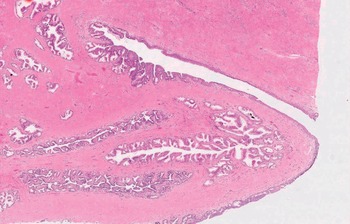
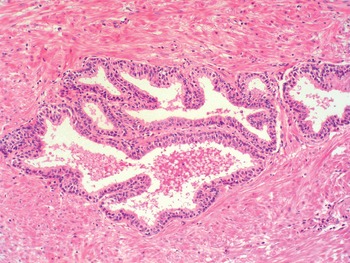
The three-zone model defines the central zone, the transition zone, and the peripheral zone. Fig 1.1
Cancer tends to arise in the peripheral zone (20% do arise in the transition zone), and BPH typically arises in the transition zone. The central zone is more resistant to disease. The prostatic urethra exits the prostate at the apex where it is continuous with the membranous urethra. Transition zone cells typically express estrogen receptors, and peripheral zone cells typically express both androgen and estrogen receptors. Figs 1.2–1.7
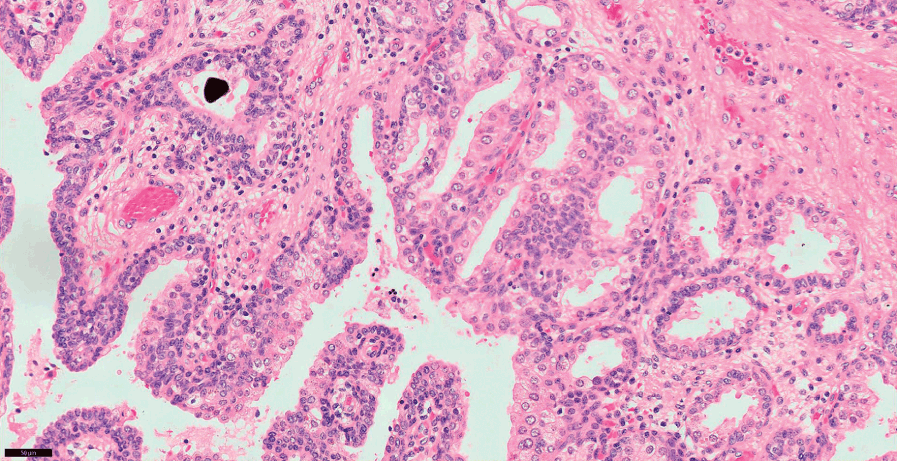
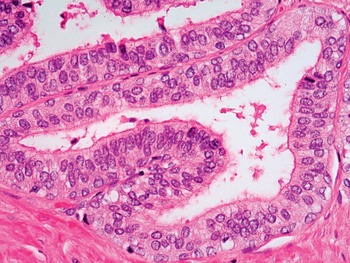
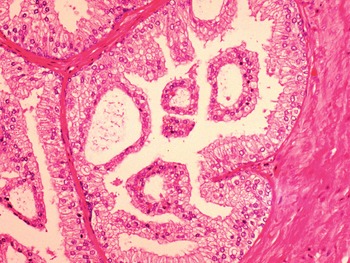
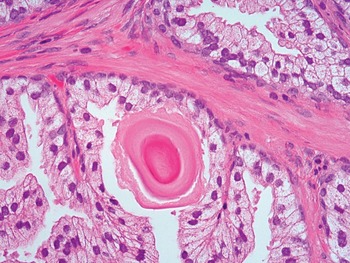
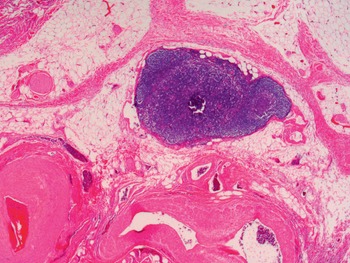
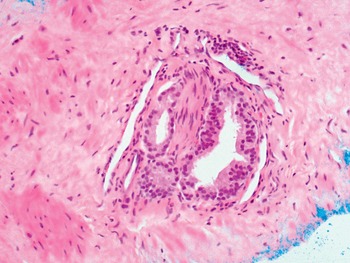
The paired ejaculatory ducts run through the central zone from the seminal vesicles to their exit at the posterior urethral protuberance, known as the verumontanum. Within the verumontanum is the prostatic utricle, located between the ejaculatory ducts. After puberty, the utricle shows a complicated and variable architecture. The epithelium no longer differs from that of the prostate glands, both morphologically and immunohistochemically. Figs 1.8–1.9
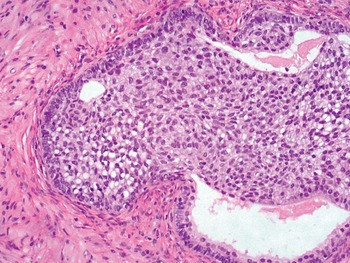
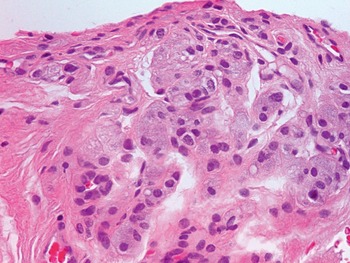
Squamous cell metaplasia and urothelial metaplasia involving the prostatic utricle have been observed occasionally in patients treated with various forms of androgen manipulation therapies for Pca and benign prostatic hyperplasia. Similar morphologic findings are also seen in the periurethral ducts. Fig 1.10
Orifices of the prostatic ducts are located in the area of the verumontanum, separately from the utricle and ejaculatory ducts. The ducts are lined by a bilayered prostatic epithelium histologically identical to that seen in the ducts and acini away from the urethra.
The opening of the ejaculatory ducts is lateral to the prostatic utricle. The epithelial lining includes a basal cell layer and a luminal cell layer. The former are morphologically and immunohistochemically similar to those of the prostate. The luminal cells show acidophilic abundant cytoplasm with a variable amount of yellow pigment, whereas the nuclei show condensed chromatin and size variation.
Occasionally, large epithelial cells with hyperchromatic nuclei can be seen. This epithelium is similar to that of the seminal vesicles. There is no morphological equivalent in the lower female genital tract.
Intraductal and invasive neoplasms originating from the periurethral ducts can grow within the urethral lumen or infiltrate the prostatic utricle. In particular, intraductal carcinoma of the prostate and prostatic ductal adenocarcinomas can be seen in the periurethral prostatic ducts. The former is similar morphologically to the counterpart duct carcinoma in situ in the female breast and the latter shows similarities with the endometrioid carcinoma of the female genital tract.
The anterior fibromuscular stroma is present anteriorly over the prostate and extends from the bladder neck to the apex of the prostate.
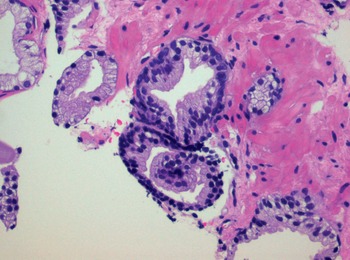
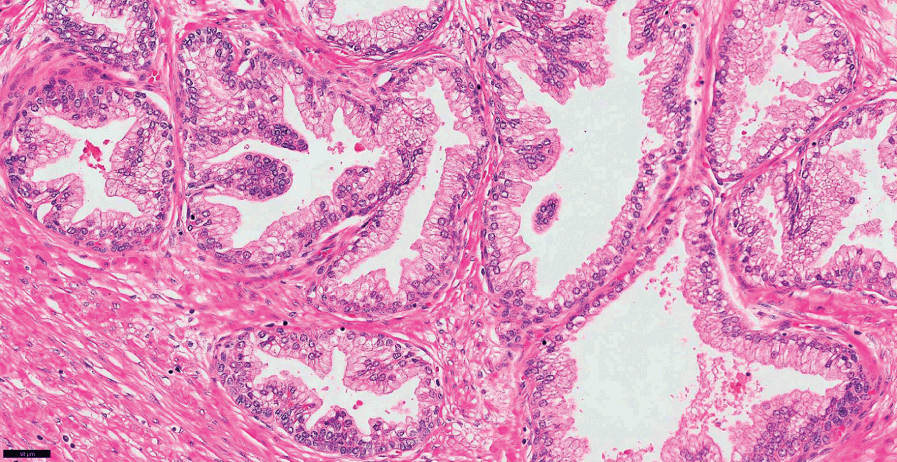
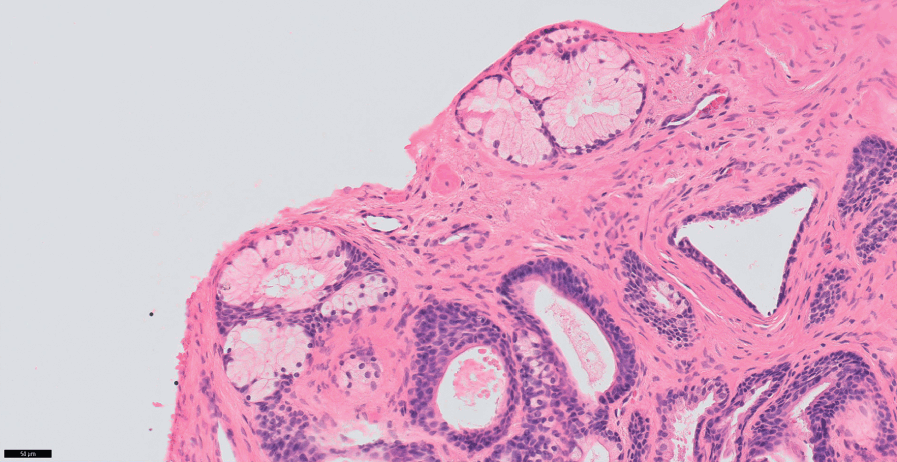
The adult prostate gland is a branching duct-acinar embedded in a fibromuscular stroma. The epithelium has two cell layers: the luminal/secretory and the basal layer with some neuroendocrine cells in the epithelium. The cytoplasm of secretory cells is clear and occasionally may exhibit yellow-brown pigment (lipofuscin). Figs 1.11–1.20
Secretory cells are positive for CKAE1/AE3, CK 8–18, PSA, PAP, PSMA, p501S (prostein) and NKX3-1. P504S (racemase) may show a focal non-circumferential positivity in normal secretory cells.
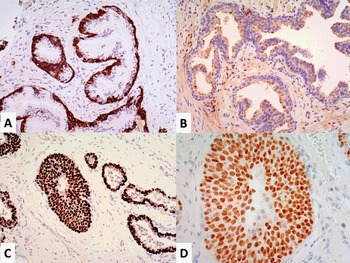
Basal cells have a dense cytoplasm and small hyperchromatic nuclei. They react with p63, p40, CK5/6 and high molecular weight CK clone 34βE12 (CK903). Fig 1.21
Neuroendocrine cells are less frequent and show an immunohistochemical profile more similar to secretory cells with variable androgen receptors and PSA/PAP expression. They express a number of immunohistochemical markers, including chromogranin, synaptophysin, neuron-specific enolase, CD56 and many other active peptides.
Urothelium is also normally present in the prostate, sometimes showing a clear cytoplasm. It is PSA negative and p63, p40, GATA3 positive.
Intraluminal contents of normal prostate glands include degenerated epithelial cells, corpora amylacea and calculi. Rarely, it may contain focal blue-tinged mucin, pink amorphous acellular secretions and crystalloids. Figs 1.22–1.23
The prostatic stroma, including fibroblasts, smooth muscle, vasculature and nerves, and rarely adipocytes (carcinoma in fat should be viewed as extraprostatic extension). There are zonal differences in prostatic fibromuscular stroma density. Figs 1.24–1.27
The prostate does not have a true capsule. The so called outer prostatic “capsule” is a band of concentrically placed fibromuscular and vascular tissue that is inseparable from prostatic stroma and surrounding fascia. This is absent in the apex. Figs 1.4; 1.7
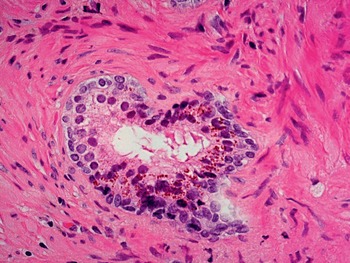
Histologically, nerves are seen in the periprostatic neurovascular bundle, in the outer fibromuscular band, and in the prostate itself. Paraganglia (clear cells that should not be mistaken as carcinoma) are usually adjacent to neurovascular bundles, but may be seen deeper. Posterolateral prostate shows abundant adipose tissue together with neurovascular bundles. Figs 1.7; 1.28–1.29
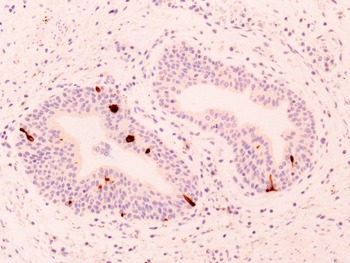
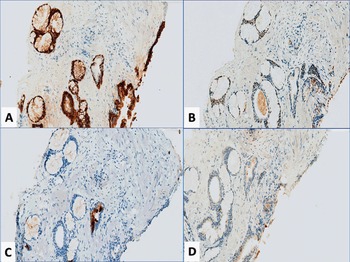
Bulbourethral Cowper glands are extrinsic to the prostate, but eventually may be seen in prostate biopsies. Histologically, they are tubule-alveolar glands with lobules of acini, admixed with excretory ducts and ductuli. Frequently, they are associated with skeletal muscle fibers. The cells are cuboidal to columnar, pale-staining mucinous cytoplasm and small, bland, basally located nuclei. A basal cell layer is not readily apparent with H&E. Immunohistochemistry shows 34βE12+/p63+ basal cells and PSA-/racemase- secretory-type cells. Figs 1.30–1.31
Other rare findings related to the prostate include distorted colorectal epithelium seen as a component of prostate biopsy that can potentially be confused with prostatic adenocarcinoma since they are negative for basal cell markers 34βE12 and p63 and positive with racemase. Mesonephric remnants may also rarely occur in the prostate and become hyperplastic, a finding that should not be misdiagnosed as carcinoma (see Chapter 2). Ectopic prostatic tissue has been reported in a number of extraprostatic locations, including epididymis and testes, urinary bladder, penile urethra, seminal vesicle and many others. In females, ectopic prostate may be seen in the cervix, ovaries, vagina, or urethra. In some cases, it may form polypoid structures typically PSA+. Paraurethral Skene glands are also PSA+.
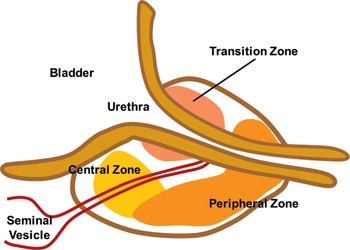
Fig 1.1 The three-zone model defines the central zone, the transition zone, and the peripheral zone.
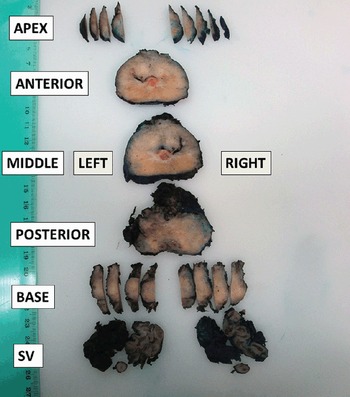
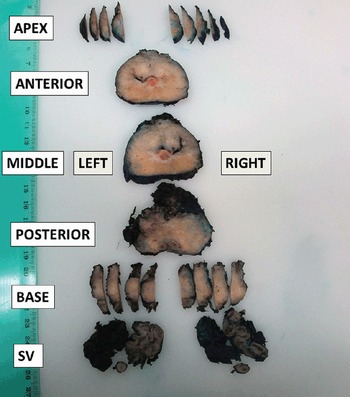
Fig 1.2 Partial sampling of the prostate after radical prostatectomy allows one to obtain information on the presence of cancer at different anatomic regions of the organ and to state laterality.
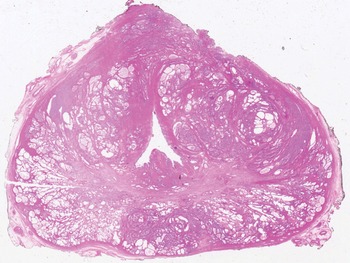
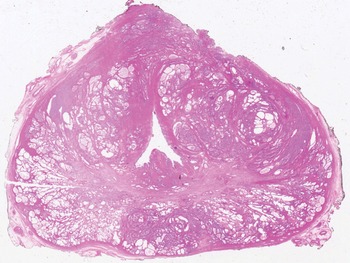
Fig 1.3 This macro-section of the prostate shows the transition zone (periurethral) and the peripheral zone.
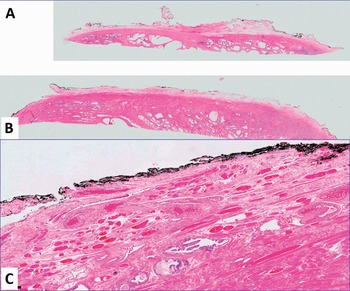
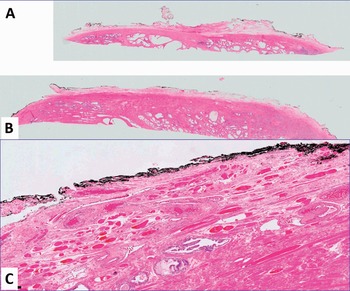
Fig 1.4 Apex (A) and (B), and anterior (C) prostate (100x).
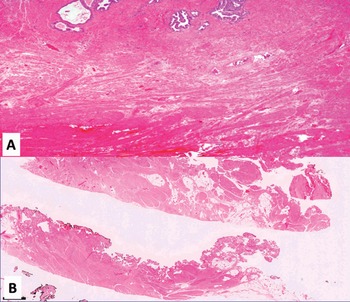
Fig 1.5 Different views (A and B) of base and posterior prostate (100x).

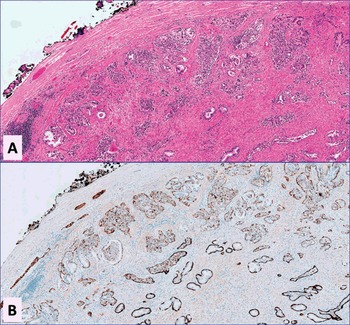
Fig 1.6 Occasionally, one may see prostate glands and acini with extensive apoptotic change on H&E (A) and with anti-cytockeratin 5/6 which highlights basal cells (B) (x100).
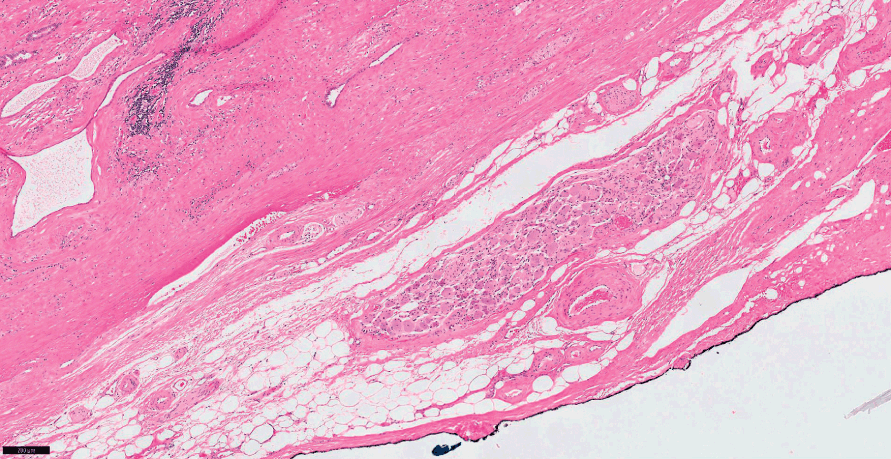
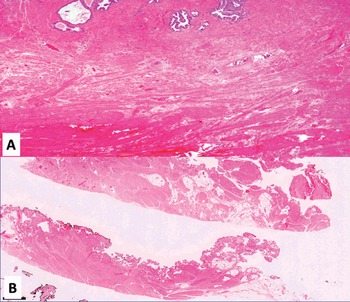
Fig 1.7 Transition between external connective tissue (so called prostate capsule) and periprostatic soft tissues with fat, vessels and nerves. The prostate tissue margin is highlighted by black ink.
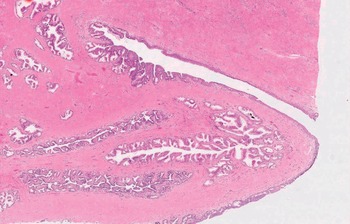
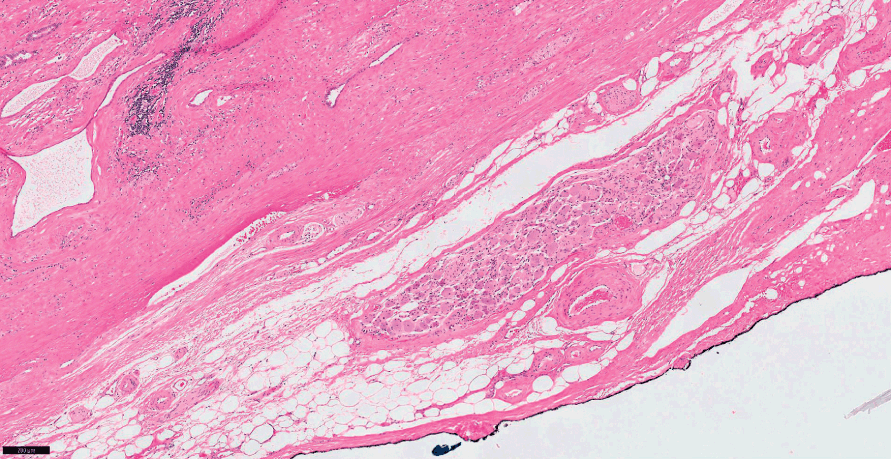
Fig 1.8 The paired ejaculatory ducts exit at the verumontanum. The prostatic utricle is seen located between the ejaculatory ducts. After puberty, the utricle shows an epithelium that no longer differs from the prostate glands.
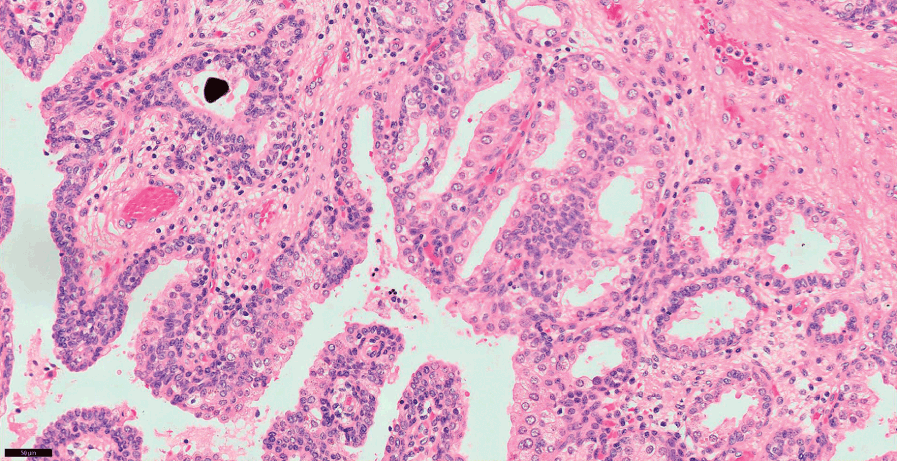
Fig 1.9 Close up view of the covering epithelium at the verumontanum (200x).
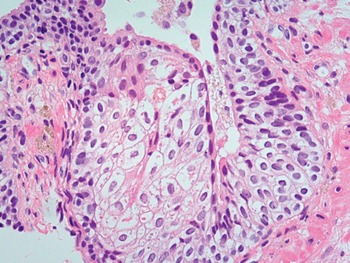
Fig 1.10 Focal squamous metaplasia in the prostate duct.
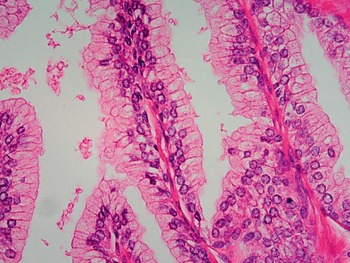
Fig 1.11 Prostate glands show basal cell layer and the secretory cells towards the lumen.
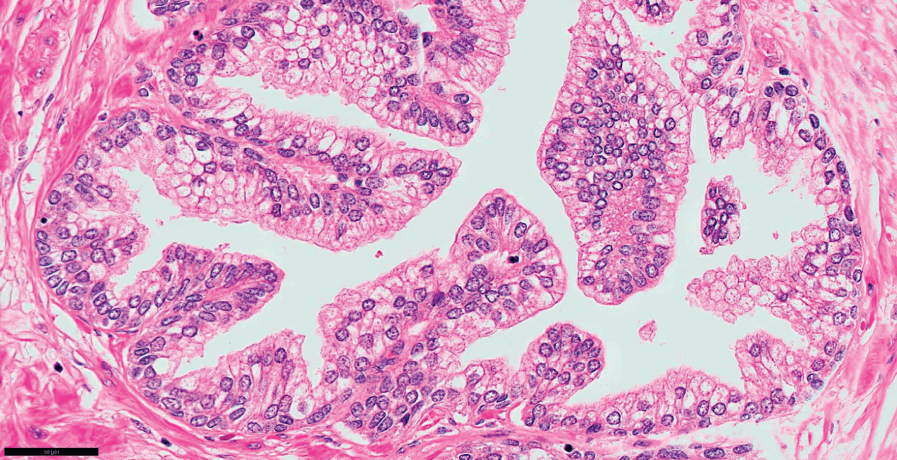
Fig 1.12 Prostate glands show secretory and basal cells surrounded by fibromuscular stroma.
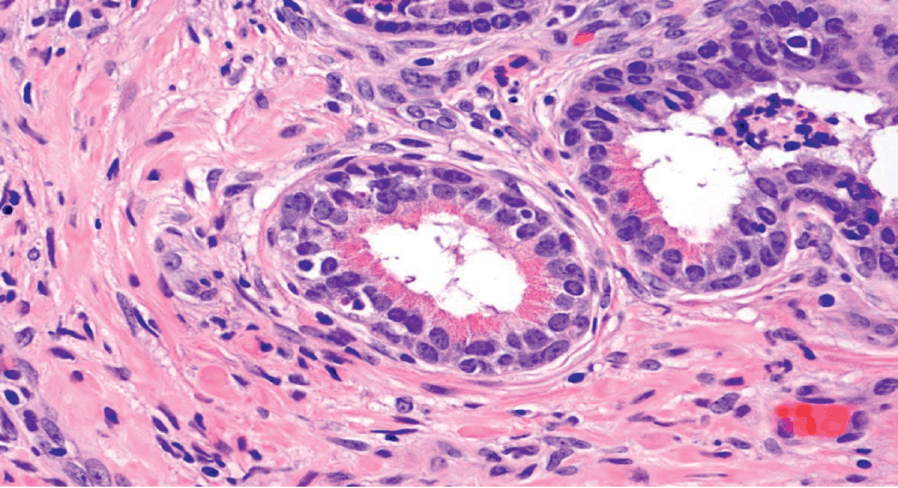
Fig 1.13 Paneth-like neuroendocrine cells may be seen in prostate glands.
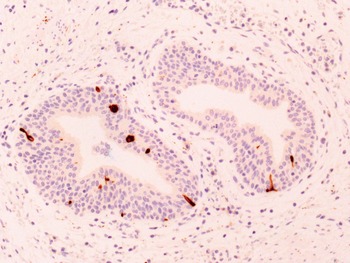
Fig 1.14 Neuroendocrine cells are present within the basal cells, but may extend to luminal surface of the gland (Anti-cromogranin antibody) (200x).

Fig 1.15 Lipofucsin pigment in secretory cells.
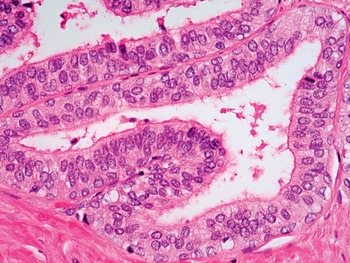
Fig 1.16 Prostate glands in the central zone show pseudostratified nuclei.

Fig 1.17 Prostate glands seen in the central zone with occasional roman-bridge structures.

Fig 1.18 Urothelial metaplasia is focally seen in some prostate glands.

Fig 1.19 “Telescoping” is occasionally seen in some normal prostate glands, a finding not to be mistaken as glomeruloid pattern of carcinoma.

Fig 1.20 Immunohistochemistry of the prostate glands and acini. Basal cells highlighted by 34βE12 antibody (A). Secretory cells showing weak racemase expression toward the lumen (B). Basal cells highlighted by p63 (C). GATA3 decorates urothelial metaplasia in the prostate.

Fig 1.21 Mucinous metaplasia may be occasionally seen in prostate glands.
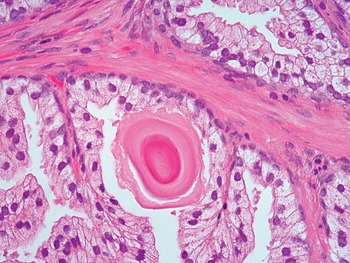
Fig 1.22 Corpora amylacea may be present in the lumen of prostate glands.
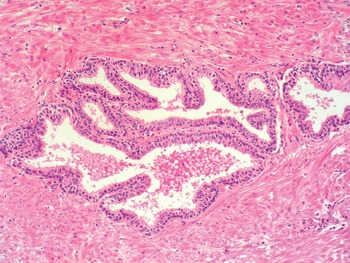
Fig 1.23 Fibromuscular stroma around glands.
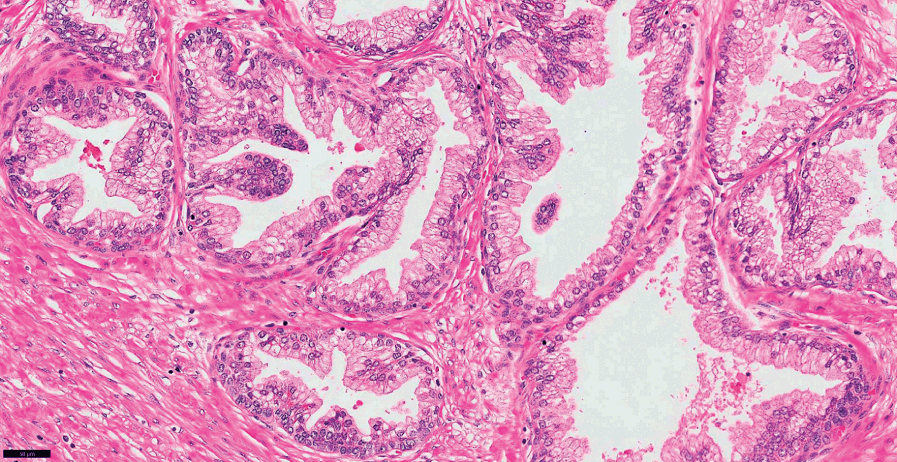
Fig 1.24 Fibromuscular stroma around glands (closer view).

Fig 1.25 Fat may be seen rarely in the prostate stroma (100x).

Fig 1.26 Small lymph nodes may be seen in the soft tissues around the prostate.

Fig 1.27 Benign glands encircling a nerve.

Fig 1.28 Paraganglia may be seen in needle prostate biopsies.
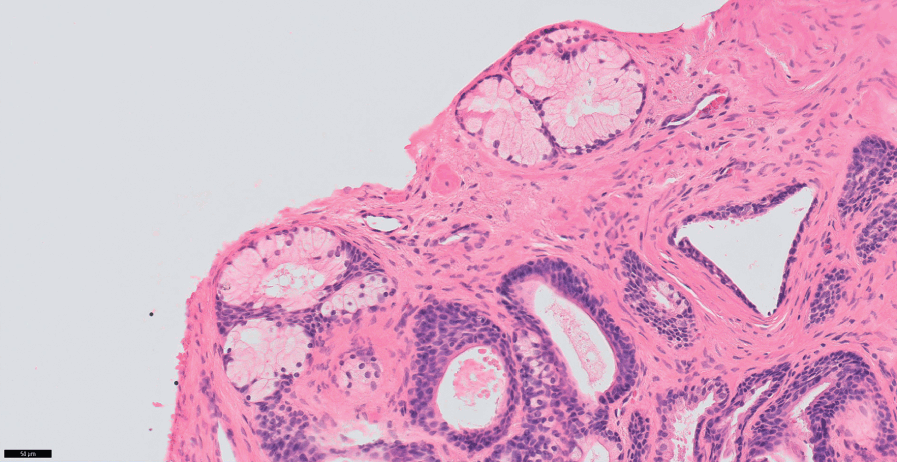
Fig 1.29 Cowper gland as seen in a needle prostate biopsy.

Fig 1.30 Immunohistochemistry of Cowper gland. 34βE12 (A) and p63 (B) positive expressions highlight basal cells. Negative PSA (C) and racemase (D).
| Atypical adenomatous hyperplasia (adenosis) |
| Atrophy |
| Cribriform hyperplasia |
| Basal cell hyperplasia |
| Nephrogenic metaplasia (nephrogenic adenoma) |
| Verumontanum mucosal gland hyperplasia |
| Squamous metaplasia |
| Urothelial metaplasia |
| Radiation atypia |
| Xanthogranulomatois prostatitis and other prostatitis |
| Malacoplakia |
| Extramedullary hematopoiesis |
| Endometriosis (rarity in men receiving estrogen therapy) |
| Atypical stromal cells related to BPH |
| Postoperative spindle cell nodule |
| Cowper glands |
| Paraganglia |
| Benign glands adjacent to nerves and skeletal muscle |
| Crowded benign acini |
| Signet ring cell change in stromal nodule |
| Seminal vesicle/ejaculatory duct epithelium |