Highlights
-
• Negative state triggers compensatory adjustments in language control processes to maintain performance comparable to that in neutral state.
-
• Compensatory mechanisms are engaged more strongly during cued than voluntary language switching across both phases of language control.
-
• The findings provide fine-grained evidence for adaptive compensatory mechanisms in bilingual language control under emotional states.
-
• They also offer new insights into the adaptability of the bilingual mind.
1. Introduction
Language control mechanism allows bilinguals to avoid interference from the nontarget languages and flexibly switch between languages. Typically, in the laboratory, this mechanism is studied independently of emotional states (see Jiang et al., Reference Jiang, Meng and Chen2024; Williams et al., Reference Williams, Srinivasan, Liu, Lee and Zhou2019, for exceptions). However, in everyday life, bilingual language production often occurs in emotional states (Dewaele, Reference Dewaele2010; Resnik, Reference Resnik2018). To better understand how bilinguals manage their languages in real-life situations, this study investigated the impact of emotional states on bilingual language control.
Furthermore, according to the influential adaptive control hypothesis (ACH; Green & Abutalebi, Reference Green and Abutalebi2013), the control system itself adapts to the social context of the communication. How, then, does this adaptive control system operate in emotional contexts? One possibility is that, in the face of emotional disruptions, the control system adaptively mobilizes compensatory top-down control according to the social contexts (Jiang et al., Reference Jiang, Meng and Chen2024). The present study aims to provide more fine-grained evidence for this adaptive compensatory mechanism using event-related potentials (ERPs).
1.1. Adaptive language control in bilinguals
According to the ACH, the control system adapts to different social contexts. For example, when Chinese–English bilinguals are conversing with English monolinguals, they are in a single-language context, where they are expected to speak exclusively in a single language (i.e., English). When talking with both Chinese and English monolinguals in the same environment, they are in a dual-language context, where they switch languages according to external cues (i.e., the interlocutors). When communicating with Chinese–English bilinguals, they are in a dense code-switching context, where they can freely switch between languages whenever they want. In this context, an opportunistic planning approach may be used, allowing bilinguals to use whichever words come to mind most readily, regardless of language. The ACH posits that a broader range of top-down control processes are involved in single- and dual-language contexts than dense code-switching contexts.
In laboratory settings, language switching is typically examined using a cued picture- or digit-naming paradigm, where participants switch between languages according to a cue indicating which language to use (Declerck & Philipp, Reference Declerck and Philipp2015; Meuter & Allport, Reference Meuter and Allport1999). However, several recent studies have also investigated language switching in a voluntary context, where participants are free to name pictures or digits in their language of choice (de Bruin et al., Reference de Bruin, Samuel and Duñabeitia2018; Gollan & Ferreira, Reference Gollan and Ferreira2009). Cued switching is most comparable to a dual-language context, while voluntary switching is more similar to a dense code-switching context (Blanco-Elorrieta & Pylkkänen, Reference Blanco-Elorrieta and Pylkkänen2018; de Bruin et al., Reference de Bruin, Samuel and Duñabeitia2018). Consistent with the ACH, cued switching has been observed to engage higher levels of control than voluntary switching.
In addition, naming in a single language throughout the entire block – which is most comparable to a single-language context – has been shown to be more effortful than freely mixing two languages (de Bruin et al., Reference de Bruin, Samuel and Duñabeitia2018; de Bruin & Xu, Reference de Bruin and Xu2023). This finding aligns with the proposal in the ACH that a single-language context involves higher levels of control than a dense code-switching context.
1.2. Adaptive compensatory mechanism in emotional contexts
To the best of our knowledge, only one behavioral study by Jiang et al. (Reference Jiang, Meng and Chen2024) has examined how the adaptive control system functions in emotional contexts. In their study, two groups of Chinese–English bilinguals performed a picture-naming task in neutral, negative and positive emotion conditions. One group named pictures in single-language and cued switching contexts, while another matched group named pictures in single-language and voluntary switching contexts.
Jiang et al. (Reference Jiang, Meng and Chen2024) proposed two possible mechanisms underlying adaptive control in emotional contexts. Specifically, it is possible that the control system functions as initially proposed in the ACH (cognitive effort account). Alternatively, it is possible that in single- and dual-language contexts, the control system recruits additional control processes to avoid language control failures (e.g., cross-language intrusions) incurred by emotional states and bring behavior more in line with the intended goal (e.g., avoiding cross-language intrusions). By contrast, in dense code-switching contexts, such compensatory adjustments in control processes would not be invoked, since increased control failures would not deviate from the intended goal of using both languages opportunistically (adaptive compensatory control account).
The results revealed that proactive control, which involves anticipating and preventing cross-language interference before it occurs, was impaired by negative state but facilitated by positive state. Most importantly, evidence was found in support of both possibilities regarding how the adaptive control system works in emotional contexts. On the one hand, negative state slowed responses to a lesser degree on voluntary nonswitch than single-language trials, suggesting that the control system may operate as initially proposed in the ACH. That is, single-language context engages more top-down control than dense code-switching context, and emotional effects become greatest with cognitively demanding tasks (Forgas, Reference Forgas1995; Seibert & Ellis, Reference Seibert and Ellis1991). On the other hand, negative state reduced the reversed language dominance effect (RLDE, slower responses in the dominant L1 than in the nondominant L2 in the mixed-language condition), an index of proactive inhibition of L1 or proactive activation of L2. This reduction in RLDE was more pronounced in voluntary than cued switching, with no difference in RLDE between the two switching types under the neutral state. Since smaller RLDE has been associated with reduced efficiency of proactive control (Stasenko et al., Reference Stasenko, Kleinman and Gollan2021), this finding indicates that negative states disrupted proactive control to a larger degree in voluntary than cued naming, and thus supports the adaptive compensatory control account. Therefore, Jiang et al. (Reference Jiang, Meng and Chen2024) concluded that the adaptive control system proposed by the ACH may function in two different, but not mutually exclusive, ways in emotional contexts.
Jiang et al. (Reference Jiang, Meng and Chen2024) provided the first empirical evidence for the adaptive compensatory mechanism, which seems to be a unique form of adaptive control that arises in emotional contexts. However, since reaction times (RTs) they collected reflect the outcome of a sequence of processes, the time course of this mechanism remains unclear. Bilingual language control occurs between language tags/schemas and between lemmas (Green, Reference Green1998; Guo et al., Reference Guo, Ma and Liu2013; Liu et al., Reference Liu, Liang, Dunlap, Fan and Chen2016). During the language schemas competition phase, the target language schema (i.e., speaking in L1 or L2) is selected. Then, during the lexical selection phase, the selected language schema inhibits lexical items in the nontarget language.
The present study aims to identify the specific phase of adaptive compensatory mechanism activation by using ERPs and separating cue and stimulus presentation. Cue-locked ERPs index the language schemas competition phase, whereas stimulus-locked ERPs reflect the lexical selection phase (Guo et al., Reference Guo, Ma and Liu2013).
1.3. Neural markers of compensatory adjustments in control processes
The core component of the compensatory mechanism lies in the recruitment of greater top-down control or cognitive effort. At the neural level, it can be reflected in enhanced brain activity (Berggren & Derakshan, Reference Berggren and Derakshan2013). For example, in neuroimaging research of attentional control, enhanced neural activities over frontal areas have been commonly associated with increased recruitment of top-down attentional control in pursuit of task goals (Berggren & Derakshan, Reference Berggren and Derakshan2013). Thus, the enhanced frontal activity under higher levels of negative emotions, together with preserved behavioral performance, has been interpreted as evidence for a compensatory mechanism, in which additional cognitive resources are mobilized to maintain high task performance (Eysenck et al., Reference Eysenck, Moser, Derakshan, Hepsomali and Allen2023).
In ERP studies of bilingual language control, the most commonly studied components are the N2 and the late positive component (LPC; Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Liu et al., Reference Liu, Zhang, Blanco-Elorrieta, He and Chen2020). The N2 is a negative, fronto-central potential that typically peaks around 200 ms after stimulus onset (Christoffels et al., Reference Christoffels, Firk and Schiller2007; Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001). Its amplitude is thought to reflect the amount of inhibition applied to the nontarget language during bilingual language production – it increases with enhanced inhibition (Guo et al., Reference Guo, Ma and Liu2013; Kang et al., Reference Kang, Ma and Guo2018). For example, switch trials (especially L2 switch trials) typically elicit increased N2 amplitudes relative to nonswitch trials (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001). This switching effect has been explained with the inhibitory processes needed to suppress the previous language and enable a successful switch. Switching into the L2 requires strong inhibition of the dominant L1, thus resulting in a robust N2 switching effect in L2 (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001). Moreover, N2 amplitudes can be higher for L1 than for L2 trials in mixed-language blocks, which is typically coupled with RLDE in RTs and has been accounted for with global inhibition of L1 (Kang et al., Reference Kang, Ma and Guo2018; Zhang et al., Reference Zhang, Li, Ma, Kang and Guo2021).
The LPC is a positive deflection that typically peaks around 600 ms after stimulus onset over parietal electrode sites (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Martin et al., Reference Martin, Strijkers, Santesteban, Escera, Hartsuiker and Costa2013). LPC amplitude is associated with the amount of cognitive effort exerted during task performance. It can increase as more top-down control is exerted, as in the case of the switching effect; namely, switch trials come with increased LPC amplitudes relative to nonswitch trials (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Liu et al., Reference Liu, Zhang, Blanco-Elorrieta, He and Chen2020; Martin et al., Reference Martin, Strijkers, Santesteban, Escera, Hartsuiker and Costa2013; Timmer et al., Reference Timmer, Grundy and Bialystok2017). Meanwhile, LPC can be enhanced when the task is relatively easy and more attentional resources are available for stimulus processing and memory updating (Timmer et al., Reference Timmer, Grundy and Bialystok2017). When RLDE was observed in RTs, LPC amplitudes were found to be higher for L2 than for L1 trials, coupled with RLDE in N2 (Jiao et al., Reference Jiao, Gao, Schwieter, Li, Zhu and Liu2022) or no language modulation in N2 (Timmer et al., Reference Timmer, Christoffels and Costa2019). This RLDE effect of LPC has been taken as evidence for inhibition of L1 (Timmer et al., Reference Timmer, Christoffels and Costa2019). In this case, the reduced LPC on L1 trials, relative to L2 trials, can be explained in terms of greater difficulty and less available cognitive resources for L1 processing, possibly due to enhanced inhibition of L1.
Therefore, compensatory adjustments in language control will be reflected in enhanced neural markers of top-down control – namely, greater switching effects and RLDE in N2 and LPC, along with preserved performance.
1.4. The present study
The present study seeks more fine-grained evidence for the adaptive compensatory language control process under emotional states using ERPs. The experiment featured a 2 (task type: cued vs. voluntary) × 3 (emotion: neutral vs. negative vs. positive) × 2 (trial type: switch vs. nonswitch trials) × 2 (language: L1 vs. L2 trials) within-participant design. We focused on the cued vs. voluntary naming comparison where previous behavioral evidence for the adaptive compensatory process was observed (Jiang et al., Reference Jiang, Meng and Chen2024).
The compensatory increases in reactive and proactive language control processes are examined. Reactive language control is implemented at a local, trial-by-trial level, resolving cross-language interference after it occurs, whereas proactive control functions at a more global level, anticipating and minimizing potential interference before it arises (Declerck, Reference Declerck2020; Ma et al., Reference Ma, Li and Guo2016). Switching cost and the RLDE are indicators of reactive and proactive language control, respectively. Switching cost refers to poorer performance, along with increased N2 and LPC amplitudes, on switch trials, where the response is in a different language than the previous trial, compared with nonswitch trials, where the response remains in the same language (Green, Reference Green1998; Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001). In contrast, the RLDE is characterized by slower responses, larger N2 and smaller LPC for L1 than for L2 trials within mixed-language blocks (Christoffels et al., Reference Christoffels, Firk and Schiller2007; Costa & Santesteban, Reference Costa and Santesteban2004; Jiao et al., Reference Jiao, Gao, Schwieter, Li, Zhu and Liu2022; Kang et al., Reference Kang, Ma and Guo2018; Timmer et al., Reference Timmer, Christoffels and Costa2019; Zhang et al., Reference Zhang, Li, Ma, Kang and Guo2021).
We hypothesize that compensatory adjustments should occur under negative state, but not under positive state, and that these adjustments should be more pronounced in cued than voluntary switching. Given that both language schema competition and lexical selection phases require top-down control (Reverberi et al., Reference Reverberi, Kuhlen, Abutalebi, Greulich, Costa, Seyed-Allaei and Haynes2015, Reference Reverberi, Kuhlen, Seyed-allaei, Greulich, Costa, Abutalebi and Haynes2018), we further hypothesize that negative state will trigger compensatory adjustments in both phases, with these adjustments being more pronounced in cued than voluntary switching. In cued switching, negative state should enhance the switching effect and RLDE in both cue- and stimulus-locked N2/LPC to a greater extent than in voluntary switching, thereby preserving behavioral performance.
The increase in switching effect should come from switch trials, while the increase in RLDE should stem from L1 trials, as top-down control underlying these effects is primarily engaged during switch trials and L1 trials, respectively.
2. Method
2.1. Participants
Sixty-seven Chinese–English bilinguals from Beijing Normal University in China participated in this study for monetary compensation. All participants had normal or corrected-to-normal vision and reported no history of neurological or psychological impairments or the use of psychoactive medication. All participants signed written informed consent. This study received ethical approval from the Committee of Protection of Participants at Beijing Normal University.
Sixty-one participants engaged in the main experimentFootnote 1. They chose no more than 65% of the time the preferred language (i.e., Chinese) in a screening test, the picture stimuli and procedure of which were similar to those used in the voluntary task of the main experiment. One participant withdrew from the main experiment, and another was excluded due to excessive electroencephalography (EEG) artifacts. The final sample for the main experiment consisted of 59 participants. An a priori power analysis using G*Power 3.1.9.6 (Faul et al., Reference Faul, Erdfelder, Lang and Buchner2007) indicated that a minimum of 29 participants was needed to detect a small to medium effect size for the critical three-way interactions (Language × Emotion × Task; Trial Type × Emotion × Task) with the following parameters: F-tests > ANOVA: repeated measures, within factors; f = .25; α = .01; power of 1-β = .99; number of groups = 1; number of measurements = 12; correlation among repeat measures = .5; nonsphericity correction ∈ = 1. However, due to the noise inherent in ERPs data, we doubled the target sample size to ensure stable estimation of effects.
Participant characteristics are reported in Table 1. The linguistic background and language proficiency were assessed using the Language History Questionnaire (LHQ, version 3; Li et al., Reference Li, Zhang, Yu and Zhao2020). All participants were exposed to Chinese (L1) from birth and learned English (L2) in a classroom setting. They had passed the College English Test Band 4 (CET-4), a standardized test administered by the Ministry of Education of China to evaluate non-English major college students’ English proficiency in writing, listening, reading and translating. None had lived in or traveled to English-speaking countries for more than three months, with three exceptions. Paired-samples t tests revealed that participants were dominant in L1 across all skills (all ts > 8.89, ps < .001). Their average Quick Placement Test (QPT) score was equivalent to the B1 (i.e., lower intermediate) level in the Common European Framework of Reference (CEFR). The relatively low average Bilingual Switching Questionnaire (BSWQ) score indicated that participants seldom engaged in code-switching in their daily lives. Meanwhile, the average Foreign Language Classroom Anxiety Scale (FLCAS) score indicated a middle level of foreign language anxiety.
Participant characteristics

a Ratings were on a scale from 1 (very poor) to 7 (excellent).
b The first part of the Quick Placement Test (QPT, version 2) was administered to obtain the objective indicator of English proficiency. Higher scores indicate higher levels of English proficiency (total score: 40).
c BSWQ (Rodriguez-Fornells et al., Reference Rodriguez-Fornells, Krämer, Lorenzo-Seva, Festman and Münte2012) is composed of 12 items measuring the frequency of switching behavior. Higher scores indicate more frequent switching (total score: 60).
d Chinese version of the Foreign Language Classroom Anxiety Scale (FLCAS; Horwitz et al., Reference Horwitz, Horwitz and Cope1986; Shao et al., Reference Shao, Yu and Ji2013) was administered to assess foreign language anxiety. Higher scores indicate higher levels of anxiety (total score: 165). Scores above 132 signify high anxiety, scores between 99 and 132 denote a middle level of anxiety, and scores below 99 imply little or no anxiety.
2.2. Materials
2.2.1. Picture naming
For the formal experiment, a total of 13 target and 6 filler black-and-white line drawings were selected from Snodgrass and Vanderwart (Reference Snodgrass and Vanderwart1980). All Chinese names were bisyllabic words, and English names were mono- or bisyllabic words, ranging from three to six letters in length. Twenty-six Chinese–English bilinguals (24 females; average age: 24.3 years, SD = 3.65) having passed the CET-4 rated their familiarity with the Chinese and English names on a five-point scale (1 = very unfamiliar; 5 = very familiar).
Paired-samples t tests showed no significant difference in familiarity between Chinese (M = 4.90, SD = 0.07) and English (M = 4.92, SD = 0.06) names of the 13 target items, t(12) = −1.10, p = .29. In addition, the naming agreement (measured by the H statistic, referring to the number of different names speakers use to refer to a given object) in Chinese (M = 0.50, SD = 0.48, range = 0–1.37; Zhang & Yang, Reference Zhang and Yang2003) and English (M = 0.37, SD = 0.32, range = 0–0.89; Snodgrass & Vanderwart, Reference Snodgrass and Vanderwart1980) was not significantly different for the 13 target items, t(12) = 0.84, p = .42.
For the screening test, a different set of 13 line drawings were selected. Familiarity ratings for Chinese (M = 4.85, SD = 0.10) and English names (M = 4.88, SD = 0.09), as well as naming agreement in Chinese (M = 0.76, SD = 0.47, range = 0–1.47) and English (M = 0.46, SD = 0.41, range = 0–1.27), again showed no significant difference (all |t|s < 1.72, all ps > .11).
2.2.2. Emotion elicitation and assessment
A total of 27 instrumental musical pieces (nine pieces for each emotion condition) were selected from the Chinese Affective Music System (CAMS, Li et al., Reference Li, Cheng, Dai, Wang and Huang2012) to elicit emotional states (for further details, see Jiang et al., Reference Jiang, Meng and Chen2024). The positive musical pieces had significantly higher arousal and pleasure ratings than neutral musical pieces (ps < .001). The negative musical pieces were significantly lower than neutral ones in both arousal and pleasure ratings (ps < .01). Participants’ emotional responses were assessed using a paper-and-pencil 9 × 9 Affect Grid (Russell et al., Reference Russell, Weiss and Mendelsohn1989), which assesses emotion along two dimensions: pleasure (1 = extremely unpleasant feelings; 9 = extremely pleasant feelings) and arousal (1 = extreme sleepiness; 9 = extremely high arousal).
2.3. Procedure
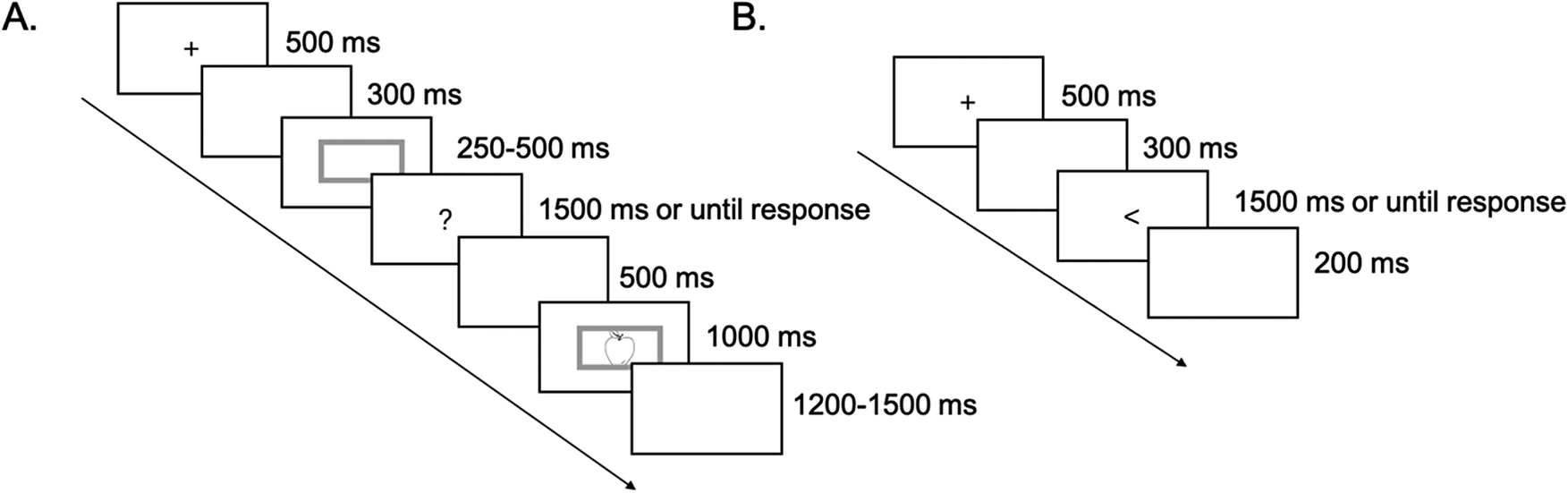
The experiment was programmed using E-Prime 2.0 (Psychology Software Tools, Inc., PA, USA). Following the screening test, participants completed the background questionnaires described above. To minimize errors related to not recognizing the pictures or using the wrong word, participants were first familiarized with the 13 target and 6 filler pictures along with their corresponding names. All participants then practiced a flanker task and the naming tasks (involving the filler stimuli), in the same order as in the main task. The practice session included eight trials each for the two naming conditions. Next, participants were instructed on how to rate their emotional state with the 9 × 9 Affect Grid (for examples of the instructions, see Jefferies et al., Reference Jefferies, Smilek, Eich and Enns2008; Russell et al., Reference Russell, Weiss and Mendelsohn1989). They were told to do this whenever a 9 × 9 Affect Grid appeared on the screen. After this instruction, participants were given earphones to wear for the duration of the experiment. Figure 1 illustrates the schematic diagram of the main experimental procedure in each emotion condition.
Schematic diagram of the procedure in each emotion condition. Participants were given a break after the flanker task and each naming condition. Each emotion induction procedure lasted 5 minutes, followed by either the picture-naming or the flanker task. Shorter versions of this procedure, that is, 2-min “boosters”, were administered between the 6-min flanker task, the 7-min cued naming section and the 7-min voluntary naming section. Participants reported their current emotional states nine times. They made their first ratings (Grid 0) before the 5-min emotion-induction procedure. Subsequent ratings (Grid 1–8) were made to precede and follow the flanker task and each of the four naming subsections. Grid = 9 × 9 Affect Grid.

2.3.1. Block and trial structures for picture-naming tasks
In each emotion condition, the picture-naming task consisted of both voluntary and cued naming sections, with their order counterbalanced between participants. In addition, the order of the picture-naming task and a flanker task (not analyzed in the present study) was also counterbalanced between participants. Unless otherwise specified, all pictures and symbols were displayed at the center of the computer screen.
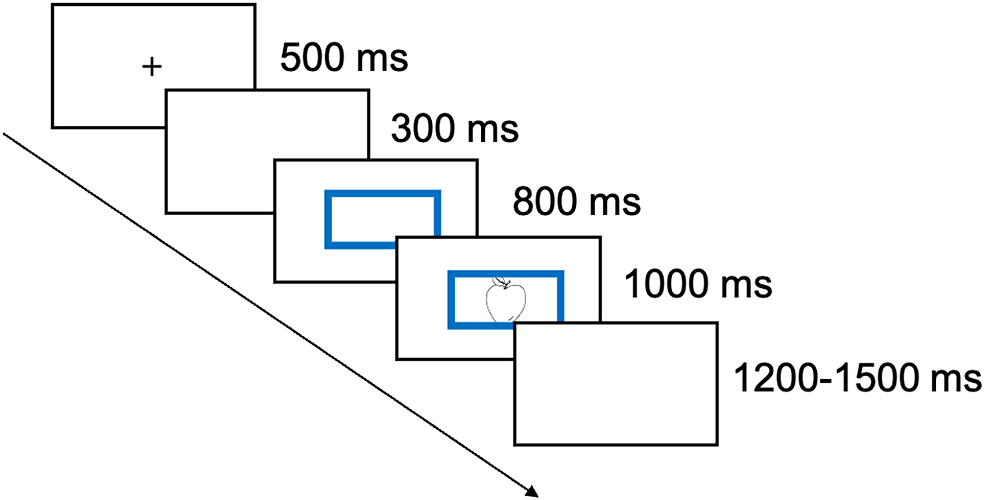
In the cued naming section, participants were required to name each picture in the language indicated by the color frame presented before the picture. The assignment of color cues (blue or red) to languages (Chinese or English) was counterbalanced between participants. Instructions were provided in Chinese on the screen. Figure 2 illustrates the trial structure for cued naming. Participants were asked to name the pictures aloud as fast and correctly as possible. Their behavioral responses were recorded on a digital recorder, and the RTs within 2,200 ms since picture onset were collected by a Serial Response Box (Psychology Software Tools, Inc., PA, USA). EEG responses time-locked to both cue and stimulus onset were recorded.
Trial structure for cued naming. Each trial started with a fixation cross “+” for 500 ms, followed by a 300 ms blank screen. A color frame then appeared, and after 800 ms, a picture to be named (stimulus) was presented inside the color frame for 1,000 ms. Next, a blank screen was presented for a duration randomly jittered between 1,200 and 1,500 ms.

In the voluntary naming section, participants were free to choose the language in which to name each picture. The trial structure was similar to that of the cued naming section, with one key difference: the blue/red color frame was replaced by the gray frame. Following Reverberi et al. (Reference Reverberi, Kuhlen, Seyed-allaei, Greulich, Costa, Abutalebi and Haynes2018), the Chinese version of this instruction was presented on the screen: “Once you see the gray frame, choose a language to name the upcoming picture. Keep your choice in mind and do not change it. Once the picture appears, use the language you’ve chosen just now to name it. Note that you must decide on the language as soon as you see the gray frame. Try to choose the language that comes to mind first, but do not use the same language throughout the whole task.” In this way, the language choice phase was set ahead of the stimulus onset to dissociate the language schemas competition and lexical selection phases, Participants’ behavioral responses and EEG responses time-locked to cue and stimulus onset were recorded.
The voluntary naming section also included two types of filler trials (involving filler pictures). Figure 3 illustrates the structure of these filler trials. Type I filler trials were used to avoid participants delaying their language choice until after the picture onset, while type II filler trials were set to avoid participants choosing the language before the gray frame appeared (e.g., immediately after naming the previous picture).
Structure for Type I (A) and Type II (B) of filler trials in voluntary naming. For Type I, each trial began with a fixation cross “+” for 500 ms, followed by a 300 ms blank screen. A gray frame then appeared, and after 250–500 ms, it disappeared and was replaced by a question mark “?”. Participants were required to verbally state, in Chinese, the language they had chosen when they saw the gray frame as fast as possible. If they did not respond within 1,500 ms, the question mark disappeared. Their responses were recorded via a digital recorder. After a 500 ms blank screen, the gray frame reappeared with a picture inside it. After 1,000 ms, a blank screen was presented for a duration randomly jittered between 1,200 and 1,500 ms. For Type II, each trial began with a fixation cross “+” for 500 ms, followed by a 300 ms blank screen. Next, an arrow (“<” or “>”) appeared. Participants were required to respond as quickly as possible to the pointing direction of the arrow by pressing the button on the keyboard (i.e., “F” button for pointing left; “J” button for pointing right). If no response was made within 1,500 ms, the arrow disappeared and a 200 ms blank screen followed.

For each naming section, 104 experimental trials were included in each emotion condition, equally distributed across two subsections. Each of the 13 pictures appeared a total of eight times per section (four times in each subsection). Each subsection began with a filler picture, which set the task for the first experimental trial (i.e., as a switch or a nonswitch trial). For the cued naming section, language switches were cued on 26 of the 104 trials (13 of the 52 trials per language), resulting in a switch rate of 25%, closely matching the voluntary switch rate of 29.70% reported in Jiang et al. (Reference Jiang, Meng and Chen2024) for this bilingual group. Each target item appeared once in a Chinese-switch trial, once in an English-switch trial, three times in Chinese-nonswitch trials and three times in English-nonswitch trials. Six pseudorandomized trial lists were created for each emotion condition based on the following constraints: (1) the same item never appeared consecutivelyFootnote 2; (2) within each subsection, each item appeared once in a switch trial and three times in nonswitch trialsFootnote 3; (3) within each subsection, each item was preceded twice by a red frame language cue and twice by a blue frame language cue. For the voluntary naming section, six pseudorandomized trial lists were created for each emotion condition, such that no item appeared consecutively. Each list also included five Type I and five Type II filler trials, distributed across the two subsections (three Type I and two Type II trials in one subsection, and two Type I and three Type II in the other). For each naming section, participants were randomly assigned to one of the six lists. In total, 18 pseudorandomized lists (six per emotion condition) were created per naming section.
2.3.2. Emotion induction procedure
A blocked design was used to induce sustained emotional states, with the order of the three emotion blocks (negative, positive and neutral) counterbalanced across the participants. In the positive and negative conditions, participants were instructed to develop a particular emotion by listening to emotionally congruent music while generating matching thoughts; for example, recalling happy events while listening to the happy music. In the neutral condition, participants listened to neutral music while imagining a series of mundane activities, such as grocery shopping, doing small tasks at home and calling a family member. The relevant music was stopped during the picture-naming tasks to prevent auditory interference with the EEG recording. Participants were fully debriefed at the end of the experiment.
2.4. EEG recording and preprocessing
Electrophysiological data during the picture-naming task were recorded using an electrode cap with 64 Ag/AgCl electrodes (NeuroScan, Inc.), arranged according to the extended 10–20 positioning system. EEGs were referenced online to CZ, with impedances maintained below 5 kΩ. The data were sampled at 1,000 Hz and band-pass filtered online between 0.05 and 100 Hz.
Offline preprocessing was conducted using EEGLAB (Delorme & Makeig, Reference Delorme and Makeig2004) and ERPLAB (Lopez-Calderon & Luck, Reference Lopez-Calderon and Luck2014) toolboxes in Matlab (MathWorks, Inc.). We re-referenced the EEG data to the average of the left and right mastoids (M1, M2) and then low-pass filtered the data at 30 Hz. Next, visual inspection was carried out to identify and remove segments of egregious artifacts, and noisy channels were interpolated to improve the accuracy of subsequent independent components analysis (ICA; Debnath et al., Reference Debnath, Buzzell, Morales, Bowers, Leach and Fox2020; Heise et al., Reference Heise, Mon and Bowman2022). ICA was then performed to identify and correct eye blink artifacts. After that, trials corresponding to naming errors, responses that could not be classified as switch or nonswitch and audio recording failures were removed offline.
Subsequently, we segmented continuous EEGs into epochs ranging from –100 ms to 700 ms relative to both cue and stimulus onset, with baseline correction performed in reference to pre-cue/stimulus activity (–100 to 0 ms). Finally, we subjected all epochs to an automatic rejection procedure where epochs containing deviations larger than 100 μV were automatically discarded. For both cue-locked and stimulus-locked data, at least 50% of epochs remained for each participant and condition. Data from one participant in the negative condition and another in the positive condition were removed for not following this constraint. Given the relatively small number of switch trials per participant (e.g., 26 cued switch trials per emotion condition initially included in the experiment), we applied a lenient 50% criterion a priori (for a discussion about lowering the threshold due to the difficulty in obtaining sufficient artifact-free epochs, see Luck, Reference Luck2014, p. 210). Notably, we used the linear mixed-effects model (LME) to analyze single-trial ERPs data. Unlike traditional mean averaging approaches that require excluding participants with too few artifact-free trials due to concerns about low signal-to-noise ratios, LME allows for the inclusion of participants even with only a limited number of usable trials (Heise et al., Reference Heise, Mon and Bowman2022). For the number of epochs per condition used in ERPs analyses, see Table A.1 in the Supplementary Material.
Both cue- and stimulus-locked ERPs were analyzed to tease apart the language schemas competition phase from the lexical selection phase. Based on previous literature (Kang et al., Reference Kang, Ma and Guo2018; Liu et al., Reference Liu, Liang, Dunlap, Fan and Chen2016, Reference Liu, Zhang, Blanco-Elorrieta, He and Chen2020; Timmer et al., Reference Timmer, Grundy and Bialystok2017) and visual inspection of the waveforms, cue-locked N2 and LPC mean amplitudes were extracted from the 250- to 350-ms and 350- to 450-ms time windows after cue onset, respectively. Meanwhile, stimulus-locked N2 and LPC mean amplitudes were extracted from the 230- to 400-ms and 400- to 600-ms time windows after stimulus onset, respectively. We also conducted complementary analyses on N2 and LPC mean amplitudes extracted from shortened stimulus-locked windows (250–350 ms and 450–550 ms), which aligned exactly with the 100-ms duration used for cue-locked ERPs. These analyses yielded an identical pattern of results.Footnote 4 Note that although the cue-locked positivity in the 350–450 ms time window occurs earlier than the classic LPC, it aligns with the LPC time window reported in earlier work (e.g., 325–400 ms in Timmer et al., Reference Timmer, Grundy and Bialystok2017) and is thus referred to as LPC in this study.
Following previous studies, we extracted stimulus-locked N2 mean amplitudes from anterior-central medial electrodes (i.e., F3, F1, FZ, F2, F4, FC3, FC1, FCZ, FC2, FC4, C3, C1, CZ, C2 and C4; Kang et al., Reference Kang, Ma and Guo2018, Reference Kang, Ma, Li, Kroll and Guo2020; Zhang et al., Reference Zhang, Li, Ma, Kang and Guo2021), and stimulus-locked LPC mean amplitudes from centro-posterior medial electrodes (i.e., C3, C1, CZ, C2, C4, CP3, CP1, CPZ, CP2, CP4, P3, P1, PZ, P2 and P4; Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Martin et al., Reference Martin, Strijkers, Santesteban, Escera, Hartsuiker and Costa2013; Timmer et al., Reference Timmer, Grundy and Bialystok2017). Cue-locked ERPs remain understudied, yet previous research has identified distinct scalp distributions of switching effect along the anteriority dimension. For instance, Verhoef et al. (Reference Verhoef, Roelofs and Chwilla2010) and Liu et al. (Reference Liu, Zhang, Blanco-Elorrieta, He and Chen2020) observed early switching effects in posterior negativity, whereas Declerck et al. (Reference Declerck, Meade, Midgley, Holcomb, Roelofs and Emmorey2021) reported early switching effects in both anterior negativity and posterior positivity. Thus, we extracted cue-locked N2 and LPC amplitudes from five predefined regions of interest (ROIs) along the anteriority dimension: prefrontal (F1, FZ, F2), fronto-central (FC1, FCZ, FC2), central (C1, CZ, C2), centro-parietal (CP1, CPZ, CP2) and parietal (P1, PZ, P2). Anteriority was then included as a fixed factor in our models of cue-locked ERPs.
2.5. Data analysis
The naming language was coded for the voluntary naming task, and the trial type (switch or nonswitch) was coded afterward. Accuracy in both cued and voluntary naming tasks was scored as follows: (A) no or late response; (B) correct response; (C) correct language but wrong word (e.g., cake instead of bread); (D) wrong language (only for the cued naming task); (E) hesitations, utterance repairs, partial responses or coughing; (F) combination of two languages (e.g., hou[zi]-monkey); (G) responses that could not be classified as a switch or nonswitch trial because they were preceded by trials of no response, or trials of Type F or I; (H) RTs recording failures; and (I) audio recording failures. Filler trials were excluded from all analyses. For RTs analyses, only trials of Type B were included. For accuracy analyses, only trials of Type I were excluded. Trials of Type A, C, D, E and F were coded as incorrect.
Statistical analyses were conducted in R (version 4.5.0). We first checked the effectiveness of the emotion induction procedure, submitting the ordinal pleasure and arousal rating data separately to cumulative link mixed models (CLMMs; package ordinal; Christensen, Reference Christensen2022). The model had treatment-coded fixed effects of Emotion Block (baseline level: neutral) and Grid (baseline level: Grid 0) as well as their interactions. The initial models were fitted with a maximal random-effects structure (Barr et al., Reference Barr, Levy, Scheepers and Tily2013): random intercepts for participants and slopes for all within-participant predictors. If this model failed to converge, we simplified the random-effects structure by removing the random slopes stepwise and compared the model fits using the likelihood ratio test. The model reduction was stopped when it resulted in a loss in goodness of fit (p < .20; Matuschek et al., Reference Matuschek, Kliegl, Vasishth, Baayen and Bates2017), unless convergence issues persisted. Next, we removed nonsignificant fixed effects based on the likelihood ratio test. The model reduction was stopped when there was a significant deterioration in model fit (p < .05). For significant interactions, follow-up pairwise comparisons were conducted (EMMEANS; package emmeans; Lenth et al., Reference Lenth, Buerkner, Herve, Jung, Love, Miguez, Riebl and Singmann2022), with p values corrected for multiple comparisons using Tukey.
Next, we analyzed the picture-naming data. RTs and single-trial ERPs mean amplitudes were submitted to the linear mixed-effects model (LMER; package lme4; Bates et al., Reference Bates, Mächler, Bolker and Walker2015). Since naming accuracy was above 98% across all emotion conditions – indicating ceiling-level performance – it was not analyzed further. RTs were log-transformed to better approximate a normal distribution.
Emotion Block (neutral, positive, negative), Language (L1, L2), Trial type (switch, nonswitch), Task (cued, voluntary) and their interactions were included as fixed effects. For the cue-locked ERPs data, ROIs (prefrontal, fronto-central, central, centro-parietal and parietal) and their interactions with other factors were also included as fixed effects. The levels of factor Language and Trial type were coded in proportion to their presence in the data so that the average weighted value was 0 (RTs model: L1 = −0.523, L2 = +0.477; nonswitch = −0.309, switch = +0.691; cue-locked ERPs model: L1 = −0.521, L2 = +0.479; nonswitch = −0.310, switch = +0.690; stimulus-locked ERPs model: L1 = −0.520, L2 = +0.480; nonswitch = −0.311, switch = +0.689). Emotion Block was treatment coded with neutral block as the baseline level, as we were mainly interested in whether the neutral block differed from the other two emotional blocks (for a discussion about using a priori contrasts to code fixed effects, see Schad et al., Reference Schad, Vasishth, Hohenstein and Kliegl2020). The factor Task was treatment-coded separately with voluntary task and cued task set as the baseline levels, in order to estimate the emotional effects in each task, as well as the interaction effects involving task. The factor ROIs was sum-coded. The initial RTs models were fitted with a maximal random-effects structure: random intercepts for participants and items, and slopes for all within-participant/item predictors. The initial ERPs models were fitted with a maximal random-effects structure: random intercepts for participants, items and channels, and slopes for all within-participant/item/channel predictors. When the initial models did not converge, correlations between random slopes were removed. If this was not sufficient, random slopes accounting for less than 1% of the variance of their associated random factors were identified and removed simultaneously. If the model still failed to converge, random slopes explaining the least variance were removed until convergence was reached. For models with significant fixed effects, p values were provided by the summary/ANOVA function of the package LmerTest (Kuznetsova et al., Reference Kuznetsova, Brockhoff and Christensen2017). Only significant results (p < .05) are discussed.
3. Results
3.1. Emotion manipulation check
Ratings were incomplete for two participants. Scores on Grid 0 in the neutral block were missing for one participant, and scores on Grid 2 in the neutral block were missing for the other participant.
There was a significant interaction between Grid and Emotion Block for both pleasure and arousal ratings [pleasure: χ 2 (16) = 260.66, p < .001; arousal: χ 2 (16) = 60.94, p < .001]. Follow-up contrasts revealed no significant differences in pleasure or arousal ratings across emotion blocks at baseline (Grid 0) (all |z|s < 1.20, all ps > .45). After the initial emotion induction procedure (Grid 1–8), the pleasure ratings were higher in the positive compared with the neutral block (all |z|s > 3.70, all ps < .001), and lower in the negative compared with the neutral block (all |z|s > 5.09, all ps < .001). The arousal ratings showed a more complex pattern after the initial emotion induction procedure (Grid 1–8). In the positive block, arousal ratings did not differ from those in the neutral block at Grid 2, 3, 5, and 6 (all |z|s < 2.31, all ps > .05), but were higher at all other time points (all |z|s > 2.95, all ps < .01). In the negative block, arousal ratings were generally comparable to those in the neutral block (all |z|s < 1.70, all ps > .21), except at Grid 3, where ratings were significantly lower in the negative block (z = 2.62, p < .05). Overall, these results indicated that the emotion manipulations effectively induced the target emotional states, at least along the pleasure dimension.
3.2. The influence of emotional states and switching contexts
3.2.1. Behavioral results
In the voluntary naming task, the total number of trials, excluding those that could not be classified as switch or nonswitch, was 3,286, 5,132, 3,443 and 6,421 for L1 switch, L1 nonswitch, L2 switch and L2 nonswitch conditions, respectively.Footnote 5 On average, participants switched on 37.81% of trials (SD = 14.65). Across the items that could be classified as switch or nonswitch trials, 46.03% (SD = 10.85) were named in L1, of which 40.12% (SD = 16.16) were switch trials. Of the items named in L2, 36.95% (SD = 16.87) were switch trials. For RTs analyses, we excluded outliers by first discarding RTs below 200 ms, and then removing RTs more than 2.5 SD above or below the mean (of the RTs per participant and condition; 4.7% of correct trials).
The significant four-way Language × Trial type × Positive × Task interaction indicated that the influence of positive state on switching cost asymmetry differed across cued and voluntary naming (E = 0.048, t = 2.13, p < .05). Under the baseline Neutral, Cued, the Language × Trial type × Positive parameter was significant (E = −0.036, t = −2.08, p < .05), suggesting that cued switching cost asymmetry was significantly modulated by positive state, with smaller cued switching cost asymmetry in positive state (Language × Trial type parameter when setting the baseline to Positive, Cued: E = −0.008, t = −0.68, p = .50) than in neutral state (E = 0.027, t = 2.18, p < .05). By contrast, under the baseline Neutral, Voluntary, the three-way Language × Trial type × Positive parameter was nonsignificant. No other emotional effect reached significance (see Table A.2 in the Supplementary Material).
In summary, we only found that cued vs. voluntary naming modulated the effect of positive state on switching cost asymmetry – positive state decreased switching cost asymmetry in cued naming but did not influence it in voluntary naming.
3.2.2. Cue-locked ERPs results
Figure 4 illustrates grand average waveforms time-locked to cue onset, and the distribution of the cue-locked language and switch-related modulation in N2 and LPC.
(A) Grand average waveforms time-locked to cue onset in prefrontal (F1, FZ, F2), fronto-central (FC1, FCZ, FC2), central (C1, CZ, C2), centro-parietal (CP1, CPZ, CP2) and parietal (P1, PZ, P2) medial electrodes. (B) The topographic plots showing the distribution of language modulation (left) and switch-related modulation (right) in cue-locked N2. Greater RLDE (L1 trials elicit a larger negativity than L2 trials) is reflected by warmer colors. Greater switching effect (switch trials elicit a larger negativity than nonswitch trials) is reflected by cooler colors. (C) The topographic plots showing the distribution of language modulation (left) and switch-related modulation (right) in cue-locked LPC. Greater RLDE (L1 trials elicit a reduced positivity than L2 trials) and switching effect (switch trials elicit a larger positivity than nonswitch trials) are reflected by warmer colors.

Cue-locked N2 mean amplitude. Table 2 shows the results for cue-locked N2 mean amplitude. The Language × Negative × Task parameter was significant, indicating that the influence of negative state on language effect differed across cued and voluntary naming. The significant Language × Negative parameter under the baseline Neutral, Cued suggested that negative state came with significantly greater RLDE compared with neutral state in cued naming (see Figure 4B; warmer colors indicate greater RLDE). Further analyses on cued naming data showed that this effect came from greater increase of N2 in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = –1.148, t = –12.45, p < .001) than in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = –0.759, t = –8.28, p < .001).
Results for cue-locked N2 mean amplitude

Note: ∗p < .05; ∗∗p < .01; ∗∗∗p < .001. Given that the effects of interest involving ROIs were nonsignificant, parameters including ROIs were not presented in the table for clarity.
Though Language × Negative parameter under the baseline Neutral, Voluntary was also significant, from Figure 4B we see that negative state yielded significantly reduced RLDE compared with neutral state in voluntary naming. Follow-up analyses on voluntary naming data revealed that the decrease in RLDE resulted from decrease of N2 in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = 0.733, t = 7.76, p < .001), rather than the modulation of N2 in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = 0.147, t =1.70, p = .09). Other emotional effects of interest were nonsignificant (ps > .09).
Cue-locked LPC mean amplitude. Table 3 presents the results for cue-locked LPC mean amplitude. The Language × Negative × Task parameter again reached significance. Though the Language × Negative parameter under the baseline Neutral, Cued was not significant, this parameter under the baseline Neutral, Voluntary reached significance, indicating that negative state incurred significantly decreased RLDE compared with neutral state in voluntary naming (see Figure 4C; warmer colors indicate greater RLDE). Further analyses on voluntary naming data revealed that the reduced RLDE was mainly driven by decreased positivity on L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = −0.358, t = −3.64, p < .001), with a slightly smaller contribution from increased positivity on L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = 0.340, t = 3.16, p < .01).
Results for cue-locked LPC mean amplitude

Note: ∗p < .05; ∗∗p < .01; ∗∗∗p < .001. Given that the effects of interest involving ROIs were nonsignificant, parameters including ROIs were not presented in the table for clarity.
Moreover, the Trial type × Negative × Task parameter was significant, revealing that the influence of negative state on switching effect differed across cued and voluntary naming. Under the baseline Neutral, Cued, the Trial type × Negative parameter was nonsignificant. However, under the baseline Neutral, Voluntary, the Trial type × Negative parameter reached significance, indicating that negative state significantly enhanced voluntary switching effect relative to neutral state (see Figure 4C; warmer colors indicate greater switching effect). Follow-up analyses of voluntary naming data revealed that this effect was mainly attributed to the enhancement of the positivity in switch trials (Negative parameter when setting the baseline to Neutral, Switch: E = 0.517, t = 4.32, p < .001), rather than the reduction of LPC in nonswitch trials (Negative parameter when setting the baseline to Neutral, Nonswitch: E = −0.359, t = −3.93, p < .001).
The Language × Trial type × Negative × Task parameter also reached significance. The three-way Language × Trial type × Negative interaction in cued naming was significant, but this interaction in voluntary naming was not significant (see the Language × Trial type × Negative parameters under the baseline Neutral, Cued and Neutral, Voluntary, respectively). Follow-up analyses of cued naming data revealed that the three-way interaction stemmed from greater influence of negative state on the difference in LPC between L1 and L2 switch trials (Language × Negative parameter when setting the baseline to Neutral, Switch: E = −0.956, t = −3.20, p < .01) compared with nonswitch trials (Language × Negative parameter when setting the baseline to Neutral, Nonswitch: E = 0.592, t = 3.45, p < .001). Further analyses on switch trials in cued naming showed that the emotional effect on the language difference across switch trials mainly came from the decrease of LPC in L2 switch trials (Negative parameter when setting the baseline to Neutral, L2: E = −2.122, t = −10.06, p < .001), rather than the modulation of LPC in L1 switch trials (Negative parameter when setting the baseline to Neutral, L1: E = −1.147, t = −5.41, p < .001). No other emotional effect of interest reached significance (ps > .05).
3.2.3. Stimulus-locked ERPs results
Figure 5 presents grand average waveforms time-locked to stimulus onset, and the distribution of the stimulus-locked language and switch-related modulation in N2 and LPC.
(A) Grand average waveforms time-locked to stimulus onset in anterior-central medial electrodes (F3, F1, FZ, F2, F4, FC3, FC1, FCZ, FC2, FC4, C3, C1, CZ, C2 and C4). (B) The topographic plots showing the distribution of language modulation (left) and switch-related modulation (right) in stimulus-locked N2. Greater RLDE (L1 trials elicit a larger negativity than L2 trials) is reflected by warmer colors. Greater switching effect (switch trials elicit a larger negativity than nonswitch trials) is reflected by cooler colors. (C) Grand average waveforms time-locked to stimulus onset in centro-posterior medial electrodes (F3, F1, FZ, F2, F4, FC3, FC1, FCZ, FC2, FC4, C3, C1, CZ, C2 and C4). (D) The topographic plots showing the distribution of language modulation (left) and switch-related modulation (right) in stimulus-locked LPC. Greater RLDE (L1 trials elicit a reduced positivity than L2 trials) and switching effect (switch trials elicit a larger positivity than nonswitch trials) are reflected by warmer colors.

Stimulus-locked N2 mean amplitude. Table 4 presents the results for stimulus-locked N2 mean amplitude. The significant Language × Negative × Task parameter suggested that the influence of negative state on language difference differed across cued and voluntary naming. The significant Language × Negative parameter under the baseline Neutral, Cued suggested that negative state incurred significantly greater RLDE compared with neutral state in cued naming (see Figure 5B; warmer colors indicate greater RLDE). Further analyses of cued naming data showed that this effect was mainly due to enhanced amplitudes in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = −0.600, t = −6.73, p < .001), rather than the decrease of N2 in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = 0.203, t = 2.28, p < .05).
Results for stimulus-locked N2 mean amplitude

Note: ∗p < .05; ∗∗p < .01; ∗∗∗p < .001.
a When setting the baseline to Neutral, Cued, the Language × Positive × Task parameter was significant (E = −1.144, t = −2.09, p < .05). However, when setting the baseline to Neutral, Voluntary, this parameter became nonsignificant (E = 1.056, t = 1.96, p = .055). Accordingly, we argue that the influence of positive state on language modulation in stimulus-locked N2 reflected by this parameter is not statistically robust.
Though Language × Negative parameter under the baseline Neutral, Voluntary was also significant, from Figure 5B we see that negative state yielded significantly reduced RLDE compared with neutral state in voluntary naming. Follow-up analyses of voluntary naming data revealed that this effect stemmed from the decrease of the negativity in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = 0.549, t = 6.03, p < .001), not in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = 0.136, t = 1.63, p = .10). Other emotional effects of interest were nonsignificant (ps > .05).
Stimulus-locked LPC mean amplitude. Table 5 shows the results for stimulus-locked LPC mean amplitude. The Language × Negative × Task parameter was again observed to be significant, suggesting a difference in the influence of negative state on language modulation in LPC across cued and voluntary naming. The significant Language × Negative parameter under the baseline Neutral, Cued suggested that negative state significantly enhanced RLDE compared with neutral state in cued naming (see Figure 5D; warmer colors indicate greater RLDE). Follow-up analyses of cued naming data revealed that this was mainly attributed to the decrease of LPC in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = −1.523, t = −13.65, p < .001), rather than the modulation of LPC in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = −1.030, t = −9.26, p < .001).
Results for stimulus-locked LPC mean amplitude

Note: ∗∗p < .01; ∗∗∗p < .001.
Language × Negative parameter under the baseline Neutral, Voluntary was also significant, but from Figure 5D we see that negative state significantly reduced RLDE compared with neutral state in voluntary naming. Follow-up analyses of voluntary naming data revealed that this was mainly driven by the increase of LPC in L1 trials (Negative parameter when setting the baseline to Neutral, L1: E = 0.518, t = 4.53, p < .001), rather than the modulation of LPC in L2 trials (Negative parameter when setting the baseline to Neutral, L2: E = −0.354, t = −3.38, p < .001).
In addition, the Trial type × Negative × Task parameter reached significance, revealing a difference in the influence of negative state on switching effect across cued and voluntary naming. Under the baseline Neutral, Cued, the Trial type × Negative parameter was significant, indicating that negative state significantly enhanced cued switching effect relative to neutral state (see Figure 5D; warmer colors indicate greater switching effect). Follow-up analyses of cued naming data revealed that this was mainly attributed to the decrease of LPC in nonswitch trials (Negative parameter when setting the baseline to Neutral, Nonswitch: E = −1.434, t = −15.82, p < .001), rather than the modulation of LPC in switch trials (Negative parameter when setting the baseline to Neutral, Switch: E = −0.788, t = −4.94, p < .001). By contrast, the Trial type × Negative parameter under the baseline Neutral, Voluntary failed to reach significance. None of the other emotional effects of interest were significant (ps > .11).
In summary, in cue- and stimulus-locked N2 and stimulus-locked LPC, negative state elicited greater RLDE in cued naming (mainly by enhancing N2 and reducing LPC in L1 trials), but came with smaller RLDE in voluntary naming (mainly by reducing N2 and enhancing LPC in L1 trials). In cue-locked LPC, negative state increased the switching effect (mainly by enhancing LPC in switch trials) and reduced RLDE (primarily driven by decreased LPC on L2 trials, with a slightly smaller contribution from increased LPC on L1 trials) in voluntary naming, while it decreased LPC on L2 switch trials in cued naming. In addition, though negative state increased the cued switching effect in stimulus-locked LPC, this effect was mainly attributed to the decreased LPC on nonswitch trials.
4. Discussion
It is unclear how the language control system adapts to the social contexts under emotional states. Jiang et al. (Reference Jiang, Meng and Chen2024) proposed that the key difference in adaptive control between non-emotional versus emotional contexts lies in the compensatory mechanism. The current study aims to identify more fine-grained evidence for this adaptive compensatory mechanism using ERPs.
The results reveal that positive state reduced behavioral switching cost asymmetry to a larger extent for cued than voluntary naming. Most importantly, though negative state did not affect behavioral performances in either task, it incurred distinct patterns of ERPs modulations across the two naming tasks.
4.1. Adaptive compensatory adjustment in language control under negative state
We hypothesized that compensatory adjustments should be triggered by negative state to a larger extent in cued than voluntary naming across both phases. Thus, we expected that negative state should enhance the switching effect and RLDE in both cue- and stimulus-locked N2/LPC to a greater extent than in voluntary switching, thereby preserving behavioral performance. Of note, the enhanced RLDE should mainly lie in L1 trials, and the enhanced switching effect should mainly stem from switch trials.
Consistent with our hypothesis, during the early part of the language schemas competition phase and throughout the lexical selection phase, negative state triggered a compensatory mechanism recruiting greater proactive control to a larger extent in cued than voluntary naming. Specifically, in both cue- and stimulus-locked N2, as well as in stimulus-locked LPC, negative state elicited greater RLDE in cued naming (mainly by enhancing N2 and reducing LPC in L1 trials), which was coupled with preserved behavioral performance. Greater RLDE in N2/LPC indicates enhanced proactive control, and the pattern of enhanced N2 and reduced LPC in L1 trials further suggests that the locus of enhanced proactive inhibition lies in L1 (Kang et al., Reference Kang, Ma and Guo2018; Timmer et al., Reference Timmer, Christoffels and Costa2019; Zhang et al., Reference Zhang, Li, Ma, Kang and Guo2021). By contrast, in voluntary naming, negative state decreased RLDE across these ERPs components (mainly by reducing N2 and enhancing LPC in L1 trials), suggesting attenuated proactive inhibition of L1.
In the following paragraphs, we will discuss findings that contradict our hypothesis in more detail. The first is the absence of emotional disruption in behavioral performance during voluntary naming. We speculate that this might be due to the compensatory effort mobilization in this naming context. This interpretation is supported by the increased cue-locked LPC switching effect under negative state during voluntary naming, suggesting that negative state triggered a compensatory mechanism recruiting greater reactive control during the late part of the language schemas competition phase. This result is inconsistent with the finding in Jiang et al. (Reference Jiang, Meng and Chen2024) that negative state reduced the voluntary RLDE in RTs. One explanation for this discrepancy between the two studies lies in the task design for voluntary naming. Specifically, in Jiang et al. (Reference Jiang, Meng and Chen2024), participants were required to use the word that came to mind first upon seeing the picture stimulus. By contrast, in this study, participants had to select a language and maintain their choice before the picture stimulus appeared, which might increase the competition between languages. In this case, the voluntary naming in the present study may have imposed increased demand for strict language control, particularly during the language schemas competition phase, thereby enhancing the need to compensate for the potential impairment of control.
Notably, in voluntary naming, evidence for enhanced (reactive) control during the language schemas competition phase emerged only in the later part of this phase, as indicated by the cue-locked LPC, but not in the earlier part, as indexed by the cue-locked N2. Given the limited research on language control that dissociates the early cue-locked N2 from the late cue-locked LPC, we tentatively suggest that this pattern may be due to the switch-related modulation of N2 – typically associated with reactive control involved in language switching when time-locked to the stimulus (Peeters & Dijkstra, Reference Peeters and Dijkstra2018) – being affected by processes related to cue feature changes when time-locked to the cue (Karayanidis et al., Reference Karayanidis, Jamadar, Ruge, Phillips, Heathcote and Forstmann2010), which are not relevant in voluntary naming. On the contrary, the switch-related modulation of the cue-locked LPC reflects reactive control processes involved in task set reconfiguration (Nicholson et al., Reference Nicholson, Karayanidis, Bumak, Poboka and Michie2006), thereby exhibiting the compensatory adjustments in top-down control.
The second finding that seems to contradict our hypothesis concerns the interaction between negative state and switching context in cue-locked LPC. Specifically, in voluntary naming, negative state increased the cue-locked LPC switching effect primarily by enhancing LPC in switch trials. However, in cued naming, negative state decreased cue-locked LPC on L2 switch trials. Larger switch-related modulation of LPC indicates greater reactive control (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Kang et al., Reference Kang, Ma and Guo2018); thus, these results suggest that the compensatory mechanism mobilizing greater reactive control was more strongly activated in voluntary than cued naming during the late part of the language schemas competition phase.
Of note, we also found a decreased positivity on L2 trials but increased positivity on L1 trials in cue-locked LPC during voluntary naming, resulting in reduced RLDE – a pattern not present in cued naming. While previous studies typically accounted for the decreased RLDE in LPC with reduced inhibition of L1 (Timmer et al., Reference Timmer, Christoffels and Costa2019), our findings suggest a different mechanism. In the current study, the decreased RLDE in cue-locked LPC was attributed not only to increased LPC in L1 trials, but more substantially to decreased LPC in L2 trials.
As discussed in the Introduction, reduced language modulation of LPC can be explained in terms of greater difficulty and less available cognitive resources for language processing. Therefore, we speculate that the decrease in LPC on L2 trials suggests greater difficulty in L2 processing. However, it remains unclear whether the greater difficulty in L2 processing is due to enhanced inhibition or reduced activation of L2 (see Declerck, Reference Declerck2020; Declerck & Koch, Reference Declerck and Koch2023, for a discussion of proactive activation of L2). Future research may delve into the specific mechanism underlying the modulation in LPC for L2 trials.
The third finding in disagreement with our hypothesis is that negative state increased the cued switching effect in stimulus-locked LPC, mainly by reducing LPC in nonswitch trials. Given that reactive control is typically engaged in switch trials (Jackson et al., Reference Jackson, Swainson, Cunnington and Jackson2001; Kang et al., Reference Kang, Ma, Li, Kroll and Guo2020), one would have expected that if the increased cued switching effect in LPC reflected the compensatory mobilization of greater reactive control, heightened control should be exerted on switch trials and enhanced LPC in switch trials should be observed. However, the observed increase in the switching effect was driven by reduced LPC in nonswitch trials. Notably, several studies have reported an enlarged N2 in nonswitch rather than switch trials. For example, Chang et al. (Reference Chang, Xie, Li, Wang and Liu2016) found that nonswitch trials exhibited a larger N2 than switch trials. They attributed this effect to a switching bias in mixed-language blocks: when participants unexpectedly needed to remain in the same language in the subsequent trial, the previously activated alternative language had to be suppressed, resulting in increased cross-language conflict and a greater need for control. Though they found a switch-related modulation of N2, instead of LPC, in nonswitch trials, we tentatively propose that the reduced LPC in nonswitch trials also reflects decreased top-down control on these trials, because LPC amplitudes typically increase with greater top-down control demands in the case of switch-related effects. On this account, the third finding suggests that the reactive control (in nonswitch trials) during the late part of the lexical selection phase in cued naming was weakened under negative than neutral state.
To summarize, the compensatory mechanism was triggered in both naming contexts. However, during the lexical selection phase, compensatory adjustment in proactive control was activated by negative state in cued rather than voluntary naming. During the language schemas competition phase, compensatory adjustment in proactive control occurred during the early stage of this phase in cued naming, whereas compensatory adjustment in reactive control was activated during the late stage of this phase in voluntary naming. Since proactive control is more cognitively demanding than reactive control (Braver, Reference Braver2012), this finding suggests that compensatory adjustment in control during the language schemas competition phase was more strongly triggered in cued naming. Overall, the pattern of results reveals that the compensatory mechanism was more strongly triggered in cued than voluntary naming across both phases, which is in line with our hypothesis.
4.2. Facilitative effect of positive state on language control
The behavioral results reveal that positive state decreased switching cost asymmetry to a larger extent in cued than voluntary naming. Reduced switching cost asymmetry has been taken as evidence for more efficient reactive control (Liu et al., Reference Liu, Liang, Dunlap, Fan and Chen2016); hence, this finding suggests that positive state facilitates reactive control to a higher degree in cued than in voluntary naming.
The more pronounced facilitative emotional effect in cued than in voluntary naming suggested that in emotional contexts, the control system might function as initially proposed in the ACH (i.e., cognitive effort account in Jiang et al., Reference Jiang, Meng and Chen2024). That is, cued naming engages more top-down control than voluntary naming, so facilitative emotional effects became greater in the former context. Consistent with this account, our data show that the behavioral switching cost asymmetry in neutral state is different across naming contexts, with larger switching cost asymmetry in cued (E = 0.027, t = 2.18, p < .05) than in voluntary naming (E = −0.011, t = −1.00, p = .32), confirming that the demand for reactive control is higher in cued than in voluntary naming. Note that compensatory effort mobilization can sometimes lead to enhanced behavioral performance (Hayes et al., Reference Hayes, MacLeod and Hammond2009; Siniatchkin et al., Reference Siniatchkin, Averkina and Gerber2006). Thus, the more pronounced facilitative effect of positive state in cued naming may also be attributed to heightened activation of compensatory mechanisms in this context. However, there was no neural evidence supporting this adaptive compensatory mechanism under positive state in this study. Further research is needed to disentangle the two possibilities underlying the greater facilitative effect of positive state in cued than in voluntary naming.
It should be noted that though the facilitative effect of positive state in the present study lies in reactive control (in cued switching), Jiang et al. (Reference Jiang, Meng and Chen2024) reported a facilitative effect on proactive control (across cued switching, voluntary switching and single-language contexts), as evidenced by faster responses in positive than in neutral state. The discrepancy may be explained with the difference in the feasibility of applying proactive control in naming tasks. Specifically, relative to the three naming tasks in Jiang et al. (Reference Jiang, Meng and Chen2024), the cued naming task in the present study might be more difficult, probably because of the more complex task design (e.g., all participants in the present study completed voluntary naming that included a separate language choice phase and two types of filler trials, in addition to the cued naming and flanker task), and thus less cognitive resources were available for language control. The higher difficulty for the (cued) naming task in the present study than the three naming tasks in Jiang et al. (Reference Jiang, Meng and Chen2024) is confirmed by a post hoc analysis,Footnote 6 which showed that the responses in neutral state were faster in the latter. Thus, it might be less feasible to improve the proactive control, which is cognitively demanding (Braver, Reference Braver2012; Moser et al., Reference Moser, Moran, Schroder, Donnellan and Yeung2013), in (cued) naming task in the present study than in the three naming tasks in Jiang et al. (Reference Jiang, Meng and Chen2024). In this case, it is more likely to observe the facilitative emotional effects on proactive control in the latter and on reactive control in the former. Accordingly, we argue that whether emotional states influence proactive or reactive language control may vary as a function of the task implementations. Future research should identify the specific task implementations that modulate the locus of emotional effects on the proactive–reactive continuum.
4.3. Theoretical implications
The present study offers a fine-grained examination of the adaptive compensatory mechanism in the regulation of language switching performance under negative states. Our findings extend the ACH in at least two aspects.
Firstly, the ACH does not explicitly incorporate a compensatory mechanism; thus, it would predict that cued language switching performance should be more severely impaired than voluntary switching when the control system faces interference. However, recent behavioral evidence (Jiang et al., Reference Jiang, Meng and Chen2024) demonstrated that cued switching performance is less impaired by negative state than voluntary switching. In the present ERPs study, we further found that compensatory mechanisms were more strongly activated in cued switching across both phases of language control. These findings go beyond the predictions of ACH by revealing the resilience of the control system in the face of (emotional) interference, suggesting that the system can dynamically adjust to compensate for disruptions rather than simply being impaired.
Secondly, the compensatory mechanism increases the likelihood of detecting the adaptability to different social contexts proposed in the ACH. For example, in the present study, the interaction between Language and Task in the neutral condition was not significant in the stimulus-locked N2 and LPC. This result might suggest comparable proactive control processes during the lexical selection phase for cued and voluntary switching, which could be interpreted as evidence against the adaptability. Nevertheless, the presence of adaptive compensatory adjustments in proactive control throughout the lexical selection phase provides clear evidence for the adaptability of the control system. Thus, studies ignoring the role of compensatory mechanisms may underestimate the adaptation in the control system (e.g., Kennis et al., Reference Kennis, Zheng, de Bruin and Piai2025). Future research may pay more attention to the compensatory mechanism in emotional or other challenging contexts to fully uncover the adaptability of the bilingual mind.
4.4. Limitations
One limitation of the present study is that participants were required to choose one language prior to stimulus picture onset in voluntary naming, since we compared the voluntary and cued naming in a fine-grained manner by adopting the cue-stimulus paradigm. It might be argued that the comparison between cued and voluntary switching in this study did not fully capture the distinction between dual-language and dense code-switching contexts as described in the ACH, because voluntary naming in our task might not engage the same mechanisms (e.g., bottom-up processes related to lexical access in the case of opportunistic planning) as the dense code-switching context. However, in our design, cued and voluntary switching differed in two key respects: whether language switching is driven by an external cue, and the amount of cognitive effort involved (i.e., greater efforts are engaged in cued than in voluntary switching, as evidenced by, for example, a larger behavioral switching cost asymmetry in neutral state for cued switching). These differences align with those distinguishing the dual-language and dense code-switching contexts in the ACH. Therefore, we argue that the cued vs. voluntary switching comparison in the present study can, at least to some extent, reflect the contrast between dual-language and dense code-switching contexts. Future neuroimaging studies should replicate our findings using a voluntary naming task where no separate language choice phase is set and the bottom-up processes are triggered to a larger extent. In addition, the present study only compared the cued vs. voluntary naming, leaving out the single-language context. Future neuroimaging research may compare the single-language and switching contexts to determine whether the observed adaptive compensatory mechanism is specific to the cued vs. voluntary switching comparison only.
Moreover, in this study, positive state facilitated performance without clear neural signatures of altered language control. This highlights the fact that neural and behavioral measures do not always converge. Previous research has also reported significant behavioral effects in the absence of significant ERPs effects (Kennis et al., Reference Kennis, Zheng, de Bruin and Piai2025). This discrepancy may arise because the behavioral effect is driven by processes not reflected in the ERPs components being measured. While this study focused on N2 and LPC amplitudes, the two most commonly examined measures in bilingual language control research, positive state may have facilitated processes not captured by these measures. Future research should delve into the specific processes that contribute to the facilitative effects of positive state on overt performance.
In addition, given the complexity of our experimental design, particularly with the inclusion of emotion manipulation, we used repeated presentation of a small set of 13 target pictures. This approach would reduce the inaccessibility of nondominant language names (Gollan et al., Reference Gollan, Kleinman and Wierenga2014) and thus the cognitive load required for naming. Excessive cognitive efforts involved in cognitive tasks can limit resources available for emotion processing, potentially diminishing reactivity to emotional information (Yates et al., Reference Yates, Ashwin and Fox2010) and thereby the effectiveness of emotion induction. Future research may consider using a larger set of pictures to increase lexical variety and ecological validity.
Furthermore, our findings may reflect the dynamics of less proficient bilinguals with moderate foreign language anxiety, rather than bilinguals more broadly. Increased proficiency has been proposed to shift reliance from top-down control to language-specific selection mechanisms during lexical selection (Costa & Santesteban, Reference Costa and Santesteban2004). On this account, highly proficient bilinguals may be able to focus their lexical selection in the target language without relying on top-down inhibition of nontarget language; thus, emotional effects on control processes may be attenuated or even absent in this group. Moreover, individuals with high foreign language anxiety have been shown to perform worse in L2 speech production (Zhang, Reference Zhang2019), which has been linked to inefficiency in shifting attention to relevant information and maintaining focus on it (Li et al., Reference Li, Yu, Guan, Li and Luo2024). Hence, compensatory recruitment in this group may be diminished or reduced by their inefficient attentional allocation or because their motivation to mobilize resources is downplayed in naming tasks involving difficult L2 production (Berggren & Derakshan, Reference Berggren and Derakshan2013). Further studies should examine how individual differences in proficiency and anxiety modulate the emotional effects on language control.
5. Conclusion
The present study seeks more fine-grained evidence for adaptive compensatory adjustments in control processes in emotional contexts using the ERPs technique. The results revealed that though compensatory mobilization in reactive control during the late part of the language schemas competition phase was elicited by negative state in voluntary naming, compensatory increases in proactive control during the early part of the language schemas competition phase and throughout the lexical selection phase were triggered in cued naming. Accordingly, we conclude that the compensatory mechanism is more strongly triggered across both language control phases in cued than in voluntary naming. Our findings extend the ACH to more naturalistic emotional settings and help to uncover the adaptability of the bilingual mind.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S1366728926101369.
Data availability statement
The data and analysis code are available via OSF at https://osf.io/z9prq.
Acknowledgments
This work was supported by the Fundamental Research Funds for the Central Universities (2024111045) and the Shanghai Philosophy and Social Science Planning Project (2025EYY006) to S.J. The authors thank the three reviewers for their valuable comments.
Competing interests
The authors declare none.