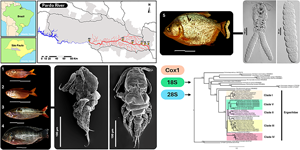
Introduction
Copepoda is a diverse monophyletic taxon of the subphylum Crustacea; it comprises over 14 000 valid species distributed throughout the world (Bernot et al., Reference Bernot, Boxshall and Crandall2021). These small crustaceans inhabit every type of aquatic habitat (i.e. from deep-sea communities to semi-terrestrial conditions such as damp moss/leaf litter in humid forests) and exhibit a wide range of body shapes with distinct life cycles (i.e. from free-living to obligate parasites) (Boxshall and Halsey, Reference Boxshall and Halsey2004). Parasitism appeared independently at least 14 times in the evolutionary history of this taxon and today over 5000 species are recognized as parasites (Bernot et al., Reference Bernot, Boxshall and Crandall2021).
Fourteen metazoan phyla, ranging from sponges to large cetaceans, act as hosts for 1 or more symbiotic stages of copepods (see Tables 3.8 and 3.9 in Boxshall and Hayes, Reference Boxshall, Hayes, Smit, Bruce and Hadfield2019). Fish (including Chondrichthyes and mainly Osteichthyes) stand out as one of the most studied host groups. In addition to a large number of known host–parasite associations with copepods (i.e. over 890 associations in the Neotropical region alone, see Luque et al., Reference Luque, Pavanelli, Vieira, Takemoto and Eiras2013), this group also includes several species that impact the economy as pests in aquaculture (Johnson et al., Reference Johnson, Treasurer, Bravo, Nagasawa and Kabata2004; Piasecki et al., Reference Piasecki, Goodwin, Eiras and Nowak2004).
In Brazil, copepods represent one of the most speciose taxa of fish ectoparasites comprising over 200 operational taxonomic units [since it includes both determined and undetermined species; see Table 1 in Luque et al. (Reference Luque, Pavanelli, Vieira, Takemoto and Eiras2013)]. In freshwater, the parasitic family Ergasilidae Burmeister, 1835 stands out as the most important due to its high diversity and harmful aquaculture potential (Piasecki et al., Reference Piasecki, Goodwin, Eiras and Nowak2004; Luque et al., Reference Luque, Pavanelli, Vieira, Takemoto and Eiras2013). These copepods are easily recognized by their prehensile antenna, the absence of maxillipeds in females and the morphology of mouthparts (Boxshall and Halsey, Reference Boxshall and Halsey2004). Most adult females (i.e. the only parasitic stage of their life cycle) can be frequently found attached to the gills, but they also occur inside nostrils, on the surface of the body, embedded into host tissues as mesoparasites or even as endoparasites inside the urinary bladder (Thatcher, Reference Thatcher2006; Tang and Kalman, Reference Tang and Kalman2008; Rosim et al., Reference Rosim, Boxshall and Ceccarelli2013).
Despite the importance of ergasilids in Brazil, several authors believe that the current biodiversity of these copepods is still underestimated (Thatcher, Reference Thatcher1998; Boxshall and Defaye, Reference Boxshall and Defaye2008; Luque et al., Reference Luque, Pavanelli, Vieira, Takemoto and Eiras2013). More than 90% of Brazilian fishes (>4000 spp.) have never been studied for their parasitic copepod fauna, leaving an enormous gap in the country for discoveries (Luque et al., Reference Luque, Pavanelli, Vieira, Takemoto and Eiras2013). During the parasitological survey of fishes from the Pardo River, São Paulo State, Brazil, copepods were found parasitizing the nostrils of 4 native and 1 introduced fish species: Astyanax lacustris (Lütken, 1875); Cyphocharax modestus (Fernández-Yépez, 1948); Psalidodon bockmanni (Vari and Castro, 2007); Serrasalmus maculatus Kner, 1858 and the introduced Coptodon rendalli (Boulenger, 1897). The use of scanning and light microscopy together with molecular and phylogenetic analyses revealed that these copepods belong to 2 distinct species of Ergasilidae: Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 and Gamispatulus schizodontis Thatcher and Boeger, Reference Thatcher and Boeger1984. Molecular data were provided (for the first time) for both copepods (COI mtDNA, 18S and 28S rDNA genes) and the newly generated sequences were used to generate a new phylogenetic hypothesis for this family. Careful analysis of present specimens of R. piranhus revealed the need to redescribe this species, which is also done here.
Materials and methods
Host sampling and morphological analysis of copepods
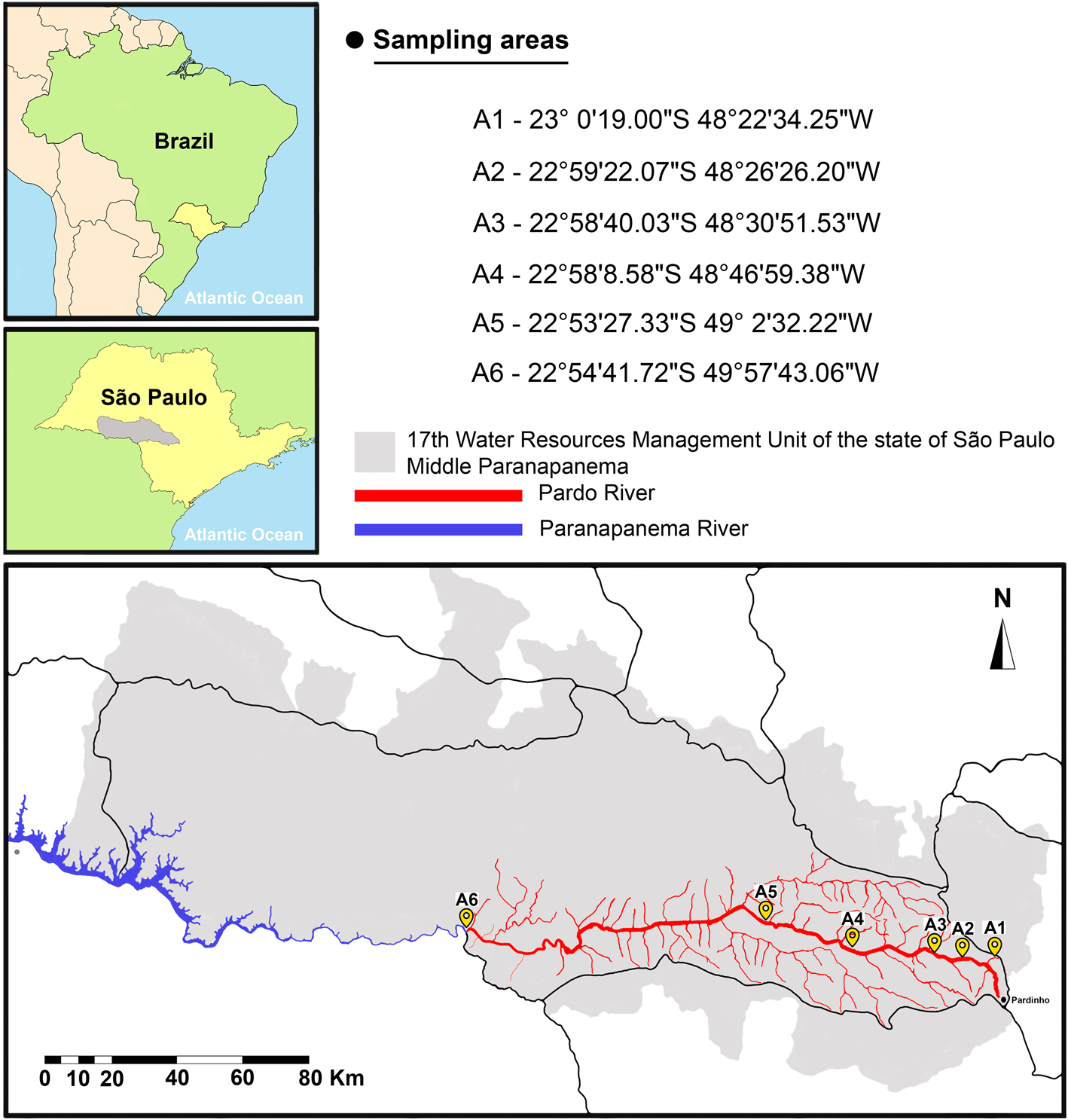
From June 2020 to December 2022, 354 fish from 5 different species were sampled along the Pardo River in São Paulo state, Brazil (Fig. 1). Fish were sampled with the aid of (1) multi-panel seine nets (3–14 cm mesh) soaked from 4 to 12 h, (2) cast nets, (3) fishing sieves and (4) fishing rods. Each fish was placed in a marked plastic bag and frozen for necropsy. In the laboratory, gills, surfaces, fins and nostrils of each fish were examined under a stereomicroscope for the presence of parasitic copepods. Fish sampling was authorized by the Instituto Chico Mendes de Conservação da Biodiversidade – ICMBio and Sistema de Autorização e Informação em Biodiversidade – SISBIO # 60640-1 and all procedures followed the recommendations of the Ethical Commission for Animal Experimentation from the São Paulo State University (Unesp), Institute of Biosciences, Botucatu, Brazil (CEUA no 9415260520). Fish were identified at the species level with the help of experts and the use of identification keys present in specialized literature (van der Sleen and Albert, Reference van der Sleen and Albert2017; Ota et al., Reference Ota, Deprá, Graça and Pavanelli2018; Ribeiro et al., Reference Ribeiro, Chagas, Thereza and Langeani2019). The scientific names of the fish cited throughout the text are in accordance with the nomenclature recommended in Eschmeyer's Catalogue of Fishes (Fricke et al., Reference Fricke, Eschmeyer and Van der Laan2024).
Sampling areas (A1 to A6) along the Pardo River, São Paulo state, Brazil.
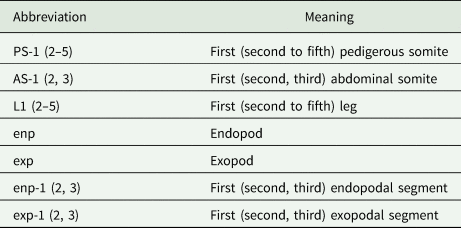
Copepods were carefully extracted from organs/tissues using fine needles and then preserved in glass vials with 70 and 96% ethanol for morphological and molecular analysis, respectively. For morphological identification, some copepods were cleared in lactic acid (10–30 min) and then mounted in Hoyer's medium. Whenever necessary, some specimens were dissected in glycerol medium and then each part was mounted on individual slides (e.g. antennules, antennae, swimming legs, etc.). Coverslips were sealed with transparent nail varnish. Morphological analysis and measurements of whole/dissected copepods were made using a compound microscope with differential interference contrast optics (Leica DMLB 5000, Leica Microsystems). Drawings were made with the aid of a compound microscope (LeicaDMLS, Leica Microsystems, Wetzlar, Germany) equipped with a drawing tube. All measurements are in micrometres (μm) and presented as the range followed by the mean in parenthesis. Morphological nomenclature used throughout the text to describe copepods was adapted from the nomenclature used by Boxshall and Halsey (Reference Boxshall and Halsey2004) and Damborenea et al. (Reference Damborenea, Rogers and Thorp2020). Abbreviations used for describing the copepod parts (e.g. body regions, segments and appendices) are summarized in Table 1. Ecological descriptors such as prevalence and mean intensity were calculated following Bush et al. (Reference Bush, Lafferty, Lotz and Shostak1997).
Abbreviations of body parts and segments used throughout the text to describe copepods
For scanning electron microscopy (SEM), copepods were dehydrated in a graded ethanol series to 100% ethanol and placed in hexamethyldisilazane. Dried specimens were mounted on carbon tape on aluminium stubs and sputter-coated with carbon (Emscope TB500, Quorum Technologies, Puslinch, ON, USA), followed by 20–30 nm gold/palladium (Eiko IB2 ion coater, Eiko, Japan). Micrographs were taken using an FEI Nova NanoSEM 450 scanning electron microscope (FEI, Hillsboro, OR, USA).
Type-material of R. piranhus (Cyclopoida: Ergasilidae) was burrowed from the Zoological Collection of the National Institute of Amazonian Research (INPA) (holotype INPA PA 309-1; paratypes INPA PA 309-2 to 309-5) (see Supplementary Fig. 1) and used for morphological comparison with present specimens. Regarding G. schizodontis, the specimens were compared with the illustrations and/or information present in the available literature (Thatcher and Boeger, Reference Thatcher and Boeger1984; Lacerda et al., Reference Lacerda, Takemoto, Lizama and Pavanelli2007; Narciso and Silva, Reference Narciso and Silva2020). Representative specimens of each species were deposited in the Zoological Collection of the Museum of Zoology of the University of São Paulo (MZUSP), municipality of São Paulo, São Paulo state, Brazil.
DNA extraction and PCR amplification
DNA extraction was performed using the posterior part of the body (=urosome) and/or the egg sacs of the copepods, except for R. piranhus, for which whole specimens were used due to their small size (<400 μm). When only a small portion of the specimens or the egg sacs was used for DNA extraction, the remainder (i.e. the entire copepod or the upper part of the body) was deposited as a hologenophore in the Zoological Collection of MZUSP. For R. piranhus, micrographs (i.e. photohologenophore) of each specimen used for DNA extraction were taken (see Supplementary Fig. 2).
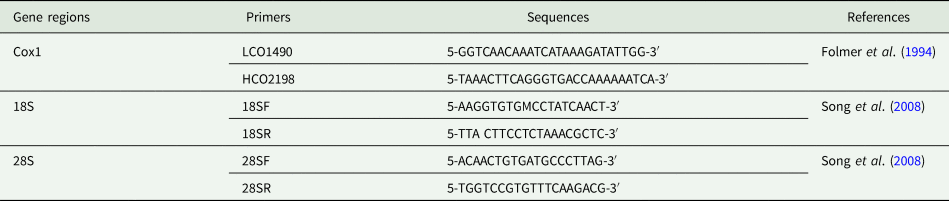
Genomic DNA was extracted from specimens fixed at 96% ethanol using the following kits: (1) NucleoSpin Tissue XS (Macherey-Nagel, Düren, Germany) Kit for whole/dissected copepods; and (2) PCRBiosystems Rapid DNA Extraction (London, England) Kit for egg sacs. Extractions followed the recommendations indicated in the manufacturer's protocol for each kit. Partial fragments of a mitochondrial gene (subunit I of cytochrome c oxidase – cox1) and 2 ribosomal genes (18S and 28S rDNA) were amplified using a set of primers available in the literature (summarized in Table 2). Polymerase chain reaction (PCR) was performed in 25 μm mixtures containing 22 μL of DreamTaq PCR Master Mix (comprising Dream Taq DNA Polymerase, optimized DreamTaq buffer, MgCl2 and dNTPs), 1.25 μL of each primer (forward and reverse) and 0.5 μL of genomic DNA under the following conditions: (i) 95°C for 5 min; (ii) 30 cycles of 95°C for 30 s; (iii) 47°C for 30 s for primer annealing; (iv) 72°C for 1 min; and (v) 72°C for 7 min for final extension (adapted from Song et al., Reference Song, Wang, Yao, Gao and Nie2008). The amplified PCR products were run on agarose gel (1%) stained with GelRedTM (Biotium Inc., Fremont City, CA, USA) and visualized with a UV transilluminator. Successful amplifications were sent for cleaning, purification and sequencing to Inqaba Biotechnical Industries (Pty) Ltd. (Pretoria, South Africa). Forward and reverse sequences were assembled, aligned and edited using Geneious 11.1.4 (Biomatters, Auckland, New Zealand). Novel sequence data have been deposited in GenBank (see Table 3).
List of primers (forward and reverse) used for DNA amplification of the partial mitochondrial (cox1) and ribosomal genes (18S and 28S rDNA) of parasitic ergasilids (Cyclopoida: Ergasilidae) found in the nostrils of fishes from Pardo River, São Paulo state, Brazil – including sequences and references
List of sequences from the partial mitochondrial (subunit I of cytochrome c oxidase or cox1) and ribosomal genes (18S and 28S rDNA) included in the phylogenetic analyses
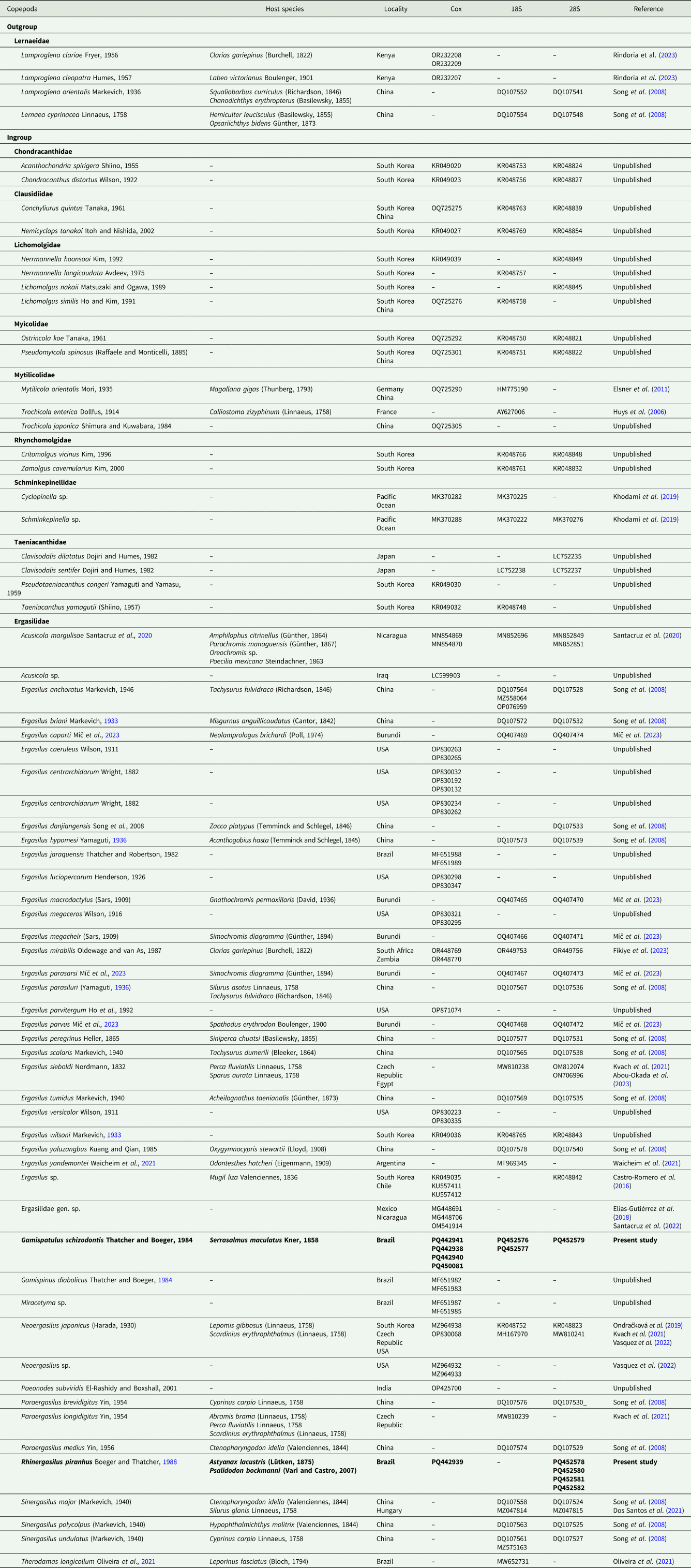
Taxa represented in bold fonts are the novel sequences provided in the present study.
DNA sequence alignment and phylogenetic reconstruction
The newly generated sequences of each gene (cox1, 18S and 28S) were assembled and inspected using ‘De Novo Assembly’ tool in Geneious Prime v2022.2. (https://www.geneious.com). The resulting consensus sequences were compared to available sequences using the Basic Local Alignment Tool (BLAST) (https://blast.ncbi.nlm.nih.gov/Blast.cgi, accessed on 2 January 2024) (Altschul et al., Reference Altschul, Gish, Miller, Myers and Lipman1990) to confirm the identity of the present specimens (if possible) and define their closest congeners. For all alignments, available sequences of the lernaeid copepods were chosen as the outgroup: Lernaea cyprinacea Hermann, 1783; Lamproglena clariae Fryer, 1956; and/or Lamproglena orientalis Markevich, 1936. Two distinct sequences of copepods were taken from 8 families of the Suborder Ergasilida and used herein as an internal group due to their phylogenetic proximity to Ergasilidae [see the phylogenetic tree present in Bernot et al. (Reference Bernot, Boxshall and Crandall2021)]. For ergasilids, all available sequences (maximum of 3 per species) were taken from the nucleotide database of the National Center for Biotechnology Information (NCBI) (https://www.ncbi.nlm.nih.gov/, accessed on 2 January 2024). Sequences shorter than 450 bp were excluded from the analyses in order to avoid the loss of informative characters. All sequences used in the analyses (including ergasilids, internal groups and outgroups) are summarized in Table 3. The alignments were made in Geneious using the default parameters of MAFFT (Katoh et al., Reference Katoh, Misawa, Kuma and Miyata2002). Extremes of each sequence were subsequently trimmed to an alignment of (i) 510 bp for cox1; (ii) 1121 bp for 18S; and (iii) 659 bp for 28S. Indels greater than 3 consecutive characters and affecting more than 5% of sequences were also removed from the final alignment. Only sequences of 18S and 28S were used for subsequent phylogenetic analyses. The best nucleotide substitution model test for maximum likelihood (ML) and Bayesian inference (BI) were determined using jModelTest v2.1.10 based on the Akaike information criterion (AIC) (Darriba et al., Reference Darriba, Taboada, Doallo and Posada2012). The chosen AIC model was GTR + I + G for all genes. The ML and BI analyses were run in RAxML v7.2.8 (Stamatakis, Reference Stamatakis2014) and MrBayes v3.2.2 (Ronquist et al., Reference Ronquist, Teslenko, Van Der Mark, Ayres, Darling, Höhna, Larget, Liu, Suchard and Huelsenbeck2012), respectively, via the CIPRES Science Gateway (available at https://www.phylo.org/) (Miller et al., Reference Miller, Pfeiffer and Schwartz2010). Nodal supports for ML analyses were estimated at 100 bootstrap replications and only nodes with values greater than 75% were considered as well-supported. The BI analyses were run for 10 million generations (ngen = 10 000 000) with 2 runs each containing 4 simultaneous Markov Chain Monte Carlo (MCMC) chains (nchains = 4) and with a sampling tree topology every 1000 generations (samplefreq = 1000). Burn-in was set to the first 25% of generations (Burninfrac = 0.25). Bayesian analyses used the following parameters: nst = 6, rates = invgamma, ngammacat = 4 and the priors' parameters of the combined dataset were set to ratepr = variable. Samples of substitution model parameters, and tree and branch lengths were summarized using the parameters ‘sump burnin = 3000’ and ‘sumt burnin = 3000’. Only nodes with posterior probabilities (pp) greater than 75% were considered as well-supported. The BI and ML trees were edited and visualized in FigTree v1.4.3 software (Rambaut, Reference Rambaut2012). Pairwise genetic distances (p-distances) were estimated in MEGA 7.0 (Kumar et al., Reference Kumar, Stecher and Tamura2016), and the number of base pair differences was calculated in Geneious.
Results
Two copepod species were found parasitizing the nostrils of 5 fish species from Rio Pardo (Fig. 2; Tables 4 and 5): (i) R. piranhus; and (ii) G. schizodontis. Rhinergasilus piranhus was found only in the first 2 sampling areas (close to the river spring) associated with both native and exotic fish. Gamispatulus schizodontis specimens were found inside the nostrils of a single host species, the native piranha S. maculatus – only at the river mouth (or A6 in Fig. 1).
Fish collected and analysed in this study, (A) Astyanax lacustris (Lütken, 1875), (B) Psalidodon bockmanni (Vari and Castro, 2007), (C) Cyphocharax modestus (Fernández-Yépez, 1948), (D) Coptodon rendalli (Boulenger, 1897), (E) Serrasalmus maculatus Kner, 1858. Scale bars = 5 cm.

Data from fish sampled along the Pardo River, São Paulo state, Brazil
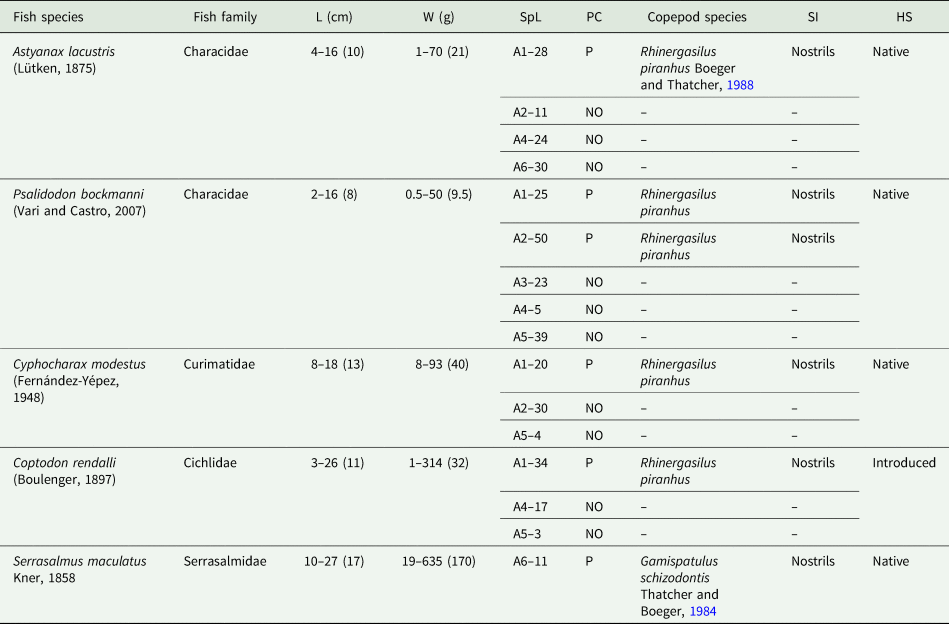
HS, host status on this river; L, total length in centimetres represented by the amplitude and the average in parentheses (cm); NO, not observed; P, present; SpL, fish specimens per locality; W, weight in grams represented by the amplitude and the average in parentheses (g).
A1 to A6 = sampling areas along the river.
Parasitological descriptors of ergasilids found in nostrils of fish from the Rio Pardo, São Paulo, Brazil

MA, average abundance of infestation; MI, mean intensity of infestation; N, number of fish analysed; P%, prevalence in percentage; R, range of parasites; s.e., standard error.
Calculations were made following the recommendations of Bush et al. (Reference Bush, Lafferty, Lotz and Shostak1997).
Morphological characterization
Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 (Figs 3–7; Tables 6 and 7)
Type host: Pygocentrus nattereri Kner, 1858 (Characiformes: Serrasalmidae).
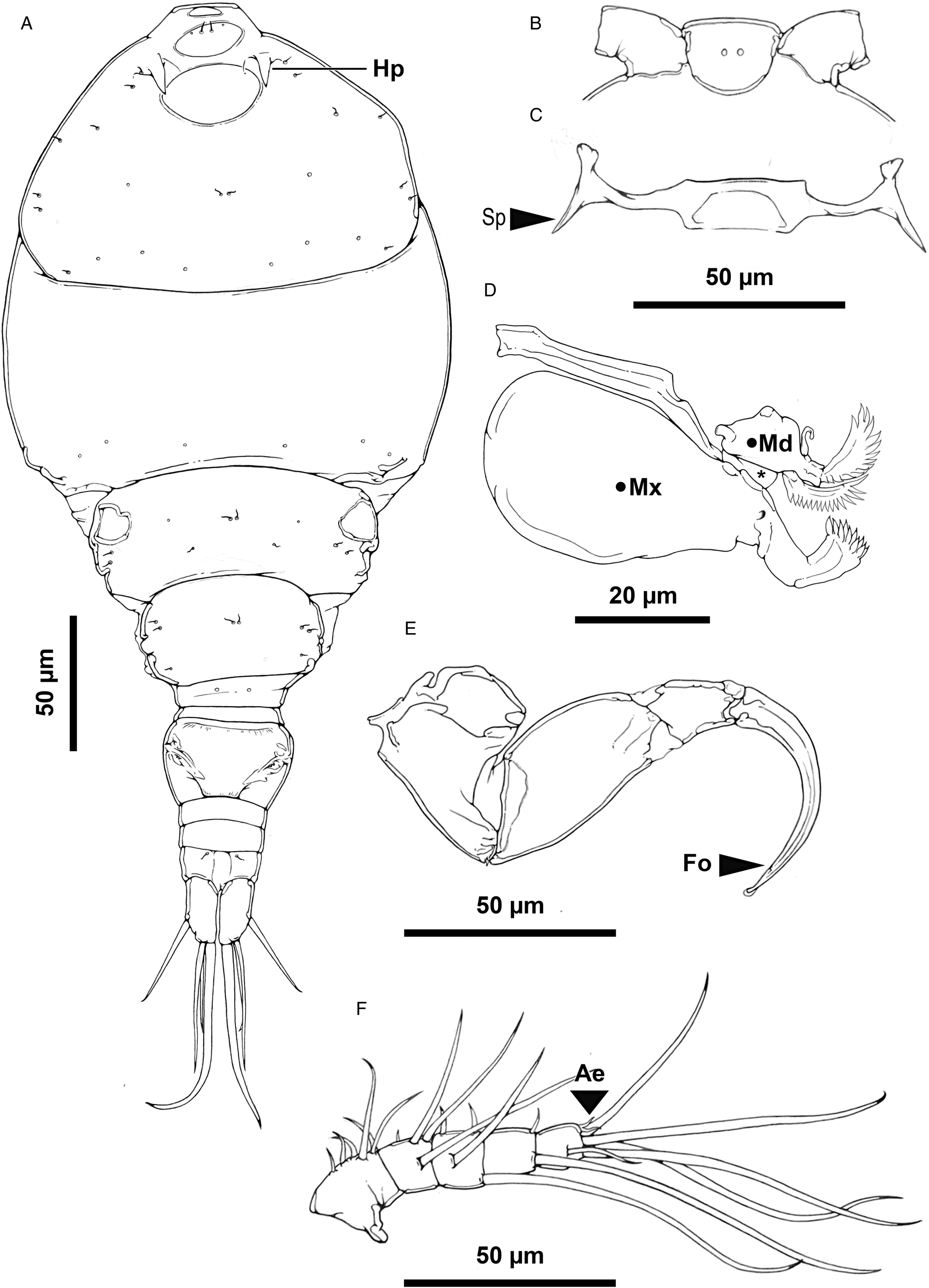
Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 – adult female, (A) body, dorsal view, cephalosome with a pair of horn-like projections (Hp), (B) rostrum, ventral view, (C) antennal support armed with lateral spine-like projections (Sp), (D) buccal apparatus comprising maxilla (Mx), maxillule (*) and mandible (Md), (E) antenna, claw with fossa (Fo) on concave margin, (F) antennule, distal segment 2 aesthetasc (Ae). Scale bars in micrometres (μm).
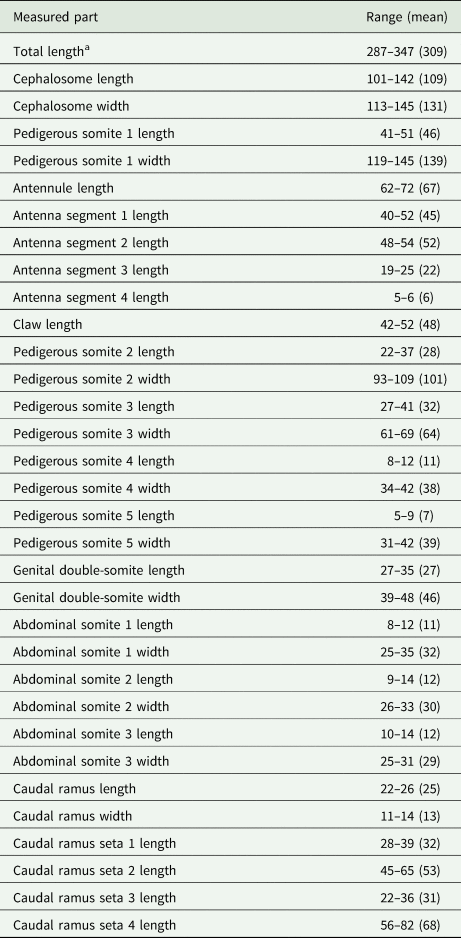
Measurements in micrometres (μm) of adult females of Rhinergasilus piranhus Boerger and Thatcher, Reference Boeger and Thatcher1988
a Excluding the length of the caudal setae.
Armature of swimming legs (L1 to L3) of Rhinergasilus piranhus Boerger and Thatcher, Reference Boeger and Thatcher1988 – adult female
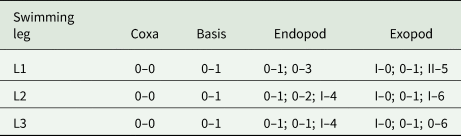
(Roman numeral = spines; Arabic numerals = setae).
Other hosts: Acestrorhynchus lacustris (Lütken, 1875) (Characiformes: Acestrorhynchidae); Acestrorhynchus falcirostris (Cuvier, 1819) (Characiformes: Acestrorhynchidae); A. lacustris (Lütken, 1875) (Characiformes: Characidae) – present study (PS); Colossoma macropomum (Cuvier, 1816) (Characiformes: Serrasalmidae); Co. rendalli (Boulenger, 1897) (Cichliformes: Cichlidae) – PS; C. modestus (Fernández-Yépez, 1948) (Characiformes: Curimatidae) – PS; Prochilodus nigricans Spix et Agassiz, 1829 (Characiformes: Prochilodontidae); P. bockmanni (Vari and Castro, 2007) (Characiformes: Characidae) – PS; and Serrasalmus altispinis Mercky, Jégu et Santos, 2000 (Characiformes: Serrasalmidae).
Type locality: Ilha da Marchantaria, Solimoes-Amazonas River near Manaus, Amazonas state, Brazil.
Sampled localities: Area 1 – initial section (close to the river spring) of the Pardo River located within the ‘Boa Vista’ Farm in the municipality of Pardinho, state of São Paulo, Brazil (22°59′59.8″S, 48°22′37.4″W); and area 2 – section of the Rio Pardo located within the ‘Paineiras’ Farm in the municipality of Pardinho, state of São Paulo, Brazil (22°59′22.1″S, 48°26′26.2″W).
Site of infestation: Nostrils.
Material examined: A total of 20 ♀♀ were examined for morphological description, all in nostrils, including (i) 7 ♀♀ from A. lacustris sampled in area 1 (4 ♀♀) and area 2 (3 ♀♀); (ii) 2 ♀♀ from Co. rendalli in area 2; (iii) 2 ♀♀ from Cy. modestus in area 2; and (iv) 9 ♀♀ from P. bockmanni in area 1 (2 ♀♀) and area 2 (7 ♀♀). Body and appendage measurements are listed in Table 6.
Additional material: 5 ♀♀ of the type-material of R. piranhus were borrowed from INPA's zoological collection and then used for morphological comparisons, including the holotype INPA PA 309-1 and 4 paratypes (INPA PA 309-2 to 309-5).
Representative DNA sequences: PQ442939, PQ452578, PQ452580-82.
Voucher material: A total of 20 whole ♀♀ (including 10 ♀♀ mounted in Hoyer's medium and 10 ♀♀ stored in glass vials with 70% ethanol) were deposited in the Zoological Collection of MZUSP (accession no. 46666-69, 46690, 46736, 46776, 46778).
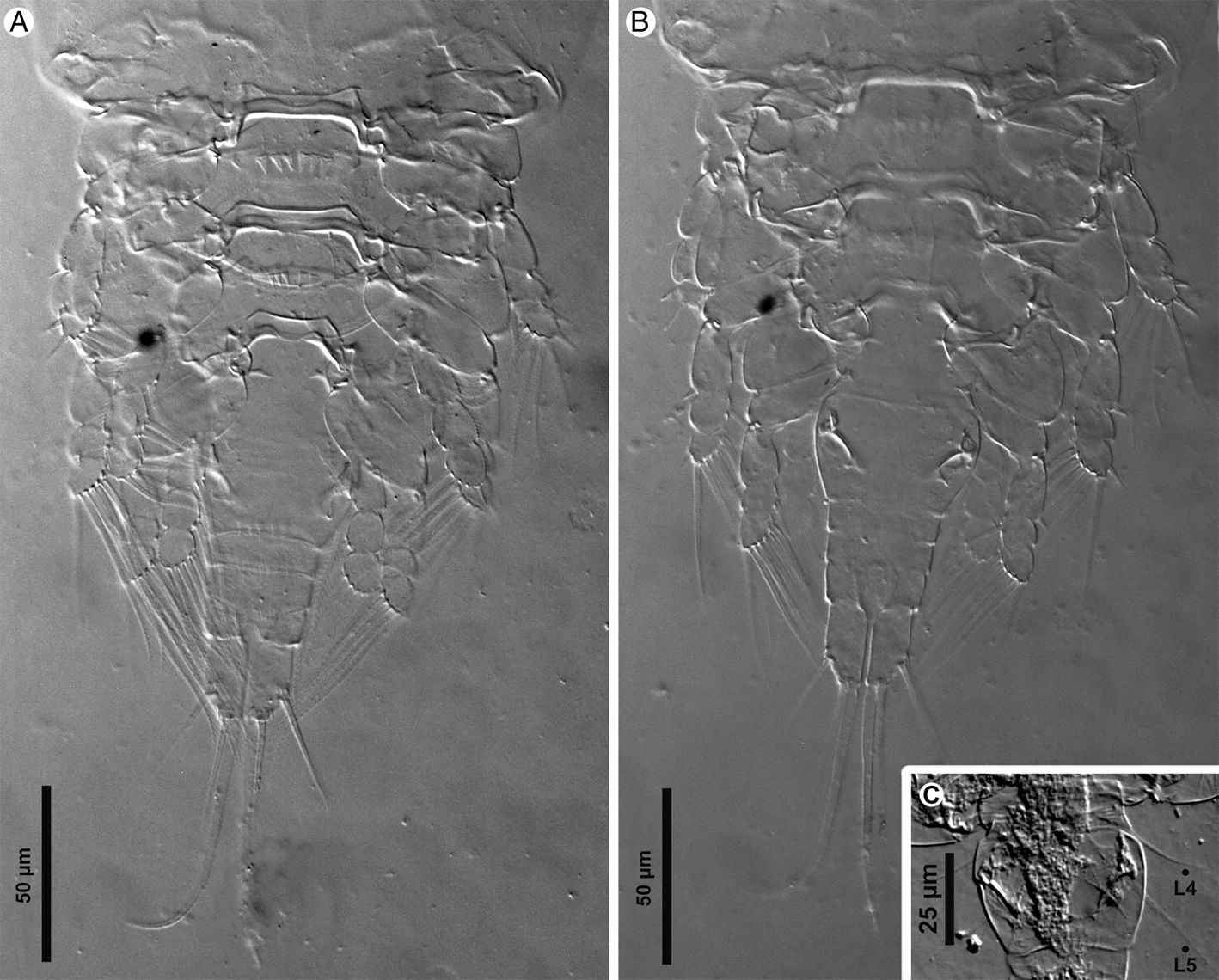
Redescription (based on adult females; male unknown): Body cyclopiform (Figs 3A and 5A), comprising prosome, urosome and caudal rami. Prosome consisting of cephalosome and PS-1; both segments separated dorsally by flexible cuticle; and 3 free pedigerous somites (PS-2 to PS-4). Cephalosome (Figs 3A and 5D) widening posteriorly, maximum width at level of buccal apparatus, anterior margin protruded, dorsal surface with 3 elliptical integumental windows (anterior, medial and posterior); posterior window with diameter about 2.0× times larger than previous window, with slightly rough surface (Figs 5E and 7H); armed with paired spine-like projections (Figs 5F and 7G), and ornamented with several pores symmetrically distributed throughout segment; pores with or without bristles. PS-1 (Figs 3A and 7C) with rounded lateral margins, ornamented with pores along posterior margin; pores lacking bristles. Rostrum (Figs 3B and 5E) with rounded posterior margin, ventrally ornamented with pair of pores. Free pedigerous somites (Figs 3A and 7B) decreasing gradually in width (on transverse axis) from anterior to posterior; each somite ornamented with 1 or more pairs of pores (Figs 3A and 7C); pores with or without bristles; PS-2 (Figs 3A and 7C) narrower than previous somite, with paired integumental windows laterally on tergite (arrowhead on Fig. 7C); PS-3 and PS-4, both lacking integumental windows; PS-4 reduced, smaller and thinner than previous prosome somites.
Urosome consisting of PS-5, genital double-somite and 3 free abdominal somites (AS-1 to AS-3). PS-5 (Figs 4A and 7C) reduced, similar in size to PS-4, unornamented. Genital double-somite (Fig. 4A) narrowing posteriorly, with rounded lateral margins, ornamented ventrally with posterior row of spinules. Abdominal somites (Figs 4A, 6B and 7E) decreasing gradually in width (on transverse axis) from anterior to posterior; each somite ornamented ventrally with posterior row of spinules; AS-3 (=anal somite) deeply incised posteriorly (=anal opening or anus), dorsal surface with a pair of pores carrying bristles located laterally to anus (highlighted in Fig. 7E).
Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 – adult female, (A) fourth pedigerous somite, urosome and caudal rami with setae (S1 to S4), ventral view: fourth and fifth somites with leg 4 (L4) and leg 5 (L5) each reduced to a single seta, respectively, (B) leg 1, (C) leg 2, (D) leg 3 with coxa ornamented with a patch of spinules near the posterior margin (dashed circle), (E) intercoxal sclerites (Isi to Isiii) and interpodal plates (Ipi to Ipiii), (F) egg sac. Scale bars in micrometres (μm).
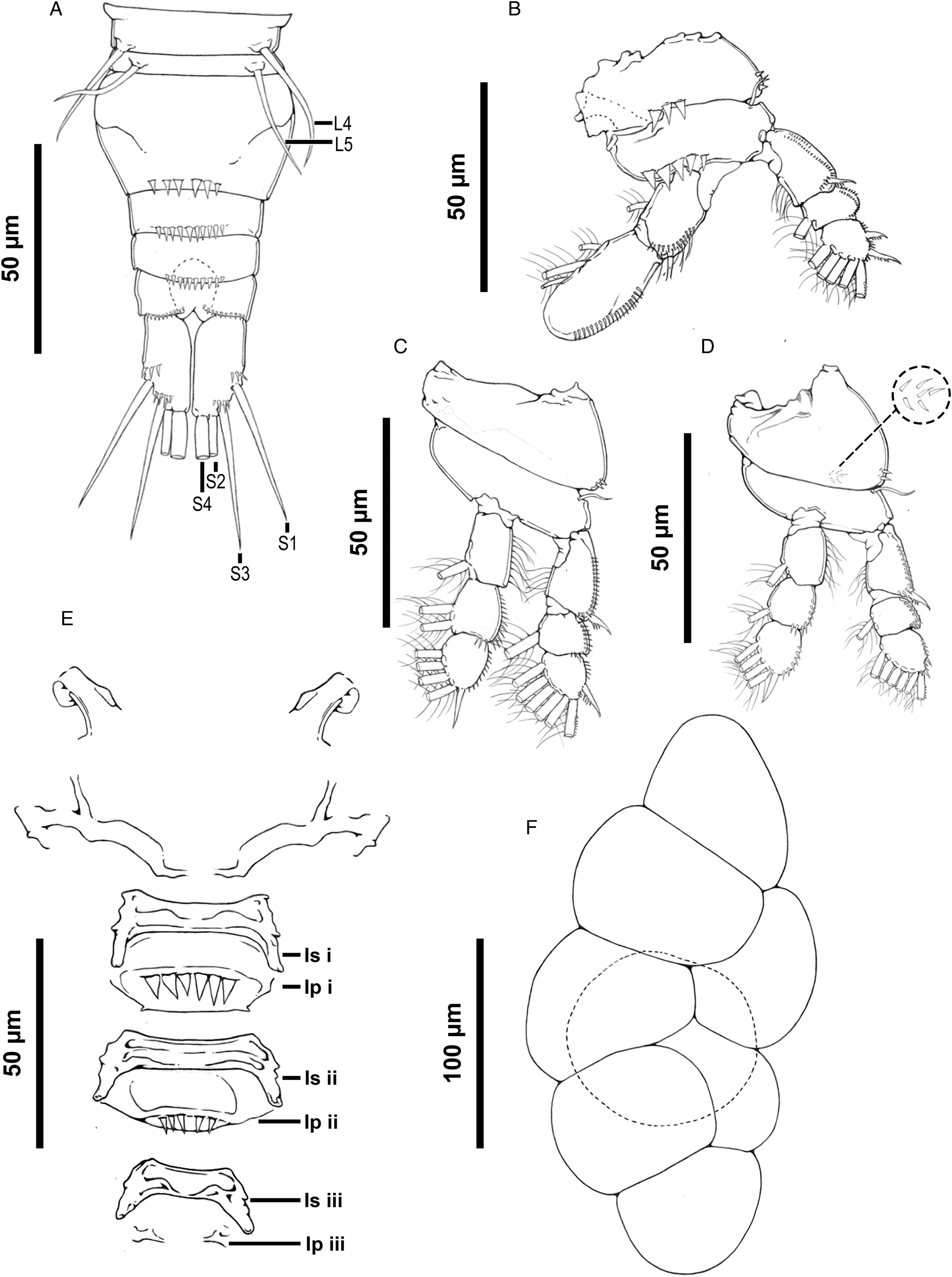
Caudal rami (Fig. 4A), each ramus ornamented with 2 rows of spinules; spinule rows located immediately before seta 1 and 3; armed with 4 bare setae (s-1 to s-4); s-1 and s-3 similar in size, both setae inserted on ventral surface; s-2 and s-4 inserted on posterior margin; s-4 longest.
Antennule 5-segmented (Figs 3F and 5B); proximal segment about 2.0 × longer than others, incompletely subdivided into 2 subsegments; setal formula: 6, 4, 4, 2, 5 + 2 ae (total 23). Antenna (Figs 3E and 5C) 4-segmented comprising coxobasis, and 3-segmented endopod (enp-1 to enp-3). Coxobasis broad, armed with short outer seta. Enp-1 slightly longer than coxobasis, unornamented. Enp-2 triangular, maximum width at level of proximal margin, unornamented. Enp-3 reduced, unornamented. Antennal claw strongly curved, similar in size to enp-1 with fossa on concave margin; tip rounded and covered by cap. Antennal support (Figs 3C and 5D) ventrally articulated with antenna by coxobasis, armed with lateral spine-like projection.
Light micrographs of Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 – adult female, (A) body, ventral view, (B) antennule segments (1–5) and the antennal support armed with lateral spine-like projection (Sp), (C) antenna with reduced third endopodal segment (enp-3) and claw with fossa (Fo) and distal hyaline cap (Cap), (D) cephalosome, ventral view, with antennal support (As), (E) cephalothorax, dorsal view, with dorsal cephalic shield with elliptical mark (Em), (F) horn-like projections (Hp). Scale bars in micrometres (μm).

Buccal apparatus (Fig. 3D) comprising mandible, maxillule and maxilla. Labrum not observed. Mandible armed with 2 blades (=anterior and posterior blade), both blades ornamented with spinules along posterior margin. Maxillule reduced, unornamented. Maxilla 2-segmented, comprising syncoxa (=first segment) and basis (=second segment); syncoxa broad, with distal pore; basis ornamented with multiple spinules.
Three pairs of biramous swimming legs (L1 to L3), each leg comprising coxa, basis, enp (=inner ramus) and exp (=outer ramus). L1 (Fig. 4B). Coxa ornamented with 2 clusters of spinules (lateral and central); central spinules arranged in horizontal line (n = 3 spinules) near posterior margin; lateral spinules minute, located on posterolateral margin (n = 3–4 spinules). Basis carrying bare outer seta and ornamented with row of spinules located immediately above inner ramus (n = ~9 spinules); spinules similar in size and shape as central spinules of coxobasis. Enp 2-segmented, all segments with spinules along outer margin; enp-1 rectangular, armed with 1 plumose seta, ornamented with bristles along outer margin; enp-2 oar-shaped, with 3 plumose setae. Exp 3-segmented, all segments with spinules along outer margin; enp-1 armed with serrated spine, ornamented with bristles along inner margin; enp-2 with plumose seta; enp-3 armed with 2 serrated spines, 1 semi-plumose seta (=serrated outer margin) and 4 plumose setae.
L2 (Fig. 4C). Coxa ornamented with minute spinules on outer margin. Basis with bare outer seta. Enp 3-segmented; enp-1 ornamented with bristles on outer margin, armed with plumose seta; enp-2 and -3 both ornamented with spinules along outer margin; enp-2 with 2 plumose setae; enp-3 with 1 spine and 4 plumose setae. Exp 3-segmented, all segments with spinules along outer margin; exp-1 armed with serrated spine, ornamented with bristles along inner margin; exp-2 with 1 plumose seta; exp-3 with 1 serrated spine, 1 semi-plumose seta and 5 plumose setae. L3 (Fig. 4D) with same ornamentation and armament described for L2, except for single plumose seta on enp-2 and lacking spine on enp-3.
L4 and L5, both legs reduced and represented by single seta (Figs 4A and 6C). Spine and setal formula of biramous swimming legs are summarized in Table 7.
Light micrographs of Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 – adult female, (A) posterior part of the body, ventral view, (B) posterior part of the body, dorsal view, (C) fourth and fifth pedigerous somites with legs reduced to a single seta (L4 and L5). Scale bars in micrometres (μm).
Intercoxal sclerites unornamented, with both lateral ends directed posteriorly (Figs 4E and 6A). Interpodal plate of L1 and L2, both ornamented with transverse row of spinules; interpodal plate of L3, unornamented. L4 and L5, absent. Egg sac (Fig. 4F) paired, multiseriate.
Remarks
The specimens studied were identified as members of the genus Rhinergasilus by having the following combination of diagnostic features: (1) PS-4 and PS-5 reduced, both somites narrower than previous ones; (2) 3 pairs of biramous swimming legs (L1 to L3); and (3) L4 and L5 reduced, each leg being represented by single seta (Boeger and Thatcher, Reference Boeger and Thatcher1988). Except for 4 genera (Abergasilus Hewitt, 1978; Brasergasilus Thatcher and Boeger, 1983; Mugilicola Tripathi, 1960; and Urogasilus Rosim et al., 2013), the reduction in the number of swimming legs allows the easy differentiation of these copepods from other ergasilids. The genera Abergasilus and Mugilicola do not occur in the Neotropical region [see Figs 4 and 5 in Amado et al. (Reference Amado, Ho and Rocha1995)] and both differ from Rhinergasilus by having the first endopod with 3 segments instead of 2. Rhinergasilus can also be distinguished from Mugilicola and Abergasilus by the absence of a long ‘neck’ separating the cephalothorax in 2 distinguished areas (i.e. post-oral and pre-oral regions) or a robust spine on the second antennal segment, respectively. Regarding the Neotropical genera, it can be differentiated from Urogasilus by the preferred attachment site (i.e. nostrils/gills instead of the urinary bladder as in Urogasilus), the shape of the maxilla (i.e. maxilla carrying 2 blades in Rhinergasilus instead of a single blade as in Urogasilus) and the absence of a posterior ‘trunk’. In Urogasilus brasiliensis Rosim et al., Reference Rosim, Boxshall and Ceccarelli2013, the final thoracic segments (PS-4 and PS-5) and genital somite are fused to form an elongated structure called ‘trunk’ (Rosim et al., Reference Rosim, Boxshall and Ceccarelli2013), which does not occur in any of the 3 known species of Rhinergasilus – i.e. even though the reduction in the size of PS-4 and PS-5 occurs in Rhinergasilus, these segments remain unfused in all species as can be seen in Fig. 7E. Brasergasilus is the Neotropical genus that most resembles Rhinergasilus by the general shape of the body, number/segmentation of legs, and attachment site. Despite that, these 2 genera can be differentiated from each other by the shape of the antennal claw and the presence/absence of the L4. In Rhinergasilus, the claw is relatively short and has a simple (sharp) tip rather than longer (i.e. usually longer than other antennal segments combined) and with a modified tip as present in Brasergasilus species. Regarding L4, in Rhinergasilus this leg is represented by a single seta, while in Brasergasilus it is completely absent.
Scanning electron micrographs of Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 – adult female, (A) body, lateral view, with first pedigerous somite (PS-1) separated from cephalosome, (B) body, dorsal view, with flexible cuticle (Fc) separating cephalosome from prosome (PS-1), (C) pedigerous somite, dorsal view, with second pedigerous somite with paired integumental windows laterally on tergite (lw), (D) detail of the patch of spinules from coxa of leg 3, (E) urosome, dorsal view, with third abdominal segment ornamented with a pair of pores carrying bristles (Po), (F) cephalothorax, lateral view, showing some parts of buccal apparatus such as the mandible (Md); (G) horn-like projections (Hp), (H) cephalothorax, dorsal view, with dorsal cephalic shield with elliptical mark (Em), (I) detail of the spinules (Se) on the first seta of third exopodal segment of leg 2. Scale bars in micrometres (μm).

Among the 3 known Rhinergasilus species (Rhinergasilus digitus Narciso et al., Reference Narciso, Brandão, Perbiche-Neves and Silva2020; R. piranhus – type-species; and Rhinergasilus unguilongus Narciso et al., Reference Narciso, Perbiche-Neves and Silva2021), the present specimens were identified as R. piranhus since they possess the same leg structure as described by Boeger and Thatcher (Reference Boeger and Thatcher1988). In R. piranhus, the L4 and L5 are both represented by a small papilla carrying a single seta and each leg arises independently from their respective somites [see Fig. 10 in Boeger and Thatcher (Reference Boeger and Thatcher1988) and Figs 4A and 6C in the present paper]. On the other hand, in the other 2 species, the papillae are apparently fused and both setae arise from the same somite (PS-5). Additionally, the present specimens also resemble R. piranhus regarding the size [287–347 (309) long in present specimens vs 237–282 (263) in the original description] and by sharing other diagnostic features with the type-species such as (1) seta-1 and -3 of caudal ramus long – each seta about ¾ the length of seta-2; (2) interpodal plates 1 and 2, coxa of L1, and base of L1 ornamented with a transverse row of spinules; (3) antenna with first endopodal segment (or enp-1) unornamented; and (4) claw being as long as enp-1 (i.e. latter segment considered the longest antennal segment).
The diagnostic features allowed the identification of the present copepods as R. piranhus; however, careful analysis of them also showed some discrepancies regarding the original description as well as the visualization of new features. Regarding the discrepancies, our specimens showed the PS-1 separated dorsally from cephalosome rather than both segments being completely fused as proposed by Boeger and Thatcher (Reference Boeger and Thatcher1988). The setation of antennules was different, in present specimens the antennule setation is 6, 4, 4, 2, 5 + 2 ae (total 23) vs 4, 1, 4, 4, 5 (total 18) as in the original description. The setation of legs also differs, in present specimens the L1 enp-3 and L3 exp-3 possess 3 and 6 plumose setae, respectively, instead of 2 and 5 as in the original description. Finally, the egg disposal varied from uni- to multiseriate instead of just uniseriate as in the original description.
Regarding the novelties, we observed new features associated with the thoracic/abdominal somites, antennae, maxilla and swimming legs. The dorsal cephalic shield is equipped with a pair of horn-like structures located immediately below the antennules (see Figs 5F and 7G). Ventrally, the antennal joint (also called antennal support here, as this structure seems to articulate the cephalosome with the antennal coxobase) carried lateral spine-like projections (see Figs 3C and 5D). Several pores (carrying bristles or without bristles) were also observed on the dorsal surface of the prosome and urosome somites – with a higher concentration of pores observed on the dorsal surface of the cephalosome (Fig. 3A). In addition to the pores, 3 elliptical windows were also observed for the first time in the cephalosome (as represented in Figs 5E and 7H). The maxilla showed spinules along the posterior margin of both blades (anterior and posterior blades) rather than only on the anterior blade as in the original description. Finally, careful analysis of the antennal claw showed the presence of fossa and a hyaline cap covering the tip. Other novelties such as the presence of serrated spines and semi-plumose setae were observed for all swimming legs.
These novelties/discrepancies noted above were not considered sufficient to propose a new taxon (as discussed in detail in the next session) hence only a redescription of this species was provided herein.
Gamispatulus schizodontis Thatcher and Boeger, Reference Thatcher and Boeger1984 (Fig. 8; Table 8)
Type host: Schizodon fasciatus Spix and Agassiz, 1829 (Characiformes: Anostomidae).
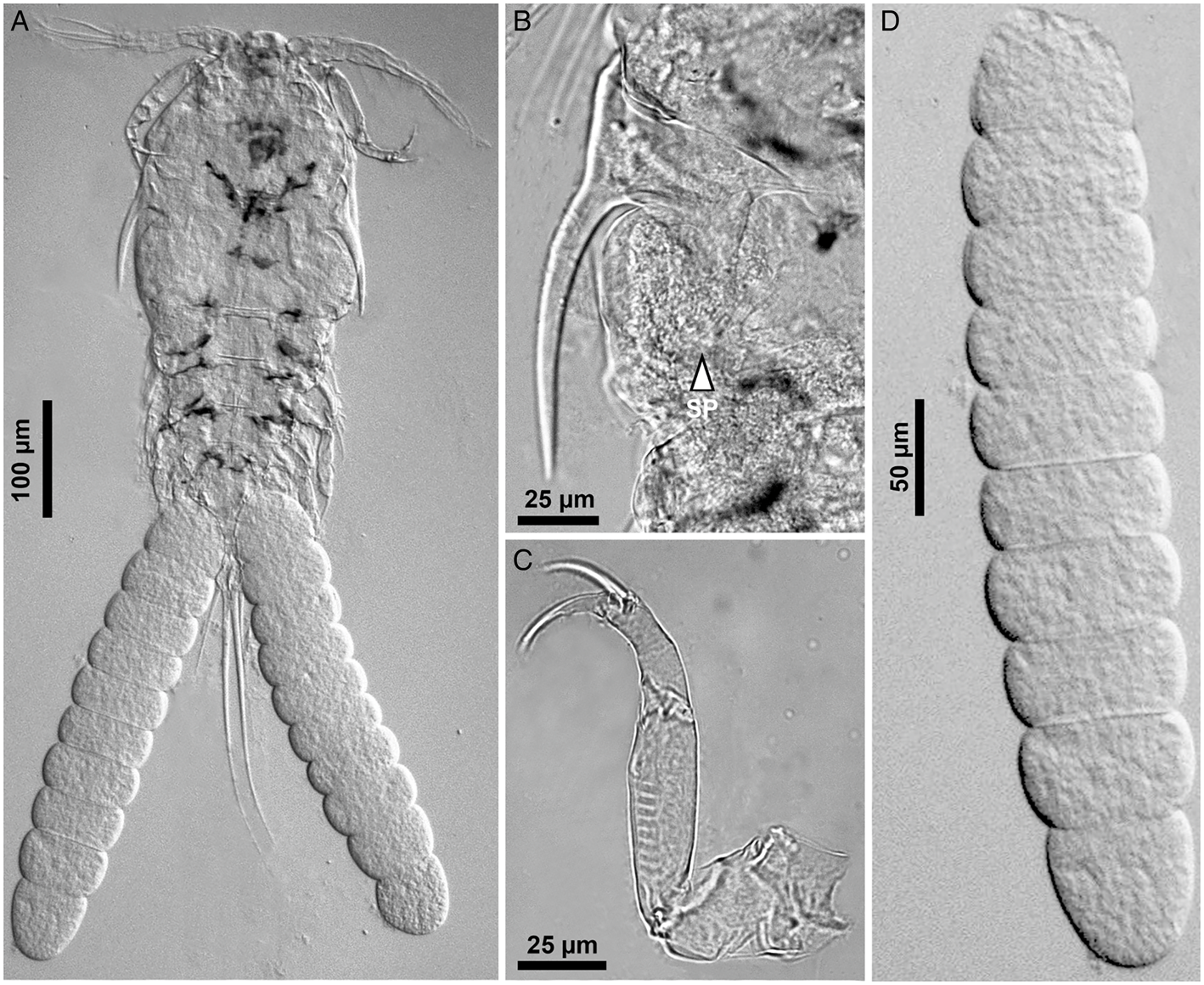
Light micrographs of Gamispatulus schizodontis Thatcher and Boeger, Reference Thatcher and Boeger1984 – adult female, (A) body, ventral view, (B) retrostylet armed with spatulate process (SP), (C) antenna, (D) uniseriate egg sac. Scale bars in micrometres (μm).
Armature of swimming legs (L1 to L4) of Gamispatulus schizodontis Boerger and Thatcher, Reference Boeger and Thatcher1988 – adult female
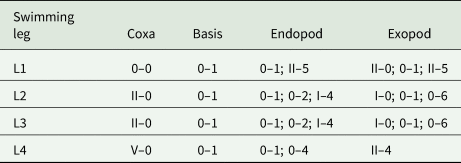
(Roman numeral = spines; Arabic numerals = setae).
Other hosts: Hoplias lacerdae Miranda Ribeiro, 1908 (Characiformes: Erythrinidae); Hoplias malabaricus (Bloch, 1794) (Characiformes: Erythrinidae); Leporinus friderici (Bloch, 1794) (Characiformes: Anostomidae); Leporinus lacustris Amaral Campos, 1945 (Characiformes: Anostomidae); Megaleporinus elongatus (Valenciennes, 1850) (Characiformes: Anostomidae); Megaleporinus obtusidens (Valenciennes, 1837) (Characiformes: Anostomidae); Pimelodus maculatus Lacepède, 1803 (Siluriformes: Pimelodidae); S. maculatus Kner, 1858 (Characiformes: Serrasalmidae) – PS; Serrasalmus marginatus Valenciennes, 1837 (Characiformes: Serrasalmidae); Schizodon borellii (Boulenger, 1900) (Characiformes: Anostomidae); and Schizodon intermedius Garavello and Britski, 1990 (Characiformes: Anostomidae).
Type locality: Ilha da Marchantaria, Solimoes-Amazonas River near Manaus, Amazonas state, Brazil.
Sampled localities: Area 6 – Mouth of the Pardo River in the municipality of Salto Grande, state of São Paulo, Brazil (22°54′41.7″S, 49°57′43.1″W).
Site of infestation: Nostrils.
Material examined: A total of 12 ♀♀ from nostrils of S. maculatus sampled in area 6 were examined for morphological description.
Representative DNA sequences: PQ442938, PQ442940-41, PQ450081, PQ452576-77, PQ452579.
Voucher material: A total of 6 whole ♀♀ stored in glass vials with 96% ethanol were deposited in the Zoological Collection of MZUSP (accession no. 46663 and 46792).
Identification (based on adult females; male unknown): Present specimens showed all diagnostic features proposed by Thatcher and Boeger (Reference Thatcher and Boeger1984) for G. schizodontis, as well as the new features observed by Narciso and Silva (Reference Narciso and Silva2020) when redescribing this species. Spine and setal formula of biramous swimming legs are summarized in Table 8.
Remarks
The copepods found inside the nostrils of S. maculatus were identified as members of the ‘vaigamid’ subgroup by having the combination of cephalosome armed with dorsolateral stylets (or retrostylets) and leg 1 with endopod 2-segmented. To date, 5 ergasilid genera are included in this subgroup, namely: Gamidactylus Thatcher and Boeger, Reference Thatcher and Boeger1984; Gamispatulus; Gamispinus Thatcher and Boeger, Reference Thatcher and Boeger1984; Pseudovaigamus Amado et al., Reference Amado, Ho and Rocha1995; and Vaigamus Thatcher and Robertson, Reference Thatcher and Robertson1984. Among all vaigamids, the present copepods were identified as members of the genus Gamispatulus by having the following combination of diagnostic features: (i) rostrum armed with a rostral spine; (ii) antennule 5-segmented and armed with 2 claws (middle and inner claw); and (iii) L4 with enp 2-segmented and exp 1-segmented.
Currently, the genus Gamispatulus consists of 2 species, G. schizodontis (type-species) and Gamispatulus ferrilongus Narciso and Silva, Reference Narciso and Silva2020. The present copepods were identified as G. schizodontis due to the possession of a spatulate process on the retrostylets (see Fig. 7) – which is an exclusive feature of this species. Additionally, they also resemble the type species by the size of the rostral spine (i.e. short as in G. schizodontis rather than long as in G. ferrilongus), the ornamentation of thoracic appendages (including antennule, antenna and swimming legs), and by the number of setae on caudal ramus (i.e. 2 setae as in G. schizodontis rather than 4 as in its congener).
Molecular and phylogenetic analyses
A total of 12 novel sequences were generated (Table 3). Several attempts were made to obtain 18S rDNA sequences for R. piranhus (i.e. including variations in the number of cycles and temperature during PCR), but no positive results were obtained. The phylogenetic reconstructions using ribosomal genes (18S and 28S) recovered the monophyly of Ergasilidae with high support values (i.e. 100 and 0.99 for ML and BI analyses, respectively) and positioned the 2 studied species (R. piranhus and G. schizodontis) as members of this family.
The final alignment of the 28S rDNA dataset comprised a total of 49 sequences and generated 659 characters for analyses (Supplementary Table 1). The estimated distance between the novel sequences of R. piranhus and G. schizodontis and other ergasilids ranged between 10.7–12.5 and 9.0–11.8%, respectively. Among the R. piranhus sequences, the intraspecific distance varied between 0.5 and 0.1%. The unique 28S rDNA sequence of G. schizodontis diverged from those of R. piranhus by 7.0–7.2%. Regarding the 18S rDNA, the final alignment comprised a total of 52 sequences and resulted in a length of 1.121 bases (Supplementary Table 2). The 2 sequences obtained from G. schizodontis diverged from each other by 1.38% and from the other ergasilids by 1.2–3.9%.
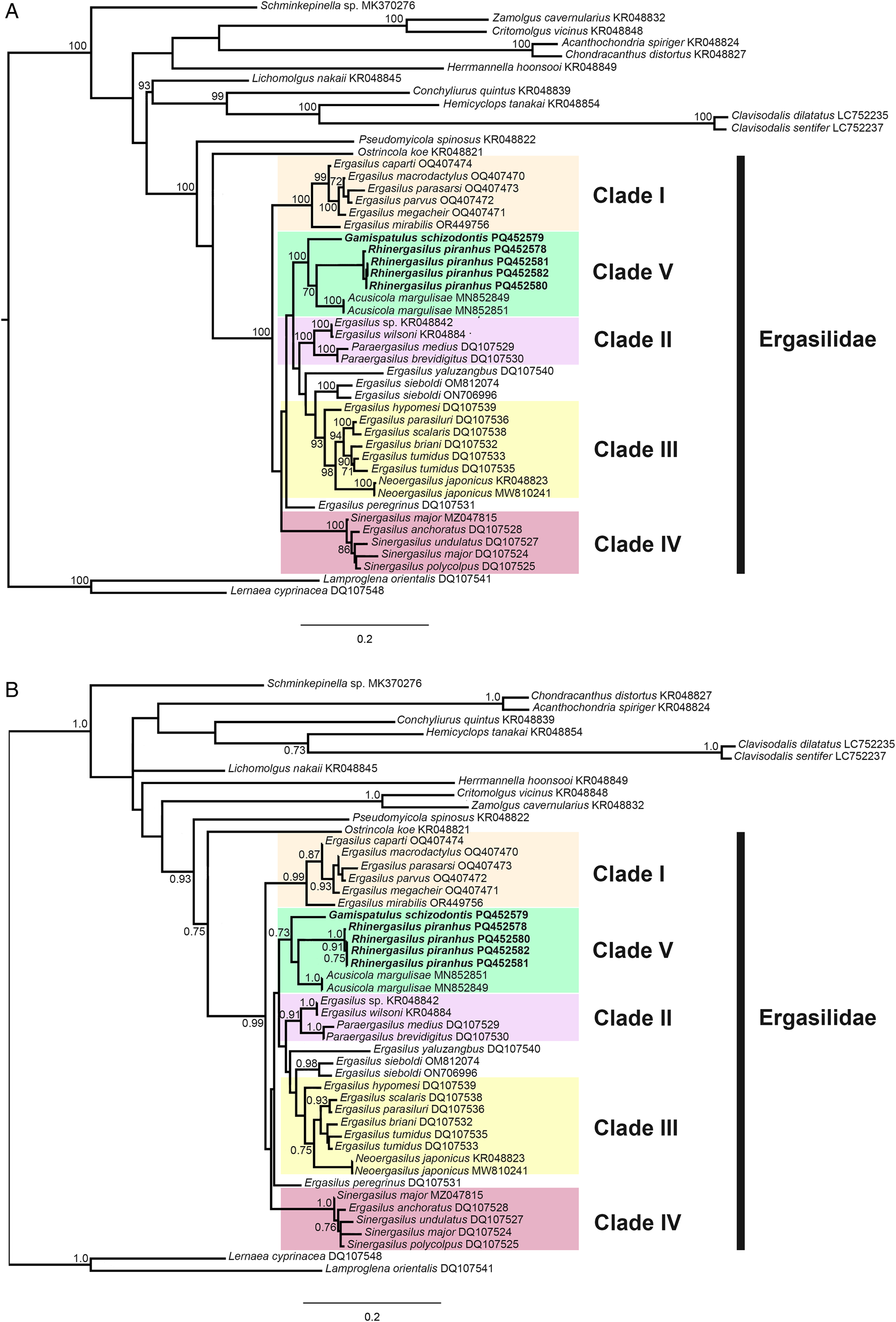
Overall, the topologies for the ML and BI analyses for both ribosomal genes (18S and 28S rDNA) were congruent. Strong support values were obtained from 5 terminal clades in Ergasilidae, but relationships between those clades showed (mostly) low support (Figs. 9 and 10). The monophyly of type-genus, Ergasilus Nordmann, 1832 was not recovered.
Phylogenetic tree of Ergasilidae copepods based on partial 28S rRNA gene alignments. Newly generated sequences for Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 and Gamispatulus schizodontis Thatcher and Boeger, Reference Thatcher and Boeger1984 are provided in bold. Nodal support presented above or below branches for (A) Bayesian inference (>0.7) and (B) maximum likelihood (>70%) analyses (BI/ML). Sequences of Lernaea cyprinacea Linnaeus, 1758 and Lamproglena orientalis Markevich, 1936 were both used as outgroup.
Phylogenetic tree of Ergasilidae copepods based on partial 18S rRNA gene alignments. Newly generated sequences for Rhinergasilus piranhus Boeger and Thatcher, Reference Boeger and Thatcher1988 and Gamispatulus schizodontis Thatcher and Boeger, Reference Thatcher and Boeger1984 are provided in bold. Nodal support presented above or below branches for (A) Bayesian inference (>0.7) and (B) maximum likelihood (>70%) analyses (BI/ML). Sequences of Lernaea cyprinacea Linnaeus, 1758 and Lamproglena orientalis Markevich, 1936 were both used as outgroup.
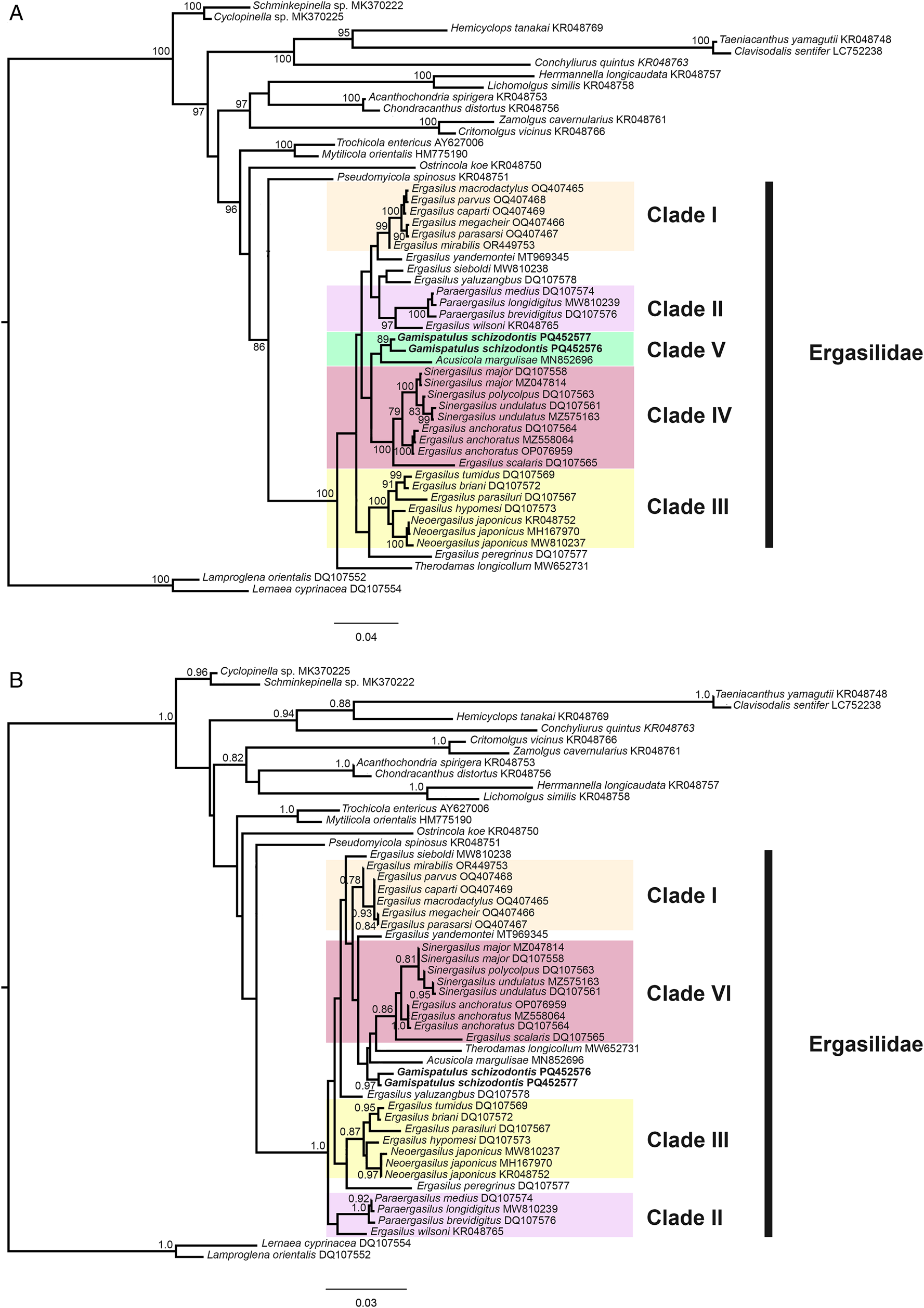
The well-supported (>0.7 ML/70 BI) terminal clades were: clade I or Sub-Saharan clade – including Ergasilus spp. from Lake Tanganyika plus Ergasilus mirabilis Oldewage and van As, 1987 from Burundi, South Africa and Zambia; clade II – including Paraergasilus spp. plus Ergasilus wilsoni Markevich, Reference Markevich1933; clade III – including Ergasilus spp. plus Neoergasilus japonicus (Harada, 1930); clade IV – constituted by Sinergasilus spp. plus Ergasilus anchoratus Markevich, 1946. For 28S, a clade composed of Neotropical species (or clade V) was also recovered – it includes the novel sequences provided herein (for R. piranhus and G. schizodontis) plus sequences of Acusicola margulisae Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020 (Fig. 9). The latter clade was also recovered for 18S but with low support (Fig. 10). Except for Ergasilus, the other multi-sequenced genera (i.e. genera with more than 1 species with available sequences in NCBI database) had their monophyly recovered with high support values, including (i) Neoergasilus Yin, 1956; (ii) Paraergasilus Markevich, 1937; and (iii) Sinergasilus.
The final cox 1 alignment comprised a total of 59 sequences and the final dataset, after trimming the extremes, resulted in 510 bases. This dataset was only used to calculate genetic difference matrices. The BI and ML analyses produced phylograms with unsupported clades and unsatisfactory placements of the sequences used in the dataset (see Supplementary Fig. 3). The intraspecific distance observed between G. schizodontis sequences ranged from 0.9 to 2.1%, while the only cox 1 sequence obtained for R. piranhus differed from other ergasilids by 20.5–21.6%. Within Ergasilidae, the observed interspecific divergence varied from 0.9 to 22.4% (see Supplementary Table 3).
Discussion
Morphological comparisons and redescription
Several morphological variations were found between the present specimens and the original description of R. piranhus. Examination of the type-material was also made; however, due to their poor state of preservation it was not possible to verify the condition of most of the new features in those copepods. Despite that, some comparisons were made concerning the morphology of the urosome, caudal rami and part of the antennae (including claw) as these parts were relatively well preserved in some of the type specimens (see Supplementary Fig. 1). In the caudal ramus of the holotype, it was possible to visualize the lateral seta (or s-1) and, as proposed herein, this seta is relatively long when compared to the same seta present in R. digitus and R. unguilongus (i.e. s-1 is several times smaller when compared to s-2 and s-4 in the latter 2 species). Something similar was also noted concerning the shape and size of the antennal claw (preserved in paratypes): in R. piranhus the claw has the same length as the first endopodal segment (or enp-1), and its internal angle is about 180 degrees which gives it a more rounded shape, whereas, in its 2 congeners, the claw has a different size (i.e. being smaller than enp-1 in R. digitus while in R. unguilongus it is the longest antennal structure) and the internal angle is always acute (apparently less than 45 degrees in both species). The above features were also observed in our specimens and agree with the original description of R. piranhus. Therefore, most of our comparisons were primarily made relying on drawings and the description text proposed for this species by Boeger and Thatcher (Reference Boeger and Thatcher1988).
Differences in the number of setae (or setation) and the segmentation of cephalothorax were the 2 main discrepancies observed between our specimens and the original description of R. piranhus. Discrepancies in setation are one of the most common discrepancies pointed out in ergasilid redescriptions (see Chernysheva and Purasjoki, Reference Chernysheva and Purasjoki1991; Kim and Nagasawa, Reference Kim and Nagasawa2006; Marques et al., Reference Marques, Boeger and Carvalho Brasil-Sato2015; Marques and Boeger, Reference Marques and Boeger2018; Narciso and Silva, Reference Narciso and Silva2020). Due to their small size, setae can be easily lost due to breakages during the copepod handling or even being covered by other structures in non-dissected specimens (Boxshall, Reference Boxshall2016). Despite the segmentation of cephalothorax being considered more evident when compared with the presence/absence of a certain seta, the correct description of this segment is also ‘tricky’ in Ergasilidae and can vary substantially depending on the taxonomist interpretation. In Marques et al. (Reference Marques, Boeger and Carvalho Brasil-Sato2015), the authors verified several incongruences in the cephalothorax segmentation when reassessing the type-material of Ergasilus species. Similar to the setation, such inconsistencies on cephalothorax may be related to the copepod handling when preparing them for morphological identification – e.g. covering copepods with a coverslip on a flat microscope slide may cause them to be crushed/smashed and consequently obscuring the real segmentation of the cephalothorax (personal observation). Less invasive mounting techniques, such as the wooden slide proposed by Humes and Gooding (Reference Humes and Gooding1964), might be viable alternatives to avoid such errors, which is particularly important for ‘big’ ergasilids – i.e. since they are more affected by such preparations. For small size species such as R. piranhus, the use of SEM proved useful to achieve a more accurate description of this somite – i.e. in the present study, the SEM allowed for the visualization of small details like the pores and elliptical marks, as well as the ‘flexible cuticle’ (or FC) that separates the cephalosome from the PS-1 which corroborates our observation that the cephalothorax in this species is subdivided (see Fig. 7B).
Our analyses also showed some novel features not previously described for R. piranhus. The dorsal cephalic shield is equipped with a pair of horn-like structures located immediately below the antennules (Figs 5F and 7G). Ventrally, spine-like projections were also observed for both sides of the antennal support (i.e. structure present on the ventral side of cephalosome that articulates with both antennae) (Fig. 5D). Although such ‘acute’ structures do not have any defined function, they can act as a complementary apparatus for its species attachment inside the nostrils – acting similarly to the retrostylets and/or rostral spine present in all ‘vaigamids’ like G. schizodontis (discussed in details below) (Narciso and Silva, Reference Narciso and Silva2020). The other 2 Rhinergasilus species were described as gill parasites and lacked such structures (Narciso et al., Reference Narciso, Perbiche-Neves and Silva2021). However, additional data such as behavioural or in situ observations of living specimens are still necessary to prove this assumption. Other minor novelties were also observed regarding the ornamentation of the body and appendages.
Similarly to the discrepancies, the previous novelties were probably also difficult to visualize due to their relatively small size in the original description. Most of the novelties were found in the cephalothorax, which is a segment that is generally not dissected and has various ‘obstacles’ that compromise its correct characterization (e.g. internal organs, presence of pigments, etc.). As noticed in the type-material (see Supplementary Fig. 1), the presence of internal lumps and/or tissues obscured our observation of most diagnostic features. In our analyses under the compound microscope, it was possible to observe most of the proposed novelties (as seen in Figs. 5 and 6), but it was only achieved due to the use of clarifying agents – e.g. lactic acid, Hoyer's medium, etc.
Descriptions that detail the ornamentation of the body somites are relatively recent and still little practiced in taxonomic studies of ergasilids. Alston et al. (Reference Alston, Boxshall and Lewis1993) provided the first detailed description of pores throughout the body of an Ergasilus species (i.e. made during the redescription of Ergasilus briani Markevich, Reference Markevich1933). In recent descriptions of Brazilian ergasilids, similar pores were also observed on the cephalothorax of different species (including species from distinct genera), which indicates that these ornaments are probably very common in the Ergasilidae (Narciso et al., Reference Narciso, Vieira and Silva2022; Couto et al., Reference Couto, Nazaré Pereira, Luque, Paschoal and Pereira2023). Regarding the antennal support, Ergasilus bryconis Thatcher, 1981 is the only species in which this structure was represented in its description. Details of the ventral side are even scarcer in Ergasilidae since most descriptions only represent/describe the dorsal side of the copepods. Despite their current low taxonomic importance, structures such as the antennal support can provide relevant diagnostic features (as presented herein) and should therefore be better explored in future descriptions.
In the Neotropical region, the cephalosome armed with dorsolateral stylets (or retrostylets) is a diagnostic feature unique to the copepods from the ‘vaigamid’ subgroup. Similar stylets were also reported for 5 species of Paraergasilus Markevich, 1937 (Paraergasilus brevidigitus Yin, 1954; Paraergasilus longidigitus Yin, 1954; Paraergasilus medius Yin, 1956; Paraergasilus mimus Yin, 1962; and Paraergasilus rylovi Markevich, 1937); however, these structures are usually described as spinous protrusions derived from the posterolateral margin of cephalosome instead of well-defined, mobile structures carrying sensory organs (i.e. pores and sensilla), as occurs in all vaigamids (Thatcher and Robertson, Reference Thatcher and Robertson1984). No developmental studies are available for representatives of both genera, making it difficult to establish the putative homology of these structures; however, other diagnostic features allow for the easy differentiation of both groups, such as (1) antenna armed with 1 or 2 claws in vaigamids vs 3 claws in Paraergasilus; (2) rostrum armed with rostral spine as present in Gamispatulus, Pseudovaigamus and Vaigamus vs lacking such structure in Paraergasilus; (3) L1 enp 2-segmented in all vaigamids vs 3-segmented in Paraergasilus; and (4) L5 represented by 1- or 2-setae in vaigamids vs 1-segmented as in Paraergasilus. The geographical distribution of these 2 groups is also different: Paraergasilus has a wide distribution, with species reported from African, European, Asian and Australian countries (El-Rashidy and Boxshall, Reference El-Rashidy and Boxshall2001a) while all vaigamids are currently restricted to the Brazilian region (Amado et al., Reference Amado, Ho and Rocha1995). The phylogenetic relationships between these 2 groups are still unclear as the available phylogenetic hypotheses (based on morphological data) are contradictory: (i) Amado et al. (Reference Amado, Ho and Rocha1995) and Tang and Kalman (Reference Tang and Kalman2008) recovered both groups in separated clades – Paraergasilus was recovered at the base of Ergasilidae acting as the most ‘ancestral-like’ ergasilid due to the retention of the tree-clawed antennae (in conjunction to other plesiomorphic characters), while vaigamids occupied a more internal position, closer to the other Neotropical genera; whereas in (ii) El-Rashidy (Reference El-Rashidy1999) both groups were recovered in a single clade at the base of the family, except for Pseudovaigamus spinicephalus who clustered inside the clade ‘Avii’ which consisted exclusively of Neotropical species. Our phylogenetic analyses (discussed in detail in the next section) recovered both groups in 2 separate clades (clade II and V, see Figs 9 and 10); however, the low ML and BI support between the terminal clades did not allow us to completely resolve their positions. Yet, the closer relationship observed between G. schizodontis with other Neotropical ergasilids (i.e. A. margulisae and R. piranhus) in clade V indicates that the proposal originally made by Amado et al. (Reference Amado, Ho and Rocha1995), which includes vaigamids alongside the other Neotropical ergasilids, is probably more accurate.
Regarding the morphology of G. schizodontis, the present copepods were consistent with the redescription proposed by Narciso and Silva (Reference Narciso and Silva2020) for this species. Novelties and/or discrepancies were not observed for this species.
Molecular phylogenetics
In the last 20 years, the number of molecular sequences available for ergasilids has increased (Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Oliveira et al., Reference Oliveira, Corrêa, Adriano and Tavares-Dias2021; Abdel-Radi et al., Reference Abdel-Radi, Rashad, Ali, Eissa, Abdelsalam and Abou-Okada2022; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024). Especially after the publication of the phylogenies based on ribosomal genes (18S and 28S rDNA) done by Song et al. (Reference Song, Wang, Yao, Gao and Nie2008), in which the relationship of Chinese ergasilids (including species of Ergasilus, Neoergasilus, Paraergasilus, Pseudergasilus and Sinergasilus) was explored for the first time. Among their main results, the polyphyletic status of the type-genus Ergasilus was achieved corroborating previous findings made by El-Rashidy (Reference El-Rashidy1999) when exploring the phylogeny of this taxon based on morphological features. In our analyses, the polyphyletic status of Ergasilus was also recovered – i.e. sequences of the type-species, Ergasilus sieboldi Nordmann, 1832 tended to appear as an isolate terminal clade while other Ergasilus spp. are scattered across different clades (Figs 9 and 10). For the MI of the 18S gene, Ergasilus yaluzangbus Kuang and Qian, 1985 appeared as a sister taxon to E. sieboldi but with low support. Since the molecular phylogeny proposed by Song et al. (Reference Song, Wang, Yao, Gao and Nie2008), E. yaluzangbus tends to ‘jump’ between clades in the available phylogenies, with no defined phylogenetic relationships. The same applies to E. sieboldi which, depending on the gene used, has different relationships within Ergasilidae [see phylogenies present in Fikiye et al. (Reference Fikiye, Smit, Van As, Truter and Hadfield2023)]. A major revision of this genus is strongly required but due to several impediments (e.g. a huge number of known species, wide distribution, difficulty in obtaining the type-material, low availability of taxonomists specialized in ergasilids, etc.) such revision is difficult to carry out. Furthermore, the uncertain relationship between E. sieboldi (type-species) and its congeners makes it even more difficult to define which are the ‘true’ members of this speciose genus. Despite this, we believe that minor revisions can be made by exploring highly supported terminal clades that include Ergasilus spp.
Connections between terminal clades had low support in our analyses making it impossible to establish reliable phylogenetic relationships between those clades. However, most of the terminal clades showed high support values, resulting in 5 main clades (clade I to V) in our analyses (Figs. 9 and 10). Clade I (or Sub-Saharan clade) comprised 6 Ergasilus species [Ergasilus caparti Míč et al., Reference Míč, Řehulková and Seifertová2023; Ergasilus macrodactylus (Sars G.O., 1909); Ergasilus megacheir (Sars G.O., 1909); E. mirabilis Oldewage and van As, 1987; Ergasilus parasarsi Míč et al., Reference Míč, Řehulková and Seifertová2023; and Ergasilus parvus Míč et al., Reference Míč, Řehulková and Seifertová2023], all of them described from African countries (mostly in Lake Tanganyika). Recently, Fikiye et al. (Reference Fikiye, Smit, Van As, Truter and Hadfield2023) redescribed E. mirabilis Oldewage and van As, 1987 sampled in South Africa and Zambia based on an integrative approach (i.e. including morphological description based on light and SEM microscopy in conjunction with molecular and phylogenetic analyses) and the same well-supported clade was recovered in their analyses. This African group of Ergasilus has an antenna with a very particular shape that is quite different from the ‘standard’ shape exhibited by other species of Ergasilus like E. sieboldi. A more detailed review of this group is necessary, but we believe that this group consists of a putative new genus for this family.
The clade II is constituted by a monophyletic group comprising Paraergasilus spp. with E. wilsoni Markevich, Reference Markevich1933 (together with Ergasilus sp. – KR048842 in 28S analyses) as its sister taxon. Paraergasilus Markevich, 1937 is considered a monophyletic genus that retains the (putative) ancestral state of the antenna, which in this taxon is armed with 3 terminal claws [i.e. loss of the lateral claws was considered a synapomorphy in Ergasilidae by Amado et al. (Reference Amado, Ho and Rocha1995), El-Rashidy (Reference El-Rashidy1999) and Tang and Kalman (Reference Tang and Kalman2008)]. To date, this genus comprises 16 valid species but only 3 of them have sequences available (for the 18S and/or 28S ribosomal genes): P. brevidigitus Yin, 1954; P. longidigitus Yin, 1954; and P. medius Yin, 1956. Latter species together with P. rylovi Markevich, 1937 (type-species) and P. mimus Yin, 1962 (the latter 2 without available sequences) can be distinguished from their congeners by having a pair of lateral projections on the cephalothorax (El-Rashidy and Boxshall, Reference El-Rashidy and Boxshall2001a). This feature resembles the ‘retrostylets’ present in Neotropical vaigamids such as G. schizodontis but due to the unclear relation between clades and the lack of sequences for the other Paraergasilus that lack such projections, it was not possible to trace the evolutionary history of this character either within the genus or family. Ergasilus wilsoni was originally described from the Black Sea, which is the marginal Mediterranean Sea located between Europe and Asia (Markevich, Reference Markevich1933). This copepod has a similar morphology to other Ergasilus which includes an antenna equipped with a single claw. Nevertheless, phylogenies based on ribosomal genes, including those of the present study, have continuously recovered E. wilsoni as a sister taxon to Paraergasilus, indicating that this species may also be a good target for future revisions.
Within clade III, 2 subclades were recovered: (i) subclade III.1 formed by 4 species of Ergasilus (i.e. 5 for 28S analyses, since Ergasilus scalaris Markevich, 1940 was also recovered as part of this subclade); and (ii) subclade III.2 formed by sequences of N. japonicus (Haradam, 1930). The position of Ergasilus hypomesi Yamaguti, Reference Yamaguti1936 is uncertain as it varies according to the gene. For 18S, this species was recovered within clade III closer to N. japonicus while for 28S it was recovered as the sister taxon of the 2 subclades (Fig. 9). Within subclade III.1, those species share some common features like the fifth leg reduced to a papilla carrying a single seta, and a tendency for somites to fuse (i.e. a feature present in all Ergasilus species once classified as Pseudergasilus Yamaguti, Reference Yamaguti1936). Kim and Nagasawa (Reference Kim and Nagasawa2006), when redescribing Pseudergasilus zacconis Yamaguti, Reference Yamaguti1936, pointed out that these 2 aforementioned features are not exclusive to Pseudergasilus in contrast to Yamaguti (Reference Yamaguti1936) proposal, which reinforced the invalidation of this genus. However, given the polyphyletic status of Ergasilus and the high support recovered for the subclade III.1, it indicates that the invalidation of Pseudovaigamus needs to be checked and this subclade can be another good target for revision in Ergasilidae.
Since the publication of Song et al. (Reference Song, Wang, Yao, Gao and Nie2008), the 3 valid species of Sinergasilus Yin, 1949 and E. anchoratus Markevich, 1946 constitute a highly supported group within Ergasilidae (see phylogenies in Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Oliveira et al., Reference Oliveira, Corrêa, Adriano and Tavares-Dias2021; Abdel-Radi et al., Reference Abdel-Radi, Rashad, Ali, Eissa, Abdelsalam and Abou-Okada2022; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023; Míč et al., Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024). The same clade is also recovered in the present study. The placement of E. anchoratus in clade IV is strongly supported; however, its relationships within the clade are still uncertain as it can act as a sister taxon for Sinergasilus species or even as an insider into the latter genus (i.e. causing Sinergasilus to be paraphyletic) (Figs 9 and 10). Song et al. (Reference Song, Wang, Yao, Gao and Nie2008) when evaluating this clade pointed out that Sinergasilus might have evolved from an ancestral Ergasilus species possibly due to host switching. Recently, the same gene arrangement in the mitochondrial genome was observed by Hua et al. (Reference Hua, Huang, Li, Feng, Qiao, Hu, Liu and Shu2024) for E. anchoratus and Sinergasilus undulatus (Markevich, 1940) providing additional evidence that supports the close relationship between those copepods in available phylogenies.
Finally, the novel sequences generated for R. piranhus and G. schizodontis formed a clade (clade V or Neotropical clade) in conjunction with A. margulisae. The latter species was described by Santacruz et al. (Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020) based on copepods found parasitizing 3 cichlids [Amphilophus citrinellus (Günther, 1864); Parachromis managuensis (Günther, 1867); and Oreochromis sp.] and 1 poecilid (Poecilia mexicana Steindachner, 1863) from a crater lake in Nicaragua. With the exception of Acusicola tenax (Roberts, Reference Roberts1965) which was originally described in Texas, USA (Roberts, Reference Roberts1965), the remaining Acusicola species occur exclusively in the Neotropical region (Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Couto et al., Reference Couto, Nazaré Pereira, Luque, Paschoal and Pereira2023). Based on the historical biogeography of freshwater Cyclopinae copepods (Suárez-Morales et al., Reference Suárez-Morales, Reid, Fiers and Iliffe2004), Acusicola probably also originated in South America and then spread northwards as a consequence of the faunal exchange in conjunction with the closure of the Panama gap during the Pliocene explaining the occurrence of this single species in the Nearctic region. Rhinergasilus and Gamispatulus are restricted to Brazil (see the checklists in Narciso and Silva, Reference Narciso and Silva2020; and Narciso et al., Reference Narciso, Brandão, Perbiche-Neves and Silva2020). The Neotropical clade was strongly supported in 28S analyses, while for 18S a similar clade was recovered but with low support. More sequences of Neotropical species are required to test the validity of clade V.
The genus Therodamas Krøyer, 1863 is also restricted to the Neotropical region. Like the other mesoparasitic genera (i.e. species whose adult females live embedded into the host tissues), Therodamas species are also easily recognized by their long ‘necks’ (El-Rashidy and Boxshall, Reference El-Rashidy and Boxshall2001b; Tang and Kalman, Reference Tang and Kalman2008). Despite their morphological similarities, the origin and/or relationships between mesoparatic taxa remain unclear in Ergasilidae – the mesoparasitism probably appeared independently at least twice in Ergasilidae given the non-homology of the necks (i.e. pre-oral neck in Mugilicola Tripathi, 1960 and Paeonodes Wilson C.B., 1944 vs post-oral neck in Majalincola Tang and Kalman, Reference Tang and Kalman2008 and Therodamas). Therodamas longicollum Oliveira et al., Reference Oliveira, Corrêa, Adriano and Tavares-Dias2021 is the only representative of the mesoparasitic ergasilids whose ribosomal sequences are available (unique sequence for 18S rDNA). In its original description, the authors recovered T. longicollum as the sister taxon of all other ergasilids with no close relationship with A. margulisae (i.e. the only Neotropical ergasilid with sequences available at that time). Our BI analyses also recovered this same basal position of T. longicollum. Interestingly, concerning the morphology of the swimming legs, all Therodamas species have the L1 with a 2-segmented enp, which is a common (and unique) diagnostic feature for all Neotropical species. On the other hand, the other 3 mesoparasitic genera, including Majalincola who shares the post-oral neck with Therodamas, remain with the L1 enp-3 with 3 segments. More data are necessary for a better understanding of the relationships between Therodamas and the other Neotropical ergasilids (or even among the 4 mesoparasitic genera). Current findings may indicate that (at least) 2 distinct lineages have colonized South America independently.
Overall, the clades found in our studies are consistent with those presented in previous phylogenetic analyses. The 5 well-supported clades observed herein can act as interesting targets for future revisions within this family, which would help to minimize big taxonomic problems like the ‘elephant in the room’ caused by the paraphyletic status of Ergasilus. However, more studies combining morphological and molecular analyses are still needed in order to achieve a better understanding of the evolutionary relationships within Ergasilidae – especially in the Neotropical region, which despite having the greatest known diversity of ergasilids in the world still has the least amount of available molecular sequences.
New records and host switching
Rhinergasilus piranhus was originally proposed based on copepods found inside the nostrils of the ‘red piranha’ P. nattereri Kner, 1858 (=Serrasalmus nattereri) from the Amazon region, Brazil. Since its description, this species has been continuously reported from the nostrils of different fishes across the country [see Table 4 in Narciso et al. (Reference Narciso, Brandão, Perbiche-Neves and Silva2020)]. A similar story also applies to G. schizodontis which was also found in the nostrils of an Amazonian fish, the anostomid S. fasciatus Spix and Agassiz, 1829 [see Table 5 in Narciso and Silva (Reference Narciso and Silva2020)]. Recently, Narciso (Reference Narciso2020), when evaluating the ergasilid fauna of fishes from 2 rivers in São Paulo State (Veados and Paranapanema rivers), reported Rhinergasilus sp.1 parasitizing the nostrils of Psalidodon fasciatus (Cuvier, 1815). Morphological analysis of the latter specimens together with comparisons with our copepods allowed us to identify these specimens as R. piranhus. Our study reaffirms the presence of R. piranhus and G. schizodontis in São Paulo (i.e. corroborating Narciso, Reference Narciso2020; and Narciso and Silva, Reference Narciso and Silva2020) and expands their distribution to a new river. Furthermore, it also broadens the list of fish hosts for both species – i.e. 5 new hosts for R. piranhus (i.e. including P. fasciatus reported by Narciso, Reference Narciso2020) and 1 for G. schizodontis.
The occurrence of R. piranhus in the nostrils of Cy. modestus and Co. rendalli expands the number of taxa (beyond species level) that act as hosts for this copepod: (i) 2 new families – Curimatidae Gill, 1858 and Cichlidae Bonaparte, 1835; and (ii) 1 new order – Cichliformes. The low parasitological indices observed for both hosts may characterize this occurrence as an erratic parasitism (i.e. when the parasite is found outside its usual host) (see Table 5). Separating each fish into individual plastic bags prevented those occurrences happened as a result of cross-infection. In contrast to other fish ectoparasites (mainly monogeneans), ergasilids tend to have a lower specificity regarding their hosts (Boxshall and Halsey, Reference Boxshall and Halsey2004). Even though it may be erratic, the occurrence of this copepod (if maintained) can give rise to new lineages that may undergo speciation in the future (Araujo et al., Reference Araujo, Braga, Brooks, Agosta, Hoberg, Von Hartenthal and Boeger2015). The occurrence of R. piranhus in Co. rendalli, which is an exotic fish, may be evidence of host switching since this parasite has never been reported outside the country.
This latter mechanism has already been observed in other ergasilids [e.g. evidence of Sinergasilus major (Markevich, 1940) switching hosts in Hungary – see Dos Santos et al., Reference Dos Santos, Avenant-Oldewage, Piasecki, Molnár, Sellyei and Székely2021] and the acquisition of native parasites by exotic species is a well-documented phenomenon in Ichthyoparasitology (Paterson et al., Reference Paterson, Townsend, Tompkins and Poulin2012, Reference Paterson, Rauque, Fernandez, Townsend, Poulin and Tompkins2013). Like the introduction of alien parasites, the acquisition of native parasites by introduced hosts can also be a problem for the local ecosystem by altering the native host–parasite dynamics, resulting in spillback or even dilution of the native infection (Kelly et al., Reference Kelly, Paterson, Townsend, Poulin and Tompkins2009). However, more studies are needed to confirm this parasite–host relationship and even the possible impacts of this association on the local fauna of Pardo River.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S003118202400129X.
Data availability statement
Sequence data are available on the NCBI GenBank database. All other necessary data are included in the article and its supplementary materials.
Acknowledgements
We thank the following agencies for the financial support to this study: Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). We thank Eduardo Meneguzzi Brambilla for his help in identifying the fish. Finally, we also thank Linda van der Spuy, North-West University for her help with the scanning electron microscopy analyses performed in this study.
Author contributions
All authors contributed to the study's conception and design. Material preparation, data collection and analysis were performed by R. B. N. The first version of the manuscript was written by R. B. N. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Financial support
We thank the following agencies for the financial support to this study: Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP – #2020/05412-9; and #2019/26831-2), and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq – 311635-2021-0). Molecular work at the North-West University Water Research Group was partially funded by the National Research Foundation (NRF) of South Africa (grant number 138573). Opinions, findings, conclusions and recommendations expressed are those of the authors, and the NRF accepts no liability whatsoever in this regard.
Competing interests
None.
Ethical standards
All applicable international, national and institutional guidelines for the care and use of animals were followed. Fish sampling was authorized by the Instituto Chico Mendes de Conservação da Biodiversidade – ICMBio and Sistema de Autorização e Informação em Biodiversidade – SISBIO # 60640-1 and all procedures followed the recommendations of the Ethical Commission for Animal Experimentation from the São Paulo State University (Unesp), Institute of Biosciences, Botucatu, Brazil (CEUA no 9415260520).