Introduction
Long-term trophectoderm (TE) cell culture provides a valuable platform for the investigation of the dynamic regulation of placenta-specific factors during early pregnancy, enabling the characterization of key placental components across developmental stages. Products secreted by binucleated cells (BNCs), such as pregnancy-associated glycoproteins (PAGs), placental lactogen, and prolactin-related proteins, are critical for pregnancy diagnosis and monitoring placental function (Green et al., Reference Green, Parks, Avalle, Telugu, McLain, Peterson, McMillan, Mathialagan, Hook, Xie and Roberts2005; Pohler et al., Reference Pohler, Geary, Johnson, Atkins, Jinks, Busch, Green, MacNeil and Smith2013, Reference Pohler, Green, Geary, Peres, Pereira, Vasconcelos, Smith, Geisert and Bazer2015). Furthermore, long-term TE culture is particularly advantageous for studying ruminant placental development through the expression of TE-specific markers and interferon-stimulated genes, including interferon tau (IFNT), which plays a central role in maternal recognition of pregnancy (Thatcher et al., Reference Thatcher, Bartol, Knickerbocker, Curl, Wolfenson, Bazer and Michael Roberts1984; Wiltbank et al., Reference Wiltbank, Sartori, Herlihy, Vasconcelos, Nascimento, Souza, Ayres, Cunha, Keskin, Guenther and Gumen2011; Dorniak et al., Reference Dorniak, Bazer and Spencer2013). This system also permits characterization of the temporal dynamics of synthesis and secretion of these factors, advancing our understanding of early placental signalling.
TE cell culture relies on two essential components: the culture medium and the extracellular matrix (ECM). For in vitro cultures to fully support cell growth and differentiation, conditions must be carefully optimized to resemble those in vivo, particularly the structural and biochemical functions of the ECM. There is evidence of feeder cells and Matrigel (Yang et al., Reference Yang, Fields, Zhang, Ozawa, Johnson and Ealy2011, Reference Yang, Liu, Xu, Wang, Wu, Shi, Xu, Dong, Wang, Lai, Zhu, Xiong, Zhu, Li, Yang, Yamauchi, Sugawara, Li, Sun, Li, Li, He, Du, Wang, Zhao, Li, Chi, Zhang, Liu, Li, Duo, Yin, Shen, Belmonte and Deng2017) used as a source of ECM capable of supporting TE cell proliferation (Talbot et al. Reference Talbot, Caperna, Edwards, Garrett, Wells and Ealy2000; Bai et al., Reference Bai, Sakurai, Someya, Konno, Ideta, Aoyagi and Imakawa2011) by working as a signalling centre and structural framework. Using substrates that promote cell adhesion, migration, and differentiation is one way to mimic and enhance ECM interactions in vitro, thereby enabling tissue-like structure and function (Fitzpatrick and McDevitt, Reference Fitzpatrick and McDevitt2015).
A recent advancement in an in vitro model for bovine TE differentiation utilizes stromal cell-conditioned medium and collagen IV-coated plates (Ortega et al., Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022), which are suitable conditions for TE cell growth. However, they can be costly, limiting the capacity to conduct extensive, long-term studies. Thus, the goal of this study was to develop cost-effective TE cell culture media along with a more affordable ECM substitute to successfully culture bovine TE cell lines, resulting in successful proliferation and differentiation.
Materials and methods
Cell culture media preparation
Base DMEM/F-12
Dulbecco’s Modified Eagle Medium Nutrient Mixture F-12 (DMEM/F-12; Gibco, Waltham, Massachusetts, US) powder media was prepared according to the manufacturer’s instructions. Medium was supplemented with 2438 mg/L sodium bicarbonate (Sigma Aldrich, St. Louis, MO, USA), 0.864 mg/L zinc sulphate (Sigma Aldrich), 400 mg/L bovine serum albumin (Sigma Aldrich), 1.9 mg/L ethanolamine (Sigma Aldrich), and 55 mg/L sodium pyruvate (Sigma Aldrich).
Modified DMEM/F-12
Base DMEM/F-12 was further supplemented with 0.5X B-27 supplement (Gibco, 12587-010), 100X insulin-transferrin-selenium (Gibco, 41400-045), 100X antibiotic-antimycotic solution (Gibco, 15240062), 1X Glutamax (Gibco, 35050-061), 1X nonessential amino acids (Sigma Aldrich, M7145), 0.1 mM 2-mercaptoethanol (Gibco, 21985023), and 0.10% bovine serum albumin (Sigma Aldrich, A4737).
Trophectoderm culture medium
Trophectoderm culture medium (TE medium) consisted of modified DMEM/F-12 (Section 2.1.2), which already contains B-27, insulin-transferrin-selenium, GlutaMAX, non-essential amino acids, and bovine serum albumin, further supplemented with 0.5× N-2 supplement (Gibco), 1.25 mM N-acetyl-L-cysteine (Millipore Sigma), 0.0005 M A83-01 (Tocris Biosciences), 500 ng/mL epidermal growth factor (EGF; PeproTech), 25 ng/mL FGF-10 (PeproTech), 20 ng/mL R-spondin (PeproTech), 20 ng/mL Noggin (PeproTech), and 10 mM nicotinamide (Millipore Sigma). This formulation is based on a previously described expansion medium used in organoid (Turco et al., Reference Turco, Gardner, Hughes, Cindrova-Davies, Gomez, Farrell, Hollinshead, Marsh, Brosens, Critchley, Simons, Hemberger, Koo, Moffett and Burton2017; Fitzgerald et al., Reference Fitzgerald, Dhakal, Behura, Schust and Spencer2019) and bovine TE outgrowth culture systems (Ortega et al., Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022).
Tissue culture gelatine coating
Gelatine from bovine skin (Sigma–Aldrich, G9391) was used to prepare a 0.1% (w/v) gelatine solution in water. The gelatine solution was autoclaved at 121°C and 15 psi for 30 min to ensure sterilization, and then stored at 4°C until use. When needed, the gelatine solution was warmed and used to coat the surface of 33 mm Petri dishes with 600 μl per well under a sterile hood, and the dishes were allowed to settle for 2 h at room temperature. The volume can be adjusted depending on the surface area of the plate. Following incubation time, the remnant solution was aspirated and discarded, and plates were rinsed with sterile 1X PBS.
In vitro production of bovine embryos
All media and supplements used for embryo production were prepared as previously described (Ortega et al., Reference Ortega, Tribulo, Siqueira, Null, Cole, Da Silva and Hansen2017b; Tríbulo et al., Reference Tríbulo, Rivera, Ortega Obando, Jannaman, Hansen and Herrick2019a), and embryo production was done following standard procedures (Ortega et al., Reference Ortega, Tribulo, Siqueira, Null, Cole, Da Silva and Hansen2017b, Reference Ortega, Moraes, Patterson, Smith, Behura, Poock and Spencer2018, Reference Ortega, Kelleher, O’Neil, Benne, Cecil and Spencer2020, Reference Ortega, Kelleher, O’Neil, Benne, Cecil and Spencer2020). Briefly, cumulus oocyte complexes (COCs) were retrieved from slaughterhouse-derived ovaries, and groups of 50 COCs were placed in individual wells containing 500 μl of oocyte maturation medium (OMM) composed of Tissue Culture Medium-199 with Earle’s salts (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS) (Sigma Aldrich), 2 µg/mL oestradiol-17β (Sigma Aldrich), 25 µg/mL Porcine purified follicle-stimulating hormone (FSH; Folltropin; Vetoquinol, Fort Worth, TX, USA), 22 µg/mL sodium pyruvate (Sigma Aldrich), 50 µg/mL gentamicin (Gibco), and 1 mM glutaMAX (Tríbulo et al., Reference Tríbulo, Rivera, Ortega Obando, Jannaman and Hansen2019b) and overlaird with 300 µl of mineral oil (FUJIFILM Irvine Scientific, Santa Ana, CA, USA) and matured at 38.5 °C, 5% CO2, and humidified air.
Fertilization medium was Tyrode’s albumin lactate pyruvate fertilization medium (IVF-TALP), and culture medium was synthetic oviduct fluid culture medium (SOF-BE2) media preparation followed that from established IVF protocols (Tríbulo et al., Reference Tríbulo, Rivera, Ortega Obando, Jannaman and Hansen2019b; Stoecklein et al., Reference Stoecklein, Ortega, Spate, Murphy and Prather2021). For fertilization, sperm were purified from frozen-thawed straws using a gradient (Isolate, FUJIFILM Irvine Scientific) and diluted to a final concentration of 1 × 106 in a 35 mm Petri dish containing 1.7 mL IVF-TALP and 80 µL of penicillamine-hypotaurine-epinephrine (PHE) solution per dish containing expanded COCs. Fertilization occurred over a 16 h period at 38.5°C, 5% CO2, and humidified air.
Putative zygotes (oocytes exposed to sperm), were denuded from their cumulus cells by vortexing in 100 μl of hyaluronidase (10,000 units/mL) for 5 min and placed in pre-equilibrated wells containing 500 μl SOF-BE2 culture medium overlaid with 300 μl of mineral oil in a humidified atmosphere containing 5% O2 and 5% CO2 for 8 days. Cleavage rate was recorded on day 3 post-fertilization, and blastocyst development on day 7.5. On day 8, embryos that reached the blastocyst stage were transferred to SOF-BE2 supplemented with 10% FBS for 2 days to promote hatching from the zona pellucida. Whole blastocysts were utilized in every experiment and embryos produced from multiple bulls were distributed across replicates and treatments to minimize potential side effects. While no lineage-specific immunostaining was performed, Ortega et al. (Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022), using similar outgrowth systems, have demonstrated that adherent outgrowths under these conditions are predominantly TE-derived, as the medium components do not support maintenance of other cell lineages.
Extracellular matrix comparison
Groups of 5 hatched, day 10 blastocysts were placed in either a collagen IV-coated plate or in a 0.1% gelatine-coated plate using 2 mL of TE medium. The day of attachment was recorded during the culture period from days 10 to 35, with half of the medium being replaced every other day. Attachment was assessed by a gentle swirling motion; those that remained steady were considered attached. Experiments were conducted over 3 independent replicates.
Trophectoderm cell culture using base DMEM/F-12
To ensure the base DMEM/F-12 medium was suitable for TE cell culture, 15 blastocysts were cultured individually in commercial Advanced DMEM/F-12, serving as a control, and 15 blastocysts were cultured individually in the base DMEM/F-12 over 3 independent replicates. During the culture period (days 10–20), the day of attachment and the TE growth area were recorded. Half of the medium was replenished every other day throughout the culture period.
Bi-phasic trophectoderm cell culture using TE media
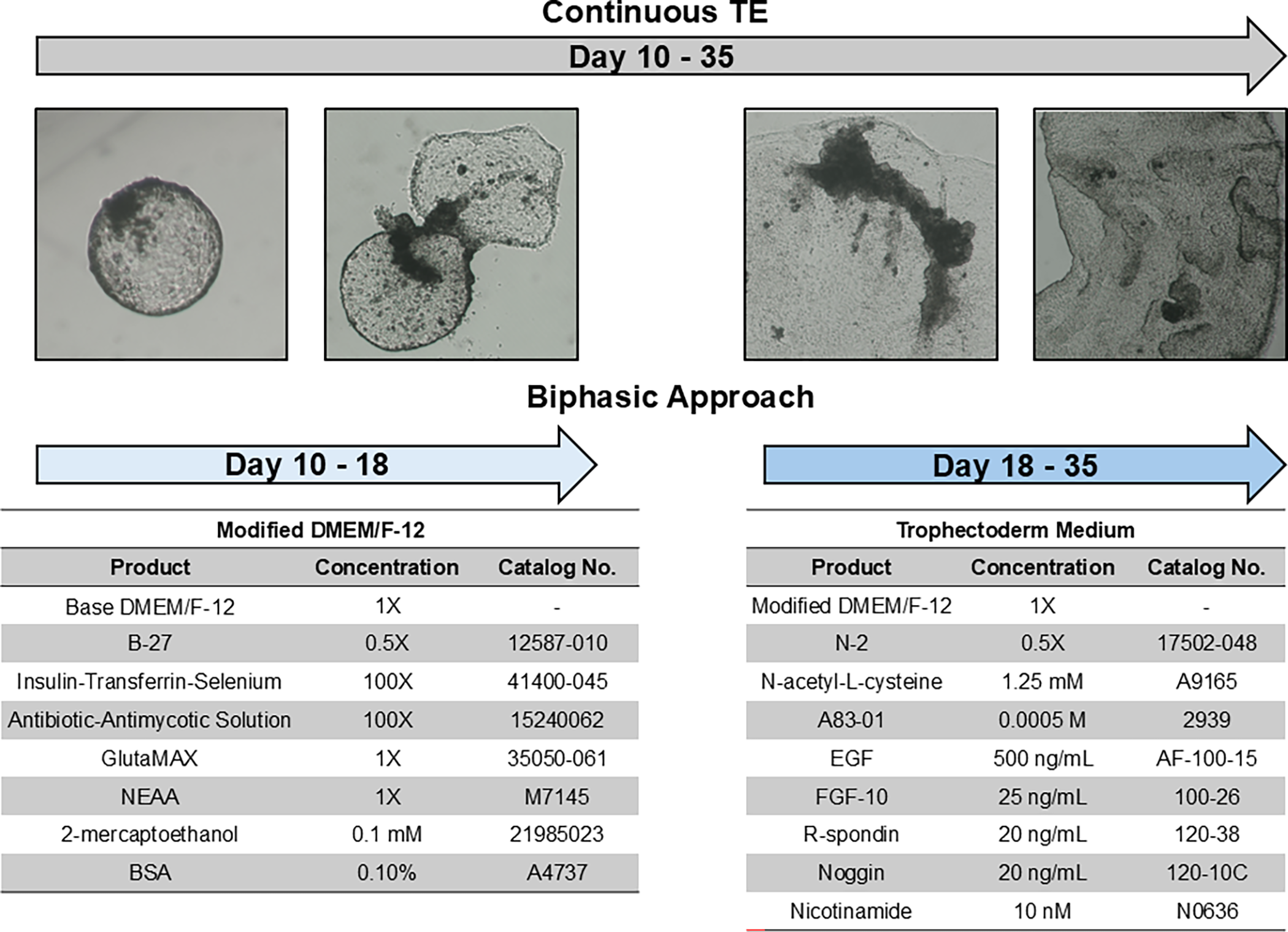
To determine whether reducing exposure to growth factor-rich medium during early culture could decrease costs without affecting TE development, a bi-phasic culture strategy was evaluated. In the experimental group, embryos were cultured in modified DMEM/F-12 from day 10 to day 18, followed by trophectoderm culture medium (TE medium) from day 18 to day 35. In contrast, embryos in the control group were cultured in TE medium continuously from day 10 to day 35 (Continuous TE; Figure 1). In all treatments, half of the medium volume in each well was replaced every other day.
Experimental design of biphasic and continuous trophectoderm (TE) culture strategies. In the biphasic approach, embryos were cultured in modified DMEM/F-12 from days 10 to 18 post-insemination, followed by complete TE medium from days 18 to 35. In the continuous TE group, embryos were cultured in TE medium for the entire culture period (days 10–35). Day 0 = day of insemination.

A total of 47 TE cell outgrowths were evaluated in the control group and 54 in the experimental bi-phasic group across three independent replicates. Representative images for each medium can be found in Supplemental Figure 1. The growth area of TE outgrowths was assessed by capturing images every two days throughout the culture period and measuring the outgrowth area using ImageJ version 1.53 (National Institutes of Health, Bethesda, MD, USA).
Quantitative PCR analysis
Placental and differentiation markers such as placenta-specific 8 gene (PLAC8), GATA-binding factor 2 (GATA2), and pregnancy-associated glycoprotein 7 (PAG7) were measured and compared between our TE cells outgrowths cultured in collagen IV and 0.1% gelatine-coated plates using specific primers (Table 1) validated following previously described procedures (Ortega et al., Reference Ortega, Kurian, McKenna and Hansen2017a). Gene expression was evaluated in TE outgrowths cultured in commercially available Advanced-DMEM/F-12 media or modified-DMEM/F-12, as well as those cultured in TE media or expansion media. RNA concentration and purity were assessed using a NanoDrop spectrophotometer. Complementary DNA (cDNA) was synthesized from total RNA using the LunaScript SuperMix Kit (New England Biolabs). Quantitative PCR reactions were performed using SYBR Green qPCR Master Mix (GlpBio) in a CFX96 Real-Time PCR Detection System (Bio-Rad). All samples were analyzed in duplicate in 10 µL reactions consisting of 0.5 µL of 10 µM primers, 5 µL of supermix, 1 µL nuclease-free water, and 3 µL of cDNA using the following PCR conditions: activation at 95°C for 2 min; 40 cycles of 95°C for 5 s; 60°C for 30 s; and 72°C for 30 s. Gene expression was calculated relative to the reference housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Delta CT (ΔCT) was estimated by subtracting the CT (cycle threshold) value for the gene of interest from the CT value for the housekeeping gene, and fold change was estimated using the formula 2−ΔCT (Ortega et al., Reference Ortega, Kurian, McKenna and Hansen2017a).
Quantitative PCR primer sequences

* Designed and validated for this study.
Statistical analyses
Statistical analyses were performed using SAS v9.4 (SAS Institute Inc., Cary, NC, USA). The embryo was considered the experimental unit. Day of attachment and gene expression data were analyzed using one-way ANOVA. TE growth area, which was measured longitudinally throughout culture (days 10–35), was analyzed using repeated measures ANOVA with treatment as a fixed effect, replicate as a random effect, and day as the repeated factor. Model assumptions were evaluated by examining residual plots for normality and homogeneity of variance, and no major deviations were observed. Differences among means were determined using the pdiff option of LSMEANS, and statistical significance was set at P ≤ 0.05.
Results
Gelatine-coated plates can be used to culture TE cells
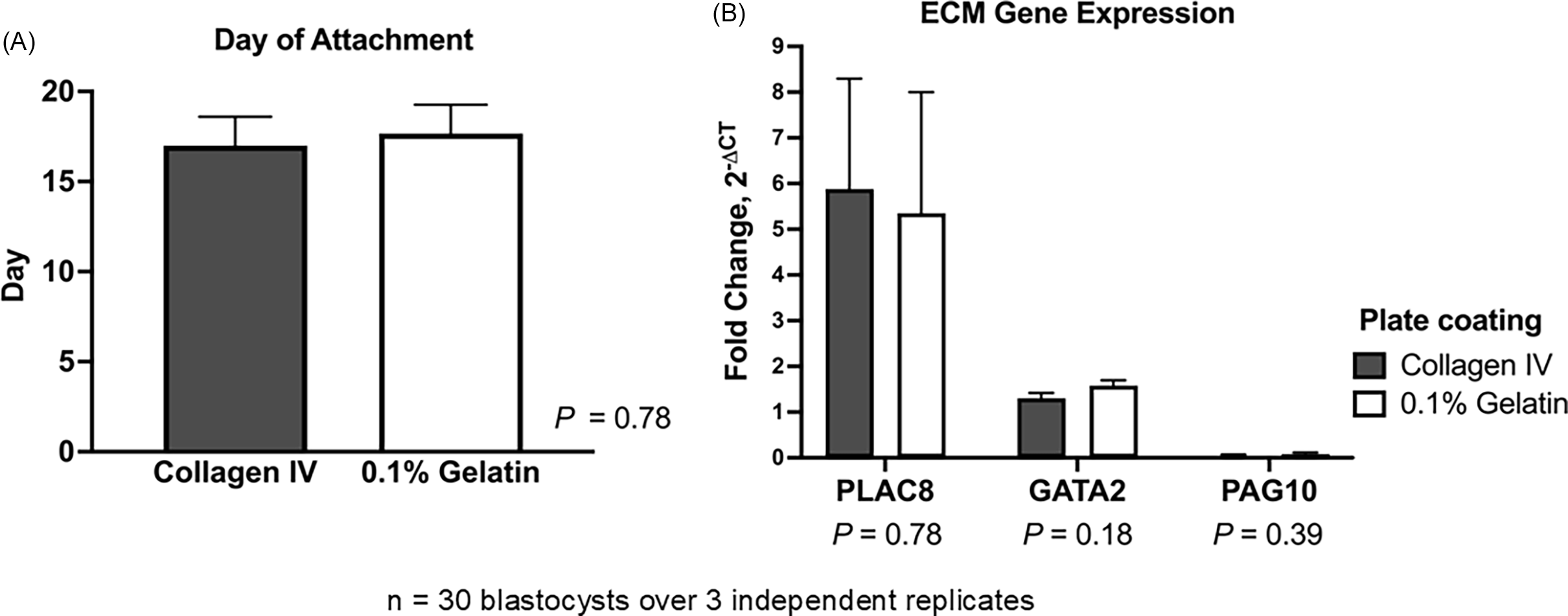
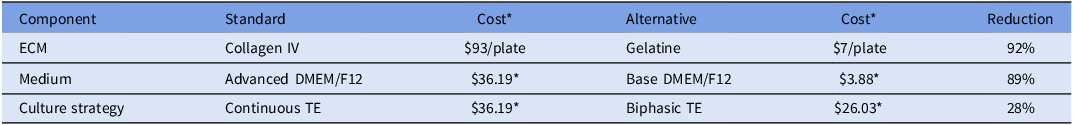
As a procedure previously employed to cultivate embryonic and TE stem cells, bovine TE cell outgrowths were cultured on plates coated with 0.1% gelatine (Pinzón-Arteaga et al., Reference Pinzón-Arteaga, Wang, Wei, Ribeiro Orsi, Li, Scatolin, Liu, Sakurai, Ye, Ming, Yu, Li, Jiang and Wu2023). Placental (PLAC8) and differentiation markers (GATA2 and PAG10) were compared between TE cell outgrowths (n = 5 blastocysts/plate, throughout 3 replicates) cultured on the two types of matrices to determine whether they can grow, proliferate, and differentiate in 0.1% gelatine-coated plates in the same manner as on collagen IV-coated plates (Ortega et al., Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022). Embryos cultured on collagen IV- and gelatine-coated plates did not differ significantly in attachment day (P = 0.78). In both matrices, embryos normally attached between days 15 and 18, considering day 0 as the day of insemination (Figure 2A). Expression of PLAC8 (P = 0.78), GATA2 (P = 0.18), and PAG10 (P = 0.39) did not change between the two types of matrices (Figure 2B). Using 0.1% gelatine ($7/24-well plate) instead of collagen IV-coated plates ($93/24-well plate) can reduce costs by over 90% while supporting comparable TE cell attachment and growth.
Effect of extracellular matrix substrate on bovine trophectoderm (TE) outgrowth. Hatched blastocysts (n = 30 over 3 independent replicates) were cultured on collagen IV- or 0.1% gelatine-coated plates. (A) Day of embryo attachment. (B) Relative mRNA expression of placental marker PLAC8 and differentiation markers GATA2 and PAG10 in TE outgrowths. Gene expression was normalized to GAPDH and calculated as fold change using 2−ΔCT. Data are presented as LSMEANS ± SEM.
TE cells can be cultured using base DMEM/F-12
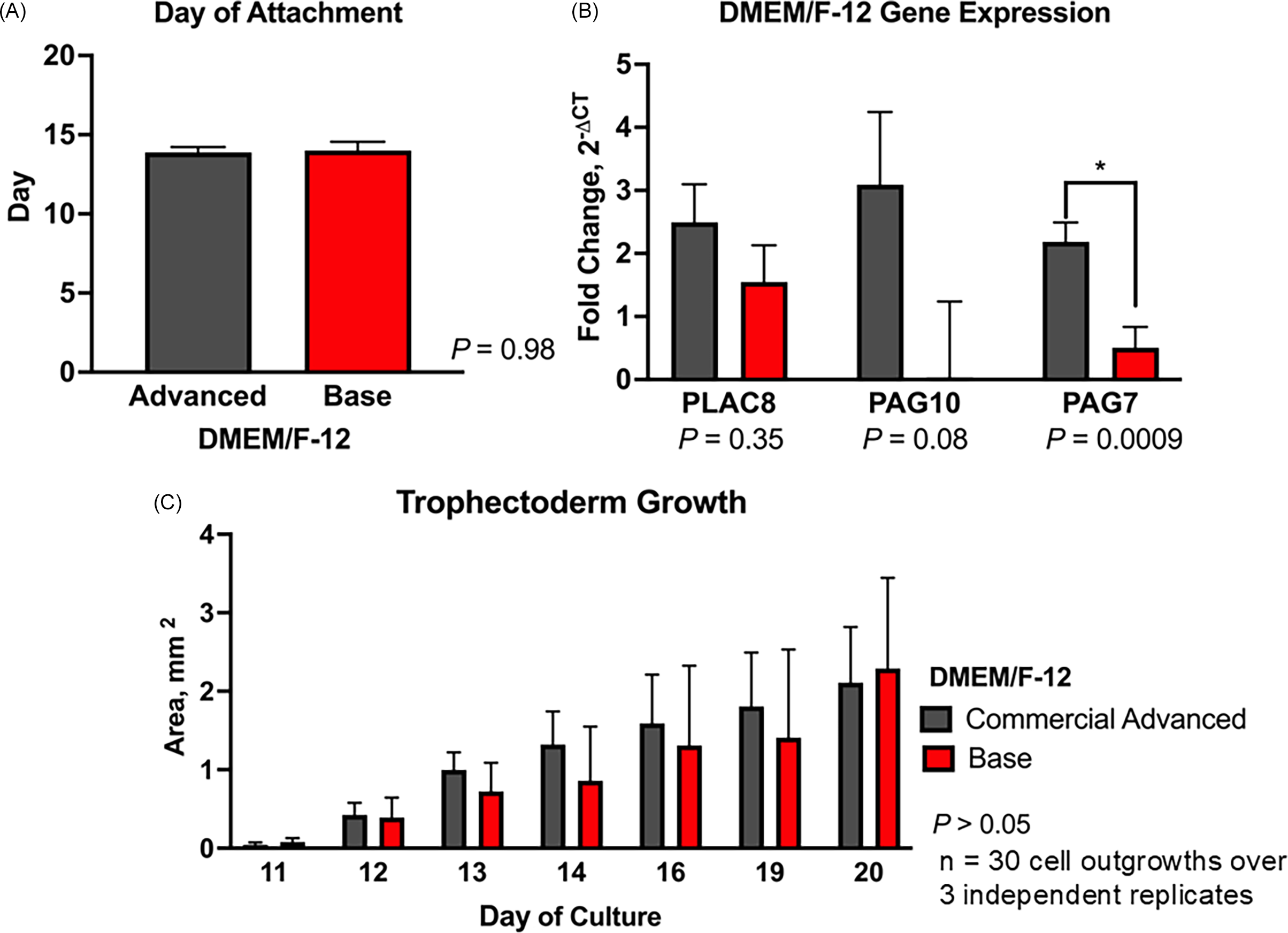
The day of attachment of embryos cultured with either commercial Advanced-DMEM/F-12 or base DMEM/F-12 did not differ (P = 0.98; Figure 3A). Despite variability, PLAC8 (P = 0.35) and PAG10 (P = 0.08) expression remained unchanged between the cell lines cultured with either commercial Advanced DMEM/F-12 or base DMEM/F-12. However, PAG7 expression was higher in embryos cultured in commercial medium compared to base DMEM/F-12 (2.18 ± 0.31 vs 0.51 ± 0.33 fold relative to GAPDH; P < 0.05; Figure 3B). Additionally, there was no difference (P > 0.05) in the growth area of TE cell lines cultured with the commercial Advanced DMEM/F-12 and the base DMEM/F-12 throughout the culture period (day 10–day 35) (Figure 3C). Using base DMEM/F-12 can reduce media costs by approximately 90% compared to commercial Advanced DMEM/F-12 ($3.88 vs. $36.19 per 100 mL), with no impact on TE cell growth or gene expression.
Effect of culture medium on bovine trophectoderm (TE) outgrowth. Hatched blastocysts (n = 30 over 3 independent replicates) were cultured individually in commercial advanced DMEM/F-12 or base DMEM/F-12. (A) Day of embryo attachment. (B) Relative mRNA expression of PLAC8, PAG7, and PAG10. Gene expression was normalized to GAPDH and calculated as fold change using 2−ΔCT. (C) Trophectoderm outgrowth area (mm2) from days 10 to 20. Data are presented as LSMEANS ± SEM. Asterisk (*) indicates P < 0.05.

Trophectoderm cells can be cultured in bi-phasic conditions using TE media
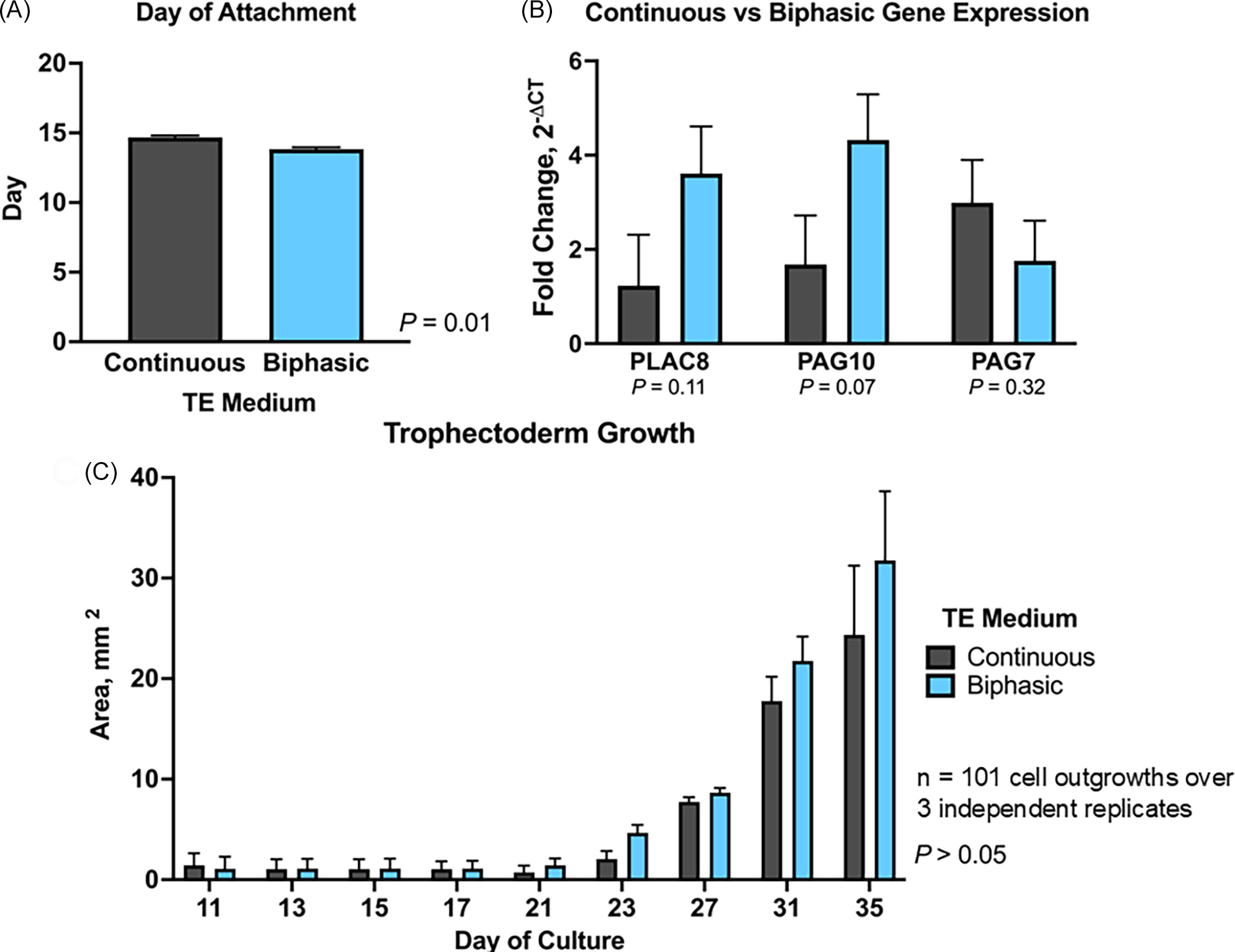
Embryos cultured in continuous TE took approximately 1 day longer to attach compared to those cultured in bi-phasic conditions with TE media (P = 0.01; Figure 4A). The expression of PLAC8 (P = 0.11), PAG7 (P = 0.32), and PAG10 (P = 0.07) did not change between TE cells cultured in the bi-phasic conditions with TE media and those cultured in continuous TE, despite variability in expression (Figure 4B). Additionally, there was no difference (P > 0.05) in TE cell growth area between cell lines cultured in bi-phasic conditions with TE media and those in continuous TE throughout the culture period (day 10–day 35) (Figure 4C). Implementing the bi-phasic TE media approach, which combines modified DMEM/F-12 from days 10–18 followed by complete TE medium through day 35, can reduce overall media costs by approximately 30% compared to continuous TE ($26.03 vs, $36.19 per 100 mL), without affecting TE cell attachment, growth, or gene expression.
Effect of biphasic versus continuous trophectoderm (TE) culture on outgrowth development. TE cell outgrowths (n = 101 over 3 independent replicates) were cultured in either biphasic (modified DMEM/F-12, days 10–18; TE medium, days 18–35) or continuous TE medium (days 10–35). (A) Day of embryo attachment. (B) Relative mRNA expression of PLAC8, PAG7, and PAG10. Gene expression was normalized to GAPDH and calculated as fold change using 2−ΔCT. (C) Trophectoderm outgrowth area (mm2) throughout the culture period (days 10–35). Data are presented as mean ± SEM. Asterisk (*) indicates P < 0.05.

Discussion
Establishing a cost-effective and scalable TE culture system is critical for advancing bovine reproductive research, particularly in modelling early placental development. This study demonstrates that a bi-phasic approach using TE media and an alternative ECM, such as gelatine, can support TE cell attachment, proliferation, and differentiation without compromising molecular integrity.
While other standard matrices for culture, such as matrigel, fibronectin, or collagen, allow development of in vitro cell culture (Bratt-Leal et al., Reference Bratt-Leal, Nguyen, Hammersmith, Singh and McDevitt2013; Qiang et al., Reference Qiang, Jin, Lijun, Shimin, Yanqin and Sam2013; Wang et al., Reference Wang, Yu, Zhou, Zhang, Zhou, Hao, Ding, Li, Gu, Ma, Qiu and Ma2022), other more cost-effective options, such as gelatine, have been recommended (Mariod and Adam, Reference Mariod and Adam2013). Its application in tissue engineering scaffolds and cell culture plates enhances cell adhesion and viability (Montero et al., Reference Montero, Vial, Nguyen, Farhand, Reardon, Pham, Tsechpenakis and Andreopoulos2012; Bratt-Leal et al., Reference Bratt-Leal, Nguyen, Hammersmith, Singh and McDevitt2013). Extended pluripotent stem cells (EPSCs) and trophectoderm stem cells (TSCs) have previously been successfully maintained on 0.1% gelatine-coated dishes (Pinzón-Arteaga et al., Reference Pinzón-Arteaga, Wang, Wei, Ribeiro Orsi, Li, Scatolin, Liu, Sakurai, Ye, Ming, Yu, Li, Jiang and Wu2023). Collagen IV-coated plates, the current standard ECM, have a cost of approximately $93 per 24-well plate. By contrast, the cost of a 24-well plate coated with 0.1% gelatine is approximately $7. The results of the present study suggest that the substitution of collagen IV with gelatine effectively reduces ECM-associated costs without compromising TE cell adhesion or proliferation.
Here, 0.1% gelatine was shown to support bovine TE outgrowth compared to collagen IV, with no significant differences in embryo attachment timing or expression of key placental markers, PLAC8, GATA2, and PAG10. This is a significant finding given the cost reduction, reducing hurdles to large-scale experimentation. Similarly, media substitutions did not have an effect on attachment or proliferation. Comparable gene expression was observed in PLAC8 and PAG10 between embryos cultured in commercial and base medium, while PAG7 expression was higher in commercial medium, suggesting a modest treatment-associated difference. The proposed culture system maintains functional outcomes while offering broader accessibility. This enables longer-term studies of trophoblast behaviour and signalling pathways, such as those involving interferon tau (IFNT), a critical signal for maternal recognition of pregnancy, and placental-derived products such as PAGs.
This work builds on previously established bovine TE cell outgrowth models (Ortega et al., Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022), which utilized collagen IV and conditioned medium from stromal cells (Hashizume et al., Reference Hashizume, Shimada, Nakano, Takahashi, Soares and Hunt2006). Although lineage-specific immunostaining was not performed to exclude epiblast or hypoblast contamination, prior characterization of this outgrowth system under identical conditions confirmed that adherent outgrowths are predominantly TE-derived (Ortega et al., Reference Ortega, Rizo, Drum, O’Neil, Pohler, Kerns, Schmelze, Green and Spencer2022). In addition to cost-effective ECM alternatives, this study evaluated the substitution of commercial Advanced DMEM/F-12 with a supplemented powdered DMEM/F-12 formulation. This was significantly more cost-effective, at $0.78 per 100 mL compared to $13.70 for the commercial formulation (Table 2). By incorporating the same additives as Advanced DMEM/F-12, a cost-efficient base DMEM/F-12 was developed that supported comparable TE cell growth and gene expression. A biphasic media approach was developed to further optimize results and costs, involving modified DMEM/F-12 used from days 10–18 post-fertilization, followed by a transition to complete TE culture medium through day 35 of culture. This strategy reflects an intentional reduction in the use of costly components during the early culture stages, while maintaining conditions necessary for continued proliferation and differentiation. Implementing the bi-phasic TE media cost was calculated at $26.03 per 100 mL compared to $36.19 per 100 mL of continuous TE used for the entirety of culture period. The biphasic strategy was designed to limit the use of growth factor-rich medium during the early attachment phase (days 10–18), when embryos are establishing TE outgrowths, while maintaining the same differentiation-supporting conditions during later stages of TE expansion.
Cost summary of standard and alternative culture practices
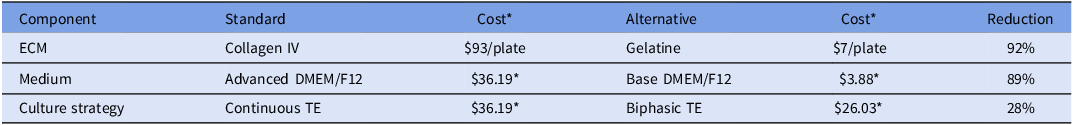
* Cost per 100 mL as of November 2025.
Collectively, these results provide strong evidence for the adoption of modified DMEM/F-12 and gelatine ECM as effective and affordable components of TE culture. For example, for a set of 24 TE outgrowths, using these media and ECM alternatives reduced overall culture costs by more than 70% compared with traditional approaches. This framework supports not only cost-conscious experimentation but also the feasibility of conducting higher outcome studies to investigate regulatory mechanisms governing early conceptus development. Future studies should evaluate additional markers, including interferon-stimulated genes (ISGs), and the downstream effects on epigenetic regulation and signalling.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S096719942610046X.
Acknowledgements
This work was supported by Agriculture and Food Research Initiative Competitive Grant no. 2021-67015-33675 from the USDA National Institute of Food and Agriculture. The authors thank Byron Chasi and Everardo Anta for their assistance with oocyte retrieval for embryo production.
Competing interests
The authors declare no conflict of interest.