Introduction
Rationale
Migraine is common, with a worldwide prevalence ranging between 8% and 18%.Reference Rasmussen1–Reference Falavigna, Teles and Velho5 Migraine impacts a person’s quality of life not only during the attack but also inter-ictally.Reference Lipton, Bigal, Kolodner, Stewart, Liberman and Steiner6–Reference Hu, Markson, Lipton, Stewart and Berger12 Migraine is ranked 2nd among all health disorders in terms of years lived with disability by the Global Burden of Disease.Reference Vos, Abajobir and Abate13 Migraine also results in significant direct and indirect costs to society.Reference Hu, Markson, Lipton, Stewart and Berger12,Reference Goldberg14
The Canadian Headache Society (CHS) Guideline for Migraine Prophylaxis was published in 2012.Reference Pringsheim, Davenport and Mackie15 The primary objective of this guideline was to assist the practitioner in choosing an appropriate prophylactic medication for an individual with episodic migraine, based on current evidence in the medical literature and expert consensus.
Since that time, there have been multiple new randomized controlled trials (RCTs) with new agents including onabotulinumtoxinA, the anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs), oral CGRP antagonists (gepants) and other oral therapies. Therefore, the decision was made to update the CHS migraine prophylaxis guidelines for episodic and chronic migraine.
This guideline is divided into two parts. Part 1 consists of evidence-based recommendations.
Part 2 comprises treatment strategies based on expert opinion.
Part 1: Evidence-Based Recommendations
Objectives
The systematic review and pairwise meta-analysis had the objective of synthesizing new randomized clinical trials and further characterizing the preventive treatment response of both old and new migraine preventive therapies in adults.
All available data of relevance to clinicians was summarized, and expert guidance on utilization of therapies for migraine prophylaxis was provided through a consensus process. This guidance is intended for Canadian neurologists and primary care providers to have an approach for managing migraine prevention guided by a systematic synthesis and interpretation of the literature available in 2023.
Methods
Population, intervention, comparator, outcome, study design question
RCTs were identified that involved adults with episodic and chronic migraine, where a treatment was evaluated against a placebo or an accepted intervention.
We aimed to answer the following questions:
-
1. Have newer therapies identified since the last guidelines shown efficacy and safety in the prevention of episodic and chronic migraine when compared to placebo or active comparators?
-
2. Is there new evidence, likely to change our previous recommendations, regarding the efficacy and safety of previously identified therapies in the preventive treatment of episodic migraine?
Eligibility criteria
The population included adults ≥18 years of age who met the International Headache Society criteria for episodic or chronic migraine. The criteria could be current or previous versions of International Classification of Headache Disorders (ICHD) criteria; specifically, we allowed ICHD-2, ICHD3 beta and ICHD3.16–18
The studies evaluated were prospective, randomized, double-blind, controlled trials (RCT), comparing a treatment to a placebo or to an active control. The active control had to be a medication known to be effective in migraine as evidenced by inclusion in previous guidelines. Both randomized parallel group and cross-over designs were allowed. This guideline is restricted to pharmacologic interventions. Notably, we did not review behavioral interventions and neuro-modulation devices, which also have an evidence base for use in migraine.Reference Burch19 This could be the subject of a future guideline.
The panel reviewed any new data on interventions reviewed in the previous guideline. Additionally, new pre-defined interventions included onabotulinumtoxinA, anti-CGRP mAbs and oral CGRP antagonists, gepants. If our review identified an intervention not previously defined, it was brought to the Steering Committee for consideration. The following additional interventions were therefore included: memantine, levetiracetam, enalapril and melatonin.
Outcomes
Efficacy outcomes included a reduction in monthly migraine days and a 50% reduction in mean migraine days per month. Where migraine days were not reported, a reduction in headache days was used as a surrogate outcome.
Safety outcomes included percentage of patients reporting adverse events (AEs), serious AEs and withdrawal due to AEs.
Information sources and strategy
A systematic search strategy was developed by an experienced information specialist in consultation with the review team. A second experienced research librarian peer-reviewed the MEDLINE search prior to execution using the PRESS checklist.Reference McGowan, Sampson, Salzwedel, Cogo, Foerster and Lefebvre20 Using the multifile option and deduplication tool available on the Ovid platform, Ovid MEDLINE® ALL, Embase and Cochrane CENTRAL were searched. The search strategy employed a combination of controlled vocabulary (e.g., “migraine,” “calcitonin gene-related peptide”) and related key words (e.g., migraine, migraine prevention, anti-CGRP mAbs, erenumab). For additional information on the search and gray literature sources, please see Appendix 1. Articles found outside the main search were identified in the Preferred Peporting Items for Systematic Review and Meta-analyses (PRISMA) flow diagram.Reference Moher, Liberati, Tetzlaff and Altman21
Study records
Data management
The search strategy identified abstracts to extract from the databases. Duplicate citations were removed, and the abstracts were imported into Covidence, a Cochrane tool for systematic review management.
Selection process
All abstracts were reviewed independently by two reviewers (IM, SC), and potentially relevant citations were selected for full review. Any disagreements as to whether studies should be included were resolved by discussion. Where multiple publications were associated with an included study, those providing the most recent data and/or unique information regarding outcomes of interest were retained. The process of study selection is described using a PRISMA flow diagram in Appendix 1.
Risk of bias in individual studies
Risk of bias (ROB) assessments were carried out on each study independently by two review team members (IM and SC) using the Cochrane ROB 2 tool. If conflicts could not be resolved by discussion, ML or CS were available to cast a final vote.
Data collection process
Data extraction was performed independently by two reviewers (IM, SC), who compared their findings and reached agreement. Data to be gathered from each study included details regarding publication characteristics, aspects of design, participant enrollment criteria and demographics, setting, interventions compared, outcomes measured and AEs from all study arms.Reference Higgins and Green22 Data was recorded using a standardized worksheet that was piloted and refined at the beginning of the data abstraction process.
Data synthesis
Data synthesis was done by one author (IM), although the data used was extracted independently and verified by two team members (IM and SC). For more details on data synthesis, please consult Appendix 1. We analyzed the doses which showed the best treatment responses, and we reported all the outcomes based on those doses. Meta-analyses were performed using random effects models where possible. We did meta-analyses where there were multiple studies with the same treatments. For all analyses, data for episodic migraine and chronic migraine was analyzed separately. To assess for publication bias, funnel plots and comparison-adjusted funnel plots were planned if sufficient studies were available; however, these were not undertaken because we did not have any treatment with more than 10 studies.Reference Guyatt, Oxman and Montori23 Findings from the review are reported based upon updated guidance from the PRISMA.Reference Page, McKenzie and Bossuyt24
Confidence in cumulative evidence
A modified form of the Grading of Recommendations Assessment, Development and Evaluation (GRADE) process was used to determine confidence in evidence for each outcome. In this process, the evidence was analyzed based on various parameters of ROB (multiple types), consistency, directness, precision and publication bias.Reference Guyatt, Oxman and Akl25 This has been the standard for neurology guidelines.Reference Gronseth, Cox, Gloss and Merilliat26,27
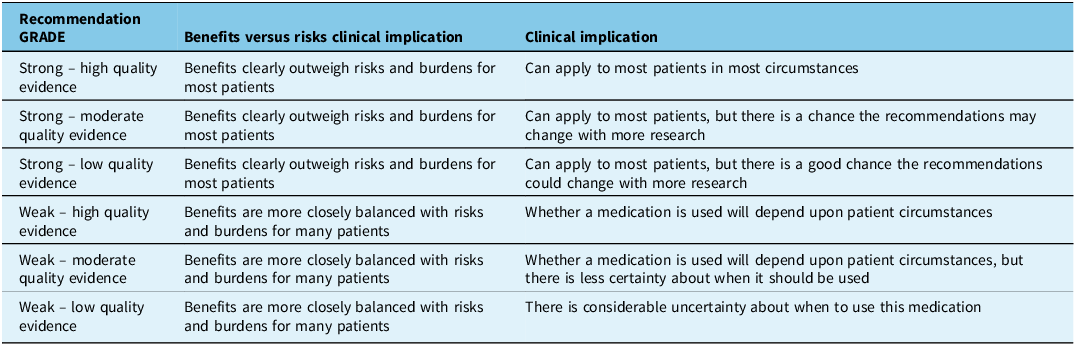
Across each intervention analyzed, we summarized all outcomes available from those we have prespecified by building a GRADE summary of finding tables,Reference Guyatt, Oxman and Akl25 created using the GRADE profiler (GRADEpro) software.28 Outcome importance was ranked a priori into the groupings of critical in all outcomes.Reference Guyatt, Oxman and Kunz29 The quality of evidence for all critical outcomes is reported in Appendix 2, but in this document, we report the lowest quality of evidence for critical outcomes. We identified the quality of evidence as high when we were confident the true effect was close to the estimate given, moderate when we felt that it was somewhat likely we were close to the true effect, low when we were not sure our estimate was close to the true effect and very low when we had little confidence in the effect estimate as we highlight in Table 1.Reference Pringsheim, Davenport and Mackie15,Reference Balshem, Helfand and Schünemann30
Level of evidence in GRADE

After arriving at the quality of evidence for the evidence base, a modified Delphi consensus processReference Hsu and Sandford31 with members of the Steering Committee and the Recommendation Committee consisting of a panel of experts within the CHS was undertaken to provide recommendations, using Welphi. Welphi is an online survey platform that implements the Delphi method. 32 Multiple rounds were circulated to the group until a 70% consensus was achieved.
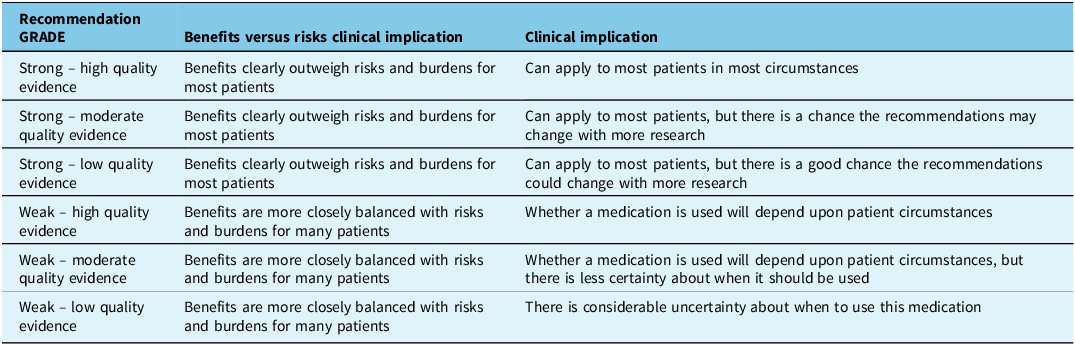
A strong recommendation was made when the Recommendation Committee members were confident that the intervention could be used for most patients and that the benefits of therapy outweigh the potential risks. A weak recommendation was made when the Recommendation Committee members were less confident that the desirable effects probably outweighed the undesirable effects. The treatment may be considered in some but may not be appropriate in others, and the consideration may depend on the patient and clinical situation. These categories are highlighted in Table 2.
GRADE recommendation and certainty of evidence explained
Results
In the abstract review stage, 4459 studies were reviewed from our search and 3 additional studies as detailed in the PRISMA flow diagram in Appendix 1. A total of 398 studies were excluded, and 442 full-length articles were reviewed. For this review, 381 studies were excluded in the full-text stage, and the reasons for exclusion are detailed in the PRISMA flow diagram in Appendix 1. Finally, 61 studies were included in this evidence update. We outline these below in the text by treatment category, and their ROB is documented in Appendix 3. The meta-analyses and summary of findings tables are outlined in Appendix 2.
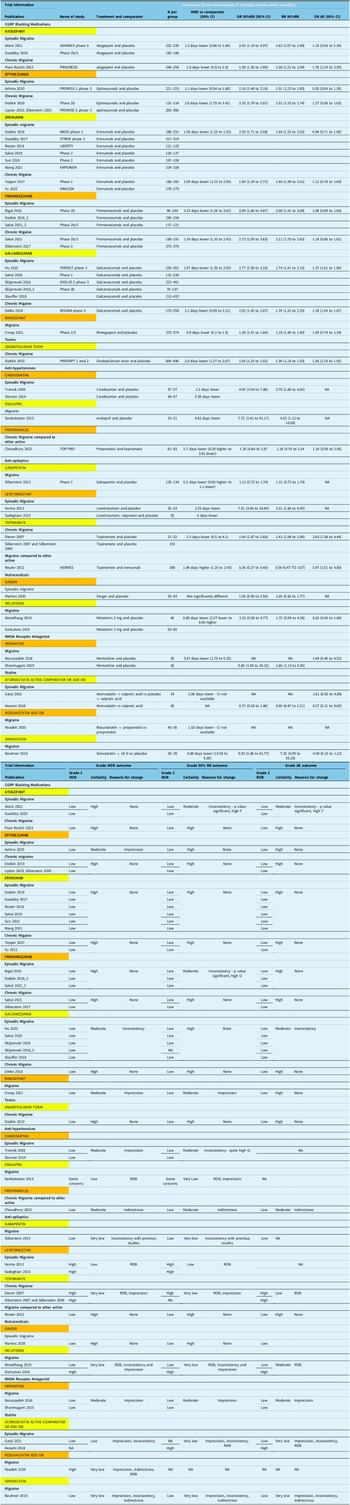
Table 3 presents all evidence incorporated into decision-making regarding the quality of evidence, from which panel came up with strength of recommendation.
Summary of evidence table
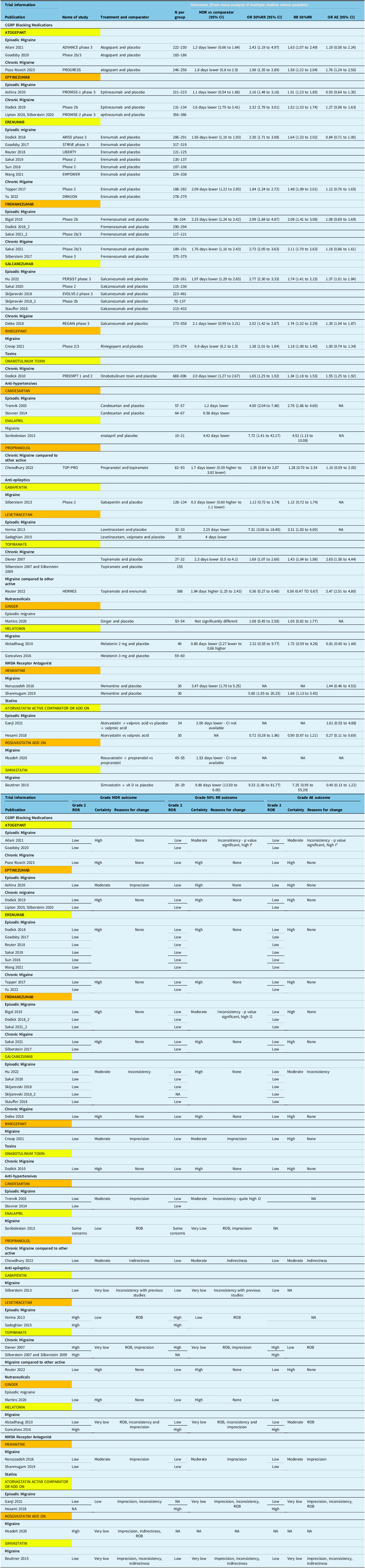
Note: N = number; MDR = migraine day reduction; OR 50%RR = odds ratio of 50% response rate; RR 50%RR = relative risk of 50% response rate; OR AE = odds ratio of adverse events; ROB = risk of bias.
More details on the systematic review and individual studies are available in Appendix 2. For all the studies, the efficacy outcomes and adverse effects are summarized in Table 2. A summary of the evidence synthesis is also provided, specifically how we arrive at the certainty of evidence and reasons for downgrade for each outcome in Table 3.
Studies were identified by the systematic review across seven different therapeutic categories as follows:
CGRP-blocking agents
Atogepant: We found two studies for episodic migraineReference Ailani, Lipton and Goadsby33,Reference Goadsby, Dodick and Ailani34 and one for chronic migraine,Reference Pozo-Rosich, Ailani and Ashina35 and one was found for treatment-resistant migraine patients.Reference Tassorelli, Nagy and Pozo-Rosich36 For episodic migraine, there was moderate certainty in evidence, and for chronic migraine, a high certainty in evidence.
Eptinezumab: We identified one study for episodic migraineReference Ashina, Saper and Cady37 and two for chronic migraine,Reference Dodick, Lipton and Silberstein38,Reference Lipton, Goadsby and Smith39 and one was found for treatment-resistant migraine patients.Reference Ashina, Lanteri-Minet and Pozo-Rosich40 For episodic migraine, there was moderate certainty in evidence and for chronic migraine, a high certainty in evidence.
Erenumab: For the treatment of episodic migraine, we identified five studies,Reference Dodick, Ashina and Brandes41–Reference Wang, Roxas and Saravia45 and for chronic migraine, two studies.Reference Tepper, Ashina and Reuter 46 ,Reference Yu, Kim and Wang 47 One study was found in treatment-resistant episodic migraine.Reference Reuter, Goadsby and Lanteri-Minet48 For episodic migraine, there was high certainty in evidence, and for chronic migraine, a high certainty in evidence.
Fremanezumab: For the treatment of episodic migraine, we identified three studies,Reference Bigal, Dodick and Rapoport49–Reference Sakai, Suzuki and Kim51 and for chronic migraine, two studies.Reference Sakai, Suzuki and Kim52,Reference Silberstein, Dodick and Bigal53 One study was found in treatment-resistant episodic migraine.Reference Ferrari, Diener and Ning54 For episodic migraine, there was moderate certainty in evidence, and for chronic migraine, a high certainty in evidence.
Galcanezumab: For the treatment of episodic migraine, we identified five studies,Reference Hu, Li and Li55–Reference Stauffer, Dodick, Zhang, Carter, Ailani and Conley59 and for chronic migraine, one study.Reference Detke, Goadsby, Wang, Friedman, Selzler and Aurora60 One study was found in treatment-resistant episodic migraine.Reference Mulleners, Kim and Lainez61 For episodic migraine, there was moderate certainty in evidence, and for chronic migraine, a high certainty in evidence.
Rimegepant: For the treatment of episodic migraine, we identified one study,Reference Croop, Lipton and Kudrow62 and although this study included some patients with chronic migraine, there was no subgroup analysis provided for the primary outcome in this group, and this was overall a small population. This data was of moderate certainty in evidence for episodic migraine patients.
Toxins
OnabotulinumtoxinA: For the treatment of chronic migraine, one study was found,Reference Dodick, Turkel and DeGryse63 and it provided high certainty in evidence. This chronic migraine study is a pooled study of Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) 1Reference Aurora, Dodick and Turkel64 and PREEMPT 2.Reference Diener, Dodick and Aurora65 We note that PREEMPT 1Reference Diener, Dodick and Aurora65 was negative on its primary outcome of change in headache episodes; however, this is not one of the outcomes we looked for in our review, nor is it a standard outcome in the field; this study was positive on all its secondary outcomes including migraine day reduction, which is one of the primary outcomes we looked for in our systematic review. There is an ongoing study in episodic migraine. The results are pending.66
Antihypertensives
Candesartan: One new study in episodic migraine was found,Reference Stovner, Linde and Gravdahl67 and one study from the previous guidelines was integrated in our analysis.Reference Tronvik, Stovner, Helde, Sand and Bovim68 This data provided a moderate certainty of evidence of this medication’s efficacy.
Enalapril: One new study was found for this treatment in episodic migraineReference Sonbolestan, Heshmat, Javanmard and Saadatnia69 and provided a very low certainty of evidence of this medication’s efficacy.
Propranolol: One new study was found for this treatment in chronic migraine;Reference Chowdhury, Bansal and Duggal70 this was a non-inferiority study with topiramate. This study provided a moderate certainty of evidence of this medication’s efficacy.
Antiepileptics
Gabapentin: One new study was found for this treatment in episodic migraine;Reference Silberstein, Goode-Sellers, Twomey, Saiers and Ascher71 this was a negative study. Previous studies reviewed in previous guideline were positive studies,Reference Mathew, Rapoport and Saper72,Reference Di Trapani, Mei, Marra, Mazza and Capuano73 but these were less well powered. This study provided a very low certainty of evidence of this medication’s lack of efficacy.
Levetiracetam: We found three studies in episodic migraine prevention: two used a placebo comparatorReference Verma, Srivastava, Kumar and Singh74,Reference Sadeghian and Motiei-Langroudi75 and one used valproic acid as a comparator.Reference Kashipazha, Ghadikolaei and Siavashi76 These studies provided low certainty of evidence of this medication’s efficacy.
Topiramate: There was a new study comparing the use of topiramate to erenumab in episodic migraineReference Reuter, Ehrlich and Gendolla77 and another new study comparing the use of topiramate to amitriptyline in episodic migraine.Reference Rodriguez-Leyva, Sanchez Aguilar and Hernandez-Sierra78 These studies showed that topiramate is less well tolerated and overall, less effective than erenumab, with a high certainty of evidence. In chronic migraine, there were three publications of two studies;Reference Diener, Bussone and Van Oene79–Reference Silberstein, Lipton and Dodick81 these studies overall provided low certainty evidence of medication’s efficacy.
Nutraceuticals
Ginger: There was a single study looking at this as a preventive treatment in episodic migraine.Reference Martins, Rodrigues and Monteze82 This study provided high certainty evidence that the treatment is not effective.
Melatonin: There were two studies looking at this preventive treatment in episodic migraine.Reference Alstadhaug, Odeh, Salvesen and Bekkelund83,Reference Goncalves, Martini Ferreira, Ribeiro, Zukerman, Cipolla-Neto and Peres84 The study with a low ROB but a lower dose of 2 mg nightly was negative,Reference Alstadhaug, Odeh, Salvesen and Bekkelund83 whereas the study with a high ROB but at a higher dose of 3 mg nightly was a positive study.Reference Goncalves, Martini Ferreira, Ribeiro, Zukerman, Cipolla-Neto and Peres84 These studies provided very low certainty of evidence of efficacy of melatonin and raised the possibility of a dose effect.
N-Methyl-D-aspartate (NMDA) receptor antagonist
Memantine: There were two studies looking at this medication for episodic migraine;Reference Noruzzadeh, Modabbernia and Aghamollaii85,Reference Shanmugam, Karunaikadal, Varadarajan and Krishnan86 these provided moderate certainty of evidence of efficacy of memantine.
Statins
We identified two studies looking at statin added to another preventiveReference Ganji, Majdinasab, Hesam, Rostami, Sayyah and Sahebnasagh87,Reference Mazdeh, Mahmudian, Vafaei, Taheri and Ghafouri-Fard88 and two studies looking at a statin versus placeboReference Buettner, Nir and Bertisch89 or another active comparator.Reference Hesami, Sistanizad, Asadollahzade, Johari, Beladi-Moghadam and Mazhabdar-Ghashghai90 Overall, these studies provided very low certainty of evidence of efficacy of statins alone or as an add-on to another preventive, and additionally, none of the therapies was used in more than a single trial.
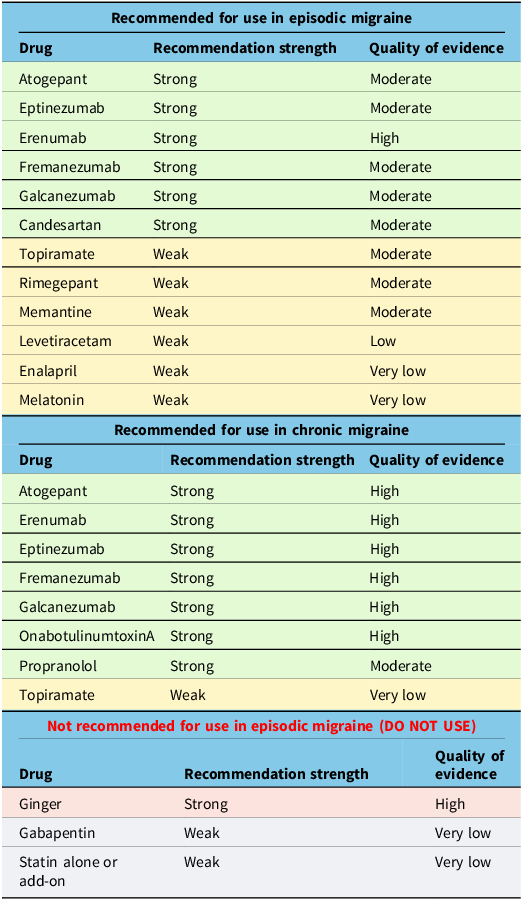
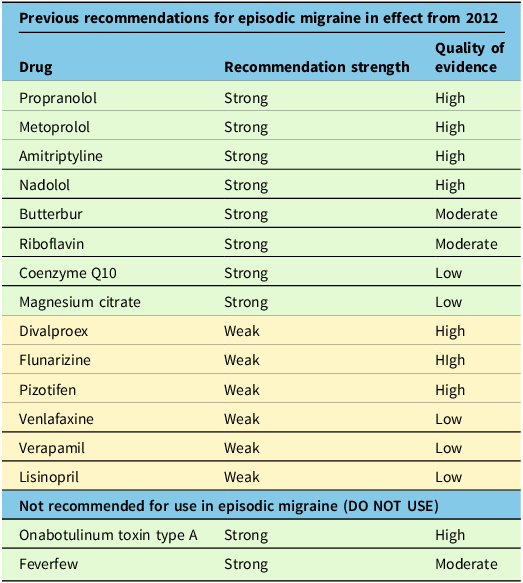
By undertaking a Delphi consensus process, the Recommendation Committee arrived at the treatment recommendations outlined below in Table 4. We also provide the previous recommendations from the 2012 guideline that have not been modified in Table 5.
New recommendations
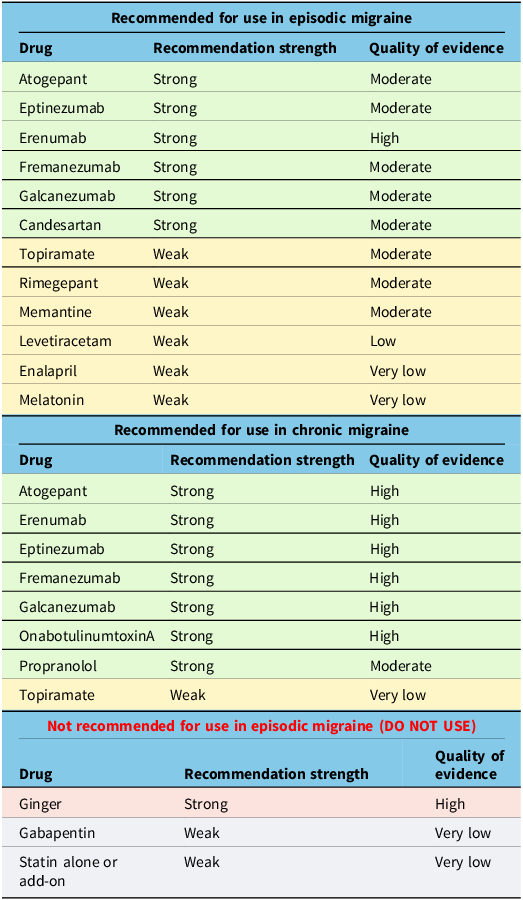
Previous recommendations still in effect
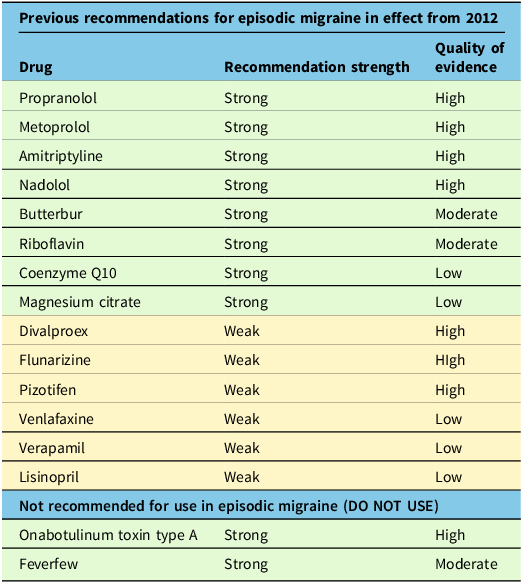
Notably, we have provided new recommendations for CGRP-blocking medications in episodic and chronic migraine that are currently in use, and most of these medications receive a strong recommendation in episodic and chronic migraine. Rimegepant is not currently approved for use in Canada as a preventive treatment but may be in the future. We have given it a weak recommendation, which could change pending future clinical studies. We upgraded candesartan to a strong recommendation for episodic migraine. We downgraded gabapentin to a weak recommendation against its use in episodic migraine. We downgraded topiramate to a weak recommendation for use in episodic migraine. Additionally, the weak recommendation for memantine, levetiracetam, enalapril and melatonin in episodic migraine is new. We additionally made recommendations for propranolol (strong) and topiramate (weak) use in chronic migraine.
Part 2: Treatment Strategies Based on Expert Consensus
Questions applicable to clinical practice were developed by the Steering Committee. These were presented to the Recommendation Committee as well as to two patient representatives. The questions were discussed, opinions exchanged and consensus obtained.
Questions for consideration in migraine prevention
1. Who should receive preventive treatment?Reference Pringsheim, Davenport and Mackie15
-
a. Patients with 4 or more moderate or severe headache days a month not responding to acute medication.
-
b. Patients with 8 or more headache days a month, even when acute medications are effective, as the risk of medication overuse headache (MOH) is increased in this group.
-
c. Patients who have migraine attacks with a significant impact on their life, despite using acute medications and trigger management/lifestyle modification strategies. The number of migraine days may be 3 or less in these situations if the impact is severe.
-
-
2. What should be considered when choosing a migraine preventive drug? Reference Pringsheim, Davenport and Mackie15
-
a. Efficacy: What is the confidence in the evidence and expected size of the effect of the drug in reducing migraine frequency?
-
b. Drug side effect profile: How safe and well tolerated is the drug?
-
c. Comorbid disorders (depression, anxiety, insomnia, obesity, hypertension, history of renal calculi, constipation, vascular disorders).
-
d. Patient disability and migraine severity.
-
e. Pregnancy planning and appropriate contraception.
-
f. Patient preference.
-
-
3. What constitutes an adequate preventive trial?
-
a. Drugs should be continued at a target dose for at least 2 months for an adequate trial unless side effects make drug discontinuation necessary.Reference Pringsheim, Davenport and Mackie15
-
b. In the case of anti-CGRP mAbs, the majority of patients can be evaluated for response after 3 months. However, especially in patients with a history of treatment-resistant chronic migraine, the improvement might be subtle over the first 3 months but become more apparent and clinically significant over 6 months.Reference Schim, Anderson and Brunner91,Reference Kuruppu, North, Kovacik, Dong, Pearlman and Hutchinson92
-
c. In the case of onabotulinumtoxinA, patients should have a minimum of two quarterly injections, and three quarterly injections could be reasonable.Reference Silberstein, Dodick and Aurora93
-
-
4. When should preventive therapy be considered effective?Reference Pringsheim, Davenport and Mackie15
-
a. Headache diaries are important in evaluating treatment effectiveness, and we recommend patients use one of the mobile applications available or a paper diary.Reference Baos, Ester and Castellanos94,Reference Nappi, Jensen, Nappi, Sances, Torelli and Olesen95
-
b. Headache frequency or intensity is reduced by 50% or more, although less reductions of headache frequency may be worthwhile, particularly if the drug is well tolerated.
-
c. Reduction in headache intensity and migraine-related disability also need to be considered.Reference Pringsheim, Davenport and Mackie15,Reference Society96
For example:
(i) Migraine Disability Assessment (MIDAS) Score
-
Reduction of ≥5 points for a baseline score between 11 and 20
-
Reduction of >30% for a baseline score >20
-
-
(ii) Headache Impact Test with 6 items (HIT-6) Score
-
Reduction of ≥5 points
-
-
-
5. How long should successful preventive therapy be continued? Reference Pringsheim, Davenport and Mackie15
This opinion is from expert consensus, as there are no randomized studies providing clear guidance on the optimal duration of migraine preventive treatments. This broadly generalized approach may not be appropriate, and this may have to be assessed on a case-by-case basis. Longer duration of treatment may be particularly important with patients with a long history of chronic migraine.Reference Sacco, Amin and Ashina97
While using conventional oral preventive drugs, it is reasonable to consider tapering off medications at 12 months, especially if patients are doing well with a meaningful response to therapy and have reverted to an infrequent episodic pattern of headache (ideally 4–6 headache days) with good control with acute therapies, no risk of medications overuse headache and minimal disability. If headaches or migraine symptoms recur as the dose is decreased or as the drug is discontinued, the dose should be increased again, or the drug be restarted.
For those on newer agents such as onabotulinumtoxinA or anti-CGRP mAbs, discontinuation can also be considered based on patient preference. We recommend this only if there is an episodic pattern of migraine with very minimal disability for a period of at least 12 months. For onabotulinumtoxinA, a proposed method of attempting this is to increase the interval between injections and see if there is no worsening, the treatment can be stopped.Reference Tassorelli, Sances and Avenali98 There are no long-term safety concerns with onabotulinumtoxinA to warrant discontinuation in patients who want to continue.Reference Blumenfeld, Stark, Freeman, Orejudos and Manack Adams99 For anti-CGRP mAbs, this can also be attempted in a similar way with increasing the interval and restarting therapy if there is a worsening of attacks. There is some evidence that stopping anti-CGRP therapies leads to increased attacks, so patients should be warned accordingly.Reference Blumenfeld, Stark, Freeman, Orejudos and Manack Adams99,Reference Al-Hassany, Lyons and Boucherie100 There are presently no known safety concerns with use that require stopping these medications after a specific treatment interval,Reference Al-Hassany, Lyons and Boucherie100 but patients should be monitored for the possibility of new onset hypertension or worsening of existing hypertension.Reference Wang, Fenton and Dao101–Reference Guerzoni, Castro, Brovia, Baraldi and Pani104
What advice should be given to the patient with medication overuse when prevention is being considered?Reference Pringsheim, Davenport and Mackie15
-
a. When preventive therapy is started, patients should be evaluated for the presence of medication overuse and instructed accordingly regarding the appropriate amount of medication to use monthly, and the frequency of acute medication use should also be followed.
-
b. Evidence suggests that in many cases, a withdrawal may not be necessary and starting preventive therapy alone may be adequate. Still, a withdrawal may be necessary for some patients.Reference Hird and Sandoe105–Reference Carlsen, Munksgaard and Nielsen107
-
c. As opioid and barbiturate-induced MOH is more likely to occur,Reference Bigal and Lipton108 and in clinical experience may be less likely to respond to prevention, we recommend taper in these situations.Reference Diener, Antonaci and Braschinsky109
Chronic migraine and overlap with high-frequency episodic migraine (HFEM)
The previous CHS Guideline of 2012 did not address chronic migraine. In the current definition of ICHD3, chronic migraine is currently defined as 15 or more headache days per month, with at least 8 days having migrainous features or response to migraine-specific medications.17 This definition is somewhat arbitrary,Reference Medrea and Christi110 and in fact chronic migraine and HFEM have a lot of similarities as we discuss below.
Recent papers have highlighted that the disability burden experienced by migraine patients is driven by the number of migraine days per month and patients with episodic migraine who have 8 or more migraine days per month experience a high degree of disability, similar to chronic migraine patients who have 8 or more migraine days per month.Reference Chalmer, Hansen, Lebedeva, Dodick, Lipton and Olesen111,Reference Ishii, Schwedt and Dumkrieger112 The degree of disability does tend to increase with the number of migraine days overall, and severe disability can start even at HFEM.Reference Ishii, Schwedt and Dumkrieger112,Reference Torres-Ferrús, Quintana, Fernandez-Morales, Alvarez-Sabin and Pozo-Rosich113 This has led to the suggestion that the requirement for 15 or more headache days be dropped from the future ICHD definition of chronic migraine.Reference Chalmer, Hansen, Lebedeva, Dodick, Lipton and Olesen111,Reference Ishii, Schwedt and Dumkrieger112
Special considerations for anti-CGRP therapies
The CGRP-blocking medications are widely used in migraine. However, their place in first-line migraine prevention may not be cost-effective in all instances.Reference Burch19,114–118 As guideline developers in constrained resource settings, we must consider this aspect.Reference Haycox, Bagust and Walley119,Reference Hill, Olson and Falck-Ytter120 Additionally, a network meta-analysis performed by the Institute for Clinical and Economic Review found these medications not to be superior in efficacy to older medications for episodic migraine prevention however using older trials that may not be comparable.Reference Ellis, Walton and Otuonye 121 For chronic migraine, only comparison possible was with topiramate. Now we have a head-to-head trial of topiramate versus erenumab, this showed the superiority of erenumab both in terms of efficacy and tolerability.Reference Reuter, Ehrlich and Gendolla77 These medications are also likely better tolerated in clinical practice when compared to oral preventives.Reference Hepp, Dodick, Varon, Gillard, Hansen and Devine 122 – Reference Schwedt, Lee and Knievel 124
In a Canadian context, Canadian Agency for Drugs and Technologies in Health (CADTH) found the use of these newer treatments first line to not be cost-effective when looking only at direct costs.114–118 For chronic migraine and likely HFEM, cost-effectiveness on direct costs is likely not the only consideration. The quite high indirect costs are important,Reference Amoozegar, Khan, Oviedo-Ovando, Sauriol and Rochdi125 along with the high burden of disability of these patientsReference Blumenfeld, Varon and Wilcox8,Reference Ishii, Schwedt and Dumkrieger112,Reference Buse, Manack and Fanning126 and the evidence of poor tolerability of older medications in the context of these patients likely needing long-term use.Reference Hepp, Dodick, Varon, Gillard, Hansen and Devine122,Reference Hepp, Dodick and Varon123 The only study in patients where no prior preventive failure was required (the population we are proposing these medications be used in) looking at indirect costs shows that these medications are cost-effective in chronic migraine patientsReference Sussman, Benner, Neumann and Menzin127 at a threshold likely acceptable in a Canadian setting.Reference Griffiths and Vadlamudi128 In this particular study, their use is not cost-effective in episodic migraine patients, but there was no separate HFEM group.Reference Sussman, Benner, Neumann and Menzin127
In patients with moderate-frequency episodic migraine (MFEM) (4 or more migraine days up to 7), we would consider it reasonable to allow the use of these medications after failure or intolerance of two preventive therapies unless data shows this to be cost-effective as first-line use.
In a Canadian context, the indirect and direct costs of chronic migraine and HFEM are similar and significantly higher than low-frequency episodic migraine.Reference Amoozegar, Khan, Oviedo-Ovando, Sauriol and Rochdi125 We believe it is reasonable to recommend that all patients with HFEM (8 or more migraine days, but less than 15 headache days) with moderate disability and all patients with chronic migraine (8 or more migraine days and 15 or more headache days) get access for first-line use of these medications with other medications, given the high indirect costs incurred by these patients, and because likely the use of these medications leads to savings overall on indirect costs.Reference Lazaro-Hernandez, Caronna and Rosell-Mirmi129 Should the ICHD incorporate HFEM into the definition of chronic migraine in the future, then we would recommend that the requirement to demonstrate moderate disability be removed.
Indication for treatment with CGRP targeting agents (atogepant, eptinezumab, galcanezumab, erenumab, fremanezumab)
-
A. MFEM (4–7 MMD)
-
(i) Intolerance/contraindication or inadequate response to an 8-week trial of at least two non-CGRP targeting oral preventive therapies.
-
-
B. HFEM (8–14 MMD)
-
(i) At least moderate disability as shown by one of:
-
a. MIDAS score ≥11
-
b. HIT-6 score >50
-
c. Clinical impression
-
-
(ii) If condition (i) met, no requirement for a trial of non-CGRP oral preventive therapies
-
-
C. Chronic migraine
No requirement for MIDAS, HIT-6 or trial of non-CGRP targeting oral preventive therapies.
Treatment options include:
-
(i) Atogepant, eptinezumab, galcanezumab, erenumab, fremanezumab
-
(ii) OnabotulinumtoxinA
-
(iii) Propranolol
-
(iv) Topiramate
-
The choice among the different treatment options for episodic and chronic migraine would depend on the healthcare practitioner’s assessment of the clinical situation as well as patient preference.
We would like to acknowledge that the possibility of unknown and perhaps serious side effects is present with many new medications up to 10 years after their introduction in up to a third of drugs, and we should remain vigilant with these newer therapies and may revise our recommendations.Reference Downing, Shah and Aminawung130,Reference Sun, Li and Xia131
Further considerations in clinical use:
a. Before starting these therapies, we recommend individualized clinical assessment of vascular disease and risk factors. Generally, anti-CGRP therapies have had a good cardiovascular and cerebrovascular safety profile in patients with no active cardiovascular or cerebrovascular disease,Reference Mathew and Klein132,Reference Wang, Liu and Sun133 but caution should be exercised especially in patients with recent cerebrovascular or cardiovascular events as this population was excluded from clinical studies.
b. There have been reports of worsening and new onset Raynaud’s phenomenaReference Sun, Li and Xia131,Reference Breen, Brumfiel and Patel134–Reference Manickam, Buture, Tomkins and Ruttledge136 and alopecia,Reference Sun, Li and Xia131 and individualized decision-making should be considered.
c. In patients with severe constipation, erenumab and atogepant should be used with caution.Reference Ailani, Lipton and Goadsby33–Reference Pozo-Rosich, Ailani and Ashina35,Reference Wang, Roxas and Saravia45,Reference Tepper, Ashina and Reuter46
d. There are reports of worsening of hypertension or de novo hypertension in some patients on erenumab and possibly other anti-CGRP therapies.Reference de Vries Lentsch, van der Arend, VanDenBrink and Terwindt103
e. Switching in cases of treatment failure should also be considered, some observational studies indicate that after the failure of one anti-CGRP therapy, it is possible that an individual may respond to another anti-CGRP therapy,Reference Overeem, Peikert and Hofacker137–Reference Ziegeler and May139 and there is also the option of class switching from a receptor antibody to a ligand antibody and vice versa.Reference Lambru, Caponnetto and Hill140,Reference Straube, Broessner and Gaul141
f. We do recommend that switching between anti-CGRP therapies in cases of side effects or patient preference:
-
– switching from erenumab or atogepant to a CGRP ligand blocker in cases of constipation
-
– switching to eptinezumab in cases of injection site reactions
-
– switching from erenumab to fremanezumab in cases of hypertensionReference de Vries Lentsch, van der Arend, VanDenBrink and Terwindt103
-
– switching to an antibody with quarterly dosing for patients preferring this option instead of monthly dosing.
-
Clinical strategies for migraine preventionReference Pringsheim, Davenport and Mackie15
-
1. First-time strategy
-
(a) Beta-blocker strategy: Propranolol, nadolol, metoprolol
-
(b) Candesartan: With caution in patients of childbearing potential regarding safety issues in pregnancy
-
(c) CGRP-blocking strategy: Erenumab *, galcanezumab*, fremanezumab *, eptinezumab *, atogepant * in HFEM (with moderate disability using MIDAS, HIT-6 or clinical impression) and chronic migraine. For the anti-CGRP mAbs, caution should be exercised in patients of childbearing age.
-
(d) Toxin strategy: OnabotulinumtoxinA should be considered first line in chronic migraine (≥8 migraine days per month and ≥15 headache days).
-
(e) Tricyclic strategy: Amitriptyline
-
-
2. Low side effect strategy
-
(a) Candesartan
-
(b) Herbal/vitamin/mineral: Magnesium citrate, riboflavin, coenzyme Q10, melatonin
-
(c) CGRP-blocking strategy: Erenumab *, galcanezumab*, fremanezumab *, eptinezumab *, atogepant *
-
(d) Toxin strategy: OnabotulinumtoxinA in chronic migraine.
-
-
3. Increased body mass index strategy
Topiramate
Atogepant *
-
4. Hypertension strategy
Propranolol, candesartan, nadolol, metoprolol
-
5. Depression/anxiety strategy
Amitriptyline, venlafaxine
-
6. Medications that can be considered in certain patients – weak recommendation
These treatments are also recommended for use as monotherapy, in addition to the strategies outlined above.
Levetiracetam* memantine* and rimegepant*, **
Topiramate, valproic acid, pizotifen, flunarizine and verapamil
*New treatments added since 2012 CHS Guideline
** Not approved for use in Canada
-
7. Refractory patient strategyReference Pringsheim, Davenport and Mackie15
Refractory migraine is defined as a condition in which symptoms cause significant interference with the ability to function or quality of life despite the use of acute and preventive treatment.Reference Pringsheim, Davenport and Mackie15,Reference Schulman, Lake and Goadsby142–Reference Martelletti, Katsarava and Lampl144 Treatment-resistant migraine is defined as a patient with a failure of properly dosed trials of medications from at least two classes of prophylactic medications.Reference Schulman, Lake and Goadsby142–Reference Martelletti, Katsarava and Lampl144 In refractory patients, there is ample evidence that anti-CGRP mAbsReference Ashina, Lanteri-Minet and Pozo-Rosich40,Reference Reuter, Goadsby and Lanteri-Minet48,Reference Ferrari, Diener and Ning54,Reference Mulleners, Kim and Lainez61 and atogepantReference Tassorelli, Nagy and Pozo-Rosich36 can be effective even after other treatments fail. In all episodic migraine patients having failed other preventive therapies, anti-CGRP mAbs and atogepant should be offered.Reference Ashina, Lanteri-Minet and Pozo-Rosich40,Reference Reuter, Goadsby and Lanteri-Minet48,Reference Ferrari, Diener and Ning54,Reference Mulleners, Kim and Lainez61
Layering of treatment can also be considered in refractory patients. There is a rationale behind the layering of drugs; it is likely that different prophylactic drugs work by different mechanisms, and therefore, the effects of two drugs may be synergistic in reducing migraine frequency. Here are some strategies to consider and rationale:
i. There are observational studies showing increased benefit from using onabotulinumtoxinA and anti-CGRP mAbs or gepants in combination.Reference Cohen, Armand, Lipton and Vollbracht145–Reference Mechtler, Saikali, McVige, Hughes, Traut and Adams147 This is reasoned to be due to the combined blockade of Adelta and C fibers CGRP signaling, likely adding synergistic benefit not seen with either therapy alone.Reference Pellesi, Do, Ashina, Ashina and Burstein148 Based on expert consensus, we recommend considering layering of anti-CGRP therapies with onabotulinumtoxinA in refractory patients.
ii. There are observational studies on layering of older therapies with anti-CGRP therapies (erenumab), and although these studies are not randomized, there have been encouraging results with improvement in migraine days and acute medication use.Reference Burch19,Reference McGowan, Sampson, Salzwedel, Cogo, Foerster and Lefebvre20 This strategy is recommended in other recent guidelines.Reference Moher, Liberati, Tetzlaff and Altman21 Based on expert consensus, we recommend considering layering of older medications with anti-CGRP therapies in refractory patients, especially in cases where onabotulinumtoxinA can’t be used.
iii. There are observational studies showing improvement looking at combinations of older therapies beta-blockers or flunarizine with topiramateReference Vos, Abajobir and Abate13,Reference Goldberg14 and also valproate and beta-blockers.Reference Pringsheim, Davenport and Mackie15 There was a negative randomized study looking at combining amitriptyline and topiramate; however, patient impression in this study was in favor of the combination.16 Combination therapy using older therapies for refractory patients has been commonly recommended by other expert groups as well.17,18 In cases where newer anti-CGRP or toxin strategies can’t be used, we recommend considering layering of older therapies, being cognizant of possible side effects and interactions.
iv. There is also evidence for the use of other strategies such as behavioral interventions and neuro-modulation, but we have not reviewed these strategies for the current guideline.Reference Higgins and Green22
For further guidance, a review should be consulted,Reference Higgins and Green22 and when possible, these patients should be considered for referral to a headache specialist for management.
These strategies are proposals. If a patient fits better in one strategy versus another, then the best medication should be used.
-
8. Migraine during pregnancy and lactation strategyReference Pringsheim, Davenport and Mackie 15
-
(a) Migraine drug prophylaxis is best avoided during pregnancy and lactation, if possible. Strategies involving trigger avoidance and lifestyle factors should be considered.
-
(b) If migraine drug prophylaxis is necessary during pregnancy or lactation, the best choice is a beta-blocker (propranolol or metoprolol), and if these are contraindicated or ineffective, amitriptyline can be considered.149,Reference Burch150
-
(c) There is some evidence on the safety of onabotulinumtoxinA in patients exposed to it during pregnancyReference Brin, Kirby and Slavotinek151–Reference Smirnoff153 and also lactation.154 In patients with disabling treatment-resistant chronic migraine, this may be considered, but we caution that this data includes a small number of patients and can’t ascertain rare AEs. We recommend clinicians consider the use of onabotulinumtoxinA during pregnancy on a case-by-case basis.
-
(d) There is some limited post-marketing data on the safety of anti-CGRP mAbs and gepants in pregnancy,Reference Noseda, Bedussi, Gobbi, Ceschi and Zecca155 but this data includes very small numbers of patients and can’t ascertain AEs. As CGRP crosses the placentaReference Bussiere, Davies and Dean156 and is involved in uteroplacental circulation,Reference Burch19 patients should not actively try to become pregnant until the treatment has been stopped for 6 months for anti-CGRP mAbs. For gepants, it should be sufficient to discontinue for a week before attempting to get pregnant based on the half-life of these agents. Patients should be advised accordingly.
-
(e) Anti-CGRP mAbs are large molecules and would likely be destroyed in the gastrointestinal tract. They are not likely to be absorbed and transferred into breast milk. They may be safe, but there is a paucity of data available,157–160 and their use in lactation is currently not recommended. For available gepants, there is no data available on transfer to breast milk and infants, and their use in lactation is currently not recommended. Rimegepant, which is not approved for prevention in Canada, shows very low secretion in breast milk.161,Reference Baker, Croop and Kamen162
-
-
9. Drugs not recommended
-
(a) OnabotulinumtoxinA is not recommended for prophylaxis of episodic migraine, but there is an ongoing study, and as such, this recommendation may need to be reconsidered when more is known about the result of this study.66
-
(b) Gabapentin is not recommended for prophylaxis of episodic migraine.
-
(c) Statins alone or add-on are not recommended for prophylaxis of episodic migraine.
-
(d) Ginger is not recommended for prophylaxis of episodic migraine.
-
Discussion
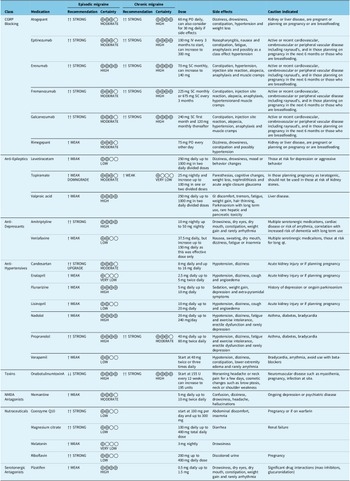
We provide updated guidelines on the treatments to be utilized in the prevention of migraine in Canada. We summarize the recommendations and the use of these medications in Table 6 above.
Recommendations for migraine prevention summary
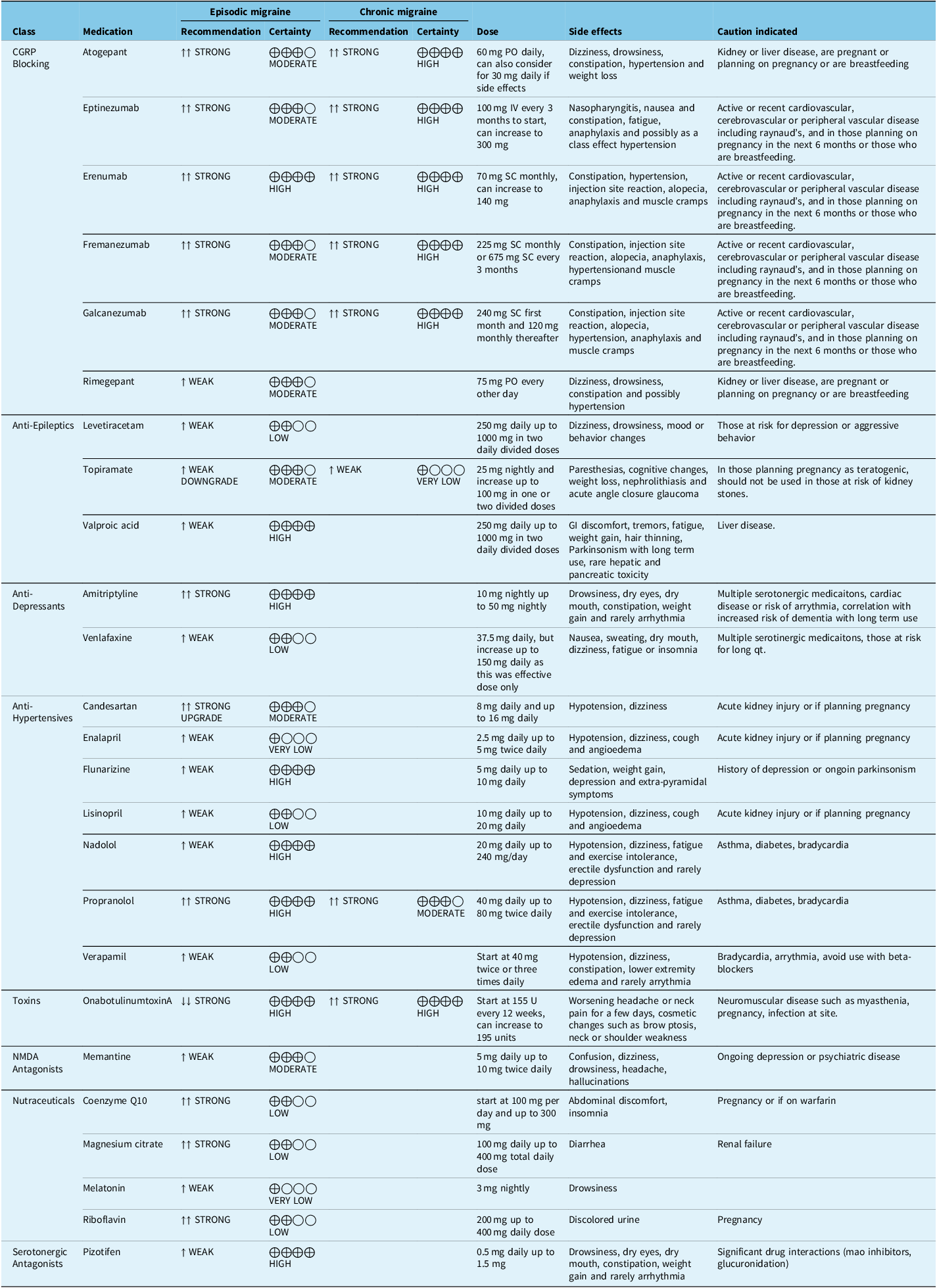
Note: In dark font are the new updated guidelines, and in light font are previous guideline recommendations, which were not updated.
PO = oral; SC = subcutaneous; IV = intravenous.
Specific strengths of our study are a well-conducted search and using GRADE methodology with two reviewers throughout all stages of the process. We opted not to update the previous recommendations and certainty of evidence for medications where there was no new evidence, so as to not duplicate the work already completed by our colleagues. We felt that it was unlikely that we were going to change the recommendations for those therapies. It would be informative and useful for future guidelines to have direct comparative studies looking at older medications such as amitriptyline or propranolol, where we have strong recommendations for their use, and seeing how they fare in non-inferiority studies with newer anti-CGRP therapies. In cases such as topiramate, gabapentin and candesartan, where there was substantial new evidence, we did undertake a revision of the previous studies and upgraded or downgraded previous recommendations.
Conclusions
In summary, we provided a systematic review of all studies in migraine prevention since the previous CHS Guideline in 2012 and significantly for all studies in chronic migraine prevention. Based on the evidence synthesis, we provide updated recommendations for the prevention of episodic and chronic migraine utilizing treatments available in Canada. The anti-CGRP agents provide new treatment options for episodic and chronic migraine. We have strong evidence for their use in all patients and in many cases first line, and we caution against denying them in any treatment-resistant patients. There is evidence for the use of propranolol, topiramate and onabotulinumtoxinA in addition to anti-CGRP agents as treatments for chronic migraine. Given the high burden of disability experienced by these patients as well as the efficacy and favorable side effect profile of the newer treatments, we have recommended that onabotulunumtoxinA and the anti-CGRP agents be considered first line among other treatments for chronic migraine. In the event of a change in the ICHD definition of chronic migraine to capture HFEM, we would make the same recommendation for HFEM. Topiramate has a weak recommendation for use, and gabapentin has a weak recommendation against its use in episodic migraine, so both have been downgraded. There is new evidence on the use of memantine, levetiracetam and enalapril in episodic migraine and in certain situations for the use of melatonin in episodic migraine.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cjn.2024.285
Author contributions
Dr I. Medrea – protocol conception and design, acquisition and analysis of data, preparation of first draft and revision of the manuscript, guarantor of review, co-chair the Steering Committee; Dr S.N. Christie – protocol conception and design, acquisition and analysis of data, revision of the manuscript, study supervision, guarantor of review, chair of the Steering Committee; Dr P. Cooper – review of the manuscript, member of the Steering Committee, member of the Recommendation Committee; Dr M. Lagman – protocol conception and design, review of the manuscript, member of the Steering Committee, member of the Recommendation Committee; Dr C.H. Sandoe – review of the manuscript, member of the Steering Committee, member of the Recommendation Committee; Dr F. Amoozegar – review of the manuscript, member of the Recommendation Committee; Dr W.M. Hussain – review of the manuscript, member of the Recommendation Committee; Dr A. C. Bradi – review of the manuscript, member of the Recommendation Committee; Dr J. Dawe – review of the manuscript, member of the Recommendation Committee; Dr M. Guay – review of the manuscript, member of the Recommendation Committee; Dr F. Perreault – review of the manuscript, member of the Recommendation Committee; Dr S. Reid – review of the manuscript, member of the Recommendation Committee; Dr C. Todd – review of the manuscript, member of the Recommendation Committee. The Steering Committee was formed prior to undertaking the guideline to help develop the process undertaken, medications considered and questions considered in our review. The Recommendation Committee comprised all members of the Steering Committee in addition to the members documented above. The Recommendation Committee provided the evidence-based recommendations in Part 1. In addition to two patient representatives, the Recommendation Committee also participated in Part 2, providing treatment strategies based on expert consensus. The Recommendation Committee also reviewed the manuscript and provided input.
Funding statement
The CHS endorsed the creation of an updated migraine preventive therapy clinical practice guideline and provided funds for this review. However, there was no involvement in study design, data analysis or interpretation and drafting/revision of this protocol. Some members of the board of the CHS (SC, PC, FA, ML, CS, WH) as well as non-board members (IM, AB, CT, SR, MG, FP, JD) have been involved in the protocol, drafting and revision of this manuscript. Their work was not compensated by the CHS. The only funds used in this review were to pay an independent contractor with systematic review experience (BS) to provide the literature search and to load the full-length papers onto Covidence that were requested after the abstract review.
Competing interests
To minimize conflicts of interest, we have undertaken a process of declaring conflicts using standard declaration sheetsReference Gronseth, Cox, Gloss and Merilliat26 and bringing these to the CHS board and the guideline panel, to obtain consensus on how to best collectively eliminate those with unmanageable conflictsReference Gronseth, Cox, Gloss and Merilliat26 and to manage these conflicts in those not deemed unmanageable.Reference Gronseth, Cox, Gloss and Merilliat26,163 Detailed auditable documents are held with the guideline panel on rules followed for conflict declarations,Reference Gronseth, Cox, Gloss and Merilliat26 ongoing conflicts in panel members and management strategies.
Disclosures
For categories not listed, the authors have nothing to disclose:
-
1. Dr Ioana Medrea
No disclosures
-
2. Dr Paul Cooper
Advisor, consultant, advisory board and/or scientific board: Teva, Lundbeck
Honoraria lecture fees, Continuing Medical Education (CME): Lilly
-
3. Dr Ana Marissa Lagman-Bartolome
Advisor, consultant, advisory board and/or scientific board:
I participated in advisory boards for TEVA, Lundbeck, Pfizer and Miravo/Search Light from 2021 to 2023. Fees from these events were submitted to Center for Headache at Women’s College Hospital until January 2023. Since January 2023, I attended one advisory board meeting for each of the following companies: TEVA, Miravo/Search Light and Lundbeck. The Center for Headache at Women’s College Hospital, where I used to work, received unrestricted educational grant from TEVA and Lundbeck.
Research Grant: I am currently the principal investigator for RCT in using erenumab for preventive migraine treatment in children and adolescents with episodic and chronic migraine at the Hospital for Sick Children sponsored by AMGEN. All fees for this trial were submitted to Research Institute of the Hospital for Sick Children. I will participate in pediatric migraine trials sponsored by Lundbeck, Abbvie and Pfizer, but none of these trials has started yet, and I did not receive any consulting fees or honoraria for these trials. Once the trials are launched, all the fees for these trials will be submitted to the Research Fund of the Division of Pediatric Neurology at the Children’s Hospital, London Health Sciences Center. These fees will not affect my salary from London Health Sciences Center, Department of Pediatrics.
Honoraria lecture fees, Continuing Medical Education (CME):
I received royalties as an author from the Canadian Pharmacists Association.
Honoraria lecture fees/speaker bureau:
I received honoraria fees for giving one teaching session for neurologists, nurse practioners and family doctors on the highlights of the 2023 American Headache Society (AHS) Scientific Meeting for TEVA and one teaching session for the family doctors and neurologists as well headache neurologist on updates in migraine trials and management presented in the 2023 AHS Scientific Meeting for Abbvie.
-
4. Dr Claire Sandoe
Advisor, consultant, advisory board and/or scientific board: Abbvie, Eli Lilly, Lundbeck, Miravo, Novartis, Pfizer, Teva
Honoraria lecture fees, CME: Eli Lilly, Master Clinician Alliance, Novartis
-
5. Dr Farnaz Amoozegar
Advisor, consultant, advisory board and/or scientific board: Abbvie, Pfizer
Honoraria lecture fees, CME): Eli Lilly, Allergan, Teva, Pfizer, Lundbeck, Miravo
-
6. Dr Wasif Hussain
Advisor, consultant, advisory board and/or scientific board: Abbvie/Allergan, Miravo, Eli Lilly, Lundbeck, Teva, Eisai.
Honoraria lecture fees, CME: Abbvie/Allergan
Honoraria lecture fees/speaker bureau: Abbvie/Allergan, Miravo, Eli Lilly, Lundbeck, Teva
-
7. Dr Ana Bradi
Honoraria lecture fees/speaker bureau: Novartis, unrelated to migraine medications
-
8. Dr Jessica Dawe
Advisor, consultant, advisory board and/or scientific board: Abbvie
Stock, stock options, patents, royalties, manuscript fees, subsidies, endowments: None
-
9. Dr Meagan Guay
Advisor, consultant, advisory board and/or scientific board: Abbvie, Searchlight
Honoraria lecture fees, CME: None
-
10. Dr Francois Perreault
Advisor, consultant, advisory board and/or scientific board: Advisory committee for Pfizer for Rimegepant
Honoraria lecture fees, CME): Moderator for a headache forum/case presentation organized by Abbvie
Stock, stock options, patents, royalties, manuscript fees, subsidies, endowments: None
-
11. Dr Candice Todd
Advisor, consultant, advisory board and/or scientific board: Pfizer, Abbvie and searchlight pharma
Honoraria lecture fees, CME: None
Honoraria lecture fees/speaker bureau: Lundbeck, Searchlight pharmaceuticals and Abbvie
-
12. Dr Stuart Reid
Advisor, consultant, advisory board and/or scientific board: Teva
Honoraria lecture fees, CME: Teva, Abbvie, Lilly, Aralez
Honoraria lecture fees/speaker bureau: Teva, Abbvie, Lilly, Aralez
-
13. Becky Skidmore
No disclosures
-
14. Dr Suzanne Christie:
Advisor, consultant, advisory board and/or scientific board: Teva Neuroscience, Abbvie, Lundbeck, Pfizer