Impact Statement
More research groups are using cryo-soft-X-ray tomography as a correlative imaging tool to study the ultrastructure of cells and tissues but very few tomograms are segmented with existing segmentation programs. Segmentation is usually a prerequisite for measuring the geometry of features in tomograms but the time- and labor-intensive nature of current segmentation techniques means that such measurements are rarely across a large number of tomograms, as is required for robust statistical analysis. Contour has been designed to facilitate the automation of segmentation and, as a result, reduce manual effort and increase the number of tomograms that can be segmented. Because it requires minimal manual intervention, Contour is not as prone to human error as programs that require the users to trace the edges of cellular features. Geometry measurements of the segmented volumes can be calculated using this program, providing a new platform to quantitate cryoSXT data. Contour also supports quantitation of volumes imported from other segmentation programs. The generation of a large sample of segmented volumes with Contour that can be used as a representative training dataset for machine learning applications is a long-term aspiration of this technique.
1. Introduction
The biology of cellular compartments has been extensively studied using high-resolution microscopy techniques. Transmission electron microscopy of thin sections of cells stained with heavy metals has been used for decades to produce images of intracellular ultrastructure and can resolve structures at the nanometer level(Reference Kourkoutis, Plitzko and Baumeister 1 ). For precise quantitation, cellular compartments of interest need to be delineated from the other ultrastructural features by segmentation. These features can be segmented manually by tracing the edges of features with Segmentation Editor in Fiji(Reference Schindelin, Arganda-Carreras and Frise 2 ) or with tools such as Amira (Thermo Scientific) that have “intelligent scissors” that predict the boundaries of the object being traced by the user( 3 ). However, these manual processes are time-consuming and the boundaries of the segmented volumes are prone to human interpretation(Reference Luengo, Darrow and Spink 4 ). Automatic tools exist, but these also have limitations. For example, Bayesian matting, wherein a Bayesian framework is used to delineate foreground objects from the background based on pixel range, is less likely to successfully segment features with textured or thin edges(Reference Chuang, Curless, Salesin and Szeliski 5 ). Similarly, “magic wand” segmentation, in which pixels of a given range of intensities are segmented if they are all connected, is less applicable to features with a broad range of intensities and where there is high noise in the background(Reference Talbot and Beare 6 , Reference Rother, Kolmogorov and Blake 7 ). Watershed segmentation is often used to separate objects by estimating the boundaries between them based on the distances between their highest intensity maxima. However, the specificity of this technique is low in noisy datasets and can lead to over-segmentation, whereby many small segments are created within a single feature(Reference Shafarenko, Petrou and Kittler 8 , Reference Romero-Zaliz, Reinoso-Gordo and Cruz Corona 9 ). As a result, segmentation tools that use machine learning and deep neural networks to distinguish features of interest from the rest of the ultrastructure have been developed for electron microscopy (e.g., Unet, Ilastik)(Reference Ronneberger, Fischer, Brox, Navab, Hornegger, Wells and Frangi 10 –Reference Liu and Ji 15 ). However, these tools require either a large representative training dataset or modified training for each micrograph.
For over a decade, the ultrastructural imaging technique known as cryo-soft-X-ray tomography (cryoSXT) has been used by cell biologists and pathologists to image the cellular compartments of unfixed whole cells in 3D(Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 , Reference Kounatidis, Stanifer and Phillips 17 ). Moreover, cryoSXT is being used as a correlative imaging technique with cryo-structured illumination microscopy (cryoSIM) to identify features in cellular ultrastructure(Reference Okolo, Kounatidis and Groen 18 , Reference Vyas, Perry and Okolo 19 ). X rays with relatively low energy (~0.5 keV)(Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 ), compared with those used for crystallography and medical imaging (~5–30 keV)(Reference Garman 20 , Reference Lewis 21 ), are used to illuminate the sample, and transmission is reduced by absorption through carbon-rich structures, such as membranous cellular compartments. As a result, the signal in cryoSXT data appears dark due to X-ray absorption, and the background appears light due to X-ray transmission. This technique is used to resolve cellular compartments to a theoretical resolution limit of 25 nm and produce 3D tomograms of whole-cell ultrastructure(Reference Kounatidis, Stanifer and Phillips 17 ). CryoSXT imaging of cells and tissues takes 5–20 min and thus a large set of tomograms—each containing cellular compartments that need to be delineated by segmentation—can be collected in a relatively short interval(Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 ). However, segmentation tools to mine information out of X-ray tomograms still need to be developed. One reason for this may be that X-ray tomograms are more difficult to segment than electron micrographs captured at room temperature with heavy metal labeling because the use of soft X rays to image the cell volume in 3D under near-native conditions produces higher noise( 22 ). CryoEM and CryoET datasets may be even more difficult to segment than cryoSXT datasets because they have a lower signal-to-noise ratio. This is because these techniques require ultra-thin sections, often generated via focused ion beam milling to produce thin 0.5–1 μm lamellae, which contain less material to scatter electrons or absorb X-rays when compared to cryoSXT datasets that capture the entire depth of the sample(Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 , Reference Turk and Baumeister 23 ).
Although manual segmentation can be used to isolate features of interest, this is more time-consuming for 3D datasets that span the entire depth of the field of view within the cell(Reference Luengo, Darrow and Spink 4 ). The development of machine learning tools for cryoSXT data could increase the rate and efficiency of segmentation. However, the resolution, density and morphology of features can vary widely between cryoSXT datasets (e.g., depending on collection date, passage number of cultured cells, sample preparation strategy)(Reference Müller, Bernard Heymann and Nagashima 24), and this lack of consistency may complicate the use of machine learning tools to segment tomograms. Currently, there is a lack of training datasets for machine learning in the form of segmented volumes from multiple tomograms. SuRVoS has been developed to circumvent the need for training datasets in this form. Instead, individual frames are segmented and used to train segmentation of the whole tomogram(Reference Luengo, Darrow and Spink 4 ). However, this strategy involves training for each tomogram, which is time-consuming, and faster segmentation methods are needed to better cope with the high rate of cryoSXT tomogram acquisition.
Here we developed Contour, a semi-automated segmentation tool for cryoSXT. This tool can be used to segment high contrast features in cryoSXT tomograms, such as mitochondria, lipid droplets, and membranous features. This is achieved by a combination of thresholding based on the projection intensity (i.e., darkness) of the features and applying a width restriction based on the size of the features. This automated procedure can be performed globally (i.e., on the entire tomogram). Some features of interest may be excluded due to the strict width restriction, but segmentation of these features can be refined locally in smaller regions of interest. Contour was developed using Python 3.7 and is available for download on Github with example datasets included (github.com/kamallouisnahas/Contour). The segmentation approach used in Contour is faster than manual segmentation tools as it does not require laborious freehand drawing and interpolation like the Segmentation Editor available in Fiji(Reference Schindelin, Arganda-Carreras and Frise 2 ).
Extracting quantitative data from cryoSXT datasets is a current challenge, and Contour can be used to measure the volume of segmented elements as well as their width along their longest axis. Contour was designed to be used alongside existing segmentation tools: for features that are difficult to segment based on projection intensity and width in Contour (e.g., cytoplasmic vesicles) other segmentation tools can be used to generate segmented volumes that can be imported into Contour for quantitation. We have used Contour in a recent preprint to study how the morphology of mitochondria and cytoplasmic vesicles change during infection with herpes simplex virus-1 (HSV-1)(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). We generated multiple-segmented volumes with Contour and found that mitochondria became more elongated, and vesicles reduced in width as the infection progressed(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). In this paper, we discuss the algorithm and applications of this segmentation tool to cryoSXT data.
2. Results
2.1. The width of cellular compartments and the projection intensity of their voxels can be exploited for semi-automated segmentation
High-contrast cellular compartments in tomograms can be segmented by applying a threshold on voxel intensity. CryoSXT Z stacks were generated for segmentation using IMOD version 5.1.2(Reference Mastronarde and Held 26 ) with a back-projection strategy, radial filtering in the form of a simultaneous iterations reconstruction technique (SIRT)-like filter being subsequently applied to reduce noise. Twenty iterations of the SIRT-like filter were applied to limit blurring and signal loss(Reference Stiller 27 ). Mitochondria have a low voxel intensity (low X-ray transmittance) compared with the cytosol, and an arbitrary threshold range determined by trial and error was used to segment them in a U2OS cell from an 8-bit reconstructed tomogram (Figure 1a)(Reference Okolo, Kounatidis and Groen 18 ). However, segmentation based solely on projection intensity was observed to be highly sensitive to voxel noise and non-specific features, such as the outline of the lipid droplets. In order to increase specificity, an additional segmentation parameter in Contour was used based on the width of the cellular compartments of interest (Figure 1b). Segmentation was first performed on a complete reconstructed cryoSXT Z stack using the global segmentation algorithm in Contour. The segmentation was later refined in smaller regions using the local segmentation algorithm.
Semi-automated segmentation by analyzing the intensity and width of cellular features. (a) The mitochondria in a tomogram of a U2OS cell were segmented by applying a voxel intensity threshold (blue arrows) LD, lipid droplet; Mito, mitochondrion. This technique was highly sensitive as most of the mitochondria were included and only a few areas were missing (white arrows). However, intensity thresholding alone led to noise and non-specific features such as the outline of lipid droplets being included in the segmented volume (orange arrows). (b) In Contour, a width restriction was applied in addition to an intensity threshold to segment the mitochondria. Any voxels included in the threshold range would only be included in the product segmented volume if they formed part of a 10 × 10 voxel area or larger (I–IV). The segmented product was specific to mitochondria, with less noise and fewer unwanted elements. However, there were more falsely-excluded areas due to the higher specificity (white arrows). (c) The remaining non-specific elements were manually erased (red box) and local regions of interest containing the excluded areas were identified (white boxes) and (d) the analysis was reattempted with a smaller width restriction of 4 voxels (green fill). (e) The final segmented volume was rendered in 3D using 3D Viewer in Fiji(Reference Schindelin, Arganda-Carreras and Frise 2 ).
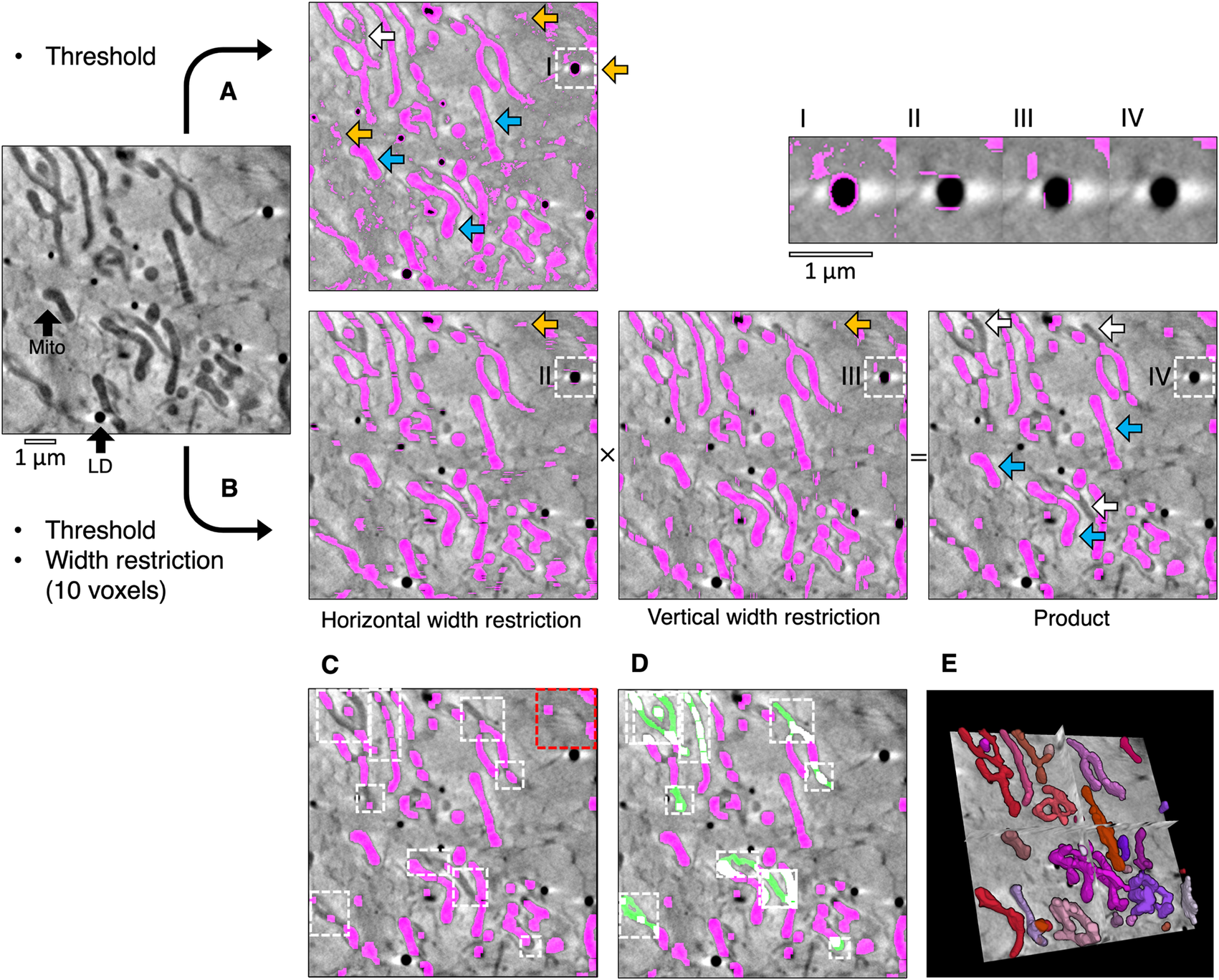
During global segmentation, the same threshold range applied in Figure 1a was applied to the tomogram in Figure 1b to isolate voxels of the desired intensity and to produce binary masks for each Z image (0 for background voxels and 1 for segmented voxels). A width restriction was determined by manually inspecting the width of the mitochondria and was applied in the second step to exclude noise and non-specific elements smaller in width than the mitochondria, such as the outline of lipid droplets. In order to apply this restriction without the slow process of iterating through each voxel, the binary masks were compressed in a lossless manner by run-length encoding(Reference Hardi, Angga, Lydia, Jaya and Tarigan 28 ). Using this compression method, the run of voxel values (e.g., 000110000) in the binary mask was compressed into a sequence where the voxel value was coupled to the number of times it appeared consecutively (e.g., (0,3), (1,2),(0,4)). The width restriction was applied to the compressed sequence by converting voxels with a value of 1 to 0 if the number of consecutive voxels was lower than the desired width. The data compression and width restriction were applied twice independently along rows and columns in the horizontal and vertical directions, respectively, and the modified sequences were decompressed into two full binary masks. Voxels segmented within the threshold range were converted into background if their width was less than the width restriction. As a result, the segmented voxels that remained appeared as stripes with a width greater than or equal to the width restriction. The stripes were horizontal or vertical depending on the direction in which the width restriction was applied (Figure 1b). The arrays of voxels that made up the horizontal and vertical binary masks were multiplied together such that only coordinates that contained a voxel of 1 in both masks (i.e., 1 × 1) were included in the product segmented volume and all other combinations were converted to background (i.e., 1 × 0, 0 × 1, and 0 × 0). This multiplication step eliminated most noise by ensuring that only rectangular matrices of dimensions width × width or larger remained. In some cases, horizontal and vertical stripes were produced from noise or non-specific features, such as the outline of lipid droplets. Voxels at the intersection between these stripes (i.e., 1 × 1) were also included after the multiplication step. The run-length encoding, width restriction, and data decompression were reapplied to the product segmented array to filter out these artifacts. The combined application of thresholding and a width restriction results in a better-defined segmentation with less noise and fewer non-specific elements. However, the increase in specificity afforded by width analysis can lead to some desired elements becoming excluded from the segmented volume. In the presented example, the global segmentation step excluded several areas based on the minimum width restriction (Figure 1c). These areas could be filled by using the local segmentation algorithm in Contour, whereby thresholding and width restriction were applied locally in a smaller 3D region of interest containing these excluded areas (Figure 1d) using a lower minimum width value (4 voxels). Given that local segmentation is performed on a smaller 3D region of interest, there is no requirement for data compression by run-length encoding before applying width restriction to improve analysis efficiency(Reference Hardi, Angga, Lydia, Jaya and Tarigan 28 ).
It is likely that local segmentation will be required following global segmentation. However, global segmentation of the complete Z stack is not required before performing local segmentations. If it is determined that the cytoplasm is too dense with high-contrast compartments to perform a global segmentation, this step can be skipped and local segmentations can be performed on the entire tomogram instead (Figure 2a and Table 1). In addition to the global and local segmentation algorithms, manual “fill” and “erase” options are available for manual adjustment of the segmented volumes (Figure 1c,d). The segmented volume can be rendered using 3D Viewer in Fiji (Reference Schindelin, Arganda-Carreras and Frise 2 ) or other appropriate visualization software (e.g., Amira [Thermo Scientific] or Chimera/ChimeraX [UCSF](Reference Goddard, Huang and Meng 29 )) (Figure 1e).
Segmentation pipeline and decision tree in Contour. (a) Global and local segmentation algorithms can be applied to delineate cellular compartments from a cryoSXT Z stack or from smaller 3D regions of interest. Global segmentation is recommended if the cellular compartments are dispersed throughout the tomogram. For smaller regions of interest, the local algorithm can be used to discriminate features in crowded areas or features excluded from the global segmentation. The threshold range and width restriction parameters can be modified to optimize the specificity and sensitivity of the global segmentation. (b) Discrete segmented elements can be differentiated and their volumes and widths can be calculated. Any elements smaller in volume than a specified number of voxels can be filtered out and this can be used to eliminate small segments of noise in one step. (c) Final touches can be applied to improve the appearance of the segmented volumes. A smoothing function can be used to smoothen blocky edges in 2D slices and a Gaussian blur can be applied to reduce the appearance of layering in between slices of the segmented volume (Figure 4). Predicted lengths of time for each process can be found in Supplementary Table S1.
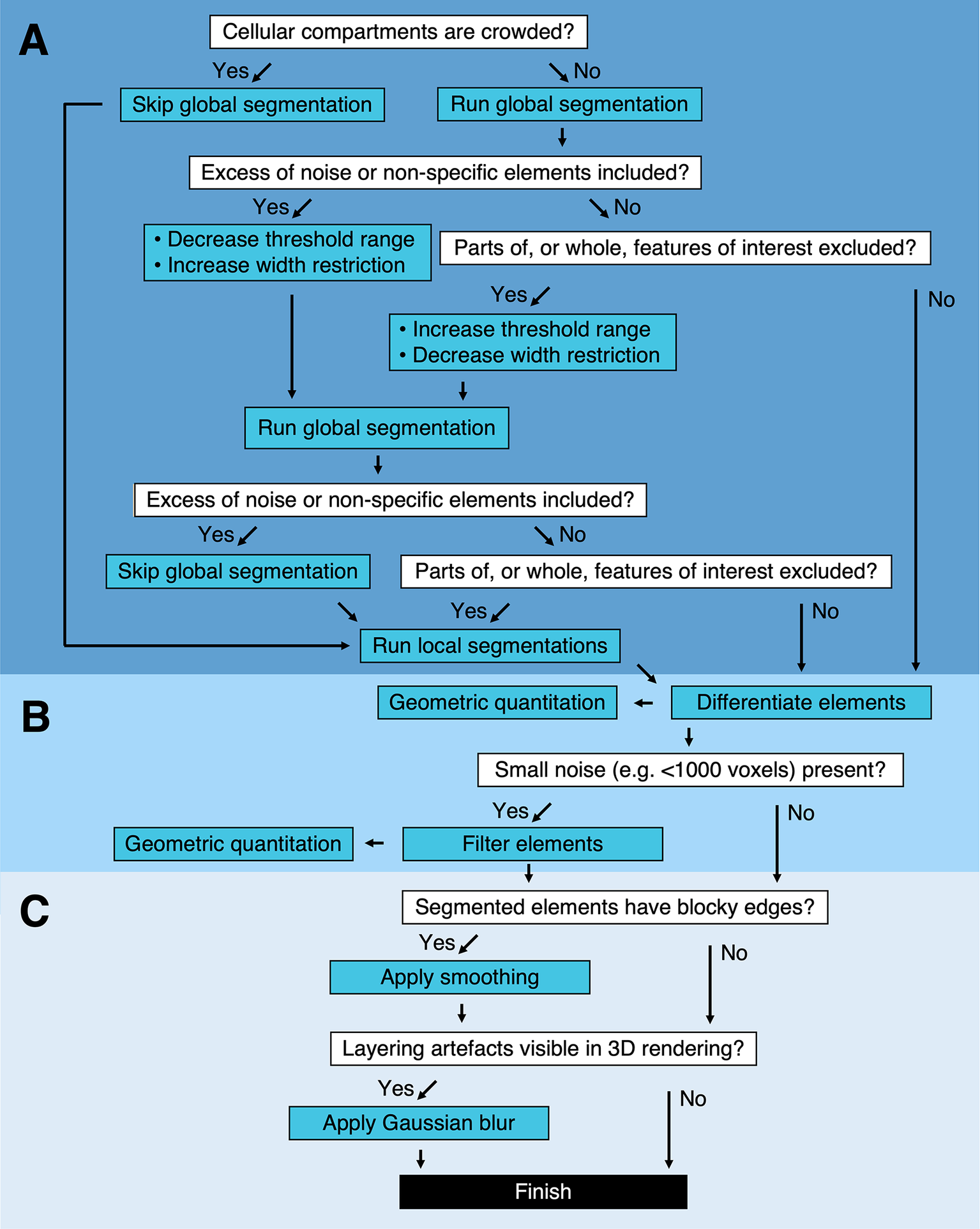
Troubleshooting segmentation in Contour.

2.2. Applications of Contour to analyze the geometry of cellular compartments
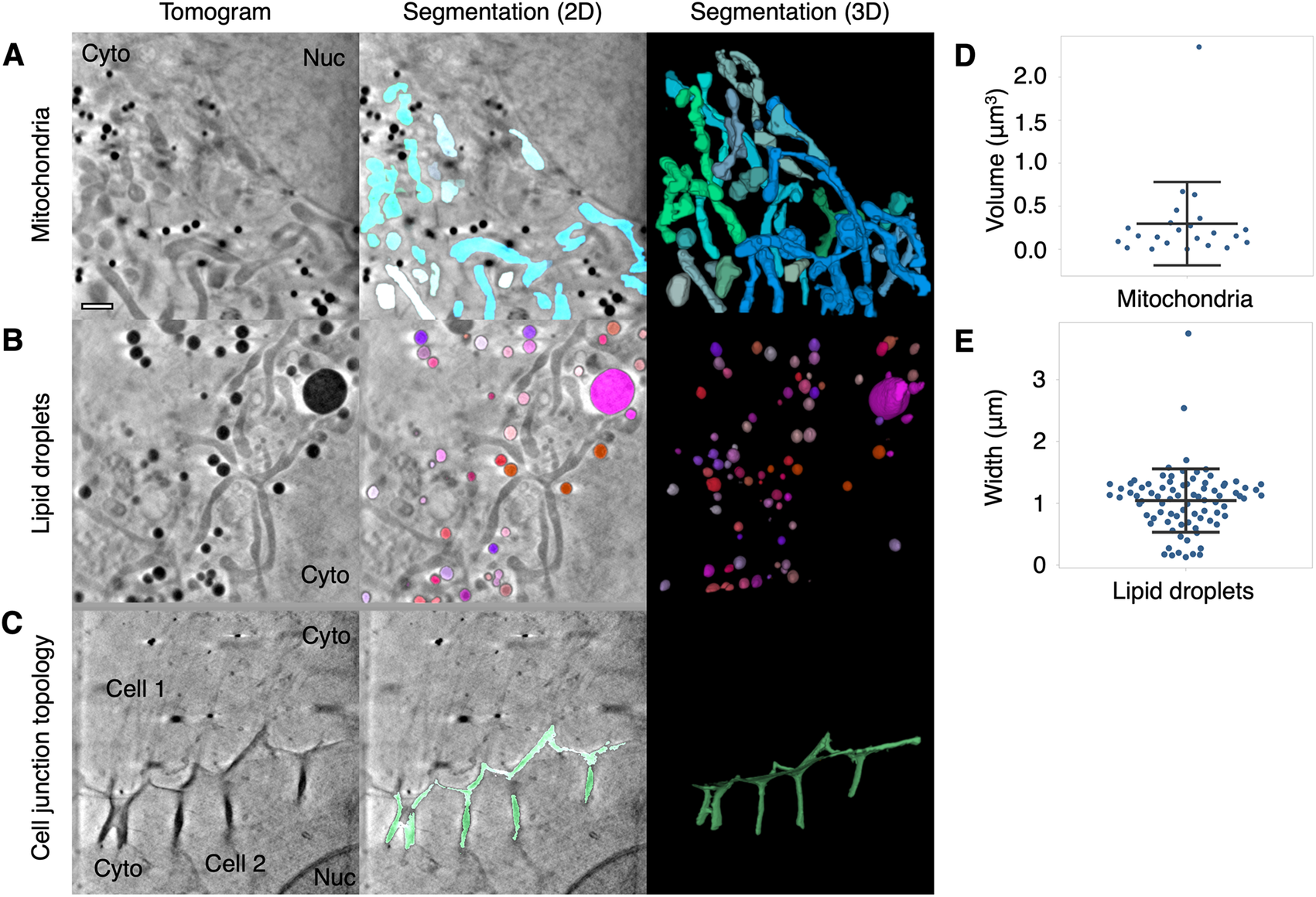
We have shown that mitochondria can be segmented using the global and local segmentation parameters based on their intensity and width (Figures 1, 2, and 3a). We have also been able to use Contour to segment mitochondria from other cell types, including a cultured human keratinocyte (HaCaT, Supplementary Figure 1a), a human foreskin fibroblast immortalized by a human telomerase reverse transcriptase (HFF-hTERT, Supplementary Figure 1b), and a green monkey kidney epithelial cell (Vero, Supplementary Figure 1c). The HaCaT cell (Supplementary Figure 1a) was captured using a 40 nm zone plate whereas the HFF-hTERT and Vero cells were captured using a 25 nm zone plate, demonstrating mitochondria can be segmented from tomograms collected using either zone plate. We were also able to segment a swollen mitochondrion from a U2OS cell containing visible cristae (Supplementary Figure 1d). We have used Contour to segment mitochondria in a recent preprint where we studied how mitochondrial morphology changes during HSV-1 infection. We found that mitochondria transitioned from a heterogenous morphology in uninfected U2OS cells to a more consistently elongated and branched formation as the infection progressed(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). Contour can be used to segment other cellular compartments based on intensity and width, such as lipid droplets (Figure 3b) and features at the cell surface or at cell–cell junctions, such as large internalizations of the plasma membrane that may resemble bulk endosomes arising from clathrin-independent endocytic events (Figure 3c)(Reference Shafaq-Zadah, Dransart and Johannes 30 ).
Segmentation and quantitation of cellular features. Contour can be used to segment high contrast features in U2OS cells such as (a) mitochondria, (b) lipid droplets, and (c) distinctive membrane topology at cell–cell junctions. Cyto, cytoplasm; Nuc, nucleus. Quantitative data can be extracted from the segmented volumes. (d) The mitochondria in this 9.46 × 9.46 μm2 field of view of a U2OS cell had a mean volume of 0.3 ± 0.48 μm3 SD. (e) The mean width along the longest axis of each lipid droplet in this 9.46 × 9.46 μm field of view of a U2OS cell was found to be 1.04 ± 0.51 μm SD. Scale bar = 1 μm. Error bars show mean ± SD.
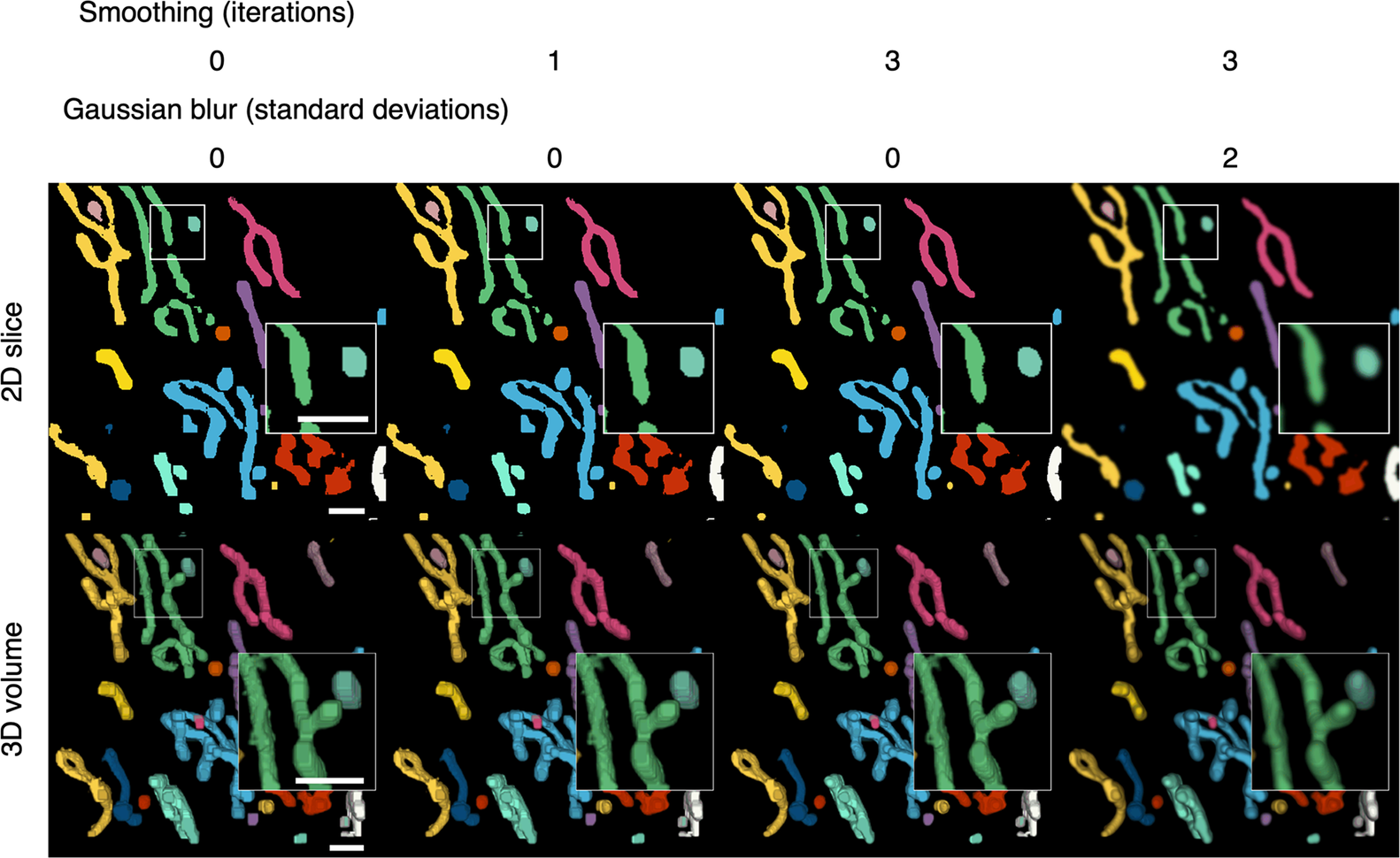
Discrete segmented elements can be differentiated from each other and color-coded to aid the discrimination of the components (Figures 2b and 4). This is achieved by assigning a common ID number to segmented voxels and their direct-contact neighbors. The inclusion criteria for direct-contact neighbors are any two voxels that are at XY coordinates that differ by one step in any of the eight cardinal (N, S, E, or W) and ordinal (NE, SE, SW, or NW) directions; or any two voxels at the same XY coordinate in tandem Z planes.
Color-coding of differentiated elements and smoothing of the 3D volume. Segmented voxels are grouped together into separate elements that can be color-coded to help distinguish them from each other. A smoothing function can be applied to 2D arrays of voxels to smooth the edges of segmented elements. Because the smoothing is applied to the 2D slices, layering artefacts can be observed in between the slices. A Gaussian blur can be applied per 2D slice to reduce the appearance of layering artefacts. Scale bars = 1 μm.
Quantitation of the geometry of cellular features is a current challenge in cryoSXT because segmentation is often a prerequisite and measurements may need to be taken at an angle distinct from the slices of the 3D projection(Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 ). Contour has the capacity to automatically calculate the volumes of cellular features (in units of voxels) along any axis once the user has differentiated these elements. For example, the mean volume of the mitochondria in a single 9.46 × 9.46 μmReference Schindelin, Arganda-Carreras and Frise 2 field of view of a U2OS cell, given a voxel size of 10 × 10 × 10 nm3, was calculated to be 0.3 ± 0.48 μm3 (mean ± SD; Figure 3d). The width of each segmented element along its longest axis, which may not be parallel with the slices of the tomographic projection, can also be calculated in this program. This is achieved by isolating the voxels at the perimeter of each segmented element in each image plane and calculating all combinations of the distance (i.e., modulus) between any two of these voxels across the complete Z stack. The longest of these moduli is presented as the width of the segmented element in units of voxels. The longest width of each lipid droplet was calculated for a 9.46 × 9.46 μm2 field of view and the droplet width was found to be 1.04 ± 0.51 μm (mean ± SD; Figure 3e). Segmented volumes generated with other segmentation tools, such as Segmentation Editor in Fiji(Reference Schindelin, Arganda-Carreras and Frise 2 ), can be imported into Contour for quantitation based on the methods described above.
2.3. Polishing the segmented volume
After the segmented elements have been differentiated, final touches can be applied to improve the appearance of the 3D volume (Figure 4). The width restriction applied during the segmentation filters out any segmented voxels that do not form part of a width × width area or larger. As a result, segmented elements may appear blocky. A smoothing function is available to smoothen the edges of segmented elements (Figure 4). Each segmented plane in the Z stack is converted into a binary mask (0 for background and 1 for segment) and is translated by one step in all eight cardinal and ordinal directions and the voxel arrays are added together such that voxels may have a value of 0 to 8. Voxels with less than a median of five, which occur at the perimeter of segmented elements, were transformed into background, resulting in the trimming of the edges of the segmented elements. A greater number of iterations of this function will increase the extent of smoothing but reduce the width of the segmented elements. A compromise of 1–3 iterations is recommended to avoid over trimming (Table 1). The smoothing function is only applied within slices of the segmented volume and layering artifacts can be observed in between slices. A two-dimensional Gaussian blur can also be applied per slice to reduce the appearance of layering artefacts and improve the 3D rendering of the volume.
3. Discussion
Here we reported the development of Contour, a segmentation tool for highly contrasting cellular features in cryoSXT tomograms that analyses the projection intensity (i.e., darkness) and width of cellular compartments. This program also calculates 3D geometric measurements from the segmented elements. We demonstrate that mitochondria, lipid droplets, and the topology of the cell surface at cell–cell junctions can be segmented using this technique. Contour was developed to accelerate segmentations of cryoSXT tomograms. Existing segmentation techniques may be time-consuming and laborious to users: manual segmentation tools require the user to trace the edges of features in periodic Z planes and interpolate between them and, although machine-learning tools such as SuRVoS are available, these tools require fresh training for each tomogram( 3 , Reference Luengo, Darrow and Spink 4 , Reference Harkiolaki, Darrow, Spink, Kosior, Dent and Duke 16 ). The algorithm used by Contour for segmentation is largely automated, allowing users to perform either a global segmentation on a complete cryoSXT Z stack or local segmentations in regions of interest. In either case, training datasets are not required, and the user does not need to trace around features, making the process less laborious and subjective(Reference Luengo, Darrow and Spink 4 ).
We have applied Contour to one study, where we investigated how HSV-1 infection alters the morphology of cellular compartments, and we were able to segment mitochondria in multiple tomograms(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). The dependency on low projection intensity and width for the segmentation does pose some limitations. For example, some cellular compartments such as mitochondria may have uneven intensities(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25). It is still possible to use Contour for these features, but successful analysis requires a greater number of local segmentations to be carried out with different threshold ranges (Table 1). The use of a width restriction parameter to distinguish features from noise complicates the application of this technique to thin cellular features, such as cytoskeletal filaments that are normally less than five voxels in width(Reference Heinrich, Bennett and Ackerman 32 ). Cytoplasmic vesicles often have a highly contrasting membrane but a light lumen, making it difficult to segment such features when applying a minimum width restriction. Although we did not use Contour to segment cytoplasmic vesicles in our recent study(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ), we used Contour to calculate the longest widths of each vesicle that we manually segmented using Segmentation Editor in Fiji(Reference Schindelin, Arganda-Carreras and Frise 2 ). We therefore show that Contour can be used in conjunction with other segmentation tools to calculate quantitative data. Our semi-automated segmentation tool could be used to generate sufficient segmented volumes of different cellular compartments to facilitate the training of machine learning algorithms in the future. CryoSXT is a growing technique and its applications are becoming more widespread in biomedical imaging, especially as a correlative imaging tool with cryoSIM(Reference Kounatidis, Stanifer and Phillips 17 –Reference Vyas, Perry and Okolo 19 ). Contour is a largely automated tool designed to significantly accelerate the segmentation of X-ray tomograms and the quantitation of geometric structures within.
4. Materials and Methods
4.1. Sample preparation
A total of 3 mm gold EM grids with a holey carbon film (R 2/2, 200 mesh; Quantifoil Cat no. AU G200F1 finder, batches Q45352 & Q45353) were glow discharged and treated with filtered poly-l-lysine for 10 minutes (Sigma Aldrich Cat no. P4832). U2OS cells (ATCC HTB-96; RRID CVCL_0042) were seeded onto the grids at 3 × 105 cells per well in a 6-well plate. The cells were cultivated overnight in Dulbecco’s Modified Eagle’s Medium (DMEM; Thermo Fisher Scientific, Cat no. 45011590366) supplemented with 10% (v/v) fetal bovine serum (FBS; Capricorn, Cat no. FBS-11A), 4 mM l-glutamine (Thermo Fisher Scientific, Cat no. 25030081), and penicillin/streptomycin (10,000 U/ml; Thermo Fisher Scientific, Cat no. 15070063). A total of 2 μL of gold fiducials (BBI Solutions; EM.GC250, batch 026935) were added to the grids as previously described (Reference Okolo, Kounatidis and Groen 18 ) and the grids were blotted with for 0.5–1 s at 30°C and 80% humidity with a Leica EM GP2 plunge freezer. The grids were plunged into liquid ethane and then transferred into liquid nitrogen. The tomograms presented in this paper were collected for a study of the effect of HSV-1 infection on the morphology of cellular compartments in U2OS cells(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). All tomograms in Figures 1– 4 were collected from uninfected cells except for Figure 3b, which was collected from a cell infected with one plaque forming unit per cell of HSV-1 as previously described(Reference Nahas, Connor, Harkiolaki, Crump and Graham 25 ). The HaCaT cells in Supplementary Figure S1 were infected with two plaque-forming units per cell of HSV-1 and were fixed with 4% paraformaldehyde. The HFF cells were infected with three plaque forming units per cell of HSV-1.
4.2. Cryo-soft-X-ray tomography
CryoSXT data were as collected at beamline B24 at the UK synchrotron Diamond Light Source using a UltraXRM-S/L220c X-ray microscope (Carl Zeiss X-ray Microscopy). Soft X-rays (500 eV, λ = 2.48 nm) were focused onto the grid sample by diffraction using a diffraction grating known as a zone plate, which can achieve a nominal resolution of 25 nm. A 1024B Pixis CCD camera (Princeton instruments) was used to collect tomographic data from U2OS cells with a 9.46 × 9.46 μm field of view by rotating the grid within the range−60° to +60° at increments of 0.5° or 1.0° and X-ray exposure times of 0.5 or 1.0 s. A single-axis alignment of the tomographic images was generated using IMOD (version 4.9.2)(Reference Mastronarde and Held 26 ). A coarse alignment with a high-frequency cut-off radius of 0.1 and a subsequent fine alignment with fiducial tracking were used to align the images. The data was reoriented in 3D using a boundary model. A final alignment was carried out using linear interpolation and tomograms were reconstructed using the back projection strategy with radial filtering to reduce noise in the form of 20 iterations of simultaneous iterations reconstruction technique (SIRT)-like filter(Reference Stiller 27 ). The tomograms were converted from a 16-bit signed format to an 8-bit format before segmentation.
4.3. Global segmentation
Tomographic images are stored as NumPy(Reference Harris, Millman and van der Walt 33 ) arrays in Python 3.7 and the images in the Z plane are stored in a list. Datasets with a field of view greater than 512 × 512 voxels were downscaled by a multiple of two to improve the efficiency of the program and the scaling was accounted for during quantitation. A threshold range with a desired minimum and maximum value was applied to produce binary masks for each image (0 for background and 1 for segmented voxels). The sequence of 0 and 1 s is compressed losslessly by run-length encoding into a paired sequence where the value is coupled to the number of times it is repeated(Reference Hardi, Angga, Lydia, Jaya and Tarigan 28 ). Values of 1 are converted to 0 if the number of repetitions is lower than the width restriction and the processed sequence is decompressed into a full array. The run-length encoding and width restrictions are applied twice independently—down columns and along rows. Both binary arrays are multiplied together so that only voxels with a value of 1 in both arrays are included in the product array. This process is repeated to remove artifacts.
4.4. Local segmentation
A cuboidal region of interest is selected from the Z stack and a threshold range is applied to produce binary masks for each image. A width restriction is applied by iterating through the voxels in each image plane in the region of interest and counting the number of repeats. If the number of repeats is lower than the width restriction the values are converted from 1 to 0. This process is run along rows to produce a new array. This new array is used as the input array to rerun the width restriction down columns. This process is repeated once along rows and columns to remove artefacts.
4.5. Quantitation and filtering
Segmented voxels were attributed with integer IDs that served to distinguish discrete elements. IDs were shared between neighboring voxels that were one position away from each other in all cardinal (N, S, E, or W) and ordinal (NE, SE, SW, or NW) directions or voxels with matching XY coordinates in tandem Z planes. Neighboring voxels were first grouped together in two dimensions in the XY planes. Any two-dimensional groups from tandem Z planes were merged into one 3D group if they contained voxels whose coordinates overlapped in XY. This process was run in ascending and descending order of Z slices to ensure that segment branches, which were separated from the main body of the segment in some slices, were not excluded from the 3D merger owing to the direction of iteration through the Z stack. The volume of each 3D segment was calculated in units of voxels. This was done by isolating all the voxels in the segmented volume with a given ID using the NumPy.argwhere function(Reference Harris, Millman and van der Walt 33 ), which produces an array of XY coordinates corresponding to these voxels per Z slice. The length of the arrays for each slice was calculated and divided by two to retrieve the number of voxels. Small segmented elements of noise were eliminated by replacing any elements with a volume of less than a desired volume threshold (e.g., 1,000 voxels) with background voxels of value 0.
The width of a 3D segmented element was calculated by finding the longest distance between any two voxels in the segment. First, the voxels present at the perimeter of elements were filtered from all the voxels in the segment by determining if any neighboring voxels have a value of 0 (background). Second, the modulus between all combinations of two perimeter voxels was calculated (equation (1)). The longest modulus was given as the width of the segment in units of voxels. Stacks of binary masks containing elements with known volumes and widths were generated to verify the quantitation functions and are available at https://github.com/kamallouisnahas/Contour/tree/main/known_quantities.
The modulus of all vectors connecting perimeter voxels a and b was calculated from coordinates x, y, and z.
4.6. Smoothing and Gaussian blur
The edges of segmented elements were smoothed by translating the arrays of voxels for each slice in the tomographic projection by one voxel in each cardinal and ordinal direction. Binary masks were used and each segmented voxel had a value of 1. A sum array was produced by adding together all eight translated arrays, such that voxels ranged from 0 to 8. A median array was calculated from the sum array by transforming voxels <5 into values of 0 and voxels ≥5 into values of 1. Several iterations of this function (up to 3) were applied to increase the extent of smoothing.
The Gaussian filter function from the SciPy library(Reference Virtanen, Gommers and Oliphant 34 ) was applied with a standard deviation of one to each of the three colors individually in RGB images of the differentiated segmented elements. Quantitation of volume and width were not affected by the smoothing and Gaussian blur functions.
4.7. Statistics
SuperPlots was used to generate scatterplots and to calculate the mean and standard deviation for the volume of mitochondria and the width of lipid droplets(Reference Lord, Velle, Mullins and Fritz-Laylin 35 ).
Acknowledgments
We thank Diamond Light Source for access to beamline B24 (mx18925, mx19958, bi21485, and bi23508) and the experimental hall coordinators for helpful support. We thank members of beamline B24 at the Diamond Light Source (Mohamed Koronfel, Ilias Kounatidis, Chidinma Okalo, and Matt Spink) for technical support with cryoSXT. We thank Thomas Fish (Diamond Light Source) for his assistance with NumPy array functions.
Competing Interests
The authors declare no competing interests exist.
Authorship Contributions
K.L.N., J.F.F., M.H., C.C., and S.G. conceptualized the study; K.L.N. curated the data and visualization; M.H., C.C., and S.G. provided funding; Investigation was performed by K.L.N., N.V., C.C., M.H., and S.G.; M.H., C.C., and S.G. administered the project; M.H. provided resources; K.L.N. provided software; M.H., C.C., and S.G. supervised the project; K.L.N. and M.H. wrote the original draft and K.L.N., J.F.F., M.H., C.C., and S.G. reviewed and edited the draft.
Funding Statement
This work was supported by a Biotechnology and Biological Sciences Research Council (BBSRC) Research Grant (CMC, BB/M021424/1), a Sir Henry Dale Fellowship, jointly funded by the Wellcome Trust and the Royal Society (SCG, 098406/Z/12/B). This work was carried out with the support of the Diamond Light Source, instrument B24 (proposals mx18925, mx19958, bi21485, and bi23508). This research was funded in whole, or in part, by the Wellcome Trust [098406/Z/12/B].
Data Availability Statement
The raw data tilt series and tomographic reconstructions of the cryoSXT datasets presented here can be accessed from the Apollo repository (University of Cambridge): https://doi.org/10.17863/CAM.78593 and the EMPIAR repository (EMBL-EBI): EMPIAR-10972. The source code is available under a GNU General Public License v3.0 from https://github.com/kamallouisnahas/Contour. The segmented volumes and quantitative data can be accessed from: https://github.com/kamallouisnahas/Contour/tree/main/repository.
Supplementary Materials
To view supplementary material for this article, please visit http://doi.org/10.1017/S2633903X22000046.






