Among micronutrients, iron plays a pivotal role in many pathophysiologic and cell processes such as the transportation of oxygen, Hb formation, DNA synthesis and energy generation(Reference Lopez, Cacoub and Macdougall1). Iron deficiency (ID) is the most common nutritional deficiency and a public health matter of great importance. It is related to several causes, including insufficient intake and poor absorption, and is associated with physical, cognitive and immune impairment(Reference Lopez, Cacoub and Macdougall1,2) . The number of ID cases worldwide increased from 984·61 million in 1990 to 1270·64 million in 2021, and by 2050, it is expected to reach 1439·99 million. Disability-adjusted life years from ID were 32·32 million in 2021 and are expected to reach 36·13 million by 2050(Reference Wang, Liang and Huangfu3). ID is one of the main causes of anaemia (ID anaemia)(Reference Kassebaum, Jasrasaria and Naghavi4), especially for high-risk populations such as the older population(Reference Stahl-Gugger, de Godoi Rezende Costa Molino and Wieczorek5). In older individuals, malnutrition, impaired absorption, coexisting chronic diseases and medication may lead to ID, which in turn has been linked to all-cause mortality and morbidity(Reference Stahl-Gugger, de Godoi Rezende Costa Molino and Wieczorek5,Reference Kehoe, Walton and Flynn6) . Thus, identifying older adults as a high-risk group for ID, as well as dietary and lifestyle factors that contribute to this risk, is crucial. Serum ferritin levels (SFL) are the most specific indicator for the diagnosis of ID, as proposed by the majority of guidelines, albeit suggested cutoffs differ(Reference Peyrin-Biroulet, Williet and Cacoub7). According to the WHO, a cutoff of SFL < 15 ng/ml is recommended for the diagnosis of ID, yet additional cutoffs are used in everyday practice such as SFL < 30 ng/ml(2,Reference Peyrin-Biroulet, Williet and Cacoub7) . SFL, as an acute-phase protein, rises during inflammation, and age-related chronic low-grade inflammation (‘inflammaging’) can obscure its portrayal of iron status(Reference Garcia-Casal, Pena-Rosas and Pasricha8). Therefore, the WHO recommends measuring an additional acute-phase marker, such as C-reactive protein (CRP), alongside SFL, with various adjustment methods proposed(2).
Traditionally, the Greek population adhered to the Mediterranean diet (MD), exerting a beneficial effect on health(Reference Koutsonida, Kanellopoulou and Markozannes9). MD contains several iron chelators, which bind to intracellular labile iron, thus protecting the cells from oxidative stress(Reference Barbouti, Lagopati and Veroutis10). However, concerns have been expressed that despite higher iron intakes from an MD(Reference Castro-Quezada, Roman-Vinas and Serra-Majem11), these may not necessarily result in higher iron status, due to restricted consumption of red meat, important for bioavailable iron. Additionally, phytate – which is found in whole-grain cereals, an important constituent of MD – inhibits iron absorption, possibly resulting in lower iron status(Reference Hurrell and Egli12).
The purpose of this secondary analysis of the HYDRIA study is to assess SFL, recognised as the most commonly used marker of iron stores(Reference Peyrin-Biroulet, Williet and Cacoub7,Reference Camaschella13) , and to determine the prevalence of ID in the older adult population in Greece and its association with several sociodemographic and lifestyle factors, including dietary iron intake and adherence to MD.
Material and methods
Study design and population
HYDRIA is a large nationally representative Greek survey that investigated food, macronutrient and micronutrient intakes in a sample of 4011 adults over 18 years of age, providing observations and conclusions on population health in relation to the nutritional status of the population. Field data were collected between June 2013 and December 2014. The survey was conducted in accordance with the guidelines set by the Helsinki Declaration and national legislation regarding data protection. The methodology of HYDRIA was based on the recommendations of the European Food Safety Authority (EFSA)(14) and the European Health Examination Survey(Reference Tolonen15). The HYDRIA survey characteristics, sampling methods, inclusion criteria and the questionnaires provided to the participants have been previously discussed in detail(Reference Martimianaki, Papatesta and Peppa16,Reference Martimianaki, Peppa and Valanou17) .
Out of the initial 4011 HYDRIA participants, SFL measurement was restricted to participants above the age of 55 years due to financial constraints (n 748). Participants who reported use of iron-containing medication, defined as iron preparations (B03A) within the Anatomical Therapeutic Chemical classification system(18) and dietary supplements of any kind, were excluded from the analysis, since consumption of such preparations could suggest a relevant health condition that would introduce bias to the analysis. Thus, the remaining sample size was 517 participants. The HYDRIA survey was approved by the Hellenic Health Foundation’s Bioethics Committee in accordance with the Declaration of Helsinki. All participants provided their informed written consent prior to their inclusion in the study.
Biochemical analysis
The blood samples were analysed by ‘Bioiatriki’ Healthcare Group. The electrochemiluminescence immunoassay, ECLIA, was used to quantify SFL. The method used for CRP was nephelometry, and high-sensitivity CRP was measured.
Dietary assessment
Dietary intake was assessed using two non-consecutive 24-h dietary recalls (24-HDR) per participant, with a time interval of approximately 15–30 d between them. During the 24-HDR, participants recalled their food and beverage intake and answered questions, among others, about the food preparation and the amounts consumed, using the Hellenic Health Foundation Nutrition Tool.
The adherence to the MD pattern was assessed using a 9-point Mediterranean diet score (MDS). For the estimation of MDS, each of the nine dietary components was scored 0 or 1 based on sex-specific median intakes. Vegetables, legumes, fruits and nuts, cereals and fish scored 1 for intakes at or above the median and 0 otherwise; meat and dairy products scored 1 for intakes below the median and 0 otherwise. Alcohol scored 1 for moderate consumption (10–50 g/d for men, 5–25 g/d for women). The monounsaturated:saturated fat ratio was used to assess fat intake (lipids). The total MDS ranged from 0 (minimal adherence) to 9 (maximal adherence)(Reference Trichopoulou, Costacou and Bamia19).
Statistical analysis
The association between SFL (ng/ml) with adherence to MD, dietary iron intake and other potential predictors was examined using linear regression models. In these models, SFL was log-transformed to better approach normality. The prevalence of ID, defined as SFL lower than 15 ng/ml and 30 ng/ml, was estimated overall, as well as by age and sex. SFL were adjusted for inflammation according to the BRINDA (Biomarkers Reflecting Inflammation and Nutritional Determinants of Anaemia) method, using only CRP, since alpha-1-acid glycoprotein was not measured in the study(Reference Luo, Geng and Zeiler20). Potential predictors of ID were also examined, in this case by logistic regression models for both thresholds.
MDS was categorised as low (0–3), intermediate (4–6) and high (7–9). Total dietary iron intake (mg/d) and dietary iron intake from haem iron sources, that is, red meat, fish and poultry (haem iron, mg/d), were also examined as predictors of SFL and ID. The dietary iron intake for each participant was estimated with the use of the mean of their two 24-HDR. Haem iron intake was estimated by considering 40 % of the total iron in meat, poultry and fish as haem iron, based on previously described methodology.(Reference Monsen, Hallberg and Layrisse21) To remove confounding by total energy intake, account for inter-individual differences in food consumption and mitigate the impact of dietary misreporting associated with 24-HDR, iron intake was energy-adjusted to 2000 kcal/d, the standard reference for adults, using the nutrient-density method (absolute nutrient intake divided by total energy)(Reference Willett, Howe and Kushi22,Reference Poslusna, Ruprich and de Vries23) .
For the evaluation of the daily iron requirement, the EFSA population reference intake was used, that is, the intake level that meets the needs of 97·5 % of the individuals in the population, which is 11 mg/dl for males and postmenopausal females(24).
Further potential predictors for both linear and logistic regression models included sex; age grouped by 10 years (55–64, 65–74, 75+); BMI (< 25, 25 to < 30, equal or > 30); level of education: low (≤ 9 years of formal education), intermediate (> 9 to ≤ 12 years) and high (> 12 years, including those with a bachelor’s degree or higher)(25); cigarette smoking (current smoker, former smoker, non-smoker); and high-sensitivity CRP. To account for skewness, high-sensitivity CRP was log-transformed when entered in regression models. The categories of ‘low’ and ‘within normal limits’ BMI (BMI < 25) comprised one larger category due to the very low number of participants with a BMI below the normal level (BMI < 18·5). Other than that, the cutoff values to determine the BMI categories followed the respective WHO recommendations(26). Physical activity was calculated in metabolic equivalents of task (MET), representing the ratio of energy expended in an activity to that when resting. Daily physical activity was calculated by multiplying the duration (h/d) of each reported activity by its corresponding MET value and adding the cumulative MET-hours across all activities(Reference Ainsworth, Haskell and Herrmann27).
All analysis has been performed considering HYDRIA’s two-stage, stratified random sampling design. The crude associations between SFL and ID and each predictor were examined by weighted univariable linear and logistic regression models. Multivariable analyses were also considered.
Level of significance was set at α equal to 0·05. The statistical analysis was performed with STATA/se 11·0 (StataCorp, College Station).
Results
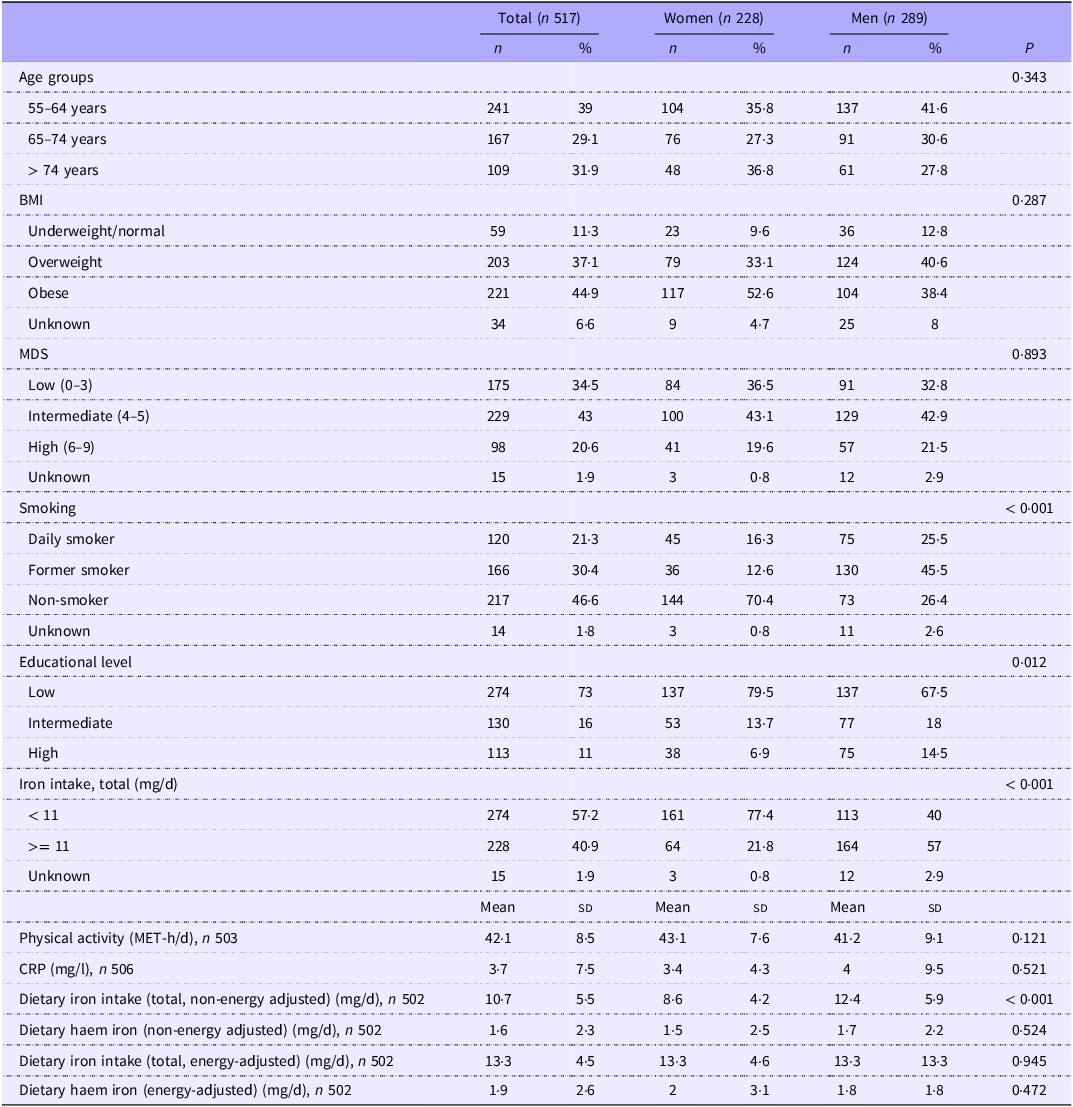
The sample includes 517 individuals, of whom 54·1 % men. The majority of the participants were overweight (37·1 %) or obese (44·9 %), and 73 % of the participants had a low educational level, while 46·6 % of them were non-smokers. The adherence to the MD paradigm was mostly intermediate (43 %). Mean CRP levels were 3·7 mg/l, and 84·8 % of the participants had normal CRP levels (i.e. < 5 mg/l). The mean dietary iron intake in mg/d was 10·7 and 1·6 for total iron and haem iron, respectively (Table 1).
Counts (n) and percentages (%) by categorical variables’ levels and mean values and sd for the continuous variables of the 517 study participants

MDS, Mediterranean diet score; MET, metabolic equivalents of task; CRP, C-reactive protein.
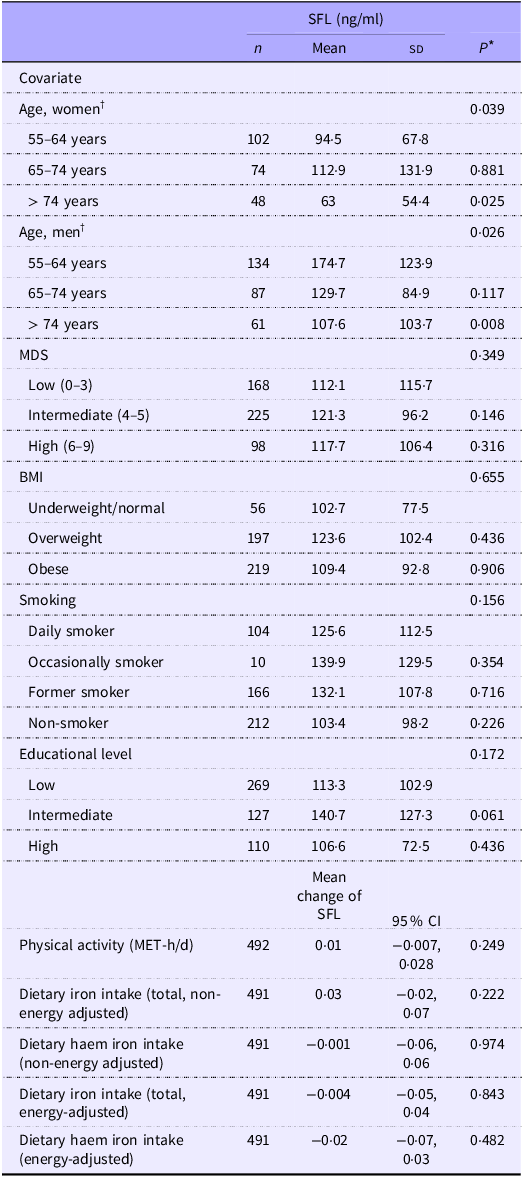
After BRINDA adjustment, mean SFL was 117 ng/ml overall (men: 142 ng/ml, women: 88 ng/ml), while the effect of age was different in men and women (age and sex interaction P-value < 0·001). Men demonstrated, on average, a more prominent and consistent decrease of SFL with increased age, with a drop of 38·4 % in the age group of 75+ (P = 0·008) compared with the 55–64 age group. Women had, on average, the highest SFL in the age group of 65–74 years, although this difference was not statistically significant when compared with women aged 55–64 years (P = 0·881). Above the age of 74, women experienced a drop of 44·2 %, compared with women 55–64 years old (P = 0·025). SFL were similar across the levels of adherence to the MD (P-value = 0·333). No association was observed between SFL and dietary total or haem iron intake. (Table 2).
Mean SFL in participants’ subgroups and mean change of SFL for one-unit increase of the continuous characteristics

MDS, Mediterranean diet score; SFL, serum ferritin levels; MET, metabolic equivalents of task.
* P-values are estimated by weighted linear regression.
† Age by sex interaction P-value: < 0·001.
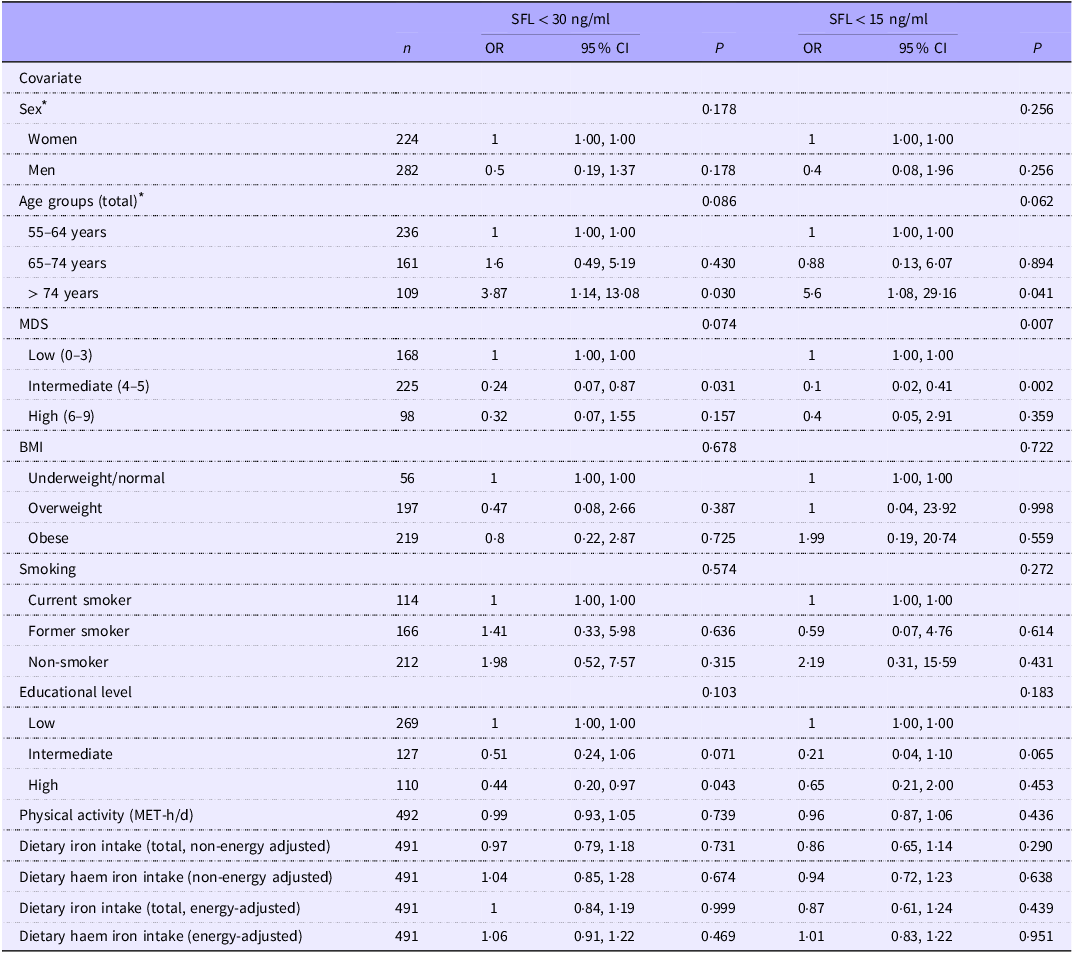
SFL < 30 ng/ml was observed in 12·7 % of the total population (9·2 % of men, 16·7 % of women), while SFL < 15 ng/ml was present in 6·5 % overall (4·0 % of men, 9·4 % of women). Two separate logistic regression models were fitted to identify significant predictors of ID using both thresholds (Table 3). Unlike the case of SFL, the age and sex interaction was not significant in either the logistic regression models (P-values: 0·272 and 0·213 for SFL < 30 ng/ml and SFL < 15 ng/ml, respectively).
Results from the weighted logistic regression models for SFL < 30 ng/ml and SFL < 15 ng/ml

SFL, serum ferritin levels; MDS, Mediterranean diet score; MET, metabolic equivalents of task.
* Age by sex interaction P-value: 0·272 and 0·213 for SFL < 30 ng/ml and SFL < 15 ng/ml, respectively.
The overall effects of sex and age on ID were not statistically significant for either threshold (SFL < 30 or < 15 ng/ml) in the total population. Nevertheless, individuals > 74 years old had 5·6 times and 3·9 times increased odds for SFL < 15 ng/ml (P = 0·041) and for SFL < 30 ng/ml (P = 0·030, respectively, compared with 55–64 years old individuals. No significant differences were observed for the 65–74 years age group.
Overall, adherence to MD was not found to be significantly associated with SFL < 30 ng/ml (P = 0·074), although intermediate adherence was associated with decreased odds for SFL < 30 ng/ml (OR = 0·24; 95 % CI 0·07, 0·87; P = 0·031) and a similar but non-significant association was observed for those with high adherence (OR = 0·32; 95 % CI 0·07, 1·55; P = 0·157). When ID was defined as SFL < 15 ng/ml, MD adherence was significantly associated with lower odds of ID (P = 0·007), mainly due to the effect of intermediate adherence (OR = 0·10; 95 % CI 0·02, 0·41; P = 0·002) but not high adherence. These associations remained significant in the presence of age and sex. Dietary indicators of iron intake were not found to have a significant effect on SFL or ID (Table 3).
Dietary iron intake was significantly lower in women (men: 12·4 mg/d v. women 8·6 mg/d, P < 0·001) and decreased significantly with increased age (P = 0·013 for 65–74 and P < 0·001 for > 75, both v. 55–64), especially in men. Thus, a significant sex by age interaction was observed. Specifically, the difference between men and women was greater in the 55–64 age group and was attenuated with advancing age (P < 0·001) (online Supplementary Table S1). Energy-adjusted (2000 kcal) total iron intake was significantly higher in the highest adherence category. In contrast, non-adjusted haem iron intake was lower in the highest MD adherence group, a trend that persisted in the energy-adjusted values, although the latter was not statistically significant (P = 0·057) (online Supplementary Table S2).
Discussion
To our knowledge, this is the first study in Greece that evaluates iron status and influencing factors in a large sample of participants of a nationally representative sample. Among the population, 6·5 % had ID, with a mean SFL of 117 ng/ml. SFL were significantly associated with sex and age, presenting lower levels in women, with individuals > 74 years old showing higher odds of ID at both thresholds, compared with those aged 55–64 years. 57·2 % of the population had dietary iron intake below the EFSA population reference intake of 11 mg/d. Dietary iron intake declined considerably with increased age and was significantly lower in women. Intermediate MD adherence was significantly associated with a reduced risk of ID at both thresholds.
Few studies have explored iron status and adequacy in specific subpopulations residing in Greece, mainly children(Reference Zikidou, Tsigalou and Trypsianis28,Reference Gompakis, Economou and Tsantali29) or army recruits(Reference Kattamis, Metaxotou-Mavromnati and Konidaris30) or other subpopulations where iron metabolism was estimated(Reference Eleftheriadis, Pissas and Remoundou31).
Overall, the mean SFL (117 ng/ml) is within normal levels for both sexes. The observed prevalence of ID indicates that ID is not extensive in the older Greek population. It is difficult to compare iron status among different countries due to differences in methodology, cutoff values, biomarkers used (i.e. SFL, serum iron levels, Hb, soluble transferrin receptor and total body iron) and age groups. Indicatively, in Slovenia, only 0·6 %(Reference Lavriša, Hristov and Hribar32) of the older population had SFL < 15 μg/l, and in Portugal, 17 %(Reference Fonseca, Marques and Robalo Nunes33) of the older population had SFL < 15 μg/l. In the older population of the USA, the prevalence of ID was 10 %(Reference Tawfik, Billingsley and Bhatt34), while in a study that included community-dwelling older adults from five European countries, it was 4·2 % based on SFL < 30 μg/l(Reference Stahl-Gugger, de Godoi Rezende Costa Molino and Wieczorek5). The mean value of SFL is in accordance with studies of similar age groups in various countries(Reference Lavriša, Hristov and Hribar32,Reference Bates, Lennox and Prentice35,Reference Cooper, Bertinato and Ennis36) .
SFL were significantly lower in women and declined with increasing age. Iron accumulation is thought to vary by age, sex and between pre- and postmenopausal women(Reference Merlo, Groothof and Khatami37). Studies have shown that hepcidin, a hormone that regulates iron metabolism in the body, is lower in women than in men and higher in postmenopausal women than in those of reproductive age, with the hypothesis that oestrogen levels alter hepcidin levels(Reference Merlo, Groothof and Khatami37,Reference Yang, Jian and Katz38) . Lower SFL in women compared with men in our study are in accordance with other studies in the literature(Reference Rushton and Barth39). The decreasing trend of SFL with advancing age was more evident in men. While older men present a drop in SFL, older women after menopause present a rise(Reference Merlo, Groothof and Khatami37). Menstrual cessation results in reduced iron losses, which raises iron stores(Reference Merlo, Groothof and Khatami37). Both sexes present a sharper decline in SFL after the age of 74 years old. This drop may be attributed to poor iron intake or lower absorption(Reference Fairweather-Tait, Wawer and Gillings40). For the age group > 74 years old, there were considerably increased odds for ID compared with the 55–64 age group.
In our study, we did not find a significant association of SFL with BMI. Conversely, several studies have shown a link between obesity and ID, which is prominent in all age groups(Reference Zhao, Zhang and Shen41). The most probable cause is obesity-related inflammation as studies indicate no differences in dietary iron intake between the obese and non-obese individuals(Reference Cepeda-Lopez, Osendarp and Melse-Boonstra42), but rather regarding iron absorption. Inflammation increases hepcidin, which blocks iron release from reticuloendothelial cells and decreases intestinal iron absorption(Reference Thurnham and McCabe43).
Infection and inflammation have an impact on several nutritional biomarkers, including iron. Inflammation results in the allocation of iron in favour of storage sites, including iron retention by macrophages and liver, resulting in decreased serum iron and increased SFL(Reference Feelders, Vreugdenhil and Eggermont44). WHO advises measuring another acute-phase reactive protein, such as CRP, along with SFL to examine the presence of inflammation(2). Following this argument, in our analysis, SFL were adjusted for inflammation using the BRINDA method. Furthermore, since almost half of our population was obese, the obesity-related inflammation underscores the need to adjust SFL for inflammation to prevent underestimation of ID.
57·2 % of the population presented inadequate daily iron intake based on the EFSA population reference intake (11 mg/d), with a mean dietary intake of 10·7 mg/d (men 12·3 mg/d; women 8·9 mg/d). Iron intakes were lower than in the Hellenic National Nutrition and Health Survey (HNNHS), which reported median iron intakes of 15 mg/d and 11·6 mg/d for men and 13·3 mg/d and 11·5 mg/d for women in the 51–70 and > 70-year age groups, and was conducted during the same period as HYDRIA(Reference Mitsopoulou, Magriplis and Michas45). Dietary iron intake ranges from 10·2 to 16 mg/d across other national representative studies(Reference Lavriša, Hristov and Hribar32,Reference Bates, Lennox and Prentice35,Reference Samaniego-Vaesken, Partearroyo and Olza46) . Dietary iron intake declined with increased age in both sexes, but men presented higher values. A higher proportion of men had adequate iron intake compared with women. In women, the mean dietary iron intake was below the population reference intake of 11 mg/d across all age groups, whereas only men over 74 years had intake below the reference value. According to EFSA, men in general present to some extent greater average daily intakes than women, primarily because they consume bigger amounts of food each day(24).
In this survey, the intake of haem iron accounted for 15·9 % of unadjusted total dietary iron. Previous findings from HYDRIA showed that men had higher consumption of products with haem iron (meat, poultry, etc.)(Reference Martimianaki, Peppa and Valanou17). Dietary haem iron was not significantly associated with SFL. This is in contrast with other published studies where haem iron is an independent factor for iron status(Reference Iglesias-Vázquez, Arija and Aranda47,Reference Fleming, Jacques and Dallal48) , although there are studies that are in accordance with ours(Reference Garry, Hunt and Baumgartner49,Reference Cooper, Greene-Finestone and Lowell50) .
Non-haem iron was not presented in our study as its absorption depends on individual iron stores and is strongly influenced by dietary enhancers and inhibitors, making reported intake an unreliable marker of actual absorption(Reference Hurrell and Egli12). In any case, non-haem iron intake (total iron minus haem iron) had a non-significant effect on BRINDA-adjusted SFL (β = 3·02; 95 % CI –0·08, 6·12; P = 0·086) and did not affect overall results, so it was not included as a primary finding.
Additionally, the MDS was evaluated in our cohort. Total iron intake, after being adjusted to a 2000 kcal diet, was significantly higher among individuals with the highest MD adherence. The unadjusted values demonstrated a similar pattern, but the difference was not statistically significant. There are a few studies that explore the relationship between adherence to MD and iron intake. Our results confirm previous studies that explored the same issue, namely, that there is a positive association regarding adherence to MD and total iron intake in the older population(Reference Jennings, Tang and Gillings51–Reference Serra-Majem, Bes-Rastrollo and Roman-Vinas53). Higher adherence to the MD has also been associated with lower iron inadequacy in children and adolescents(Reference Castro-Quezada, Roman-Vinas and Serra-Majem11). Adherence to MD did not significantly affect mean SFL. Although the intake of highly bioavailable haem iron decreased with increasing adherence to the MD, iron status did not differ significantly. This finding implies that the MD offers enough bioavailable iron despite lower haem iron. Its combination of plant-based iron sources and dietary enhancers (such as vitamin C) possibly improves non-haem iron absorption(Reference Jennings, Tang and Gillings51,Reference Mesias, Seiquer and Munoz-Hoyos54) . In a study with male adolescents, iron absorption was enhanced when a Mediterranean-based diet was consumed(Reference Mesias, Seiquer and Munoz-Hoyos54). Conversely, a study that included older men from Crete, where MD used to prevail, has demonstrated lower SFL compared with men from Zutphen in the Netherlands(Reference Buijsse, Feskens and Moschandreas55).
It is noteworthy that in our study, adherence to MD was associated with decreased odds for ID, considering SFL < 15 ng/ml. The intermediate category of MD adherence had a statistically significant protective impact against low SFL with both cutoffs. Since the effect of iron intake was not significant for ID, further studies are needed to explore this tendency. The associations observed may partly reflect heterogeneity in the dietary compounds contributing to the MDS. Individuals with similar MDS values may achieve comparable scores through different combinations of foods. For instance, some may consume higher amounts of fish and fruits, while others may emphasise cereals and legumes. Such variability may differentially affect iron intake and bioavailability, given the varying contributions of non-haem iron sources and dietary enhancers or inhibitors of iron absorption(Reference Hurrell and Egli12). Additionally, in the HYDRIA study, higher MD adherence was more prevalent among older participants(Reference Martimianaki, Peppa and Valanou17), who may experience reduced intestinal iron absorption, potentially attenuating the expected benefits of higher adherence levels.
This study presents several strengths and limitations. Its major strength is that the sample is drawn from a national representative study, allowing the findings to be generalised to the population of older persons who do not use iron medication or supplements. There is an abundance of carefully selected and recorded characteristics, such as sociodemographic and lifestyle factors, and blood samples were collected from the entire population > 55 years old in our study. Moreover, dietary intake was based on 2 × 24-HDR, which is considered the appropriate and more accurate approach according to the recommendations of the European Health Examination Survey and EFSA. Additionally, to account for the potential impact of chronic low-grade inflammation on SFL and correctly estimate its levels, particularly in older adults and the obese population, the BRINDA method was applied. Nevertheless, limitations should be noted. Although SFL, according to WHO, is a reliable indicator of iron stores and can be utilised to detect ID, additional indicators of iron status, such as soluble transferrin receptor and transferrin saturation, were not available. Furthermore, the contribution of individual food groups to total iron intake was not assessed, nor were dietary factors that could affect iron absorption, including potential enhancers (i.e. vitamin C) and inhibitors (i.e. phytates and polyphenols). Also, since it is a cross-sectional study, causal associations cannot be established.
Conclusion
In conclusion, the number of older adults who are iron deficient in a large Greek sample is low. There was a clear trend for increased risk of ID with age. There was no association between iron status and dietary iron intake, nor intake of haem iron. Adherence to the MD was associated with lower odds for ID but had no effect on SFL. Given that ID is a public health concern, further research is needed to determine the components of dietary iron consumption and its relation to iron biomarkers to quantify both the enhancers and inhibitors of iron absorption and to identify the most suitable biomarker to reflect the iron status of specific demographic groups, such as the older population.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114526106540.
Acknowledgements
The National Health and Nutrition Survey – HYDRIA – was co-financed by the European Social Fund of the European Union, national resources and the Hellenic Health Foundation. The funders had no role in the design, analysis or writing of this article.
E. M. P., G. V., E. P., I. P., A. M. P. and A. T.: conceptualisation, visualisation and methodology; E. M. P. and EP: investigation; E. M. P., E. P. and A. T.: project administration; G. V. and I. P.: data curation and formal analysis; A. T.: supervision; E. M. P., G. V., E. P., I. P. and A. M. P.: writing the original draft; E. M. P., G. V., E. P., I. P., A. M. P. and A. T.: writing – review and editing. All authors read and approved the final manuscript.
The authors declare no conflict of interest.