Introduction
The tetraclitid genus Tesseropora Pilsbry, 1916 (Cirripedia, Thoracica, Balanomorpha) currently comprises seven valid extant species (WoRMS [World Register of Marine Species] 2024a). These species inhabit the Indo-Pacific region and the shores of various Atlantic islands (Newman and Ross Reference Newman and Ross1977; Southward Reference Southward1998; Wirtz et al. Reference Wirtz, Araújo and Southward2006; Young Reference Young1998). Fossil representatives of the genus have been described from multiple regions, including the Mediterranean-Tethyan realm (Oligocene–Miocene), the eastern Atlantic (Late Pliocene), and the eastern Pacific (Pliocene) (Carriol Reference Carriol2005, Reference Carriol2008; Zullo Reference Zullo1968). Among these is Tesseropora canariana (Hornung Reference Hornung, Wiese, Reich and Arp2014), described from Lower Pliocene shoreline deposits in western Fuerteventura (Canary Islands) (Hornung Reference Hornung, Wiese, Reich and Arp2014).
Newman and Ross (Reference Newman and Ross1977) identified Tesseropora atlantica (Newman and Ross Reference Newman and Ross1977) as a living relict species with origins tracing back to the Oligocene. Type specimens were collected from Bermuda (northwestern Atlantic, approximately 32º15’N) and the Azores Islands (north-central Atlantic, approximately 38º39’N). Earlier records of this species include Bermuda (Verrill Reference Verrill1901, as Tetraclita porosa; Henry Reference Henry1958, as Tetraclita squamosa stalactifera) and the Azores (Baker Reference Baker and Anon1967, as Tetraclita squamosa var. elegans).
Subsequently, T. atlantica was recorded at St. Paul’s Rocks (approximately 0º55’N) in the equatorial central Atlantic (Edwards and Lubbock Reference Edwards and Lubbock1983). Young (Reference Young1998) described Tesseropora arnoldi as a new endemic species to the Azores; however, Southward (Reference Southward1998), after examining fresh specimens from both the Azores and Bermuda, concluded that the previously reported morphological differences were not supported. His analysis confirmed the presence of T. atlantica in both archipelagos.
Wirtz et al. (Reference Wirtz, Araújo and Southward2006) provided a comprehensive list of cirripede species from Madeira Island and adjacent deep waters (northeastern Atlantic), based on specimens housed at the Funchal Natural History Museum (MMF), references cited in the literature, and new field collections. Their study reported the first record of T. atlantica in Madeira, with specimens collected from both the northern (approximately 32º49’30”N) and southern (approximately 32º39’N) coasts of the island. Given that A. J. Southward was a co-author, the morphological identification of T. atlantica from Madeira was considered reliable (P. Wirtz, pers. comm.).
The first account of thoracian barnacles from the Canary Islands, including notes on distribution and ecology, was provided by González et al. (Reference González, Martín, Herrera, González-Lorenzo, Espino, Barquín-Diez and Southward2012). That study documented intertidal, shallow-water, and oceanic species collected from various animals and floating substrates, from the surface down to approximately 150 m depth. Later, González et al. (Reference González, Shalaeva, Martín-García, Lorenzo and Southward2017) expanded on this work with a report on the deep-sea stalked barnacles in the Canary Islands, updating the regional checklist of thoracian cirripeds and their hosts. To date, T. atlantica has not been reported in either of these studies or any other published source from the Canary Islands.
In the present study, we collected fresh specimens of Tesseropora from the Canarian Islands of Lanzarote and Fuerteventura, and from Madeira for comparative purposes. Morphological examination and comparison of the samples confirmed the presence of T. atlantica in the Canary Islands.
Materials and methods
Fieldwork in the Canary Islands was carried out between July 2013 and June 2023 along the coastlines of Lanzarote and Fuerteventura, the easternmost islands in the archipelago. Sampling in Madeira Island was conducted in October 2022. In both archipelagos, specimens were collected from the intertidal zone on rocky shores and tidal pools, specifically beneath medium-sized stones. Tesseropora specimens were photographed in situ and manually collected using a shellfish scraper. Following collection, individuals were photographed fresh and preserved in 80% ethanol for subsequent morphological identification and DNA barcoding. Voucher specimens were labelled, curated, catalogued, and deposited in the ICCM study collection at the University of Las Palmas de Gran Canaria and in the collections of the Funchal Natural History Museum (MMF).
Specimens were preliminarily identified as Tesseropora atlantica based on morphological characters, with reference to Newman and Ross (Reference Newman and Ross1977) and Costa and Jones (Reference Costa and Jones2000). The rostro-carinal diameter (rc) was measured using callipers with a precision of ± 0.1 mm.
Material examined. – Tesseropora atlantica. Canary Islands. Voucher code: ICCM527. Thirty individuals, ranging from 6.5 to 9.5 mm rc. Collection locality: Arrecife – Playa Honda, eastern coast of Lanzarote, intertidal zone, collected in Jul. 2013, Apr. 2015, Mar. 2018, Feb. 2019, and Mar. 2021 by R. Ramírez (third author). Voucher code: ICCM528. Ten individuals, ranging from 6.9 to 9.2 mm rc. Collection locality: Puerto Lajas, northeastern coast of Fuerteventura, intertidal zone, collected in Sep. 2020 by R. Ramírez (third author). Two of these individuals were used for genetic analyses (ICCM528-1 and ICCM528-2). Specimens co-occurred with Chthamalus stellatus (Poli) and C. montagui Southward on both islands. Madeira Island. Voucher code: MMF50527. Five individuals, ranging from 2.1 to 4.4 mm rc. Collection locality: Caniço de Baixo, Reis Magos, southern coast, intertidal zone, collected on 12 Oct. 2022 by R. Araújo (fourth author). Specimens co-occurred with Balanus trigonus Darwin and Chthalamus stellatus. Two individuals were used for genetic analyses (MF50527-1 and MF50530).
DNA sequences were obtained from two specimens of T. atlantica from the Canary Islands and two specimens from Madeira. Total genomic DNA was extracted from internal tissue using a modified 10% Chelex protocol following Estoup et al. (Reference Estoup, Largiadèr, Perrot and Chourrout1996).
Target mitochondrial genes (16S rRNA and COI) were amplified via polymerase chain reaction (PCR) using the following thermal cycling conditions: initial denaturation at 95°C for 2 min; 40 cycles of 95°C for 20 s, annealing at 45°C (COI) or 48°C (16S) for 20 s, extension at 72°C for 47 s (COI) or 45 s (16S); and a final extension at 72°C for 5 min. For the 16S gene, primers 1472 (5′-AGA TAG AAA CCA ACC TGG-3′) (Crandall and Fitzpatrick Reference Crandall and Fitzpatrick1996) and 16L2 (5′-TGC CTG TTT ATC AAA AAC AT-3′) (Schubart et al. Reference Schubart, Cuesta and Felder2002) were used to amplify a 538-bp fragment. For the COI gene, primers COH6 (5′-TAD ACT TCD GGR TGD CCA AAR AAY CA-3′) and COL6b (5′-ACA AAT CAT AAA GAT ATY GG-3′) (Schubart and Huber Reference Schubart and Huber2006) were used to amplify a 663 bp fragment.
PCR products were purified and bidirectionally sequenced by Stab-Vida Laboratories. Sequence chromatograms were edited using Chromas version 2.0. Final sequences were compared with existing records using BLAST searches on the NCBI website. All sequences were deposited in GenBank under accession numbers PQ594194–PQ594196 (16S) and PQ5935–PQ593517 (COI).
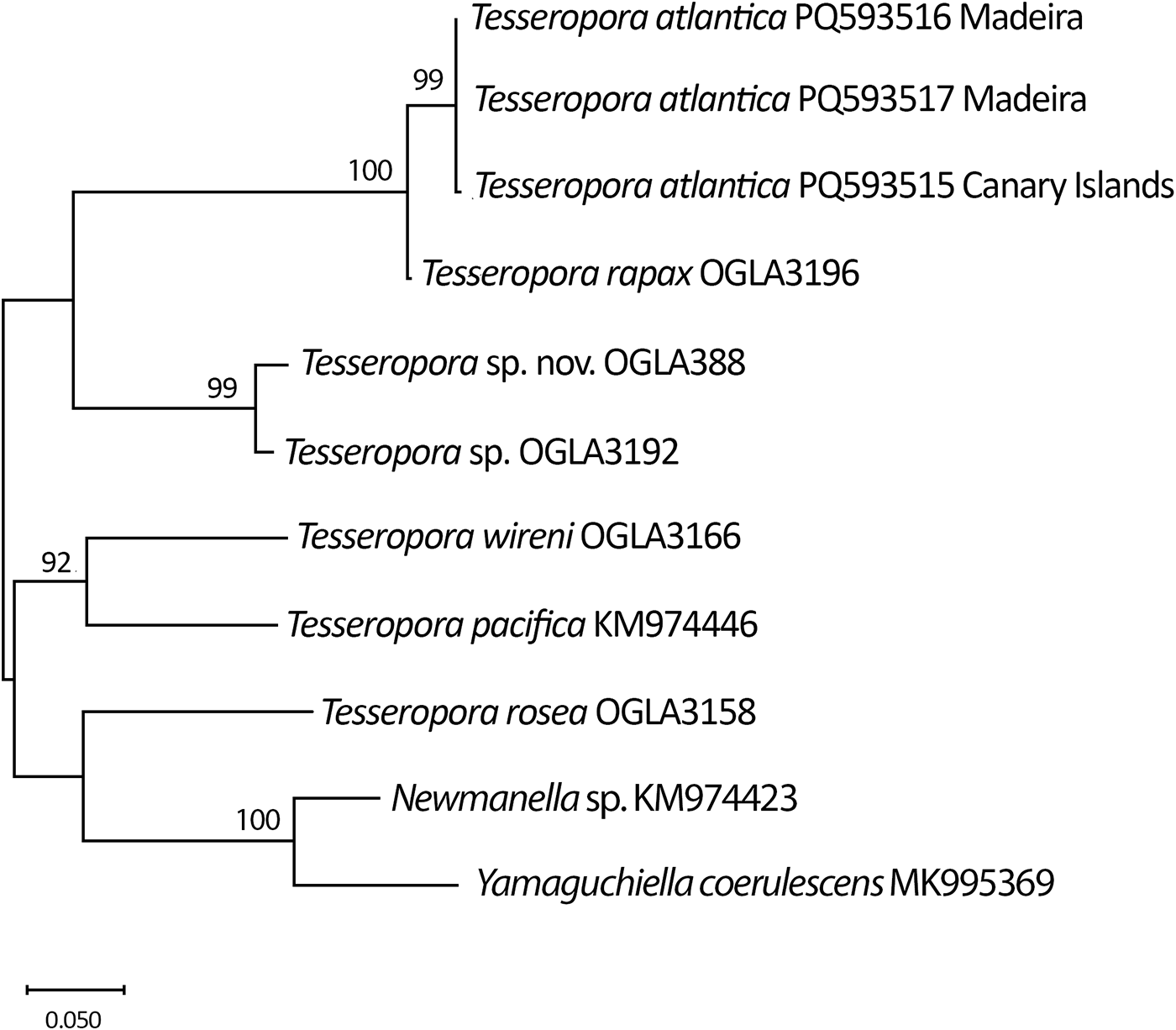
Alignments of 16S and COI sequences included both newly generated sequences and congeneric sequences downloaded from GenBank (http://www.ncbi.nlm.nih.gov). The best-fitting nucleotide substitution models for 16S and COI were selected using MEGA X (Kumar et al. Reference Kumar, Stecher, Li, Knyaz and Tamura2018), based on the corrected Akaike information criterion (AICc). Phylogenetic analysis for 16S was performed using the Neighbour-Joining (NJ) method with the p-distance model, and nodal support was assessed via 2000 bootstrap replicates (Figure 1). The COI dataset was analysed using Maximum Likelihood (ML) in MEGA X, with topological robustness assessed by 2000 non-parametric bootstrap replicates (Figure 2).
Neighbour-joining (NJ) phylogenetic tree based on mitochondrial 16S rRNA sequences of Tesseropora species, constructed using 2000 bootstrap replicates. Bootstrap support values > 60 are shown at nodes. Yamaguchiella coerulescens (Spengler) and Newmanella sp. were used as outgroups. GenBank accession codes are indicated for each sequence.

Maximum likelihood (ML) phylogenetic tree based on mitochondrial cytochrome c oxidase subunit I (COI) sequences of Tesseropora species, constructed using 2000 bootstrap replicates. Bootstrap support values > 60 are shown at nodes. Yamaguchiella coerulescens and Newmanella sp. were used as outgroups. GenBank and BOLD accession codes are indicated for each sequence.

Results
Morphological comparisons of the collected specimens confirmed the presence of Tesseropora atlantica in the easternmost Canary Islands, specifically Lanzarote and Fuerteventura (Figure 3A, B). This represents the first documented occurrence of the species in the Canary Islands and establishes a new southern range limit for T. atlantica in the eastern Atlantic Ocean.
The tetraclitid barnacle Tesseropora atlantica (Newman and Ross Reference Newman and Ross1977) from the Canary Islands. (A) Live individuals in their natural habitat. (B) Dorsal view of an ethanol-preserved specimen (ICCM527).

Morphological identification
The assignment of the examined barnacles to the genus Tesseropora was based on several diagnostic features: the presence of intraparietal septa connecting to the inner lamina at approximately right angles, the development of only 2–3 rows of parietal tubes in large (adult) specimens, and the presence of a calcareous basal plate (e.g., Hornung Reference Hornung, Wiese, Reich and Arp2014). These characteristics distinguish Tesseropora from the morphologically similar genus Tetraclita Schumacher, 1817, as noted by Newman and Ross (Reference Newman and Ross1977).
Specimens from Lanzarote and Fuerteventura (eastern Canary Islands), as well as those from Madeira Island, closely matched the original species description by Newman and Ross (Reference Newman and Ross1977). Tesseropora atlantica is a steeply conical barnacle with a basal diameter of up to 10 mm. It displays a uniformly intense white colouration across the wall, sheath, filling, and opercular valves. The radii and alae are moderately developed, with summits running parallel to the base. Irregular parietal tubes pores are arranged in a single row. The scutal adductor ridge is aligned with the articular ridge, and the crest of the labrum lacks conspicuous teeth (Costa and Jones Reference Costa and Jones2000; Newman and Ross Reference Newman and Ross1977).
Distinguishing features between T. arnoldi and T. atlantica include the development and pattern of ribbing from the base to the sheath – T. arnoldi exhibits prominent ribs interspersed with numerous fine ribs, whereas T. atlantica displays only small, fine ribs. Additional differences involve the relative position of the scutal adductor ridge to the articular ridge and the presence of conspicuous teeth on the labral crest in T. arnoldi (Costa and Jones Reference Costa and Jones2000; Young Reference Young1998).
The most prominent morphological difference between T. atlantica and other non-Atlantic congeners lie in the alignment of the scutal adductor ridge with the articular ridge. In T. atlantica, the adductor ridge aligns almost continuously with the articular ridge, while in other species it typically overlaps the ridge (Costa and Jones Reference Costa and Jones2000). In T. arnoldi, the adductor ridge is distinctly separated from the articular ridge (Costa and Jones Reference Costa and Jones2000; Young Reference Young1998).
All diagnostic features described above were carefully examined and verified by the authors.
DNA barcoding
The 16S alignment consisted of 538 base pairs (bp), with Tamura 3-parameter with invariant sites and gamma-distributed rates for the variables sites (T92 + G + I) selected as best-fitting nucleotide substitution model. For COI, the final alignment comprised 663 bp, and the best-fitting model was the General Time Reversible model, also with invariant sites and gamma-distributed rates for the variable sites (GTR + G + I).
Unfortunately, publicly available genetic data for Tesseropora species remain limited. For 16S, sequences are available only for T. pacifica (Pilsbry) and T. rosea (Krauss). In the case of COI, sequences are known for these two species, as well as for T. rapax Jones, T. wireni (Nilsson-Cantell), an indeterminate Tesseropora sp., and an undescribed species listed as Tesseropora n. sp. – based on unpublished records deposited in BOLD by Dr Andrew Hosie. No genetic data are currently available for T. alba Ren & Liu or T. arnoldi Young.
Phylogenetic analyses based on 16S and COI sequences (Figures 1 and 2) indicate that T. atlantica clusters within the Tesseropora clade and, based on COI data, appears to be most closely related to T. rapax. The T. atlantica specimens collected from the Canary Islands and Madeira showed identical sequences for 16S and differed by only a single nucleotide in the COI gene.
Discussion
Tesseropora atlantica is an amphi-Atlantic species with a preference for warmer waters, and it has not been recorded from the continental coasts of the eastern Atlantic (Costa and Jones Reference Costa and Jones2000; Wirtz et al. Reference Wirtz, Araújo and Southward2006; WoRMS [World Register of Marine Species] 2024b). This species exhibits suppressed larval development, retaining larvae within the mantle cavity until they reach the cyprid stage. Consequently, its larval dispersal capabilities are likely limited (Newman and Ross Reference Newman and Ross1977; Southward Reference Southward1998; Young Reference Young1998). Southward (Reference Southward1998) reported that T. atlantica cyprids began exploratory behaviour within 24 h of release and continued for an additional 24 h, although no settlement or metamorphosis was observed. Despite this short dispersal window, the species has a broader distribution than expected (Costa and Jones Reference Costa and Jones2000). The absence of a prolonged dispersal phase in insular barnacle species is often considered an adaptation to maintain isolated island populations by reducing propagule loss (Costa and Jones Reference Costa and Jones2000; Newman and Ross Reference Newman and Ross1977).
Giving its presence in the Azores and Madeira, the occurrence of T. atlantica in the Canary Islands may reflect a natural range extension, as might be expected for a species with some planktonic dispersal. This scenario would be consistent with larval transport via the Canary Current, which flows predominantly from north to south and has been implicated in the dispersal of planktonic and rafting organisms across the eastern-central Atlantic (e.g., Barton et al. Reference Barton, Arístegui, Tett, Cantón, García-Braun, Hernández-León, Nykjaer, Almeida, Almunia, Ballesteros, Basterretxea, Escánez, García-Weill, Hernández-Guerra, López-Laatzen, Molina, Montero, Navarro-Pérez, Rodríguez, van Lenning, Vélez and Wild1998).
However, in light of the species’ limited larval dispersal potential, alternative mechanisms must be considered. In sessile organisms such as barnacles, long-distance dispersal often occurs through rafting on natural or anthropogenic floating substrates, or via transport on ship hulls and offshore structures such as oil platforms (González et al. Reference González, Martín, Herrera, González-Lorenzo, Espino, Barquín-Diez and Southward2012; Southward Reference Southward, Crothers and Hayward2008). Triay-Portella et al. (Reference Triay-Portella, Pajuelo, Manent, Espino, Ruiz-Díaz, Lorenzo and González2015) documented the arrival of alien marine species to the Canary Islands associated with oil platforms towed from the Americas between 2010 and at least 2015. Notably, according to one of the anonymous reviewers, T. atlantica has been recorded on an oil platform towed from the Gulf of Mexico in route to Ireland, suggesting that such structures may serve as effective vectors. The presence of T. atlantica in the Canary Islands, therefore, could plausibly result from a recent anthropogenic introduction.
Given the species’ distinctive morphology and white colouration, its colonization of the northeastern Canary Islands (Lanzarote and Fuerteventura) likely occurred within the past few decades. Although T. atlantica tends to inhabit shaded microhabitats beneath rocks – making it easy to overlook – these areas are frequently explored by divers. The relatively recent discovery of this species in Madeira (Wirtz et al. Reference Wirtz, Araújo and Southward2006) further supports the hypothesis of recent range expansion. Continued surveys are recommended to assess the extent of its colonization in the Canary Islands and to evaluate potential ecological interactions with native intertidal species.
Acknowledgements
We are sincerely grateful Chris Hauton and two anonymous reviewers for their insightful comments and constructive suggestions, which have significantly improved the quality of this manuscript.
Author contributions
J.A.G. conceived and designed the study. R.R., M.B., and R.A. conducted the field samples. J.A.G. analysed the morphological data. J.A.C made the molecular laboratory work and analysed the sequences and prepared the tree figures. R.R. made the photograph of the specimens. M.B. managed the specimens of the collections. J.A.G. and J.A.C. made the first draft of the manuscript. All authors read and approved the final version of the manuscript.
Funding
This work received no specific financial support from any funding agency or institution.
Competing interests
None.
Ethical standards
This study does not contain sampling procedure and technique involving vertebrates and regulated invertebrates performed by any of the authors.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article itself. The DNA sequences generated in the present study are deposited in the NCBI/GenBank database (accession nos. PQ594194–PQ594196 and PQ5935–PQ593517).