Metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD), is the most common cause of liver disease worldwide(Reference Byrne and Targher1). Its prevalence continues to rise, with recent global estimates exceeding 30 %(Reference Powell, Wong and Rinella2,Reference Younossi, Golabi and Paik3) . MASLD encompasses a spectrum ranging from bland steatosis to metabolic dysfunction-associated steatohepatitis, which risks progression to cirrhosis and liver failure(Reference Maurice and Manousou4). MASLD is further associated with an increased risk of major adverse cardiovascular events, including myocardial infarction, stroke and heart failure(Reference Chung, Yu and Yoo5,Reference Raverdy, Tavaglione and Chatelain6) .
The pathophysiology of MASLD encompasses many metabolic pathogenic pathways, ranging from circulating lipids, adipose-related compounds, pancreatic and gut hormones to muscle and liver-related compounds(Reference Petta, Gastaldelli and Rebelos7). Genetic, hormonal and dietary factors further modulate disease expression and severity(Reference Carr, Oranu and Khungar8). The generation of reactive oxygen species are also key contributors to the inflammatory and fibrotic progression of MASLD(Reference Delli Bovi, Marciano and Mandato9).
Flavonoid-rich diets have emerged as a promising area of interest due to their anti-inflammatory and antioxidant properties(Reference Wang, Tan and Liu10). In the USA, tea is the predominant dietary source of flavonoids, accounting for approximately 75 % of total intake and over 90 % of flavan-3-ol intake. Other major sources include wine, berries and grapes for anthocyanidins, citrus fruits and juices for flavanones, vegetables for flavonols and flavones, and soy products for isoflavones (online Supplementary Table 1)(Reference Chun, Chung and Song11,Reference Sebastian, Goldman and Moshfegh12) . Because flavonoid-rich foods, particularly fruits and vegetables, are core components of healthy dietary patterns, flavonoid intake is correlated with overall diet quality. This collinearity raises the question of whether observed flavonoid-MASLD associations reflect independent effects or are confounded by the broader dietary context. Currently, the epidemiological literature on the association between flavonoid-rich diets and MASLD remains inconclusive.
A meta-analysis on twelve randomised controlled trials found that flavonoid supplementation demonstrated favourable effects on liver biomarkers and a likely reduction in NAFLD(Reference Li, Ji and Du13). These trials typically supplemented one flavonoid subclass for 8–12 weeks in small groups of approximately eighteen to fifty-five individuals. Moreover, these trials often lacked the dietary context necessary to elucidate whether flavonoid effects are independent of overall diet quality. These limitations underscore the continued need for population-based analyses. Currently, cross-sectional data from the 2017–2018 National Health and Nutrition Examination Survey (NHANES) cohort showed inverse associations between flavonoid subclasses of anthocyanins and isoflavones and vibration-controlled transient elastography-derived NAFLD(Reference Tong, Zeng and Xie14). Similarly, a prospective cohort study in the UK Biobank found that a greater flavonoid intake was associated with reduced risk of incident NAFLD(Reference Bell, Jennings and Thompson15).
Clinically, lifestyle modifications, including effective dietary regimen, are the cornerstone of MASLD management(Reference Chalasani, Younossi and Lavine16). Evidence from randomised controlled trials supports the benefits of Mediterranean diet interventions for reducing hepatic steatosis, while meta-analyses of five trials suggest the Dietary Approaches to Stop Hypertension (DASH) diet may be effective in MASLD management(Reference Hasan, Singh and Garcia17–Reference Yaskolka Meir, Rinott and Tsaban19). Similarly, observational studies have reported that greater DASH and Mediterranean-style diet score (MDS) scores were associated with a reduced risk of MASLD and subsequent hepatic fibrosis(Reference Petermann-Rocha, Carrasco-Marin and Boonpor20,Reference Romero-Gómez, Zelber-Sagi and Trenell21) . Prior work using both the NHANES and the Framingham Heart Study (FHS) has demonstrated that higher adherence to the Alternative Healthy Eating Index (AHEI), DASH and MDS is associated with lower obesity and reduced hepatic steatosis and may further modulate progression from steatosis to fibrosis(Reference Gao, Long and Singh22). Although these three indices differ in how individual components are weighted, they share substantial overlap (online Supplementary Table 2) in emphasising fruits, vegetables and whole grains that are all key dietary sources of flavonoids, suggesting that following any of these patterns is likely to yield broadly similar flavonoid intake profiles. In addition, these dietary scores reflect, to some extent, the synergistic effects of different food components. Thus, while flavonoid intake has been studied in relation to incident MASLD, the potential impacts of overall diet quality on the observed protective association of flavonoid consumption remain unclear. To bridge this knowledge gap, we evaluate how adherence to three a priori diet quality scores – AHEI, DASH and MDS – modifies the associations between total and subclass flavonoid intake and liver steatosis.
Methods
Study participants
In this cross-sectional study, study participants were taken from the FHS, a multigenerational prospective cohort study initiated in 1948 to study the incidence of CHD as well as to identify risk factors. We analysed data from participants in the FHS Offspring cohort and Third Generation (Gen3) cohort(Reference Tsao and Vasan23). The offspring cohort began in 1971 by enrolling 5124 adults, and the Gen3 cohort was initiated in 2002 with enrolment of 4095 adults. After excluding individuals with missing dietary data, abdominal computed tomography (CT) imaging or heavy alcohol consumption (> 3 drinks/d for men and > 2 drinks/d for women), the final analytic sample included 2815 participants (online Supplementary Figure 1). All participants provided written informed consent. Study protocols for FHS were approved by the Boston University Medical Center Institutional Review Board, and the present analyses were approved by the Tufts University Institutional Review Board.
Multidetector CT for measurement of hepatic steatosis
Hepatic steatosis was assessed using multidetector CT with 8-slice technology (LightSpeed Ultra, General Electric). A calibration phantom (Image Analysis) containing a water-equivalent compound and calcium hydroxyapatite (0, 75 and 150 mg/cm3) was positioned beneath each participant. Three regions of interest from the liver and one from the phantom were measured; the mean liver attenuation was then used to calculate a liver:phantom ratio (LPR). This CT-based method of quantifying hepatic steatosis has been shown to correlate strongly with other hepatic steatosis measures(Reference Speliotes, Massaro and Hoffmann24). Aligning with prior FHS studies(Reference Speliotes, Massaro and Hoffmann25,Reference Weinstein, Davis-Plourde and Himali26) , we define hepatic steatosis as LPR < 0·33.
Dietary assessment
Dietary intake over the year preceding the examination was assessed using a previously validated, self-administered, semi-quantitative FFQ, the Harvard 2007 grid FFQ(Reference Rimm, Giovannucci and Stampfer27). Using this FFQ, we calculated three a priori dietary quality scores: the DASH score, AHEI and MDS(Reference Kim, Lu and Ho28–Reference Chiuve, Fung and Rimm31). For all three scores, higher values indicate greater adherence to a healthy dietary pattern and reflect higher overall diet quality. Components of the three scores are described in online Supplementary Table 1.
Flavonoid exposures
We used the USDA flavonoid content of foods and the proanthocyanidin databases to derive flavonoid intake information(Reference Bhagwat and Haytowitz32). Intakes of individual flavonoid compounds were calculated by multiplying the consumption frequency of each food by the flavonoid content for a standard portion size and summing across all foods. We derived intakes for six major flavonoid subclasses commonly consumed in the USA diet: flavanones (eriodictyol, hesperetin and naringenin), anthocyanins (cyanidin, delphinidin, malvidin, pelargonidin, petunidin and peonidin), flavan-3-ols (catechins and epicatechins), flavonoid polymers (proanthocyanidins, theaflavins and thearubigins), flavonols (quercetin, kaempferol, myricetin and isorhamnetin) and flavones (luteolin and apigenin). This classification and methodology was described previously(Reference Shishtar, Rogers and Blumberg33,Reference Cassidy, O’Reilly and Kay34) . Total flavonoid intake was calculated as the sum of these six subclasses.
Covariate assessment
Sociodemographic and anthropometric information was obtained using standard FHS questionnaires(Reference Splansky, Corey and Yang35,Reference Kannel, Belanger and D’Agostino36) . Age was a continuous variable, and sex was a binary variable classified as self-reported biological sex assigned at birth. BMI (continuous, kg/m2) was calculated as measured weight in kilograms divided by the square of measured height in metres. The physical activity index (continuous) was calculated from questionnaire-derived duration and intensity of habitual activities, following established FHS protocols with demonstrated reliability and validity(Reference Kannel, Belanger and D’Agostino36–Reference He, Ogden and Bazzano38). Alcohol intake (in continuous scale) was expressed in servings per week, with one USA standard drink defined as 14 g of ethanol(Reference Ma, Huang and Yan39). Smoking status followed established FHS definitions: participants were classified as current smokers if they reported smoking 1 or more cigarette per day within the past year, and as non-current smokers if they had quit smoking (with cessation age recorded) or reported no prior regular cigarette use(Reference Freund, Belanger and D’Agostino40,Reference Samet, Thun and Berrington de Gonzalez41) . Education was included as a four-category variable: no high school degree, high school graduate, some college or college graduate or above.
Statistical analysis
Differences in characteristics across quartile categories of flavonoid intake were assessed using one-way ANOVA for continuous variables and chi-square tests with continuity correction for categorical variables. Logistic regression analyses were performed to examine the association between hepatic steatosis, defined by LPR < 0·33, and quartiles of total and subclass-specific flavonoid intake. Models were adjusted for sex, age, alcohol consumption, total energy intake, smoking status, physical activity and education. To assess whether diet quality confounds the association between flavonoid intake and MASLD, additional models included further adjustment for one of three dietary quality indices: AHEI, DASH or MDS. Additionally, to assess for a linear trend across flavonoid intake quartiles, logistic regression models were constructed using the median value of each flavonoid and total flavonoid quartile as continuous predictors against LPR-defined hepatic steatosis.
To assess the extent to which flavonoids contribute to the protective association between overall diet quality and hepatic steatosis, we performed an exploratory attenuation analysis. Separate logistic regression models were first fit for each dietary index (AHEI, MDS and DASH), adjusted for the same covariates as described above. Total flavonoid intake and individual flavonoid subclasses were then added to each model, and the proportional reduction in the dietary index β-coefficient was calculated.
To evaluate potential effect modification by sex, we conducted sex-stratified subgroup analyses for all flavonoid subclasses and total flavonoids. Interaction was formally tested by including a multiplicative sex by flavonoid quartile term in the base models. We used the likelihood ratio test to derive P for the interaction term. Sex-stratified models were adjusted for the same covariates as the primary analysis.
All analyses were performed using R version 4.5.0 with a two-sided 0·05 alpha used to declare significance.
Results
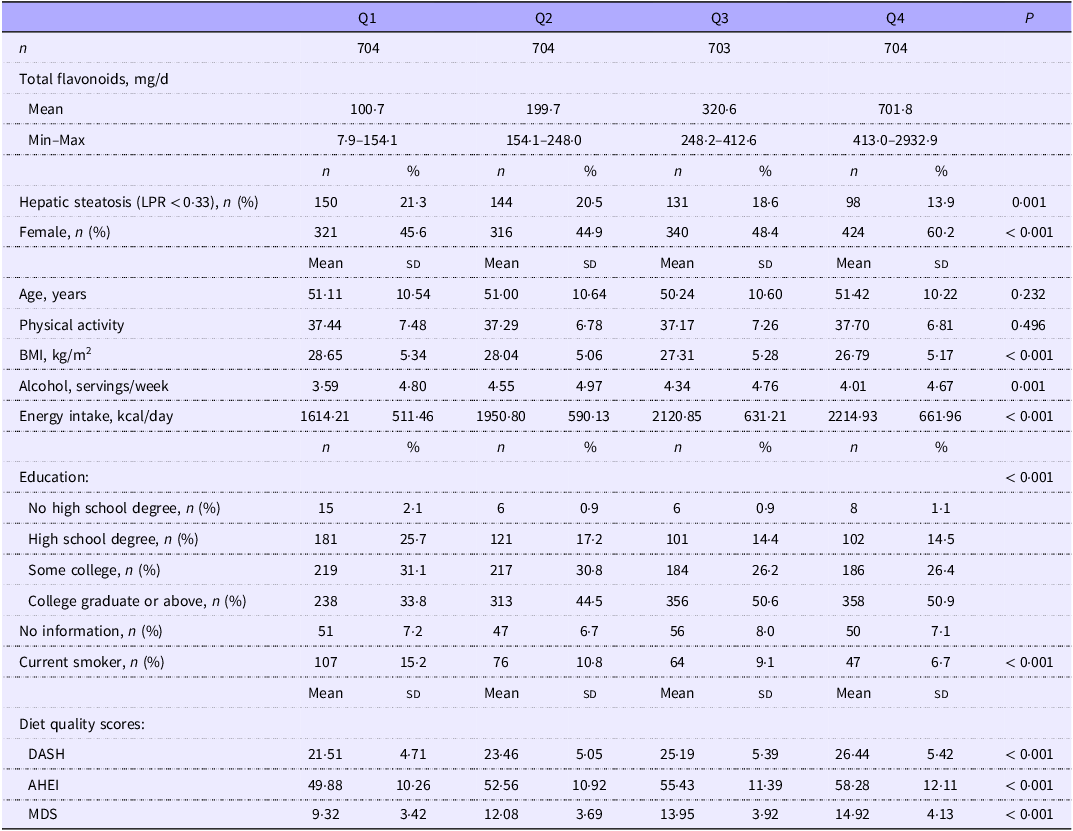
Baseline characteristics for participants stratified by total flavonoid intake quartiles are shown in Table 1. Statistically significant differences were observed across quartiles for age, BMI, total energy intake, sex, education and smoking status. Physical activity did not differ significantly by flavonoid intake. Women were more likely to be in higher flavonoid intake quartiles than men. Participants in the lowest flavonoid quartile had the highest mean BMI, while those in the highest quartile reported the lowest mean alcohol consumption (drinks/week). Higher flavonoid intake was also associated with greater educational attainment. Furthermore, all three dietary quality scores (DASH, MDS and AHEI) significantly trended upwards across flavonoid quartiles (P < 0·001).
Characteristics of FHS participants according to quartile categories of total flavonoids

Values are expressed as mean (sd) or proportion (%).
FHS, Framingham Heart Study; LPR, liver:phantom ratio; DASH, Dietary Approaches to Stop Hypertension; AHEI, Alternative Healthy Eating Index; MDS, Mediterranean-style diet.
Multivariate-adjusted correlations between total flavonoid and subclasses and hepatic steatosis
After adjusting for covariates but not dietary scores, higher intake of total flavonoids and several subclasses, specifically flavonols, flavan-3-ols, anthocyanidins, and flavonoid polymers, was significantly associated with lower odds of hepatic steatosis, with clear dose–response trends (P trend < 0·05; Table 2). No significant associations were observed for flavones or flavanones. Overall, the strongest inverse associations were consistently observed in the highest intake quartiles across the protective subclasses.
Adjusted OR (95 % CI) for hepatic steatosis by quartile of flavonoid intake

All models are adjusted for age, sex, alcohol intake, smoking status, energy intake, physical activity and education. P trend models assess linear trends using the median value of each quartile as a continuous predictor.
Association between flavonoid intake and hepatic steatosis is attenuated by adjustment for overall diet quality
After additional adjustment for overall diet quality using the DASH, AHEI and MDS scores, the inverse associations between higher flavonoid intake and reduced odds of hepatic steatosis were attenuated and largely lost statistical significance (Figure 1, Table 3). Particularly, all significant associations in trend analysis disappeared with adjustment of the DASH index. For example, the P trend for flavonols increased from 0·005 to 0·13 (mean OR attenuation of 14·3 %) and the P trend for total flavonoids increased from 0·002 to 0·06 (mean OR attenuation of also 14·3 %) after adjusting for the DASH score (Table 3). Similar attenuations were observed across all dietary indices. Consistent with the trend analysis, the effect size estimations for individual quartiles were also reduced with adjustment for the overall dietary quality scores (Table 3).
Total flavonoid intake and hepatic steatosis: Attenuation of OR with dietary adjustment. OR and 95 % CI for hepatic steatosis (defined by LPR < 0·33) comparing the second through fourth quartiles (Q2–Q4) of total flavonoid intake with the lowest quartile (Q1, reference). Estimates are presented for diet-unadjusted models and models adjusted for dietary quality using the Alternative Healthy Eating Index (AHEI), Mediterranean Diet Score (MDS) and Dietary Approaches to Stop Hypertension (DASH) indexes. All models are additionally adjusted for age, sex, alcohol intake, smoking status, energy intake, physical activity and education.

Diet-adjusted OR (95 % CI) for hepatic steatosis by quartile of flavonoid intake

DASH, Dietary Approaches to Stop Hypertension; AHEI, Alternative Healthy Eating Index; MDS, Mediterranean-style diet.
All models are adjusted for age, sex, alcohol intake, smoking status, total energy intake, physical activity and education, with additional adjustment for one of three a priori dietary scores (DASH, AHEI or MDS). P trend models assess linear trends using the median value of each quartile as a continuous predictor.
To estimate the extent to which flavonoids contributed to the protective association between overall diet quality and hepatic steatosis, attenuation of dietary index β-coefficients was evaluated after adding flavonoid variables to each diet-adjusted model (online Supplementary Table 3). Across all three dietary scores, flavonoid subclasses produced only minimal attenuation, generally within 0–6 %. The only exception was anthocyanidins, which reduced the dietary score coefficients by approximately 10–15 %.
Exploratory analyses for sex–flavonoid interaction were not statistically significant for any subclass (all P–interaction > 0·17; online Supplementary Table 4). Nonetheless, among men, diet-unadjusted base models found that higher anthocyanidin, flavan-3-ol, flavonoid polymers and total flavonoid intake were associated with lower odds of hepatic steatosis. While in women, flavonols demonstrated an inverse association. After adjustment for dietary quality indices, all associations were attenuated except for flavonols in women, which was with associated lower odds of hepatic steatosis in MDS adjusted model at P < 0·05
Discussion
In this cross-sectional study of hepatic steatosis and flavonoid intake in the FHS, higher flavonoid intakes of flavonols, flavan-3-ols, anthocyanins, flavonoid polymers and overall total flavonoids were associated with decreased hepatic steatosis prior to adjustment for diet quality, with the strongest associations observed in the highest quartiles of intake. These diet-unadjusted findings are generally consistent with previous prospective and cross-sectional studies over various cohorts that have demonstrated a higher flavonoid diet and greater intake of flavonoid-rich foods being associated with a lower MASLD risk(Reference Tong, Zeng and Xie14,Reference Bell, Jennings and Thompson15,Reference Zhong, Wu and Xiong42,Reference Xie and Zhang43) . However, accounting for diet quality largely attenuated these associations. This attenuation suggests that the observed protective effects of these flavonoid subclasses may be partially explained by their role as components of an overall healthy dietary pattern. Notably, flavonols and flavan-3-ols showed the most robust associations prior to adjustment, but these associations diminished in magnitude and, in most cases, lost statistical significance after dietary quality was accounted for.
There are two plausible interpretations of this pattern. First, the attenuation of associations does not necessarily negate a potential protective effect of flavonoids. Rather, it may reflect that higher flavonoid intake is an especially important element of a broader healthy diet. As such, flavonoids may mediate the protective effect of overall dietary quality on MASLD risk. Alternatively, the observed associations could be confounded by components of a healthy diet which may independently reduce hepatic steatosis risk.
If the former hypothesis is true, the unique bioactive properties in flavonoids may be the key drivers. Flavonols have been linked to many benefits, most notably its antioxidant properties(Reference Hollman and Katan44,Reference Hollman and Katan45) . In preclinical NAFLD models, fisetin, a flavonol, has demonstrated hepatoprotective effects through suppressing of hepatic lipid accumulation and anti-inflammatory Poly ADP-ribose polymerase pathways(Reference Gaballah, El-Horany and Helal46). Similarly, several preclinical studies have shown that flavan-3-ol supplementation improves metabolic conditions by modulating the expression of genes involved in hepatic lipid accumulation, as well as enhancing insulin sensitivity and thermogenesis(Reference Vauzour, Rodriguez-Ramiro and Rushbrook47,Reference Osakabe, Hoshi and Kudo48) . Additionally, cyanidin-3-O-glucoside, an anthocyanin (glycoside of anthocyanidin), has been shown to induce mitophagy – albeit through different pathways – thereby reducing hepatic oxidative stress in preclinical NAFLD models(Reference Cao, Wang and Zhang49,Reference Li, Shi and Zhu50) .
Overall, our findings contrast with those of Wang et al., who reported a significant inverse association between the highest tertile of total flavonoid intake and MASLD in the NHANES 2017–2018 cohort, even after several covariate adjustments along with the HEI-2015 dietary index(Reference Wang, Li and Zhang51). Their study found protective associations for anthocyanins, flavones and flavanones, which were not observed in our analysis. A separate study by Tong et al. using the same NHANES cycle reported inverse associations between anthocyanin and isoflavone intake and MASLD after adjusting for HEI-2015(Reference Tong, Zeng and Xie14). Additionally, another NHANES-based study only found anthocyanin intake to be protective against NAFLD, particularly among non-Hispanic whites(Reference Xiang, Li and Li52).
Several factors may account for these differences. NHANES and FHS use different dietary assessment instruments: NHANES relies on 24-hour dietary recalls, which may capture short-term intake; FHS uses a semi-quantitative FFQ, which ideally reflects usual long-term dietary patterns. A previous comprehensive review by Peterson et al. demonstrated that flavonoid intakes vary greatly within and between populations(Reference Peterson, Dwyer and Jacques53). Data from NHANES exhibit a much wider and more heterogeneous intake range, as reflected in the study by Tong et al. (Reference Tong, Zeng and Xie14) and Xiang et al. (Reference Xiang, Li and Li52) There, anthocyanidin Q4 ranged from approximately 11–644 mg/day while this FHS cohort demonstrated a more moderate distribution (Table 2). When comparing FHS with the UK Biobank cohort (where 24-hour recall was also utilised)(Reference Bell, Jennings and Thompson15), overall intake distributions were similar in magnitude and right-skewedness, but certain subclass-specific differences were observed: anthocyanidins, flavonols and flavan-3-ols of the highest-quartile median intake and ranges were markedly higher in UK Biobank (online Supplementary Table 5). In contrast, flavones and flavanones were mildly higher in FHS, further underscoring the cohort-specific dietary differences. Additionally, these cohorts differ in liver fat assessment modality: the NHANES studies mentioned previously utilised vibration-controlled transient elastography(Reference Tong, Zeng and Xie14,Reference Wang, Li and Zhang51,Reference Xiang, Li and Li52) , our current FHS analyses employ CT-based LPR, and the aforementioned UK Biobank study(Reference Bell, Jennings and Thompson15) identifies incident MASLD through ICD-10 codes. These methods vary substantially in sensitivity, dynamic range and diagnostic precision, which may further contribute to differences in effect estimates across studies(Reference Ndaa, Pandya and Swensson54,Reference Talebi, Zeraattalab-Motlagh and Rahimlou55) .
Randomised controlled trials of flavonoid supplementation in patients with NAFLD have generally demonstrated mild improvements in hepatic enzymes, lipids or markers of oxidative stress(Reference Li, Ji and Du13,Reference Liu, Luan and Duan56) . However, these intervention doses far exceed average intake of flavonoids by approximately 3- to 15-fold(Reference Peterson, Dwyer and Jacques53). In contrast, population-based dietary studies – including the present analysis – have yielded more heterogeneous findings. This discrepancy may reflect differences in dose, as pharmacologic supplementation delivers concentrated flavonoids at levels difficult to achieve through diet alone. Also, supplement trials typically enrol participants with established MASLD which may explain why they report more benefits on liver biomarkers, while population-based studies yield more heterogeneous findings that are sensitive to dietary pattern adjustments. Additionally, food-based intake delivers flavonoids within a complex mixture of fibre, antioxidants and micronutrients that may act synergistically or modulate bioavailability(Reference Egert and Rimbach57,Reference Ross and Kasum58) , and these interactions have not been thoroughly studied.
While diet quality may represent much of the flavonoid-steatosis relationship in the general population, this does not diminish the broader potential benefits of flavonoid intake in the prevention of other diseases and pathologies. Several population studies have suggested potential benefits of flavonoid subclasses in cardiometabolic outcomes(Reference Ross and Kasum58–Reference Rahimlou, Grau and Banaie-Jahromi63). As MASLD is linked to major adverse cardiovascular events(Reference Maurice and Manousou4,Reference Chung, Yu and Yoo5,Reference Rahimlou, Grau and Banaie-Jahromi63,Reference Mousavi, Rayyani and Heshmati64) , it is imperative to fully evaluate the efficacy and mechanisms of diet and flavonoid and subclass-specific intake in the prevention and treatment of MASLD and its broader comorbidities.
Strengths of this study include the use of a well-characterised cohort with CT imaging-based assessment of hepatic steatosis and comprehensive dietary data. There are several limitations to this study. The cross-sectional analysis may not support the causal effects. Additional studies, especially experimental investigations, are needed to confirm the protective effects of flavonoids on the occurrence and development of MASLD. Second, the FFQ may not accurately capture flavonoid intakes based on recall of diet and use of a predefined food list. This residual confounding may bias our observations. Bias due to differential recall is unlikely as participants were unaware of their MASLD status at the time of dietary reporting. Future epidemiological studies may potentially benefit from incorporating biomarkers of flavonoid intake, such as urinary concentrations of flavonoid metabolites(Reference Nielsen, Freese and Kleemola65,Reference Linseisen and Rohrmann66) , although these are also still lacking in validity and consensus(Reference Peterson, Dwyer and Jacques53). Next, although several subclass associations with hepatic steatosis were nominally significant (P < 0·05), none survived correction for multiple testing, and our limited sample size may have constrained statistical power to detect truly robust effects. Fourthly, the FHS cohort is composed almost entirely of non-Hispanic white individuals, and these findings may not be generalisable to more racially and ethnically diverse populations. Additional large-scale prospective research with considerations for genetic polymorphisms in flavonoid metabolism and in MASLD susceptibility(Reference Chen, Kuppa and Oliveri67), as well as potential confounding by medication use that affect hepatic lipid metabolism are also warranted.
Ultimately, given the global burden of MASLD and the lack of pharmacologic treatments, identifying specific dietary components that confer hepatic protection is highly critical. Our results indicate that the protective associations between flavonoid intake and hepatic steatosis are largely reduced by overall diet quality and that a healthier diet may be an effective initial intervention to reduce the risk of MASLD. Whether or not flavonoids exert direct protective effects or simply serve as markers of a healthier dietary pattern, the implication remains the same: encouraging adherence to an overall healthy diet rich in fruits, vegetables and other flavonoid-containing foods may be beneficial in reducing the burden of MASLD. Further studies should explore flavonoid dose–response relationships and assess whether these subclasses modify hepatic outcomes in randomised controlled trials. Additional large-scale prospective research with considerations for genetic polymorphisms in flavonoid metabolism and potential confounding by medication use are is warranted.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114526106825
Acknowledgements
None.
The Framingham Heart Study was supported by NIH contracts N01-HC-25195, HHSN268201500001I and 75N92019D00031. P. F. J. was supported in part by the USA Department of Agriculture, Agricultural Research Service Cooperative Agreement #58-8050-3-003.
The authors’ contributions were as follows – V. G. conducted the research, preformed statistical analysis and wrote the manuscript; J. M. and P. F. J. designed research, analysed data and had primary responsibility for final content; all authors read and approved the final manuscript.
The authors declare no conflicts of interest.
The Framingham Heart Study datasets analysed in the present study are available at the dbGAP repository phs000007.v32.p13 (https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs000007.v30.p11) via the Controlled Access Data.
The views and opinions expressed in this manuscript are those of the authors and do not necessarily represent the views of the National Heart, Lung and Blood Institute, the National Institutes of Health, or the USA. Department of Health and Human Services.