During pregnancy, a reduction in macrovascular endothelial function (measured by flow-mediated dilatation (FMD)) is associated with hypertensive syndromes, whereas an increase in FMD reduces the risk of CVD( Reference Green, Jones and Thijssen 1 ). In addition, preserved microvascular endothelial function in pregnancy is necessary for adequate tissue oxygenation. For example, impairments in uteroplacental vascular function in pregnancy lead to increased vascular resistance and decreased blood flow across the placenta, which reduce the placental capacity for nutrient and gas exchange( Reference Cottrell, Tropea and Ormesher 2 ). The low oxygen availability may compromise oxygen delivery to the fetus and favour the development of fetal growth restriction (FGR)( Reference Savvidou, Hingorani and Tsikas 3 ). The NO molecule is critically involved in maintaining uteroplacental blood flow in pregnancies as NO regulates vascular tonus. Thus, the reduction of NO bioavailability has been associated with FGR( Reference Cottrell, Tropea and Ormesher 2 , Reference Krause, Carrasco-Wong and Caniuguir 4 , Reference Chaddha, Viero and Huppertz 5 ).
The regulation of vascular tonus varies according to anatomical location (conduit artery or resistance vessel) owing to different vascular morphologies( Reference Aird 6 ). Measuring FMD is a way of evaluating macrovascular endothelial function in the conduit artery (i.e. brachial artery), whereas microvascular function (microcirculation) may be evaluated by near-IR spectroscopy (NIRS), a technology that observes changes in tissue oxygen saturation (StO2), allowing for the evaluation of tissue oxygen extraction (oxidative capacity) and microvascular function (tissue arterioles function) during the occlusion and reperfusion periods, respectively( Reference Masschelein, Van Thienen and Wang 7 , Reference Bailey, Winyard and Vanhatalo 8 ).
Beetroot has gained the attention of the scientific community owing to its beneficial effects on cardiovascular health. The function of beetroot consumption on the risk of CVD has been studied and clinical trials have shown that beetroot is linked to improvement at both the macro-circulatory and the micro-circulatory levels( Reference Masschelein, Van Thienen and Wang 7 – Reference Bakker, Engan and Patrician 13 ). Beetroot has beneficial effects on cardiovascular health owing to the nitrate present in this food. Nitrate can be converted to nitrite in the mouth by action of commensal facultative anaerobic bacteria that express nitrate reductase enzymes. In the acidic stomach and with low oxygen availability, nitrite is rapidly converted to NO( Reference Bailey, Winyard and Vanhatalo 8 , Reference Bakker, Engan and Patrician 13 – Reference Lundberg, Gladwin and Weitzberg 15 ).
Effects of beetroot juice (BJ) consumption on macrovascular endothelial function have been studied, demonstrating improvements in endothelial function (by FMD) in elderly people with risk factors for CVD (after ingestion of a single dose of beetroot-based nutritional gel containing approximately 12 mmol nitrate( Reference Oliveira, Morgado and Pierucci 9 )); in healthy subjects after consuming 500 ml of BJ( Reference Webb, Patel and Loukogeorgakis 10 ); in treated and untreated hypertensive subjects after 28d of BJ consumption containing approximately 6·4 mmol nitrate( Reference Kapil, Khambata and Robertson 11 ); and in hypercholesterolaemic adult patients after 6 weeks of BJ consumption containing approximately 6 mmol nitrate( Reference Velmurugan, Gan and Rathod 12 ). The effects of BJ consumption at microcirculatory levels have also been evaluated by NIRS measurement. In healthy subjects, 6 d of ingestion of BJ containing approximately 0·07 mmol nitrate/kg body promoted an improvement in muscle oxygenation status during submaximal and maximal exercise in severe acute hypoxia( Reference Masschelein, Van Thienen and Wang 7 ). Also in healthy subjects, 6 d of 500 ml of BJ consumption containing 11·2 mmol of nitrate promoted an improvement in indexes of muscle oxygenation during exercise( Reference Bailey, Winyard and Vanhatalo 8 ).
Although evidence assessing the effect of BJ consumption on macrovascular function and StO2 parameters exists, no study in pregnant women has been performed. Therefore, the present study investigated whether a single dose of BJ would improve FMD and StO2 parameters in pregnant women. A single dose of BJ was hypothesised to result in increased NO bioconversion, consequently promoting beneficial changes in FMD and StO2 parameters in pregnant women.
Methods
Subjects
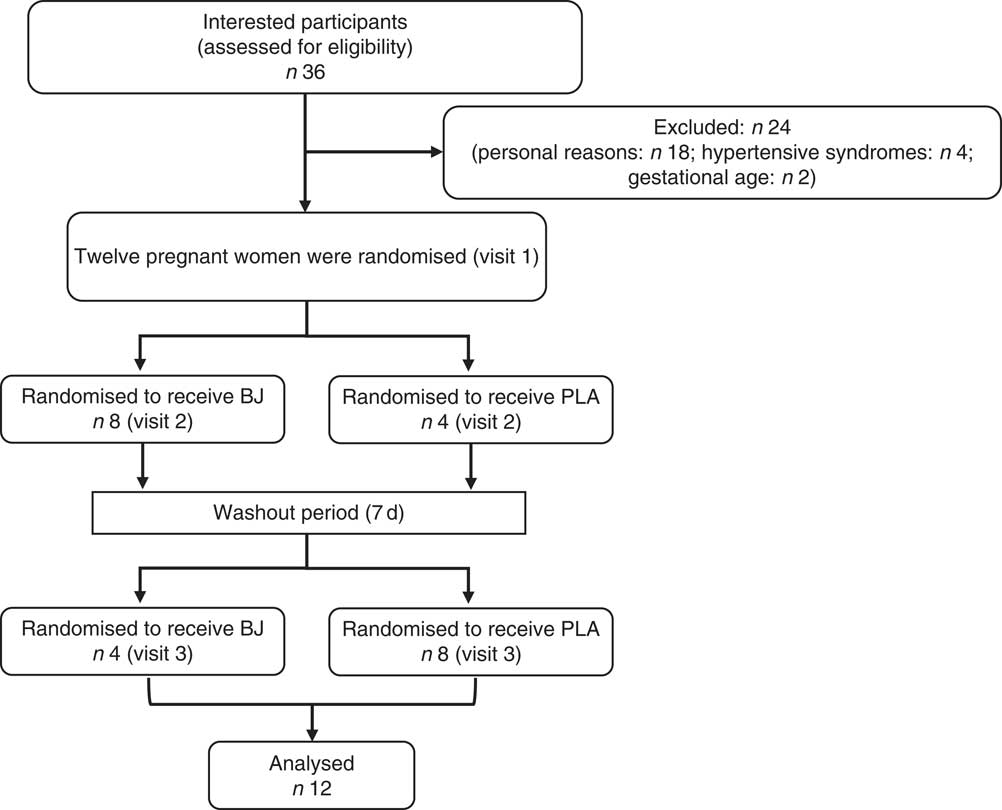
This study was conducted in accordance with the guidelines set forth in the Declaration of Helsinki, and the research ethics committee of the Universidade Federal do Rio de Janeiro, Campus Macaé, Brazil (protocol no. 56732216.5.0000.5699), approved all procedures involving human subjects. The study was registered with the Clinical Trials Registry (NCT03315806). All pregnant women gave their written informed consent. A total of thirty-six pregnant women were recruited through announcements in flyers and advertisements during community events in the Macaé City, Rio de Janeiro Estate, Brazil; twenty-four recruits withdrew for personal reasons or were ineligible according to the inclusion criteria; and twelve recruits were randomised (Fig. 1). Pregnant women were selected if they were between 20 and 35 weeks of gestation and were over 18 years of age. Exclusion criteria included the presence of diabetes mellitus and gestational diabetes, hypertensive syndromes, HIV positive, liver or kidney disease, beetroot allergy, use of antioxidants and medication that alters endothelial function (angiotensin-converting enzyme inhibitor), use of antibiotics up to 2 weeks before the beginning of the study.
Schematic of participant recruitment of the study. BJ, beetroot juice; PLA, placebo.
Study design
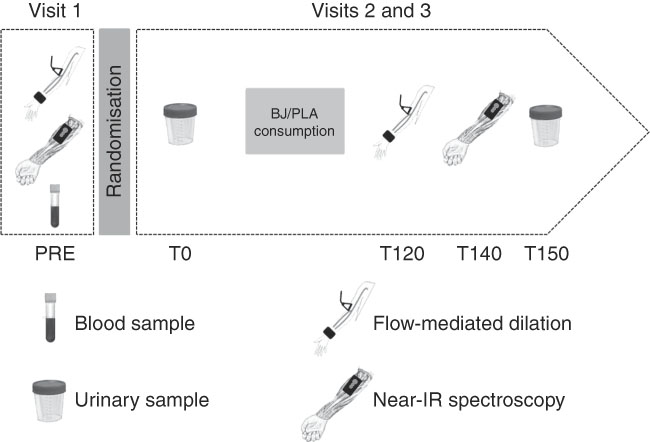
A randomised, double-blind, crossover and placebo (PLA)-controlled study was conducted from October 2016 to May 2017. All subjects reported to the Dynamic Vascular Laboratory of Federal University of Rio de Janeiro, Macaé City, Brazil, on three occasions, with at least a 1-week interval between visits. The first visit was used to register basal (PRE) macrovascular and microvascular function measurements, blood sample collection for the characterisation of pregnant women, followed by randomisation (2:1 BJ:PLA). The BJ production requires several steps, including sanitation, centrifugation, pasteurisation and portioning. Production of small quantities of BJ for only one pregnant women was impracticable because it requires considerable time for production. In the randomisation 1:1, the remaining BJ would be lost as no preservatives were added, hence its short shelf life. Furthermore, it is important to point out that refrigerating (at 4°C) and freezing (at −20°C) the juice for 1 week could reduce the nitrate content by 15 and 14 %, respectively( Reference Corleto, Singh and Jayaprakasha 16 ). Thus, randomisation 2:1 (BJ:PLA) was more convenient in guaranteeing no nitrate losses. The participants were randomised and assigned by laboratory staff to ensure blind measurement. In the second and third visit, sample urine was drawn before (T0) BJ or PLA consumption. Afterward, macrovascular endothelial function, StO2 parameters and urine samples were collected, respectively, at 120 min (T120), 140 min (T140) and 150 min (T150) after BJ or PLA consumption (Fig. 2). The three visits were held between 07.00 and 12.00 hours. The participants were instructed to fast for at least 8 h before each visit; to restrict carbonated mineral water; to avoid the intake of foods rich in nitrate and nitrite; to not use any mouthwash; to not consume coffee or teas; and not to exercise for at least 12 h before the visits.
Experimental design. BJ, beetroot juice; PLA, placebo.
Beetroot/placebo juice preparation
The beetroots (Beta vulgaris L.) were washed in tap water, sanitised with a chlorine solution and centrifuged (Model CE700; Black & Decker). The resulting juice was pasteurised, and 140 ml of juice was distributed in two bottles of dark colour. For preparation of the PLA (control nitrate-depleted juice), the nitrate was removed from BJ by using a specific anion-exchange resin A-520E (Purolite®). The contents of nitrate of the BJ and PLA consumed by the subjects of this study were 8·95 (sd 0·28) and 0·27 (sd 0·02) mm/140 ml, respectively.
Urinary nitrate
Nitrate analysis in urine was performed as previously described by Croitoru( Reference Croitoru 17 ) by using a HPLC system. A volume of 100 µl of sample was added to 100 µl of acetonitrile (v/v) and centrifuged (10 min at 14 000 rpm) for deproteinisation. Thereafter, 150 µl of supernatant was mixed with 150 µl of mobile phase (v/v) (5 mm tetrabutylammonium hydroxide) and the solution was mixed. Finally, 150 µl was injected in HPLC. Sodium nitrate was used for quantification, and the results were expressed as mmol/mmol creatinine for nitrate.
Macrovascular endothelial function analysis
The evaluation of the FMD was performed in a quiet air-conditioned room (constant temperature of 22–25°C) 120 min (T120) after BJ/PLA consumption. All examinations were conducted by the same investigator. Pregnant women rested quietly with an angle of 90° between the femur and the trunk. Endothelial function was assessed in accordance with Oliveira et al.( Reference Oliveira, Morgado and Pierucci 9 ). FMD is calculated as the percentage dilated at the maximum peak brachial artery diameter after cuff deflation, relative to maximum baseline brachial artery diameter (BBD). All analyses were conducted by a single investigator who was blinded to the participant and supplementation order. CV was intraday 5·1 (sd 0·31) % and interday 5·1 (sd 0·12) %.
Oxygen saturation parameters analysis
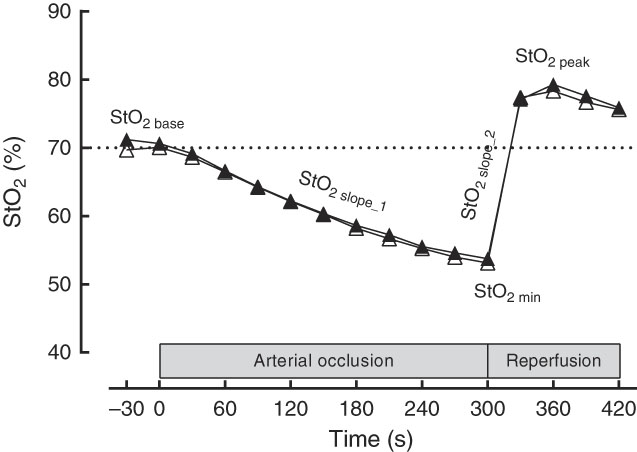
Changes in tissue StO2 were detected by a NIRS device (PortaMon; Artinis Medical Systems) that was placed on the flexor muscles of each subject’s dominant forearm. After the StO2 signals had been stable for 30 s, the cuff was rapidly inflated to suprasystolic levels (i.e. 250 mmHg), to obstruct blood flow to forearm muscles and a 5-min arterial occlusion was performed. The reperfusion phase after cuff release was recorded for more 2 min. During all tests, the NIRS system was connected to a personal computer via Bluetooth for data acquisition (10 Hz), analogue-to-digital conversion and subsequent analysis of the raw data (i.e. no filter was used) using native software (Oxysoft version 2.1.6; Artinis Medical Systems). The software calculates absolute changes in light absorption at the different wavelengths (750 and 850 nm) of oxyHb (O2Hb) and deoxyHb (HHb) using the spatially resolved spectroscopy method. StO2 was calculated as (O2Hb/O2Hb+HHb)×100. The following NIRS parameters were considered for statistical analysis: baseline StO2 (StO2 base), which represents the average of 30 s of StO2 before arterial occlusion; desaturation rate (StO2 slope_1), which corresponds to the slope of StO2 (the rate of change in StO2 per time) during the arterial occlusion phase; minimum StO2 (StO2 min), which corresponds to the lowest StO2 value during the arterial occlusion phase; resaturation rate (StO2 slope_2), which corresponds to the slope of StO2 during the reperfusion phase; maximal StO2 (StO2 max), which corresponds to the highest StO2 value during the reperfusion phase; time to maximal StO2 (StO2 tmax), which corresponds to the time to reach the StO2 max. CV was intraday 1·6 (sd 0·9) % for baseline, 2·0 (sd 1·6) % for occlusion and 1·6 (sd 1·1) % for reperfusion and interday 1·8 (sd 0·2) % for baseline, 2·9 (sd 0·7) % for occlusion and 1·5 (sd 0·1) % for reperfusion.
Statistical analysis
An a priori power analysis was conducted (G*Power version 3.0.1) for an F test (ANOVA, repeated measures, within factors). On the basis of statistical power (1−β) of 0·80, an effect size of 0·35 and an overall level of significance of 0·05, at least sixteen participants were needed to detect a statistical difference. Although this study was unable to achieve the sample size of sixteen participants, the effect size was taken into account in order to determine the magnitude of the BJ ingestion effect on endothelial function and muscle StO2 parameters( Reference Sullivan and Feinn 18 ). In addition, previous studies have observed statistical difference in StO2 parameters with ≤12 volunteers after beetroot supplementation( Reference Bailey, Winyard and Vanhatalo 8 , Reference Oliveira, Morgado and Pierucci 9 , Reference Horiuchi, Endo and Dobashi 19 ).
The normality and homogeneity of variances of the data were examined with the Shapiro–Wilk and Mauchly tests, respectively, and were violated only for StO2 base. To identify differences in the FMD and StO2 parameters between PRE, PLA and BJ interventions, a repeated-measures one-way ANOVA was performed. When the assumption of normality and homogeneity of variances were violated, a Friedman test was used. To identify differences in urinary nitrate between BJ and PLA, a two-way ANOVA was used. In addition, ANOVA with repeated measurement was also performed to verify whether the order of BJ or PLA condition affected the results in this study. The effect size to observe the magnitude of the effect of BJ ingestion compared with PLA was calculated by Cohen’s d, where a value <0·2 is considered trivial, 0·2–<0·5 is a small effect, 0·5–<0·8 is a moderate effect and ≥0·8 is a large effect. Statistical significance was set at the 0·05 level of confidence. All analyses were performed using a commercially available statistical package (IBM SPSS Statistics version 23 for Mac) and the results were expressed as means and standard deviations.
Results
Of the thirty-six assessed for eligibility, twelve (33 %) were randomised and completed the study (Fig. 1). Furthermore, the StO2 data of one pregnant woman had to be excluded owing to technical reasons regarding the loss of NIRS signal. Therefore, only eleven volunteers were considered for analysis of this variable. The pregnant women were adult (27 (sd 6·51) years), sedentary, with a gestational age of 20–35 weeks, singleton pregnancies and the pregnant women did not use medications. The baseline volunteers’ characteristics are shown in Table 1.
Baseline demographic and clinical characteristics (Mean values and standard deviations)
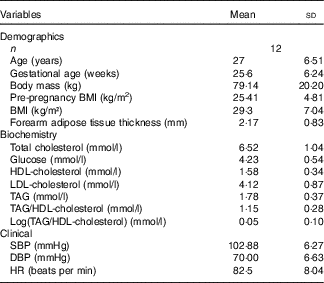
SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate.
Urinary nitrate
T0 values of urinary nitrate (mmol/mmol creatinine) were not different between groups (PLA: 0·02 (sd 0·00) v. BJ: 0·02 (sd 0·00), P=0·866). A significant main effect for time was observed (P=0·001). Post hoc test revealed that urinary nitrate increased 10-fold in T150 after BJ consumption compared with T0 (T150: 0·20 (sd 0·13) v. T0: 0·02 (sd 0·00), P=0·000), but not for PLA consumption compared with T0 (T150: 0·02 (sd 0·00) v. T0: 0·02 (sd 0·00), P=0·992). Furthermore, there was a significant treatment×time interaction effect for urinary nitrate (P=0·001). Post hoc test revealed that urinary nitrate increased 10-fold in T150 after BJ consumption compared with PLA (P=0·001). In addition, BJ and PLA have a large effect size (d=1·95).
Endothelial function
The endothelial function parameters are shown in Fig. 3 and Table 2. FMD (%) increased 2-fold after BJ consumption (11·00 (sd 1·67)) compared with PLA (5·34 (sd 1·31)) and PRE (5·53 (sd 1·17)). BJ and PLA have a large effect size (d=3·77). There were no changes on BBD and PDB after BJ compared with PRE and PLA. In addition, there was no effect on the supplementation order (BJ→PLA v. PLA→BJ) for FMD (P=0·547), BBD (P=0·673) and PDB (P=0·658).
Mean flow-mediated dilatation (FMD) after beetroot juice (BJ) and placebo (PLA) intervention. Values are means, with standard deviations represented by vertical bars. * Mean value was significantly different from baseline (PRE) and PLA (P<0·001).

Macrovascular endothelial function parameters at baseline and after beetroot juice (BJ) and placebo (PLA) consumption (Mean values and standard deviations)

PRE, baseline; FMD, flow-mediated dilation; BBD, maximum baseline brachial artery diameter; PBD, maximum peak brachial artery diameter.
***Statistical difference compared with PRE and PLA (P<0·001; one-way ANOVA followed by Bonferroni’s post hoc test).
Microvascular function
The microvascular function parameters are shown in Fig. 4 and Table 3. There were no changes in StO2 base, StO2 slope_1, StO2 min, StO2 slope_2, StO2 max and StO2 tmax after BJ compared with PRE and PLA. In addition, there was no effect on the supplementation order (BJ→PLA v. PLA→BJ) for StO2 base (P=0·784), StO2 slope_1 (P=0·720), StO2 min (P=0·784), StO2 slope_2 (P=0·518), StO2 max (P=0·671) and StO2 tmax (P=0·386). BJ/PLA have a trivial effect size for StO2 base (d=0·29), StO2 slope_1 (d=0·03), StO2 min (d=0·04), StO2 slope_2 (d=0·04), StO2 max (d=0·06) and StO2 tmax (d=0·03).
Near-IR spectroscopy-derived profile of typical comportment of forearm muscle oxygen saturation (StO2, %) and the parameters evaluated. StO2 base, baseline oxygen saturation; StO2 slope_1, desaturation rate; StO2 slope_2, resaturation rate; StO2 min, minimum oxygen saturation. ![]() , Placebo;
, Placebo; ![]() , beetroot juice.
, beetroot juice.
Oxygen saturation (StO2) parameters at baseline and after beetroot juice (BJ) and placebo (PLA) consumption* (Mean values and standard deviations)
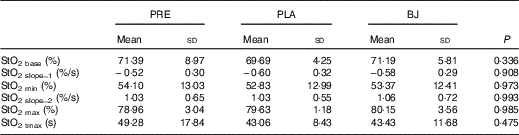
PRE, baseline; StO2 base, baseline StO2; StO2 slope_1, desaturation rate; StO2 min, minimum StO2; StO2 slope_2, resaturation rate; StO2 max, maximal StO2; StO2 tmax, time to maximal StO2.
*One-way ANOVA followed by Bonferroni’s post hoc test.
Discussion
The aim of this study was to examine the effects of a single dose of BJ on macrovascular endothelial function FMD and StO2 parameters in pregnant women. This is the first investigation into the effects of the single dose of BJ on FMD and StO2 parameters (StO2 base, StO2 min, StO2 slope_1, StO2 max, StO2 slope_2 and StO2 tmax) in pregnant women. The major finding of this study is that a single dose of a BJ improves FMD in pregnant women.
FMD is a non-invasive test of endothelial function, and changes in FMD values provide important prognostic information( Reference Harris, Nishiyama and Wray 20 ). The FMD technique promotes increased blood flow and vasodilation owing to changes in shear stress, and this phenomenon is mediated by release of NO. Subjects with CVD have lower FMD values (endothelial dysfunction), whereas increased FMD decreases the risk of CVD( Reference Green, Jones and Thijssen 1 ). Low FMD values in pregnancy are associated with health complications of the mother and fetus, such as pre-eclampsia and FGR. It has been shown that pregnant women who developed pre-eclampsia and FGR have shown FMD values of 3·5 and 6·2 %, respectively, whereas pregnant women who did not develop pre-eclampsia and FGR presented FMD values of 8 %( Reference Savvidou, Hingorani and Tsikas 3 ). The authors concluded that endothelial function is impaired in pregnant women who develop pre-eclampsia and FGR. In the present study, although the pregnant women did not develop pre-eclampsia and/or FGR, a low baseline FMD (5 %) was observed, suggesting impaired endothelial function. After BJ consumption, the FMD values increased from 5 to 11 % (meaning a 110 % of increase), thereby reaching FMD values similar to pregnant women who did not develop pre-eclampsia and FGR( Reference Savvidou, Hingorani and Tsikas 3 ).
The results of FMD in the present study are in accordance with previous findings( Reference Oliveira, Morgado and Pierucci 9 , Reference Kapil, Khambata and Robertson 11 , Reference Asgary, Afshani and Sahebkar 21 ). Oliveira et al. ( Reference Oliveira, Morgado and Pierucci 9 ) found improvements on FMD after ingestion of a single dose of beetroot-based nutritional gel rich in nitrate (containing approximately 12 mmol nitrate in 100 g) in the elderly with risk factors for CVD. Kapil et al. ( Reference Kapil, Khambata and Robertson 11 ) demonstrated increased FMD after 28 d of ingestion of 250 ml of BJ (containing approximately 6·4 mmol of nitrate in 250 ml) in hypertensive subjects. Asgary et al. ( Reference Asgary, Afshani and Sahebkar 21 ) observed improvements on FMD after 2 weeks of consumption of 250 ml of BJ and 250 g of cooked beet in hypertensive subjects. Therefore, the results of these studies suggest that the effect of a single dose of BJ on FMD may be the same regardless of the population studied.
Increase in urinary nitrate is in accordance with previous findings( Reference Oliveira, Morgado and Pierucci 9 , Reference Baião, Conte-Junior and Paschoalim 22 ). Baião et al. ( Reference Baião, Conte-Junior and Paschoalim 22 ) demonstrated an increase in urinary nitrate concentration via bioconversion after ingestion of a single dose of 100 ml of BJ (containing approximately 1·6 mmol of nitrate). Oliveira et al. ( Reference Oliveira, Morgado and Pierucci 9 ) showed an increase in urinary nitrate concentration after ingestion of a single dose of beetroot-based nutritional gel rich in nitrate (containing approximately 12 mmol nitrate in 100 g). Siervo et al. ( Reference Siervo, Oggioni and Jakovljevic 23 ) showed an increase in nitrate concentration after ingestion of 140 ml of BJ (containing 12 mmol of nitrate) during 7d.
StO2 parameters during the arterial occlusion period (StO2 min and StO2 slope_1) provide information about tissue oxygen extraction( Reference Kolb, Orbegozo and Creteur 24 ). Dipla et al. ( Reference Dipla, Triantafyllou and Grigoriadou 25 ) evaluated recently StO2 parameters during arterial occlusion in pregnant women with gestational diabetes mellitus (GDM). In GDM, the StO2 min value was greater when compared with the control group of pregnant women (approximately 55 % for GDM and 40 % for control), suggesting that the GDM group exhibited a reduced capacity for O2 extraction at the skeletal muscle level. In the present study, pregnant women demonstrated an StO2 min value near the GDM group in the Dipla et al.( Reference Dipla, Triantafyllou and Grigoriadou 25 ) study, suggesting that the pregnant women in the present study possibly presented an impaired capacity for O2 extraction. It is already known that elevated blood glucose increases cell oxidative stress( Reference Asmat, Abad and Ismail 26 , Reference Rochette, Zeller and Cottin 27 ), which impairs oxygen extraction by tissues (mitochondrial dysfunction)( Reference Pedersen, Baekgaard and Quistorff 28 ). In this study, the pregnant women presented normal blood glucose (<5·55 mmol/l); however, they presented elevated cholesterol (>5 mmol/l). Elevated blood cholesterol has also been shown to be associated with increased oxidative stress( Reference Rasool, Abdul Rahman and Abd Ghaffar 29 ), which may impair oxygen extraction by tissues( Reference Asmat, Abad and Ismail 26 , Reference Rochette, Zeller and Cottin 27 ). In addition, a significant correlation between StO2 min and cholesterol (date not showed) was observed, which may explain the greater StO2 min value in pregnant. It was expected that BJ consumption would improve StO2 parameters during arterial occlusion owing to the effect of nitrate on mitochondrial function. The nitrate–nitrite–NO pathway has been shown to modulate O2 consumption in mitochondria( Reference Larsen, Schiffer and Borniquel 30 ). In complex IV of the electron transport chain, NO competes with O2 for cytochrome c oxidase (COX), whereby NO binds to COX when oxygen is available( Reference Larsen, Schiffer and Borniquel 30 ). However, no changes were observed in StO2 parameters during arterial occlusion. Bioconversion of nitrite to NO requires approximately 50 % Hb-saturated oxygen( Reference Lundberg, Weitzberg and Gladwin 31 ). The present study observed that approximately 53 % Hb was saturated with oxygen, which indicates that the environment was favourable for nitrite-to-NO bioconversion, although no significant difference was observed in muscle oxygen extraction.
During the reperfusion phase, StO2 parameters (StO2 max, StO2 slope_2 and StO2 tmax) reflect the ability of recruiting greater arteriole numbers( Reference Ryan, Brophy and Lin 32 ). After arterial disocclusion, there is an increase in blood flow in the ischaemic limb, which promotes an increase in vasoactive substances( Reference Oliveira, Morgado and Conte-Junior 33 ). An improvement in StO2 parameters after BJ consumption owing to vasodilator propriety of NO had been expected. However, there was no significant difference in StO2 parameters after BJ consumption. The StO2 max and StO2 slope_2 in the present study was near the control group (healthy pregnant) values in Dipla et al.( Reference Dipla, Triantafyllou and Grigoriadou 25 ) study, suggesting that pregnant women present a sufficient ability to recruit greater arteriole numbers. Hence, the integrity of microvascular reactivity in pregnant women in the present study may be a possible reason for the neglible effect on these parameters after BJ consumption. It was speculated that NO should be delivered only to the areas with impaired microvascular perfusion to avoid diffuse dilation in perfused capillaries. As microvascular reactivity is preserved in pregnant women of this study, the capillaries are sufficiently perfused and do not require increase in perfusion.
This study was unable to achieve a large sample size owing to physiological changes inherent in pregnancy, such as sea sickness and vomiting, as well as to the protocol of the study that requires periods of fasting. However, the strengths of the experimental study design are that all pregnant women were submitted to both BJ or PLA ingestion (crossover design), the muscle StO2 measure presented good repeatability (CV approximately 3 %), the pregnancies were within the gestational age of 20–35 weeks, the study included adults (27 (sd 6·51) years) and the ingestion of the food containing nitrate and nitrite before all experimental visits was controlled. In addition, the BJ ingestion is a potent nutritional supplement, which has been demonstrated to provoke changes in muscle StO2 parameters( Reference Barry, Szucs and Reyes 34 ). Therefore, the very small effect size reported in the present study suggests the weak magnitude of the BJ ingestion effect on muscle StO2 parameters( Reference Engelke, Halliwill and Proctor 35 ).
In addition, the differences among the responses observed in macro-circulatory and micro-circulatory levels after BJ consumption can be explained by cell heterogeneity. Differences in endothelial cells are reported at the level of morphology, function, gene expression and antigen composition( Reference Aird 6 ). Furthermore, endothelial cells exhibit differences depending on where the type of vessel or organ is located( Reference Potente and Mäkinen 36 ). Therefore, NO action seems to be different in macrovasculature and microvasculature.
In summary, the effect of a single dose of BJ consumption, a source of dietary nitrate, on macrovascular endothelial function and StO2 parameters was investigated. The results of the present study show that BJ improves macrovascular endothelial function, but not StO2 parameters. Furthermore, BJ consumption increases urinary nitrate. The beneficial effect on macrovascular endothelial function observed after BJ consumption may be an important nutritional intervention for pregnant women aiming to reduce the risk of developing CVD. However, future studies are necessary to address and evaluate the effects of BJ consumption in pregnancy with CVD.
Acknowledgements
The authors thank Ricky Toledano for preparing the English version of the manuscript.
This work was supported by the Research Foundation of the State of Rio de Janeiro – FAPERJ (process no. E-26/111.297/2013 and E-26/110.309/2014). Funding was also provided from The Brazilian National Council for Scientific and Technological Development – CNPq (process no. 442977/2014-0).
M. V.-S. and G. V. d. O. contributed to volunteers’ recruitment and physiological analysis. T. S. A. reviewed the manuscript. M. V.-S. contributed substantially to data acquisition, statistical analysis and data interpretation, and was the manuscript writer. All authors read and approved the final manuscript.
The authors have no conflicts of interest that are directly relevant to the content of this manuscript.









