Introduction
Mood and anxiety disorders affect 15%–25% of adolescents (Racine et al., Reference Racine, McArthur, Cooke, Eirich, Zhu and Madigan2021) and are associated with social impairment, poor academic performance, substance use, obesity, and death by suicide (Bitsko et al., Reference Bitsko, Holbrook, Ghandour, Blumberg, Visser, Perou and Walkup2018). Sleep disturbances occur in 60%–80% of adolescents with major depressive disorder (MDD) and generalized anxiety disorder (GAD) (Chase & Pincus, Reference Chase and Pincus2011; Liu et al., Reference Liu, Buysse, Gentzler, Kiss, Mayer, Kapornai, Vetró and Kovacs2007), with experimental evidence suggesting poor sleep may be one causal factor underlying affective dysregulation in this age group (Baum et al., Reference Baum, Desai, Field, Miller, Rausch and Beebe2014; Booth et al., Reference Booth, Carskadon, Young and Short2021). It is also well-established that sex differences in rates of mood and anxiety disorders emerge during adolescence (Angold et al., Reference Angold, Costello, Erkanli and Worthman1999), increasing in relative prevalence to approximately twice the risk for female adolescents. These findings suggest that sleep disturbances during adolescence may contribute to sex differences in mood pathology. While prior studies have established insufficient sleep duration as a predictor of affective dysfunction in mixed sex samples (Johri et al., Reference Johri, Pillai, Kulkarni and Balkrishnan2025), research is critically lacking in several areas, including: (1) a priori examination of sex-specific effects; (2) consideration of additional sleep and sleep-related characteristics that are directly influenced by puberty, such as sleep regularity and circadian timing; and (3) characterizing outcomes by specific stages of adolescence, particularly the pubertal transition, where vulnerabilities to mood and anxiety disorders emerge.
The pubertal transition
As children move from prepuberty into puberty, their natural sleep and wake times shift later, reflecting an average delay in circadian timing (Laberge et al., Reference Laberge, Petit, Simard, Vitaro, Tremblay and Montplaisir2001). This delay occurs at an earlier age in female compared with male adolescents, and is associated with altered brain development (Lunsford-Avery et al., Reference Lunsford-Avery, Damme, Engelhard, Kollins and Mittal2020) and mood symptoms (Liu et al., Reference Liu, Buysse, Gentzler, Kiss, Mayer, Kapornai, Vetró and Kovacs2007). Additionally, several sex differences in sleep function emerge during puberty, with adolescent females experiencing higher rates of primary sleep disorders (Marver & McGlinchey, Reference Marver and McGlinchey2020), shorter sleep duration (Maslowsky & Ozer, Reference Maslowsky and Ozer2014), poorer sleep quality (Galland et al., Reference Galland, Gray, Penno, Smith, Lobb and Taylor2017), and higher levels of daytime fatigue (Forest et al., Reference Forest, Gaudreault, Michaud and Green-Demers2022). These differences can be attributed in part to activational (or acute) effects of gonadal steroid hormones – estradiol/progesterone for females, testosterone for males – as these hormones directly modulate sleep processes (Mong & Cusmano, Reference Mong and Cusmano2016). Further, interactions between gonadal hormones, the central circadian pacemaker, and the hypothalamic-pituitary-adrenal (HPA) axis, which coordinates physiological responses to the psychosocial and other stressors experienced during adolescence, play a major role in sleep and affective function (reviewed in [Bailey & Silver, Reference Bailey and Silver2014]). Experimental evidence suggests that this integrated system may be more susceptible to dysregulation in females, particularly in the context of heightened stress (Chen et al., Reference Chen, Zorrilla, Smith, Rousso, Levy, Vaughan, Donaldson, Roberts, Lee and Vale2006; Verma et al., Reference Verma, Hellemans, Choi, Yu and Weinberg2010).
Endogenous (e.g., hormone exposure) and exogenous (e.g., life experience) processes also exert organizational effects during puberty (Schulz et al., Reference Schulz, Molenda-Figueira and Sisk2009), shaping physiological systems in a longer-lasting fashion during sensitive developmental windows. Organizational effects during puberty are highest near the pubertal transition (Schulz et al., Reference Schulz, Molenda-Figueira and Sisk2009) and are necessary for healthy maturation of brain networks governing emotion-regulation and cognition. Studies have suggested that adolescent sleep disturbance can not only produce acute deficits in mood, but may also adversely affect developmental trajectories and increase the risk of mental illness in adulthood (Roane & Taylor, Reference Roane and Taylor2008; Uccella et al., Reference Uccella, Cordani, Salfi, Gorgoni, Scarpelli, Gemignani, Geoffroy, De Gennaro, Palagini, Ferrara and Nobili2023), consistent with direct organizational effects of sleep disturbance. The mechanisms underlying these effects – for instance, how they interact with/influence normative pubertal organizational effects of gonadal steroids to yield sex differences in pathology – are poorly understood. Nonetheless, understanding the sex-specific influence of sleep and circadian processes during sensitive periods such as puberty may have substantial implications for both treatment and prevention of mental illness.
Sleep-related processes and mood
Sleep is facilitated by two parallel processes: (1) a homeostatic drive for sleep and (2) circadian rhythms (Borbély et al., Reference Borbély, Daan, Wirz-Justice and Deboer2016). The former increases with time spent awake; sleep of insufficient duration does not fully reset the homeostatic drive, resulting in excessive daytime sleepiness (Durmer & Dinges, Reference Durmer and Dinges2005). Circadian rhythms increase and decrease wakefulness within ∼ 24-h oscillations anchored to environmental cues (e.g., light and daily routines) (Rosenwasser & Turek, Reference Rosenwasser and Turek2015), and altered circadian rhythms may manifest as wakefulness or arousal at atypical times (e.g., late evening – a circadian phase delay, or very early in the morning – a phase advance) (Pavlova, Reference Pavlova2017).
Sleep deprivation and altered circadian timing have both been associated with mood impairment during adolescence (Johri et al., Reference Johri, Pillai, Kulkarni and Balkrishnan2025). Additionally, individual variability in circadian entrainment, known as chronotype, can interact with circadian timing to exacerbate phase disturbances and associated behavioral outcomes (Zou et al., Reference Zou, Zhou, Yan, Yao and Lu2022). Chronotype can generally be categorized into morningness (a tendency toward greater arousal during the morning) and eveningness (a tendency toward greater arousal during the evening). Substantial evidence (rev. in [Kivelä et al., Reference Kivelä, Papadopoulos and Antypa2018]) has linked eveningness chronotype with psychopathology (e.g., depression, anxiety, psychosis, and maladaptive eating behaviors). Multiple biological systems have been implicated in affective dysfunction related to circadian phase misalignment, including changes in hypothalamic-pituitary-adrenal (HPA) axis (Nicolaides et al., Reference Nicolaides, Vgontzas, Kritikou, Feingold, Adler and Ahmed2000), inflammatory (Zielinski & Gibbons, Reference Zielinski and Gibbons2022), and melatoninergic (Honma et al., Reference Honma, Revell, Gunn, Davies, Middleton, Raynaud and Skene2020) function.
Sleep regularity and adolescent mental health
Another emerging feature of sleep that appears important for affective function is sleep regularity, defined by the sleep regularity index (SRI) as the probability of an individual being in the same wake/sleep state at any two time points 24 h apart (Fischer et al., Reference Fischer, Klerman and Phillips2021). Irregular sleep patterns are common in adolescence (e.g., decreasing weekday sleep to meet academic demands, compensating by increasing weekend sleep), and reduced sleep regularity during adolescence has been associated with psychiatric symptoms (Bei et al., Reference Bei, Manber, Allen, Trinder and Wiley2017; Castiglione-Fontanellaz et al., Reference Castiglione-Fontanellaz, Schaufler, Wild, Hamann, Kaess and Tarokh2023), including depression (Lunsford-Avery et al., Reference Lunsford-Avery, Wang, Kollins, Chung, Keller and Engelhard2022). Sleep regularity has been shown to improve prediction of adolescent behavioral health outcomes beyond shortened/fragmented sleep (Lunsford-Avery et al., Reference Lunsford-Avery, Wang, Kollins, Chung, Keller and Engelhard2022), and inconsistent associations with other sleep measures suggests that sleep regularity is regulated by unique physiological processes (which may bear specific interactions with puberty). Though not utilizing SRI specifically, one study found that variability in sleep minutes predicted trajectories of internalizing and externalizing symptoms in a longitudinal study of children and adolescents, consistent with the idea that sleep regularity may have long-term effects on affective development (Thompson et al., Reference Thompson, Gillis, Hinnant, Erath, Buckhalt and El-Sheikh2024). It should be noted that some research suggests that irregular sleep across the lifespan may be related to desynchrony of circadian rhythms (Murray et al., Reference Murray, Phillips, Magee, Sletten, Gordon, Lovato, Bei, Bartlett, Kennaway, Lack, Grunstein, Lockley, Rajaratnam, Armstrong, Chohan, Djavadkhani, Dodds, Gunaratnam, Hardy and Yu2019; Phillips et al., Reference Phillips, Clerx, O’Brien, Sano, Barger, Picard, Lockley, Klerman and Czeisler2017).
Given the apparent influence of sleep on short- and long-term behavioral outcomes, sleep regularity during puberty may serve as an important metric for predicting both the development of acute mood symptoms as well as risk of adulthood psychiatric disease. However, no studies to date have examined sleep regularity in relation to mood during puberty specifically. Further, despite strong evidence that sleep modulates biophysiological function (Morris et al., Reference Morris, Aeschbach and Scheer2012), little work has directly examined how sleep regularity influences the circadian release of hormones such as cortisol and melatonin. Lastly, relationships between sleep regularity, biological sex, and mood have not been established.
The current study
The present study assesses relationships between sleep regularity, mood, and circadian rhythms using actigraphy-based sleep data obtained from a larger study examining hormone-mood relationships in early post-menarchal (within 15 months of menarche) female adolescents. The primary aims of this sub-study were to examine: (1) sleep regularity as a predictor of depressive symptoms; (2) the impact of chronotype on the relationship between mood and sleep regularity; and (3) sleep regularity in relation to hormone measures of circadian function, including diurnal cortisol and 6-sulphatoxymelatonin (aMT6s), a urinary metabolite of melatonin. We hypothesized that sleep regularity would predict average depression scores across the study period, and that chronotype would moderate the effects of sleep regularity and mood given the generally protective effect of morningness against affect dysregulation (Zou et al., Reference Zou, Zhou, Yan, Yao and Lu2022) and findings suggesting that chronotype may moderate sleep effects on behavior/cognition (Wang et al., Reference Wang, Huo, Yao, Zhang, Ma, Nie, Wang, Zhou, Xu, Weng, Xiang, Hu and Liu2024). For circadian hormones, we hypothesized that reduced sleep regularity would be associated with a flattened diurnal cortisol curve, reduced cortisol awakening response, and decreased morning aMT6s levels (which reflect melatonin from the previous evening).
Methods
Participants
Adolescents (n = 66) aged 11–14 who were assigned female sex at birth and undergoing a healthy pubertal transition were recruited using flyers, mass emails to community members, and electronic school communications. Participants were mid-to-late puberty (Tanner Stage 4) according to self-assessment via line drawings and within 15 months post-menarche, as indicated initially by caregivers during screening, and confirmed by self-report on the Pubertal Development Scale (PDS). Psychotic symptoms, bipolar disorder, or active suicidal ideation were exclusionary, as were medications or supplements known to alter hormones, mood, and neurological function (e.g., hormonal contraceptives, antidepressants, anxiolytics, and stimulants).
Procedure
After establishing eligibility on a parent-reported screening form, eligible adolescents underwent an initial enrollment, which included an abbreviated Structured Clinical Interview for DSM-V (SCID) to screen for psychotic symptoms, bipolar disorder, and current suicidal ideation. During enrollment, participants and their parents were instructed on the daily and weekly assessment protocols and provided with filter paper strips for dried urine (cortisol and melatonin) collections. Starting seven days after menses onset, participants completed a one-week sleep and circadian rhythm assessment involving self-reported sleep behaviors, 24-h objective sleep monitoring using wrist actigraphy, and serial cortisol and melatonin (aMT6s) collections for four days to capture circadian rhythms during the mid-to-late follicular phase. The 8-day assessment period began 7 days after menses onset for all participants (Figure 1).
Overview of Study Design. Participation began on Day 1 of participants’ first menstrual period following enrollment (Period 1) and continued for a total of 6 weeks. Starting on Day 1 of Period 1, participants completed 6 weekly surveys assessing mood, stress, and sleep. Sleep analysis began on Day 7 of their second menstrual period following enrollment, during which participants wore an actigraphy wristwatch and completed twice-daily sleep diaries for 8 days and collected serial cortisol via dried urine samples for 4 days (Days 3-6 of sleep analysis). Created in BioRender. Jensen, K. (2025) https://BioRender.com/t88t094.

Study approval.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the UNC Chapel Hill Institutional Review Board. Parents and adolescents provided written consent and assent prior to participating. Participants received prorated compensation for participation.
Measures
Screening
Caregivers of prospective participants completed a brief online screen, followed by a phone screen. Screening questions included age, demographics, medical and psychiatric history, medications, and questions pertaining to pubertal development.
Baseline (enrollment) assessments
Participants completed a series of questionnaires assessing mood, interpersonal stress exposure, and sleep patterns at enrollment.
Pubertal staging.
Participants self-reported menarche status, breast development, pubic hair growth, height or growth spurt, and skin changes via the PDS using a 4-point scale, ranging from 1 (“No Development/No Menses”) to 4 (“Completed/Menses”). Self-assessment of Tanner Staging was completed using line drawings. A gonadal score was calculated as the sum of the height, breast development, and menarche items, according to (Shirtcliff et al., Reference Shirtcliff, Dahl and Pollak2009). A category score was computed as the total sum of the scale values above, which, using cutoffs as described in (Carskadon & Acebo, Reference Carskadon and Acebo1993), can be used as a measure of pubertal progress (e.g., prepuberty, early-/mid-/late-puberty, and postpuberty).
Recent life events.
The Coddington Life Events Record (CLER) (Coddington, Reference Coddington1972) is a 26-item inventory of stressful life events adolescents may experience (e.g., parents’ divorce, gaining a sibling, a change in parents’ financial status, a major move, the death of a loved one). Participants selected items that they had experienced in the past six months. A sum score was calculated based on the total number of items selected. Higher scores indicate a greater number of stressful life events in the six months before enrollment.
Sleep characteristics.
Participants completed the 10-item Sleep/Wake Problems Behavior Scale (Wolfson & Carskadon, Reference Wolfson and Carskadon1998) to assess sleep-related functional impairment (e.g., sleeping past noon) in the previous two weeks (scale: 1 (“never”) to 5 (“every day/night”), range: 10–50).
Chronotype.
The 10-item Superscience Morningness/Eveningness Scale (Carskadon et al., Reference Carskadon, Vieira and Acebo1993) assessed chronotype (i.e., morningness vs. eveningness). Individual items were summed to obtain a total scale score ranging from 10 to 42, with higher scores indicating greater morningness and lower scores indicating greater eveningness.
Weekly self-reported assessments
Participants completed six weekly assessments of mood, sleep, and stress beginning on Day 1 of the first menstrual cycle following enrollment.
Mood report.
The Center for Epidemiologic Studies Depression Scale for Children (CES-DC) (Roberts et al., Reference Roberts, Andrews, Lewinsohn and Hops1990) is a well-validated 20-item self-report assessing depressive symptoms in adolescents (scale: 0 (“Not At All”) to 3 (“A Lot”), range: 0 to 60). Higher scores indicate greater depressive symptoms, and a score of 15 indicates clinically significant depressive symptoms and likely diagnosis of MDD (Fendrich et al., Reference Fendrich, Weissman and Warner1990) – though it is important to note that the CESD-C is a screening tool and not validated as a diagnostic measure.
Sleep report.
The PROMIS Sleep Disturbance Short Form 4a and Sleep-Related Impairment Short Form 4a (Bevans et al., Reference Bevans, Meltzer, De La Motte, Kratchman, Viél and Forrest2019; Forrest et al., Reference Forrest, Meltzer, Marcus, de la Motte, Kratchman, Buysse, Pilkonis, Becker and Bevans2018) assessed self-reported perceptions of sleep disturbance and daytime sleepiness, respectively, (scale: 1 (“not at all”) to 5 (“very much”), range: 4 to 20).
Perceived stress.
The Perceived Stress Scale (PSS) (Cohen et al., Reference Cohen, Kamarck and Mermelstein1983) is a 10-item instrument assessing stress perception (scale: 0 (“never”) to 4 (“very often”), range: 0 to 40). The PSS consists of two subscales, one assessing perceived helplessness (negatively worded items) and the other assessing perceived self-efficacy (positively worded items) (Harris et al., Reference Harris, Gaffey, Schwartz, Krantz and Burg2023).
Daily self-reported assessments
Participants completed twice-daily sleep diaries (sleep–wake diary), where they self-reported bedtime, waketime, and variables that could impact sleep, such as caffeine use, exercise, social media use, stress, and daytime naps (Manber et al., Reference Manber, Bootzin, Acebo and Carskadon1996).
Actigraphic sleep assessment
Following completion of the six-week main protocol (during which the above assessments were collected), participants wore an actigraphy wristwatch (Philips Respironics Actiwatch Spectrum Plus) for eight consecutive days to provide measures of sleep/wake indices, physical activity, and light exposure. The eight-day sampling period began one week following the start of a confirmed menstrual period, and allowed for inclusion of at least one full weekend in the SRI calculation. Participants were instructed to press the event marker button on the Actiwatch device upon waking each morning and just before falling asleep each night. Participants only removed the watch for water-related activities (e.g., bathing).
Actigraphy analysis and sleep characteristics.
Spectrograms were generated from the actigraphy data, which detailed activity, light levels, event markers, off-wrist periods, and sleep periods for each day (Philips Respironics, 2014). Rest intervals were confirmed by study personnel using a well-established procedure based on (a) self-reported bedtime/waketime, (b) activity drop/rise, (c) Actiwatch event markers, and (d) ambient light data (Dean et al., Reference Dean, Goldberger, Mueller, Kim, Rueschman, Mobley, Sahoo, Jayapandian, Cui, Morrical, Surovec, Zhang and Redline2016). Individual 30-second epochs coded as either wake (1) or sleep (0) were generated using the Oakley algorithm (Oakley, Reference Oakley1997). Epochs outside of the main rest intervals that could not be determined automatically (referred to as excluded periods) were manually recoded as either wake or sleep, using the following procedure: (1) if the duration of the excluded period was less than 15 min, it was recoded as wake; (2) if the duration of the excluded period was greater than 15 min, the available self-report data regarding naps was used. When there was no evidence of naps, all excluded periods were recoded as wake. When a nap was reported, epochs were recoded as sleep beginning at the reported start time of the nap and continuing for the maximum reported duration of the nap (nap durations were reported as ranges [e.g., 15–30 mins]). Excluded epochs falling outside of the reported nap time were recoded as wake.
Cleaned sleep/wake epochs from the actigraphy files were used to calculate the SRI in Python using previously described methods (Lunsford-Avery et al., Reference Lunsford-Avery, Engelhard, Navar and Kollins2018). Other sleep markers were extrapolated from these epochs as well, including total sleep time (difference between waketime and bedtime, waketime-bedtime), sleep efficiency (percentage of time spent asleep while in bed), wake/bedtime variability (standard deviation of wake/bedtimes), wakefulness after sleep onset (WASO) (time spent awake between the bedtime and morning waketime), and sleep midpoint (midpoint between bedtime and waketime).
Circadian rhythm measurement
Cortisol and 6-sulphatoxymelatonin.
On Days 3–6 of the sleep study (Days 9–12 of Cycle 2), participants provided four urine collections daily on filter paper: immediately upon waking (T1), 30 min after waking (T2), before dinner (T3), and before bed (T4). Participants were instructed to saturate the strip, record the exact date and time of collection, let the sample dry completely, and store samples in their home freezers until study personnel picked them up. Samples were then stored in the lab and kept frozen at -80°C until they were sent to ZRT Laboratory for analysis (ZRT Laboratory, Oregon, USA). Liquid chromatography-tandem mass spectrometry (LC-MS/MS) ensured the most sensitive and accurate quantification of urinary free cortisol and aMT6s. Average inter- and intra-assay precision for cortisol was 17.72% and 16.29%, respectively, and the reportable range was 0.7 to 275ng/mL. Average inter- and intra-assay precision for aMT6s was 8.83% and 6.26%, respectively, and the reportable range was 0.5 to 1588 ng/mL. Creatinine levels were used to correct values for hydration status.
Analysis plan
Data coding and preparation
Sixty-six adolescents (ages 11–14, M = 12.8, SD = 0.96) provided data for the present analyses. Fifty-nine participants had acceptable sleep actigraphy (minimum six days total with at least one weekend day), and 62 participants provided cortisol/aMT6s samples for at least one day. Of the 62 participants with circadian rhythm data, 79% (N = 49) of participants collected the first sample within 30 min of verified waketime and the second sample within 45 min of the first to calculate the cortisol awakening response. Seven participants did not have actigraphy or self-reported waketimes to verify accurate collection times, and two participants had samples collected within 15 min of the verified waketime without a second sample within 45 min. Across all participants, 349 days of actigraphy data were collected with an average difference between actigraphy-measured and self-reported bedtimes and waketimes of 36.14 min (SD = 45.18) and 26.42 min (SD = 36.87), respectively. For CESD, missing values on individual questionnaires were imputed using mean imputation. Measure completion was 83% (327/396) across all study participants. Average cortisol and aMT6s levels at each time point are shown in Supp. Figure 1. Cronbach’s alpha was calculated as a measure of internal consistency reliability for relevant measures.
Diurnal cortisol/aMT6s
The collection time for each sample at T1 was compared to the validated waketime for the given day. Samples collected more than 30 min after the validated waketime were excluded from analysis. We calculated the diurnal slope (regression of values across the day onto hours since awakening, without T2) and the awakening response (CAR: difference between T2 and T1, T2-T1) (Ross et al., Reference Ross, Murphy, Adam, Chen and Miller2014) for cortisol. Samples were excluded from the CAR analysis if the time between T1 and T2 was greater than 45 min. For aMT6s, the level at T1 represented an estimate of overnight levels, as has been demonstrated previously (Arendt, Reference Arendt2006).
Statistics
A linear mixed effects model (PROC MIXED, SAS OnDemand for Academics) was used to examine relationships between depressive symptoms and SRI. Covariates included sleep duration, social jetlag, and age at menarche. Intercept was included as a random effect. A second linear mixed effects model assessed SRI-chronotype interactions using the same fixed effects as the direct model plus additional fixed effects of chronotype, self-reported stress, and the SRI*chronotype term. Restricted maximum likelihood estimation was used to estimate variance components. No additional imputations were made for missing data, as mixed models appropriately handle dependent variable data missing at random (Powney et al., Reference Powney, Williamson, Kirkham and Kolamunnage-Dona2014). Linear regression models were used to examine effects of cortisol (diurnal slope and CAR) and morning a6MTS levels on SRI.
All variables were treated as continuous for the primary analyses. For all models, assumptions of linearity, normality, and homoscedasticity were checked. The significance level was set at α =.05. False discovery rate was used to correct p values for multiple comparisons. Pearson correlations between sleep regularity with other sleep characteristics, including bedtime variability, total sleep time, sleep efficiency, sleep midpoint, WASO, and self-reported sleep impairment were conducted as exploratory analyses.
Power analysis.
With 66 participants, and α =.05, G*power (version 3.1) confirmed that we had 90% power to detect medium main effects (f2 = 0.15) of sleep regularity on affective symptoms and diurnal rhythms (cortisol, aMT6s), and 80% power to detect medium interaction effects of SRI with chronotype and on affective symptoms.
Results
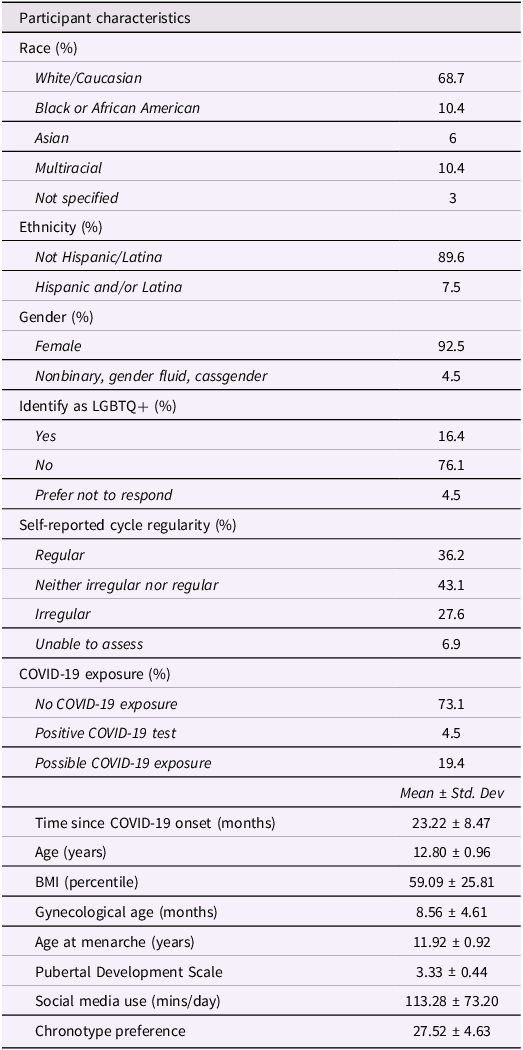
Demographics and participant characteristics
Demographic and participant characteristics are presented in Table 1. Participants were predominantly White (69%), not Hispanic/Latina (90%), identified as female gender (90%), and were on average 8.6 months post-menarche. Average participant BMI percentile was 59.09 (SD = 25.81). Data were collected between March 2020 and December 2023, with an average of 23 months since the beginning of the COVID-19 pandemic at enrollment.
Demographics

Measure reliability
Reliability analysis demonstrated acceptable to good reliability across all measures, including CESD-C (α = .87), SuperScience Morningness/Eveningness (α = .78), PROMIS Sleep Disturbance (α = .82), PROMIS Sleep Impairment (α = .90), and PSS (α = .80).
SRI and depressive symptoms
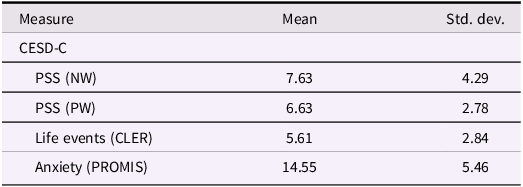
Summary statistics for mood measures are depicted in Table 2. In our direct model, reduced sleep regularity (SRI) was a significant predictor of weekly CES-DC depressive symptoms (F [1,276] = 9.74, p = < .002). No covariates in the direct model were significant predictors of depressive symptoms (p > .05).
Descriptive statistics for assessments
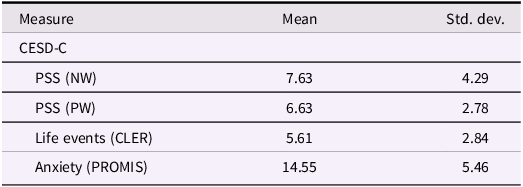
CESD-C = Center for Epidemiologic Studies Depression Scale – Child Version; PSS = perceived stress scale, negative worded (NW = distress subscale), (PW = coping subscale); Life events = number of stressful life events on the Coddington Life Events Record (CLER); PROMIS =Patient-Reported Outcomes Measurement Information System.
Chronotype as a moderator of SRI-affect
The inclusion of stress, chronotype, and the stress × chronotype interaction increased model fit (AIC decreased from 2043.8 to 2001.2 compared to direct model). In terms of individual predictors, SRI (F [1,273] = 18.65, p = < .0001), chronotype (F [1,273] = 21.13, p = < .0001), and the interaction between SRI and chronotype (F [1,273] = 16.54, p = < .0001) were all significant predictors of CESD scores in the interaction model (Figure 2). Main effects of sleep duration (F [1,273] = 6.25, p = .01) and life stress (F [1,273] = 22.82, p = < .0001) were also observed.
Depression vs. Sleep Regularity Index. Sleep regularity index reflects the probability of an individual being in the same wake/sleep state at any two timepoints 24 hours apart. Higher sleep regularity was associated with lower depressive symptoms.

Sleep regularity and diurnal cortisol/aMT6s
Linear regression models testing the effects of the diurnal cortisol curve, cortisol awakening response, and aMT6s at timepoint one demonstrated no significant effects of endocrine circadian function on SRI (p > .05).
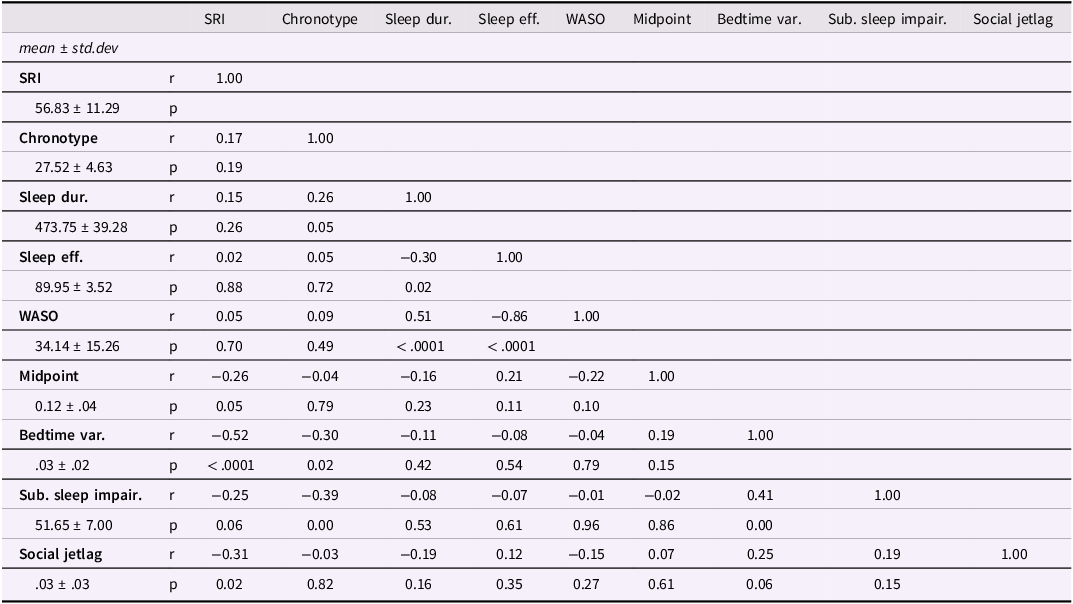
Sleep regularity versus other sleep characteristics
See Table 3. Weak correlations were observed between SRI and TST (r = 0.15, p = .47), sleep efficiency (r = .02, p = .90), and WASO (r = .05, p = .87). Weak negative correlations were observed between SRI and sleep midpoint (r = −.26, p = .17), SRI and subjective sleep impairment (r = −.25, p = .18), and SRI and social jetlag (r = −.31, p = .10) (the absolute value of the average difference between weekday and weekend sleep midpoint (Sun et al., Reference Sun, Yang, Yu, He, Luo, Zhang, Chen and Tung2025)). A moderate negative correlation was observed between SRI and bedtime variability (r = −.52, p = .001).
Correlation matrix of sleep measures

Note. r = Pearson correlation coefficient; SRI = sleep regularity index; Chronotype = Chronotype preference (Superscience morningness/eveningness); Sleep Dur = sleep duration; Sleep Eff = sleep efficiency, total time asleep/total time in bed; WASO = wake after sleep onset; Bedtime Var = standard deviation in actigraphic bedtimes; Sub. Sleep Impair = subjective sleep impairment, sleep diary; Social Jetlag = absolute value of the difference between average weekend sleep midpoints and average weekday sleep midpoints.
Discussion
Summary of present findings
The current study examined sleep regularity in relation to mood and circadian rhythms in a cohort of early post-menarchal female adolescents. In line with previous findings, sleep regularity was shown to be a predictor of depressive symptoms. Unlike prior findings, however, greater sleep duration predicted greater depressive symptoms across the study period in our interaction model.
Sleep regularity/duration and depressive symptoms
This study contributes to a growing body of research showing that sleep regularity plays a key role in adolescents’ emotional health. That this effect is present in early post-menarchal girls highlights the therapeutic potential of sleep regularity both to target acute mood dysregulation in the context of immature endocrine function around menarche, as well as to establish healthy trajectories for affective function to adolescence and adulthood. Consistent with other work (Phillips et al., Reference Phillips, Clerx, O’Brien, Sano, Barger, Picard, Lockley, Klerman and Czeisler2017), sleep regularity was only weakly correlated with sleep duration, supporting the idea that sleep regularity and sleep duration should be considered as independent factors contributing to mood.
The longer sleep duration that was associated with higher depressive symptoms in our interaction model was likely a result of the eight days of sleep data being collected after the six-week mood assessment period, meaning we could not directly evaluate how changes in sleep duration related to mood at the time of assessment (i.e., sleep duration may have been different during the six-week mood assessment period). Additionally, adverse mood effects have been reported following experimental sleep deprivation (Johri et al., Reference Johri, Pillai, Kulkarni and Balkrishnan2025), which utilizes more severe restriction than participants experienced in this study (the average sleep duration in this study was nearly 8 h).
Sleep regularity, chronotype, and depressive symptoms
Regarding chronotype, eveningness preference was shown to be a risk factor for more severe depressive symptoms, consistent with prior studies (Murray et al., Reference Murray, Phillips, Magee, Sletten, Gordon, Lovato, Bei, Bartlett, Kennaway, Lack, Grunstein, Lockley, Rajaratnam, Armstrong, Chohan, Djavadkhani, Dodds, Gunaratnam, Hardy and Yu2019). Additionally, an interaction between sleep regularity and chronotype were observed, such that eveningness and reduced sleep regularity together predicted higher depression. Dividing participants into high and low sleep regularity and chronotype at their respective medians suggests that depression scores were worse predominately for participants in the bottom 50th percentile for both sleep regularity and chronotype (Figure 3), meaning that female adolescents with an eveningness chronotype and sleep irregularity may stand to gain the most from a hypothetical treatment targeting SRI (though these findings should be interpreted with caution given the limitations of dichotomizing SRI in this manner). Given moderate inverse correlations between SRI and bedtime variability, increasing consistency of bedtimes may be one approach to improving sleep irregularity in “night owls.” Previous research has also suggested that earlier bedtimes in adolescents with delayed circadian phase may help normalize sleep function (Crowley et al., Reference Crowley, Velez, Killen, Cvengros, Fogg and Eastman2023), though our data demonstrated only weak correlations between SRI and sleep midpoint. A more individualized approach to parsing the effects of chronotype and behaviors that interfere with sleep timing might involve consideration of sleep phase angle – the time between the onset of evening melatonin secretion, which varies according chronotype (Taillard et al., Reference Taillard, Sagaspe, Philip and Bioulac2021), and bedtime.
Interaction between Sleep Regularity and Chronotype in Predicting Depression. A. Interaction between sleep regularity index and chronotype in predicting depression scores. For visualization purposes, chronotype preference was categorized as “evening” or “morning” based on a median split of the continuous chronotype scores that were used in the primary analysis. B. Depression vs Sleep Regularity Index (categorical), grouped by chronotype *P<.005. For visualization purposes, categorical values for chronotype preference (morning/evening) and sleep regularity index (high/low) were obtained using a median split of their respective continuous variables (primary analysis used all variables in continuous form).

Sleep regularity and circadian function
No significant association was observed for sleep regularity and steepness of diurnal cortisol slopes, cortisol awakening response, or overnight melatonin levels. Changes in cortisol dynamics (reduced CAR, flattened diurnal slopes) are thought to represent nonspecific stress-related dysregulation of endogenous circadian pacemakers (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017) and have been linked to a host of mental and physical health conditions, including depression (Doane et al., Reference Doane, Mineka, Zinbarg, Craske, Griffith and Adam2013). Stress-induced alterations in HPA-axis function can disrupt circadian systems (Buckley & Schatzberg, Reference Buckley and Schatzberg2005), contributing to mood dysregulation – further research is needed to clarify how sleep regularity relates by stress and stress-induced alterations in circadian rhythms.
Sleep regularity and other sleep characteristics
Sleep regularity demonstrated a moderate inverse correlation with bedtime variability (about 27% of the variance), i.e., the consistency in participants’ bedtimes, and weak correlations were observed between SRI and sleep midpoint, sleep efficiency, and social jetlag, suggesting that other factors (behavioral, environmental, endogenous, etc.) contribute to sleep regularity, or that SRI reflects the combined effects of multiple influences.
Strengths and limitations
The present study expands on prior work demonstrating relationships between sleep regularity, circadian timing, and mood (Lunsford-Avery et al., Reference Lunsford-Avery, Wang, Kollins, Chung, Keller and Engelhard2022; Murray et al., Reference Murray, Phillips, Magee, Sletten, Gordon, Lovato, Bei, Bartlett, Kennaway, Lack, Grunstein, Lockley, Rajaratnam, Armstrong, Chohan, Djavadkhani, Dodds, Gunaratnam, Hardy and Yu2019; Walsh et al., Reference Walsh, Maddox, Smith, Olino, Zee, Nusslock and Alloy2025). Study strengths include the use of actigraphy as an objective measure of sleep, frequent assessment of mood and self-reported sleep characteristics, and the sampling of cortisol and melatonin (a6MTS) as a measure of circadian function. Despite the strengths discussed above, there are several limitations to address. Limitations include the relatively short study period without simultaneous ovarian steroid collection to confirm menstrual cycle phase, and a hormone sampling protocol with (a) a relatively small number of daily collections and (b) variability in collection times, due to home/participant-driven collection in adolescent participants. While all participants had a confirmed menstrual period one week prior to starting the study, reproductive hormone levels may have varied based on whether the cycle during which data was collected was ovulatory or anovulatory, which was not assessed in this study. In general, counting methods based on last menstrual period can be adequate for menstrual staging in regularly-cycling adults (Gloe et al., Reference Gloe, Russman Block, Klump, Beltz and Moser2023), but are less useful in early post-menarchal samples given the high rates of anovulatory cycles; ovulation and/or hormone testing should be used when possible in this population. Recruitment was restricted to female adolescents to examine biobehavioral factors that may contribute to the increased prevalence of affective illness in females starting at puberty, which limited the generalizability of the results. Sex differences in sleep regularity, circadian function, and depressive symptoms will need to be examined in future studies. Additionally, though we included proximal measures of circadian rhythms in this study (cortisol, sleep midpoint, chronotype), direct assessment of the circadian clock, which requires serial melatonin assessment to establish dim-light melatonin onset, was not performed. Despite not employing the gold standard techniques for sleep assessment (polysomnography) or hormone sampling (venipuncture in a lab setting, allowing for greater control over sampling times and the ability to define hormone dynamics more precisely), we were nonetheless able to obtain high-quality sleep and endocrine data with a nonburdensome alternative with superior feasibility and compliance in adolescent participants. We intend to refine and expand this protocol for use in future experimental research.
Future directions
Future research considerations include the use of experimental designs to elucidate causality, as well as testing potential treatments targeting sleep regularity. For instance, experimental paradigms for social stress (e.g., the Trier Social Stress Test), sleep deprivation, and circadian phase manipulation can be employed alone or in combination to assess effects on the HPA axis, sleep regularity, and affect. Further, combining ovarian steroid levels, circadian function, and sleep actigraphy across an adolescent menstrual cycle would offer important insight on the biobehavioral indices of depression risk in females. Samples that include a broader range of pubertal stages, as well as male participants, can increase the generalizability of findings. Denser hormone sampling protocols and the inclusion of melatonin would allow for more detailed assessment of circadian functioning, including cortisol awakening responses, melatonin curves, and phase angle, which can subsequently be examined in relation to sleep regularity. Longitudinal assessments can provide insight into the kinetics of mood, sleep regularity, and HPA-axis function, and may help us identify early stages of depressive illness that are amenable to sleep-based interventions.
From a clinical standpoint, the emergence of sleep regularity as a modifiable risk factor for affective function offers new possibilities for predicting and treating adolescent mental illness. Smartphone-based sleep tracking technologies may allow practitioners to roughly estimate sleep regularity in an office setting, providing insight into behavioral issues that adolescent patients may not be able to articulate precisely. Sleep regularity data may also help predict the onset of mood episodes and can be considered in addition to standard queries about sleep duration and subjective sleep impairment. From an intervention perspective, no treatments that target sleep regularity currently exist. However, interventions that address circadian timing, such as the Transdiagnostic Intervention for Sleep and Circadian Intervention, have been shown to have beneficial effects on mental health in adolescents with delayed circadian phase (Asarnow et al., Reference Asarnow, Soehner, Dolsen, Dong and Harvey2023); given the purported relationship between circadian timing and sleep regularity, this and other chronotherapeutic interventions (such as bright light therapy) may stand to improve sleep regularity as well.
Conclusion
The present study extends prior findings demonstrating that sleep regularity is an important risk factor for depressive symptoms in early post-menarchal female adolescents. Findings suggest that sleep regularity interacts with chronotype to influence mood outcomes. No statistically significant relationships were observed between sleep regularity and endocrine circadian rhythms, and future research may consider more intensive hormone sampling approaches. Interventions that target sleep regularity during peripuberty may directly improve adolescent depression, and positively impact long-term mental health trajectories.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0954579426101242.
Acknowledgments
The Python syntax for computing the Sleep Regularity Index was generously provided by Dr Matthew Engelhard. Data from this study was presented at the Society for Biological Psychiatry’s annual meeting, San Diego, CA, 2023.
Author contributions
We, the undersigned authors, declare that we have each made substantial contributions to the conception and design of the work, the acquisition of data, or the analysis and interpretation of data. Each author has participated in drafting or revising the manuscript critically for important intellectual content and has approved the final version to be published. We affirm that we have disclosed any potential conflicts of interest and have adhered to ethical guidelines in conducting and reporting this research.
Kayla Jensen: Data collection and curation, manuscript preparation.
Chris Sikes-Keilp: Formal analysis, visualization, manuscript preparation.
Elizabeth Wilson: Manuscript preparation.
Jessica Lunsford Avery: Methodology, resources, manuscript review and editing.
Elizabeth Andersen: Conceptualization, methodology, resources, manuscript preparation, supervision
All authors have agreed to be listed in this order and are accountable for the accuracy and integrity of the work.
Funding statement
This study was supported by the National Institute of Mental Health, the National Institute of Health, and the Foundation of Hope for Research and Treatment of Mental Illness.
Competing interests
The authors declare no conflicts of interest.
Ethical standards
This research was approved by the UNC Biomedical Institutional Review Board, date of approval 11/30/2020, reference # 20-2,816.
Pre-registration
Analyses were not pre-registered.
Data availability
The data and code necessary to reproduce the analyses presented here are publicly accessible, as are the materials necessary to attempt to replicate the findings. Data, code, and materials for this research are available at the following URL: https://osf.io/nmu5t/?view_only=a139ceca23a943be8fd42255952bb7d6.
AI
No AI tools were used in the preparation of this manuscript.
Patient consent statement
Parent/guardian consent was obtained in addition to participant assent for all participants.