Introduction
B. longum ssp. infantis (B. infantis) is an early-life colonizer of the infant gut that is well adapted to metabolize human milk oligosaccharides (HMOs), a group of complex indigestible sugars present in human milk (Underwood et al., Reference Underwood, German, Lebrilla and Mills2015; Frese et al., Reference Frese, Hutton, Contreras, Shaw, Palumbo, Casaburi, Xu, Davis, Lebrilla, Henrick, Freeman, Barile, German, Mills, Smilowitz and Underwood2017). HMO metabolism by B. infantis produces lactic and acetic acids and other short-chain fatty acids that contribute to a lower pH in the gut lumen, inhibit proliferation of potential pathogens, and may influence immunoregulation and immune development (Fukuda et al., Reference Fukuda, Toh, Hase, Oshima, Nakanishi, Yoshimura, Tobe, Clarke, Topping, Suzuki, Taylor, Itoh, Kikuchi, Morita, Hattori and Ohno2011; Frese et al., Reference Frese, Hutton, Contreras, Shaw, Palumbo, Casaburi, Xu, Davis, Lebrilla, Henrick, Freeman, Barile, German, Mills, Smilowitz and Underwood2017; Huda et al., Reference Huda, Ahmad, Alam, Khanam, Kalanetra, Taft, Raqib, Underwood, Mills and Stephensen2019; Henrick et al., Reference Henrick, Rodriguez, Lakshmikanth, Pou, Henckel, Arzoomand, Olin, Wang, Mikes, Tan, Chen, Ehrlich, Bernhardsson, Mugabo, Ambrosiani, Gustafsson, Chew, Brown, Prambs, Bohlin, Mitchell, Underwood, Smilowitz, German, Frese, Brodin and Brodin2021; Laursen et al., Reference Laursen, Sakanaka, Von Burg, Mörbe, Andersen, Moll, Pekmez, Rivollier, Michaelsen, Mølgaard, Lind, Dragsted, Katayama, Frandsen, Vinggaard, Bahl, Brix, Agace, Licht and Roager2021). B. infantis colonization is hypothesized to improve infant health, including linear and ponderal growth outcomes, possibly by reducing the risk of common morbidities such as diarrhoeal illness, and through effects of short-chain fatty acids on host metabolism, including gluconeogenesis (Boets et al., Reference Boets, Gomand, Deroover, Preston, Vermeulen, De Preter, Hamer, Van Den Mooter, De Vuyst, Courtin, Annaert, Delcour and Verbeke2017).
However, there is a lack of epidemiological evidence in support of an effect of early postnatal B. infantis colonization on infant growth. Few studies have been conducted in low- and middle-income countries (LMICs); in one study of infants in Bangladesh, there was a positive association between B. infantis and infant weight-for-age z-score (WAZ) and weight-for-length z-score (WLZ) in the first year after birth (Vatanen et al., Reference Vatanen, Ang, Siegwald, Sarker, Le Roy, Duboux, Delannoy-Bruno, Ngom-Bru, Boulange, Strazar, Avila-Pacheco, Deik, Pierce, Bullock, Dennis, Sultana, Sayed, Rahman, Ahmed, Modesto, Mattarelli, Clish, Vlamakis, Plichta, Sakwinska and Xavier2022), but in a multivariable-adjusted analysis of cohorts in Bangladesh, Tanzania, and Pakistan, there were no associations between neonatal B. infantis abundance and length-for-age z-score (LAZ) or WAZ at 24 months of age (Colston et al., Reference Colston, Taniuchi, Ahmed, Ferdousi, Kabir, Mduma, Nshama, Iqbal, Haque, Ahmed, Ali Bhutta, Kosek and Platts-Mills2022). A key consideration in the interpretation of epidemiological studies is the wide global variation in B. infantis abundance – in general, it is rare or even absent among infants in high-income countries but frequently detected in LMICs (Lawley et al., Reference Lawley, Otal, Moloney-Geany, Diana, Houghton, Heath, Taylor and Tannock2019; Taft et al., Reference Taft, Lewis, Nguyen, Ho, Masarweh, Dunne-Castagna, Tancredi, Huda, Stephensen, Hinde, Von Mutius, Kirjavainen, Dalphin, Lauener, Riedler, Smilowitz, German, Morrow and Mills2022; Shao et al., Reference Shao, Wang, Gichuki, Stares, Rozday, Kumar, Browne, Dawson, Njunge, Tigoi, Ngao, Chisti, Singa, Kariuki, Diallo, Saleem, Ali, Mupere, Mbale, Tickell, Voskuijl, Lancioni, Bandsma, Ahmed, Walson, Berkley and Lawley2026), settings where infant growth faltering is most common (Victora et al., Reference Victora, Christian, Vidaletti, Gatica-Domínguez, Menon and Black2021; UNICEF, 2023). Therefore, while B. infantis abundance might be found to explain between-individual variations in linear or ponderal growth within a study population; these associations may not explain why nearly all infants in LMICs grow more slowly than expected for their chronological age based on international growth standards (Roth et al., Reference Roth, Krishna, Leung, Shi, Bassani and Barros2017).
In a cohort of generally healthy and mostly term-born infants in Dhaka, Bangladesh, we previously found B. infantis was highly abundant in stool by 2 months of age and persisted to at least 6 months of age; however, it was generally absent in the immediate postnatal period and there was substantial variation in abundance in the first month (neonatal period) (Freitas et al., Reference Freitas, Li, Shawon, Qamar, Pell, Kabir, Oduaran, Puebla-Barragan, Bassani, O’Callaghan, Onuora, Loutet, Heasley, Starke, Mahmud, Hamer, Pullenayegum, Hossain, Siddiqui, Sherman, Shah, Gaffar, Sultana, Morris, Ahmed, Haque, Sarker, Roth and Roth2025). To explore the potential role of postnatal B. infantis colonization in modulating growth in early infancy, we examined the associations between B. infantis abundance in the neonatal period and anthropometric outcomes up to 6 months of age in the same cohort. In further analyses, we estimated associations of B. infantis with stool pH and infant morbidity (diarrhoea and hospitalizations), considering that such relationships might represent pathways by which B. infantis could influence infant growth.
Methods
Study design and participants
This secondary-use study was based on data from the Synbiotics for the Early Prevention of Severe Infections in Infants (SEPSiS) observational cohort study (ClinicalTrials.gov ID: NCT04012190). Approval for the original data collection was provided by The Hospital for Sick Children Research Ethics Board (REB# 1000063899) and the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b) Research Ethics Committee (PR-19045). Approval for this secondary use study was obtained from The Hospital for Sick Children (REB#1000082019). Further details on the design and implementation of the SEPSiS observational cohort study have been described elsewhere (Freitas et al., Reference Freitas, Li, Shawon, Qamar, Pell, Kabir, Oduaran, Puebla-Barragan, Bassani, O’Callaghan, Onuora, Loutet, Heasley, Starke, Mahmud, Hamer, Pullenayegum, Hossain, Siddiqui, Sherman, Shah, Gaffar, Sultana, Morris, Ahmed, Haque, Sarker, Roth and Roth2025; Fung et al., Reference Fung, Heasley, Pell, Bassani, Shah, Morris, Hamer, Islam, Mahmud, Pullenayegum, Saha, Haque, Hossain, Chen, Emdin, O’Callaghan, Loutet, Sultana, Billah, Gaffar, Karim, Sayed, Yeasmin, Hoque, Ahmed, Sarker and Roth2025). Briefly, between November 2020 and February 2022, infants were enrolled at two government healthcare facilities in Dhaka, Bangladesh: Maternal and Child Health Training Institute and the Mohammadpur Fertility Services and Training Centre. Infants were eligible for enrolment if they were delivered at a study hospital, aged zero (day of birth) to 4 days old, orally feeding at the time of screening, and their caregivers were planning to maintain residence in the study catchment area until the infant reached at least 60 days of age. Infants were ineligible for enrolment if they weighed <1500 g at birth, had a major congenital anomaly of the gastrointestinal tract, death or major surgery were likely within the first week after birth, were on mechanical ventilation or cardiac support, were receiving or prescribed parenteral antibiotics, were enrolled in an ongoing clinical trial involving the administration of probiotics and/or prebiotics, resided in the same household as another infant under 60 days of age (excluding twins), prenatal or postpartum/postnatal non-dietary probiotic supplements were used by the infant’s mother or provided directly to the infant, multiple gestation of the mother exceeded two, or the mother was diagnosed with HIV infection and/or history of receiving anti-retroviral drug(s) for a presumed HIV infection. For each model assessed, this study included all SEPSiS observational cohort participants who had relevant data.
Stool sample collection
Study personnel collected infant stool samples using standardized procedures according to one of three schedules (A, B, or C). Infants in schedules A and B had up to 11 routine stool samples collected (days 0 or 1, 3 or 4, 6 or 7, 10, 14, 21, 28, 35, 60, 90, and 180), whereas infants in schedule C had up to three routine stool samples collected (on the day of enrolment and twice more on randomly assigned visits up to day 60). Infants on schedules A and B were combined in this analysis because they used the same stool sample collection schedules, had a similar number of samples per infant, and had no other differences in the protocol that would influence the current analysis. In the hospital or the infant’s home, stool samples were collected using a wrapped plastic sheet or plastic wrap-lined diaper, then manually homogenized and aliquoted into appropriate vials. For samples collected by study workers (all schedule A/B samples, 46% of schedule C samples), aliquots intended for qPCR were placed in the vapour phase of liquid nitrogen within 20 minutes of defecation, and aliquots for stool pH measurements were stored in a cold box at 2–8 °C (never frozen). For schedule C samples that could not be collected directly by study workers (54% of samples), the infant’s caregiver was instructed to place the stool sample in a cold box (maintained at 2–8 °C) within 20 minutes of defecation until it was transported to the field office by study staff. All stool aliquots were placed into the vapour phase of liquid nitrogen within 6 hours of defecation, before transport to icddr,b for storage at ≤−70 °C until analysis.
Sample processing and laboratory analyses
Stool sample selection and quantification of B. infantis absolute abundance have been described in detail elsewhere (Freitas et al., Reference Freitas, Li, Shawon, Qamar, Pell, Kabir, Oduaran, Puebla-Barragan, Bassani, O’Callaghan, Onuora, Loutet, Heasley, Starke, Mahmud, Hamer, Pullenayegum, Hossain, Siddiqui, Sherman, Shah, Gaffar, Sultana, Morris, Ahmed, Haque, Sarker, Roth and Roth2025). Briefly, total DNA was extracted from 100 to 150 mg of stool using QIAamp Fast DNA Stool Mini Kit (QIAGEN, Germany). Total DNA mass was quantified using a Nanodrop 2000 spectrophotometer (Thermo Scientific, USA). B. infantis was quantified using qPCR with primer-probes specific to B. infantis (Lawley et al., Reference Lawley, Munro, Hughes, Hodgkinson, Prosser, Lowry, Zhou, Makrides, Gibson, Lay, Chew, Lee, Wong and Tannock2017). Cell counts were estimated using standard curves generated using pure cultures of B. infantis ATCC 15697, and absolute abundance was normalized to total DNA mass and expressed as log B. infantis cells per μg total DNA. For samples with undetectable B. infantis (i.e., did not amplify or had cell counts below the limit of detection of the qPCR assay), a value of 2.5 log cells/μg DNA was imputed for the absolute abundance. This value corresponds to the median of half the limit of detection normalized to each undetected sample DNA mass.
Aliquots for measuring stool pH were brought to room temperature before mixing 1 g of stool with deionized water to achieve a total sample volume of 10 mL. Stool pH was measured twice per sample using portable pH testers (Hanna, USA); if the difference between the two pH measurements was greater than 0.5 pH units, a third measurement was obtained. The mean of the two pH values (or the mean of the two closest values if three were available) was considered the pH measurement of each stool sample used in further analyses described below.
Clinical and anthropometric data collection
Routine study visits were conducted five times at the infant’s home or via phone call up to and including 14 days of age, then weekly until 60 days, then at 3 and 6 months. Study staff collected information about feeding practices (7-day recall), infant receipt of antibiotics (since last study visit), diarrhoeal episodes, and hospital admissions (Fung et al., Reference Fung, Heasley, Pell, Bassani, Shah, Morris, Hamer, Islam, Mahmud, Pullenayegum, Saha, Haque, Hossain, Chen, Emdin, O’Callaghan, Loutet, Sultana, Billah, Gaffar, Karim, Sayed, Yeasmin, Hoque, Ahmed, Sarker and Roth2025). Diarrhoeal episodes were based on a caregiver response of “yes” when asked if the infant had diarrhoea described as the “frequency of stool is too often, the quantity of stool is too much, and/or the consistency is too loose compared to the usual,” since the prior study visit or in the preceding 7 days, including the day of the encounter. Hospitalizations were verified and recorded by Study Medical Officers and were defined on the basis of admission to a hospital for any reason other than injury or elective surgery.
Anthropometric measurement methods were adapted from standardized procedures from the INTERGROWTH-21st study (Cheikh Ismail et al., Reference Cheikh Ismail, Knight, Bhutta and Chumlea2013). Trained study staff measured infant length and weight on the day of enrolment (days 0–4) and during scheduled home visits at postnatal ages 2, 3, and 6 months. Length was measured using a wooden length board to the nearest 1 mm (Shorr board, Weight and Measure LLC, USA), and weight was measured with a Seca 334 or 354 digital scale to the nearest 1 g (Seca, USA). Anthropometric measurements were obtained twice and averaged; if the difference between two measurements was greater than 7 mm or 50 g for length and weight, respectively, a third measurement was taken. If a third measurement was taken, the two closest measurements were used to calculate the average.
For measurements on days 0–2 of age, baseline LAZ and WAZ were derived using INTERGROWTH-21st Newborn Size growth curves (The International Fetal and Newborn Growth Consortium for the 21st Century, 2024a). For measurements obtained beyond 2 days of age, including at the 2, 3, and 6 months of age visits, the WHO Child Growth Standards were used to derive LAZ and WAZ for term infants (gestational age ≥ 259 days) (WHO, 2006), whereas for preterm infants (gestational age < 259 days) older than 2 days postnatal age, LAZ and WAZ were generated using the INTERGROWTH-21st Postnatal Growth for Preterm Infants growth curves (The International Fetal and Newborn Growth Consortium for the 21st Century, 2024b). Gestational age at birth was based on recalled last menstrual period (LMP) or prenatal ultrasound reports, following American College of Obstetricians and Gynecologists guidelines (American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, American Institute of Ultrasound in Medicine, Society for Maternal–Fetal Medicine, 2017). All WLZs were derived from the WHO Child Growth Standards as the index is independent of age; the baseline WLZ was based on concurrent weight and length measured at enrolment (days 0–4), but was not generated if the length was <45 cm. In instances of implausible values (infant length or weight decreasing over time or LAZ/WAZ ≥ ±6 or WLZ ≥ ±5), values and trajectories were examined independently by two reviewers to determine if any measurements should be removed. In total, 92 (4.8%) infants were flagged for investigation, among whom 51 had discrete length and 14 had weight measurements that were removed from analysis. For primary analyses, anthropometric z-scores were grouped according to the following age intervals: baseline (days 0–4), 2 months (days 59–75), 3 months (days 76–110), and 6 months (days 160–200).
Statistical analysis
Baseline characteristics were described using medians (with 25th and 75th centiles) for continuous data and counts (proportions) for categorical data. Scatterplots with LOESS curves were generated for all B. infantis-anthropometric associations investigated to visualize trends in bivariate relationships.
B. infantis absolute abundance in the first 28 days after birth was summarized at the infant level using the mean of each infant’s qPCR abundance data from all analysed samples collected from 0–28 days (neonatal period) and is referred to as ‘neonatal B. infantis absolute abundance’. Several summary measures of neonatal B. infantis absolute abundance were tested, and the mean was selected for primary analyses (see Supplementary Methods, Supplementary Table S1 and Supplementary Figures S1–S3). Associations between neonatal B. infantis absolute abundance and anthropometric indices at specified time points were estimated using linear regression models. All models with anthropometric outcomes included adjustment for the same anthropometric index measured at baseline. Sample sizes for the otherwise unadjusted models included only those infants with complete covariate data and who were included in the corresponding multivariable-adjusted analysis. Additional covariates in the adjusted regression models included: breastfeeding status assessed at or closest to age 28 days, infant sex, hospital enrolment site, mode of delivery, gestational age at birth, neonatal infant systemic antibiotics (ever/never), maternal BMI at enrolment, maternal education, maternal age, the number of infants within the household under 5 years of age, and household asset index quintile. The asset index was calculated for all infants enrolled in the SEPSiS observational cohort and a related clinical trial (Pell et al., Reference Pell, Qamar, Bassani, Heasley, Funk, Chen, Shawon, O’Callaghan, Pullenayegum, Hamer, Haque, Kabir, Ahmed, O’Kelly, Hossain, Khan, Loutet, Islam, Morris, Shah, Sherman, Sultana, Mahmud, Saha, Sarker and Roth2025) using principal component analysis of a checklist of household assets and scored based on the first principal component, which was then converted to quintiles. The primary outcome was LAZ, and the secondary outcomes were WAZ and WLZ.
In sensitivity analyses, we used the following alternative approaches to define neonatal B. infantis abundance: (1) area under the curve (of absolute abundance by age, as a continuous variable); (2) B. infantis absolute abundance expressed as a categorical dichotomous variable defined as undetectable (all stool samples within the first 28 days of age with undetectable B. infantis) or detectable (at least one stool sample with detectable B. infantis within the first 28 days of age). We also conducted sensitivity analyses in which we modified the period during which B. infantis absolute abundance was summarized for each infant (mean of available data for 0–14 or 0–60 days of age), and a cross-sectional sensitivity analysis using the single absolute abundance measurement that was closest in time to the 2-month anthropometric measurement (and collected within the interval of 59–75 days). We repeated the primary analysis in a subset of the cohort restricted to infants assigned to specimen collection schedules A/B. Lastly, we estimated the association between baseline anthropometric indices and neonatal B. infantis abundance as the outcome, and we performed the primary analyses without adjustment for the baseline measures.
To estimate the cross-sectional association between B. infantis absolute abundance and stool pH (using corresponding values generated for the same stool sample), we used unadjusted linear mixed effects regression models including all stool samples collected in the neonatal period. Using the same modelling approach, we also compared the stool pH of samples with detectable versus undetectable B. infantis.
For each infant, “neonatal stool pH” was defined as the median of their pH measurements among all stool samples collected from 0 to 28 days. Associations of neonatal stool pH with LAZ, WAZ, and WLZ at 2, 3, and 6 months were assessed using multivariable linear regression models with the same covariates as described above. In a sensitivity analysis, we conducted a cross-sectional analysis of the association of stool pH with anthropometric outcomes using the pH measurement of the single stool sample collected closest in time to each anthropometric measurement (and within the same age interval used for the corresponding anthropometric value).
To estimate associations of neonatal B. infantis abundance with diarrhoeal events from 29 to 60 days (2 months) of age and hospitalizations (excluding trauma/injury or elective surgeries) from 29 to 180 days (6 months) of age, infants were categorized as never/ever having had a diarrhoeal event or hospitalization during the defined period. Modified Poisson regression models (with robust standard errors) were used to estimate relative risks of diarrhoea and hospitalization by neonatal B. infantis abundance. In adjusted models, covariates included breastfeeding status assessed on or closest to 28 days of age, mode of delivery, gestational age, neonatal antibiotic exposure, maternal education, and household asset index quintile.
We considered the use of path analysis to examine if any associations between B. infantis abundance and anthropometric indices could be explained by mediating roles of stool pH or clinical events, but these analyses were not pursued due to null associations of B. infantis abundance with anthropometric indices.
The Holm method was used to account for multiple comparisons and maintain an overall α of 0.05 for each set of analyses (one set refers to all analyses presented within each table). All analyses were conducted using R statistical software (Version 4.4.1) (R Core Team, 2024). All statistical code used in analyses for this study was independently reviewed by a second data analyst.
Results
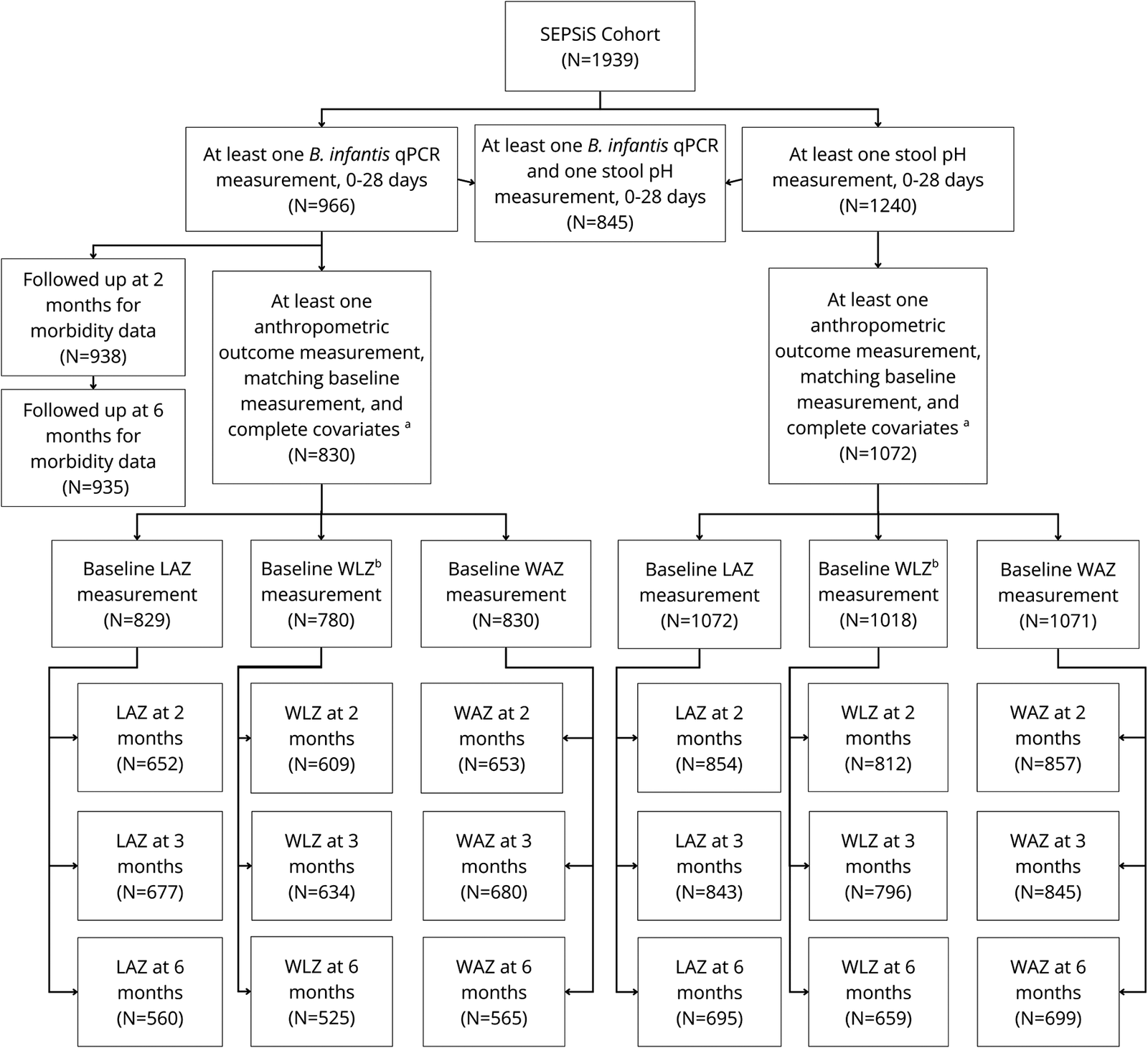
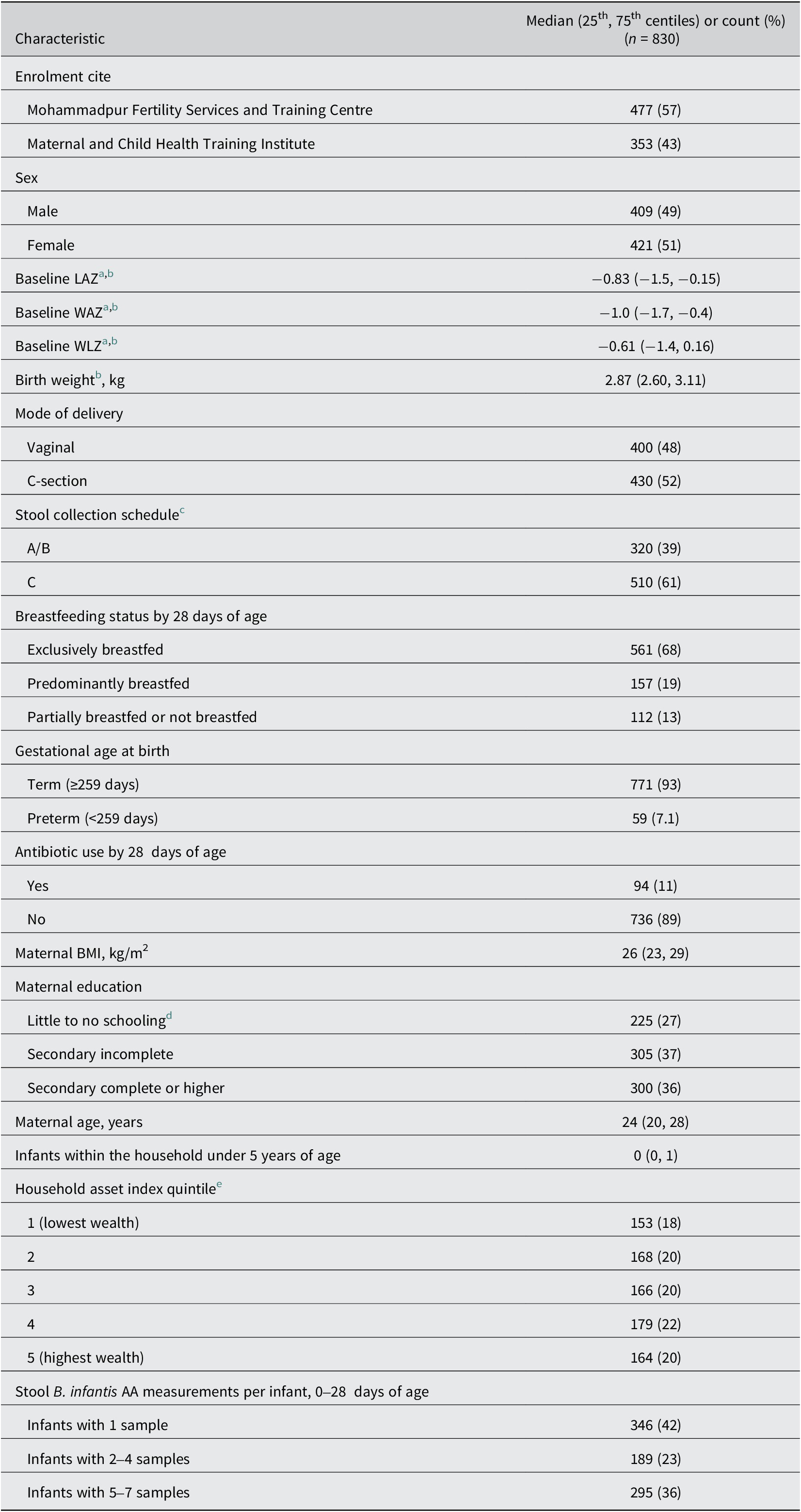
Out of 1939 infants enrolled in the SEPSiS observational cohort, 966 infants contributed 3903 stool samples in the neonatal period, of which 2982 (76%) underwent B. infantis qPCR testing. From 0 to 28 days of age, infants assigned to schedules A/B and C contributed a median (25th, 75th centiles) of 6.0 (2.0, 7.0) and 1.0 (1.0, 3.0) stool samples for qPCR analysis respectively. There were 830 infants (43% of 1939 enrolled) with at least one B. infantis qPCR measurement (0–28 days), at least one anthropometric outcome measure (2, 3, and 6 months) and a corresponding baseline anthropometric measure, and complete covariates, who were therefore included in at least one of the primary analyses of associations of B. infantis abundance and anthropometric outcomes (Figure 1). Eligible infants were predominantly born full-term (93%), via C-section (52%), and 68% were exclusively breastfed up to 28 days of age (Table 1, Supplementary Table S2 stratified by infant sex). There were 1072 infants (55%) with at least one stool pH measurement in the neonatal period, at least one anthropometric outcome measurement in one of the age intervals assessed, and complete covariates, who were therefore included in at least one of the primary analyses of associations of stool pH and anthropometric outcomes (Figure 1). There were 938 and 935 infants (48%) with a neonatal B. infantis absolute abundance measurement who also had morbidity-related data up to 2 months or hospitalization data up to 6 months of age respectively.
Study flow diagram. Numbers of infants from the SEPSiS observational study cohort who contributed to the primary and secondary analyses in this study. Note: aComplete covariates include: enrolment site, infant sex, breastfeeding status at 28 days of age (if available) or at age most proximal to 28 days, mode of delivery, gestational age at birth, infant antibiotic exposure by 28 days of age, maternal BMI, maternal education, maternal age, number of infants within the household under 5 years of age, and household asset index quintile. bSample sizes for WLZ were smaller than for the corresponding LAZ and WAZ datasets because WLZ was derived using the WHO child growth standards, whereby WLZ was not generated for some baseline measurements if length and weight were not measured at the same visit, or when length was <45 cm; however, the INTERGROWTH-21st standards enabled baseline LAZ to be generated for newborn lengths below 45 cm.

Baseline characteristics of infants included in primary analyses of the associations of neonatal B. infantis absolute abundance and anthropometric outcomes

Abbreviations: AA, absolute abundance; BMI, body mass index; LAZ, length-for-age z-score; qPCR, quantitative polymerase chain reaction; WAZ, weight-for-age z-score; WLZ, weight-for-length z-score.
a Mean and SD of anthropometric z-score distributions are shown in Supplementary Table S3.
b n < 830: baseline LAZ, n = 829; baseline WLZ, n = 780. Baseline WAZ (n = 830) and WLZ were calculated from weights measured at enrolment by study staff, whereas birthweights (n = 830) were extracted from the hospital records.
c Infants in schedule A/B had up to seven routine stool samples collected in the first month of age (days 0 or 1, 3 or 4, 6 or 7, 10, 14, 21, and 28). Infants in schedule C had up to three routine stool samples collected (day of enrolment and twice more on randomly assigned visits up to 60 days of age).
d Little to no schooling was defined as: no schooling at all; having completed qawmi madrasa but no other schooling; or, up to 5 years of schooling or alia madrasa.
e Quintiles of a score derived using the first component of principal component analysis from a checklist of household assets from all SEPSiS infants (observational cohort and associated trial). The higher quintile corresponds to higher asset ownership.
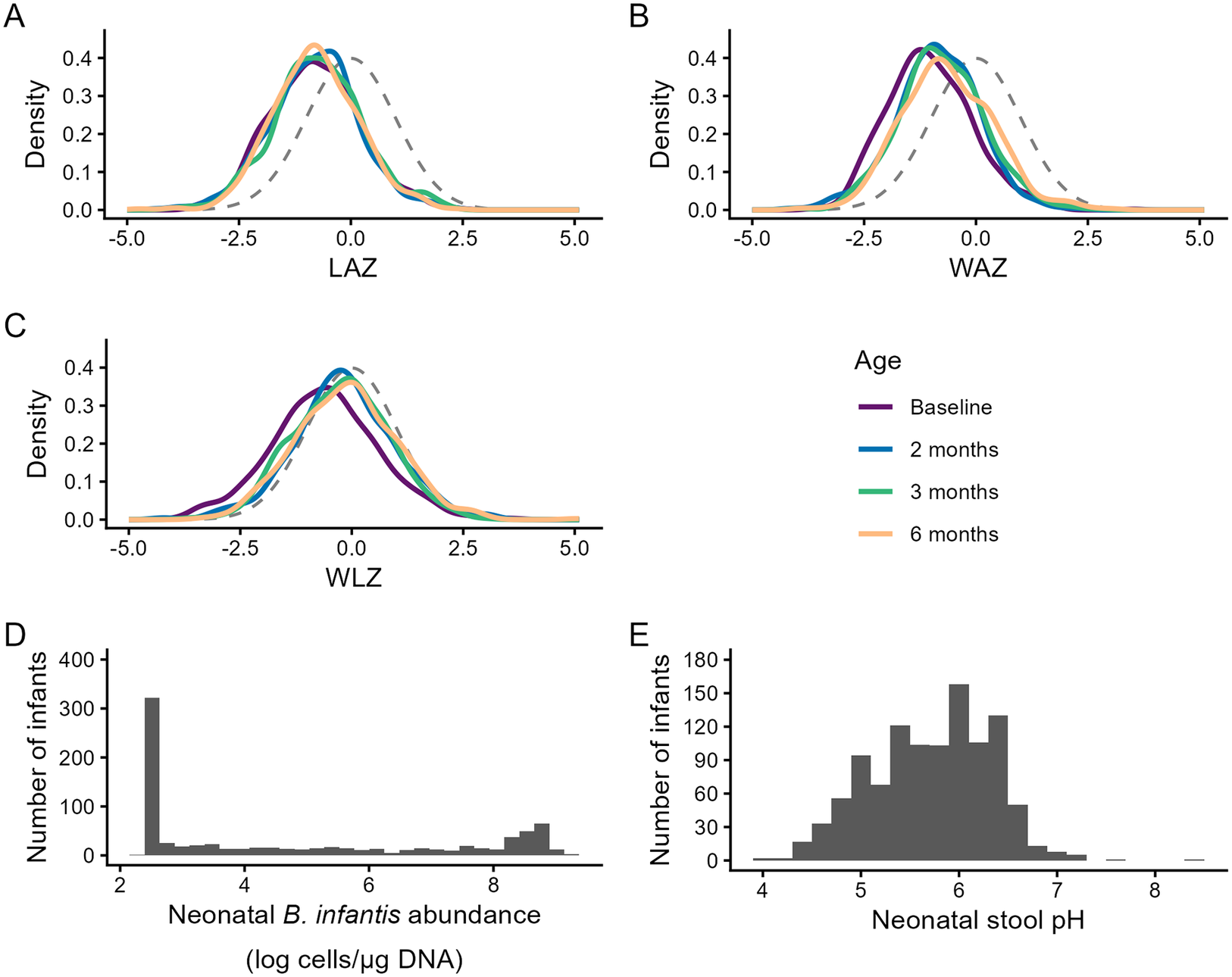
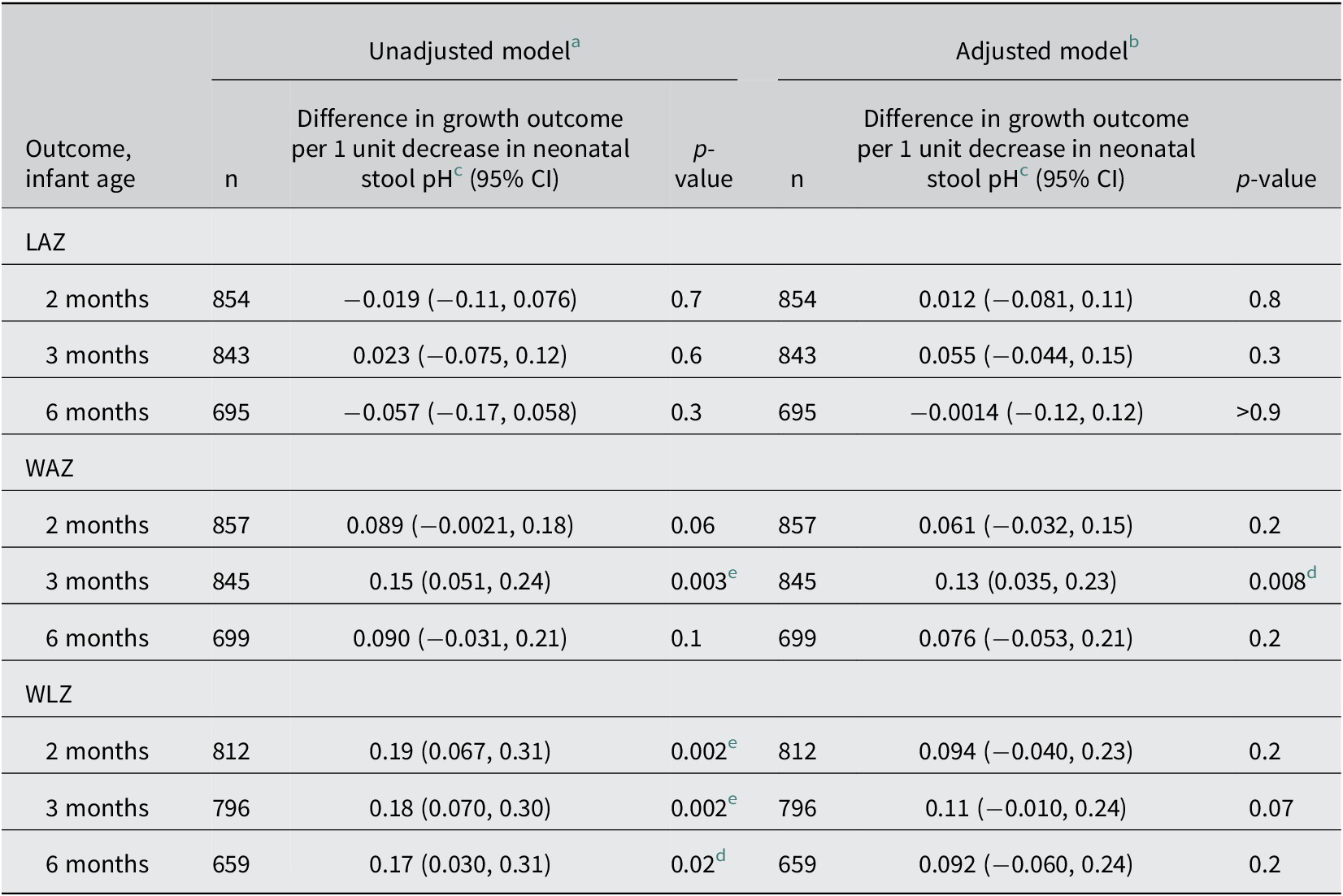
The distribution of neonatal B. infantis absolute abundances was bimodal (N infants = 830), with a median (25th and 75th percentiles) of 3.7 log cells/μg DNA (2.5, 8.8) (Figure 2). When the age range was reduced to 0–14 days, this median reduced to 2.9 (2.5, 6.9) log cells/μg DNA, and when expanded to 0–60 days, it increased to 5.4 (2.5, 8.2) log cells/μg DNA. Among the 830 infants in the primary analyses, 524 (63%) had B. infantis detected in at least one stool sample. The median (25th, 75th) neonatal abundance among infants with detectable B. infantis was 6.4 log cells/μg DNA (2.5, 8.8); conversely, among infants with undetectable B. infantis in all stool samples from 0 to 28 days of age (37%), neonatal B. infantis abundances were all uniformly 2.5 log cells/μg DNA (corresponding to the imputed lower limit of quantification). The mean (SD) neonatal stool pH (n = 1072 infants) was 5.7 (0.6).
Distributions of anthropometric indices, neonatal B. infantis absolute abundance, and neonatal stool pH. Note: Kernel density plots for infants with at least one B. infantis qPCR measurement and complete covariates: (A) LAZ (Baseline, n = 829; 2 months, n = 652; 3 months, n = 677; 6 months, n = 560), (B) WAZ (Baseline, n = 830; 2 months, n = 653; 3 months, n = 680; 6 months, n = 565), and (C) WLZ (Baseline, n = 780; 2 months, n = 609; 3 months, n = 634; 6 months, n = 525). Standard normal distributions (mean = 0, SD = 1) are shown as dashed grey lines in each panel. Histograms of (D) neonatal B. infantis absolute abundance (n = 830) and (E) neonatal stool pH (n = 1072). Abbreviations: LAZ, length-for-age z-score; WAZ, weight-for-age z-score; WLZ, weight-for-length z-score.
Anthropometric z-scores were approximately normally distributed with means below zero at all timepoints (Figure 2 and Supplementary Table S3). Infants included in this sub-study were generally representative of the overall SEPSiS observational cohort with respect to growth patterns (Supplementary Table S3).
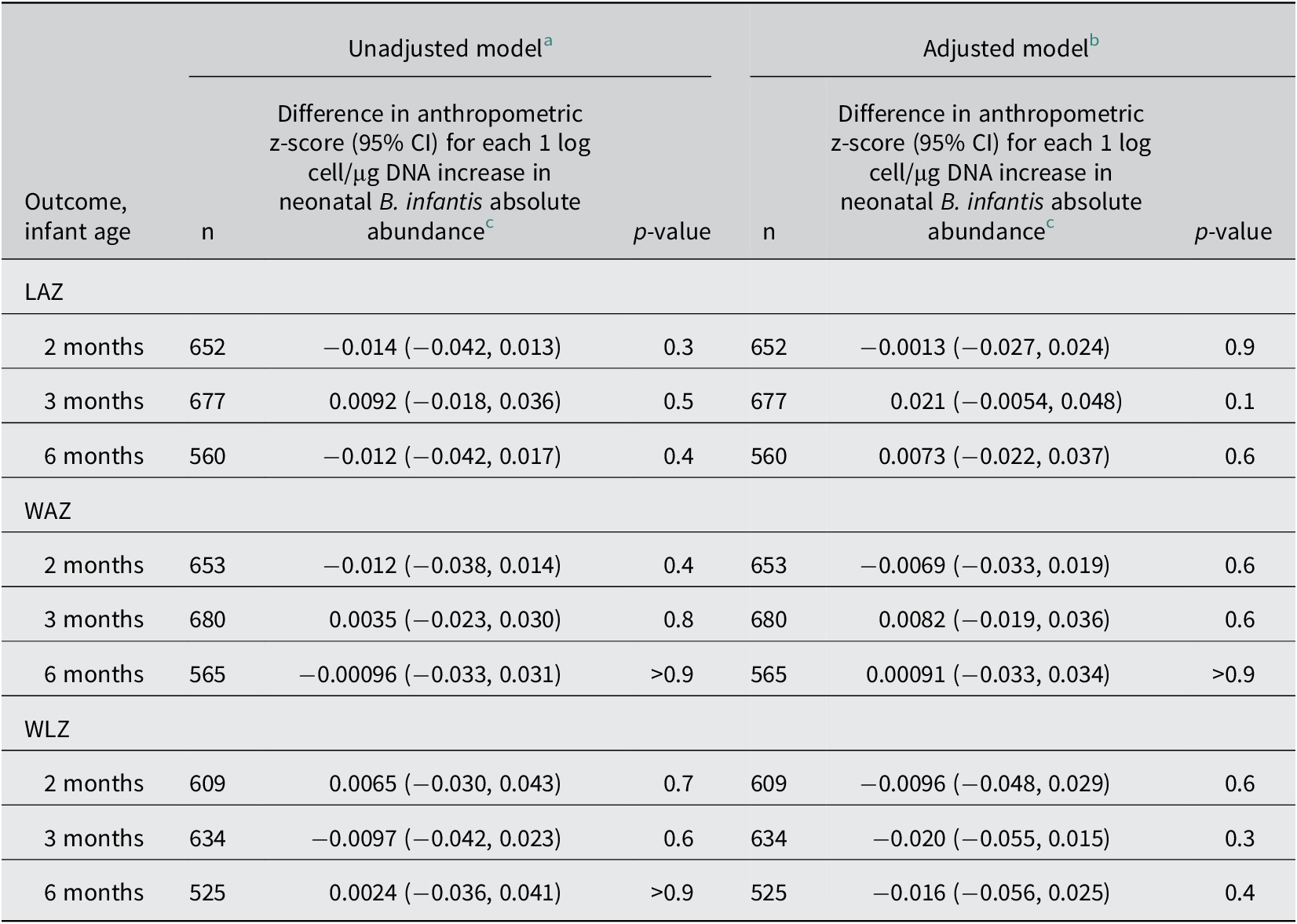
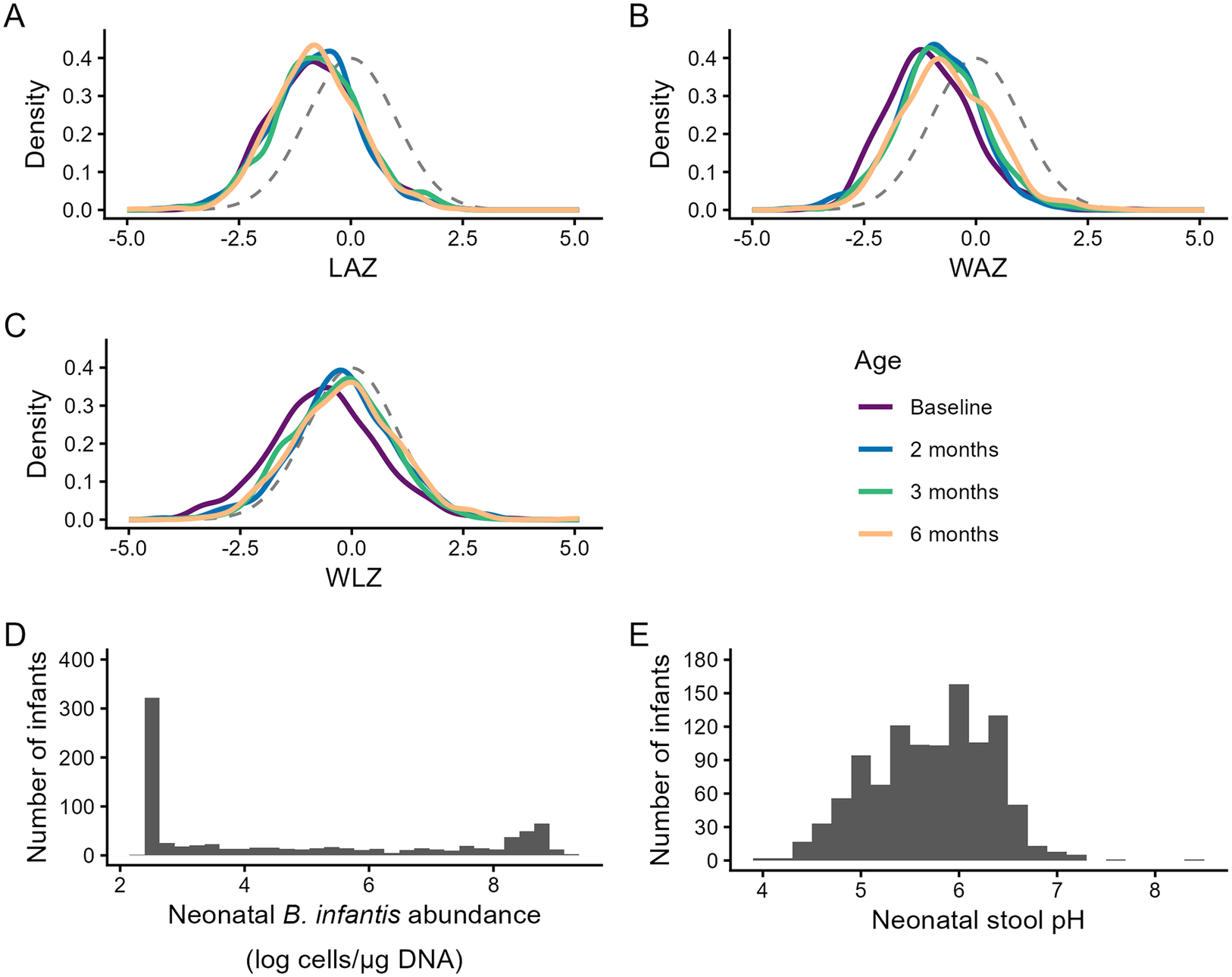
There were no associations between neonatal B. infantis absolute abundance and LAZ, WAZ, or WLZ, at 2, 3, or 6 months of age (Table 2; Supplementary Figure S4). Neonatal B. infantis absolute abundance was not associated with baseline LAZ, WAZ, or WLZ (Supplementary Table S4). Inferences remained unchanged when participants were stratified by sex (Supplementary Table S5 ), and in sensitivity analyses (Supplementary Tables S6–S11) with these exceptions: in the analysis restricted to infants enrolled in schedules A/B, there was a significant positive association between neonatal B. infantis abundance and LAZ at 3 months of age and a significant negative association between B. infantis and WLZ at 2 months of age, but both associations were attenuated and no longer significant at 6 months of age (Supplementary Table S12). To investigate if the higher number of samples in schedule A/B may have influenced the associations, the analysis was rerun using two random samples per infant to resemble the total sample numbers in schedule C, with selection simulated 1000 times. Based on this further analysis, we did not find evidence that the greater number of stool samples per infant in schedules A/B during the 0–28 day period explained the discrepancy in findings between the sub-analysis of schedule A/B and the primary analysis (Supplementary Table S13).
Associations of neonatal B. infantis absolute abundance (0–28 days of age) and infant anthropometric indices at 2, 3, and 6 months of age
Abbreviations: CI, confidence interval; LAZ, length-for-age z-score; WAZ, weight-for-age z-score; WLZ, weight-for-length z-score.
a Baseline (enrolment) measurement of the same parameter as the outcome was included as a covariate in all linear regression models.
b Additional covariates in adjusted models: enrolment site, infant sex, breastfeeding status at 28 days of age (if available) or at age most proximal to 28 days, mode of delivery, gestational age at birth, infant antibiotic exposure by 28 days of age, maternal BMI, maternal education, maternal age, number of infants within the household under 5 years of age, and household asset index quintile.
c Neonatal B. infantis absolute abundance was defined at the individual infant level as the mean of the infant’s B. infantis qPCR abundance data from all analysed stool samples collected from 0 to 28 days (as described in the main text).
In individual neonatal stool samples (n = 2118 samples), there was a 0.093 (95% CI: 0.084, 0.10; p < 0.001) decrease in stool pH per one unit increase in log B. infantis cells/μg of DNA. There was evidence that the relationship between B. infantis absolute abundance and stool pH may be non-linear (Supplementary Figure S5). When B. infantis abundance was dichotomized, the mean (SD) pH of stool samples with detectable and undetectable B. infantis was 5.5 (0.68) and 6.0 (0.54) respectively (p < 0.001).
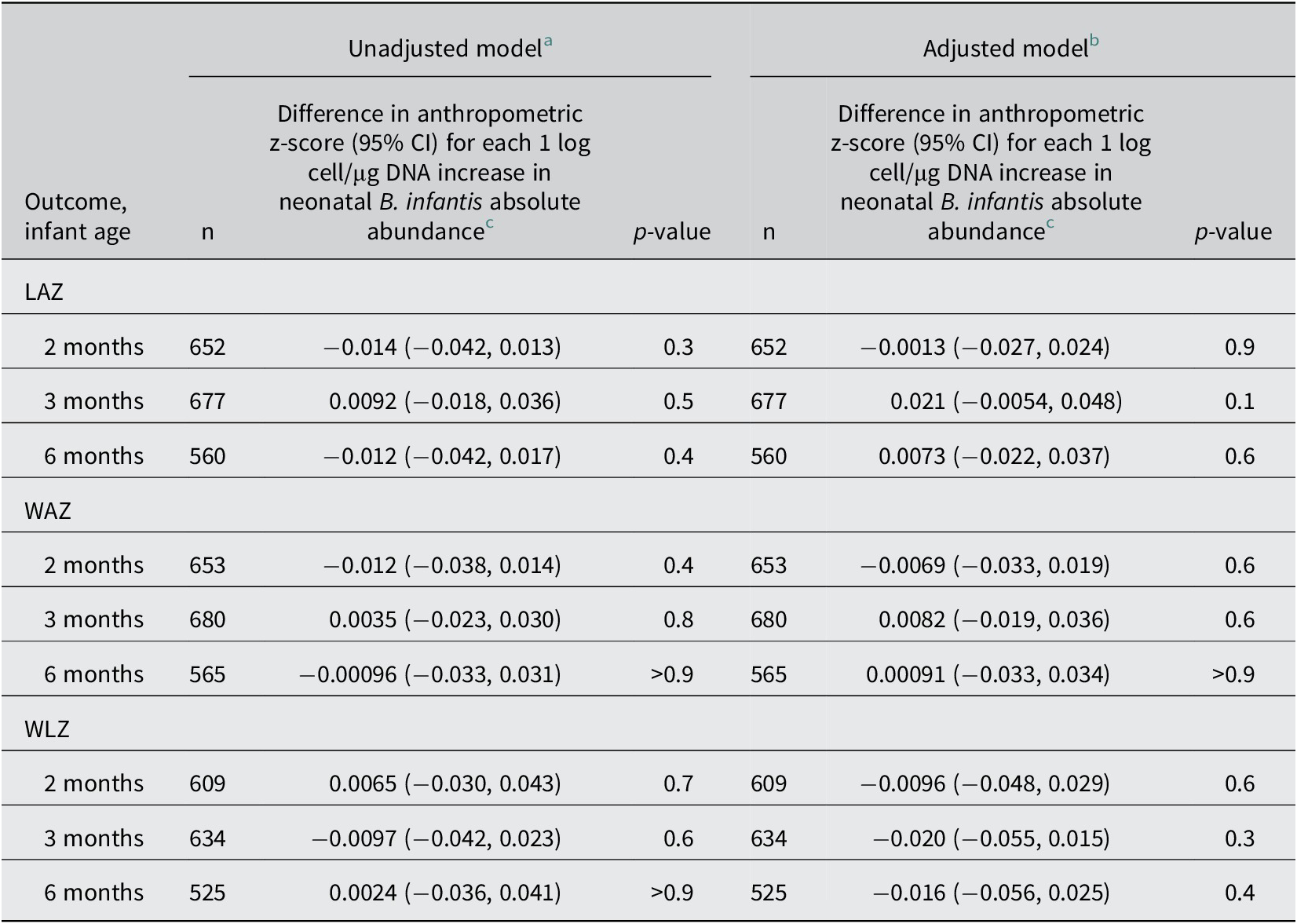
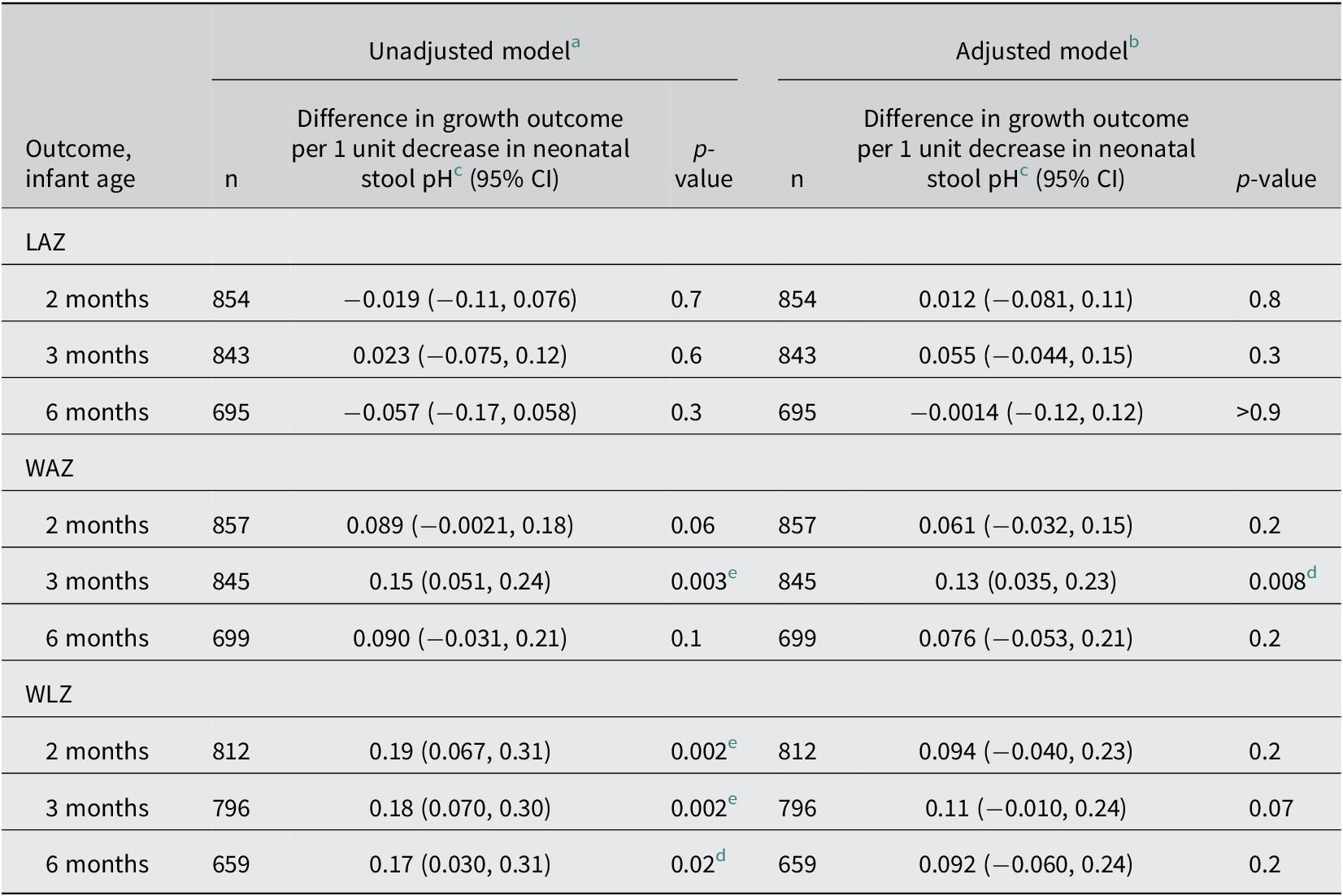
Neonatal stool pH up to 28 days after birth was significantly associated with WAZ at 3 months and WLZ at 2 and 3 months of age in unadjusted models (Table 3 and Supplementary Figure S6), but none of the associations were statistically significant after covariate adjustment and accounting for multiple testing (Table 3). There was no association between neonatal stool pH and LAZ at any time point (Table 3). Stool pH was not associated with any anthropometric indices in cross-sectional analyses (Supplementary Table S14).
Associations of neonatal stool pH (0–28 days of age) and infant anthropometric indices at 2, 3, and 6 months of age
Abbreviations: CI, confidence interval; LAZ, length-for-age z-score; WAZ, weight-for-age z-score; WLZ, weight-for-length z-score.
a Baseline (enrolment) measurement of the same parameter as the outcome was included as a covariate in all linear regression models.
b Additional covariates in adjusted models: enrolment site, infant sex, breastfeeding status at 28 days of age (if available) or at age most proximal to 28 days of age, mode of delivery, gestational age at birth, infant antibiotic exposure by 28 days of age, maternal BMI, maternal education, maternal age, number of infants within the household under 5 years of age, and household asset index quintile.
c Neonatal stool pH was defined for each infant as the median of their pH measurements among all stool samples collected from 0 to 28 days.
d No longer statistically significant after Holm’s procedure.
e Remained statistically significant after Holm’s procedure.
Of the infants who had available B. infantis absolute abundance data and had not exited the study by the end of 2 or 6 months, 36 (3.8%) infants had at least one diarrhoeal event between 29 and 60 days of age (n = 938), and 78 (8.3%) had at least one hospitalization event between 29 and 180 days of age (n = 935). Neonatal B. infantis absolute abundance was not associated with the risk of an infant experiencing a diarrhoeal event (adjusted RR = 0.96 (95%CI: 0.82, 1.1) per 1 log B. infantis cells/μg of DNA increase; p = 0.5) or hospitalization (adjusted RR = 1.0 (95%CI: 0.90, 1.1) per 1 log B. infantis cells/μg of DNA increase; p > 0.9).
Discussion
In a cohort of generally healthy infants in Dhaka, Bangladesh, B. infantis stool absolute abundance in the neonatal period was not associated with anthropometric indices up to 6 months of age. Stool pH was inversely associated with stool B. infantis abundance, consistent with previous observations (Frese et al., Reference Frese, Hutton, Contreras, Shaw, Palumbo, Casaburi, Xu, Davis, Lebrilla, Henrick, Freeman, Barile, German, Mills, Smilowitz and Underwood2017), but our results did not corroborate prior findings of an inverse association between stool pH and LAZ in Bangladeshi infants (Hossain et al., Reference Hossain, Das, Gazi, Alam, Haque, Mahfuz, Ahmed and Damman2019). Although there was weak evidence of an inverse association of stool pH with infant WAZ in early infancy, the magnitude of the association was small given the narrow range of observed pH values, the association was not significant after accounting for multiple testing, and it was not sustained to 6 months of age. Additionally, this relationship could not be explained by a pH-lowering effect of B. infantis given the absence of an association of WAZ with stool B. infantis abundance; therefore, if the relationship between stool pH and weight represents a causal pathway, it may be attributable to other components of the microbiome, or it may be confounded by unmeasured aspects of the diet or medications that influence both stool pH and weight gain (Yamamura et al., Reference Yamamura, Inoue, Nishino and Yamasaki2023).
The null associations between B. infantis abundance and anthropometric indices in the first 6 months postnatal age were broadly consistent with a prior study that did not find relationships between neonatal B. infantis abundance and LAZ (n = 292) or WAZ (n = 472) at 24 months of age in Bangladesh, Pakistan, and Tanzania (Colston et al., Reference Colston, Taniuchi, Ahmed, Ferdousi, Kabir, Mduma, Nshama, Iqbal, Haque, Ahmed, Ali Bhutta, Kosek and Platts-Mills2022). In those Bangladeshi and Tanzanian cohorts, there was a positive association of enrolment (i.e., birth) LAZ with B. infantis abundance (Colston et al., Reference Colston, Taniuchi, Ahmed, Ferdousi, Kabir, Mduma, Nshama, Iqbal, Haque, Ahmed, Ali Bhutta, Kosek and Platts-Mills2022), which raises the possibility of reverse causality. However, we did not observe a similar association in the SEPSiS observational cohort, and our inferences were the same in models with and without adjustment for baseline LAZ.
Recent systematic reviews of the effects of probiotics in infants have included few studies of B. infantis, or when included, were generally administered in combination with other probiotic species (Catania et al., Reference Catania, Pandit, Ehrlich, Zaman, Stone, Franceschi, Smith, Tanner-Smith, Zackular, Bhutta and Imdad2021; Guo et al., Reference Guo, Fan, Hou, Li, Wang, Wang, Peng, Wang, Wu, Zhang, Guo, Fan, Hou, Li, Wang, Wang, Peng, Wang, Wu and Zhang2023; Panchal et al., Reference Panchal, Athalye-Jape, Rao and Patole2023), or focused on B. infantis but did not address growth outcomes (Batta et al., Reference Batta, Rao, Patole, Batta, Rao and Patole2023). Two trials of the B. infantis probiotic EVC001 conducted in the United States did not find effects on achieved weight/weight gain or length among term exclusively breastfed infants (Smilowitz et al., Reference Smilowitz, Moya, Breck, Cook, Fineberg, Angkustsiri and Underwood2017) or preterm infants (Bajorek et al., Reference Bajorek, Duar, Corrigan, Matrone, Winn, Norman, Mitchell, Cagney, Aksenov, Melnik, Kopylova and Perez2022). Similarly, two trials in Spain showed no effects of B. infantis probiotics on infant weight or length (Manzano et al., Reference Manzano, De Andrés, Castro, Rodríguez, Jiménez and Espinosa-Martos2017; Escribano et al., Reference Escribano, Ferré, Gispert-Llaurado, Luque, Rubio-Torrents, Zaragoza-Jordana, Polanco, Codoñer, Chenoll, Morera, Moreno-Muñoz, Rivero and Closa-Monasterolo2018). However, in a single-blind randomized controlled trial involving 2- to 6-month-old infants with severe acute malnutrition in Dhaka, Bangladesh (n = 67), infants treated with B. infantis EVC001 daily for 28 days had a significantly higher WAZ and mid-upper arm circumference compared to the placebo group at 28 days post-intervention, despite no effect on LAZ (Barratt et al., Reference Barratt, Nuzhat, Ahsan, Frese, Arzamasov, Sarker, Islam, Palit, Islam, Hibberd, Nakshatri, Cowardin, Guruge, Byrne, Venkatesh, Sundaresan, Henrick, Duar, Mitchell, Casaburi, Prambs, Flannery, Mahfuz, Rodionov, Osterman, Kyle, Ahmed and Gordon2022; Nuzhat et al., Reference Nuzhat, Hasan, Palit, Islam, Mahfuz, Islam, Alam, Flannery, Kyle, Sarker and Ahmed2023). Some studies of probiotics with multiple species, including B. infantis reported positive effects on weight gain in very preterm infants in Australia and New Zealand (Jacobs et al., Reference Jacobs, Tobin, Opie, Donath, Tabrizi, Pirotta, Morley and Garland2013) or very low birth weight infants in the United States (Al-Hosni et al., Reference Al-Hosni, Duenas, Hawk, Stewart, Borghese, Cahoon, Atwood, Howard, Ferrelli and Soll2012) and Germany (Härtel et al., Reference Härtel, Pagel, Spiegler, Buma, Henneke, Zemlin, Viemann, Gille, Gehring, Frommhold, Rupp, Herting and Göpel2017), but it is uncertain whether the effects were specifically attributable to B. infantis. In a sensitivity analysis in the present study, which was restricted to a subset of infants who had stool sampled more frequently (schedule A/B; 40–42% of infants in primary analyses), B. infantis was positively associated with LAZ and negatively associated with WLZ at 3 months of age but these associations were substantially attenuated by 6 months of age and were not explained by a greater precision of B. infantis abundance measurement due to higher sample numbers. Therefore, we did not consider these findings to have altered the overall conclusion that neonatal B. infantis abundance is unrelated to growth in the first 6 months postnatal age.
A meta-analysis of trials investigating the effect of probiotics containing B. infantis on preterm and/or very low birth weight infants found that administering probiotics reduced all-cause mortality (14 trials with 4292 infants), necrotizing enterocolitis (15 trials with 3626 infants), and late onset sepsis (13 trials with 4123 infants) (Batta et al., Reference Batta, Rao, Patole, Batta, Rao and Patole2023). However, only five of the 16 studies included 100 or more infants in each trial arm, and all studies used a combination of probiotics that included B. infantis, rather than B. infantis alone. In the present study, we did not find associations between B. infantis neonatal abundance and either diarrhoeal events or hospitalizations; however, we acknowledge that these analyses may have been underpowered due to a low number of events. We did not assess associations with cause-specific hospitalizations due to the low number of events related to discrete causes. However, we acknowledge that the use of all-cause hospitalization as the outcome measure may have biased the association towards the null if only certain illnesses leading to hospitalization were truly associated with B. infantis abundance.
Examining individual-level risk factors, such as B. infantis abundance, may identify sources of inter-individual variations in growth within a population; however, inter-individual variations may not provide insights into a population-level phenomenon (Rose, Reference Rose1985; Roth et al., Reference Roth, Krishna, Leung, Shi, Bassani and Barros2017). Growth faltering in LMICs is not restricted to a subset of infants with stunting within a population, but rather the whole population distributions of anthropometric indices are shifted downwards (Roth et al., Reference Roth, Krishna, Leung, Shi, Bassani and Barros2017; Victora et al., Reference Victora, Christian, Vidaletti, Gatica-Domínguez, Menon and Black2021), as observed in this study cohort. Given the high prevalence of B. infantis colonization in populations with endemic postnatal growth faltering (Lawley et al., Reference Lawley, Otal, Moloney-Geany, Diana, Houghton, Heath, Taylor and Tannock2019; Taft et al., Reference Taft, Lewis, Nguyen, Ho, Masarweh, Dunne-Castagna, Tancredi, Huda, Stephensen, Hinde, Von Mutius, Kirjavainen, Dalphin, Lauener, Riedler, Smilowitz, German, Morrow and Mills2022; Shao et al., Reference Shao, Wang, Gichuki, Stares, Rozday, Kumar, Browne, Dawson, Njunge, Tigoi, Ngao, Chisti, Singa, Kariuki, Diallo, Saleem, Ali, Mupere, Mbale, Tickell, Voskuijl, Lancioni, Bandsma, Ahmed, Walson, Berkley and Lawley2026), and the absence of associations of B. infantis with growth at the infant level as observed here, there is unlikely to be an important role for early postnatal B. infantis colonization in mitigating growth-faltering in such settings.
Several limitations of the present study should be considered. Stool B. infantis abundance was used as a proxy of intestinal B. infantis abundance, but stool abundance of specific microbes may not reflect abundance within the intestinal lumen (Zmora et al., Reference Zmora, Zilberman-Schapira, Suez, Mor, Dori-Bachash, Bashiardes, Kotler, Zur, Regev-Lehavi, Brik, Federici, Cohen, Linevsky, Rothschild, Moor, Ben-Moshe, Harmelin, Itzkovitz, Maharshak, Shibolet, Shapiro, Pevsner-Fischer, Sharon, Halpern, Segal and Elinav2018); however, methods for obtaining luminal samples from infants’ intestinal tracts rely on procedures that would be invasive, infeasible and unethical in a large cohort study in this age group. We focused our analysis on B. infantis colonization in the neonatal period because most infants were eventually colonized with B. infantis by 1–2 months of age (Freitas et al., Reference Freitas, Li, Shawon, Qamar, Pell, Kabir, Oduaran, Puebla-Barragan, Bassani, O’Callaghan, Onuora, Loutet, Heasley, Starke, Mahmud, Hamer, Pullenayegum, Hossain, Siddiqui, Sherman, Shah, Gaffar, Sultana, Morris, Ahmed, Haque, Sarker, Roth and Roth2025); therefore, we were unable to examine the effects of longer-term colonization. Additionally, we only explored the associations between infant growth and the abundance of naturally occurring B. infantis without consideration of strain-level distinctions. Beneficial effects from commensals or probiotics may be strain-specific; for example, HMO gene clusters are highly conserved across B. infantis strains, but the presence of specific genes within clusters differs among B. infantis strains (Locascio et al., Reference Locascio, Desai, Sela, Weimer and Mills2010; Duar et al., Reference Duar, Casaburi, Mitchell, Scofield, Ortega Ramirez, Barile, Henrick and Frese2020; Shao et al., Reference Shao, Wang, Gichuki, Stares, Rozday, Kumar, Browne, Dawson, Njunge, Tigoi, Ngao, Chisti, Singa, Kariuki, Diallo, Saleem, Ali, Mupere, Mbale, Tickell, Voskuijl, Lancioni, Bandsma, Ahmed, Walson, Berkley and Lawley2026). Infant feeding pattern at 28 days was included as a covariate in analyses, but we acknowledge that the associations might be modified by feeding pattern, which was not assessed in the present study. The study population had a high C-section rate, which influences early gut microbial succession patterns (Freitas et al., Reference Freitas, Li, Shawon, Qamar, Pell, Kabir, Oduaran, Puebla-Barragan, Bassani, O’Callaghan, Onuora, Loutet, Heasley, Starke, Mahmud, Hamer, Pullenayegum, Hossain, Siddiqui, Sherman, Shah, Gaffar, Sultana, Morris, Ahmed, Haque, Sarker, Roth and Roth2025); however, this rate of C-section is comparable to other studies in this setting (National Institute of Population Research and Training (NIPORT) and ICF, 2024; Nuzhat et al., Reference Nuzhat, Hasan, Palit, Islam, Mahfuz, Islam, Alam, Flannery, Kyle, Sarker and Ahmed2023). Furthermore, as in any observational study, the estimates should not be interpreted as causal effects as they may be biased by residual confounding due to a wide range of unmeasured factors related to the infant’s health status, components of the infant’s diet, and microbiome composition.
Conclusion
Stool absolute abundance of B. infantis in the first month after birth was not associated with linear or ponderal growth up to 6 months of age in generally healthy infants in Dhaka, Bangladesh. Based on these findings, promotion of early B. infantis colonization through interventions such as B. infantis probiotic administration in the neonatal period may not be expected to improve growth outcomes in generally healthy infants born in settings with high prevalence of natural B. infantis colonization and where postnatal growth faltering is widespread.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/gmb.2026.10023.
Acknowledgements
We thank the families who participated in the SEPSiS cohort study, as well as the other SEPSiS project co-investigators and study personnel who were involved in data collection and laboratory analysis.
Author contribution
Conceptualization, C.K.H., V.S., L.G.P., D.G.B., K.M.OC., P.S.S., and D.E.R.; Data Curation, C.K.H., C.F., A.C.F., and G.L.; Formal Analysis, C.K.H.; Funding Acquisition, D.E.R.; Methodology, C.K.H. and D.E.R.; Supervision, D.E.R.; Validation, C.F., A.C.F., and G.L.; Writing—Original Draft, C.K.H., and V.S.; Writing—Review and Editing, C.K.H., V.S., C.F., A.C.F., G.L., L.G.P., D.G.B., K.M.OC., P.S.S., J.S., S.M.A.G., R.H., S.A.S., and D.E.R.
Funding
This research was funded by The Bill and Melinda Gates Foundation, grant number INV-007389 to The Hospital for Sick Children and grant GR-02268 to the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b). The funders had no role in the study design, data collection, statistical analysis, preparation of the manuscript, or the decision to submit the manuscript for publication.
Disclosure statement
The authors report no conflict of interest.
Research transparency and reproducibility
All data and code used for the analyses are available on Borealis: https://doi.org/10.5683/SP3/PWPMGK