Dietary polyphenols are found in fruits and vegetables, and in products derived from plants such as fruit beverages, tea, coffee and dark chocolate. Epidemiological evidence shows that consumption of polyphenol-rich foods reduces the risk of CVD and associated conditions(Reference Hooper, Kroon and Rimm1), and human intervention studies have supported this association(Reference Williamson and Manach2). The absorption and metabolism of flavonoids is quite well described for some compounds, such as quercetin and (–)-epicatechin(Reference Scholz and Williamson3). However, although the absorption of certain other phenols, polyphenols and tannins (PPT) such as procyanidins, chlorogenic acids and anthocyanins has been described in the literature, the levels of the parent compounds in blood after a high dose, or a large amount of PPT-rich food, are very low compared with other flavonoids(Reference Vitaglione, Donnarumma and Napolitano4, Reference Nurmi, Mursu and Heinonen5). In contrast, intervention studies show that procyanidin-rich, chlorogenic acid-rich or anthocyanin-rich foods affect certain biomarkers, but because the concentration of parent compounds to be expected in blood after consumption is too low to affect these biomarkers(Reference Jensen, Wu and Patterson6), other bioactive substances such as metabolites may be responsible and must be identified. It is proposed that these ‘missing’ components are the colonic catabolites, and that they are potentially important compounds mediating some of the biological activities and health benefits of polyphenol-rich foods.
In excess of 8000 phenolic compounds have been reported, and they are widely dispersed throughout the plant kingdom. The nature of those occurring in foods and beverages has been reviewed(Reference Crozier, Jaganath and Clifford7, Reference Crozier, Clifford and Ashihara8). Briefly, they can be subdivided into flavonoids (anthocyanins, chalcones and dihydrochalcones, flavanols or catechins, flavanones, flavones, flavonols, isoflavones and proanthocyanidins or condensed tannins) and non-flavonoids (benzoic acids, cinnamic acids and cinnamic acid conjugates, such as the chlorogenic acids, and gallic acid esters or hydrolysable tannins). To these must be added the ‘derived polyphenols’, substances that are characteristic of many traditionally processed foods and beverages (coffee, black tea, matured red wine, etc.) but which do not occur in healthy intact plant tissue. Collectively, these three major groups will in this review be referred to as PPT. In addition, this review will discuss the aromatic and phenolic acids produced from these PPT by the gut microflora, many of which do not occur preformed in the diet. These acids can be classified by the number of carbons in the side chain (one to five), and whether the side chain is saturated or unsaturated, and whether it carries an aliphatic hydroxyl. Specimen structures are shown in Fig. 1.
Structures of selected phenols, polyphenols and tannins, and selected gut microflora catabolites. A and B indicate the A- and B-ring, respectively, of a typical flavonoid. Typical structures are shown for flavanols (catechins) (I); flavanones (II); flavones (III); dihydrochalcones (IV); chlorogenic acids (cinnamate conjugates) (V); cinnamic acids (C6–C3) (VI); anthocyanidins (VII); phenylvaleric acids (C6–C5-γ-OH) (VII); phenylpropionic acids (C6–C3) (IX. XIII); phenylacetic acids (C6–C2) (X, XIV); flavonols (XI); benzoic acids (C6–C1) (XII, XV). Note: it is not possible to show all possible substrates, intermediates and pathways but those shown are representative.

Polyphenol breakdown in the colon
Bacterial composition of the human gut
The first part of this review is concerned primarily with the capability of the microflora in the distal gastro-intestinal tract (GIT) to transform PPT provided by the diet. A detailed discussion of the complex nature and composition of this microflora is beyond the scope of this review, but some basic statistics are instructive. The GIT is home to between some 10 and 100 trillion microbes(Reference Bäckhed, Ley and Sonnenburg9), approximately 10-times as many cells as found in the human body(Reference Savage10). The corresponding microbiome is said to contain more than 100-times as many genes as the human genome(Reference Bäckhed, Ley and Sonnenburg9). Each portion of the GIT (mouth, oesophagus, stomach, small intestine and large intestine) has a distinctive flora, but the majority of the organisms are located in the distal GIT, where the concentration of some 500 bacterial species may reach 1011–1012 colony forming units/g, and account for some 35–50 % of the contents(Reference Salminen, Isolauri and Salminen11). The flora contains bacteria, archaea and eukarya, and the bacterial component has been studied the most extensively. Three genera, Bacteroides spp., Clostridium spp. and Eubacterium spp., each account for approximately 30 % of the organisms present(Reference Bäckhed, Ley and Sonnenburg9), but the colon also has a significant content of Fusobacterium spp., Peptostreptococcus spp. and Bifidobacterium spp.(Reference Salminen, Isolauri and Salminen11, Reference Tuohy, Gibson, Crozier, Clifford and Ashihara12) These bacteria are accompanied by a single methanogenic archaeon, Methanobrevibacter smithii (Reference Bäckhed, Ley and Sonnenburg9, Reference Gill, Pop and Deboy13). The eukarya include protozoa, but those found in the GIT seem never to have been studied with reference to PPT catabolism.
It is generally accepted that colonisation of the GIT of neonates starts immediately after birth. The type of delivery (passage through the birth canal v. caesarean section), type of diet (breast v. formula feeding) and environment affect the colonisation pattern(Reference Gronlund, Lehtonen and Eerola14, Reference Harmsen, Wildeboer-Veloo and Raangs15). The composition of the gut flora of a 2-year-old child is essentially the same as that of an adult(Reference Conway, Gibson and Macfarlane16), but the variations in microflora composition associated with age, diet and health status are still not fully determined(Reference Eckburg, Bik and Bernstein17). A study of faecal samples from 230 volunteers, aged 20–50 years, from four European countries has demonstrated marked country–age interactions. The proportions of bifidobacteria were 2- to 3-fold higher in the Italian study population than in any other study group, and this effect was independent of age. Higher proportions of enterobacteria were found in all elderly volunteers independent of the location(Reference Mueller, Saunier and Hanisch18). There is good evidence that comparatively minor changes in the diet of rats can produce significant changes in the catabolites eventually produced from dietary PPT(Reference Phipps, Stewart and Wright19), but the extent to which the human microflora can be modulated by diet is unknown.
Models used to study colonic catabolism
The distal GIT is conveniently viewed as an anaerobic fermentation vessel in which polysaccharides, proteins, lipids and xenobiotics such as PPT can be transformed. Several strategies have been used to investigate these transformations. In vitro studies frequently use either a defined bacterial strain(Reference Wang, Hur and Lee20, Reference Raimondi, Roncaglia and De Lucia21), ileostomy fluid(Reference Knaup, Kahle and Erk22) or a flora from freshly voided human(Reference Aura, Martin-Lopez and O'Leary23–Reference Gonthier, Remesy and Scalbert27) or animal faeces(Reference Labib, Hummel and Richling28–Reference Forester and Waterhouse30) cultured in a suitable medium to which a defined substrate has been added, and monitor (i) disappearance of substrate, (ii) formation of catabolites or (iii) changes in the microflora(Reference Lee, Jenner and Low31, Reference Tzounis, Vulevic and Kuhnle32). Rarely all three parameters are studied at the same time. Typically controls would include (i) incubations containing test substrate but lacking the microbial culture to investigate purely chemical transformation, (ii) incubations containing test substrate and chloroform-inactivated culture to investigate substrate losses associated with cell binding and transformations associated with enzymes released after lysis of the micro-organisms and (iii) incubations containing basal medium and microbial culture in order to monitor catabolites arising from basal metabolism and to distinguish these from catabolites produced from the test substance. The culture may be static(Reference Gonthier, Remesy and Scalbert27) or dynamic(Reference Gao, Xu and Krul33). The various types of study used to examine microbial transformation of PPTs are shown in Table 1.
Studies concerned with the transformation of phenols, polyphenols and tannins (PPT) by gut flora micro-organisms

It is impossible to know how well these in vitro models reflect the human GIT in vivo but studies using gnotobiotic rats that have been associated with a specific GIT micro-organism do not produce the same catabolites as that of the organism incubated in an artificial medium(Reference Peppercorn and Goldman34) and one must assume that the models are imperfect. In particular, micro-organisms that bind to the GIT surface might be very different to those found in the lumen, and it is these ‘unbound’ organisms that might predominate in the voided stool from which the culture is prepared(Reference Eckburg, Bik and Bernstein17, Reference Hooper and Gordon35). The micro-organisms that survive the culturing process will depend on how rigorously oxygen was excluded and the nature of the medium employed. Media for anaerobic micro-organisms are typically rich in protein or polypeptides and these can be incompatible with the recovery of PPT that bind strongly to protein thus preventing proper analysis of the transformations. Studies in the authors’ laboratories suggest that the catabolism of PPT that are not strongly bound to protein is not significantly different from their catabolism in protein-free media, and protein-free media have been used to investigate the catabolism of procyanidins that would bind irreversibly to protein(Reference Stoupi, Williamson and Drynan36). One advantage of these models is the requirement for comparatively small amounts of a scarce and costly pure substrate.
A second approach has been the use of laboratory animals, commonly rodents, but occasionally pigs. The rodents might be either (i) normal laboratory animals with their typical flora(Reference He, Magnuson and Giusti37), (ii) animals treated with antibiotics (typically neomycin) to destroy the flora, (iii) gnotobiotic animals delivered under sterile conditions, and housed in sterile conditions and fed only on γ-irradiated diets(Reference Peppercorn and Goldman34, Reference Scheline and Midtvedt38) or (iv) gnotobiotic animals associated with a human-type flora(Reference Tamura and Saitoh39). The use of such animals has allowed the effects of the rodent flora and human-type flora to be compared, and the effects of the flora and mammalian metabolism to be distinguished. Radio-labelled test substances can be used and full pharmacokinetic studies can be performed with these animals, and the contents (microbial and chemical) can be compared at different sites in the GIT(Reference He, Magnuson and Giusti37). However, gnotobiotic animal facilities, are very expensive to maintain. An extensive review of the in vitro and animal models is available(Reference Rumney and Rowland40). The early studies using these procedures, but focusing on flavonoids, have been reviewed(Reference de Eds, Florkin and Stotz41–Reference Berry, Francis and Bollag43).
A third approach is to use volunteers, either free-living or following prescribed diets, and to collect faeces(Reference Knust, Erben and Spiegelhalder44), faecal water(Reference Jenner, Rafter and Halliwell45) and urine for analysis(Reference Vitaglione, Donnarumma and Napolitano4). Normal volunteers can be compared with volunteers who have undergone an ileostomy but who are otherwise healthy enabling metabolism in the small intestine to be distinguished from that in the large intestine. These are discussed in more detail as follows.
Transformation of polyphenols by colonic microflora
The transformations of which the microflora are capable include O- and C-deglycosylation, the hydrolysis of esters and amides, and deglucuronidation of excreted mammalian metabolites. The aglycones are susceptible to aromatic dehydroxylation, demethoxylation and demethylation, and hydrogenation, α-oxidation and β-oxidation of the aliphatic elements generated following rupture of the aromatic ring(Reference Peppercorn and Goldman34, Reference Scheline and Midtvedt38, Reference de Eds, Florkin and Stotz41–Reference Berry, Francis and Bollag43, Reference Hattori, Shu and El Sedawy46–Reference Kroon, Faulds and Ryden49). Most investigations have focused on phenolic catabolites, i.e. those that retain at least one phenolic hydroxyl, but it is clear that complete dehydroxylation can occur producing aromatic catabolites(Reference Phipps, Stewart and Wright19, Reference Goodwin, Ruthven and Sandler50). Benzoic acid, however, can also be produced in human subjects by GIT microflora aromatisation of quinic acid(Reference Adamson, Bridges and Evans51). It should also be noted that there is a significant, but often unquantified, yield of non-aromatic products, including oxaloacetate, CO2, etc.(Reference Walle, Walle and Halushka52). A key step in the catabolism of flavonoids is the fission of the A-ring and loss of carbons C5 to C8 as oxaloacetate that is ultimately metabolised to CO2(Reference Das and Griffiths53). Similarly, an oral-dosing volunteer study using [4-14C]-quercetin demonstrated that approximately 50 % of the radioactivity was eliminated as CO2(Reference Walle, Walle and Halushka52), but the fate of the A-ring and B-ring was not investigated.
Dehydroxylation of ortho-dihydroxy and ortho-trihydroxy substrates can occur at either a meta or para position, but it is accepted that the hydroxyl at the meta position is removed less readily. Accordingly, ortho-dihydroxy and ortho-trihydroxy substrates yield predominantly meta-hydroxy and meta-dihydroxy catabolites, respectively(Reference Griffiths and Smith54, Reference Smith and Griffiths55). Absence of a para-hydroxyl in the B-ring of flavonoids is considered to slow the degradation by the GIT microflora(Reference Labib, Hummel and Richling28, Reference Griffiths and Smith56). Studies using 2H-labelled substrates and crude human gut flora have established that with 3-(4′-hydroxyphenyl)-propionic acid and 4-hydroxyphenyl-lactic acid, the aliphatic side chain may be moved about the aromatic ring, thus converting a 4-hydroxy substrate to the corresponding 3-hydroxy isomer(Reference Curtius, Mettler and Ettlinger57, Reference Fuchs-Mettler, Curtius and Baerlocher58). The enzymes/micro-organisms responsible have not been characterised and their substrate specificity is unknown. Although it is possible that such an isomerisation might explain the apparent preference for removal of the para-hydroxyl from a 3,4-dihydroxy substrate, it would not explain the tendency for 3,4,5-trihydroxy substrates to be converted to 3,5-dihydroxy products.
Aromatic demethylation by rodent, pig and human GIT microflora has been observed in vitro, for example demethylation of 3′-O-methyl-(+)-catechin(Reference Gott and Griffiths59), 4′-O-methylgenistein (angolensin)(Reference Blaut, Schoefer and Braune60), 6-methoxyapigenin(Reference Labib, Hummel and Richling28) and phenolic acids formed from the B-ring of anthocyanins(Reference Keppler and Humpf24). In contrast, the gut microflora of the rat, mouse and marmoset are unable to degrade 3-O-methyl-(+)-catechin(Reference Hackett and Griffiths61), suggesting that the they cannot attack the C-ring aliphatic O-methyl group. Similarly, there is no evidence for demethylation of this compound when given orally to volunteers(Reference Hackett, Griffiths and Wermeille62).
Comparatively few of the enzymes associated with these transformations have been characterised in microbes associated with the GIT, and the subtleties of substrate specificity, enzyme inhibition, etc., remain largely unknown. At least with regard to polysaccharide catabolism there appears to be few structures that cannot be degraded(Reference Bäckhed, Ley and Sonnenburg9), and this situation might well apply also to PPT.
It has been demonstrated that some strains of the GIT micro-organism Eubacterium oxidoreducens can convert pyrogallol to dihydrophloroglucinol and 3-hydroxy-5-oxohexanoate. The phloroglucinol reductase and pyrogallol–phloroglucinol isomerase have been isolated and characterised(Reference Krumholz and Bryant63–Reference Haddock and Ferry66). This isomerase can be viewed as creating a new hydroxyl at C2 in the phloroglucinol (1,3,5-trihydroxybenzene) equivalent to creating a hydroxyl at either C6 or C8 in the flavonoid A-ring. Such a hydroxylation of the flavonoid A-ring has been demonstrated with Pseudomonas spp.(Reference Jeffrey, Jerina and Self67, Reference Jeffrey, Knight and Evans68) but the general relevance of this observation to GIT transformation of PPT is uncertain because these facultative anaerobes are not typical of the GIT microflora.
Eubacterium ramulus has been reported at 4·4 × 10Reference Crozier, Jaganath and Clifford7 to 2·0 × 109 colony forming units/g (n 20) of human faecal dry mass(Reference Simmering, Kleessen and Blaut69), and its growth is stimulated in vivo by dietary flavonoids(Reference Simmering, Pforte and Jacobasch70). Eubacterium spp. are well known for their ability to degrade flavonoids anaerobically, variously possessing deglycosylating activity, the ability to split the C-ring, chalcone isomerase and phloretin hydrolase activity(Reference Braune, Engst and Blaut26, Reference Schneider and Blaut71–Reference Herles, Braune and Blaut76). These chalcone isomerase(Reference Herles, Braune and Blaut76) and phloretin hydrolase(Reference Braune, Engst and Blaut26, Reference Schoefer, Braune and Blaut75) enzymes convert flavanones to the isomeric dihydrochalcones and the dihydrochalcones to a C6–C3 acid and phloroglucinol, respectively. The phloroglucinol will be rapidly converted to aliphatic products as described earlier. It is not known whether the retro-chalcone associated with anthocyanins in media with pH>7(Reference Clifford77) are substrates for the phloretin hydrolase.
Eubacterium limosum demethylates isoflavones such as biochanin A, formononetin and glycitein(Reference Hur and Rafii78), and Eubacterium A-44 has a novel arylsulfotransferase but whether mammalian flavonoid sulphates can serve as donors is not known(Reference Kim, Konishi and Kobashi79). Eubacterium sp. SDG-2 and Peptostreptococcus sp. SDG-1 convert seco-isolariciresinol diglucoside to mammalian lignans(Reference Wang, Meselhy and Li80), and this Eubacterium degrades various (3R)- and (3S)-flavanols and their 3-O-gallates to diaryl-propan-2-ols, 3,5-dihydroxyphenylvalerolactone and 3-hydroxyphenyl-valerolactone. Interestingly, this organism can remove the para-hydroxyl from 3R flavanols such as (–)-epicatechin and (–)-catechin but not from the 3S flavanols such as (+)-catechin and (+)-epicatechin(Reference Wang, Meselhy and Li81), and it has been demonstrated in vitro that the conversion of (+)-catechin to C6–C5 and C6–C3 catabolites required its initial conversion to (+)-epicatechin(Reference Tzounis, Vulevic and Kuhnle32).
Some Clostridium spp. can cleave the C-ring of flavonoids, converting flavonols to a phenylacetic acid and presumably phloroglucinol. Activity with flavanones was much weaker and could not be detected with flavanols(Reference Winter, Moore and Dowell82, Reference Winter, Popoff and Grimont83). Some Fusobacterium spp. and Bacteroides spp. possess α-l-rhamnosidase(Reference Jang and Kim84, Reference Bokkenheuser, Shackleton and Winter85), β-d-glucosidase(Reference Kim, Sohng and Kobashi86, Reference Kim, Kim and Park87), β-d-galactosidase(Reference Bokkenheuser, Shackleton and Winter85) and β-d-glucuronidase(Reference Sung, Kang and Yoon88) activities.
The PPT catabolites most frequently reported are aromatic/phenolic acids having 0, 1, 2 or 3 aromatic hydroxyls, 0, 1 or 2 methoxyls, and a side chain of between one and five carbons, which might be unsaturated and might bear an aliphatic hydroxyl. For most flavonoids, the acids produced retain the intact B-ring, but anthocyanins also yield 2,4,6-trihydroxybenzoic acid and the corresponding benzaldehyde (phloroglucinaldehyde) both derived from the A-ring(Reference Keppler and Humpf24, Reference Schneider, Schwiertz and Collins72, Reference Justesen, Arrigoni and Larsen89, Reference Kim, Jung and Sohng90). There is evidence that these phloroglucinol derivatives might form from anthocyanins purely by chemical degradation at the prevailing pH value in the GIT(Reference Kay, Kroon and Cassidy91). Whether of chemical or microbial origin, these aromatic/phenolic acids are common to most PPT substrates so far investigated, although some variation of ring substitution and side chain length are substrate specific as summarised in Table 2. Phenolic acids can be decarboxylated to the corresponding hydrocarbons(Reference Labib, Hummel and Richling28, Reference Peppercorn and Goldman34, Reference Griffiths and Smith56, Reference Justesen, Arrigoni and Larsen89, Reference Peppercorn and Goldman92, Reference Indahl and Scheline93), but the corresponding alcohols seem not to have been reported.
The nature of the aromatic and phenolic acids produced by the gut microflora from pure phenols, polyphenols and tannins (PPT) substrates

* C6–C3 βOH (phenylhydracrylic acids) probably arise from mammalian metabolism of a microbial catabolite rather than directly by microbial catabolism.
† May occur also as lactones.
‡ In the case of isoflavones the aryl substituent is attached to C2 of the C6–C3 dihydro side chain. For all other PPT, it is attached to C3.
§ C6–C1 acids may form also by a purely chemical mechanism at the prevailing pH value.
Certain larger mass intermediates have been characterised only during the catabolism of flavanols and proanthocyanidins (e.g. diaryl-propan-2-ols)(Reference Groenewoud and Hundt94, Reference Meselhy, Nakamura and Hattori95), and catabolites such as S-equol, diarylbutanes and urolithins(Reference Doyle and Griffiths96–Reference Seeram, Aronson and Zhang99) are specific to the isoflavones, lignans and ellagitannins, respectively. Faecal water from free-living volunteers also contains some flavonoid aglycones indicating that aglycone degradation is not necessarily complete(Reference Jenner, Rafter and Halliwell45).
Models to study absorption in the colon compared with the small intestine
Although absorption is traditionally associated with the small intestine, the colon is also very capable of absorbing compounds; normally it is the supply of nutrients that limits absorption in the colon. There are several different models for studying absorption at these different sites. The most commonly used is to give a single bolus dose of a food or compound to volunteers and collect blood and urine over the next 24–48 h period. Compounds which are absorbed in the small intestine normally appear in the plasma within an hour and the maximum concentration of a compound at any time point in a pharmacokinetic study is usually less than 2·5 h, although this depends on how full the stomach is at the start, i.e. whether a meal was given or not. Compounds that appear after 3 h with T max (a time point corresponding to maximum concentration of a compound at any time point in a pharmacokinetic study) >5 h are generally considered to have been absorbed mainly in the colon. The latter situation involves, almost without exception, a chemical or microbial transformation which converts the compound from non-absorbable to absorbable. The microflora can catalyse two basic steps at this stage: removal of a conjugated chemical moiety such as a sugar or organic acid, and breakdown of the PPT itself. These interactions are illustrated by the flavonol quercetin. Conjugation with a glucose moiety, such as quercetin-4′-O-glucoside found in onions, leads to absorption in the small intestine after deglycosylation by the brush border enzyme lactase phloridzin hydrolase(Reference Day, Cañada and Díaz100), and the T max is < 1 h(Reference Hollman, Buijsman and van Gameren101). In contrast, if the quercetin is conjugated with a rhamnose moiety as in rutin (quercetin-3-O-rhamnoglucoside), a sugar which is not a substrate for the brush border enzyme lactase phloridzin hydrolase, then the compound is untouched in the small intestine and reaches the colon intact, where colonic microflora remove the terminal rhamnose and the glucose moieties(Reference Hollman, Buijsman and van Gameren101). The immediate product, quercetin aglycone, is then either absorbed intact and appears in the plasma as methylated, sulfated and/or glucuronidated conjugates, or is broken down by microflora as described previously. The inferior absorption of the quercetin component of rutin is thus due to degradation by microflora rather than less efficient colon absorption per se. This illustrates the dual role of the colon microflora for absorption and catabolism.
A second approach is to use healthy ileostomists, i.e. subjects who do not have a colon, because it has been removed by colectomy as a result of ulcerative colitis, Crohn's disease, familial polyposis or colon cancer complications. The end of the ileum is brought through the abdominal wall to form a stoma, usually on the lower right side of the abdomen. The GIT contents pass out of the body through the stoma and are collected in an individually fitted drainable pouch, which is worn at all times. The ileal fluids are easily accessible, their nature makes them easier to analyse than some other biological fluids, and ileostomists are a good tool for comparing the catabolism of a given PPT presented in different foods or matrices. There are some limitations: the terminal ileum develops a (minimal) gut microflora, only disappearance is measured, and some of the subjects have increased gut permeability, electrolyte imbalance and a lower production of urine. Nevertheless, this model has been used successfully to examine the absorption of quercetin(Reference Walle, Otake and Walle102–Reference Olthof, Hollman and Buijsman104), flavanols and hydroxycinnamic acids(Reference Olthof, Hollman and Buijsman104, Reference Auger, Mullen and Hara105), apple polyphenols(Reference Kahle, Kraus and Scheppach106), blueberry polyphenols(Reference Kahle, Kraus and Scheppach107), cider dihydrochalcones(Reference Marks, Mullen and Borges108) and coffee hydroxycinnamates(267).
An alternative approach is to use sections of rat intestine; these are everted and then suspended in tissue culture, where they exhibit functionality for a short time ( < 1 h). These rat everted sacs can then be tested for transport of various substances as required. The difference between absorption of phenolic acids in the small intestine and colon of the rat was examined using this model(Reference Poquet, Clifford and Williamson109). Ascending and descending colon transported dihydrocaffeic and dihydroferulic acids to the serosal (blood) side 1·5- and 3-fold faster than jejunal segments, implying a more efficient absorption in the colon for these catabolites, both major compounds derived from microbial catabolism of hydroxycinnamic acids and some flavonoids (Table 2). In addition, the two colon segments were much more efficient at effluxing glucuronides back to the luminal side(Reference Poquet, Clifford and Williamson109).
The Caco-2 cell model is very commonly used as a model of small intestinal absorption. This cell line is very well characterised(Reference Hayeshi, Hilgendorf and Artursson110–Reference Sambuy, De Angelis and Ranaldi112) but, unlike the small intestine and especially the colon, Caco-2 cells do not secrete mucus. The cell line was derived originally from colonic cells but monolayers of the cells differentiate to produce a small intestine-like morphology, with expression of sucrase and maltase, markers of the small intestine. An alternative colon model using combinations of Caco-2 (76 %) and mucus-secreting HT-29 goblet cells (24 %) has been reported(Reference Poquet, Clifford and Williamson109). The presence of mucus in the co-culture system reduced ferulic acid transport by 23 %, and the HT29 cell component alone was responsible for glucuronidation of the supplied ferulic acid. The co-culture system also reduced the hydroxycinnamic acids to the respective dihydrocinnamic acids, and in this system, dihydroferulic acid was more efficiently transferred to the basolateral side than dihydrocaffeic acid, in contrast to the system using rat everted sacs(Reference Poquet, Clifford and Williamson109).
Polyphenol content of blackcurrants and most abundant predicted catabolites from colonic microflora
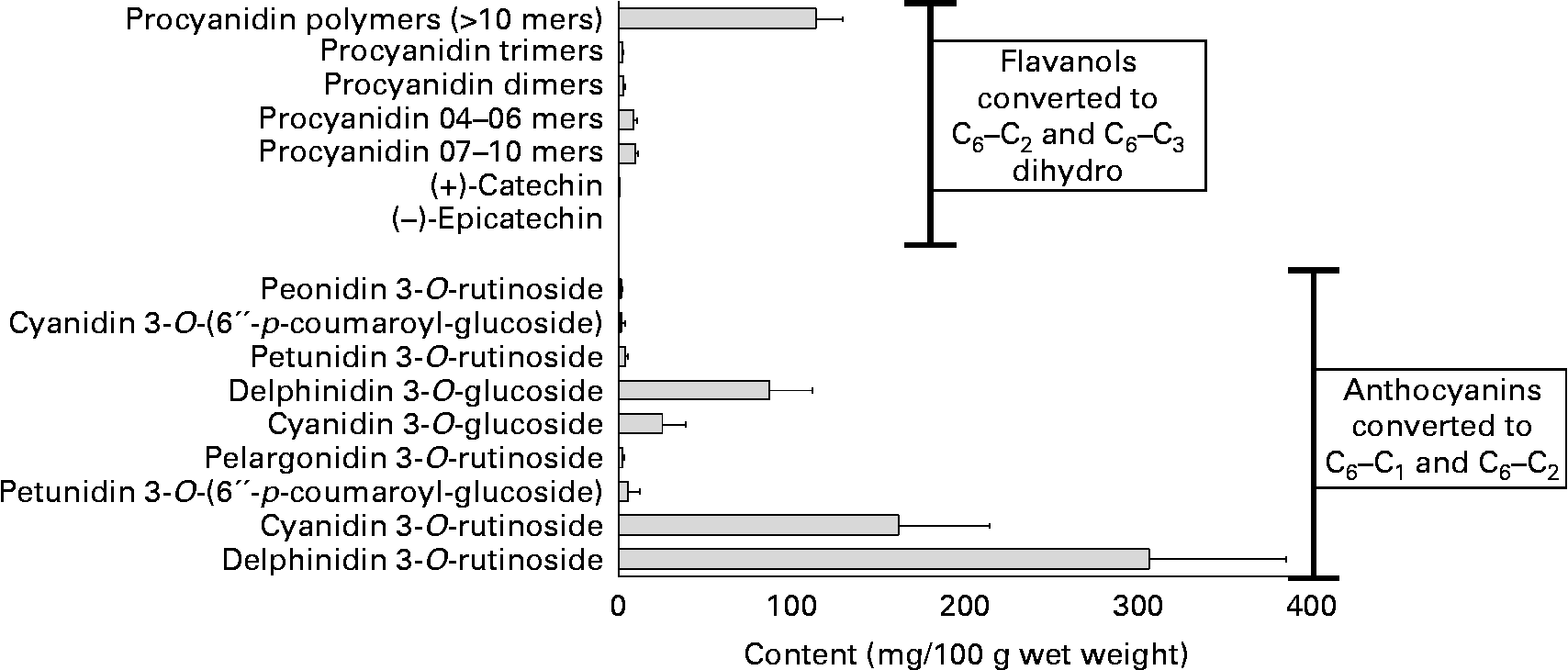
The PPT content of the diet has been reviewed(Reference Crozier, Jaganath and Clifford7, Reference Lindsay and Clifford113–Reference Crozier, Yokota, Jaganath, Crozier, Clifford and Ashihara115). Berries are among the foods richest in polyphenols, many having an especially high content of anthocyanins, and some (e.g. blueberries) being rich also in chlorogenic acids. Blackcurrants contain various anthocyanins at a high-total concentration (approximately 600 mg/100 g with the 3-O-glucosides and 3-O-rutinosides of cyanidin and delphinidin usually dominant), a relatively high level of proanthocyanidins (approximately 140 mg/100 g, consisting of procyanidins and prodelphinidins), some flavonols (approximately 14 mg/100 g) and hydroxycinnamates (approximately 13 mg/100 g), and lower levels of hydroxybenzoic acids (approximately 1·5 mg/100 g) and catechins (approximately 1·2 mg/100 g)(Reference Maatta, Kamal-Eldin and Torronen116, Reference Gu, Kelm and Hammerstone117) (Figs. 2 and 3).
Content of anthocyanidins and flavanols in blackcurrants (http://www.phenol-explorer.eu/, accessed October 2009).
Content of flavonols, cinnamates, benzoic and gallic acid derivatives in blackcurrants (http://www.phenol-explorer.eu/, accessed October 2009).

The next section of this review on the GIT transformation of dietary PPT is concerned with the potential for blackcurrants and blackcurrant-derived products to influence human health. Much of the information is derived from studies of similar PPT from other food sources. Table 2 shows that the classes of aromatic/phenolic acids that might be expected to form in the GIT tract is largely independent of the classes of PPT consumed. The dominance in blackcurrants of anthocyanins and proanthocyanidins will result primarily in the production of C6–C1 and C6–C3 dihydro acids, with some C6–C5-γ-OH products to be expected early in the degradation of the proanthocyanidins and an increasing yield of C6–C1 acids at later time points as the side chain is progressively shortened either by the microflora or by the mammalian enzyme systems. A percentage of the cinnamic acids might be absorbed as such but these also are reduced to the C6–C3 dihydro acids: the flavonols will yield C6–C2 acids, and these might also form from α-oxidation of C6–C3 dihydro acids and yield C6–C1 acids by the same mechanism.
The ring (C6) hydroxylation pattern depends primarily on the B-ring hydroxylation and the precise biochemical competence of the gut microflora. As summarised in Table 3, the dominance of cyanidin and delphinidin glycosides suggests that initially protocatechuic and gallic acid are likely to be the dominant C6–C1 acids. However, in human subjects, gallic acid consumed as flavanol-3-O-gallates is rapidly converted to 3-O-methylgallic acid, 4-O-methylgallic acid and 3,4-O-dimethylgallic acid(Reference Hodgson, Morton and Puddey118, Reference Shahrzad and Bitsch119). Although free gallic acid has been detected in human plasma after an oral dose (50 mg)(Reference Shahrzad and Bitsch119), this mammalian metabolism is consistent with the failure to observe gallic acid in plasma after volunteers had consumed delphinidin(Reference Nurmi, Mursu and Heinonen5).
The expected B-ring fragments for the common anthocyanidins and their known mammalian metabolites

Evidence for microbial biotransformations from animal and human intervention studies
Anthocyanins
The absorption and catabolism of anthocyanins from blackcurrants in rats and human subjects have been studied(Reference Matsumoto, Inaba and Kishi120). Four anthocyanins, delphinidin-3-O-rutinoside, cyanidin-3-O-rutinoside, delphinidin-3-O-glucoside and cyanidin-3-O-glucoside, were absorbed and excreted as the glycosylated forms in both rats and human subjects. In human subjects, only 0·05 % of the ingested anthocyanin dose was found in urine. Although, unexpectedly, the 3-O-rutinosyl anthocyanins were directly absorbed, the amount found in the urine was very low(Reference Matsumoto, Inaba and Kishi120). Co-administration of anthocyanins with phytic acid enhances plasma concentration significantly by an unknown mechanism(Reference Matsumoto, Ito and Yonekura121). In all studies on healthy subjects consuming anthocyanin-rich food, it is now well established that < 2 % of the total anthocyanin dose is absorbed intact, as estimated from the amount of anthocyanin and conjugates either in urine(Reference Felgines, Talavera and Gonthier122) or in blood(Reference Mullen, Edwards and Serafini123). Using ileostomist subjects, a high proportion of the anthocyanins from blueberry (up to 85 %, depending on the attached sugar moiety) traversed the small intestine unchanged and were found in the ileostomy bags; this amount would therefore reach the colon under physiological conditions and be subject to microbial degradation(Reference Kahle, Kraus and Scheppach107). In vitro studies using human microflora in an anaerobic chamber have indicated that protocatechuic acid is one of the most likely major degradation products from anthocyanins having a 3,4-dihydroxy B-ring(Reference Aura, Martin-Lopez and O'Leary23). In a human intervention study on blood orange juice, the C max in blood for cyanidin-3-O-glucoside was 1·9 nm, whereas the value for protocatechuic acid was approximately 250-fold higher (492 nM). It was calculated that the main product, protocatechuic acid, accounted for up to 70 % of the anthocyanin intake. Only 1·2 % of the anthocyanin dose ultimately appeared in urine, whereas urinary protocatechuic acid represented 28 % of the total anthocyanin dose. The protocatechuic acid appeared to be unconjugated(Reference Vitaglione, Donnarumma and Napolitano4). Protocatechuic acid was also found in rat plasma after feeding cyanidin-3-O-glucoside(Reference Tsuda, Horio and Osawa124) and after deglycosylation, cyanidin can breakdown spontaneously to give protocatechuic acid (very pronounced at physiological pH)(Reference Kay, Kroon and Cassidy91). After ingestion of black raspberries by pigs, the profile of compounds in the gastrointestinal tract was analysed. In the entire gut, protocatechuic acid was the major phenolic, followed by 4-hydroxycinnamic, caffeic, ferulic and 3-hydroxybenzoic acids. These accounted for approximately 6 % of the ingested anthocyanin(Reference Wu, PittmanIii and Hager125) which is considerably less than the human study described previously.
After consumption of a high dose of strawberries by volunteers, the main phenolic acids in urine after 5 h were 4-hydroxybenzoic acid (10·4 mg/l), protocatechuic acid (0·7 mg/l), vanillic acid (1 mg/l) and genistic acid (1 mg/l)(Reference Russell, Scobbie and Labat126). Strawberry anthocyanins are primarily pelargonidin glycosides with lesser amounts of cyanidin glycosides(Reference Felgines, Talavera and Gonthier122, Reference Hollands, Brett and Dainty127) that would be expected to yield 4-hydroxybenzoic acid and protocatechuic acid (Table 3), but these acids are normal preformed constituents of strawberries(Reference Stohr and Herrmann128).
Consumption of oats added to a purée of bilberries (glycosides of delphinidin, accompanied by lesser amounts of malvidin, peonidin and petunidin glycosides)(Reference Ichiyanagi, Kashiwada and Ikeshiro129–Reference Ichiyanagi, Hatano and Matsugo131) and lingonberries (cyanidin glycosides)(Reference Ek, Kartimo and Mattila132) by volunteers resulted in urinary excretion of 3-methoxy-4-hydroxyphenylacetic (homovanillic) and vanillic acid, low amounts of syringic acid (from malvidin glycosides) but no gallic acid (which would have been expected from delphinidin glycosides). Urinary excretion of these acids was maximal at 4–6 h and intact urinary anthocyanins comprised < 0·01 % of the dose(Reference Nurmi, Mursu and Heinonen5).
Procyanidins
When rats were fed procyanidin dimer B3, the major urinary catabolites were 3-(3′-hydroxyphenyl)-propionic, 3-hydroxycinnamic, 4-hydroxybenzoic and vanillic acids (total 6·5 % of intake). Feeding of the procyanidin trimer C3 or a mixture of procyanidin polymers gave in addition 3-hydroxyphenylvaleric acid (C6–C5)(Reference Gonthier, Donovan and Texier133). When rats were fed 14C-labelled procyanidin B2(Reference Stoupi, Williamson and Viton134), approximately 80 % of the 14C-radiolabel was absorbed and appeared in the urine. The 14C-radiolabel appeared rapidly in the blood at low levels, but did not reach a maximum until 6 h, indicative of a major contribution of catabolism by colonic microflora. These two studies indicate that the majority (>70 %) of the colonic catabolites of procyanidin dimers are unknown. In human subjects, after consumption of procyanidin and catechin-rich chocolate, the main urinary catabolites were 3-(3′-hydroxyphenyl)-propionic acid, ferulic acid, 3,4-dihydroxyphenylacetic acid, 3-hydroxyphenylacetic acid, vanillic acid and 3-hydroxybenzoic acid(Reference Rios, Gonthier and Remesy135).
Hydroxycinnamic acids
Hydroxycinnamic acids are most commonly found linked to a quinic acid moiety in fruits and foods. These compounds are called chlorogenic acids and the most common are caffeoyl-quinic acids, found at high levels in coffee. Fibre such as wheat bran is another source, where the hydroxycinnamic acids, particularly ferulic acid, are covalently linked to an arabinose sugar unit. Much of the understanding of the metabolism of hydroxycinnamates is derived from work on coffee. Chlorogenic acids are poorly hydrolysed by conditions in the stomach or small intestine. When coffee is given, there is a relatively small absorption of caffeic and ferulic acids in the small intestine and a low absorption of intact chlorogenic acids. The major absorption occurs in the colon, where dihydroferulic and dihydrocaffeic acids, products of microbial biotransformation, are the major products(Reference Stalmach, Mullen and Barron136, Reference Renouf, Guy and Marmet137). The important involvement of the colon is also supported by studies on ileostomists, where approximately 67 % of the dose of chlorogenic acids reaches the colon(Reference Olthof, Hollman and Katan138). Dihydrocaffeic acid is one of the major phenolic acids in human faecal water(Reference Jenner, Rafter and Halliwell45) and has been detected in the plasma of coffee drinkers(Reference Goldstein, Stull and Markey139), in urine as the free form and mainly conjugated in human plasma after ingestion of artichoke leaf extracts(Reference Wittemer, Ploch and Windeck140), in human urine after chocolate intake(Reference Rios, Gonthier and Remesy135) and in rat urine after ingestion of polyphenol-rich wine extract(Reference Gonthier, Cheynier and Donovan141). In summary, the major products from ingestion of chlorogenic acids are dihydrocinnamic acids in the plasma and urine. Studies on rats showed that the major 5-caffeoylquinic acid-derived phenolic acids, 3-hydroxybenzoic, 3-hydroxybenzoylglycine (3-hydroxyhippuric) and 3-hydroxycinnamic acids were from the caffeic acid moiety but that a significant portion of the hippuric acid (benzoylglycine) was produced from the quinic acid moiety(Reference Gonthier, Verny and Besson142).
Some older work on the tissue distribution of radiolabelled cinnamic acids is worth noting. Injection of 14C-cinnamic acid to rats gave distribution in organs as shown in Fig. 4, with the remaining radioactivity in urine (48 %), faeces (25 %) and exhaled CO2 (0·3 %)(Reference Teuchy and van Sumere143). There was a substantial amount in skin and gonads. A later study showed that 82–90 % of orally administered trans-(3-14C)-cinnamic acid was absorbed in rats as indicated by the amount present in urine after 24 h(Reference Nutley, Farmer and Caldwell144).
Distribution of radiolabel in rat tissues after injection of 14C-cinnamic acid to rats. The remaining radioactivity was in urine (48 %), faeces (25 %) and exhaled CO2 (0·3 %)(Reference Teuchy and van Sumere143).

Once formed by microbial biotransformation, compounds must pass the colonic epithelium and enter the bloodstream. Mammalian enzymes will further transform the microbial products, mainly by conjugation, but also by β-oxidation. Conjugation can involve methylation (catalysed by the enzyme catechol-O-methyl transferase), sulfation (sulfotransferases), β-glucuronidation (UDP-glucuronosyl transferases), glycinylation (via a CoA thioester) and glutaminylation (by conjugation with glutamine). Many of the enzymes involved exist as multiple isoforms with different but overlapping specificities, typical of those acting on xenobiotics.
Properties of benzoate derivatives (C6–C1) (e.g. protocatechuic and 4-hydroxybenzoic acids)
Absorption and metabolism of C6–C1
Both protocatechuic acid and phloroglucinaldehyde are transported by Caco-2 cells, and then metabolised to sulphate and glucuronide conjugates by Caco-2 cells(Reference Kay, Kroon and Cassidy91) (Fig. 5). Studies with Caco-2 cells have shown that benzoic acid and the three mono-hydroxybezoic acid isomers are substrates for the monocarboxylate transporter(Reference Haughton, Clifford and Sharp145). 3-Methoxy-4-hydroxyphenylacetic (homovanillic) acid is a substrate for the rat organic anion transporter(Reference Mori, Takanaga and Ohtsuki146). Following absorption, catechol-O-methyl transferase methylates protocatechuic acid to vanillic acid(Reference Cao, Zhang and Ma147). Of the conjugating enzymes, UDP-glucuronosyl transferases 1A6 in human liver microsomes is active on protocatechuic aldehyde(Reference Liu, Liu and Zhang148). In addition, protocatechuic aldehyde is converted to approximately 70 % protocatechuic acid by guinea pig liver slices by the enzymes aldehyde oxidase, xanthine oxidase and aldehyde dehydrogenase(Reference Panoutsopoulos and Beedham149), but approximately 20 % was unidentified polar conjugates of protocatechuic acid. The conversion of caffeic acid to protocatechuic acid did not occur in the absence of a gut microflora in germ-free rats(Reference Peppercorn and Goldman34).
Summary of absorption and metabolic pathways of C6–C1 and C6–C3 compounds in the gastrointestinal tract.

Biological effects of C6–C1
In a rat model of induced carcinogenesis, protocatechuic acid prevented 4-nitroquinoline-1-oxide-induced oral carcinogenesis, N-methyl-N-nitrosourea-induced carcinogenesis in the glandular stomach, azoxymethane-induced carcinogenesis in the colon and diethylnitrosamine-induced carcinogenesis in the liver(Reference Tanaka, Kawamori and Ohnishi150, Reference Tanaka, Kojima and Kawamori151). The doses of protocatechuic acid were expressed only as a percentage of the diet, but can be estimated as approximately 10–40 mg/kg body weight per d assuming that a 200 g rat typically consumes 20 g diet per d.
Several papers have reported in vitro activity of protocatechuic acid on cultured cells, and in some investigations, this microbial catabolite is more potent than cyanidin glycoside, an anthocyanin that gives rise to substantial levels of protocatechuic acid in vivo (Reference Vitaglione, Donnarumma and Napolitano4). For example, in human neuronal SH-SY5Y cells, protocatechuic acid (100 μm) is more potent than cyanidin-3-O–glucoside (100 μm) at inhibiting hydrogen peroxide-induced apoptotic events, including mitochondrial function and DNA fragmentation. However, neither was effective below 25 μm(Reference Tarozzi, Morroni and Hrelia152). Protocatechuic acid induces c-jun N-terminal kinase-dependent hepatocellular carcinoma cell death at concentrations>50 μm(Reference Yip, Chan and Pang153).
Protocatechuic acid at concentrations above 500 μm promoted time-dependent and concentration-dependent migration of human adipose-tissue derived stromal cells in vitro possibly via effects on matrix metalloproteinase-2(Reference Wang, Liu and Guan154), and promoted cell proliferation of cultured rat neural stem cells, with reduced apoptosis possibly via repression of caspase-3 activation(Reference Guan, Ge and Liu155). Significantly lower production of reactive oxygen species was seen following treatment with protocatechuic acid (6 μm for 7 d or 30 μm for 4 d) but suppression of caspase-3 required 30 μm for both durations. The relative effectiveness of lower doses longer term is encouraging and suggests that dietary exposure over a long period might perhaps promote better health over the subsequent years.
Several investigations have addressed lipid and cholesterol metabolism. Protocatechuic acid, the major component of the Chinese functional medicine, Danshen, has reported anti-angina efficacy(Reference Cao, Zhang and Ma147). Protocatechuic acid is methylated and then diffuses into mitochondria where it is conjugated with CoA. The result is that fatty acid oxidation is decreased, as shown by a lower acyl CoA/CoA ratio in heart, which in turn activates pyruvate dehydrogenase, a key and irreversible step in carbohydrate oxidation. This could switch heart energy substrate preference from fatty acid to glucose that would be beneficial for ischaemic heart conditions. There was no detectable accumulation of protocatechuic acid in tissues(Reference Cao, Zhang and Ma147). Protocatechuic acid at 0·5 g/kg diet given to rats (estimated as approximately 10 mg/kg body weight per d) produced changes in cholesterol and lipid metabolism. The serum total cholesterol, HDL-cholesterol and VLDL-cholesterol were all lower in the protocatechuic acid-treated group, suggested to be partly as a result of induction (estimated by mRNA) of hepatic LDL receptor, apoB, apoE, lecithin-cholesterol acyltransferase and hepatic TAG lipase(Reference Tamura, Fukushima and Shimada156).
Properties of phenylacetate derivatives (C6–C2)
Absorption and metabolism of C6–C2
Phenylacetate is an endogenous product of phenylalanine metabolism, is present at low levels in the mammalian circulation and is conjugated with glutatmine during metabolism. Its pharmacokinetic parameters have been well studied in a clinical setting in patients owing to its proposed effects on cancer. When phenylacetate was administered as an intravenous infusion as part of a phase I trial in children with refractory cancer, the half life was approximately 1 h, and phenylacetate was conjugated with glutatmine to form phenylacetylglutatmine(Reference Thompson, Balis and Serabe157). After oral consumption of phenylacetate, the concentration of phenylacetate peaked at 2 h and 40 % was excreted in the urine after 40 h(Reference Wajngot, Chandramouli and Schumann158). Intravenous radiolabelled 14C-phenylacetate was rapidly taken up by rat brain and converted into 14C-acetate(Reference Inoue, Hosoi and Momosaki159). Phenylacetate was well tolerated when infused into patients twice per d for weeks at a high dose (125 mg/kg body weight), and at these levels, phenylacetate induced its own clearance by 27 % during this period(Reference Thibault, Samid and Cooper160). Both phenylketonuric and normal subjects eliminated an oral dose (80 mg) of [14C]-phenylacetic acid in the urine almost entirely as phenacetylglutamine, showing that the glutamine conjugation mechanism is not defective in phenylketonuria and that it is able to cope with the large amounts of phenylacetic acid produced in this disorder(Reference James and Smith161). The contents of the colon, examined as human faecal water, contained phenylacetic acid, 3-phenylpropionic acid, 3-hydroxyphenylacetic acid, 3,4-dihydroxyphenylacetic acid, 3-(4′-hydroxyphenyl)-propionic acid and 4-hydroxy-3-methoxycinnamic (ferulic) acid at high levels, up to 400 μm for phenylacetic acid and 3-phenylpropionic acid in one individual. The mean levels were 188, 197, 110, 64, 61 and 10 μm, respectively(Reference Karlsson, Huss and Jenner162).
Biological effects of C6–C2
Phenylacetic acid (administered as sodium phenylacetate) is one component of a drug (together with sodium benzoate) which is given for the treatment of acute hyperammonemia(Reference MacArthur, Altincatal and Tuchman163). As discussed previously, the pharmacokinetic behaviour is well characterised(Reference MacArthur, Altincatal and Tuchman163), and phenylacetylglutatmine and hippuric acids are products excreted in the urine after an intravenous dose. Phenylbutyrate, a prodrug that is metabolised into its active component, phenylacetate, in vivo, has been reported to extend lifespan in Drosophila (Reference Kang, Benzer and Min164). Large doses of phenylbutyrate can have some toxic side effects(Reference Kasumov, Brunengraber and Comte165), and it has been shown that phenylacetate and phenylbutyrate modulate medulloblastoma(Reference Li, Parikh and Shu166). Some C6–C2 compounds may modulate cholesterol metabolism (see below). When given orally, a single high dose of phenylacetate did not affect plasma glucose concentration nor gluconeogenesis in type II diabetic patients(Reference Wajngot, Chandramouli and Schumann158). Phenylacetate affects cell growth and proliferation(Reference Li, Parikh and Shu166–Reference Shibahara, Onishi and Franco168), inhibits prenylation and has been tested in both phase I and II clinical trials. 3,4-Dihydroxyphenylacetic acid exhibited antiproliferative activity on prostate and colon cancer cells(Reference Gao, Xu and Krul33). 3-Phenylpropionic acid, 3-hydroxyphenylacetic acid and 3-(4′-hydroxyphenyl)-propionic acid, found at high concentrations in faecal water, decreased the expression of cyclo-oxygenase-2 in HT29 cells at physiologically relevant concentrations(Reference Karlsson, Huss and Jenner162).
Properties of phenylpropionate derivatives (C6–C3) (e.g. dihydrocaffeic and dihydroferulic acids)
Absorption and metabolism of C6–C3
As summarised in Table 2, C6–C3 acids can be formed from many PPT substrates including proanthocyanidins. However, information on the absorption and metabolism of C6–C3 acids has come largely from studies of chlorogenic acids, conjugates of cinnamic acids with quinic acid. Such conjugates are widespread in foods and beverages, but coffee, artichoke, apples and blueberries are particularly rich(Reference Crozier, Jaganath and Clifford7, Reference Clifford169, Reference Clifford170) Approximately, 1 h after the consumption of coffee, ferulic, caffeic and isoferulic acids appear in the plasma at low levels indicating absorption in the small intestine(Reference Renouf, Guy and Marmet137). This early appearance was characterised in more detail, and the circulating compounds included caffeic acid-3-O-sulphate and ferulic acid-4-O-sulphate, and sulphates of 3- and 4-caffeoylquinic acid lactones(Reference Stalmach, Mullen and Barron136). These lactones are formed from the chlorogenic acids during coffee roasting(Reference Clifford169) and are rarely found in other foods or beverages. Several hours later (T max >4 h), greater concentrations of dihydroferulic acid, dihydroferulic acid-4-O-sulphate and dihydrocaffeic acid-3-O-sulphate appear in blood, indicating colonic absorption after microbial transformation(Reference Renouf, Guy and Marmet137). Dihydroferulic acid, dihydroferulic acid-4-O-sulphate, dihydrocaffeic acid-3-O-sulphate, ferulic acid-4-O-sulphate, dihydroferulic acid-4-O-glucuronide and feruloyl-glycine were major urinary components. These catabolites collectively represented approximately 29 % of the chlorogenic acid dose, indicating good absorption of the microbial catabolites of chlorogenic acids(Reference Stalmach, Mullen and Barron136).
The colon pH value ranges from pH 5·7 to 6·7(Reference Fallingborg171), and unless there are localised pockets of significantly lower pH value, these phenolic acids with pK values in the range pH 4–5(Reference Jovanovic, Steenken and Tosic172) will be extensively ionised. The absorption of C6–C3 catabolites has been examined using Caco-2 cells and rat everted intestinal sacs (Fig. 5). Dihydrocaffeic acid is absorbed mainly by the passive transcellular route and enters the circulation mainly as the free form, although a small proportion is glucuronidated(Reference Poquet, Clifford and Williamson109). The dihydrocaffeic acid is either rapidly sulphated, or methylated then sulphated, by the liver(Reference Poquet, Clifford and Williamson173), so that the major metabolites in the circulation are free dihydroferulic acid, dihydrocaffeic acid-3-O-sulphate or dihydroferulic acid-4-O-sulphate. Dihydroferulic acid in the gut is absorbed partially by the passive transcellular route, but with some involvement of a monocarboxylic transporter(Reference Poquet, Clifford and Williamson109, Reference Konishi and Kobayashi174). 3-Hydroxycinnamic acid and its dihydroform, 3-(3′-hydroxyphenyl)-propionic acid, are both absorbed at least partially by the monocarboxylic transporter in Caco-2 cells(Reference Konishi and Kobayashi175).
Once absorbed in the colon, sulphate conjugation occurs primarily in the liver. In human subjects, sulphotransferase 1A1 is the most active isoform for sulfation of caffeic, isoferulic and dihydrocaffeic acids, whereas sulphotransferase 1E1 is most active towards the other methylated forms, i.e. ferulic and dihydroferulic acids(Reference Wong, Meinl and Glatt176). Glucuronidation occurs to a much lesser extent. The levels of dihydrocaffeic acid-3-O-sulphate were approximately 45-fold greater than the corresponding glucuronides, and the amount of dihydroferulic acid-4-O-sulphate in urine was much greater than the amount of dihydroferulic acid-4-O-glucuronide. The only cinnamic acid for which the glucuronides predominated was isoferulic acid(Reference Wong, Meinl and Glatt176).
Cinnamic acids, as distinct from the dihydrocinnamic acids, have rarely been reported as gut flora catabolites of flavonoids. Rats excrete 4-hydroxycinnamic acid in urine after the consumption of apigenin, phloridzin and naringenin(Reference Griffiths and Smith56).
Biological effects of C6–C3
Excess solar UV radiation produces damage and initiates immune response and inflammation in skin, sometimes leading to cancer. Dihydrocaffeic and caffeic acids, but not dihydroferulic and ferulic acids, reduced the cytotoxicity and pro-inflammatory cytokine production (IL-6 and -8) in HaCaT cells, a keratinocyte model, following UV radiation(Reference Poquet, Clifford and Williamson177), and dihydrocaffeic and 4-hydroxycinnamic acids inhibited UV-B damage in human conjunctival cells as assessed using the DNA damage marker, 7,8-dihydro-8-oxo-2′-deoxyguanosine(Reference Larrosa, Lodovici and Morbidelli178). 4-Hydroxycinnamic acid, but not protocatechuic acid, decreases basal oxidative DNA damage in rat colonic mucosa(Reference Guglielmi, Luceri and Giovannelli179). In CCD-18 colon fibroblast cells stimulated with IL-1β, dihydrocaffeic, dihydroferulic and dihydroxyphenylacetic acids attenuated PG E2 demonstrating an anti-inflammatory effect in this system. In addition, dihydrocaffeic acid diminished the expression of the cytokines IL-1β, IL-8 and TNF-α, reduced malonyldialdehyde levels and reduced oxidative DNA damage (measured as 7,8-dihydro-8-oxo-2′-deoxyguanosine) in distal colon mucosal tissue in the dextran sodium sulphate-induced colitis model in rats(Reference Larrosa, Luceri and Vivoli180). Animal and studies in vitro also suggest that some C6–C2(Reference Bhat and Ramasarma181) and especially C6–C3 (Reference Lee, Choi and Jeon182) catabolites interfere with various enzymes in the mevalonate pathway including 3-hydroxy-3-methylglutaryl-CoA reductase, the rate limiting enzyme in hepatic cholesterol biosynthesis, albeit at concentrations unlikely to occur in plasma. However, these observations are of interest since commodities rich in PPT that would yield such catabolites, and the catabolites when given per os, have been shown to inhibit platelet aggregation(Reference Yasuda, Takasawa and Nakazawa183) or to have cholesterol-lowering activity in animal(Reference Lee, Choi and Jeon182, Reference Bok, Lee and Park184–Reference Yamakoshi, Kataoka and Koga188) and human studies(Reference Kurowska, Spence and Jordan189), and such gut flora catabolites may have contributed to the in vivo effect. Interference in the mevalonate pathway, particularly 3-hydroxy-3-methylglutaryl-CoA reductase inhibition, may have broader human significance(Reference Mo and Elson190).
Properties of phenylvalerate derivatives (C6–C5)
Following the consumption of green tea flavanols or proanthocyanidin-rich extracts, several C6–C5 catabolites have been detected in human plasma and urine as methyl and/or glucuronide/sulphate conjugates(Reference Lee, Maliakal and Chen191–Reference Duweler and Rohdewald193).
Prebiotic effects of polyphenols on the microflora
Although the fact that biotransformation of dietary PPT by the gut microflora occurs has been known for many years, the possibility that either the untransformed PPT or the phenolic acid catabolites might modify the composition and biochemical competence of the gut microflora has attracted comparatively little attention. Changes over 20 d in the catabolites produced from punicalagin have been attributed to changes in the GIT microflora(Reference Cerda, Llorach and Ceron97). There is evidence from in vitro models(Reference Lee, Jenner and Low31, Reference Tzounis, Vulevic and Kuhnle32) and in vivo studies using human subjects, pigs and sheep that PPT and/or their catabolites influence the composition of the gut microflora, for example lowering the colonic pH value, suppressing bacteroides and pathogenic Clostridium perfringens and Clostridium difficile, and increasing the proportion of bifidobacteria and eubacteria without inhibiting lactic acid bacteria(Reference Lee, Jenner and Low31, Reference Goto, Kanaya and Nishikawa194–Reference Okubo, Ishihara and Oura196), but the exact mechanisms are uncertain(Reference Min, Attwood and Reilly197). The concentrations of benzoic acid, phenylacetic acid, phenylpropionic acid and 3-(3′-hydroxyphenyl)-propionic acid in human faecal water can each reach millimolar concentrations(Reference Knust, Erben and Spiegelhalder44, Reference Jenner, Rafter and Halliwell45) which has the obvious potential to modulate bacterial growth.
Caffeic acid, a comparatively minor component, can reach concentrations (approximately 50 μm)(Reference Jenner, Rafter and Halliwell45) well in excess of the concentration shown in vitro to produce 50 % inhibition in the growth of the opportunistic pathogen Listeria monocytogenes (Reference Ramos-Niño, Ramìrez-Rodriguez and Clifford198). Various flavonoid aglycones are also found in faecal water, but at low concentrations ( < 3 μm). There is some evidence also that some pathogenic protozoa are inhibited by some flavonoids (IC50 or EC50 values in the range 15–50 μm)(Reference Gargala, Baishanbo and Favennec199, Reference Kerboeuf, Riou and Guegnard200) but the effect of the aromatic/phenolic acid catabolites does not seem to have been investigated. Although the yield of phenolic/aromatic acids is variable (up to × 10) between individuals and is not necessarily normally distributed(Reference Knust, Erben and Spiegelhalder44, Reference Jenner, Rafter and Halliwell45, Reference Li, Lee and Sheng201, Reference Clifford, Copeland and Bloxsidge202), the potential for suppressing some pathogens and for a prebiotic effect in some individuals clearly exists.
Summary, conclusions and recommendations for future research
The colonic microbiota transform a very complex range of PPT substrates that will vary between individuals and occasions. These transfomations can be extensive, and while some microbial catabolites are substrate-specific (equol, urolithins, mammalian lignans), certain catabolites are common to many of the major substrates, and this implies that the spectrum of catabolites produced is less complex and qualitatively less variable than the spectrum of substrates consumed. The catabolites most likely to dominate are the C6–C1, C6–C2 and C6–C3 dihydro acids derived from cinnamates, anthocyanins and most other flavonoids. These microbial catabolites are often better absorbed than the parent compounds, because of the mechanism of absorption, the large absorptive area available in the colon and the high concentrations (approaching millimolar) in the colonic lumen.
Therefore, we propose that these microbial catabolites could be responsible for a significant proportion of the biological activity derived from consumption of fruits, vegetables and other plant derived products such as fruit drinks, wine, coffee and tea.
The biological activity of these microbial catabolites would be in addition to the biological activity of any absorbed parent compound and its mammalian metabolites, and the potential for synergy between microbial catabolites and the parent compound and its mammalian metabolites could be important but has not been studied. Because of the potential importance of the microbial catabolites, we need to understand better the factors controlling (i) their production and whether this can be modulated advantageously, (ii) their absorption and mammalian metabolism and (iii) their biological activities both in vitro and in vivo.
The existing epidemiological data must be revisited and extended so as to take account of substances not present in the diet per se. For a given substrate consumption, there is often a large inter-individual variation (approximately × 10) in catabolite yields, subsequent plasma concentrations and also variation in the concentration–time profile. It is essential to define the factors responsible, whether manifest in the microflora, in the host or in both. Similarly it is important to determine the potential of PPT and their catabolites to modulate the gut microflora and hence potentially their own production. The answers to these are crucial if we are to understand and exploit the effect of PPT consumption on human health.
Acknowledgements
We acknowledge funding from GlaxoSmithKline to G. W. and M. N. C. for writing this review. There are no conflicts of interest. G. W. and M. N. C. contributed 50 % each to the writing of the present paper.









