It is frequently assumed that dementia is only a disorder of cognitive impairment; however, dementia is often accompanied by significant motor disability due in part to paratonia, a form of increased muscle tone. First observed by Dupre in 1910, Reference Dupre1 it is characterized as an inability to relax muscles in the setting of cognitive impairment.
Paratonia has been estimated to be present in 5% of those with mild cognitive impairment and 100% in those with advanced dementia. Reference Souren, Franssen and Reisberg2 Initially, in its earliest stages, paratonia may manifest as facilitatory (mitgehen), where a person is unable to relax and “helps” the examiner when a limb is moved passively. Reference Kral3 Resistive paratonia (geggenhalten) increases proportionately as dementia severity increases. Reference Kleist4 Paratonia exerts its effects by increasing muscle resistance reflexively when a limb is moved passively, resulting in contraction of the muscles. As paratonia can fluctuate in severity depending on level of relaxation, the variability in resistance can often erroneously be interpreted as refusal of care and perceived as voluntary, with negative consequences including conflict between patient and caregiver, Reference Backhouse, Jeon, Killett and Mioshi5 as well as inappropriate utilization of anti-psychotics or other sedative medications to manage the perceived hostile behavior. 6
In advanced dementia, paratonia may also exist at rest, with constant muscle contraction leading to contractures. When muscle is in a prolonged shortened state, rheologic changes lead to physical shortening of the muscle and other structures such as fascia, nerves, blood vessels and skin; this state defines muscle contracture, Reference Franssen, Kluger, Torossian and Reisberg7 which is a fixed orthopedic deformity. As such, provision of care may cause pain and suffering for the person with dementia, as well as increased caregiver burden for the family, professional caregiver and the healthcare system due to costly complications.
Symptomatic paratonia, a manifestation of advanced dementia, is especially prevalent in long-term care (LTC) facilities as it is estimated that ∼ 70% of residents of LTC in Canada have dementia. 8 In addition to difficulty in bathing/provision of hygiene, dressing, feeding, transferring and general care, contractures may lead to skin breakdown and decubitus ulcers, infection and spontaneous fractures. Approximately 30% of residents of non-acute care settings in Canada have been estimated to manifest pressure ulcers, Reference Woodbury and Houghton9 with a cost to the Canadian Health Care System of $2.1 billion annually. 10 Though paratonia occurs frequently and is disabling, there is minimal research regarding its etiology, natural history or strategies to treat or prevent it. In 2014, we published a randomized, placebo-controlled double-blind pilot study of botulinum toxin A (BoNT-A) administration in patients with dementia and paratonia causing involuntary postures in the upper limbs. Results confirmed a significant reduction in caregiver burden and demonstrated an increase in range of motion in upper limbs, as hypothesized. Reference Kleiner-Fisman, Khoo and Moncrieffe11 BoNT-A injections to address paratonia is not an approved indication and its use is off-label.
In this study, we sought to identify knowledge and real-world practices of injectors of BoNT-A in Canada for the indication of paratonia in dementia. We conducted a national survey of Canadian physiatrists and neurologists who are established BoNT-A injectors for muscle hypertonia, to determine awareness and recognition of paratonia, “real-world” practices, and determine gaps and barriers in care, to inform future study priorities.
Survey recipients were selected based on convenience sampling methods and the following inclusion criteria: (a) Neurologist or Physiatrist in Canada and (b) BoNT-A injector for muscle hypertonia. The identification of eligible survey recipients meeting these criteria included the leveraging of the study authors’ clinical and research networks of Canadian BoNT-A injectors, and reviewing literature of authors of Canadian BoNT-A Clinical Guidelines publications in spasticity and dystonia, to identify eligible study participants. The initial survey items were developed and reviewed by the study authors for relevant content in an iterative process. Beta-testing of the survey was conducted using exhaustive sample responses to ensure flow, functionality and to avoid survey fatigue. Introduction to the project was sent to survey recipients by email, along with a link to the survey (supplementary material 1) hosted on the Qualtrics XM platform. Survey recipients were invited to respond anonymously.
The purpose and content of the survey was approved by the Baycrest Health Sciences Hospital Research Ethics Board. No identifiable data were collected and participants responded to the survey anonymously. Survey recipients were informed of the purpose of the survey, conflict of interest disclosures, estimated length of time to complete the survey (10 minutes), and were asked to consent to the use of the data in the aggregate in a research publication or as part of a commercial development program. Survey recipients were informed that completion of the survey would be viewed as consent to participate in the research.
The survey was distributed on three occasions over a period of 4 weeks, with each round eliciting an additional number of respondents. The survey was distributed to 67 physiatrists and neurologists. There were 36 (54%) respondents to the survey. However, of these 36 respondents, only 27 (40%) were included in the analysis due to some respondents not completing a sufficient number of items for analysis. The participant tree is provided in Supplementary Material 2. Descriptive statistics were performed to summarize survey responses across the final dataset of 27 respondents.
Of the 27 respondents included in the analysis, there were 18 (67%) physiatrists and nine (33%) neurologists. Figure 1a displays the Canadian provinces where the respondents practice and their distribution, separately for physiatrists and neurologists. Almost 60% percent of respondents are from Ontario. Figure 1b presents the settings where physiatrists and neurologists treat paratonia patients including LTC, Clinic/Office, Rehabilitation Facility and Acute Care Hospital. 78% of physiatrists and 29% of neurologists that treat paratonia work in LTC. Conversely, 100% of neurologists and 33% of physiatrists work in clinic or office settings. 44% of physiatrists work in a rehabilitation facility. No neurologists work in a rehabilitation facility. Only 11% of physiatrists and 14% of neurologists treat paratonia in acute-care settings. These responses were not discrete as respondents were able to select multiple settings.
a: Canadian province where physiatrist and neurologist respondents practice (N = 27). b: Treatment setting where physiatrists and neurologists treat patients with paratonia with botulinum toxin A (BoNT-A) (N = 25). c: Treatment modalities used to treat paratonia (N = 25). *PRMT: passive range of motion therapy. d: Years of experience treating paratonia with botulinum toxin A (BoNT-A) (N = 23).

Survey respondents were asked what modalities they used to treat paratonia. Respondents could enumerate all the treatment modalities they employed. The frequency of each modality is reported in Figure 1c. The most frequent modality used to treat paratonia was BoNT-A (92%), followed by positioning (80%), and passive range of motion therapy (72%). Figure 1d summarizes respondents’ years of experience treating paratonia with BoNT-A.
Respondents were asked to list all the indications for which they injected BoNT-A in LTC and in what proportion they injected for each indication, with all responses equaling a total of 100% per respondent. Spasticity (100%) and paratonia (91%) were the most frequently endorsed indications for injection in LTC. The frequency and proportion of BoNT-A injections in LTC across each indication is provided in Table 1S supplementary material 3.
While a consensus definition of paratonia created through a rigorous Delphi process has been published, Reference Hobbelen, Koopmans, Verhey, Van Peppen and de Bie12 we were interested in documenting whether respondents were aware of this definition, and if not, how paratonia was being defined in contrast to spasticity, another form of hypertonia. The question posed was “How is paratonia different from spasticity?”. In Table S2 supplementary material 3 we outline the paratonia definition elements (themes) enumerated in the free text responses, and the frequency of which these themes were reported. The most common themes defining paratonia and distinguishing it from spasticity were resistance to passive movement, tone increases with increasing movement, and absence of velocity dependence.
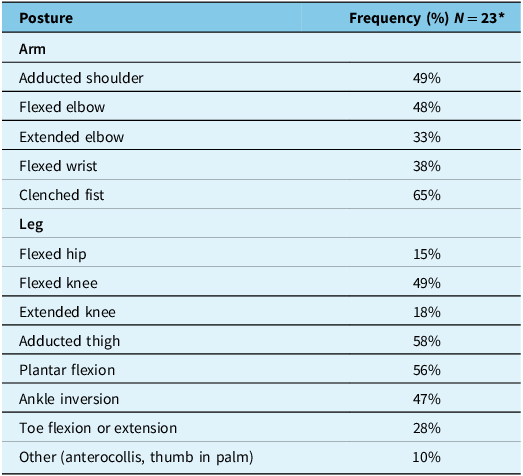
Respondents were asked what factors contributed to their decision to inject BoNT-A for paratonia. The most frequently endorsed reason was pain when the limb was being manipulated (100%). All responses are provided in Table 3S supplementary material 3. We also sought to determine what postures were most commonly injected for paratonia. Respondents were asked to enumerate all the postures they injected and the frequency at which they injected for each posture. All postures injected with BoNT-A for paratonia are listed in Table 1. Clenched fist (65%) and adducted thigh (58%) were the most common postures treated with BoNT-A.
Frequency of postures injected with botulinumtoxin-A (BoNT-A) for paratonia

* 2 respondents don’t treat paratonia and 2 respondents don’t treat paratonia with BoNT-A.
Respondents were asked how many paratonia patients they treated with BoNT-A on average, each month, across the settings they provided BoNT-A injections. All results are displayed in Table S4 supplementary material 3. In LTC, 53% of respondents reported injecting 1–5 paratonia patients/month, with 20% treating between 6–10 and 21–30 patients/month. Sixty seven percent of respondents reported treating 1–5 paratonia patients/month in the Clinic or Office setting. Two injectors who treated patients with BoNT-A in a Rehabilitation Facility reported injecting 1–5 paratonia patients/month. All respondents reported using onabotulinumtoxinA. Six respondents reported using all 3 brands (onabotulinumtoxinA, incobotulinumtoxinA and abobotulinumtoxinA) which are available in Canada. The frequency and proportion of each brand used and average doses of BoNT-A injected for treatment of paratonia are listed in Table S5 supplementary material 3.
Sixty percent of respondents injecting BoNT-A for off-label paratonia reported having access to funding, with 40% unable to access funding. Strategies used to obtain access to BoNT-A included using a limited use code (Ontario) for stroke or transient ischemic attack (68%), applying to an exceptional access program from the provincial Ministry of Health (31%), obtaining the drug from a compassionate use program from a pharmaceutical company (18%), applying private insurance (15%) and patients paying privately (8%).
We asked respondents what outcome measures they used to determine effectiveness of BoNT-A injections for paratonia. All outcome measures used to determine effectiveness of injections for paratonia are outlined in Table 6S supplementary material 3. The most common measure was subjective impression of the caregiver (87%) followed by subjective impression of the clinician (78%). Thirteen percent of those injecting BoNT-A for paratonia reported a significant clinically relevant improvement; 74% endorsed a moderate clinically relevant improvement; 13% endorsed a slight clinically relevant improvement. No one reported absence of effect of BoNT-A injections or worsening of clinical status following injections.
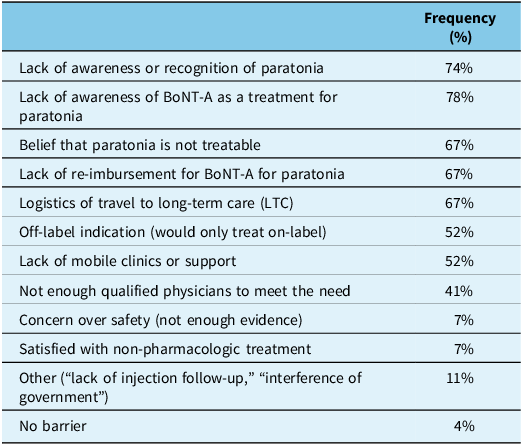
Sixty-five percent of respondents who injected BoNT-A for paratonia reported no side-effects with treatment. Twenty-seven percent reported bruising at the site of injections and 26% endorsed pain during injections. There were no reported serious side-effects from injections in this elderly population. Ninety percent of respondents indicated they perceived barriers existed for treatment of paratonia with BoNT-A. The reasons provided for the perceived barriers are displayed in Table 2.
Frequency of perceived barriers to treating paratonia with botulinumtoxin-A (BoNT-A)

This survey was conducted to identify real-world practices of injectors of BoNT-A in Canada for the indication of paratonia in dementia. Despite variability in knowledge, understanding and access to BoNT-A for paratonia, the survey demonstrates that paratonia is being treated widely across Canada, though the majority of respondents confirmed there to be a significant barrier to treatment with BoNT-A. While the survey was distributed to a convenience sample and did not include all Canadian BoNT-A injectors of hypertonia, the results of the survey suggest that BoNT-A may be a safe and effective treatment to address this debilitating complication of dementia. Given the number of Canadians affected by dementia and paratonia, and the human and economic costs associated with paratonia, a comprehensive road map for research is needed to address current gaps in knowledge and practice.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cjn.2024.46.
Acknowledgements
The authors would like to acknowledge Joanna Grace van den Broek B.Sc., PT, Dr Alex Benham PhD and Mr. Adrian Robertson MPhil, Consultant PT, for additional collaboration and support.
Author contributions
There is no objection to the above details being published. No person who had contributed substantially to the production of this manuscript has been excluded from authorship. The authors affirm that conflicts of interests have been declared. No funding was received from any source to support this project. We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this manuscript is consistent with those guidelines.
1. Research Project.
A. Conception.
B. Organization.
C. Execution.
2. Survey Preparation.
A. Design.
B. Execution.
C. Review and Critique.
3. Results.
A. Data analysis.
B. Data interpretation.
C. Table and figure generation.
4. Manuscript Preparation.
A. Writing of the first draft.
B. Review and Critique.
GK: 1A, 1B, 1C, 2A, 2B, 4A.
FI: 1B, 1C, 2C, 4B.
OK: 1B, 1C, 2C, 4B.
CP: 3A, 3B, 3C, 4B.
LT: 3A, 3B, 3C, 4B.
CB: 1B, 1C, 2C, 2B, 4B.
Funding statement
None.
Competing interests
The authors whose names are listed immediately below report the following details of affiliation or involvement in an organization or entity with a financial or non-financial interest in the subject matter or materials discussed in this manuscript.
Galit Kleiner: In partnership with Baycrest Health Sciences GK owns a use patent for the indication of botulinum toxin for paratonia. GK has received payment of patent maintenance fees (February 2021, April 2022) while patent was considered for licensing (declined). No current relationship. GK has received consulting fees and Honoraria from Abbvie and Ipsen. GK has received sample botulinum toxin A (BoNT-A) from Abbvie, Merz and Ipsen.
Farooq Ismail: FI has received consulting fees and Honoraria from Abbvie, Merz and Ipsen. FI has received sample BoNT-A from Abbvie, Merz and Ipsen.
Omar Khan: OK has received consulting fees and Honoraria from Abbvie, Merz, Ipsen and Pendopharm. OK has received educational grants from Abbvie and Ipsen to attend conferences. OK has participated in advisory board meetings with AbbVie, Merz, Ipsen and Pendopharm. OK has received sample BoNT-A from Abbvie, Merz and Ipsen.
Christopher Pilieci: No conflicts to declare.
Linda Truong: No conflicts to declare.
Chris Boulias: CB has received consulting fees and Honoraria from Abbvie, Merz and Ipsen. CB has received sample BoNT-A from Abbvie, Merz and Ipsen.