Introduction
Land-use changes and direct exploitation are the primary drivers of population declines and species extinction risk (Nic Lughadha et al., Reference Nic Lughadha, Bachman, Leão, Forest, Halley and Moat2020). The Global Strategy for Plant Conservation comprises 21 voluntary, complementary actions related to plant conservation, supporting the Kunming–Montreal Global Biodiversity Framework’s 23 targets for halting biodiversity loss (CBD, 2022, 2024). These actions can inform decision-making in biodiversity conservation in areas such as Brazil’s Atlantic Forest, a centre of plant endemism and richness that was reduced to 8.5% of its original tree cover in the 1980s but has since recovered to c. 27% through forest regeneration and reforestation (Myers et al., Reference Myers, Mittermeier, Mittermeier, Fonseca and Kent2000; Rezende et al., Reference Rezende, Scarano, Assad, Joly, Metzger and Strassburg2018; SOS Mata Atlântica & INPE, 2023). Approximately 82% of tree species in the Atlantic Forest are threatened with extinction (Lima et al., Reference Lima, Dauby, Gasper, Fernandez, Vibrans and Oliveira2024), one of which is Paubrasilia echinata (Lam.) Gagnon, H.C.Lima & G.P.Lewis (= Caesalpinia echinata Lam.), family Leguminosae, commonly referred to as brazilwood. It is Brazil’s national tree, is endemic to the coastal region of the Atlantic Forest (Gagnon et al., Reference Gagnon, Lewis and Lima2024), and is categorized as Endangered on national Red Lists (Martinelli & Moraes, Reference Martinelli and Moraes2013; MMA, 2022) and the IUCN Red List (Varty, Reference Varty1998). It has been exploited since the 16th century as a raw material for red dye (brazilin) in the textile industry and gained new importance in the 19th century as the preferred material for violin bows (Lima et al., Reference Lima, Lewis, Bueno and Bueno2002). However, because of the impacts of logging, trade of the species has been regulated by CITES since 2007 (CITES, 2022).
Phylogenomic analysis of P. echinata has identified five groups based on genotype: arruda-BA, arruda-RJ, café, laranja and north (Rees et al., Reference Rees, Neaves, Lewis, Lima and Gagnon2023). However, these groups or lineages are not easily distinguished based on morphology (Macedo et al., Reference Macedo, Lima, Souza, Gonçalves, Costa and Barros2019). The species’ southernmost populations (sensu Ricklefs, Reference Ricklefs2007, see Methods) occur in the state of Rio de Janeiro, comprising the entirety of the arruda-RJ lineage. The species exhibits high genetic differentiation and low gene flow, demonstrating geographical isolation along the Brazilian coast (Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003; Cardoso et al., Reference Cardoso, Provan, Lira, Pereira, Ferreira and Cardoso2005). Our fieldwork during 2004–2024 has provided new insights into the geographical distribution of arruda-RJ, complementing knowledge of remnant populations in 13 forest fragments (Pereira et al., Reference Pereira, Cardoso, Lima and Kruel2007). Since the late 20th century the rarity of arruda-RJ has attracted conservation efforts, including within protected areas, conservation planning, urban tree projects and forest restoration (Fundação Margaret Mee, 1997; Rio de Janeiro, Reference de Janeiro2016). However, the resulting data are scattered and outdated, and an update is required to assess the surviving populations.
To assess the current conservation status of arruda-RJ, we investigated its current geographical distribution and habitat, assigned a conservation priority (high, medium or low) to each forest fragment and, given this prioritization, identified the most effective management actions. We conducted field surveys and inventoried, measured and analysed the conservation status of forest fragments where arruda-RJ persists, to assess the effectiveness of protection. Additionally, we developed and applied a qualitative prioritization protocol to classify each fragment based on its importance for conservation. We recommend a range of conservation actions as part of a broader conservation plan for arruda-RJ. This approach was designed as a model potentially applicable to other lineages of P. echinata, and as a contribution to the Global Biodiversity Framework and the Global Strategy for Plant Conservation (CBD, 2022, 2024).
Study area
The study was conducted in the coastal zone of the state of Rio de Janeiro, between the municipalities of Rio de Janeiro and Armação dos Búzios (Fig. 1). The areas surveyed were three Atlantic Forest formations: Lowland Semi-deciduous Forest, Restinga Forest and Dense Ombrophilous Submontane Forest (Rio de Janeiro, Reference de Janeiro2011; Machado et al., Reference Machado, Nascimento, Barros, Sartori, Bohrer and Pennington2021). The climate is tropical and hot semi-arid (Barbiére & Kronemberger, Reference Barbiére and Kronemberger1994).

Fig. 1 Distribution of the Paubrasilia echinata arruda-RJ lineage in the state of Rio de Janeiro, Brazil, showing forest fragments containing the lineage in the (a) south, (b) central and (c) north regions, and indicating individual records and those forest fragments were we newly recorded the lineage. Forest fragments were categorized as high, medium or low priority using our prioritization classification protocol (see text for details).
Methods
Data collection
We collated all occurrence records of P. echinata and its synonyms (sensu Gagnon et al., Reference Gagnon, Lewis and Lima2024) by querying the Global Biodiversity Information Facility (GBIF, 2024), JABOT (JBRJ, 2024), REFLORA (2024) and SpeciesLink (2024) platforms with no limits on the search period.
We conducted fieldwork during 2004–2024 across 35 municipalities in the states of Rio de Janeiro and São Paulo (Supplementary Table 1), guided by our verification of occurrence records in herbarium collections and bibliographic sources, and by the presence of suitable fragments of relevant native flora communities and phytophysiognomies. We also considered reports of natural populations from local informants, including residents, park rangers, reserve managers and biologists involved in environmental impact assessments. We performed random walk surveys for brazilwood in forest fragments (Filgueiras et al., Reference Filgueiras, Nogueira, Brochado and Guala1994), complemented by high-resolution imagery obtained with an unmanned aerial vehicle (Mavic Mini 2 drone, DJI, China) in areas with limited accessibility (Nyberg, Reference Nyberg2019). Voucher specimens of P. echinata were lodged in the herbarium RB (Rio de Janeiro Botanical Gardens, Brazil). We were able to confirm the presence of brazilwood populations in 11 municipalities. We adopted Ricklefs’ (Reference Ricklefs2007) definition of a population as ‘the individuals of one species in an area. Each population lives mainly in patches of adequate habitat’. Thus, forest fragments supporting a discrete population of P. echinata were our units for collecting data on demography, threats, and conservation actions and opportunities.
Table 1 The scoring system used to produce a prioritization classification protocol for forest fragments containing Paubrasilia echinata arruda-RJ lineage populations in the state of Rio de Janeiro, Brazil (Fig. 1). Scores were determined for five factors. Scores ranged from 0 to 3 (0, absent or unknown; 1, minor impact; 2, medium impact; 3, major impact). Threats were based on the IUCN Threats Classification Scheme (IUCN, 2012a): 1, residential-commercial development; 2, agriculture and livestock; 3, energy production/mining; 4, transportation; 5, biological resource use; 6, human disturbance; 7, natural system modifications; 8, invasive species-diseases; 9, pollution. Opportunities are activities compatible with biodiversity conservation, sustainable practices and/or those with the potential to benefit biodiversity conservation (MMA, 2023).

1 Quilombola communities are self-identified ethnic groups of predominantly rural or urban Black populations, defined by ties to land, territory, ancestry and cultural traditions.
We adapted the classification of Rodrigues et al. (Reference Rodrigues, Abreu, Barcellos, Lima and Scarano2009) in our demographic survey of the age distribution of brazilwood. We classified seedlings as ≤ 50 cm in height with a basal diameter < 5–10 mm, foliage ranging from first leaves to >10 pairs of mature leaves, a stem with small thorns and occurring in the understorey; juveniles as 51–200 cm in height with a basal diameter 10.1–30.0 mm, a stem with abundant thorns and occurring in the understorey; and adults as ≥ 201 cm in height with a basal diameter ≥ 30.1 mm, with sparsely thorned stems and occurring in the understorey or near the canopy, or without thorns but with peeling bark and pustular lenticels, and occurring in the canopy (Plate 1).

Plate 1 Life stages and characteristics of Paubrasilia echinata arruda-RJ lineage trees recorded in the state of Rio de Janeiro, Brazil: (a) mature tree (forest fragment 1), (b) flowers (forest fragment 38), (c) seedling (forest fragment 3), (d) juvenile (forest fragment 4), (e) young adult, stem with thorns (forest fragment 27), and (f) mature adult, stem without thorns, peeling bark with pustular lenticels (forest fragment 27).
During fieldwork we documented any observed threats to populations of brazilwood and Atlantic Forest fragments and inferred threats to other populations from the literature for each forest fragment. We categorized threats using the IUCN Threat Classification (IUCN, 2012a). Conservation actions followed the IUCN Conservation Actions Classification (2012b), which we identified through field observations, a literature review and by identifying protected areas (INEA, 2023). We defined opportunities as either activities or supportive local communities with the potential to contribute to biodiversity conservation and/or sustainability (adapted from MMA, 2023). We also overlaid a map of the forest fragments with Brazil’s priority areas for conservation and Rio de Janeiro’s ecological corridors to identify fragments located within designated conservation areas (SEAS, 2017; MMA, 2023).
Data analysis
We defined forest fragments by excluding cultivated trees, records of doubtful validity and duplicates from our total of 361 records, resulting in 197 exclusions. The final database comprised 164 records of P. echinata in our study area. We mapped these records using Google Earth (Google, 2024) onto native forest fragments within the Atlantic Forest, delineated by roads, clearings, rivers, beaches, urban areas or agricultural zones. We verified, corrected or assigned the geographical coordinates of occurrence records through locality interpretation (Martinelli et al., Reference Martinelli, Martins, Moraes, Loyola and Amaro2018) using Google Earth and QGIS 3.30.3 (QGIS Development Team, Reference Development Team2023). We determined whether arruda-RJ records were native trees based on their occurrence in remnant fragments in areas of secondary or primary forest, the presence of other species typical of the Atlantic Forest, local knowledge or label information on herbarium records. Records of cultivated trees were those representing anthropogenic introductions such as planting in botanical gardens, household gardens and urban green spaces. We categorized forest fragments by size as small, 0.003–5 km2, medium, 5.01–10 km² or large, 10.01–15 km².
We adopted a prioritization classification protocol based on the relative priority of each forest fragment for conservation. We scored five factors (demographic composition, fragment size, presence in protected areas, threats and opportunities) for each fragment, on a scale from 0 to 3, where 0 is absent or unknown, 1 is minor impact, 2 is medium impact, and 3 is major impact. The qualitative and quantitative descriptors corresponding to each score for each factor are given in Table 1. We aggregated the scores of the five qualitative factors across all forest fragments. The results were classified using Sturges’ formula in the first instance (Milone & Angelini, Reference Milone and Angelini1995), resulting in six classes. Following inspection of these classes, the lower, middle and upper pairs were merged to form three diagnosable classes: low priority (3–5 points), medium priority (6–11 points) and high priority (12–14 points). This prioritization classification protocol provides a transparent, simple, cost-effective and species-specific approach, offering a method to rank forest fragments by their conservation or restoration value for arruda-RJ, even where there are significant threats. It weighs all factors equally, reflects current population status and considers each fragment’s individual potential. The protocol integrates principles of conservation biology, field data and literature on the species’ ecology, reproduction, genetic diversity and responses to habitat fragmentation (Primack & Rodrigues, Reference Primack and Rodrigues2001; Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003; Cardoso et al., Reference Cardoso, Provan, Lira, Pereira, Ferreira and Cardoso2005; Pereira et al., Reference Pereira, Cardoso, Lima and Kruel2007; Zaidan et al., Reference Zaidan, Chui, Aidar, Buckerigde, Ribeiro, Barbedo, Alves, Domingos and Braga2008; Rodrigues et al., Reference Rodrigues, Abreu, Barcellos, Lima and Scarano2009; CBD, 2022).
Results
Geographical distribution
Our results confirmed the presence of native populations of P. echinata within the state of Rio de Janeiro, corresponding to the arruda-RJ lineage previously inferred through studies of population genetic structure and phylogenomic analysis (Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003; Rees et al., Reference Rees, Neaves, Lewis, Lima and Gagnon2023; Fig. 1, Supplementary Table 2). We identified 43 populations in forest fragments distributed across 11 municipalities, in primary or secondary vegetation. These populations include 30 occurrences of arruda-RJ in forest fragments not previously reported in the peer-reviewed scientific literature, all in the coastal region of the state. Once mapped in Google Earth, the forest fragments were clearly divisible into three areas: south, central and north (Fig. 1; Supplementary Table 2). The three areas are compatible with the documented population genetic structure (Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003). The total area of forest fragments containing arruda-RJ was 106,439 km², corresponding to just 0.24% of the land area of the state. We documented 35 small, six medium-sized and two large forest fragments across the south, central and north regions. Mean fragment size was 2.47 km². We observed forest fragments in Dense Ombrophilous Submontane Forest areas close to Rocky Outcrops and Inselbergs in the south and central regions, and fragments in Restinga Forest and Lowland Semi-Deciduous Forest in the north region.
Threats to populations of arruda-RJ
Forty of the 43 forest fragments (93%) faced threats of varying severity. Residential and commercial development was the most frequently observed threat, in 32 forest fragments (74%), reflecting the impacts of urbanization in the three regions, followed by energy production and mining in rural areas in the central and north of the state, as well as roads and transmission lines, also in the north (13 forest fragments, 30%). Conversion of native vegetation to agriculture and livestock farming affected 10 forest fragments (Supplementary Table 2). Other threats included anthropogenic disturbances such as recreational activities in forest fragments in the south and central regions (12 forest fragments), invasive species (eight forest fragments), hunting, and harvesting of tree species other than brazilwood (seven forest fragments) and pollution (six forest fragments) and forest fires (four forest fragments). We did not document any threats in three forest fragments: one was within a fully protected area where we observed no threats during our visit, and we did not visit two forest fragments.
Conservation actions
We identified 62 conservation actions and opportunities for the protection of arruda-RJ in 29 of the 43 forest fragments, mostly in existing protected areas (Supplementary Table 2). A total of 29 forest fragments are in 24 Full Protection areas, where almost all anthropogenic disturbances are prohibited although not entirely prevented (Supplementary Table 2). Four forest fragments are in protected areas where sustainable land use is permitted, and one forest fragment is in a private reserve (Supplementary Tables 2 & 3). We did not identify any conservation actions for 14 forest fragments in the central and north regions. We identified opportunities for initiatives to protect P. echinata in 22 forest fragments. Four forest fragments are within areas classified as extremely high priority by the Brazilian Ministry of the Environment (MMA, 2023; Supplementary Table 2). Seven fragments are in areas designated as ecological corridors by the state government, one in the central region and six in the north (SEAS, 2017). We identified ongoing environmental education activities, including hiking, school field trips and urban afforestation, in 17 fragments. Nine fragments are in sites where there are ongoing restoration activities, such as marking mother trees, seed collection, seed banking, seedling production or reforestation through the planting of brazilwood and other species (Supplementary Table 2). In addition, the ex situ mother tree at the Rio de Janeiro Botanical Garden arboretum serves as a resource for research, education and seed banking.
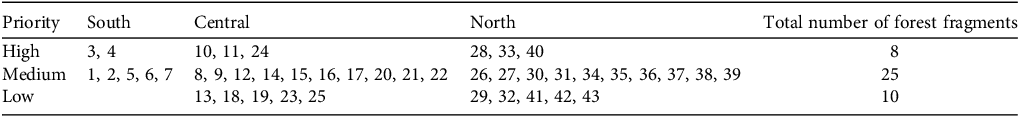
Table 2 Prioritization of 43 forest fragments containing the P. echinata arruda-RJ lineage in the three regions in the state of Rio de Janeiro (south, central, north; Fig. 1) using the prioritization classification protocol (see text for details) to assign priority levels as high, medium or low (Supplementary Table 2). Numbers identify individual forest fragments.

Demographic characteristics of populations
We conducted demographic surveys in 29 (68%) of the 43 forest fragments and identified 1,358 individuals, including all life stages (Plate 1). Of the censused fragments, 18 (62%) contained all age classes (seedlings, juveniles, adults), nine (31%) contained only adults, and two (7%) contained juveniles and adults but no seedlings. The 18 fragments with all age classes spanned the three regions and are all in protected areas, although they still face threats including urban expansion, fire and, to a lesser extent, mining. Sixteen forest fragments are in areas subject to management actions, including the deployment of park rangers, monitoring, forest restoration, environmental education and ecotourism. The nine forest fragments containing only adult individuals are all small and are distributed across all three regions. They are threatened by urban expansion and uncontrolled tourism; seven are in protected areas and two are unprotected. The two forest fragments containing only juvenile and adult individuals are threatened by urbanization and livestock grazing.
Conservation prioritization of forest fragments
In applying our prioritization classification protocol, eight forest fragments were classified as high priority, 25 as medium priority and 10 as low priority (Table 2, Fig. 1, Supplementary Table 3). The high-priority forest fragments spanned all three regions, and included all three age classes. They are located in Full Protection areas, are mostly medium-sized or large, have benefitted from ecological restoration projects, occur in areas with active protected area management, ecotourism and environmental education but are threatened by urbanization and fire (Fig. 1, Table 1, Supplementary Table 3). The 25 medium-priority forest fragments also spanned all three regions but they vary in conservation status. Most are small forest fragments (92%) and occur in Full Protection areas (64%) but only 10 forest fragments contain all three age classes. All are exposed to multiple threats and few present opportunities for restoration or environmental education. The 10 forest fragments classified as low priority are small, occur in the central and north regions, are on private land, outside protected areas, exposed to urbanization or agricultural threats, and lack any conservation actions.
Discussion
Through targeted field surveys across a fragmented tropical landscape, we identified 30 previously unknown sites harbouring the arruda-RJ lineage of brazilwood. Preserving these forest fragments will most probably require in situ conservation, land-use management, ecological restoration and enrichment of degraded forest fragments to maintain extant genetic diversity, and ex situ actions to prevent extinction of native populations (CBD, 2022, 2024). Our study shows how floristic inventories can provide data useful for prioritizing forest fragments harbouring brazilwood. Our approach may also be applicable to conservation planning for other tree species. Moreover, we find grounds for optimism concerning the possibility that undocumented populations of other rare trees may persist in inaccessible areas.
Historical and current distribution
Rio de Janeiro state harbours 32% of the remaining Atlantic Forest (MapBiomas, 2024). In south and central Rio de Janeiro, the P. echinata arruda-RJ lineage currently persists in forest fragments in steep areas that are of little value for real estate. In contrast, intensive urbanization began relatively recently, within the past 50 years, in the north region, which may explain the persistence of brazilwood populations in this area (Scarano et al., Reference Scarano, Costa, Freitas, Lima, Martinelli, Nascimento, Bergallo, Fidalgo, Rocha, Uzêda, Costa and Alves2009).
The arruda-RJ lineage of brazilwood is known to occur in Seasonally Dry Tropical Forests in central and northern Rio de Janeiro (Pennington et al., Reference Pennington, Lavin and Oliveira-Filho2009; Machado et al., Reference Machado, Nascimento, Barros, Sartori, Bohrer and Pennington2021). Our observations and those of Peixoto et al. (Reference Peixoto, Martins, da Silva and Silva2004) suggest that the coastal massifs in the Dense Ombrophilous Submontane Forest in southern Rio de Janeiro may also form areas of Seasonally Dry Tropical Forest because of edaphic conditions. Low tolerance to cold probably prevents P. echinata from expanding further south (Zaidan et al., Reference Zaidan, Chui, Aidar, Buckerigde, Ribeiro, Barbedo, Alves, Domingos and Braga2008; Guerra et al., Reference Guerra, Bezerra, Jorge and Fullen2013). In contrast, the northern limit of arruda-RJ may be influenced by humidity from the mouth of the Paraíba do Sul River and the Restinga forest formed during the Holocene (Machado et al., Reference Machado, Nascimento, Barros, Sartori, Bohrer and Pennington2021).
Recent fieldwork has led to the rediscovery of several species previously undocumented in Rio de Janeiro state (Rosa et al., Reference Rosa, Baez, Martins and Martinelli2018; Lírio et al., Reference Lírio, Barbosa, Freitas, Rosa, Negrão and Pietro2023). Our discovery of 30 previously undocumented forest fragments harbouring brazilwood is encouraging. Although observer bias cannot be ruled out, our surveys were conducted by trained teams capable of identifying individuals across life stages (Plate 1). Nevertheless, access to 14 forest fragments was not possible because of landowner restrictions or safety concerns, particularly in areas under control of drug trafficking groups. It is therefore possible that additional, unrecorded populations may persist in forest fragments with similar vegetation types. Despite these limitations, we believe our findings have utility for conservation planning for this species, which faces continued habitat fragmentation and genetic isolation (Cardoso et al., Reference Cardoso, Provan, Lira, Pereira, Ferreira and Cardoso2005). With adequate management, these areas may support the recovery of the P. echinata arruda-RJ lineage. The species is currently categorized as Endangered on the IUCN Red List (Varty, Reference Varty1998), although a recent reassessment suggests its status may be Critically Endangered based on population size reduction and generation length (Lima et al., Reference Lima, Dauby, Gasper, Fernandez, Vibrans and Oliveira2024). We found that brazilwood was often absent in suitable habitats, perhaps as a result of historical logging.
Alignment of brazilwood conservation with the Global Biodiversity Framework
The Kunming–Montreal Global Biodiversity Framework proposed 23 targets towards halting biodiversity loss. Target 4, ‘Halt Species Extinction, Protect Genetic Diversity, and Manage Human–Wildlife Conflicts’, has an associated indicator, A4, based on the ‘Proportion of populations within species with an effective population size > 500’. As effective population size is typically c. 10% of census size (Hoban et al., Reference Hoban, Silva, Hughes, Hunter, Stroil and Laikre2024), and no forest fragments we censused contained 500 adults (Supplementary Table 1), our results suggest that P. echinata arruda-RJ falls short of the threshold for this indicator. We found 1,358 individuals of arruda-RJ, of which 38% were adults (Supplementary Table 2). A previous study of six populations found 2,745 individuals, of which 10% were adults (Pereira et al., Reference Pereira, Cardoso, Lima and Kruel2007). The low number may reflect past logging of mature trees (Lima et al., Reference Lima, Lewis, Bueno and Bueno2002; Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003; Rodrigues et al., Reference Rodrigues, Abreu, Barcellos, Lima and Scarano2009). Management practices that could contribute to increasing the genetic diversity of arruda-RJ include forest restoration in medium-priority and low-priority forest fragments using seeds from mother trees and seedling transplants from high-priority forest fragments.
In addition, Target 3 of the Global Biodiversity Framework calls for at least 30% of terrestrial areas ‘especially areas of particular importance for biodiversity’ to be ‘effectively conserved and managed’ through ‘well-connected and equitably governed systems of protected areas and other effective area-based conservation measures’ (CBD, 2022). If the remaining forest fragments containing arruda-RJ are considered areas of particular importance for biodiversity, they represent an important contribution towards meeting Target 3 in terms of area. Of the 43 forest fragments we documented, 28 comprising 92% (9,867 km²) of the total area are in Full Protection or Sustainable Use protected areas. However, not all aspects of Target 3 are met, as the protected areas where arruda-RJ occurs are not well-connected and some Sustainable Use protected areas lack management plans.
Target 2 of the Global Biodiversity Framework aims for 30% of areas of degraded ecosystems to be under effective restoration by 2030. Conserving forest fragments where arruda-RJ persists could help meet this target. Forest cover in Rio de Janeiro increased from c. 30% to 32% of the state area over the last 4 decades, mainly through restoration and natural regeneration (MapBiomas, 2024), but is primarily carried out in Montane and Ombrophilous areas and mangroves (Brasil, 2006; Rezende et al., Reference Rezende, Scarano, Assad, Joly, Metzger and Strassburg2018), and few forest fragments with arruda-RJ have been restored. Coastal forests that harbour P. echinata arruda-RJ could be added to the State Ecological Restoration Plan (Rio de Janeiro, Reference de Janeiro2019), focusing on enrichment with seedlings, forest restoration and creating ecological corridors between forest fragments here classified as medium or low priority. The establishment of such corridors could increase population size and genetic diversity (Lira et al., Reference Lira, Cardoso, Ferreira, Cardoso and Provan2003; Pereira et al., Reference Pereira, Cardoso, Lima and Kruel2007).
The Global Strategy for Plant Conservation (actions 4 c,d) recommends that threatened plant species are restored using ex situ methodologies that achieve adequate levels of genetic diversity (CBD, 2024). Seed banks are an efficient and economically viable action for protecting populations of P. echinata. High- and medium-priority forest fragments from each region include potential mother trees that could contribute seeds. An innovative proposal is to establish seed orchards aimed at conserving genetic diversity, combined with improvement for commercial production (Sebben et al., Reference Sebben, Freitas, Zanatto, Moraes and Moraes2007). For arruda-RJ this would offer significant benefits for restoration and ex situ conservation but also presents challenges related to cost, genetic diversity and environmental vulnerability. However, seed orchards could usefully be established at universities, within buffer zones of protected areas and along ecological corridors connecting medium- and low-priority forest fragments (SEAS, 2017). Furthermore, the cultivation of trees in urban greening projects and botanical gardens could enhance environmental education, which is another tenet of the Global Strategy (Rio de Janeiro, Reference de Janeiro2016; CBD, 2024).
Conclusion
The previously undocumented sites for Brazil’s national tree reported here are cause for cautious optimism for the survival of the P. echinata arruda-RJ lineage. Our methodology offers a model applicable to other lineages of P. echinate (and brazilwood is a candidate indicator of the ecological integrity of Seasonally Dry Tropical Forests; Carignan & Villard, Reference Carignan and Villard2002). We hope our findings will inform a National Conservation Plan for brazilwood, as well as contributing to Global Biodiversity Framework Targets and the Global Strategy for Plant Conservation (CBD, 2022, 2024). In addition, our findings may be useful for conservation status assessments of lineages of species for inclusion in the IUCN Red List of Threatened Species and national Red Lists, subject to scientific consensus on such approaches (Blair et al., Reference Blair, Sterling and Hurley2011; Bertola et al., Reference Bertola, Quinn, Hanghøj, Garcia-Erill, Rasmussen and Balboa2024).
The principal data requirements for our prioritization classification protocol, which could also be applied to other threatened tree species in fragmented tropical forest landscapes, are the age classes in extant forest fragments and the level of threat and protection. Fieldwork is essential to locate remnant populations and to collect the necessary demographic information. However, much of the analysis of threats and protection levels can be conducted remotely using GIS and secondary data, which makes the approach adaptable to other tropical trees. Our study suggests that tropical hardwoods such as brazilwood can persist in small forest fragments in inaccessible areas such as steep hillsides that may not have been previously inventoried.
Supplementary material
The supplementary material for this article is available at doi.org/10.1017/S0030605325102020
Author contributions
Conception, data collection, literature review, data analysis, writing: PR, HCL, ENL, TF; data analysis and interpretation: JF; data collection, theoretical contributions, revision: NGA, CFL, CFCS, CBdAB, HGRD, GPL.
Acknowledgements
We thank Andressa Novaes, Beto, Cláudio Santana, Christopher Quinn, Davi Machado, Diego Monsores, Domingos Cardoso, Fabio, Jailton Costa, João Braga, Juan Gomes, Luana Bianquini, Luiz Pedreira, Marcelo Costa, Nicky van Luijk, Pablo Feliz, Pedro Mireles, Rodrigo Castro, Tânia Sampaio Pereira, Sebastião Silva Neto and Viviane Fonseca-Kruel for their assistance. This research was supported by grants from CAPES (88887.603047/2021-00) and Funbio Brasil–Bolsas para o Futuro–2021 (019/2022), Instituto Humanize and Eurofins Foundation. We dedicate this article to the memory of biologist Robson Daumas Ribeiro for his commitment to locating forest fragments with Paubrasilia echinata in the state of Rio de Janeiro.
Conflicts of interest
None.
Ethical standards
We obtained a licence for specimen collection from the Instituto Chico Mendes de Conservação de Biodiversidade (83134–2), Instituto Estadual do Ambiente–Inea (026/2022) and the Secretaria Municipal de Meio Ambiente da Prefeitura do Rio de Janeiro (MAB-PRO-2022/01555). This research abided by the Oryx guidelines on ethical standards.
Data availability
Data are available from the authors upon reasonable request.