Drug use has significant social and health consequences, including the development of addictive disorders in a percentage of vulnerable individuals. Substance use disorder (SUD) is a frequently relapsing mental disorder characterised by compulsive substance-seeking, loss of control and negative emotional states in the absence of the substance. Reference Sachdev, Blacker, Blazer, Ganguli, Jeste and Paulsen1 The prevention and treatment of SUDs, as well as the negative consequences associated with drug use, are currently one of the main challenges facing the World Health Organization. 2 SUDs are often associated with multiple psychiatric comorbidities that present with additional health and social complications. In this context, long-term substance use has been linked to ongoing cognitive decline and cumulative brain damage throughout life. Reference Lappin and Sara3
It is estimated that between 30 and 80% of individuals with SUDs, or who regularly use drugs, develop some degree of cognitive impairment. Reference Toledo-Fernández, Marín-Navarrete, Villalobos-Gallegos, Salvador-Cruz, Benjet and Roncero4,Reference Bruijnen, Dijkstra, Walvoort, Markus, VanDerNagel and Kessels5 This can affect one or more functions, such as executive and attentional functions, visuospatial functions or long-term and working memory. Reference Mahoney6 Although each drug has its own unique characteristics, the literature shows common cognitive impairments that could be mediated by shared neurophysiological mechanisms. Reference Fernández-Serrano, Pérez-García and Verdejo-García7 The magnitude of the deficits varies depending on factors such as the type and dose of the drug, duration of use, age of onset, the presence of poly-drug use or the assessment protocol. Reference Valls-Serrano, Verdejo-García and Caracuel8,Reference Vonmoos, Hulka, Preller, Jenni, Baumgartner and Stohler9 Furthermore, although prolonged abstinence can partially reverse these impairments, cases of irreversible impairment have also been described. Reference Parvaz, Rabin, Adams and Goldstein10 These cognitive deficits often affect brain networks related to reward, decision-making and impulse control, which play a key role in maintaining addiction, creating a cycle in which impaired executive control makes it even more difficult to stop using the drug and remain abstinent. Reference Volkow, Wang, Fowler and Tomasi11
Furthermore, these cognitive problems significantly affect adherence to treatment programmes and increase the risk of relapse, complicating social and professional reintegration. Reference D’Souza12 Individuals often require external help to perform everyday activities such as remembering appointments, taking medication or maintaining care of the home. Furthermore, conventional addiction therapies require cognitive functions that are often impaired in these people. Therefore, the degree of cognitive impairment is an important predictor of therapeutic success, because greater deficits are associated with a higher probability of abandonment or relapse. Reference Sømhovd, Hagen, Bergly and Arnevik13,Reference Verdejo-García, Betanzos-Espinosa, Lozano, Vergara-Moragues, González-Saiz and Fernández-Calderón14 Therefore, the early assessment and detection of cognitive impairment in SUD individuals would have important consequences for diagnosis and treatment. However, this scientific knowledge has not yet been transferred to clinical practice, because current addiction treatment programmes do not implement neuropsychology. Reference Verdejo-Garcia, Rezapour, Giddens, Khojasteh Zonoozi, Rafei and Berry15
There is also currently insufficient information on gender differences in SUD-induced cognitive deficits. Women and men seem to show different vulnerabilities to cognitive decline when they use alcohol, cocaine, methamphetamine or other substances; these differences appear in areas such as memory, attention and executive function. Reference Fama, Le Berre and Sullivan16,Reference Shrestha, Huedo-Medina and Copenhaver17 The underlying causes of these gender differences include variations in brain structure, hormone levels, substance metabolism and neuroplasticity. Reference Luquiens, Rolland, Pelletier, Alarcon, Donnadieu-Rigole and Benyamina18 Understanding these gender-specific patterns can improve the effectiveness of interventions and facilitate personalised treatment planning.
Nevertheless, despite the fact that cognitive assessment in SUD individuals should be mandatory, a complete neuropsychological assessment is a complex and time-consuming process that can be performed only by specialists. Reference Ruiz Sánchez de León, Pedrero Pérez, Rojo Mota, Llanero Luque and Puerta García19 However, it is possible to implement cognitive screening tests in addiction treatment centres because these are brief, easy to administer and can be performed by health professionals. In this sense, the Montreal Cognitive Assessment (MoCA), developed by Nasreddine in 1996 in Montreal (Quebec, Canada), has become a popular screening tool for general cognition in both clinical and research settings. Reference Nasreddine, Phillips, Bédirian, Charbonneau, Whitehead and Collin20 Various cognitive screening tests have been validated in SUD individuals, such as the Neuropsychological Assessment Battery-Screening Module, Addenbrooke’s Cognitive Examination-Revised and the Mini-Mental State Examination (MMSE). Reference Ridley, Batchelor, Draper, Demirkol, Lintzeris and Withall21,Reference Grohman and Fals-Stewart22 However, MoCA has emerged as a key tool in evaluation of cognitive impairment among SUD individuals because it assesses many cognitive domains (executive function, attention, language, memory and orientation) in a short time and is especially sensitive for the detection of mild cognitive impairment. Reference Zhuang, Yang and Gao23 In fact, in the SUD population, MoCA outperforms other cognitive impairment screening tests such as MMSE. Specifically, Ridley et al reported that MoCA achieved a higher diagnostic accuracy, with an area under the receiver operating characteristic (ROC) curve (AUC) of 0.84, compared with 0.78 for MMSE, reflecting greater discriminative capacity. MMSE also exhibited lower sensitivity, leading to a higher rate of missed cases of cognitive impairment, which underlines the advantage of MoCA as a screening tool in this clinical context. Reference Ridley, Batchelor, Draper, Demirkol, Lintzeris and Withall21 The use of MoCA in SUD individuals could serve not only to detect cognitive deficits, but also to elucidate its potential applications for the creation of personalised therapeutic interventions, the prevention of therapeutic abandonment and monitoring of the recovery process.
Taking all the foregoing into account, the aim of the present study was to investigate the utility of MoCA to detect cognitive decline, examine the reversibility of cognitive functions with prolonged abstinence, study gender differences in cognitive function and predict the abandonment of addiction treatment. In this way, it was intended to demonstrate that the application of MoCA is useful for the treatment of addictions because it provides essential information to adapt and monitor the treatment of those with SUD.
Method
Recruitment and screening of participants
SUD participants were recruited at Fundación CESMA – Proyecto Hombre (Malaga, Spain), an organisation dedicated to the prevention and treatment of addictions through a biopsychosocial therapeutic approach. This treatment focuses on the individual to achieve maturity and growth through a therapeutic-educational method. Its main characteristics are: a comprehensive approach that addresses both personal and emotional and social problems; restoration of the individual’s autonomy and responsibility; involvement of the family and social environment; personalised treatment based on each individual’s needs; and a variable duration that can range from a single session to several months. The main objectives are the recovery of mental and physical health and reintegration into family and social relationships. The SUD group were required to meet the following inclusion criteria: 18–65 years of age; >2 weeks abstinent at the time of the neuropsychological evaluation (and a maximum of 1.5 months); alcohol, cocaine and/or cannabis use disorder; absence of serious medical or mental illness; and willingness to participate in the study after signing the consent form.
Control participants were included through the collaboration of staff working in collaborating centres (Proyecto Hombre-Malaga, Fundación Héroes, University of Malaga, IBIMA-Plataforma Bionand (Malaga, Spain)). The control group was comparable to the SUD group in age, gender and body mass index (BMI). The inclusion criteria for the control group were: 18–65 years of age; no history of drug use; or meeting the criteria for a SUD, absence of serious medical or mental illness and willingness to participate in the study after signing the consent form.
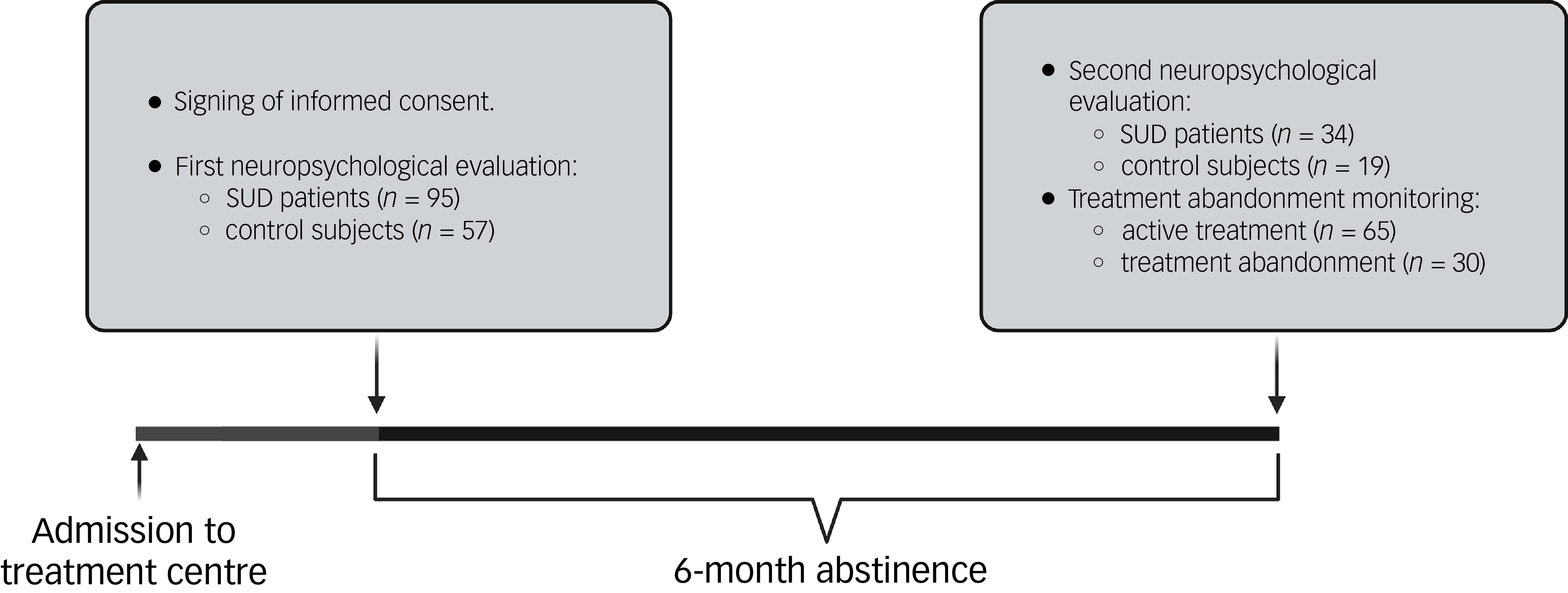
Figure 1 shows that 95 SUD patients and 57 control subjects were evaluated for the first time at the beginning of the study. All participants were recalled after 6 months for a second evaluation. Only 34 participants in the SUD group were evaluated for a second time, because 21 participants were not tested on account of the study having ended (12 months), 25 patients dropped out, 12 were excluded for having another treatment that interfered with cognition, 2 did not want to participate in the study again and 1 had completed treatment. In addition, we evaluated 19 control subjects after 6 months to control for the potential learning effect of the MoCA test. Finally, Proyecto Hombre-Malaga provided us with information on the therapeutic adherence of SUD participants after 6 months, finding that 46.15% of them had abandoned treatment.
Diagram of the study design. SUD, substance use disorder.

Ethical standards
Written informed consent was obtained from each participant following complete description of the study. All participants had the opportunity to discuss any questions or issues. This confirmed that any participant had consented to the inclusion of material pertaining to themselves, that they acknowledged that they could be identified via the manuscript and that they had been fully anonymised by the author. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 1975 as revised in 2013. All procedures involving human subjects/patients were approved by the Ethics and Clinical Research Committee of The Regional University Hospital of Malaga (no. SICEIA-2024-000878). All collected data were given code numbers to maintain privacy and confidentiality.
Psychiatric and neuropsychological evaluation
The Spanish version of the Psychiatric Research Interview for Substance and Mental Diseases (PRISM) diagnostic interview was used for the evaluation of SUDs (section 2: evaluation of substances), according to DSM-IV-TR criteria. This interview provided us with sociodemographic data, the diagnosis of SUD (yes/no) and addiction-related variables (severity (DSM-IV criteria), age of onset of consumption (age), age of onset of SUD (age), duration with SUD (years), number of relapses and duration of current abstinence (days)). PRISM is a semi-structured interview with good psychometric properties in the evaluation of SUDs. Reference Hasin, O’Brien, Auriacombe, Borges, Bucholz and Budney24
MoCA is a screening test that assesses general cognitive status, and has been shown to be especially sensitive to cognitive impairment induced by drugs of abuse, Reference Copersino, Fals-Stewart, Fitzmaurice, Schretlen, Sokoloff and Weiss25 including in a Spanish population with SUD. Reference Rojo-Mota, Pedrero-Perez, Ruiz-Sanchez de Leon, Llanero-Luque and Puerta-Garcia26 It assesses multiple cognitive domains through seven subtests: visuospatial/executive, identification, attention, language, abstraction, memory and orientation (Table 1). It can be administered in just 10 min, is freely accessible and can be used by any health professional after completing online training (https://mocacognition.com). For test correction, the original scoring standards were used, adding one more point to the total score if the individual had <12 years of education, and using the cut-off point of <26 to detect cognitive impairment.
Description of cognitive domains and subtests of Montreal Cognitive Assessment screening test

The evaluation was carried out in a quiet room by a psychologist specialised in neuropsychology. The evaluation was approximately 30 min in length, and participants could leave at any time if they so wished.
Statistical analysis
All data in the tables are expressed as either the number and percentage of subjects (N (%)) or mean and standard deviation (mean (s.d.)). Significance of differences in categorical variables was determined using Fisher’s exact test (chi-square test). Student’s t-test for independent samples and two related samples was used to assess the cognitive status of participants at baseline and after 6 months. Welch’s t-test was used when Levene’s test indicated inequality of variances. An analysis of covariance was performed to examine the effects of addiction-related variables (i.e. lifetime alcohol severity) on participants’ cognitive status, controlling for other SUD comorbidities (i.e. cocaine and cannabis). A multiple linear regression analysis was performed, checking the normality of the residuals by inspecting histograms and P–P plots; linearity and homoscedasticity using scatter plots of the standardised residuals; and the independence of errors using the Durbin–Watson statistic. A binary logistic regression analysis was performed using Pearson’s chi-square (χ 2) test with the Hosmer–Lemeshow test. The absence of multicollinearity was assessed using tolerance values and the variance inflation factor (VIF), with VIF <10 and tolerance >0.20 considered acceptable. The receiver operating characteristic (ROC) curve was used to evaluate the ability to discriminate therapeutic abandonment in SUD participants. Statistical analyses were performed using GraphPad Prism version 9 for Windows (GraphPad Software, San Diego, California, USA; https://www.graphpad.com), and IBM SPSS Statistics version 28 for Windows (IBM Corp., Armonk, New York, USA; at https://www.ibm.com/products/spss-statistics). Statistical analyses were carried out using GraphPad Prism version 9 and IBM SPSS Statistic version 28 (IBM, Armonk, New York, USA). A P-value <0.05 was considered statistically significant.
Results
Sociodemographic characteristics of the study sample
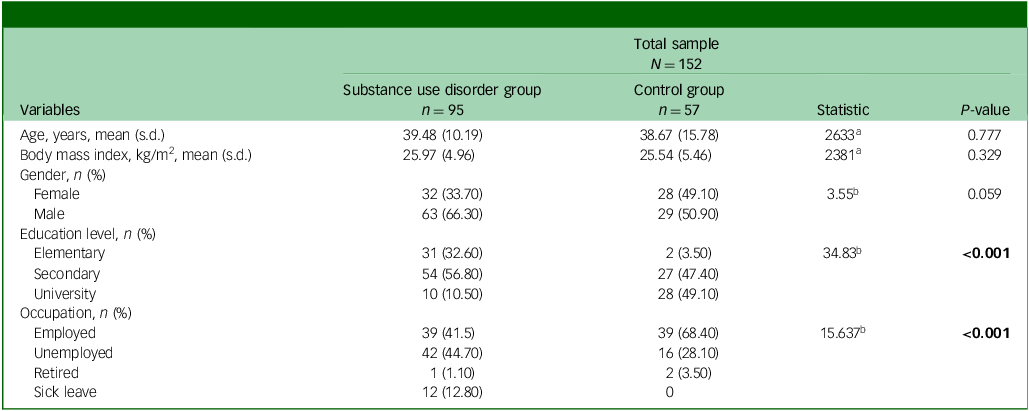
We recruited 95 abstinent patients from drug use out-patient programmes and 57 healthy control subjects. The sociodemographic description of the study participants is summarised in Table 2. We found that the SUD group were mostly 40-year-old male patients with a BMI of 26 kg/m2, a secondary education level (57%) and unemployed (45%). The control group were half men and half women aged 39, with a BMI of 26 kg/m2, a university education (50%) and employed (68%). Although the SUD and control groups were matched by gender, age and BMI, significant differences were observed in both educational level (P < 0.001) and occupation (P = 0.030).
Sociodemographic characteristics of the sample

a. Value calculated using the chi-squared test.
b. Value calculated using the Mann–Whitney U-test.
Bold values are statistically significant when P < 0.050.
Clinical characteristics of the SUD group
The clinical characteristics of the SUD group are shown in Table 3. The results indicated that 74% of participants had sought help for the first time for problematic substance use. Among participants attending out-patient treatment for substance abuse, alcohol use disorder was the most prevalent (68%), followed by cocaine (65%) and cannabis use disorders (48%). In addition, there was a high prevalence of other comorbid SUDs related to poly-drug use patterns (68%). There was a high medical comorbidity among these participants (66%), the most frequent being osteoarticular diseases (23%), metabolic diseases (20%), cardiovascular diseases (15%) and respiratory diseases (14%). Approximately 39% of participants had a diagnosis of mental disorder throughout their lives, the most common being depressive (24%) and anxiety (17%) disorders. Furthermore, 50% of participants had received psychiatric medication during the past year, most frequently being prescribed anxiolytics (32%) and antidepressants (30%).
Clinical characteristics of the substance use disorder (SUD) group

ADHD, Attention-deficit hyperactivity disorder.
Cognitive function at baseline and after 6 months
MoCA scores revealed that 72.60% of the SUD group showed deficits in global cognition at baseline. In the SUD group compared with the control group at baseline, we found significantly lower scores for total MoCA (t = −3.99, P < 0.001; Fig. 2(a)), visuospatial/executive (t = −2.37, P = 0.019; Fig. 2(b)), cube-drawing (t = −2.34, P = 0.021; Fig. 2(c)), subtracting (t = −2.38, P = 0.019; Fig. 2(d)), language (t = −2.08, P = 0.039; Fig. 2(e)), fluency (t = −3.06, P = 0.003; Fig. 2(f)) and memory (t = −2.81, P = 0.006; Fig. 2(h)).
Changes in global and domain-specific cognitive performance in substance use disorder patients compared with controls at baseline (Pre) and following 6 months of abstinence (Post). (a) Total Montreal Cognitive Assessment (MoCA) scores. (b) Visuospatial/executive scores. (c) Cube score. (d) Subtraction score. (e) Language score. (f) Fluency score. (g) Abstraction score. (h) Memory score. Mean values and standard deviation are represented in the bar graphs. *P < 0.001, **P < 0.010, ***P < 0.001.

Regarding intra-subject analysis, SUD patients had significantly improved scores in total MoCA (t = −3.16, P = 0.004; Fig. 2(a)), subtracting (t = −2.07, P = 0.046; Fig. 2(d)) and memory (t = −2.54, P = 0.016; Fig. 2(h)) after 6 months of abstinence compared with baseline. We did not observe any learning effect in the control group on overall and domain-specific MoCA scores following its repetition 6 months later. Finally, MoCA scores revealed that 50% of the SUD group showed deficits in global cognition after 6 months. We observed significantly lower scores in total MoCA (t = −2.36, P = 0.022; Fig. 2(a)) and abstraction (t = −2.10, P = 0.044; Fig. 2(g)) in the SUD group compared with the control group after 6 months.
Gender differences in cognitive function in the SUD group at baseline and after 6 months
MoCA scores revealed that 76.20% of men and 65.60% of women had deficits in global cognition at baseline. We observed that only men had significantly lower scores in abstraction compared with women at baseline (t = −2.22, P = 0.029; Fig. 3(c)).
Changes in global and domain-specific cognitive performance in male and female patients at baseline (Pre) and following 6 months of abstinence (Post). (a) Total Montreal Cognitive Assessment scores. (b) Attention score. (c) Abstraction score. (d) Memory score. Mean values and standard deviation are represented in the bar graphs. *P < 0.001.

Regarding intra-subject analysis, men showed significant improvement in total MoCA (t = −2.44, P = 0.024; Fig. 3(a)), attention (t = −2.23, P = 0.038; Fig. 3(b)), abstraction (t = −2.83, P = 0.010; Fig. 3(c)) and memory (t = −2.39, P = 0.029; Fig. 3(d)) after 6 months of abstinence compared with baseline (Fig. 3). However, we did not observe significant differences in overall and domain-specific MoCA scores in women after 6 months of abstinence compared with baseline.
MoCA scores revealed that 47.60% of men with SUD and 53.80% of women with SUD had deficits in global cognition following 6 months of abstinence. However, we did not find significant differences in overall and domain-specific MoCA scores between men and women following 6 months of abstinence.
Differences in addiction-related variables according to cognitive function at baseline and after 6 months
We analysed alcohol, cocaine and cannabis use, because these were the most prevalent substances in our sample (see Table 3). We did not find significant differences between the prevalence of cognitive impairment at baseline and the presence or absence of a diagnosis of alcohol, cocaine and cannabis use disorder. However, when we analysed specific addiction-related variables while controlling for substance, we observed that participants in the SUD group with cognitive impairment at baseline, compared with the control group at baseline, had significantly greater lifetime alcohol severity (F 1,61 = 5.37, P = 0.024; Fig. 4(a)), and developed cannabis use disorder significantly earlier (F 1,41 = 4.40, P = 0.042; Fig. 4(b)) than patients without cognitive impairment. After 6 months of abstinence, no significant differences were found in cognitive impairment according to the presence of either alcohol, cocaine or cannabis use disorder or other addiction-related variables.
Associations between substance use disorders (SUDs) and addiction-related variables with cognitive outcomes in SUD patients. (a) Lifetime alcohol severity and cognitive impairment. (b) Onset of cannabis use disorder and cognitive impairment. (c) Cocaine use disorder and cognitive recovery, based on Montreal Cognitive Assessment (MoCA) scores. (d) Linear regression analysis to predict cognitive recovery based on the presence or absence of cocaine use disorder. Mean and standard deviation are represented in the bar graphs. Fitted regression line and 95% confidence interval represent the linear regression plot. *P < 0.001.

We also explored differences in the recovery of cognitive function in alcohol, cocaine and cannabis use disorders, as measured by the difference in MoCA total scores between baseline and 6-month assessments (Δ = 6 months–baseline). We observed that only patients with cocaine use disorder showed a lower change in MoCA score than those without cocaine use disorder, even when alcohol and cannabis consumption were controlled for (F 3, 30 = 5.74, P = 0.023; Fig. 4(c)). A simple linear regression was performed to examine whether cocaine use disorder predicted cognitive change. The model was significant (F 1, 32 = 6.28, P = 0.018), explaining 16.40% of the variance in cognitive change according to R 2. Patients with cocaine use disorder showed less improvement in MoCA scores (0.33 points, B = −1.67, s.e. = 0.67, β = −0.41, P = 0.018) compared with those without cocaine use disorder, who improved by 1.67 points on average (Fig. 4(b)).
Differences in cognitive function and addiction-related variables according to addiction treatment abandonment
Although there were no statistically significant differences in the prevalence of treatment abandonment between participants with and without cognitive impairment (Supplementary Table 1 available at https://doi.org/10.1192/bjo.2026.10986), those who discontinued treatment showed significantly lower scores in language (U = 677, P = 0.010) and fluency (U = 800, P = 0.047) at baseline compared with those who remained under treatment over the 6 months. In addition, we found a significantly higher prevalence of treatment abandonment in participants with cocaine use disorder (38.70%) compared with those without this disorder (18.20%) (χ 2 = 4.20, P = 0.040). However, no significant differences were found in treatment dropout rates based on the presence of a diagnosis of alcohol or cannabis use disorder, nor in other variables related to addiction (Supplementary Table 1).
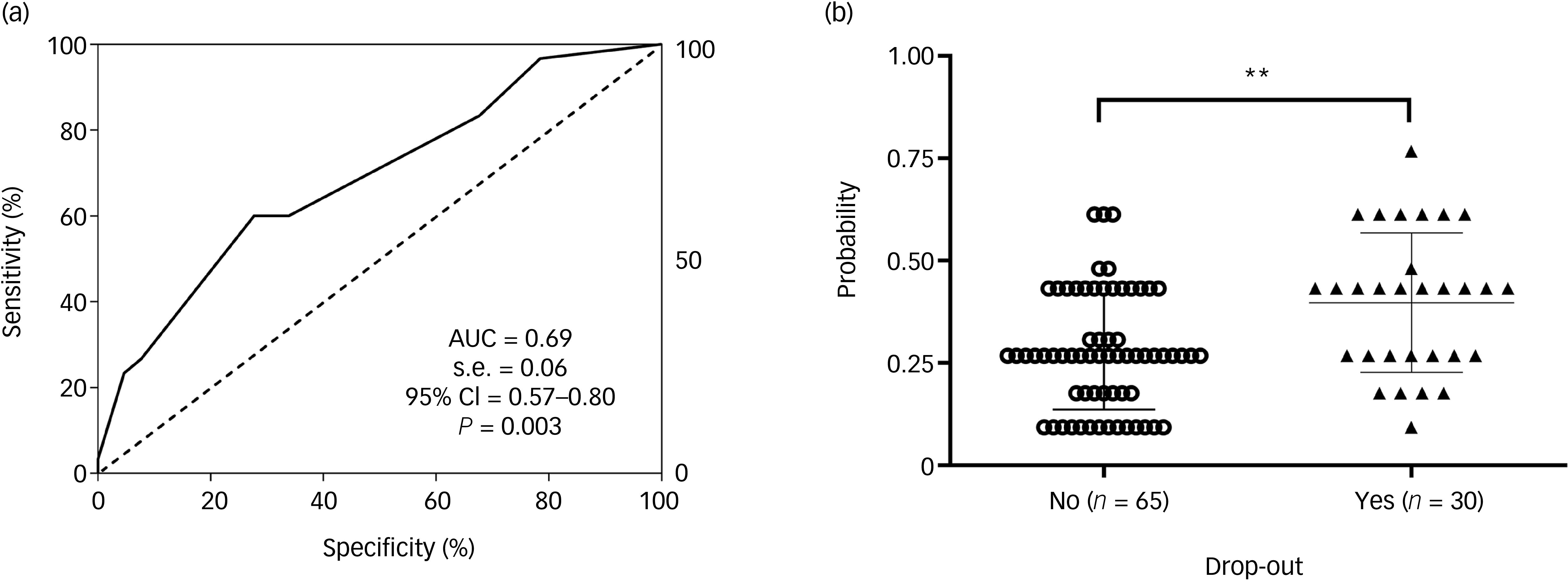
A binary logistic regression model was performed to examine whether cocaine use disorder and language and fluency scores could predict treatment dropout for addiction. Fluency scores were excluded because these did not reach statistical significance for the model. The model was statistically significant and showed good calibration (Hosmer–Lemeshow χ 2 = 4.71, P = 0.319), with a Nagelkerke R 2 of 0.162, indicating a modest but acceptable model fit for a logistic regression within a clinical context. The final model had a sensitivity (true positive rate) and a specificity (false negative rate 100% − specificity percentage) for correct prediction of SUD participants with and without treatment abandonment in 60 and 72.30%, respectively, of cases (overall percentage correct 68.40%, cut-off value 0.40). Having a diagnosis of cocaine use disorder increased the likelihood of dropping out of treatment by 3.57-fold (odds ratio 3.57, 95% CI = 1.19–10.73, P = 0.023). For each additional point on the language subtest, the probability of dropping out of treatment decreased by approximately 52% (odds ratio 0.48, 95% CI = 0.28–0.83, P = 0.009). In addition, ROC analysis (AUC = 0.69, 95% CI = 0.57–0.80) indicated a moderate discrimination power (Fig. 5(a)). The predicted probability also indicated significant differences between SUD participants with and without treatment abandonment (U = 607, P < 0.010; Fig. 5(b)).
Receiver operating characteristic (ROC) analysis and scatter plots of the multivariate predictive model for treatment dropout. (a) ROC curve for the model whose variables were language and cocaine use disorder. (b) Scatter plot of the predictive probabilities for model A. Mean values ± standard deviation are represented in the scatter plot. AUC, area under the ROC curve. **P < 0.010.

We wanted to evaluate variables that could have biased the model, such as sociodemographic variables (e.g. educational level), clinical variables (e.g. medical and psychiatric comorbidity) and pharmacological variables (e.g. anxiolytics). We found that only those participants who had discontinued treatment had a lower educational level (P < 0.029) and a higher disulfiram intake (P < 0.003) (Supplementary Table 1). However, educational level was not significant for the model when it was included along with the variables language and cocaine use disorder. The disulfiram variable was not included in the model because it yielded a high error rate due to the small sample size.
Discussion
Using MoCA as a brief screening tool, the present study provides new evidence on the cognitive deficits associated with SUD, the partial recovery of cognitive functions during sustained abstinence, the role of gender differences and the predictive value of cognitive variables for treatment adherence. Our main findings are: (a) a large proportion of SUD patients showed deficits on MoCA at baseline (72.6 %); (b) cognitive scores improved significantly overall following 6 months of abstinence, although 50% of the SUD group still had detectable cognitive impairment; (c) men’s cognitive function improved following 6 months of abstinence, but women’s cognitive performance appeared to be stable over time; (d) among addiction-related variables, patients with cognitive impairment at baseline had significantly greater lifetime alcohol severity and developed cannabis use disorder earlier; (e) patients with cocaine use disorder showed a lower change in MoCA score after 6 months, which was able to explain 16.40% of the variance in cognitive recovery; and (f) having a diagnosis of cocaine use disorder and low language scores increased the likelihood of dropping out of treatment and, together, had a predictive capacity of 69%.
Cognitive impairment in SUD
In line with previous reports, the prevalence of cognitive impairment in our SUD sample at baseline was high (73%). Bruijnen et al found that 54.90% of patients in addiction care settings had a substance-induced neurocognitive disorder as assessed by MoCA. Reference Bruijnen, Dijkstra, Walvoort, Markus, VanDerNagel and Kessels5 In a Spanish population, Rojo-Mota et al found, using MoCA, that 70.90% of people with addiction were under treatment and had scores below the normal threshold. Reference Rojo-Mota, Pedrero-Perez, Ruiz-Sanchez de Leon, Llanero-Luque and Puerta-Garcia26
Our results suggest that SUD patients had a worse overall cognitive status than healthy control subjects at baseline, especially in visuospatial/executive functions, language, abstraction and memory. It is widely known that substance use and SUDs can cause impairment of cognitive functions, such as processing speed, attention, executive functions and episodic memory, which negatively impacts the quality of life. Reference Bhusri, Bathla, Gupta and Singh27 The pattern of deficits found in this study supports the notion of a generalised cognitive impairment that is not limited to one specific domain, but rather reflects a broad dysfunction in frontostriatal and frontoparietal circuits, which are particularly vulnerable to drug-induced neurotoxicity. Reference Volkow, Wang, Fowler and Tomasi11 These deficiencies affect the regulation of motivational processes such as the desire for reward and the ability to self-regulate and control impulses, making it more difficult to break the vicious cycle of addiction and remain abstinent. Reference Nautiyal28
In addition, SUD patients showed difficulties in particular items such as drawing a cube, subtractiing and fluency. The inability to draw a cube suggests problems in visuoconstructive planning, which has been linked to maintaining stable employment in alcohol-dependent individuals. Reference Moriyama, Mimura, Kato, Yoshino, Hara and Kashima29 We also found that SUD patients had trouble performing a simple mathematical operation by retaining the calculations mentally. This difficulty could imply deficits in visuospatial working memory and phonological memory, because these are skills essential for calculations and problem-solving, which are closely related to worse clinical outcomes. Reference Peng, Namkung, Barnes and Sun30 Furthermore, poor performance in phonemic fluency could indicate dysexecutive problems such as planning and inhibition of inappropriate responses. Reference Rosenkranz, Kircher, Nagels, Erdeljac and Sekulić Sović31 Deficits in verbal fluency can affect independence, self-care, social interaction and general functionality in those with cognitive deficits such as dementia and schizophrenia. Reference Martyr, Clare, Nelis, Marková, Roth and Woods32,Reference Bowie, Harvey, Moriarty, Parrella, White and Davis33
Recovery of cognitive functions during abstinence
We observed that the prevalence of cognitive impairment decreased from 72.60% at baseline to 50% following 6 months of abstinence, representing an absolute reduction of 22.6 percentage points (31.10% relative reduction). This result indicates a partial reversibility of drug-induced cognitive deficits, showing that abstinence plus addiction treatment can promote neural recovery through processes of neuroplasticity and functional compensation. Reference Parvaz, Rabin, Adams and Goldstein10 Similarly, Pelletier et al found that MoCA scores improved significantly following 4 weeks of abstinence in 236 alcohol-dependent patients, increasing the rate of unimpaired patients from 15.80% at admission to 53.80% at discharge. Reference Pelletier, Nalpas, Alarcon, Rigole and Perney34
The longitudinal follow-up also revealed significant improvements in MoCA total scores, as well as in specific domains such as subtraction and memory. It is important to highlight the fact that we have ruled out the possibility that this cognitive improvement following abstinence is due to the learning effect of the MoCA test, because we did not observe this effect in the control subjects. Functional imaging studies have demonstrated that abstinence is associated with increased activity in the prefrontal cortex and anterior cingulate, regions crucial for executive control and decision-making, which may underpin the behavioural improvements observed during recovery. Reference Goldstein and Volkow35
Although we observed cognitive improvement in SUD patients following 6 months of abstinence, they still had worse global cognitive performance and abstraction ability than the control group. This suggests that cognitive deficits either require a prolonged recovery time (>6 months) or that recovery from them is not entirely complete, particularly following long-term or high-dose substance exposure. In this regard, Le Berre et al found that patients with alcohol use disorder may show improvements with prolonged abstinence in processing speed and attention (first weeks/months), working memory (6–12 months) and certain aspects of executive function (long-term), whereas episodic memory and social cognition skills tend to be less reversible. Reference Berre, Fama and Sullivan36 However, persistent impairments may also result from pre-existing vulnerabilities and cognitive alterations that predispose individuals to addiction and that are not entirely reversible following abstinence. Reference Ersche, Jones, Williams, Turton, Robbins and Bullmore37
Gender differences in cognitive function and recovery
Gender differences in the neurocognitive effects of substance use have received increasing attention, given the biological and hormonal variations between men and women that influence brain structure, neurotransmission and metabolism. Reference Luquiens, Rolland, Pelletier, Alarcon, Donnadieu-Rigole and Benyamina18 At baseline, 76.2% of men and 65.6% of women showed deficits in global cognition, indicating a higher prevalence (but not significant) of cognitive impairment among men. This could be explained by the neuroprotective effects of oestrogen on the inflammatory response that can prevent brain damage. Reference Berre, Fama and Sullivan36 However, overall and domain-specific MoCA scores were relatively similar between men and women at baseline (except in the abstraction domain).
Following 6 months of abstinence, the reduction in cognitive impairment prevalence was greater (but not significant) in men than in women. Men showed a 28.6 percentage point absolute reduction (37.50% relative reduction), whereas women showed an 11.8 percentage point absolute reduction (18% relative reduction). Thus, men showed significant cognitive recovery when compared with baseline, with improvements observed in global cognition, attention, abstraction and memory. These effects were not observed in women, suggesting that their cognitive state is more stable over time and might require longer periods or specific interventions to achieve comparable recovery levels. This is consistent with findings that long-term sobriety in alcoholic women is associated with significant recovery of short-term memory and psychomotor speed, but deficits in postural stability persist even following prolonged abstinence, indicating that certain cognitive and motor functions may require extended recovery times or targeted rehabilitation. Reference Roseribloom, Pfefferbaum and Sullivan38
In this regard, studies have shown that women have lower activity of the gastric enzyme alcohol dehydrogenase compared with men, which can result in higher blood alcohol concentrations and neurotoxic effects. Reference Chrostek, Jelski, Szmitkowski and Puchalski39 Oestrogens also enhance dopaminergic sensitivity and reinforcing effects towards drugs, which could explain the more rapid progression to dependence in women (the ‘telescoping effect’). Reference Becker and Hu40 The faster maturation of girls’ brains could also be related to a greater susceptibility to the neurotoxic effects of early substance use during adolescence. Reference Noorbakhsh, Afzali, Boers and Conrod41 In addition, women have greater comorbidity with depression and anxiety, as well as worse social conditions (e.g. violence, caregiving roles, stigmatisation, socioeconomic disadvantages) than men, Reference Fernández, Miranda, Pastor and Muñoz42 which further complicates cognitive recovery. In this context, women often initiate treatment later than men, which may result in more pronounced cognitive impairment due to prolonged substance use. Reference Greenfield, Back, Lawson and Brady43 The absence of significant differences in global and specific cognitive scores between men and women after 6 months indicates that recovery in women is not necessarily poor, but may follow a different trajectory or require more specific interventions.
Addiction-related variables and cognitive function
We observed that patients with baseline cognitive impairment showed greater lifetime alcohol severity than those without cognitive impairment. Meta-analytic studies have demonstrated that chronic and severe alcohol consumption impairs executive functions, verbal and visual memory, processing speed, attention and language. Reference Stavro, Pelletier and Potvin44 A systematic review by Wang et al expands on this relationship by demonstrating that alcohol use disorder, especially in its severe forms, is a significant risk factor for cognitive impairment and the development of neurodegenerative disorders such as dementia. Reference Wang, Li, Vance and Li45
On the other hand, we found that patients with baseline cognitive impairment developed cannabis use disorder at a younger age than those without cognitive impairment. Scientific literature is in agreement that the early initiation of cannabis use, especially during adolescence, is one of the factors most strongly associated with a higher risk of cognitive impairment, due to its interference with brain development. A clear example is the longitudinal study by Meier et al, in which individuals who began using cannabis regularly before the age of 18 and maintained their use over time showed a substantial decline in total IQ (8-point drop), verbal memory, sustained attention, processing speed and executive skills from adolescence to middle adulthood. This cognitive impairment persisted even in those who stopped using cannabis for years, leading the authors to suggest that the damage could be long-lasting or irreversible. Furthermore, individuals who began using cannabis after adolescence or who used it intermittently did not show significant cognitive declines, reinforcing the importance of the neurological period of brain development during adolescence. Reference Meier, Caspi, Ambler, Harrington, Houts and Keefe46
Finally, patients with cocaine use disorder showed poorer recovery of cognitive function following 6 months of abstinence (16% of explained variance), as evidenced by a significantly smaller change in MoCA scores. This result aligns with previous evidence indicating that cocaine use produces pronounced and sometimes enduring deficits in attention, working memory and executive functions, due to the marked alteration of dopamine signalling and vasoconstrictive effects on the prefrontal cortex. Reference Bachi, Mani, Jeyachandran, Fayad, Goldstein and Alia-Klein47,Reference Briand, Flagel, Garcia-Fuster, Watson, Akil and Sarter48 The diminished cognitive recovery observed in cocaine users underscores the need for targeted cognitive rehabilitation strategies for this population, which may include cognitive training, mindfulness and music and art interventions aimed at enhancing neuroplasticity. Reference Sampedro-Piquero, Ladrón de Guevara-Miranda, Pavón, Serrano, Suárez and Rodríguez de Fonseca49
Cognitive and clinical predictors of treatment adherence
A meta-analysis that included more than 26 000 participants found that dropout rates in face-to-face psychotherapy treatments for SUDs averaged 30.4%. This study highlighted that dropout rates were significantly higher in studies focused on cocaine and other stimulant drugs, and that greater cocaine use was associated with increased treatment dropout rates. Accordingly, our results showed that 46.15% of patients dropped out of treatment and that those with cocaine use disorder had a higher dropout prevalence (38.7%) than those without this diagnosis (18.2%), increasing the risk of dropping out by 3.57-fold. It is important to highlight that patients with cocaine use disorder also showed reduced cognitive recovery, which could be involved in treatment adherence. In this regard, executive function has been shown to predict lower treatment retention in individuals with cocaine dependence receiving residential treatment. Reference Verdejo-García, Betanzos-Espinosa, Lozano, Vergara-Moragues, González-Saiz and Fernández-Calderón14
Furthermore, we observed that patients who dropped out of treatment had worse language and fluency scores than those who remained active. In addition, for each additional point in the language subtest, the probability of dropping out decreased by approximately 52%. In this regard, Aharonovich et al found that specific language patterns in the initial discourse of cocaine patients could predict relevant clinical variables such as craving levels, relapse likelihood, duration of abstinence and subsequent use up to 1 year later. Specifically, less coherent language with a greater presence of negative or ambivalent terms was associated with a higher risk of relapse and a shorter duration of abstinence, whereas richer and more fluent discourse was linked to better treatment outcomes and longer periods of abstinence. Reference Aharonovich, Amrhein, Bisaga, Nunes and Hasin50 Language difficulties can hinder patients’ ability to effectively communicate their needs, thoughts and emotions, potentially resulting in frustration, therapeutic disengagement and feelings of misunderstanding within the treatment context. These communicative issues may result in patients facing challenges in adapting to the demands of psychosocial addiction treatments.
Finally, ROC analysis based on the presence of cocaine use disorder and language showed moderate discriminatory power (69%), and the predicted probabilities differed significantly between patients with and without treatment dropout. Therefore, MoCA cannot predict relapse on its own – only when accompanied by other clinical variables that complement the patient’s cognitive profile.
Limitations
Although this study provides valuable insights, several limitations should be acknowledged. First, the sample size is limited, especially for the longitudinal follow-up, which was reduced because participants dropped out of the treatment pathway; this may have limited the statistical power and generalisability of the results. Second, although MoCA is a sensitive screening tool, it is not a complete neuropsychological battery and may not detect all specific cognitive deficits or subtle impairments. Future research should complement MoCA assessments with comprehensive tests of executive functions and memory. Third, patients and controls were not matched for educational level, which can skew cognitive outcomes. Furthermore, other factors such as sleep quality, nutrition and stress could also influence cognitive performance. Finally, because the participants were recruited from a specific therapeutic community (Proyecto Hombre, Malaga), the findings may not generalise to other populations or treatment modalities.
Future directions
Future studies should aim to expand on these findings by incorporating neuroimaging methods to link cognitive performance with structural and functional brain changes during abstinence. Longitudinal designs with multiple follow-up points could provide a more detailed characterisation of the dynamics of cognitive recovery and relapse risk. Although MoCA is a useful tool for clinical screening, its sensitivity and specificity are limited without biological correlates. Therefore, biomarkers such as vitamin levels, neurotrophic factors (e.g. Brain-Derived Neurotrophic Factor) and indicators of mild systemic inflammation (e.g. C-reactive protein, tumour necrosis factor-α) would be beneficial in improving diagnostic accuracy and potentially helping to identify individuals at higher risk of persistent cognitive impairment. Additionally, interventions specifically targeting cognitive enhancement (such as computerised cognitive training, exercise programmes, mindfulness-based therapies or pharmacological agents) should be tested for their ability to improve both cognitive function and treatment adherence. It would also be valuable to explore whether the integration of MoCA results into personalised feedback for patients could increase motivation and engagement in therapy. Finally, gender differences deserve further investigation, particularly regarding the hormonal and neurobiological factors that modulate recovery. Understanding these mechanisms could guide the development of gender-specific interventions to optimise cognitive rehabilitation in both men and women with SUD.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjo.2026.10986
Data availability
The data that support the findings of this study are available on request from the corresponding author.
Acknowledgements
We thank the Proyecto Hombre institution for their active participation in the recruitment of patients and control subjects for the study. We also thank the team at the Fundación Héroes comprehensive rehabilitation centre, specifically therapists Adrián Pavón Lara and José Carlos Sánchez Romero and coordinator Silvia García Sánchez, for their active participation in the recruitment of control subjects for the study.
Author contributions
N.R.-O. participated in recruiting the participant sample, conducted neuropsychological assessments, scored the tests, compiled the database, performed the statistical analyses and drafted the manuscript. M.C.M.-P. provided support in conducting the neuropsychological assessments. N.S.-Á. developed the statistical plan, participated in the data analysis and reviewed the manuscript. P.S.-P. provided information for the funding application and study design, selected the outcome measures, supervised the study and reviewed the manuscript. E.C.-O. secured funding, initiated and supervised collaboration between the treatment centre and research team, selected the outcome measures for analysis, interpreted the results and reviewed the manuscript.
Funding
This study was funded by the CAIXA-Conecta Project of the Social Observatory of the ‘la Caixa’ Foundation (C21-0260). This work was supported by a Juan de la Cierva Fellowship from the Spanish Ministry of Science, Innovation and Universities awarded to N.R.-O. (JDC2023-051204-I). Funding for the open access charge was received from the University of Malaga.
Declaration of interest
None.








eLetters
No eLetters have been published for this article.